Highly Active and Carbon-Resistant Nickel Single-Atom Catalysts for Methane Dry Reforming
Abstract
1. Introduction
2. Experimental Section
2.1. Catalysts Preparation
2.1.1. Synthesis of Hydroxyapatite (HAP) Support
2.1.2. Deposition of Ni on HAP
2.2. Catalysts Characterization
2.2.1. Inductively Coupled Plasma Spectrometry-Atomic Emission Spectrometry (ICP-AES)
2.2.2. Specific Surface Areas (SSA)
2.2.3. X-ray Diffraction (XRD)
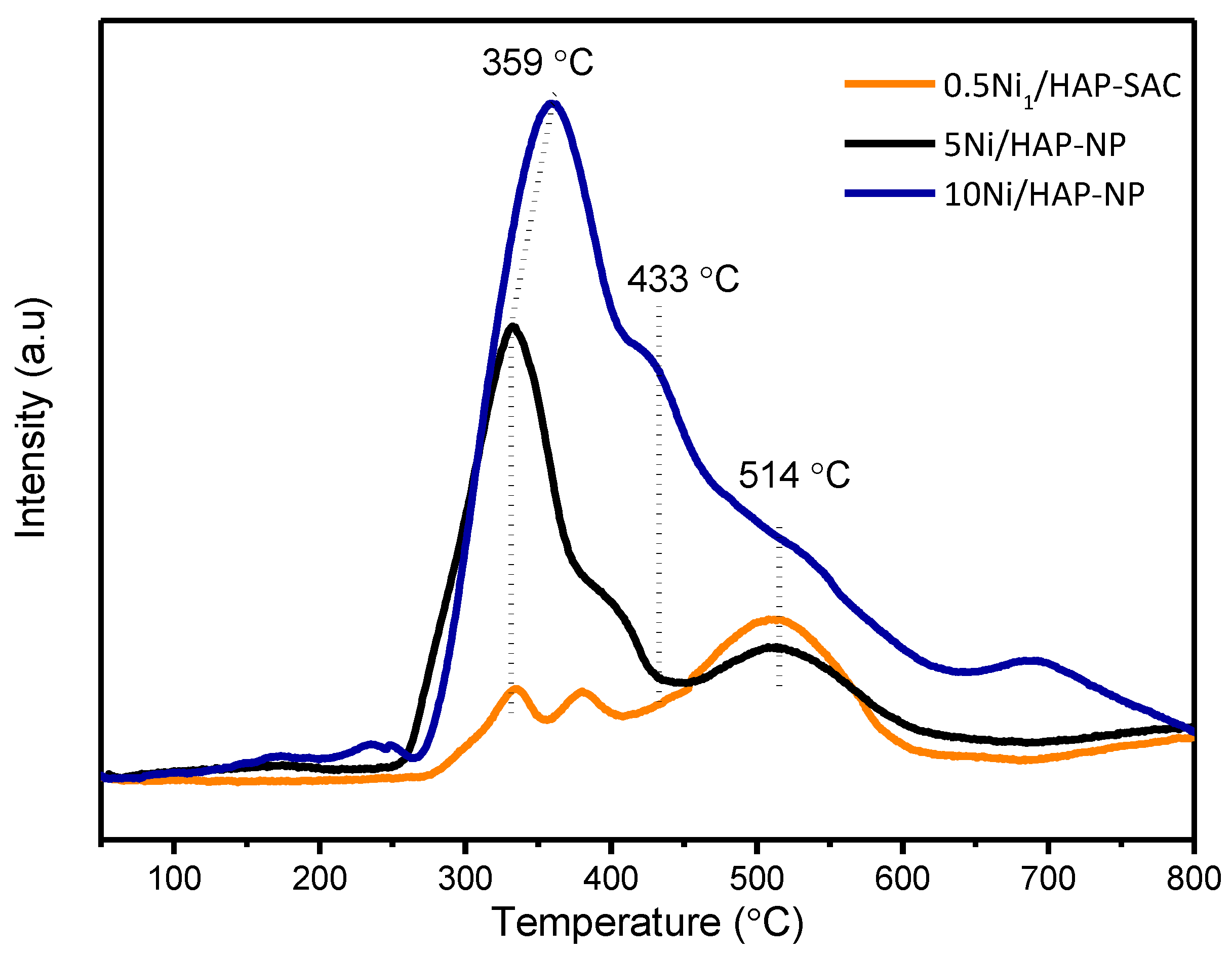
2.2.4. Temperature-Programmed Reduction (H2-TPR)
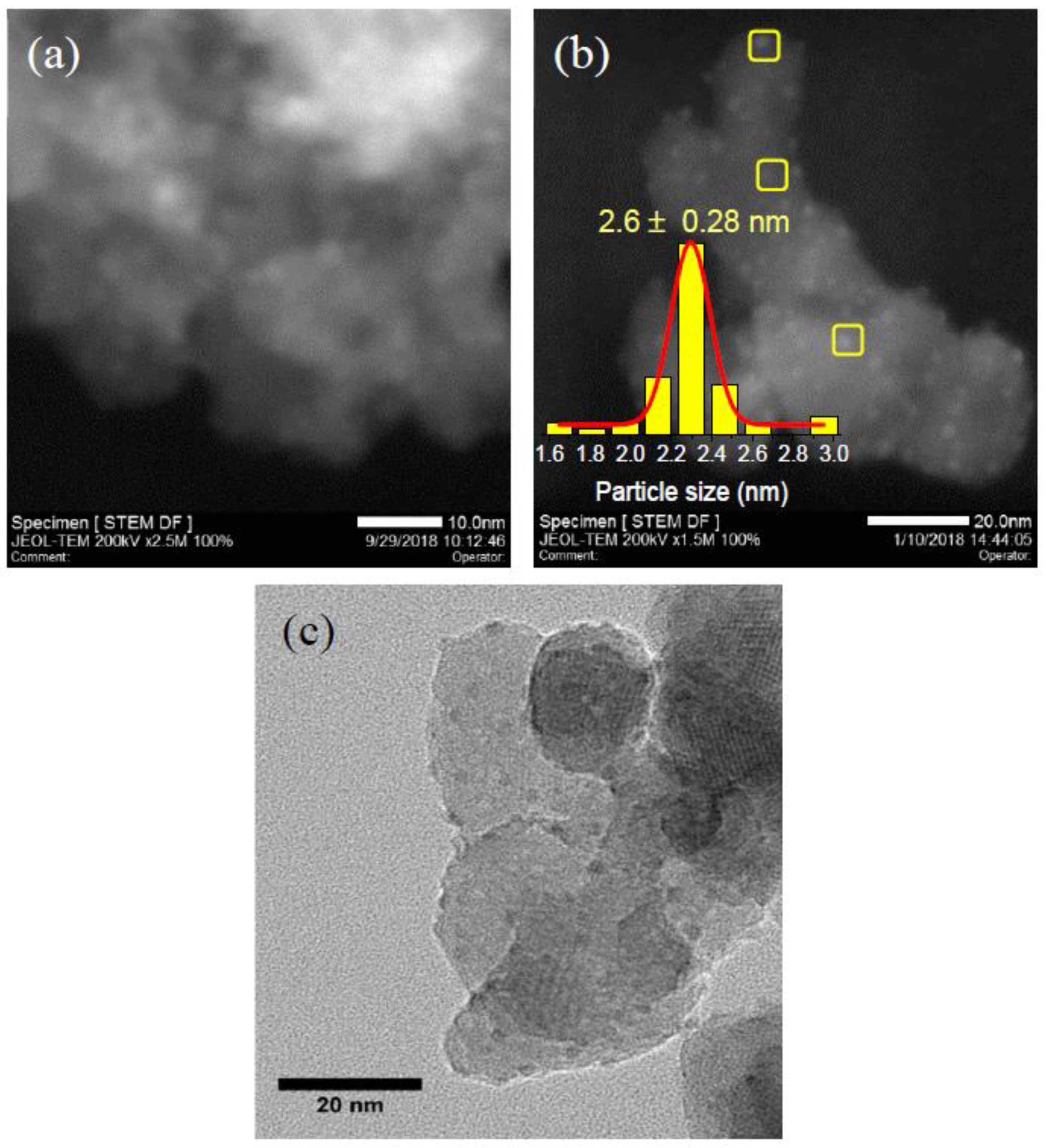
2.2.5. Scanning Transmission Electron Microscopy (STEM)
2.2.6. X-ray Photoelectron Spectroscopy (XPS)
2.2.7. Raman Spectroscopy
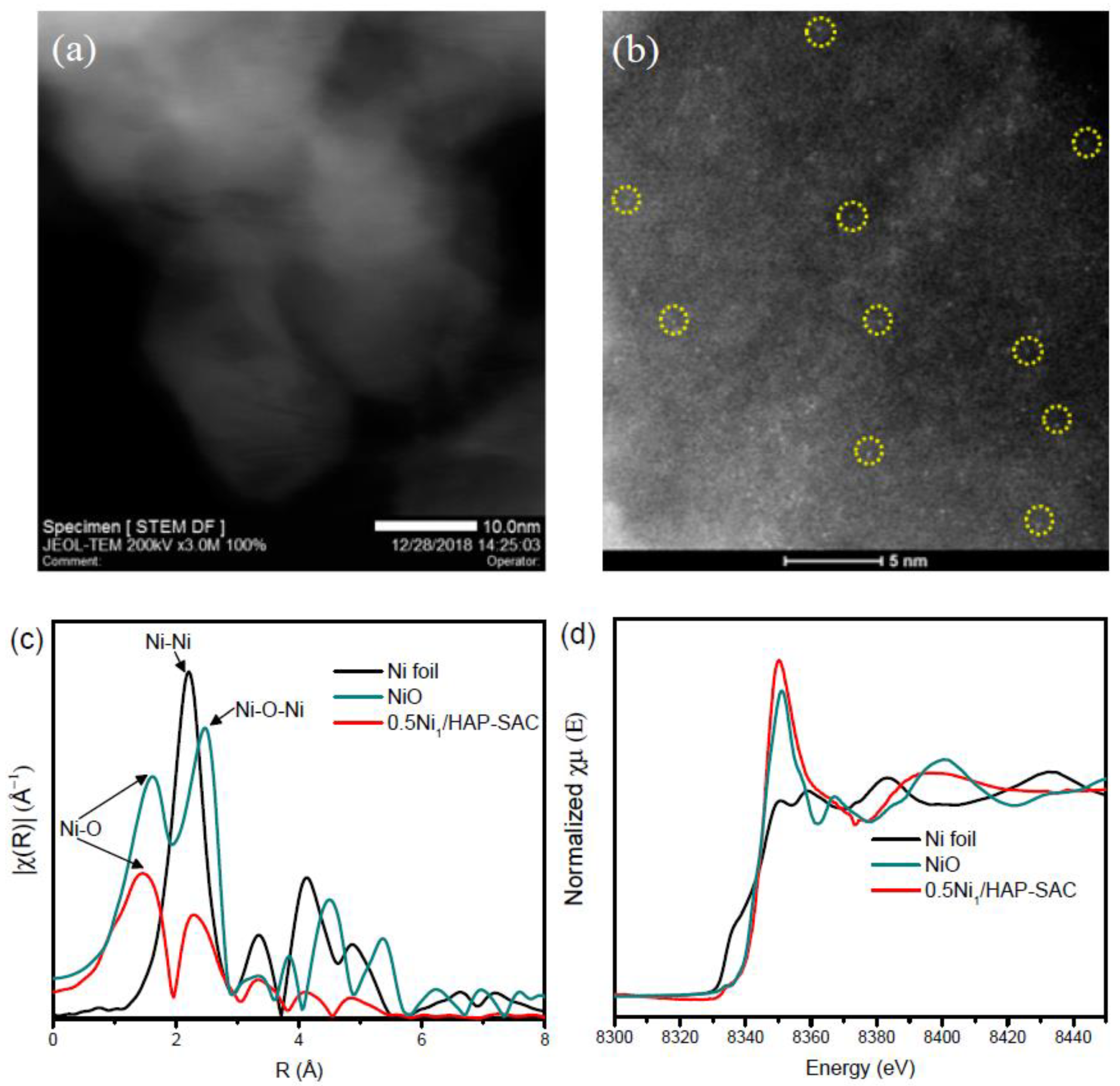
2.2.8. X-ray Absorption Fine Structure (XAFS)
2.3. Catalytic Test
3. Results and Discussions
3.1. Textural Proprieties
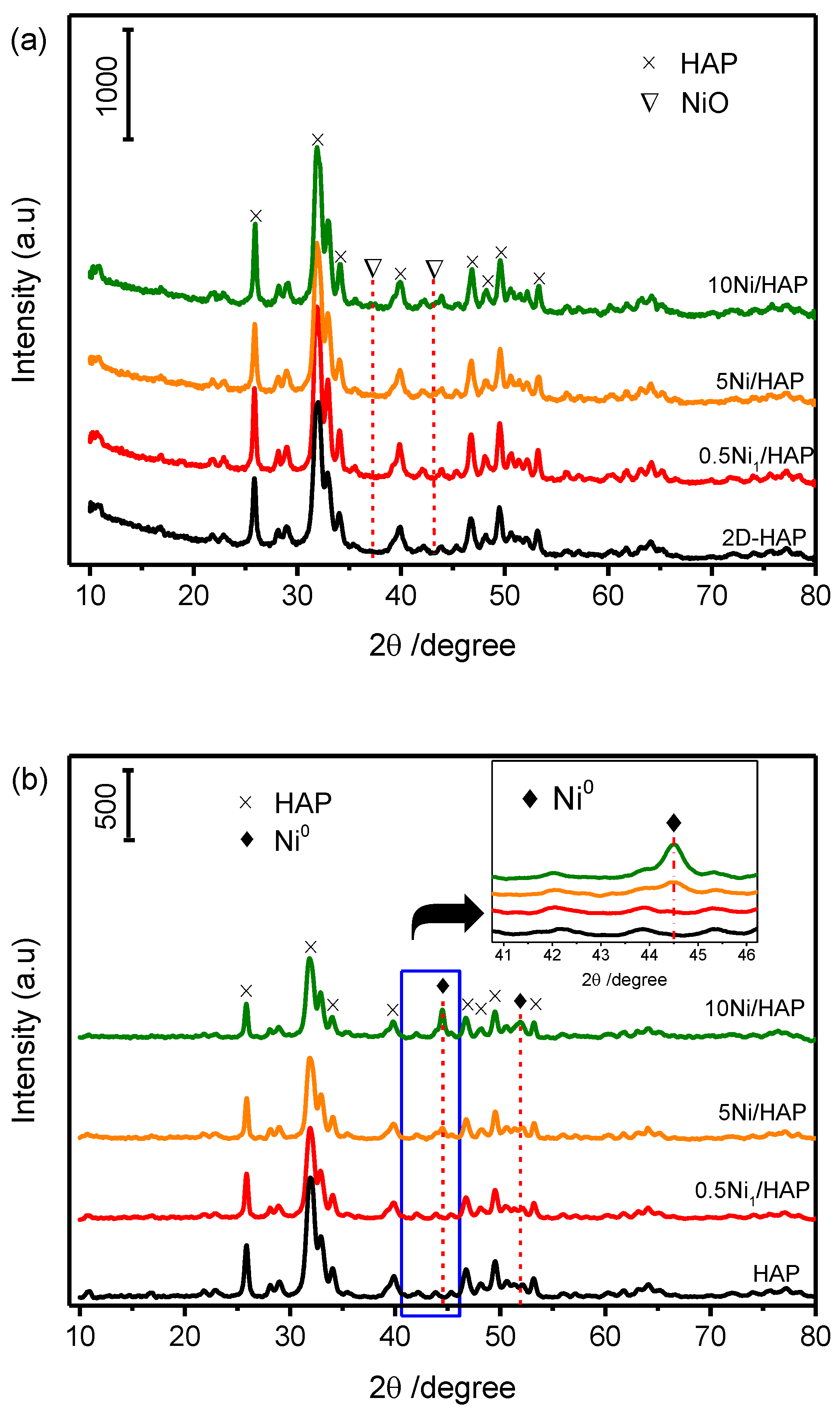
3.2. Structural Properties
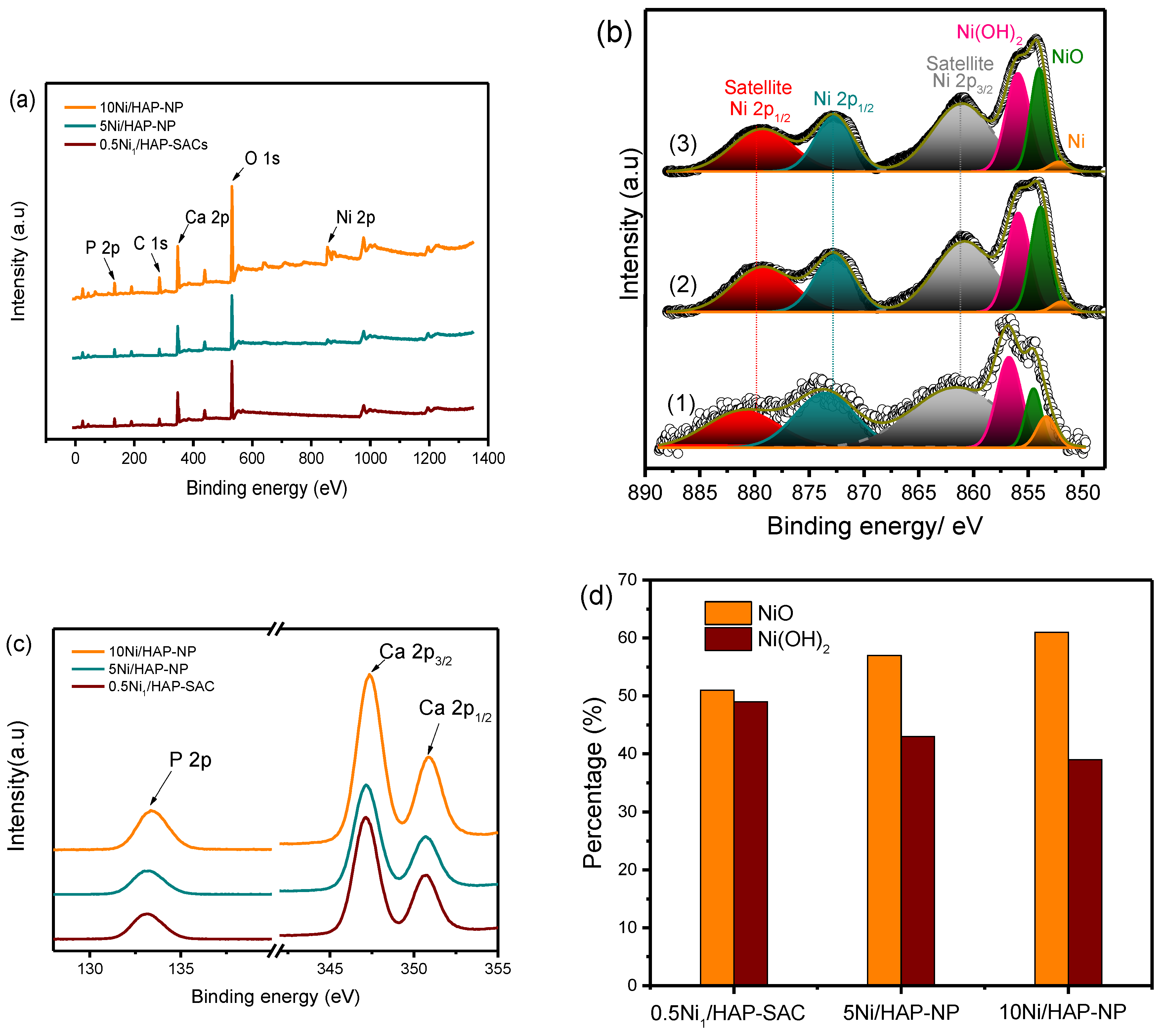
3.3. Chemical States of Ni over Various Catalysts
3.4. Morphology and Microstructure
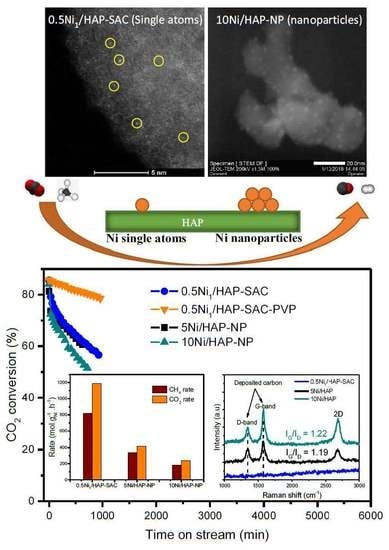
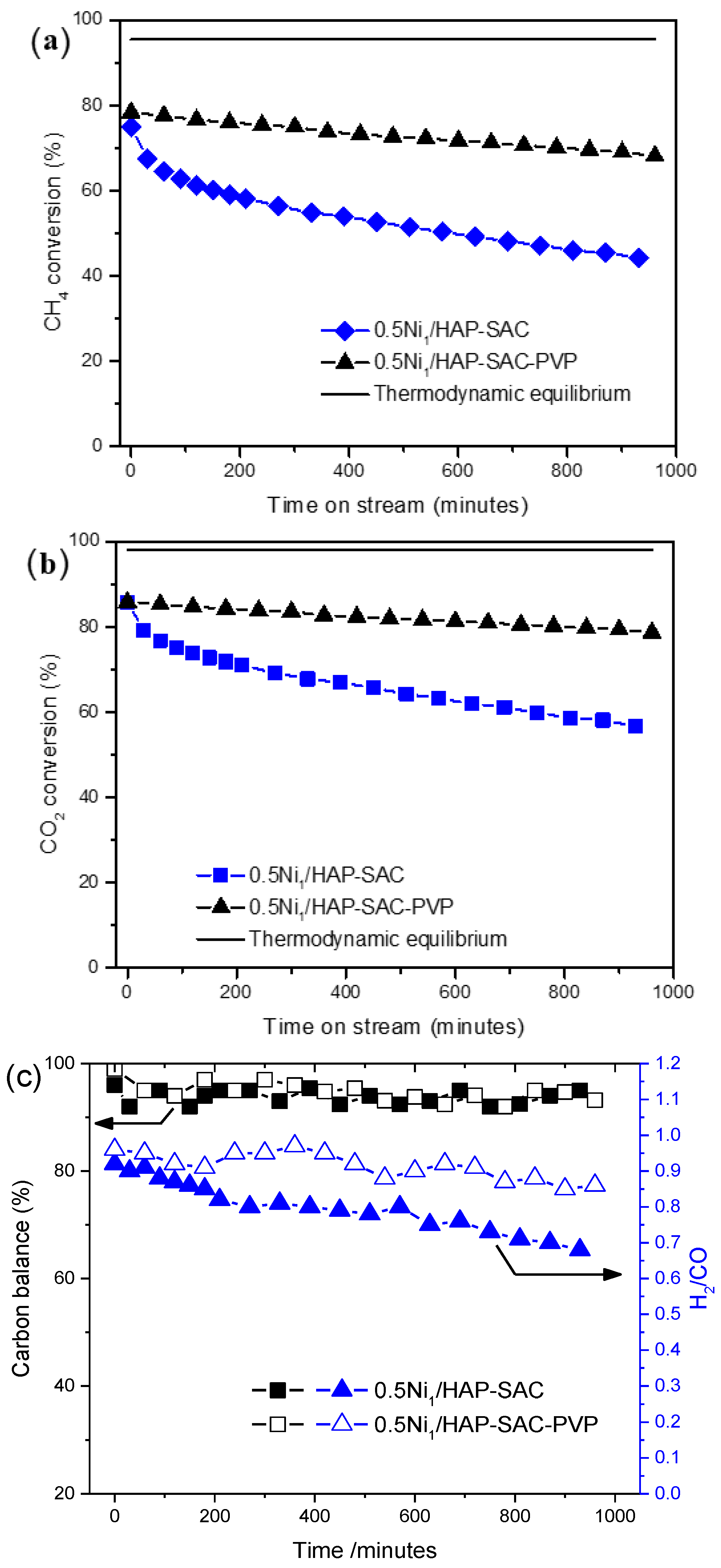
3.5. Catalysts Performance
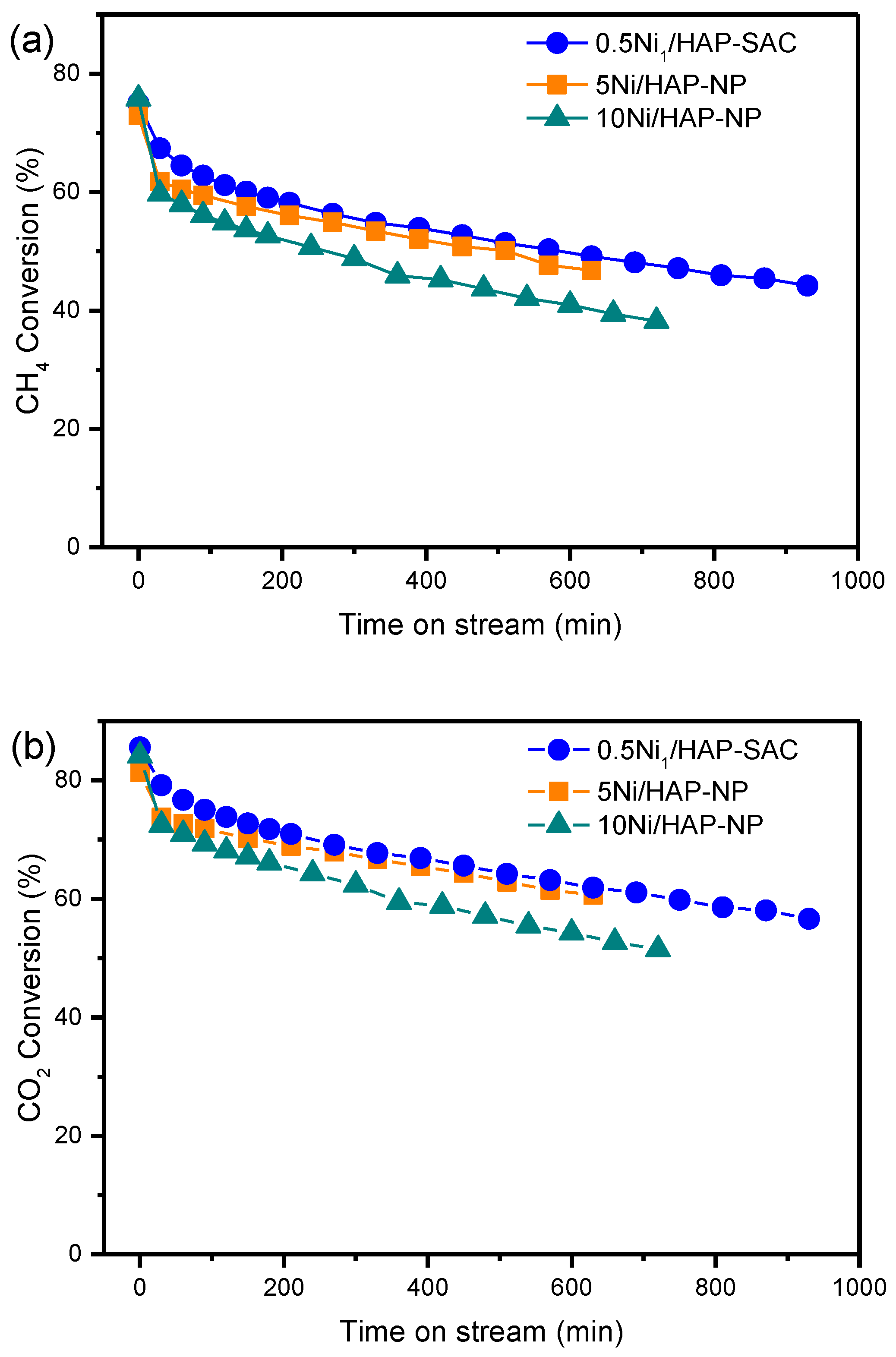
3.5.1. Influence of Ni Loading
3.5.2. Influence of the Synthesis Method of 0.5Ni/HAP
3.5.3. Intrinsic Activity and Comparison with Published Papers
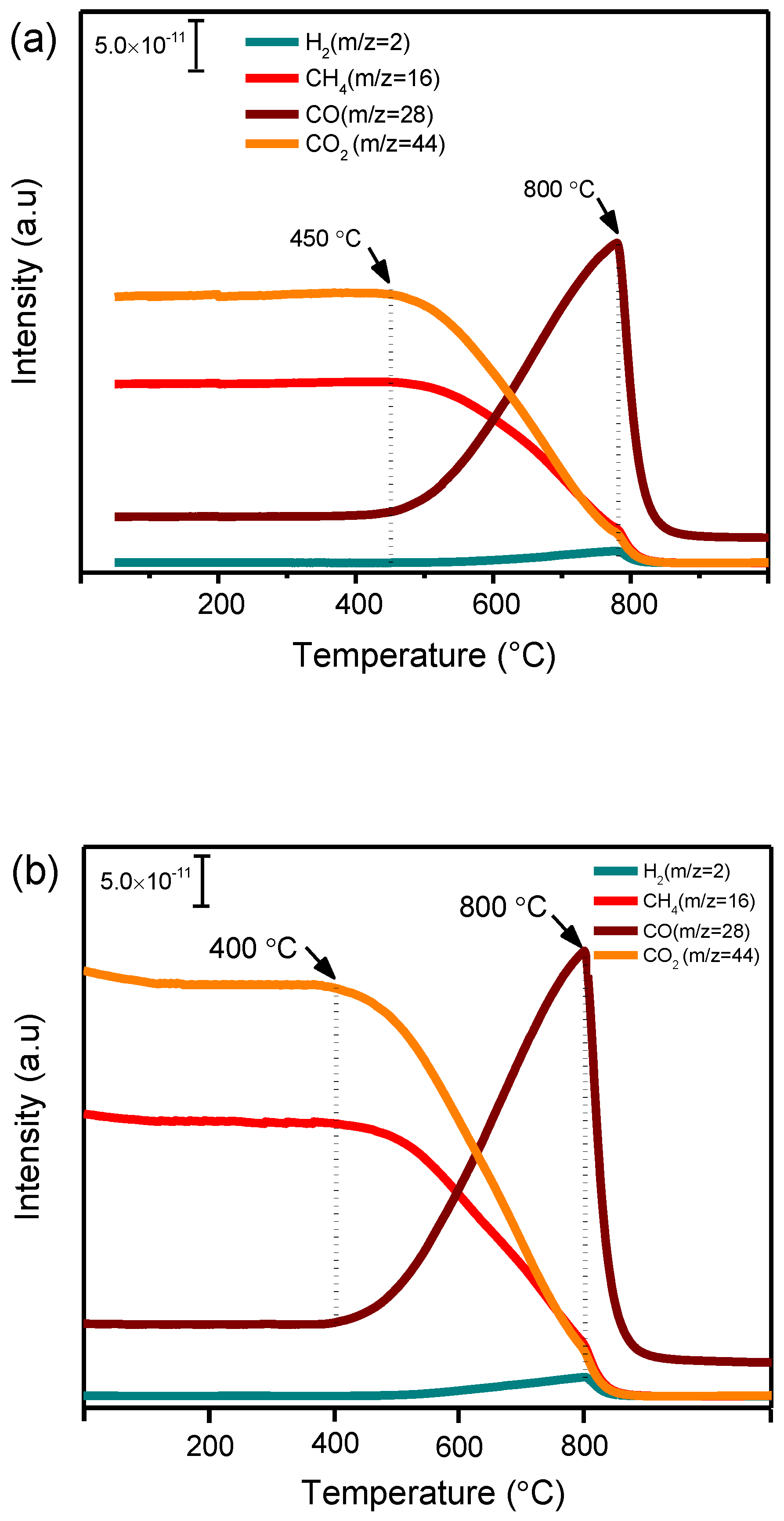
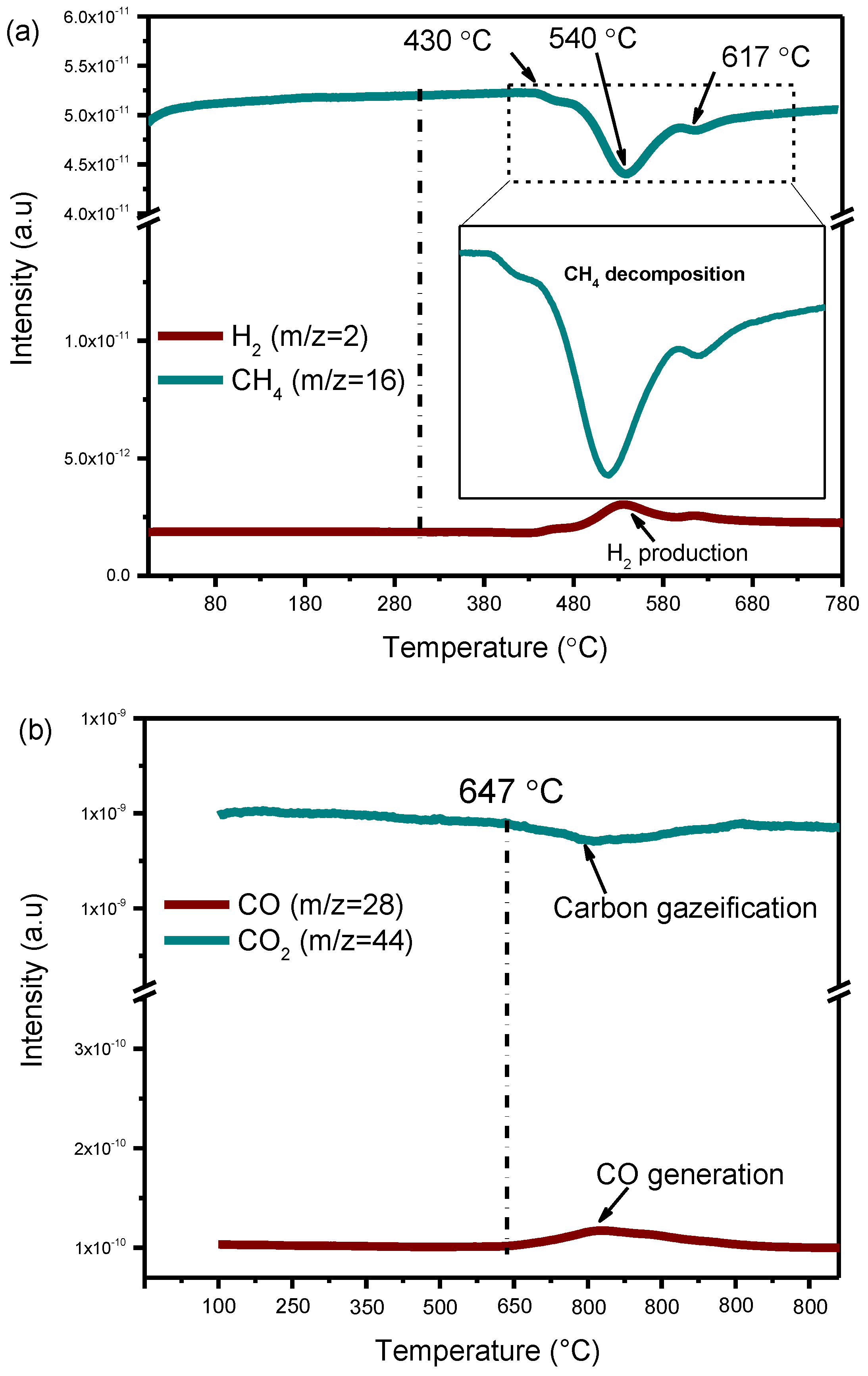
3.5.4. Carbon Deposition and Its Reactivity with CO2 over Nickel Nanoparticle Catalysts
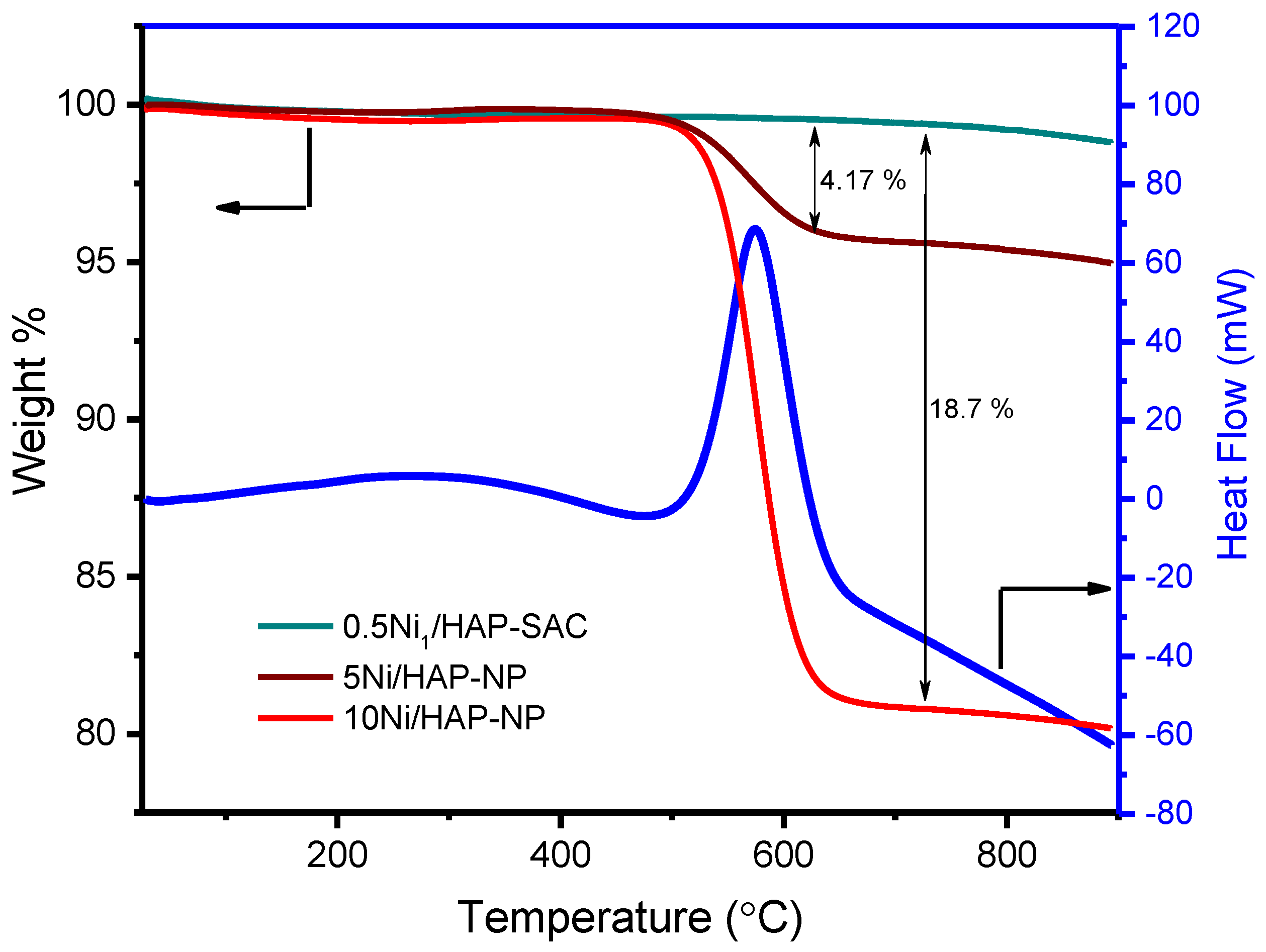
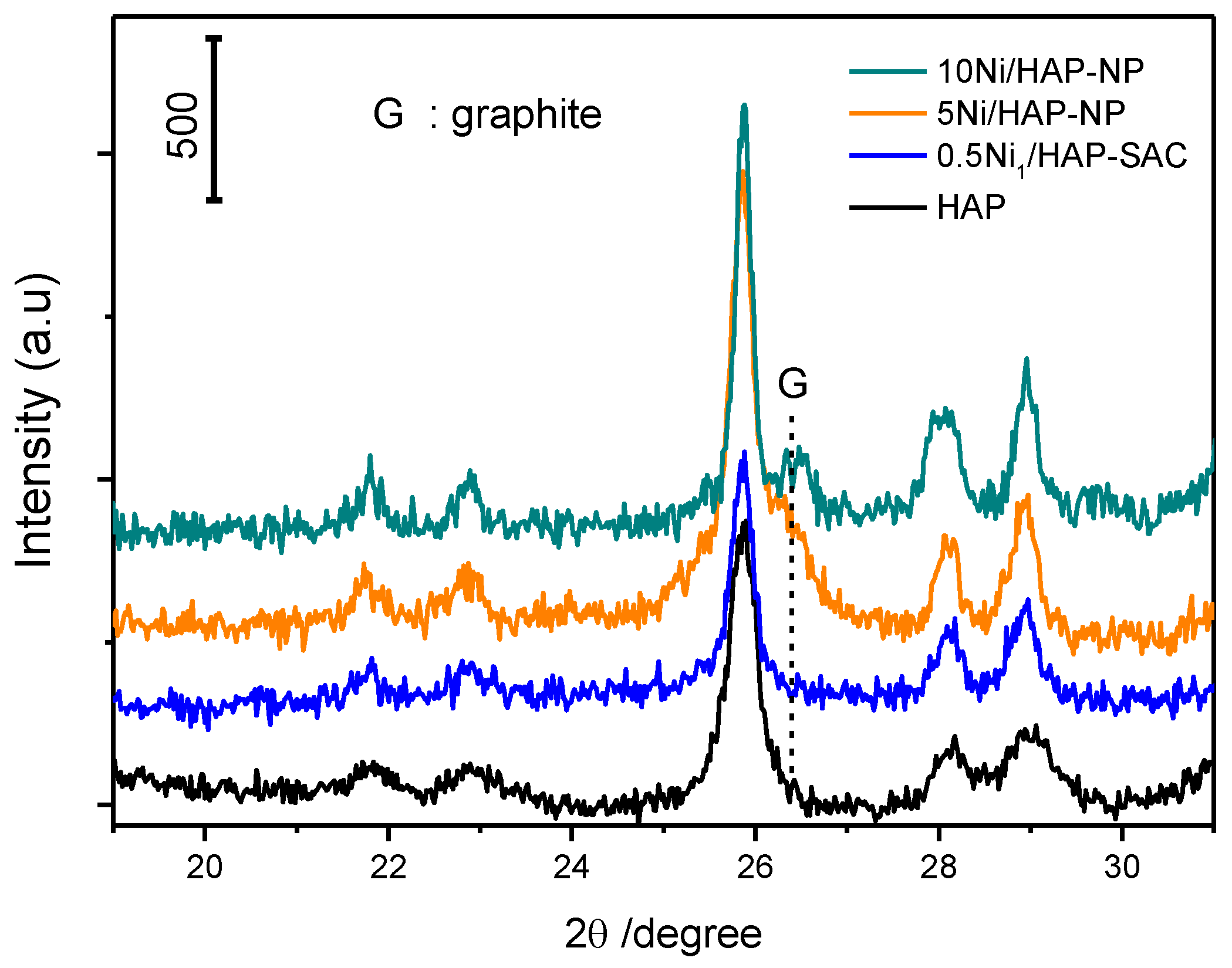
3.6. Characterization after Reaction
3.6.1. TDA-TGA Analysis
3.6.2. X-ray Diffraction (XRD)
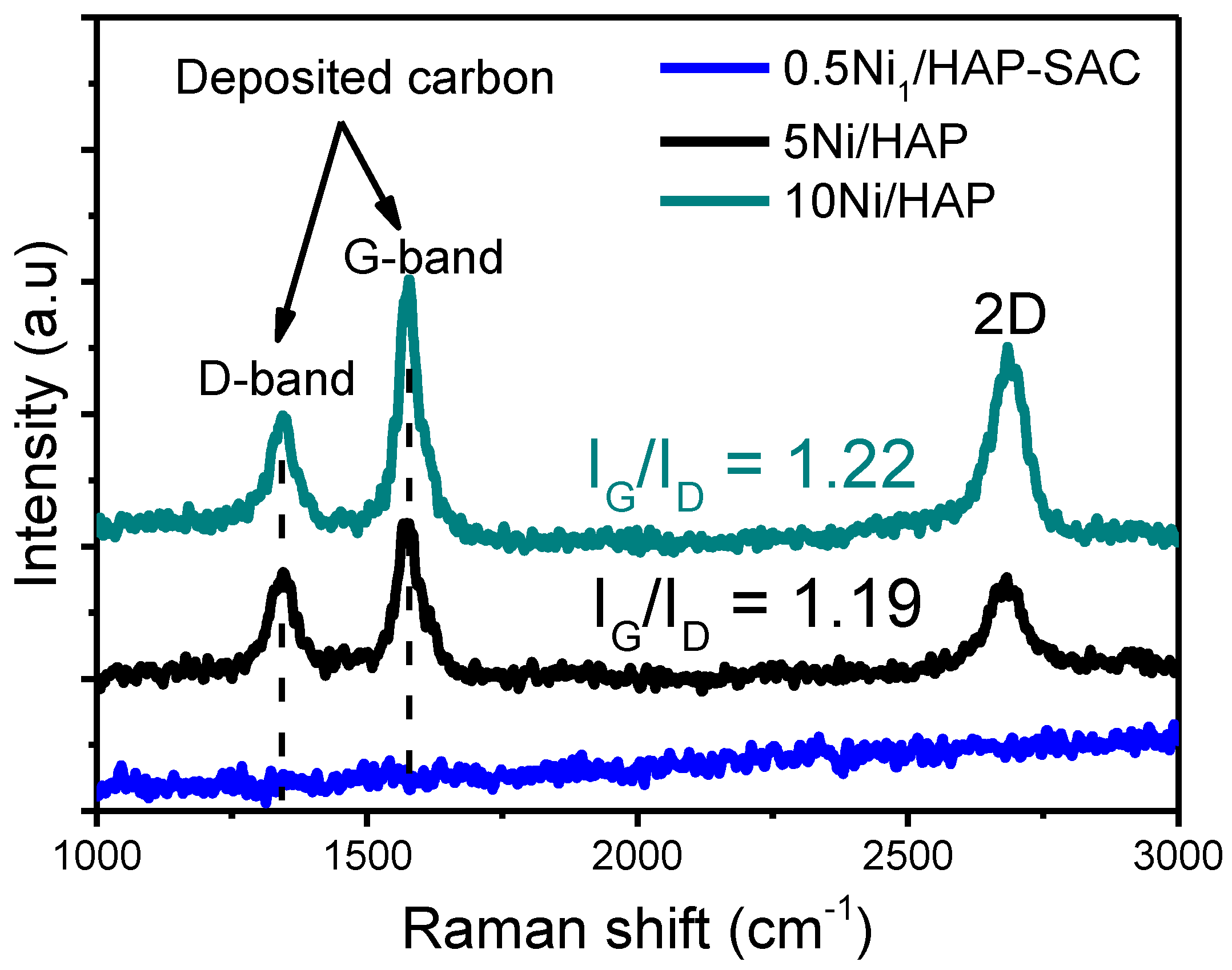
3.6.3. RAMAN Spectroscopy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, D.; Guo, X.; Xiao, B. What causes growth of global greenhouse gas emissions? Evidence from 40 countries. Sci. Total Environ. 2019, 661, 750–766. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, J.-M. Review on dry reforming of methane, a potentially more environmentally-friendly approach to the increasing natural gas exploitation. Front. Chem. 2014, 2, 81. [Google Scholar] [CrossRef] [PubMed]
- Kathiraser, Y.; Oemar, U.; Saw, E.T.; Li, Z.; Kawi, S. Kinetic and mechanistic aspects for CO2 reforming of methane over Ni based catalysts. Chem. Eng. J. 2015, 278, 62–78. [Google Scholar] [CrossRef]
- Jang, W.-J.; Shim, J.-O.; Kim, H.-M.; Yoo, S.-Y.; Roh, H.-S. A review on dry reforming of methane in aspect of catalytic properties. Catal. Today 2019, 324, 15–26. [Google Scholar] [CrossRef]
- Abdullah, B.; Ghani, N.A.A.; Vo, D.-V.N. Recent advances in dry reforming of methane over Ni-based catalysts. J. Clean. Prod. 2017, 162, 170–185. [Google Scholar] [CrossRef]
- Usman, M.; Daud, W.M.A.W.; Abbas, H.F. Dry reforming of methane: Influence of process parameters—A review. Renew. Sustain. Energy Rev. 2015, 45, 710–744. [Google Scholar] [CrossRef]
- Tsyganok, A.I.; Inaba, M.; Tsunoda, T.; Hamakawa, S.; Suzuki, K.; Hayakawa, T. Dry reforming of methane over supported noble metals: A novel approach to preparing catalysts. Catal. Commun. 2003, 4, 493–498. [Google Scholar] [CrossRef]
- Liu, X.; Wang, H.; Ye, G.; Zhou, X.; Keil, F.J. Enhanced performance of catalyst pellets for methane dry reforming by engineering pore network structure. Chem. Eng. J. 2019, 373, 1389–1396. [Google Scholar] [CrossRef]
- Akri, M.; Chafik, T.; Granger, P.; Ayrault, P.; Batiot-Dupeyrat, C. Novel nickel promoted illite clay based catalyst for autothermal dry reforming of methane. Fuel 2016, 178, 139–147. [Google Scholar] [CrossRef]
- Akri, M.; Pronier, S.; Chafik, T.; Achak, O.; Granger, P.; Simon, P.; Trentesaux, M.; Batiot-Dupeyrat, C. Development of nickel supported La and Ce-natural illite clay for autothermal dry reforming of methane: Toward a better resistance to deactivation. Appl. Catal. B Environ. 2017, 205, 519–531. [Google Scholar] [CrossRef]
- Akri, M.; Achak, O.; Granger, P.; Wang, S.; Batiot-Dupeyrat, C.; Chafik, T. Autothermal reforming of model purified biogas using an extruded honeycomb monolith: A new catalyst based on nickel incorporated illite clay promoted with MgO. J. Clean. Prod. 2018, 171, 377–389. [Google Scholar] [CrossRef]
- Gallego, G.S.; Batiot-Dupeyrat, C.; Barrault, J.; Florez, E.; Mondragón, F. Dry reforming of methane over LaNi1−yByO3±δ (B=Mg, Co) perovskites used as catalyst precursor. Appl. Catal. A Gen. 2008, 334, 251–258. [Google Scholar] [CrossRef]
- Gallego, G.S.; Marín, J.G.; Batiot-Dupeyrat, C.; Barrault, J.; Mondragón, F. Influence of Pr and Ce in dry methane reforming catalysts produced from La1−xAxNiO3−δ perovskites. Appl. Catal. A Gen. 2009, 369, 97–103. [Google Scholar] [CrossRef]
- Pakhare, D.; Spivey, J. A review of dry (CO2) reforming of methane over noble metal catalysts. Chem. Soc. Rev. 2014, 43, 7813–7837. [Google Scholar] [CrossRef]
- Arora, S.; Prasad, R. An overview on dry reforming of methane: Strategies to reduce carbonaceous deactivation of catalysts. RSC Adv. 2016, 6, 108668–108688. [Google Scholar] [CrossRef]
- Muraza, O.; Galadima, A. A review on coke management during dry reforming of methane. Int. J. Energy Res. 2015, 39, 1196–1216. [Google Scholar] [CrossRef]
- Aramouni, N.A.K.; Zeaiter, J.; Kwapinski, W.; Ahmad, M.N. Thermodynamic analysis of methane dry reforming: Effect of the catalyst particle size on carbon formation. Energy Convers. Manag. 2017, 150, 614–622. [Google Scholar] [CrossRef]
- Han, J.W.; Park, J.S.; Choi, M.S.; Lee, H. Uncoupling the size and support effects of Ni catalysts for dry reforming of methane. Appl. Catal. B Environ. 2017, 203, 625–632. [Google Scholar] [CrossRef]
- Xin, J.; Cui, H.; Cheng, Z.; Zhou, Z. Bimetallic Ni-Co/SBA-15 catalysts prepared by urea co-precipitation for dry reforming of methane. Appl. Catal. A Gen. 2018, 554, 95–104. [Google Scholar] [CrossRef]
- Liu, J. Catalysis by supported single metal atoms. ACS Catal. 2017, 7, 34–59. [Google Scholar] [CrossRef]
- Liu, L.; Corma, A. Metal catalysts for heterogeneous catalysis: From single atoms to nanoclusters and nanoparticles. Chem. Rev. 2018, 118, 4981–5079. [Google Scholar] [CrossRef] [PubMed]
- Akri, M.; Zhao, S.; Li, X.; Zang, K.; Lee, A.F.; Isaacs, M.A.; Xi, W.; Gangarajula, Y.; Luo, J.; Ren, Y.; et al. Atomically dispersed nickel as coke-resistant active sites for methane dry reforming. Nat. Commun. 2019, 10, 5181. [Google Scholar] [CrossRef]
- James, T.E.; Hemmingson, S.L.; Campbell, C.T. Energy of supported metal catalysts: From single atoms to large metal nanoparticles. ACS Catal. 2015, 5, 5673–5678. [Google Scholar] [CrossRef]
- Tang, Y.; Wei, Y.; Wang, Z.; Zhang, S.; Li, Y.; Nguyen, L.; Li, Y.; Zhou, Y.; Shen, W.; Tao, F.F.; et al. Synergy of single-atom Ni1 and Ru1 sites on CeO2 for dry reforming of CH4. J. Am. Chem. Soc. 2019, 141, 7283–7293. [Google Scholar] [CrossRef] [PubMed]
- Qiao, B.; Liu, J.; Wang, Y.-G.; Lin, Q.; Liu, X.; Wang, A.; Li, J.; Zhang, T.; Liu, J. Highly efficient catalysis of preferential oxidation of CO in H2-Rich stream by gold single-atom catalysts. ACS Catal. 2015, 5, 6249–6254. [Google Scholar] [CrossRef]
- Chen, Y.; Lin, J.; Li, L.; Qiao, B.; Liu, J.; Su, Y.; Wang, X. Identifying size effects of pt as single atoms and nanoparticles supported on FeOx for the water-gas shift reaction. ACS Catal. 2018, 8, 859–868. [Google Scholar] [CrossRef]
- Gu, X.-K.; Qiao, B.; Huang, C.-Q.; Ding, W.-C.; Sun, K.; Zhan, E.; Zhang, T.; Liu, J.; Li, W.-X. Supported single Pt1/Au1 atoms for methanol steam reforming. ACS Catal. 2014, 4, 3886–3890. [Google Scholar] [CrossRef]
- Lin, J.; Wang, A.; Qiao, B.; Liu, X.; Yang, X.; Wang, X.; Liang, J.; Li, J.; Liu, J.; Zhang, T. Remarkable performance of Ir1/FeOx single-atom catalyst in water gas shift reaction. J. Am. Chem. Soc. 2013, 135, 15314–15317. [Google Scholar] [CrossRef]
- Lang, R.; Xi, W.; Liu, J.-C.; Cui, Y.-T.; Li, T.; Lee, A.F.; Chen, F.; Chen, Y.; Li, L.; Li, L.; et al. Non defect-stabilized thermally stable single-atom catalyst. Nat. Commun. 2019, 10, 234. [Google Scholar] [CrossRef]
- Qiao, B.; Wang, A.; Yang, X.; Allard, L.F.; Jiang, Z.; Cui, Y.; Liu, J.; Li, J.; Zhang, T. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 2011, 3, 634. [Google Scholar] [CrossRef]
- Boukha, Z.; Kacimi, M.; Pereira, M.F.R.; Faria, J.L.; Figueiredo, J.L.; Ziyad, M. Methane dry reforming on Ni loaded hydroxyapatite and fluoroapatite. Appl. Catal. A Gen. 2007, 317, 299–309. [Google Scholar] [CrossRef]
- Tang, H.; Wei, J.; Liu, F.; Qiao, B.; Pan, X.; Li, L.; Liu, J.; Wang, J.; Zhang, T. Strong metal–support interactions between gold nanoparticles and nonoxides. J. Am. Chem. Soc. 2016, 138, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Boukha, Z.; Yeste, M.P.; Cauqui, M.Á.; González-Velasco, J.R. Influence of Ca/P ratio on the catalytic performance of Ni/hydroxyapatite samples in dry reforming of methane. Appl. Catal. A Gen. 2019, 580, 34–45. [Google Scholar] [CrossRef]
- Pizzala, H.; Caldarelli, S.; Eon, J.-G.; Rossi, A.M.; Laurencin, D.; Smith, M.E. A solid-state NMR study of lead and vanadium substitution into hydroxyapatite. J. Am. Chem. Soc. 2009, 131, 5145–5152. [Google Scholar] [CrossRef] [PubMed]
- Cawthray, J.F.; Creagh, A.L.; Haynes, C.A.; Orvig, C. Ion Exchange in hydroxyapatite with lanthanides. Inorg. Chem. 2015, 54, 1440–1445. [Google Scholar] [CrossRef]
- Jiao, L.; Regalbuto, J.R. The synthesis of highly dispersed noble and base metals on silica via strong electrostatic adsorption: I. Amorphous silica. J. Catal. 2008, 260, 329–341. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Williams, R.T. Physisorption hysteresis loops and the characterization of nanoporous materials. Adsorpt. Sci. Technol. 2004, 22, 773–782. [Google Scholar] [CrossRef]
- Xu, B.-Q.; Wei, J.-M.; Wang, H.-Y.; Sun, K.-Q.; Zhu, Q.-M. Nano-MgO: Novel preparation and application as support of Ni catalyst for CO2 reforming of methane. Catal. Today 2001, 68, 217–225. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, J.; Wu, Y.; Pan, J.; Zhang, Q.; Tan, Y.; Han, Y. Insight into the effects of the oxygen species over Ni/ZrO2 catalyst surface on methane reforming with carbon dioxide. Appl. Catal. B Environ. 2019, 244, 427–437. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, Q.; Zhang, T.; Wang, Y.; Wang, J.; Long, K.; Song, Z.; Liu, X.; Ning, P. Facile one-pot synthesis of highly dispersed Ni nanoparticles embedded in HMS for dry reforming of methane. Chem. Eng. J. 2017, 313, 1370–1381. [Google Scholar] [CrossRef]
- Xie, T.; Shi, L.; Zhang, J.; Zhang, D. Immobilizing Ni nanoparticles to mesoporous silica with size and location control via a polyol-assisted route for coking- and sintering-resistant dry reforming of methane. Chem. Commun. 2014, 50, 7250–7253. [Google Scholar] [CrossRef] [PubMed]
- Rabelo-Neto, R.C.; Sales, H.B.E.; Inocêncio, C.V.M.; Varga, E.; Oszko, A.; Erdohelyi, A.; Noronha, F.B.; Mattos, L.V. CO2 reforming of methane over supported LaNiO3 perovskite-type oxides. Appl. Catal. B Environ. 2018, 221, 349–361. [Google Scholar] [CrossRef]
- Silva, C.G.; Passos, F.B.; da Silva, V.T. Influence of the support on the activity of a supported nickel-promoted molybdenum carbide catalyst for dry reforming of methane. J. Catal. 2019, 375, 507–518. [Google Scholar] [CrossRef]
- Gili, A.; Schlicker, L.; Bekheet, M.F.; Görke, O.; Penner, S.; Grünbacher, M.; Götsch, T.; Littlewood, P.; Marks, T.J.; Stair, P.C.; et al. Surface carbon as a reactive intermediate in dry reforming of methane to syngas on a 5% Ni/MnO catalyst. ACS Catal. 2018, 8, 8739–8750. [Google Scholar] [CrossRef]
- Jabbour, K.; Massiani, P.; Davidson, A.; Casale, S.; el Hassan, N. Ordered mesoporous “one-pot” synthesized Ni-Mg(Ca)-Al2O3 as effective and remarkably stable catalysts for combined steam and dry reforming of methane (CSDRM). Appl. Catal. B Environ. 2017, 201, 527–542. [Google Scholar] [CrossRef]

| Catalysts | Ni Loading (wt.%) a | Rate (Mol. gNi−1.h−1) | TOF (s−1) | T (°C) | References | ||
|---|---|---|---|---|---|---|---|
| CH4 | CO2 | CH4 | CO2 | ||||
| 0.5Ni1/HAP | 0.51 | 816.5 | 1186.2 | 13.3 | 19.3 | 750 | This work |
| 5Ni/HAP | 4.14 | 339.7 | 414.4 | 11.1 | 14.4 | 750 | This work |
| 10Ni/HAP | 6.98 | 185.2 | 238.6 | 12.1 | 15.5 | 750 | This work |
| Ni/MgO | 7.1 | 16.6 | - | 2.4 | - | 757 | Ref [38] |
| Ni/ZrO2 (3NZH) | 3 | 11.09 | - | 3.32 | - | 700 | Ref [39] |
| Ni/HMS | 6.21 | - | - | 4.98 | 700 | Ref [40] | |
| Ni/ZeO2 | 0.32 | - | - | 29.5 | 37.8 | 800 | Ref [18] |
| Ni/TiO2 | 0.21 | - | - | 12.9 | 18.1 | 800 | Ref [18] |
| Ni/SBA-15-EG | 6.1 | 0.51 | - | 0.06 | - | 450 | Ref [41] |
| LaNiO3 | 23.7 | 1.08 | - | 0.87 | - | 550 | Ref [42] |
| LaNiO3/Al2O3 | 6.9 | 1.9 | - | 1.55 | - | 550 | Ref [42] |
| Ni0.4MoCx/SiC | 4.9 | - | - | 28.4 | - | 800 | Ref [43] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akri, M.; El Kasmi, A.; Batiot-Dupeyrat, C.; Qiao, B. Highly Active and Carbon-Resistant Nickel Single-Atom Catalysts for Methane Dry Reforming. Catalysts 2020, 10, 630. https://doi.org/10.3390/catal10060630
Akri M, El Kasmi A, Batiot-Dupeyrat C, Qiao B. Highly Active and Carbon-Resistant Nickel Single-Atom Catalysts for Methane Dry Reforming. Catalysts. 2020; 10(6):630. https://doi.org/10.3390/catal10060630
Chicago/Turabian StyleAkri, Mohcin, Achraf El Kasmi, Catherine Batiot-Dupeyrat, and Botao Qiao. 2020. "Highly Active and Carbon-Resistant Nickel Single-Atom Catalysts for Methane Dry Reforming" Catalysts 10, no. 6: 630. https://doi.org/10.3390/catal10060630
APA StyleAkri, M., El Kasmi, A., Batiot-Dupeyrat, C., & Qiao, B. (2020). Highly Active and Carbon-Resistant Nickel Single-Atom Catalysts for Methane Dry Reforming. Catalysts, 10(6), 630. https://doi.org/10.3390/catal10060630