Human Papillomavirus Genotype Distribution in Invasive Cervical Cancer in Pakistan
Abstract
:1. Introduction
2. Results
2.1. General Characteristics
2.2. HPV Prevalence and Type Distribution
3. Discussion
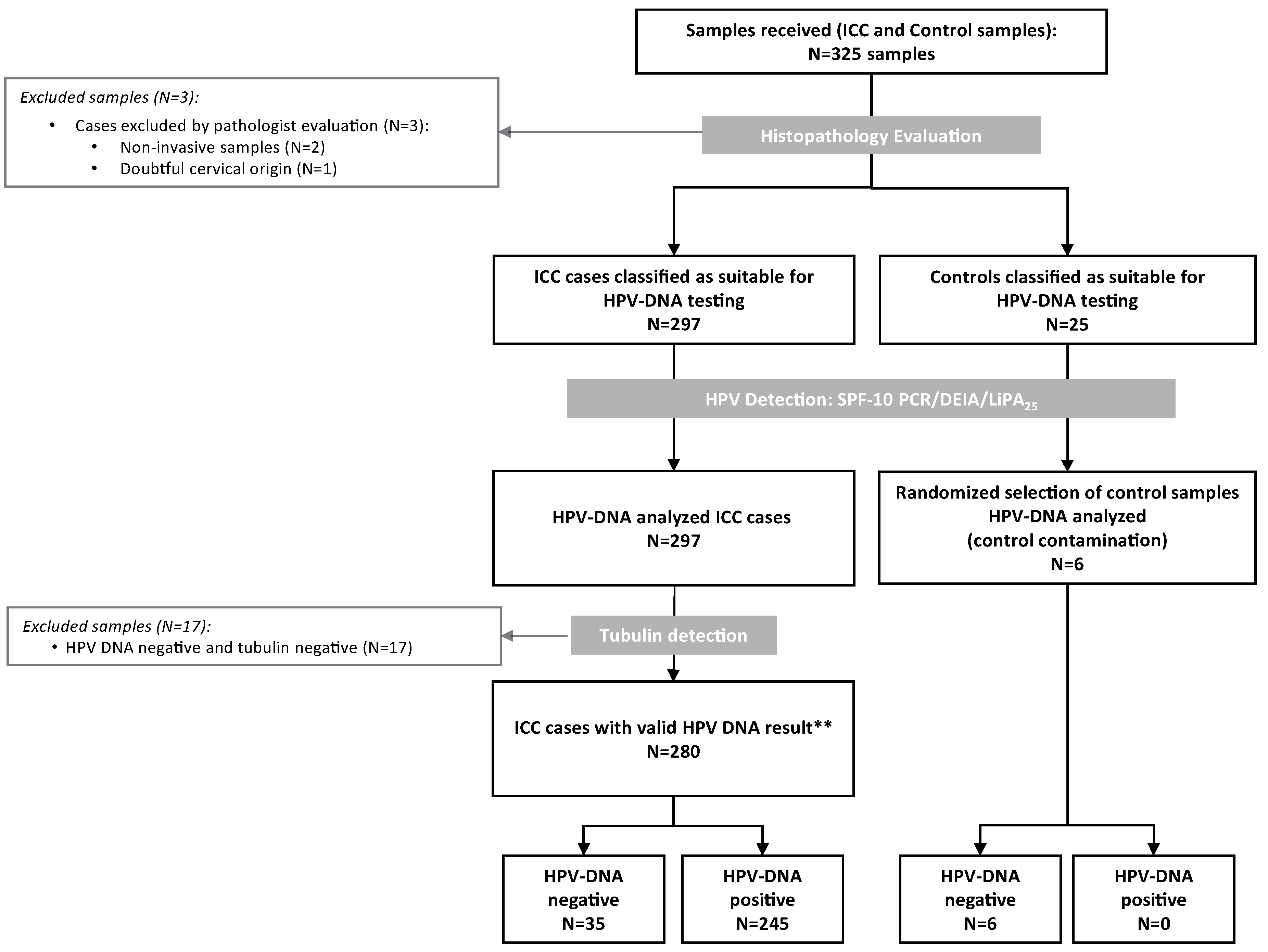
4. Materials and Methods
4.1. Study Design and Materials
4.2. Pathology and Laboratory Procedures: Formalin Fixed Paraffin Embedded Blocks Processing, Histopathological Evaluation, HPV DNA Detection and Typing
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ADC | Adenocarcinoma |
| ADSC | Adenosquamous carcinoma |
| AF | Attributional Fraction |
| CIN | Cervical Intraepithelial Neoplasia |
| DEIA | DNA Enzyme Immunoassay |
| H&E | Hematoxylin and Eosin |
| HPV | Human Papillomavirus |
| HR | High Risk |
| ICC | Invasive Cervical Cancer |
| ICO | Institut Català d’Oncologia (Catalan Institute of Oncology) |
| LR | Low Risk |
| N | Number |
| PCR | Polymerase Chain Reaction |
| RC | Relative Contribution |
| SKMCH & RC | Shaukat Khanum Memorial Cancer Hospital and Research Centre |
| SCC | Squamous Cell Carcinoma |
| Sd | Standard Deviation |
| VIA | Visual Inspection using Acetic acid |
| VLP | Virus Like Particles |
| 2vHPV | bivalent HPV vaccine |
| 4vHPV | quadrivalent HPV vaccine |
| 9vHPV | ninevalent HPV vaccine |
| 95% CI | 95% Confidence Interval |
References
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 11. International Agency for Research on Cancer: Lyon, France, 2013. Available online: http://globocan.iarc.fr (accessed on 15 February 2016).
- De Martel, C.; Ferlay, J.; Franceschi, S.; Vignat, J.; Bray, F.; Forman, D.; Plummer, M. Global burden of cancers attributable to infections in 2008: A review and synthetic analysis. Lancet Oncol. 2012, 13, 607–615. [Google Scholar] [CrossRef]
- IARC. Monographs on the Evaluation of Carcinogenic Risks to Humans. A Review of Human Carcinogens. Part B: Biological Agents, Volume 100. International Agency for Research on Cancer: Lyon, France, 2011; Available online: http://monographs.iarc.fr/ENG/Monographs/vol100B/mono100B.pdf (accessed on 15 February 2016).
- Castellsagué, X.; Díaz, M.; de Sanjosé, S.; Muñoz, N.; Herrero, R.; Franceschi, S.; Peeling, R.W.; Ashley, R.; Smith, J.S.; Snijders, P.J.F.; et al. Worldwide human papillomavirus etiology of cervical adenocarcinoma and its cofactors: Implications for screening and prevention. J. Natl. Cancer Inst. 2006, 98, 303–315. [Google Scholar]
- Walboomers, J.M.; Jacobs, M.V.; Manos, M.M.; Bosch, F.X.; Kummer, J.A.; Shah, K.V.; Snijders, P.J.F.; Peto, J.; Meijer, C.J.L.M.; Muñoz, N. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 1999, 189, 12–19. [Google Scholar] [CrossRef]
- Li, N.; Franceschi, S.; Howell-Jones, R.; Snijders, P.J.F.; Clifford, G.M. Human papillomavirus type distribution in 30,848 invasive cervical cancers worldwide: Variation by geographical region, histological type and year of publication. Int. J. Cancer. 2011, 128, 927–935. [Google Scholar] [CrossRef] [PubMed]
- De Sanjose, S.; Quint, W.G.; Alemany, L.; Geraets, D.T.; Klaustermeier, J.E.; Lloveras, B.; Tous, S.; Felix, A.; Bravo, L.E.; Shin, H.R.; et al. Human papillomavirus genotype attribution in invasive cervical cancer: A retrospective cross-sectional worldwide study. Lancet Oncol. 2010, 11, 1048–1056. [Google Scholar] [CrossRef]
- Guan, P.; Howell-Jones, R.; Li, N.; Bruni, L.; de Sanjosé, S.; Franceschi, S.; Clifford, G.M. Human papillomavirus types in 115,789 HPV-positive women: A meta-analysis from cervical infection to cancer. Int. J. Cancer. 2012, 131, 2349–2359. [Google Scholar] [CrossRef] [PubMed]
- United Nations: Department of Economic and Social Affairs, Population Division. World Population Prospects. 2015. The 2015 Revision. Available online: http://www.un.org/en/development/desa/population/events/other/10/index.shtml (Accessed on 15 February 2016).
- Gul, S.; Murad, S.; Javed, A. Prevalence of High risk Human Papillomavirus in cervical dysplasia and cancer samples from twin cities in Pakistan. Int. J. Infect. Dis. 2015, 34, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Siddiqa, A.; Zainab, M.; Qadri, I.; Bhatti, M.F.; Parish, J.L. Prevalence and genotyping of high risk human papillomavirus in cervical cancer samples from Punjab, Pakistan. Viruses 2014, 6, 2762–2777. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.A.; Franceschi, S.; Pallardy, S.; Malik, F.R.; Avan, B.I.; Zafar, A.; Pervez, S.; Serajuddaula, S.; Snijders, P.J.F.; van Kemenade, F.J.; et al. Human papillomavirus infection in women with and without cervical cancer in Karachi, Pakistan. Br. J. Cancer 2010, 102, 1657–1660. [Google Scholar] [CrossRef] [PubMed]
- Yousuf, S.; Syed, S.; Moazzam, A.; Lucky, M.H. Frequency of high risk human papillomavirus types in squamous cell carcinoma of cervix among women. J. Pak. Med. Assoc. 2010, 60, 193–196. [Google Scholar] [PubMed]
- Khan, S.; Jaffer, N.N.; Khan, M.N.; Rai, M.A.; Shafiq, M.; Ali, A.; Pervez, S.; Khan, N.; Aziz, A.; Ali, S.H. Human papillomavirus subtype 16 is common in Pakistani women with cervical carcinoma. Int. J. Infect. Dis. 2007, 11, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Sancho-Garnier, H.; Khazraji, Y.C.; Cherif, M.H.; Mahnane, A.; Hsairi, M.; El Shalakamy, A.; Osgulg, N.; Tuncerg, M.; Jumaanh, A.O.; Seoudi, M. Overview of cervical cancer screening practices in the extended Middle East and North Africa countries. Vaccine 2013, 31, G51–G57. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Barrionuevo-Rosas, L.; Albero, G.; Aldea, M.; Serrano, B.; Valencia, S.; Brotons, M.; Mena, M.; Cosano, R.; Muñoz, J.; et al. Human Papillomavirus and Related Diseases in Pakistan, Summary Report 2015-12-23; ICO Information Centre on HPV and Cancer (HPV Information Centre): Barcelona, Spain, 2015. [Google Scholar]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Ronco, G.; Dillner, J.; Elfström, K.M.; Tunesi, S.; Snijders, P.J.F.; Arbyn, M.; Kitchener, H.; Segnan, N.; Gilham, C.; Giorgi-Rossi, P.; et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: Follow-up of four European randomised controlled trials. Lancet 2014, 383, 524–532. [Google Scholar] [CrossRef]
- Arbyn, M.; Ronco, G.; Anttila, A.; Meijer, C.J.L.M.; Poljak, M.; Ogilvie, G.; Koliopoulos, G.; Naucler, P.; Sankaranarayanan, R.; Peto, J. Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer. Vaccine 2012, 30, F88–F99. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Human Papillomavirus Vaccines WHO Position Paper. World Health Organization: Geneva, Switzerland, 2014. Available online: http://www.who.int/wer/2014/wer8943.pdf?ua=1 (accessed on 15 February 2016).
- Press Announcements—FDA approves Gardasil 9 for Prevention of Certain Cancers Caused by Five Additional Types of HPV. Available online: http://www.fda.gov/newsevents/newsroom/pressannouncements/ucm426485.htm (accessed on 15 February 2016).
- European Medicines Agency—News and Events—Gardasil 9 Offers Wider Protection against Cancers Caused by Human Papillomavirus (HPV). Available online: http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2015/03/news_detail_002295.jsp&mid=WC0b01ac058004d5c1 (Accessed on 15 February 2016).
- Shepherd, J.P.; Frampton, G.K.; Harris, P. Interventions for encouraging sexual behaviours intended to prevent cervical cancer. Cochrane Database Syst. Rev. 2011, 13, CD001035. [Google Scholar]
- Badar, F.; Anwar, N. Vaccination against human papillomavirus for prevention of cervix uteri cancer in Pakistan—A public health perspective and debate. J. Ayub Med. Coll. Abbottabad. 2011, 23, 186. [Google Scholar] [PubMed]
- Insinga, R.P.; Liaw, K.L.; Johnson, L.G.; Madeleine, M.M. A systematic review of the prevalence and attribution of human papillomavirus types among cervical, vaginal, and vulvar precancers and cancers in the United States. Cancer Epidemiol. Biomarkers Prev. 2008, 17, 1611–1622. [Google Scholar] [CrossRef] [PubMed]
- Wentzensen, N.; Schiffman, M.; Dunn, T.; Zuna, R.E.; Gold, M.A.; Allen, R.A.; Zhang, R.; Sherman, M.E.; Wacholder, S.; Walker, J.; et al. Multiple human papillomavirus genotype infections in cervical cancer progression in the study to understand cervical cancer early endpoints and determinants. Int. J. Cancer 2009, 125, 2151–2158. [Google Scholar] [CrossRef] [PubMed]
- Serrano, B.; Alemany, L.; Tous, S.; Bruni, L.; Clifford, G.M.; Weiss, T.; Bosch, F.X.; de Sanjosé, S. Potential impact of a nine-valent vaccine in human papillomavirus related cervical disease. Infect. Agents Cancer 2012, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Safaeian, M.; Schiffman, M.; Gage, J.; Solomon, D.; Wheeler, C.M.; Castle, P.E. Detection of precancerous cervical lesions is differential by human papillomavirus type. Cancer Res. 2009, 69, 3262–3266. [Google Scholar] [CrossRef] [PubMed]
- Bhurgri, Y.; Nazir, K.; Shaheen, Y.; Usman, A.; Faridi, N.; Bhurgri, H.; Kayani, N.; Pervez, S.; Hasan, S.H.; Setna, F.; et al. Patho-epidemiology of Cancer Cervix in Karachi South. Asian Pac. J. Cancer Prev. 2007, 8, 357–362. [Google Scholar] [PubMed]
- The DHS Program—Pakistan: DHS, 2012-13—Final Report (English). Available online: http://dhsprogram.com/publications/publication-fr290-dhs-final-reports.cfm (accessed on 15 February 2016).
- Goldie, S.J.; O’Shea, M.; Diaz, M.; Kim, S.Y. Benefits, cost requirements and cost-effectiveness of the HPV16,18 vaccine for cervical cancer prevention in developing countries: Policy implications. Reprod. Health Matters 2008, 16, 86–96. [Google Scholar] [CrossRef]
- Human Papillomavirus Vaccine Support—Gavi, the Vaccine Alliance. Available online: http://www.gavi.org/support/nvs/human-papillomavirus/ (accessed on 15 February 2016).
- Brown, D.R.; Kjaer, S.K.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Koutsky, L.A.; Tay, E.H.; Garcia, P.; et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic non vaccine HPV types in generally HPV-naive women aged 16–26 years. J. Infect. Dis. 2009, 199, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, C.M.; Castellsagué, X.; Garland, S.M.; Szarewski, A.; Paavonen, J.; Naud, P.; Salmerón, J.; Chow, S.N.; Apter, D.; Kitchener, H.; et al. Cross-protective efficacy of HPV-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by non-vaccine oncogenic HPV types: 4-year end-of-study analysis of the randomised, double-blind PATRICIA trial. Lancet Oncol. 2012, 13, 100–110. [Google Scholar] [CrossRef]
- Malagón, T.; Drolet, M.; Boily, M.C.; Franco, E.L.; Jit, M.; Brisson, J.; Brisson, M. Cross-protective efficacy of two human papillomavirus vaccines: A systematic review and meta-analysis. Lancet Infect. Dis. 2012, 12, 781–789. [Google Scholar] [CrossRef]
- Mahmood, S.; Faraz, R.; Yousaf, A.; Asif, H.; Badar, F. Cancer Registry and Clinical Data Management (CRCDM)—Shaukat Khanum Memorial Cancer Hospital and Research Center (SKMCH & RC)—Report Based on Cancer Cases Registered at SKMCH & RC from December 1994–December 2014 and in 2014. Released June 2015. Available online: www.shaukatkhanum.org.pk (accessed on 15 May 2016).
- Alemany, L.; Saunier, M.; Alvarado-Cabrero, I.; Quirós, B.; Salmeron, J.; Shin, H.R.; Pirog, E.C.; Guimerà, N.; Hernandez-Suarez, G.; Felix, A.; et al. Human papillomavirus DNA prevalence and type distribution in anal carcinomas worldwide. Int. J. Cancer 2015, 136, 98–107. [Google Scholar] [CrossRef] [PubMed]
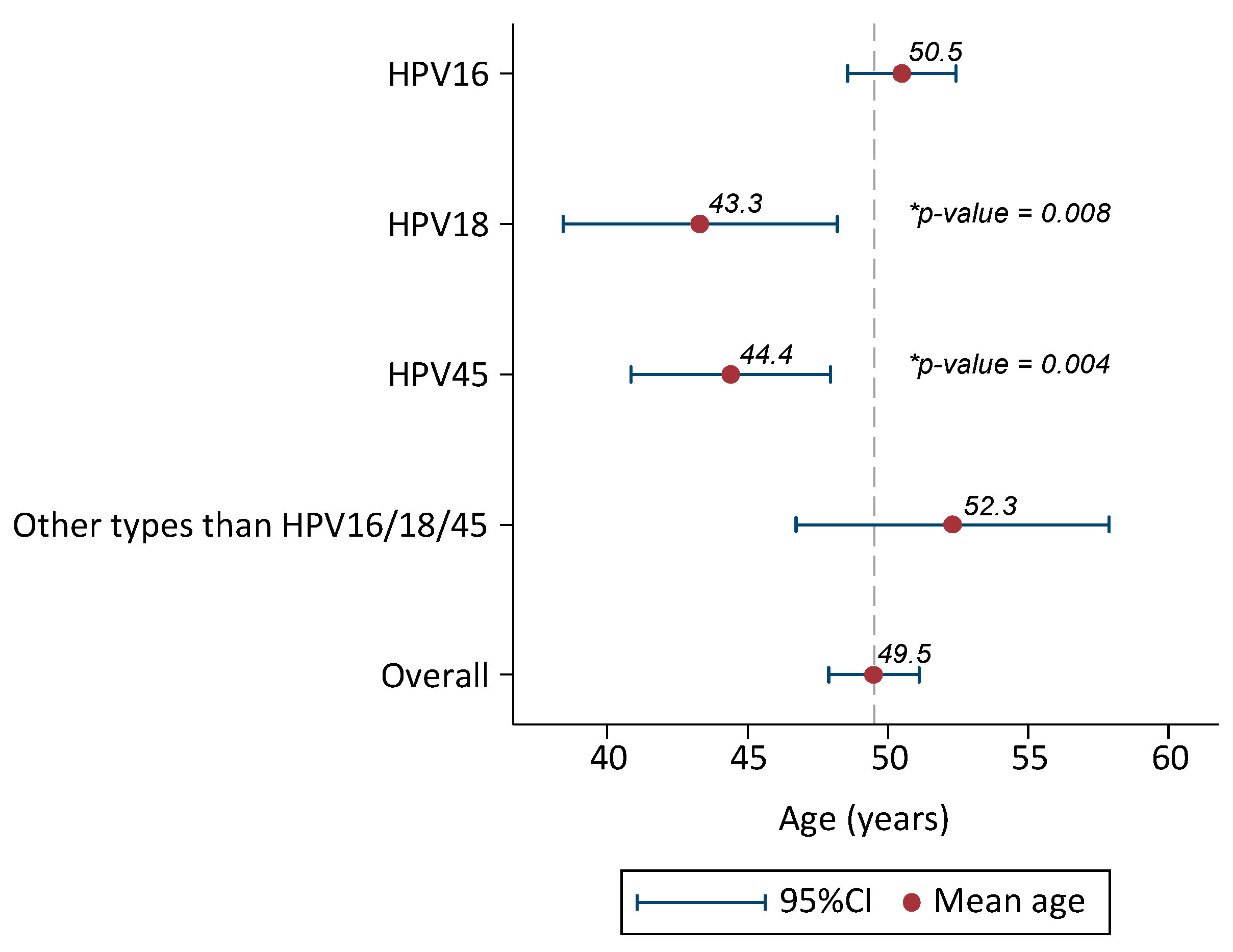
| Reference | Period of Study | Cities | Age (Years) | PCR/Primers | Tested (N) | Prevalence (%, 95CI) | Multiple Infections (%) | HPV Tested (RC%) | Details on Histology (N) |
|---|---|---|---|---|---|---|---|---|---|
| Gul et al., 2015 [10] | 2010–2013 | Islamabad, Rawalpindi | 21–80 | GP5/GP6; TS16; TS18; Beta globin | 56 | 91.1 (80.4–97.0) | Not specified | HPV16 (45.1%). HPV18 (43.1%). other types (11.8%) | SCC (33), ADC (23) |
| Siddiqa et al., 2014 [11] | 2007–2010 | Punjab | 25–70 | GP5+/GP6+; TS16; TS18; C16E7; C18E7, Beta globin | 47 | 97.9 (88.7–99.9) | 34.0 | HPV16 (32.6%). HPV18 (28.3%). HPV1618 (34.8%). other types (4.3%) | SCC (43), ADC(1); ADSC (3) |
| Raza et al., 2010 [12] | 2004–2008 | Karachi | 15–59 | GP5+/6+; Beta globin | 91 | 91.2 (83.4–96.1) | 3.3 | HPV16 (83.1%). HPV18 (7.2%). HPV33 (1.2%). HPV42 (1.2%). HPV45 (4.8%). HPV56 (2.4%). HPV59 (1.2%). HPV66 (1.2%). HPV69 (1.2%) | SCC (79), ADC (3), Small cell (4), Other (5) |
| Yousuf et al., 2010 [13] | 2003–2008 | Karachi | 25–90 | My09/My11; GP5+/6+ | 50 | 18.0 (8.6–31.4) | Not specified | HPV16 (55.6%). unknown (44.4%) | SCC (50) |
| Khan et al., 2007 [14] | 1991–2005 | Karachi | 20–60 | GP5/GP6, TS16; TS18; Beta globin | 60 | 98.3 (91.1–100) | Not specified | HPV16 (94.9%). HPV18 (1.7%). other types (3.4%) | SCC, ADC (N not specified) |
| ICC Cases | HPV-DNA Prevalence | Chi-Squared/Fisher’s Exact Test | |||
|---|---|---|---|---|---|
| Characteristics | N | % a | N+ | % b | p-value |
| Age at diagnosis | |||||
| Mean (Sd.) | 49.5 (12.4) | HPV-DNA +: 49.5 (12.5) | |||
| Year of diagnosis | |||||
| 2005 | 44 | 15.7 | 38 | 86.4 | |
| 2006 | 51 | 18.2 | 41 | 80.4 | |
| 2007 | 53 | 18.9 | 46 | 86.8 | |
| 2008 | 47 | 16.8 | 44 | 93.6 | |
| 2009 | 26 | 9.3 | 24 | 92.3 | 0.500 |
| 2010 | 59 | 21.1 | 52 | 88.1 | 0.246# |
| Histological evaluation | |||||
| Squamous cell carcinoma | 269 | 96.1 | 236 | 87.7 | |
| Adenocarcinoma | 2 | 0.7 | 1 | 50.0 | |
| Other diagnosis: | 9 | 3.2 | 8 | 88.9 | 0.304 |
| Undifferentiated carcinoma | 4 | 44.4 | 3 | 75.0 | |
| Neuroendocrine carcinoma | 3 | 33.3 | 3 | 100.0 | |
| Lymphoepithelioma-like carcinoma | 1 | 11.1 | 1 | 100.0 | |
| Trophoblastic carcinoma | 1 | 11.1 | 1 | 100.0 | |
| % invasion | |||||
| ≤50% | 96 | 34.3 | 84 | 87.5 | |
| >50% | 184 | 65.7 | 161 | 87.5 | 1.000 |
| % necrosis | |||||
| ≤50% | 275 | 98.2 | 242 | 88.0 | |
| >50% | 5 | 1.8 | 3 | 60.0 | 0.119 |
| Presence of pre-neoplastic lesions adjacent to the neoplastic lesion | |||||
| No | 270 | 96.4 | 235 | 87.0 | |
| CIN3 | 10 | 3.6 | 10 | 100.0 | 0.619 |
| Total | 280 | 100.0 | 245 | 87.5 | |
| HPV Type | ICC Cases | |
|---|---|---|
| N-Positive | RC (%) | |
| Single HPV infections | 235 | 95.9 |
| HPV16 | 165 | 67.3 |
| HPV18 | 25 | 10.2 |
| HPV45 | 18 | 7.3 |
| HPV56 | 5 | 2.0 |
| HPV68or73 | 5 | 2.0 |
| HPV31 | 3 | 1.2 |
| HPV52 | 3 | 1.2 |
| HPV33 | 2 | 0.8 |
| HPV35 | 2 | 0.8 |
| HPV39 | 2 | 0.8 |
| HPV59 | 2 | 0.8 |
| HPV66 | 2 | 0.7 |
| HPV58 | 1 | 0.4 |
| Multiple HPV infections | 2 | 0.8 |
| HPV16 & HPV18 | 1 | 0.4 |
| HPV16 & HPV45 | 1 | 0.4 |
| HPVX | 8 | 3.3 |
| Total | 245 | 100.0 |
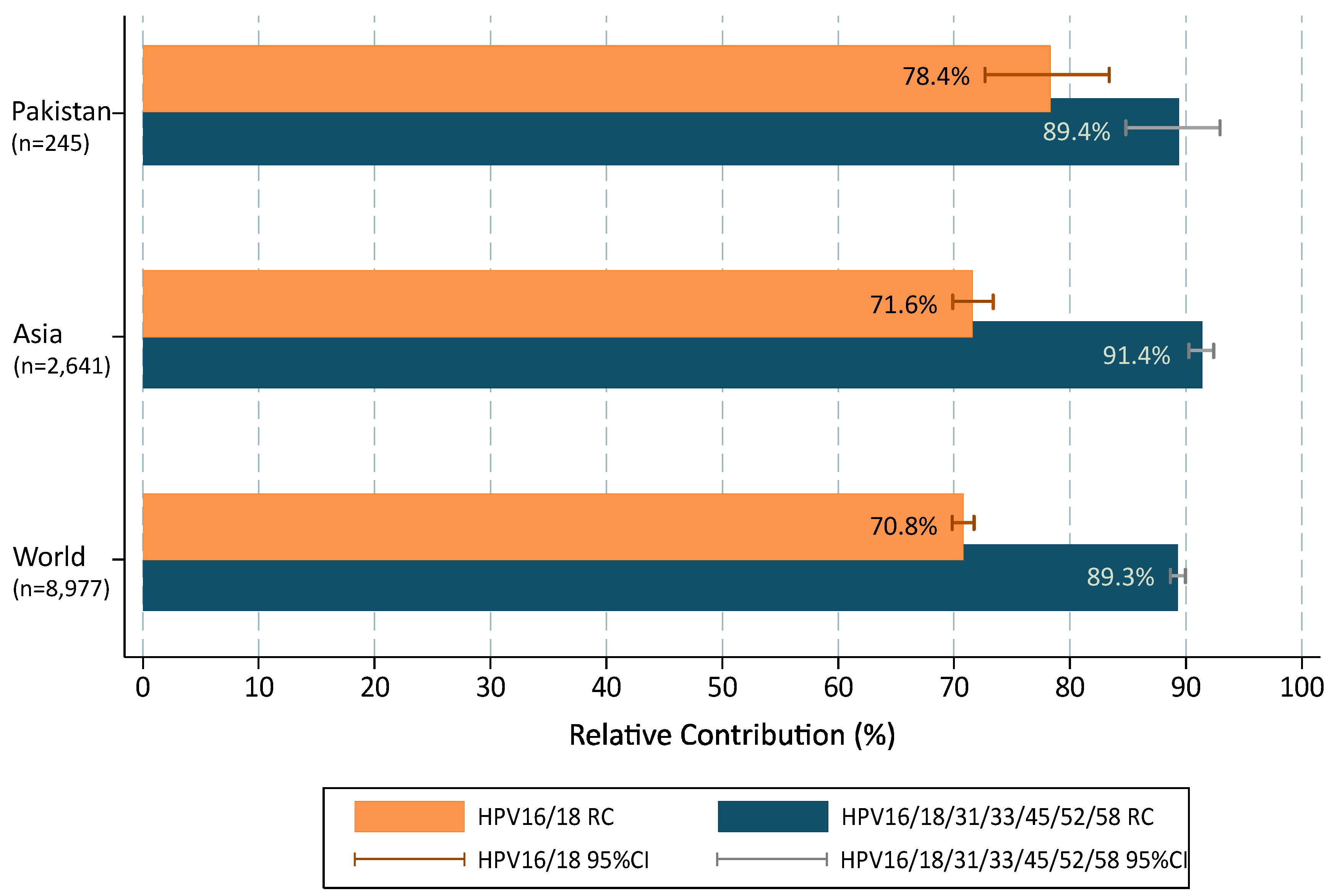
| HPV Types of ICC Cases Potentially Preventable through Vaccination | Pakistan | Asia | World | ||||||
|---|---|---|---|---|---|---|---|---|---|
| (N = 300; N-pos = 245) | (N = 2994; N-pos = 2641) a | (N = 10,575; N-pos = 8977) a | |||||||
| N-pos | RC (%) | 95%CI | N-pos | RC (%) | 95%CI | N-pos | RC (%) | 95%CI | |
| Cases infected with HPV types targeted by prophylactic vaccines (HPV16/18) | 192 | 78.4 | 72.7–83.3 | 1892 | 71.6 | 69.9–73.4 | 6357 | 70.8 | 69.9–71.8 |
| Cases infected with HPV types non-targeted by prophylactic vaccines potentially preventable by cross-protection conferred by 4vHPV vaccine (HPV31/33/35/39/45/51/52/56/58/59) (including co-infection HPV16/18) b | 12 | 5.1 | 2.6–8.4 | 212 | 8 | 7.0–9.1 | 736 | 8.2 | 7.6–8.8 |
| Cases infected with HPV types non-targeted by prophylactic vaccines potentially preventable by cross-protection conferred by 2vHPV vaccine (HPV31/33/35/39/45/51/52/56/58/59/68) (including co-infection HPV16/18) c | 24 | 9.9 | 6.4–14.2 | 381 | 14.4 | 13.1–15.8 | 1306 | 14.5 | 13.8–15.3 |
| Cases infected with HPV types targeted by prophylactic 9vHPV vaccine (HPV6/11/16/18/31/33/45/52/58) | 219 | 89.4 | 84.8–92.9 | 2416 | 91.5 | 90.4–92.5 | 8032 | 89.5 | 88.8–90.1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loya, A.; Serrano, B.; Rasheed, F.; Tous, S.; Hassan, M.; Clavero, O.; Raza, M.; De Sanjosé, S.; Bosch, F.X.; Alemany, L. Human Papillomavirus Genotype Distribution in Invasive Cervical Cancer in Pakistan. Cancers 2016, 8, 72. https://doi.org/10.3390/cancers8080072
Loya A, Serrano B, Rasheed F, Tous S, Hassan M, Clavero O, Raza M, De Sanjosé S, Bosch FX, Alemany L. Human Papillomavirus Genotype Distribution in Invasive Cervical Cancer in Pakistan. Cancers. 2016; 8(8):72. https://doi.org/10.3390/cancers8080072
Chicago/Turabian StyleLoya, Asif, Beatriz Serrano, Farah Rasheed, Sara Tous, Mariam Hassan, Omar Clavero, Muhammad Raza, Silvia De Sanjosé, F. Xavier Bosch, and Laia Alemany. 2016. "Human Papillomavirus Genotype Distribution in Invasive Cervical Cancer in Pakistan" Cancers 8, no. 8: 72. https://doi.org/10.3390/cancers8080072
APA StyleLoya, A., Serrano, B., Rasheed, F., Tous, S., Hassan, M., Clavero, O., Raza, M., De Sanjosé, S., Bosch, F. X., & Alemany, L. (2016). Human Papillomavirus Genotype Distribution in Invasive Cervical Cancer in Pakistan. Cancers, 8(8), 72. https://doi.org/10.3390/cancers8080072







