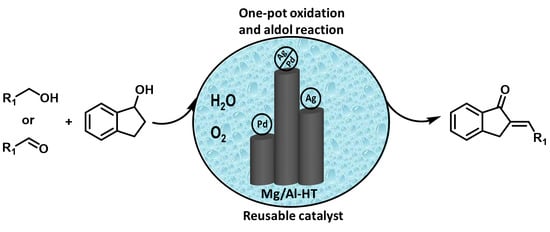
Hydrotalcite-Supported Ag/Pd Bimetallic Nanoclusters Catalyzed Oxidation and One-Pot Aldol Reaction in Water
Abstract
1. Introduction
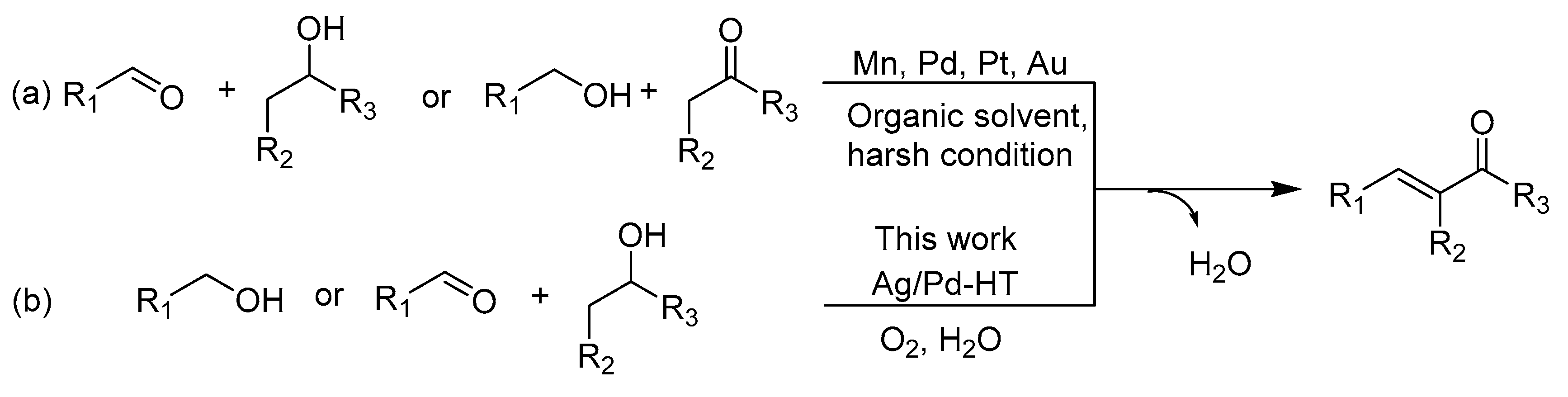
2. Results and Discussion
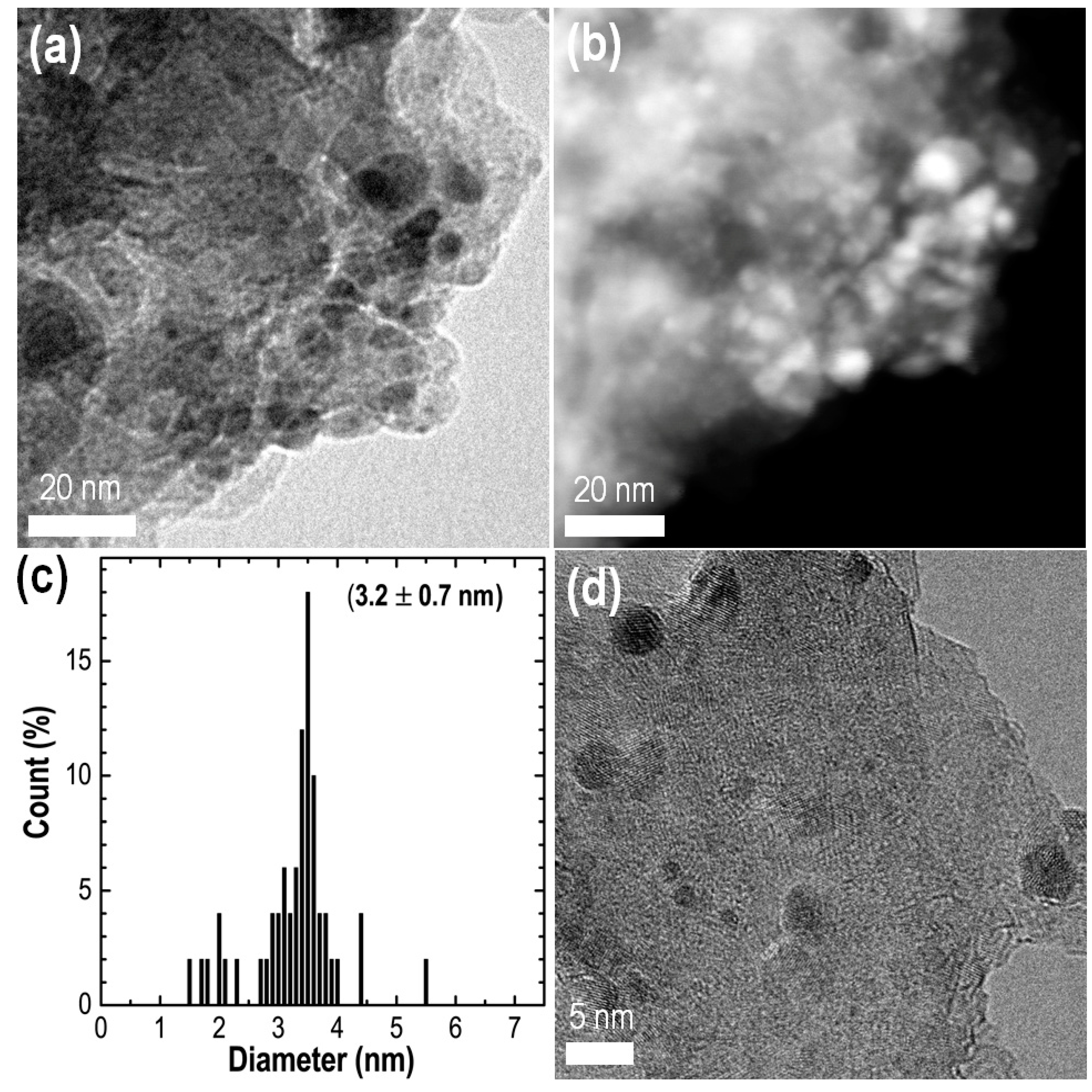
2.1. Synthesis of Catalyst
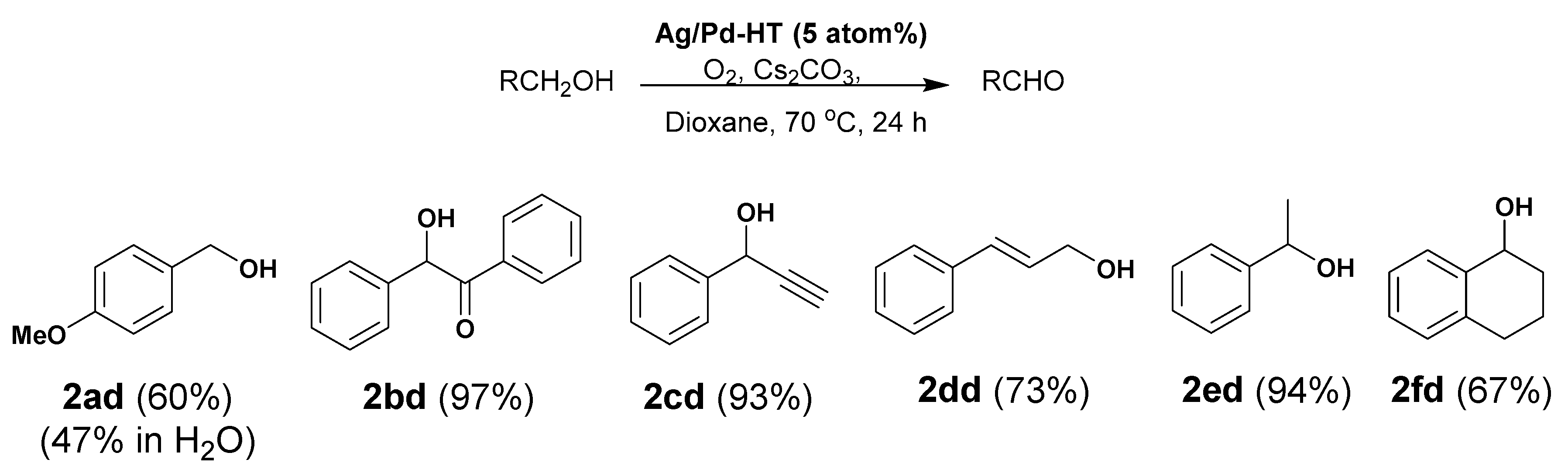
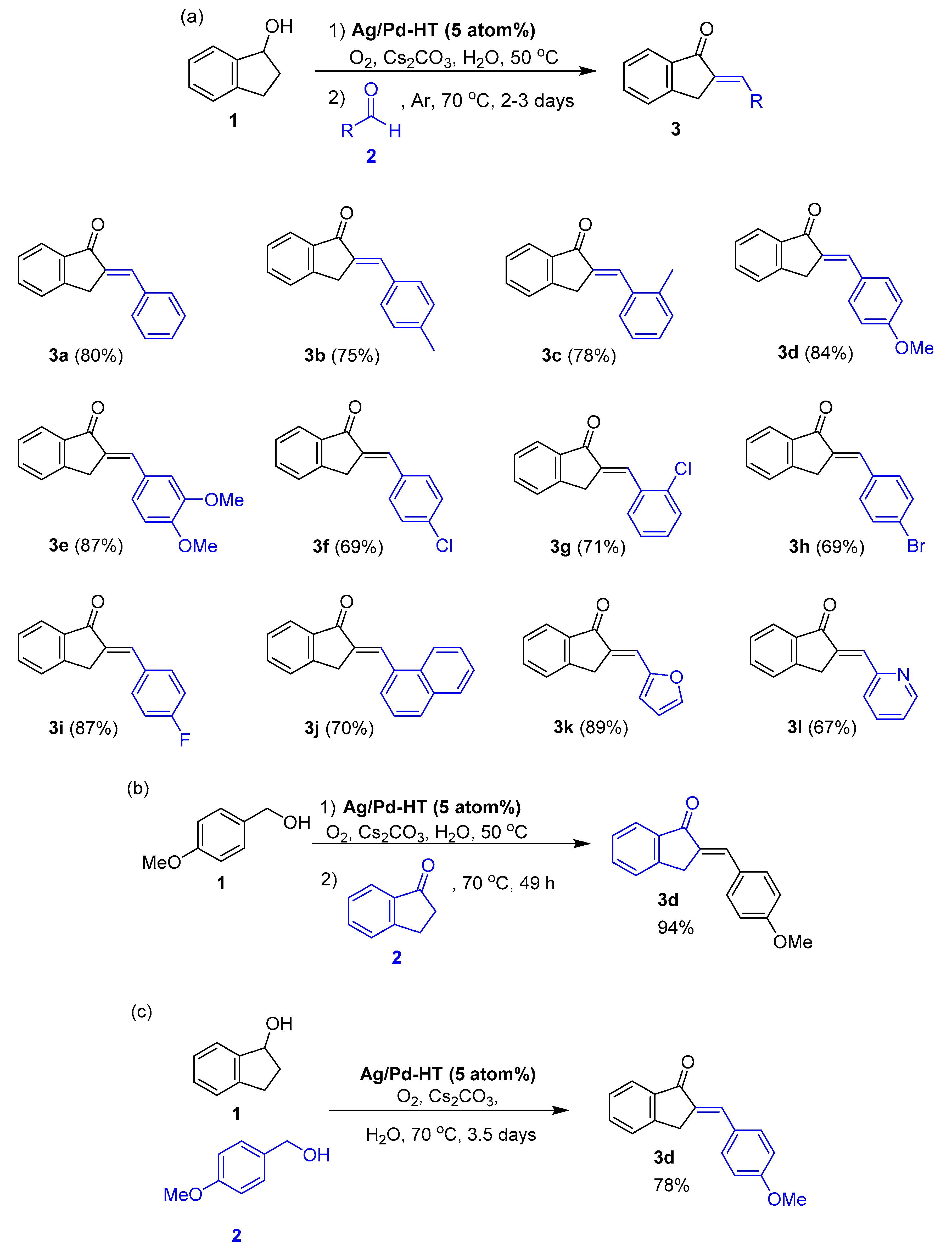
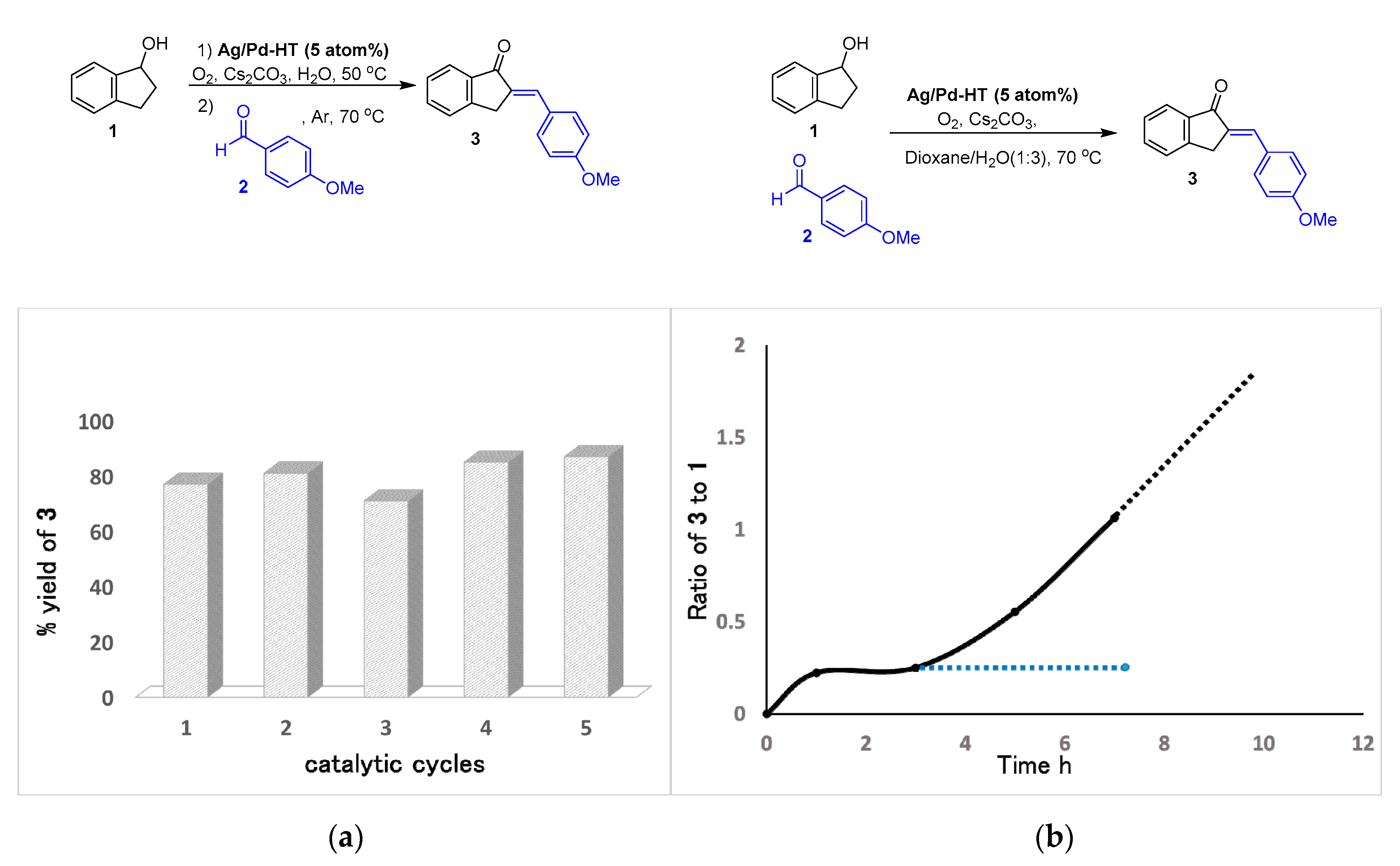
2.2. Catalytic Performance
3. Materials and Methods
3.1. General Procedure for Oxidation
3.2. General Procedure for Oxidation and One-pot Aldol Reaction in Water
3.2.1. Method A
3.2.2. Method B
3.2.3. Method C
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nel, M.S.; Petzer, A.; Petzer, J.P.; Legoabe, L.J. 2-Heteroarylidene-1-indanone derivatives as inhibitors of monoamine oxidase. Bioorgan. Chem. 2016, 69, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Varma, R.S.; Kabalka, G.W.; Evans, L.T.; Pagni, R.M. Aldol condensations on basic alumina: The facile syntheses of chalcones and enones in a solvent-free medium. Synth. Commun. 1985, 15, 279–284. [Google Scholar] [CrossRef]
- Guida, A.; Lhouty, M.H.; Tichit, D.; Figueras, F.; Geneste, P. Hydrotalcites as base catalysts. Kinetics of Claisen-Schmidt condensation, intramolecular condensation of acetonylacetone and synthesis of chalcone. Appl. Catal. 1997, 164, 251–264. [Google Scholar] [CrossRef]
- Climent, M.J.; Corma, A.; Iborra, S.; Primo, J. Base catalysis for fine chemicals production: Claisen-Schmidt condensation on zeolites and hydrotalcites for the production of chalcones and flavanones of pharmaceutical interest. J. Catal. 1995, 151, 60–66. [Google Scholar] [CrossRef]
- Solhy, A.; Tahir, R.; Sebti, S.; Skouta, R.; Bousmina, M.; Zahouily, M.; Larzek, M. Efficient synthesis of chalcone derivatives catalyzed by re-usable hydroxyapatite. Appl. Catal. A Gen. 2010, 374, 189–193. [Google Scholar] [CrossRef]
- Gawali, S.S.; Pandia, B.K.; Gunanathan, C. Manganese(I)-catalyzed α-Alkenylation of Ketones Using Primary Alcohols. Org. Lett. 2019, 21, 3842–3847. [Google Scholar] [CrossRef]
- Kwon, M.S.; Kim, N.; Seo, S.H.; Park, I.S.; Cheedrala, R.K.; Park, J. Recyclable Palladium Catalyst for Highly Selective α-Alkylation of Ketones with Alcohols. Angew. Chem. 2005, 117, 7073–7075. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Y.; Wang, M.; Lu, J.; Zhang, C.; Li, L.; Jianga, J.; Wang, F. The cascade synthesis of α,β-unsaturated ketones via oxidative C–C coupling of ketones and primary alcohols over a ceria catalyst. Catal. Sci. Technol. 2016, 6, 1693–1700. [Google Scholar] [CrossRef]
- Kim, S.; Bae, S.W.; Lee, J.S.; Park, J. Recyclable gold nanoparticle catalyst for the aerobic alcohol oxidation and C–C bond forming reaction between primary alcohols and ketones under ambient conditions. Tetrahedron 2009, 65, 1461–1466. [Google Scholar] [CrossRef]
- Kakiuchi, N.; Maeda, Y.; Nishimura, T.; Uemura, S. Pd(II)−Hydrotalcite-catalyzed oxidation of alcohols to aldehydes and ketones using atmospheric pressure of air. J. Org. Chem. 2001, 66, 6620–6625. [Google Scholar] [CrossRef]
- Karimi, B.; Abedi, S.; Clark, J.H.; Budarin, V. Highly efficient aerobic oxidation of alcohols using a recoverable catalyst: The role of mesoporous channels of SBA-15 in stabilizing palladium nanoparticle. Angew. Chem. Int. Ed. 2006, 45, 4776–4779. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.S.; Kim, N.; Park, C.M.; Lee, J.S.; Kang, K.Y.; Park, J. Palladium nanoparticles entrapped in aluminum hydroxide: Dual catalyst for alkene hydrogenation and aerobic alcohol oxidation. Org. Lett. 2005, 7, 1077–1079. [Google Scholar] [CrossRef] [PubMed]
- Uozumi, Y.; Nakao, R.; Rhee, H. Development of an amphiphilic resin-dispersion of nanopalladium catalyst: Design, preparation, and its use in aquacatalytic hydrodechlorination and aerobic oxidation. J. Organomet. Chem. 2007, 692, 420–427. [Google Scholar] [CrossRef]
- Biffis, A.; Minati, L. Efficient aerobic oxidation of alcohols in water catalysed by microgel-stabilised metal nanoclusters. J. Catal. 2005, 236, 405–409. [Google Scholar] [CrossRef]
- Lucchesi, C.; Inasaki, T.; Miyamura, H.; Matsubara, R.; Kobayashi, S. Aerobic oxidation of alcohols under mild conditions catalyzed by novel polymer-incarcerated, carbon-stabilized gold nanoclusters. Adv. Synth. Catal. 2008, 350, 1996–2000. [Google Scholar] [CrossRef]
- Sun, F.; Zhou, J.; Zhou, W.; Pan, J.; Qian, J.; He, M.; Chena, Q. Efficient aerobic oxidation of alcohols catalyzed by NiGa hydrotalcites in the absence of any additives. New J. Chem. 2018, 42, 4029–4035. [Google Scholar] [CrossRef]
- Hou, W.; Dehm, N.A.; Scott, R.W.J. Alcohol oxidations in aqueous solutions using Au, Pd, and bimetallic AuPd nanoparticle catalysts. J. Catal. 2008, 253, 22–27. [Google Scholar] [CrossRef]
- Abad, A.; Almela, C.; Corma, A.; Garcia, H. Efficient chemoselective alcohol oxidation using oxygen as oxidant. Superior performance of gold over palladium catalysts. Tetrahedron 2006, 62, 6666–6672. [Google Scholar] [CrossRef]
- Sheldon, R.A. Green and sustainable manufacture of chemicals from biomass: State of the art. Green Chem. 2014, 16, 950–963. [Google Scholar] [CrossRef]
- Deng, Q.; Xu, J.; Han, P.; Pan, L.; Wang, L.; Zhang, X.; Zou, J.-J. Efficient synthesis of high-density aviation biofuel via solvent-free aldol condensation of cyclic ketones and furanic aldehydes. Fuel Process. Technol. 2016, 148, 361–366. [Google Scholar] [CrossRef]
- West, R.M.; Liu, Z.Y.; Peter, M.; Gärtner, C.A.; Dumesic, J.A. Carbon–carbon bond formation for biomass-derived furfurals and ketones by aldol condensation in a biphasic system. J. Mol. Catal. A Chem. 2008, 296, 18–27. [Google Scholar] [CrossRef]
- Chheda, J.N.; Huber, G.W.; Dumesic, J.A. Liquid-phase catalytic processing of biomass-derived oxygenated hydrocarbons to fuels and chemicals. Angew. Chem. Int. Ed. 2007, 46, 7164–7183. [Google Scholar] [CrossRef]
- Karanjit, S.; Kashihara, M.; Nakayama, A.; Shrestha, L.K.; Ariga, K.; Namba, K. Highly active and reusable hydrotalcite-supported Pd(0) catalyst for Suzuki coupling reactions of aryl bromides and chlorides. Tetrahedron 2018, 74, 948–954. [Google Scholar] [CrossRef]
- Choudary, B.M.; Madhi, S.; Chowdari, N.S.; Kantam, M.L.; Sreedhar, B. Layered double hydroxide supported nanopalladium catalyst for Heck-, Suzuki-, Sonogashira-, and Stille-type coupling reactions of chloroarenes. J. Am. Chem. Soc. 2002, 124, 14127–14136. [Google Scholar] [CrossRef] [PubMed]
- Aramendıa, M.A.; Aviles, Y.; Borau, V.; Luque, J.M.; Marinas, J.M.; Ruiz, J.R.; Urbano, F.J. Thermal decomposition of Mg/Al and Mg/Ga layered-double hydroxides: A spectroscopic study. J. Mater. Chem. 1999, 9, 1603–1607. [Google Scholar] [CrossRef]
- Gracia, T.; Agouram, S.; Dejoz, A.; Sanchez-Royo, J.F.; Torrente-Murciano, L.; Solsona, B. Enhanced H2O2 production over Au-rich bimetallic Au–Pd nanoparticles on ordered mesoporous carbons. Catal. Today 2015, 248, 48–57. [Google Scholar] [CrossRef]
- Long, J.; Liu, H.; Wu, S.; Liao, S.; Li, Y. Selective oxidation of saturated hydrocarbons using Au–Pd alloy nanoparticles supported on metal–organic frameworks. ACS Catal. 2013, 3, 647–654. [Google Scholar] [CrossRef]
- Enache, D.I.; Edwards, J.K.; Landon, P.; Solsona-Espriu, B.; Carley, A.F.; Herzing, A.A.; Watanabe, M.; Kiely, C.J.; Knight, D.W.; Hutchings, G.J. Solvent-free oxidation of primary alcohols to aldehydes using Au-Pd/TiO2 catalysts. Science 2006, 311, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Brett, G.L.; He, Q.; Hammond, C.; Miedziak, P.J.; Dimitratos, N.; Sankar, M.; Herzing, A.A.; Conte, M.; Lopez-Sanchez, J.A.; Kiely, C.J.; et al. Selective oxidation of glycerol by highly active bimetallic catalysts at ambient temperature under base-free conditions. Angew. Chem. Int. Ed. 2011, 50, 10136–10139. [Google Scholar] [CrossRef]
- Dhital, R.N.; Kamonsatikul, C.; Somsook, E.; Bobuatong, K.; Ehara, M.; Karanjit, S.; Sakurai, H. Low-temperature carbon–chlorine bond activation by bimetallic gold/palladium alloy nanoclusters: An application to Ullmann coupling. J. Am. Chem. Soc. 2012, 134, 20250–20253. [Google Scholar] [CrossRef]
- Karanjit, S.; Jinasan, A.; Samsook, E.; Dhital, R.N.; Motomiya, K.; Sato, Y.; Tohji, K.; Sakurai, H. Significant stabilization of palladium by gold in the bimetallic nanocatalyst leading to an enhanced activity in the hydrodechlorination of aryl chlorides. Chem. Commun. 2015, 51, 12724–12727. [Google Scholar] [CrossRef] [PubMed]

| Entry | Catalyst (Atom%) | Solvent | Base | Temperature (°C) | Yield (%) b |
|---|---|---|---|---|---|
| 1 | Ag-HT (5) | dioxane | Cs2CO3 | 70 | 60 |
| 2 | Pd-HT (5) | dioxane | Cs2CO3 | 70 | 38 |
| 3 | Ag/Pd-HT(1:1) (5) | dioxane | Cs2CO3 | 70 | 93 |
| 4 | Ag/Pd-HT(3:7) (5) | dioxane | Cs2CO3 | 70 | 28 |
| 5 | Ag/Pd-HT(7:3) (5) | dioxane | Cs2CO3 | 70 | 51 |
| 6 | Ag/Pd-HT(1:1) (3) | dioxane | Cs2CO3 | 50 | 92 |
| 7 | Ag/Pd-HT(1:1) (3) | H2O | Cs2CO3 | 50 | 87 |
| 8 c | Ag/Pd-HT(1:1) (3) | H2O | Cs2CO3 | 50 | 30 |
| 9 | HT | H2O | Cs2CO3 | 50−70 | ― |
| 10 | No catalyst | H2O | Cs2CO3 | 50−70 | ― |
| 11 | Ag/Pd-HT(1:1) (3) | H2O | NaOH | 50 | 90 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karanjit, S.; Tamura, A.; Kashihara, M.; Ushiyama, K.; Shrestha, L.K.; Ariga, K.; Nakayama, A.; Namba, K. Hydrotalcite-Supported Ag/Pd Bimetallic Nanoclusters Catalyzed Oxidation and One-Pot Aldol Reaction in Water. Catalysts 2020, 10, 1120. https://doi.org/10.3390/catal10101120
Karanjit S, Tamura A, Kashihara M, Ushiyama K, Shrestha LK, Ariga K, Nakayama A, Namba K. Hydrotalcite-Supported Ag/Pd Bimetallic Nanoclusters Catalyzed Oxidation and One-Pot Aldol Reaction in Water. Catalysts. 2020; 10(10):1120. https://doi.org/10.3390/catal10101120
Chicago/Turabian StyleKaranjit, Sangita, Ayumu Tamura, Masaya Kashihara, Kazuki Ushiyama, Lok Kumar Shrestha, Katsuhiko Ariga, Atsushi Nakayama, and Kosuke Namba. 2020. "Hydrotalcite-Supported Ag/Pd Bimetallic Nanoclusters Catalyzed Oxidation and One-Pot Aldol Reaction in Water" Catalysts 10, no. 10: 1120. https://doi.org/10.3390/catal10101120
APA StyleKaranjit, S., Tamura, A., Kashihara, M., Ushiyama, K., Shrestha, L. K., Ariga, K., Nakayama, A., & Namba, K. (2020). Hydrotalcite-Supported Ag/Pd Bimetallic Nanoclusters Catalyzed Oxidation and One-Pot Aldol Reaction in Water. Catalysts, 10(10), 1120. https://doi.org/10.3390/catal10101120