Cr-Free, Cu Promoted Fe Oxide-Based Catalysts for High-Temperature Water-Gas Shift (HT-WGS) Reaction
Abstract
1. Introduction
2. Results
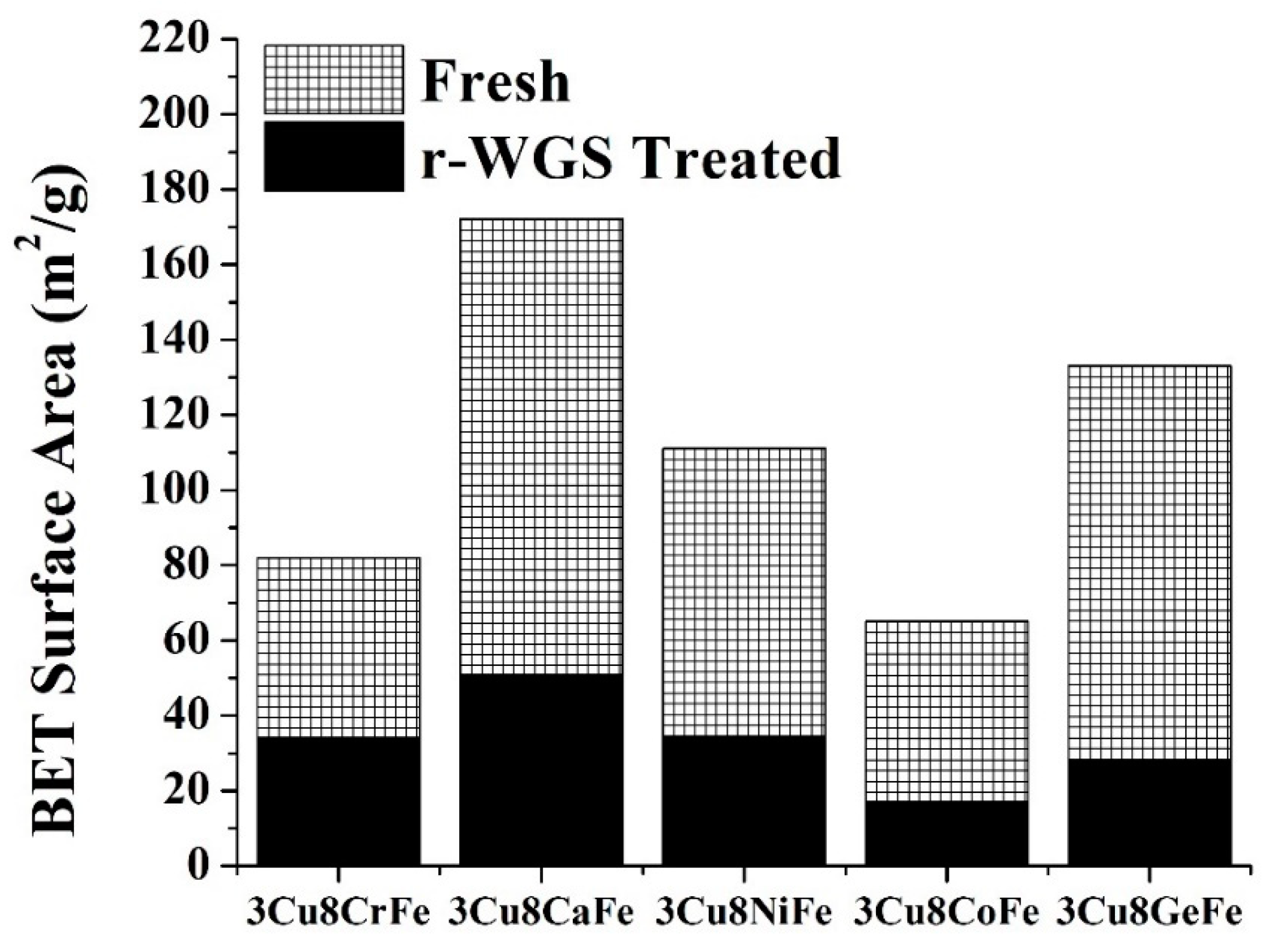
2.1. BET Surface Area
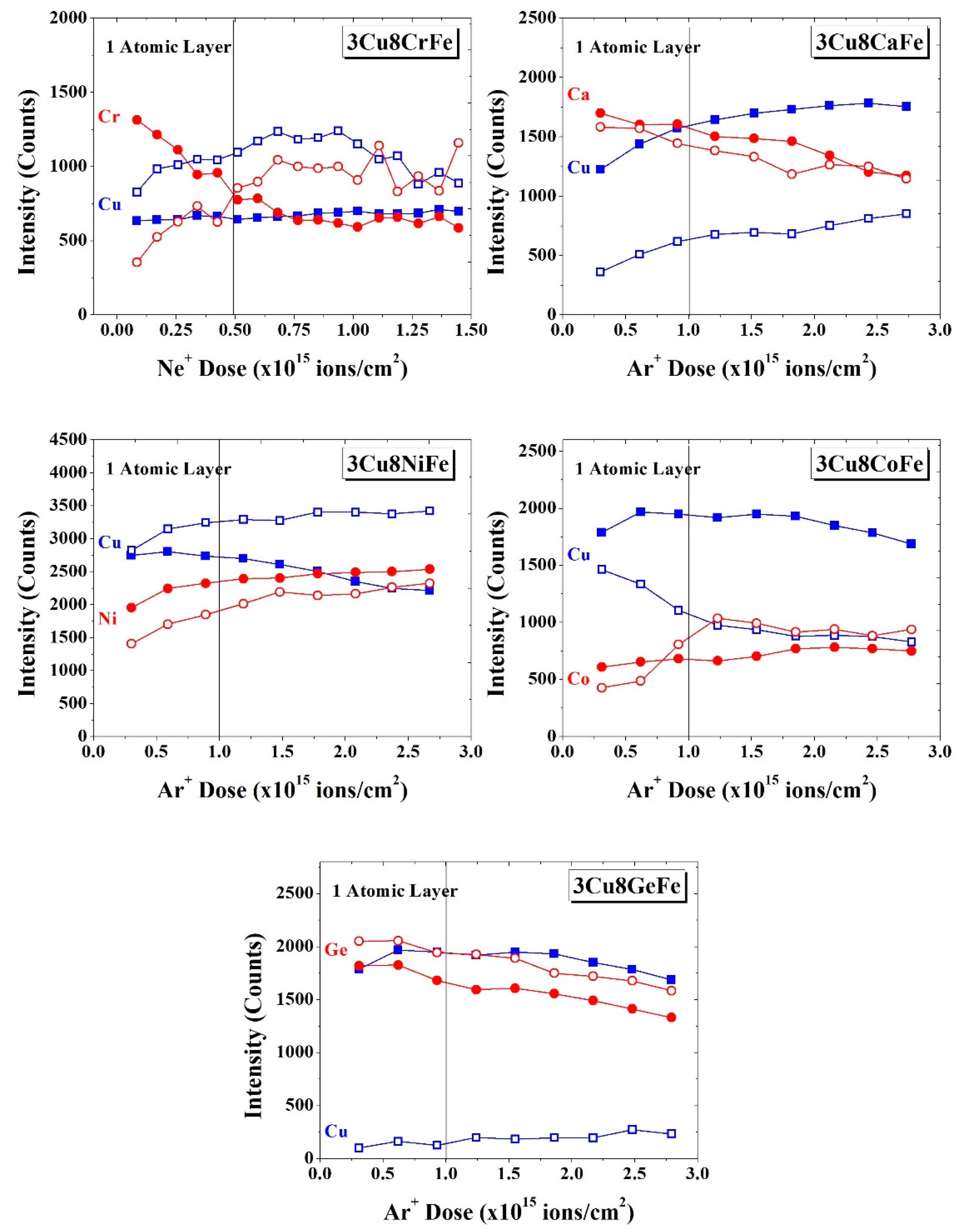
2.2. High Sensitivity Low Energy Ion Scattering (HS-LEIS)
2.3. CO-Temperature Programmed Reduction (TPR)
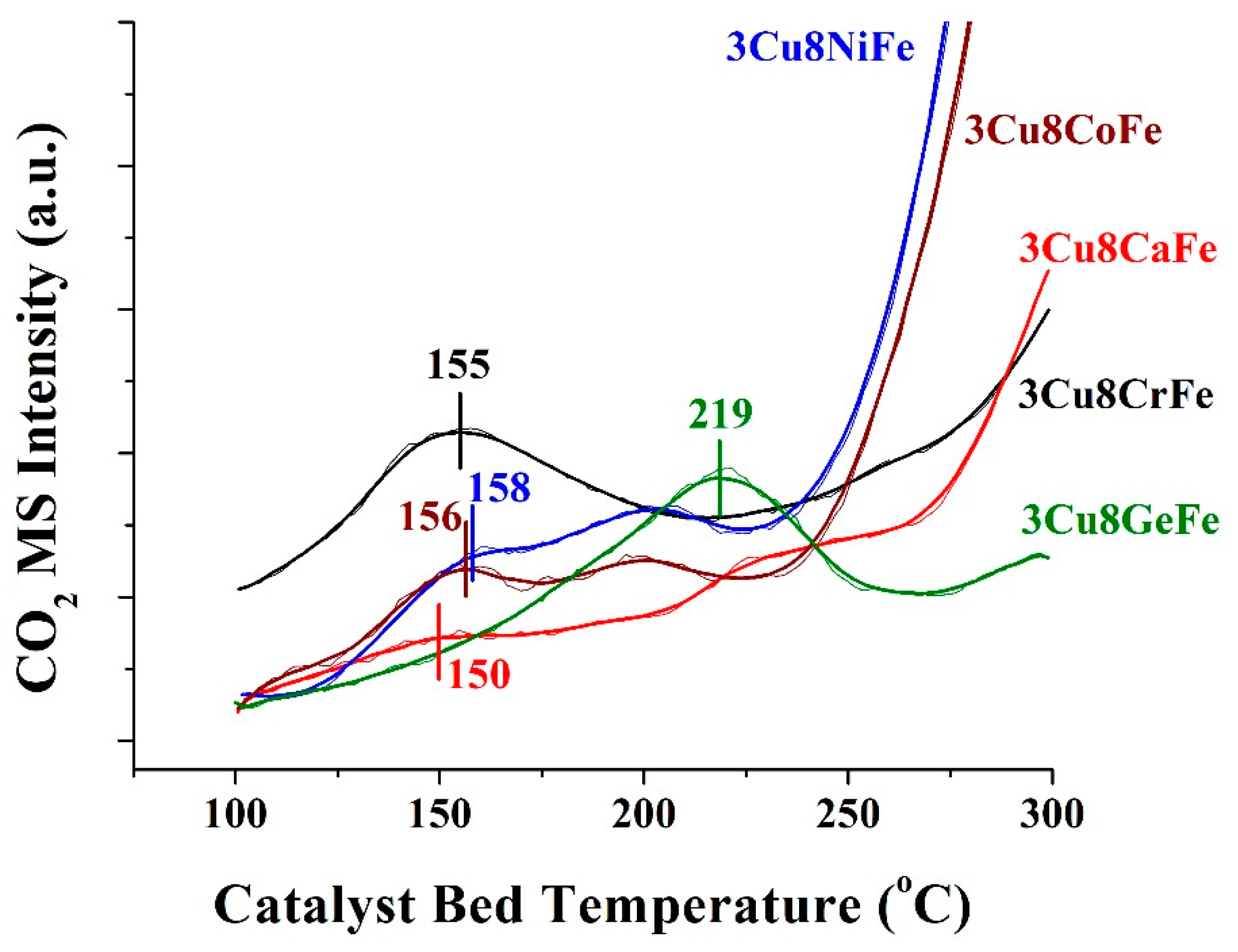
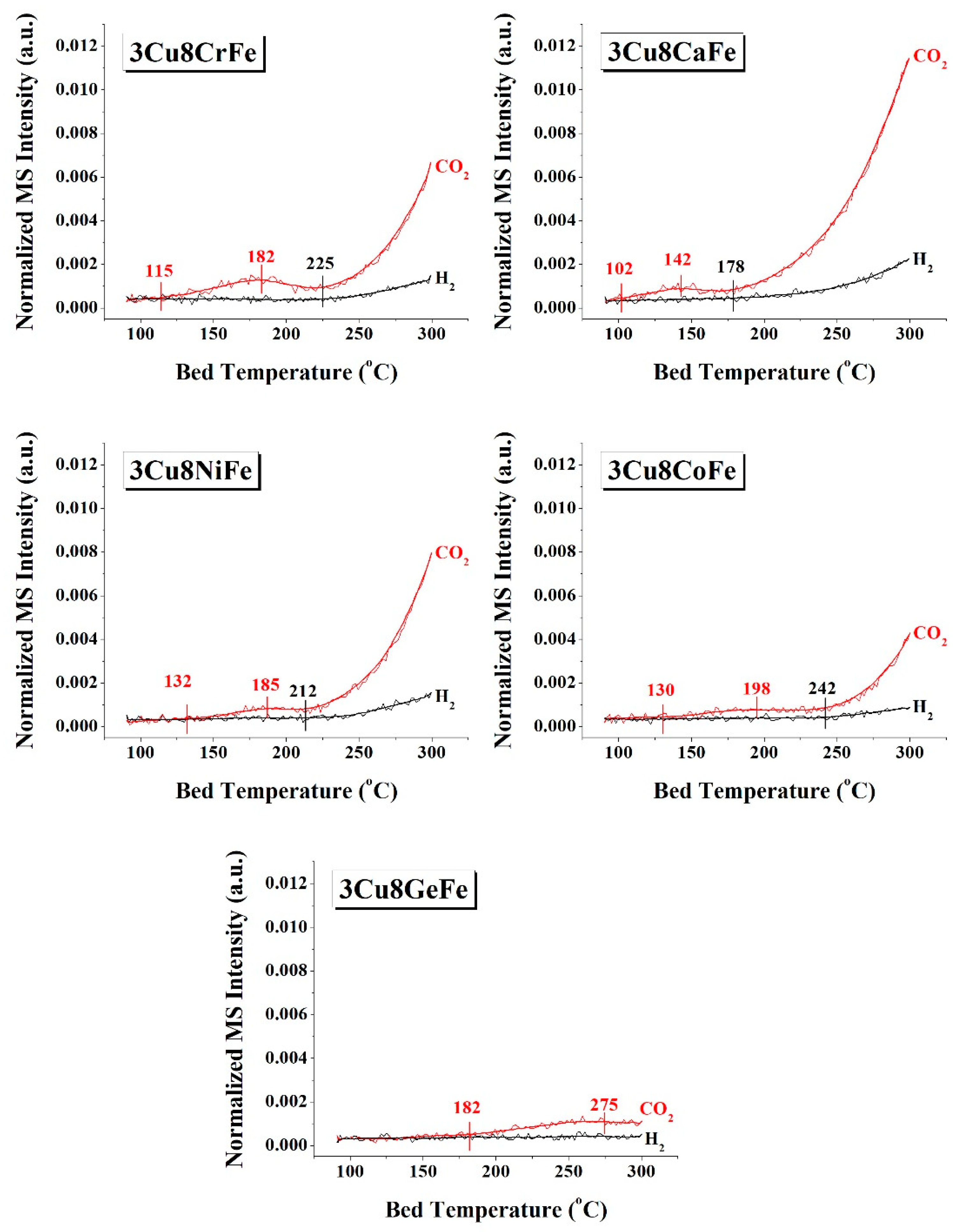
2.4. CO+H2O Temperature Programmed Surface Reaction (TPSR)
2.5. SS-WGS Activity
3. Discussion
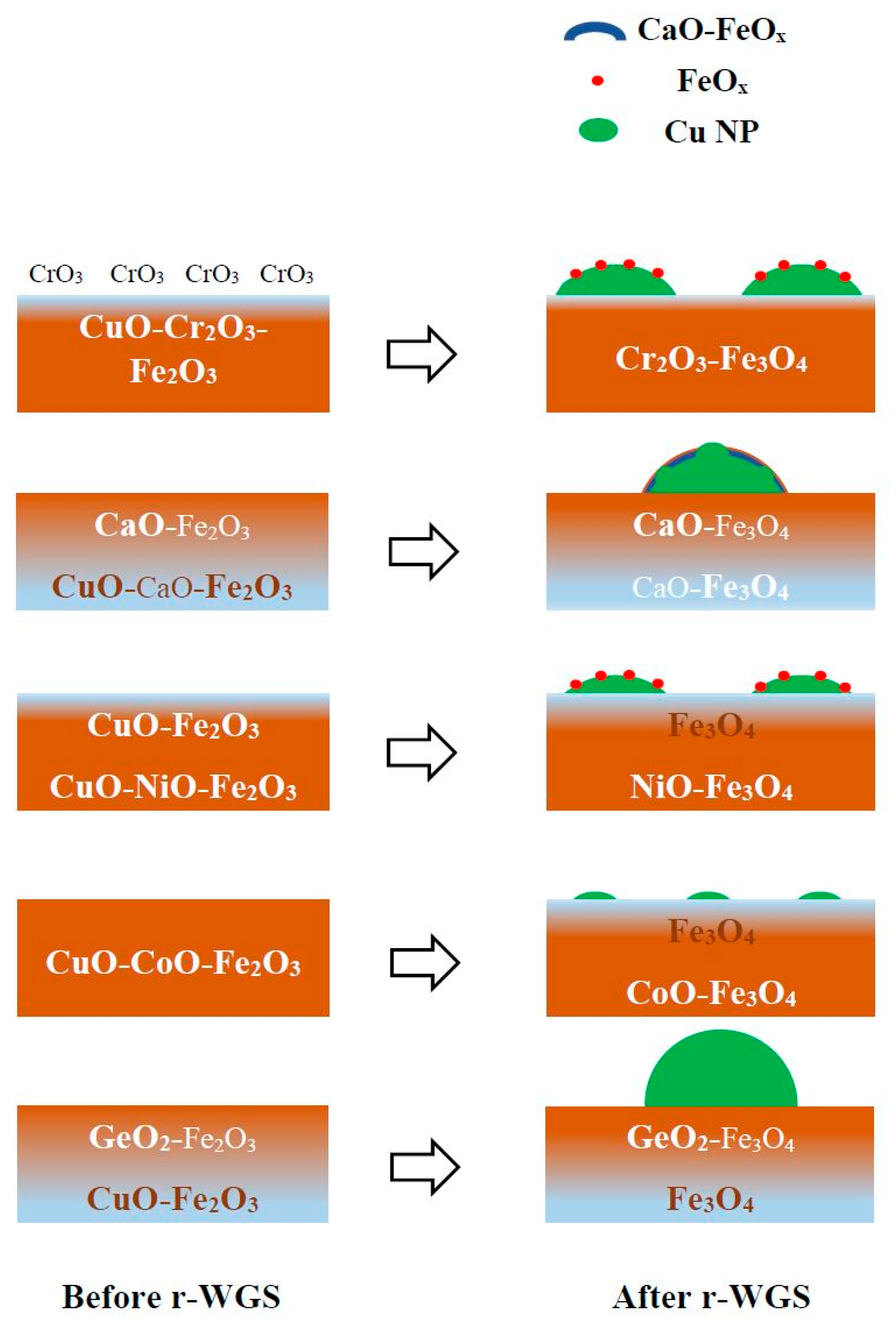
3.1. Structure of Catalysts after Calcination and Activation with r-WGS Reaction
3.2. Number of Catalytic Active Sites
3.3. Reducibility and Redox Characteristics of Catalytic Active Sites
3.4. TOF Values
3.5. HT-WGS Reaction Mechanism
- (i)
- The evolution of CO2 and H2 occur at two different temperatures (redox mechanism) rather than simultaneously being involved from a common surface reaction intermediate (associative mechanism).
- (ii)
- At a relatively high temperature, when both CO2 and H2 evolution occur, the H2 formation is always retarded, compared to CO2 production; H2/CO2 ratio is less than 1, which becomes ~1 at a much higher temperature (>500 °C) [37].
- (iii)
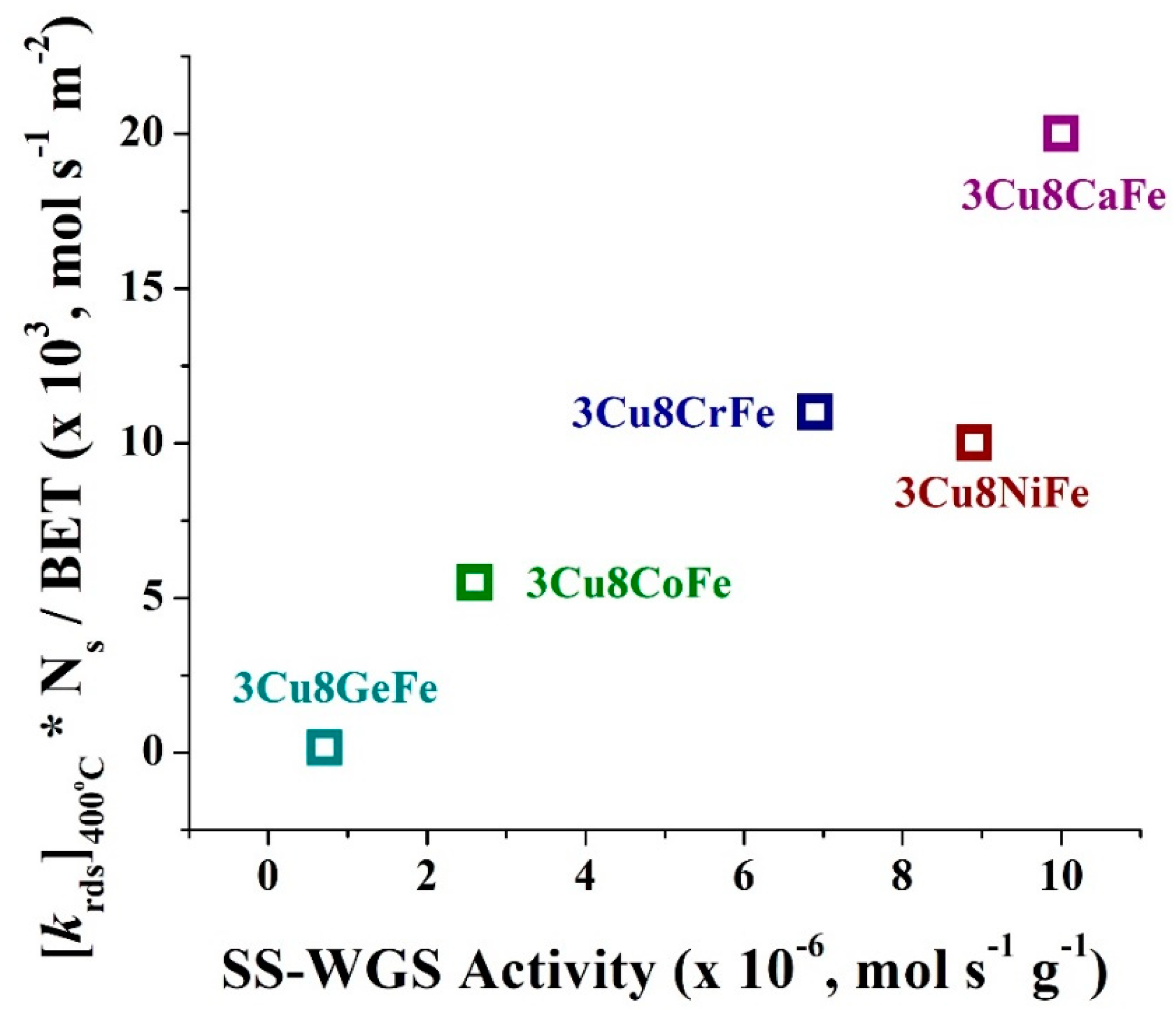
- The comparison of SS-activity performance exhibited by all the catalysts show that the activity values follow the same trend (see Figure 5) as the degree of redox ability of the active sites observed from the CO+H2O TPSR experiments, which is 3Cu8CaFe > 3Cu8NiFe ~ 3Cu8CrFe > 3Cu8CoFe >> 3Cu8GeFe.
4. Experimental
4.1. Catalyst Synthesis and Activation
4.2. Brunauer-Emmett-Teller (BET) Surface Area Determination
4.3. High-Sensitivity Low Energy Ion Scattering (HS-LEIS) Spectroscopy
4.4. CO-TPR and CO+H2O TPSR Spectroscopy
4.5. SS-WGS Activity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ridler, D.E.; Twigg, M.V. Catalyst Handbook, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1996. [Google Scholar]
- Satterfield, C.N. Heterogeneous Catalysis in Industrial Practice, 2nd ed.; McGraw Hill Book Co.: New York, NY, USA, 1991. [Google Scholar]
- Newsome, D.S. The Water-Gas Shift Reaction. Catal. Rev. 1980, 21, 275–318. [Google Scholar] [CrossRef]
- Ratnasamy, C.; Wagner, J.P. Water Gas Shift Catalysis. Catal. Rev. 2009, 51, 325–440. [Google Scholar] [CrossRef]
- Chinchen, G.C.; Logan, R.H.; Spencer, M.S. Water-Gas Shift Reaction over an Iron Oxide/Chromium Oxide Catalyst.: II: Stability of Activity. Appl. Catal. 1984, 12, 89–96. [Google Scholar] [CrossRef]
- Patlolla, A.; Carino, E.V.; Ehrlich, S.N.; Stavitski, E.; Frenkel, A.I. Application of Operando XAS, XRD, and Raman Spectroscopy for Phase Speciation in Water Gas Shift Reaction Catalysts. ACS Catal. 2012, 2, 2216–2223. [Google Scholar] [CrossRef]
- Zhu, M.; Rocha, T.C.R.; Lunkenbein, T.; Knop-Gericke, A.; Schlögl, R.; Wachs, I.E. Promotion Mechanisms of Iron Oxide-Based High Temperature Water–Gas Shift Catalysts by Chromium and Copper. ACS Catal. 2016, 6, 4455–4464. [Google Scholar] [CrossRef]
- Keturakis, C.J.; Zhu, M.; Gibson, E.K.; Daturi, M.; Tao, F.; Frenkel, A.I.; Wachs, I.E. Dynamics of CrO3–Fe2O3 Catalysts during the High-Temperature Water-Gas Shift Reaction: Molecular Structures and Reactivity. ACS Catal. 2016, 6, 4786–4798. [Google Scholar] [CrossRef]
- Zhu, M.; Yalçın, Ö.; Wachs, I.E. Revealing Structure-Activity Relationships in Chromium Free High Temperature Shift Catalysts Promoted by Earth Abundant Elements. Appl. Catal. B Environ. 2018, 232, 205–212. [Google Scholar] [CrossRef]
- Tauster, S.J. Strong Metal-Support Interactions. Acc. Chem. Res. 1987, 20, 389–394. [Google Scholar] [CrossRef]
- Zhu, M.; Tian, P.; Kurtz, R.; Lunkenbein, T.; Xu, J.; Schlögl, R.; Wachs, I.E.; Han, Y.-F. Strong Metal–Support Interactions between Copper and Iron Oxide during the High-Temperature Water-Gas Shift Reaction. Angew. Chem. Int. Ed. 2019, 58, 9083–9087. [Google Scholar] [CrossRef]
- Zhu, M.; Wachs, I.E. Determining Number of Active Sites and TOF for the High-Temperature Water Gas Shift Reaction by Iron Oxide-Based Catalysts. ACS Catal. 2016, 6, 1764–1767. [Google Scholar] [CrossRef]
- Polo-Garzon, F.; Fung, V.; Nguyen, L.; Tang, Y.; Tao, F.; Cheng, Y.; Daemen, L.L.; Ramirez-Cuesta, A.J.; Foo, G.S.; Zhu, M.; et al. Elucidation of the Reaction Mechanism for High-Temperature Water Gas Shift over an Industrial-Type Copper–Chromium–Iron Oxide Catalyst. J. Am. Chem. Soc. 2019, 141, 7990–7999. [Google Scholar] [CrossRef] [PubMed]
- Domka, F.; Łaniecki, M. The Role of Cr2O3 in Catalyst for Water-Gas Shift Reaction. Z. Anorg. Allg. Chem. 1977, 435, 273–283. [Google Scholar] [CrossRef]
- Lee, D.-W.; Lee, M.S.; Lee, J.Y.; Kim, S.; Eom, H.-J.; Moon, D.J.; Lee, K.-Y. The Review of Cr-Free Fe-Based Catalysts for High-Temperature Water-Gas Shift Reactions. Catal. Today 2013, 210, 2–9. [Google Scholar] [CrossRef]
- Zhu, M.; Wachs, I.E. Iron-Based Catalysts for the High-Temperature Water–Gas Shift (HT-WGS) Reaction: A Review. ACS Catal. 2016, 6, 722–732. [Google Scholar] [CrossRef]
- Zhu, M.; Wachs, I.E. A Perspective on Chromium-Free Iron Oxide-Based Catalysts for High Temperature Water-Gas Shift Reaction. Catal. Today 2018. [Google Scholar] [CrossRef]
- Chinchen, G.C. EP Patent EP0062410 B1, 1986.
- Rethwisch, D.G.; Dumesic, J.A. The Effects of Metal-Oxygen Bond Strength on Properties of Oxides: II. Water-Gas Shift over Bulk Oxides. Appl. Catal. 1986, 21, 97–109. [Google Scholar] [CrossRef]
- Pereira, A.L.; dos Santos, N.A.; Ferreira, M.L.; Albornoz, A.; do Carmo Rangel, M. Effect of Cobalt on the Activity of Iron-Based Catalysts in Water Gas Shift Reaction. In Natural Gas Conversion VIII.; Bellot Noronha, F., Schmal, M., Falabella Sousa-Aguiar, E., Eds.; in Studies in Surface Science and Catalysis; Elsevier: San Diego, CA, USA, 2007; Volume 167, pp. 225–230. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, D.-W.; Lee, K.-Y.; Wang, Y. Cr-Free Fe-Based Metal Oxide Catalysts for High Temperature Water Gas Shift Reaction of Fuel Processor Using LPG. Catal. Today 2009, 146, 260–264. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, D.-W.; Hong, Y.-K.; Lee, K.-Y. The CO Removal Performances of Cr-Free Fe/Ni Catalysts for High Temperature WGSR under LNG Reformate Condition without Additional Steam. Int. J. Hydrog. Energy 2011, 36, 8173–8180. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, D.-W.; Lee, M.S.; Lee, K.-Y. Cs-Promoted Ni/Fe Catalyst as a Cr-Free, High Temperature Shift Catalyst for Steam Methane Reformate without Additional Supply of Steam. Catal. Commun. 2011, 15, 37–40. [Google Scholar] [CrossRef]
- Jha, A.; Jeong, D.-W.; Shim, J.-O.; Jang, W.-J.; Lee, Y.-L.; Rode, C.V.; Roh, H.-S. Hydrogen Production by the Water-Gas Shift Reaction Using CuNi/Fe2O3 Catalyst. Catal. Sci. Technol. 2015, 5, 2752–2760. [Google Scholar] [CrossRef]
- de Araújo, G.C.; do Carmo Rangel, M. An Environmental Friendly Dopant for the High-Temperature Shift Catalysts. Catal. Today 2000, 62, 201–207. [Google Scholar] [CrossRef]
- Jeong, D.-W.; Subramanian, V.; Shim, J.-O.; Jang, W.-J.; Seo, Y.-C.; Roh, H.-S.; Gu, J.H.; Lim, Y.T. High-Temperature Water Gas Shift Reaction Over Fe/Al/Cu Oxide Based Catalysts Using Simulated Waste-Derived Synthesis Gas. Catal. Lett. 2013, 143, 438–444. [Google Scholar] [CrossRef]
- Jang, W.-J.; Shim, J.-O.; Jeon, K.-W.; Na, H.-S.; Kim, H.-M.; Lee, Y.-L.; Roh, H.-S.; Jeong, D.-W. Design and Scale-up of a Cr-Free Fe-Al-Cu Catalyst for Hydrogen Production from Waste-Derived Synthesis Gas. Appl. Catal. B Environ. 2019, 249, 72–81. [Google Scholar] [CrossRef]
- Damma, D.; Jampaiah, D.; Welton, A.; Boolchand, P.; Arvanitis, A.; Dong, J.; Smirniotis, P.G. Effect of Nb Modification on the Structural and Catalytic Property of Fe/Nb/M (M = Mn, Co, Ni, and Cu) Catalyst for High Temperature Water-Gas Shift Reaction. Catal. Today 2019. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses; John Wiley & Sons: Hoboken, NJ, USA, 2003. [Google Scholar]
- Redhead, P.A. Thermal Desorption of Gases. Vacuum 1962, 12, 203–211. [Google Scholar] [CrossRef]
- Laidler, K.J. The Development of the Arrhenius Equation. J. Chem. Educ. 1984, 61, 494. [Google Scholar] [CrossRef]
- Cornell, R.M.; Giovanoli, R. The Influence of Copper on the Transformation of Ferrihydrite (5Fe2O3 9H2O) into Crystalline Products in Alkaline Media. Polyhedron 1988, 7, 385–391. [Google Scholar] [CrossRef]
- Bernstein, L.R.; Waychunas, G.A. Germanium Crystal Chemistry in Hematite and Goethite from the Apex Mine, Utah, and Some New Data on Germanium in Aqueous Solution and in Stottite. Geochim. Cosmochim. Acta 1987, 51, 623–630. [Google Scholar] [CrossRef]
- Shannon, R.D.; Prewitt, C.T. Effective Ionic Radii in Oxides and Fluorides. Acta Crystallogr. Sect. B 1969, 25, 925–946. [Google Scholar] [CrossRef]
- Singh, B.; Sherman, D.M.; Gilkes, R.J.; Wells, M.; Mosselmans, J.F.W. Structural Chemistry of Fe, Mn, and Ni in Synthetic Hematites as Determined by Extended X-Ray Absorption Fine Structure Spectroscopy. Clays Clay Miner. 2000, 48, 521–527. [Google Scholar] [CrossRef]
- de Sitter, J.; Govaert, A.; de Grave, E.; Chambaere, D.; Robbrecht, G. A Mössbauer Study of Ca2+-Containing Magnetites. Phys. Status Solidi 1977, 43, 619–624. [Google Scholar] [CrossRef]
- Zhu, M.; Wachs, I.E. Resolving the Reaction Mechanism for H2 Formation from High-Temperature Water–Gas Shift by Chromium–Iron Oxide Catalysts. ACS Catal. 2016, 6, 2827–2830. [Google Scholar] [CrossRef]
| Catalyst | Number of Active Sites from CO-TPR (Ns) x10−3 (mol/g) | Kinetic Parameters from CO+H2O-TPSR | Activity (340 °C) x10−6 (mol/s g) | TOF (Activity/Ns) x10−3 (s−1) | Activation Energy (Ea) (kJ/mol) | |
|---|---|---|---|---|---|---|
| [Tp]rds (°C) | [krds]400 °C x108 (s−1) | |||||
| 3Cu8CrFe | 0.74 | 182 | 5.1 | 6.9 | 9.3 | 69 |
| 3Cu8CaFe | 0.22 | 142 | 47.5 | 10.0 | 45 | 50 |
| 3Cu8NiFe | 0.76 | 185 | 4.3 | 8.9 | 12 | 60 |
| 3Cu8CoFe | 0.45 | 198 | 2.1 | 2.6 | 5.8 | 78 |
| 3Cu8GeFe | 1.52 | 275 | 0.03 | 0.71 | 0.5 | 36 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sourav, S.; Wachs, I.E. Cr-Free, Cu Promoted Fe Oxide-Based Catalysts for High-Temperature Water-Gas Shift (HT-WGS) Reaction. Catalysts 2020, 10, 305. https://doi.org/10.3390/catal10030305
Sourav S, Wachs IE. Cr-Free, Cu Promoted Fe Oxide-Based Catalysts for High-Temperature Water-Gas Shift (HT-WGS) Reaction. Catalysts. 2020; 10(3):305. https://doi.org/10.3390/catal10030305
Chicago/Turabian StyleSourav, Sagar, and Israel E. Wachs. 2020. "Cr-Free, Cu Promoted Fe Oxide-Based Catalysts for High-Temperature Water-Gas Shift (HT-WGS) Reaction" Catalysts 10, no. 3: 305. https://doi.org/10.3390/catal10030305
APA StyleSourav, S., & Wachs, I. E. (2020). Cr-Free, Cu Promoted Fe Oxide-Based Catalysts for High-Temperature Water-Gas Shift (HT-WGS) Reaction. Catalysts, 10(3), 305. https://doi.org/10.3390/catal10030305





