Reduced Cytokine Release in Ex Vivo Response to Cilengitide and Cetuximab Is a Marker for Improved Survival of Head and Neck Cancer Patients
Abstract
:1. Introduction
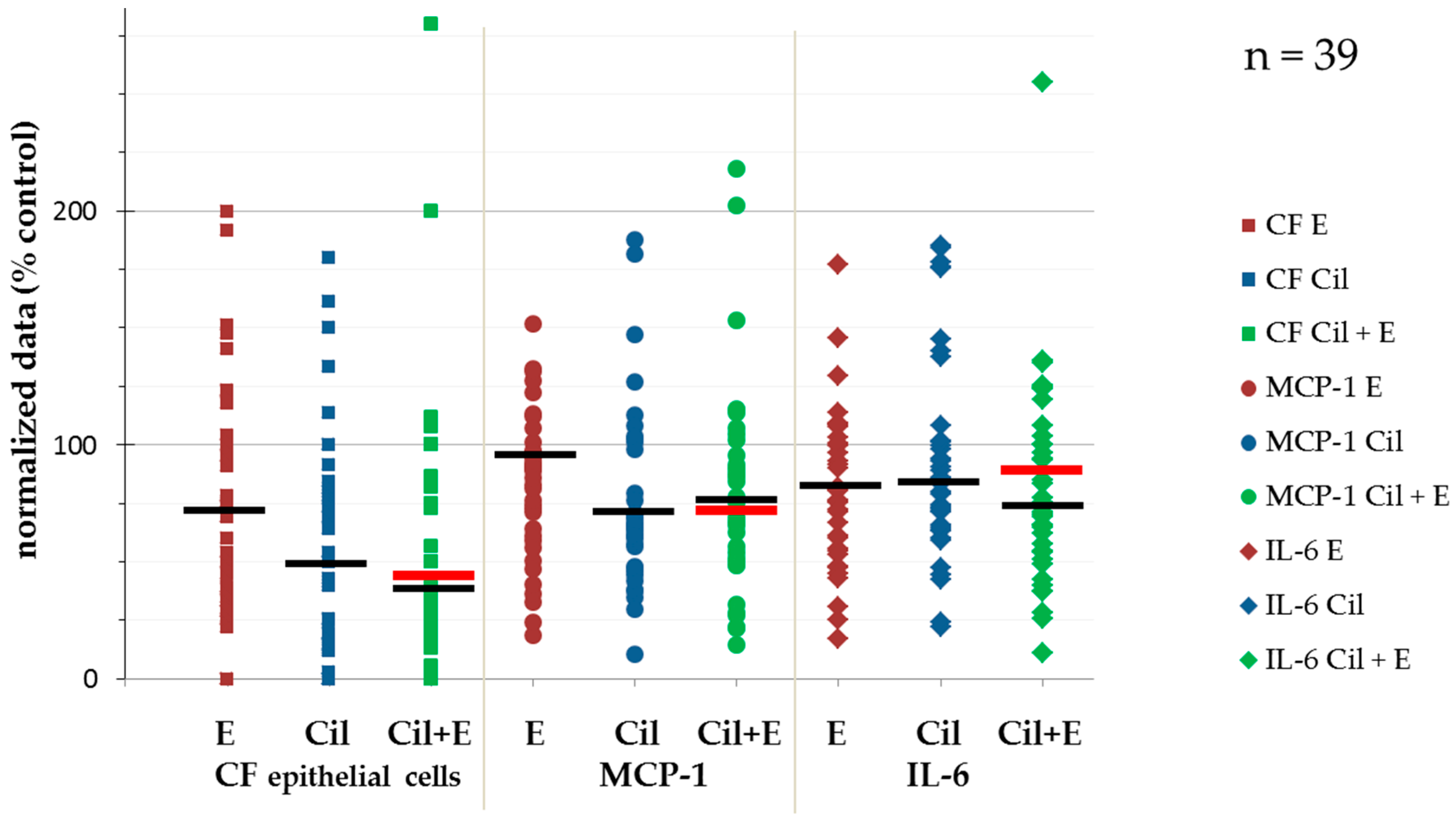
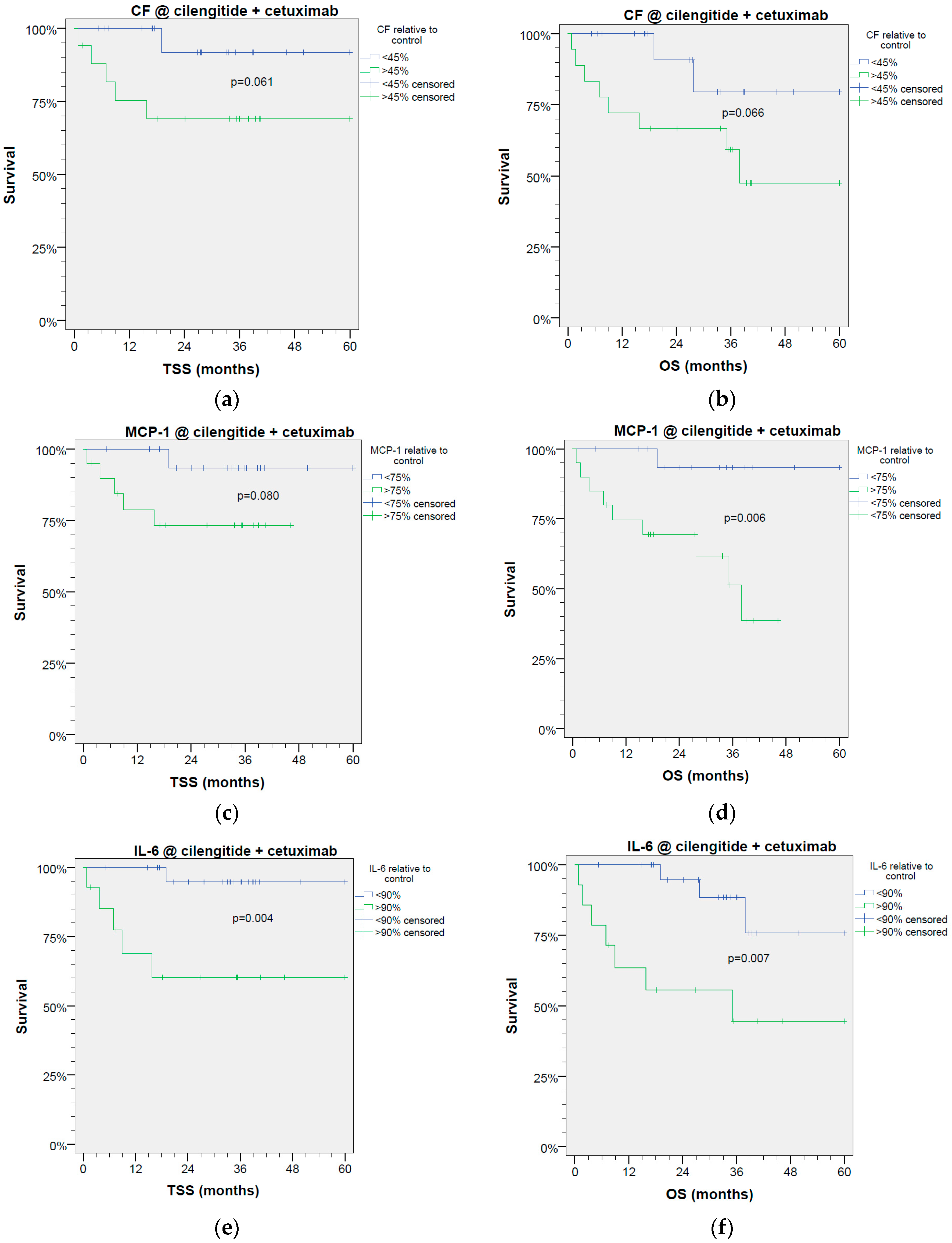
2. Results
3. Discussion
4. Materials and Methods
4.1. Patient Characteristics
4.2. FLAVINO-Assay
4.3. ELISA
4.4. Statistical Analysis
4.5. Ethical Approval
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ahmedah, H.T.; Patterson, L.H.; Shnyder, S.D.; Sheldrake, H.M. RGD-binding integrins in head and neck cancers. Cancers 2017, 9, 56. [Google Scholar] [CrossRef] [PubMed]
- Mas-Moruno, C.; Rechenmacher, F.; Kessler, H. Cilengitide: The first anti-angiogenic small molecule drug candidate design, synthesis and clinical evaluation. Anti-Cancer Agents Med. Chem. 2010, 10, 753–768. [Google Scholar] [CrossRef]
- Goodman, S.L.; Holzemann, G.; Sulyok, G.A.G.; Kessler, H. Nanomolar small molecule inhibitors for alphav(beta)6, alphav(beta)5, and alphav(beta)3 integrins. J. Med. Chem. 2002, 45, 1045–1051. [Google Scholar] [CrossRef] [PubMed]
- Dechantsreiter, M.A.; Planker, E.; Matha, B.; Lohof, E.; Holzemann, G.; Jonczyk, A.; Goodman, S.L.; Kessler, H. N-Methylated cyclic RGD peptides as highly active and selective alpha(V)beta(3) integrin antagonists. J. Med. Chem. 1999, 42, 3033–3040. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kishimoto, T. The biology and medical implications of interleukin-6. Cancer Immunol. Res. 2014, 2, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Lippitz, B.E. Cytokine patterns in patients with cancer: A systematic review. Lancet Oncol. 2013, 14, e218–e228. [Google Scholar] [CrossRef]
- Riedel, F.; Zaiss, I.; Herzog, D.; Gotte, K.; Naim, R.; Hormann, K. Serum levels of interleukin-6 in patients with primary head and neck squamous cell carcinoma. Anticancer Res. 2005, 25, 2761–2765. [Google Scholar] [PubMed]
- Nagasaki, T.; Hara, M.; Nakanishi, H.; Takahashi, H.; Sato, M.; Takeyama, H. Interleukin-6 released by colon cancer-associated fibroblasts is critical for tumour angiogenesis: Anti-interleukin-6 receptor antibody suppressed angiogenesis and inhibited tumour-stroma interaction. Br. J. Cancer 2014, 110, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, G.; Cedra, S.; Schlegel, D.; Kolb, M.; Wiegand, S.; Boehm, A.; Hofer, M.; Dietz, A. Cilengitide and cetuximab reduce cytokine production and colony formation of head and neck squamous cell carcinoma cells ex vivo. Anticancer Res. 2017, 37, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, G.; Korner, C.; Boehm, A.; Mozet, C.; Dietz, A. Stimulation by monocyte chemoattractant protein-1 modulates the ex vivo colony formation by head and neck squamous cell carcinoma cells. Anticancer Res. 2015, 35, 3917–3924. [Google Scholar] [PubMed]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte chemoattractant protein-1 (MCP-1): An overview. J. Interferon Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.-T.; Chen, H.-R.; Lin, C.-H.; Lee, J.-W.; Lee, C.-C. Monocyte chemotactic protein 1 (MCP-1) modulates pro-survival signaling to promote progression of head and neck squamous cell carcinoma. PLoS ONE 2014, 9, e88952. [Google Scholar] [CrossRef] [PubMed]
- Salcedo, R.; Ponce, M.L.; Young, H.A.; Wasserman, K.; Ward, J.M.; Kleinman, H.K.; Oppenheim, J.J.; Murphy, W.J. Human endothelial cells express CCR2 and respond to MCP-1: Direct role of MCP-1 in angiogenesis and tumor progression. Blood 2000, 96, 34–40. [Google Scholar] [PubMed]
- Wang, H.; Zhang, Q.; Kong, H.; Zeng, Y.; Hao, M.; Yu, T.; Peng, J.; Xu, Z.; Chen, J.; Shi, H. Monocyte chemotactic protein-1 expression as a prognosic biomarker in patients with solid tumor: A meta-analysis. Int. J. Clin. Expe. Pathol. 2014, 7, 3876–3886. [Google Scholar]
- Abdollahi, A.; Griggs, D.W.; Zieher, H.; Roth, A.; Lipson, K.E.; Saffrich, R.; Grone, H.-J.; Hallahan, D.E.; Reisfeld, R.A.; Debus, J.; et al. Inhibition of alpha(v)beta3 integrin survival signaling enhances antiangiogenic and antitumor effects of radiotherapy. Clin. Cancer Res. 2005, 11, 6270–6279. [Google Scholar] [CrossRef] [PubMed]
- Buerkle, M.A.; Pahernik, S.A.; Sutter, A.; Jonczyk, A.; Messmer, K.; Dellian, M. Inhibition of the alpha-nu integrins with a cyclic RGD peptide impairs angiogenesis, growth and metastasis of solid tumours in vivo. Br. J. Cancer 2002, 86, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Burke, P.A.; DeNardo, S.J.; Miers, L.A.; Lamborn, K.R.; Matzku, S.; DeNardo, G.L. Cilengitide targeting of alpha(v)beta(3) integrin receptor synergizes with radioimmunotherapy to increase efficacy and apoptosis in breast cancer xenografts. Cancer Res. 2002, 62, 4263–4272. [Google Scholar] [PubMed]
- MacDonald, T.J.; Taga, T.; Shimada, H.; Tabrizi, P.; Zlokovic, B.V.; Cheresh, D.A.; Laug, W.E. Preferential susceptibility of brain tumors to the antiangiogenic effects of an alpha(v) integrin antagonist. Neurosurgery 2001, 48, 151–157. [Google Scholar] [PubMed]
- Mitjans, F.; Meyer, T.; Fittschen, C.; Goodman, S.; Jonczyk, A.; Marshall, J.F.; Reyes, G.; Piulats, J. In vivo therapy of malignant melanoma by means of antagonists: V integrins. Int. J. Cancer 2000, 87, 716–723. [Google Scholar] [CrossRef]
- Raguse, J.-D.; Gath, H.J.; Bier, J.; Riess, H.; Oettle, H. Cilengitide (EMD 121974) arrests the growth of a heavily pretreated highly vascularised head and neck tumour. Oral Oncol. 2004, 40, 228–230. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Hegi, M.E.; Gorlia, T.; Erridge, S.C.; Perry, J.; Hong, Y.-K.; Aldape, K.D.; Lhermitte, B.; Pietsch, T.; Grujicic, D.; et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071–22072 study): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2014, 15, 1100–1108. [Google Scholar] [CrossRef]
- Wang, J.T.; Liu, Y.; Kan, X.; Liu, M.; Lu, J.G. Cilengitide, a small molecule antagonist, targeted to integrin alphanu inhibits proliferation and induces apoptosis of laryngeal cancer cells in vitro. Eur. Arch. Otorrinolaringol. 2014, 271, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Heiduschka, G.; Lill, C.; Schneider, S.; Seemann, R.; Kornek, G.; Schmid, R.; Kotowski, U.; Thurnher, D. The effect of cilengitide in combination with irradiation and chemotherapy in head and neck squamous cell carcinoma cell lines. Strahlenther. Onkol. 2014, 190, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Vermorken, J.B.; Guigay, J.; Mesia, R.; Trigo, J.M.; Keilholz, U.; Kerber, A.; Bethe, U.; Picard, M.; Brummendorf, T.H. Phase I/II trial of cilengitide with cetuximab, cisplatin and 5-fluorouracil in recurrent and/or metastatic squamous cell cancer of the head and neck: Findings of the phase I part. Br. J. Cancer 2011, 104, 1691–1696. [Google Scholar] [CrossRef] [PubMed]
- Vermorken, J.B.; Peyrade, F.; Krauss, J.; Mesia, R.; Remenar, E.; Gauler, T.C.; Keilholz, U.; Delord, J.P.; Schafhausen, P.; Erfan, J.; et al. Cisplatin, 5-fluorouracil, and cetuximab (PFE) with or without cilengitide in recurrent/metastatic squamous cell carcinoma of the head and neck: Results of the randomized phase I/II ADVANTAGE trial (phase II part). Ann. Oncol. 2014, 25, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, G.; Dietz, A. Präklinische Modelle zur Etablierung innovativer Therapiestrategien: Ex vivo-Testung der Chemo und Immunresponse von Kopf-Hals-Tumoren. HNO 2016, 64, 460–469. [Google Scholar] [CrossRef] [PubMed]
| n | (%) | n | (%) | ||
|---|---|---|---|---|---|
| Localization | Tumor Stage | ||||
| hypopharynx/larynx | 12 | (30.8%) | UICC I | 2 | (5.1%) |
| oropharynx | 24 | (61.5%) | UICC II | 6 | (15.4%) |
| nasopharynx | 1 | (2.6%) | UICC III | 7 | (17.9%) |
| oral cavity | 2 | (5.1%) | UICC IV | 24 | (61.5%) |
| T Category | Lifetime Tobacco Exposure (Pack Years, py) | ||||
| T1 | 5 | (12.8%) | 0 py | 8 | (20.5%) |
| T2 | 7 | (17.9%) | 1–20 py | 3 | (7.7%) |
| T3 | 14 | (35.9%) | 21–40 py | 14 | (35.9%) |
| T4a | 13 | (33.3%) | 41–60 py | 8 | (20.5%) |
| T4b | 0 | (0.0%) | >60 py | 4 | (10.3%) |
| no information | 2 | (5.1%) | |||
| N Category | Alcohol Consumption (g/day) | ||||
| N0 | 16 | (41.0%) | 0 | 4 | (10.3%) |
| N1 | 3 | (7.7%) | <30 | 14 | (35.9%) |
| N2a | 1 | (2.6%) | 31–60 | 8 | (20.5%) |
| N2b | 6 | (15.4%) | ≥60 | 11 | (28.2%) |
| N2c | 12 | (30.8%) | no information | 2 | (5.1%) |
| N3 | 1 | (2.6%) | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cedra, S.; Wiegand, S.; Kolb, M.; Dietz, A.; Wichmann, G. Reduced Cytokine Release in Ex Vivo Response to Cilengitide and Cetuximab Is a Marker for Improved Survival of Head and Neck Cancer Patients. Cancers 2017, 9, 117. https://doi.org/10.3390/cancers9090117
Cedra S, Wiegand S, Kolb M, Dietz A, Wichmann G. Reduced Cytokine Release in Ex Vivo Response to Cilengitide and Cetuximab Is a Marker for Improved Survival of Head and Neck Cancer Patients. Cancers. 2017; 9(9):117. https://doi.org/10.3390/cancers9090117
Chicago/Turabian StyleCedra, Susan, Susanne Wiegand, Marlen Kolb, Andreas Dietz, and Gunnar Wichmann. 2017. "Reduced Cytokine Release in Ex Vivo Response to Cilengitide and Cetuximab Is a Marker for Improved Survival of Head and Neck Cancer Patients" Cancers 9, no. 9: 117. https://doi.org/10.3390/cancers9090117
APA StyleCedra, S., Wiegand, S., Kolb, M., Dietz, A., & Wichmann, G. (2017). Reduced Cytokine Release in Ex Vivo Response to Cilengitide and Cetuximab Is a Marker for Improved Survival of Head and Neck Cancer Patients. Cancers, 9(9), 117. https://doi.org/10.3390/cancers9090117






