1. Introduction
Bladder cancer accounts for 3% of all malignant neoplasms, making it the 10th most common cancer (6th among men and 17th among women) [
1]. Globally, approximately 500,000 new cases of this cancer were reported in 2020. At the same time, this disease caused 2.1% of cancer-related deaths, translating into around 210,000 deaths worldwide. The incidence of bladder cancer increases with age, with a median age at diagnosis of 73 years. Approximately 98% of cases occur after the age of 45 [
2].
The key cause of bladder cancer development is carcinogenesis induced by the presence of xenobiotics in urine and their mutagenic effect on the urothelial mucosa [
3]. The primary risk factor responsible for bladder cancer development is smoking (it is estimated to account for over 50% of cases) [
4]. Other significant risk factors include occupational exposure to chemical carcinogens, particularly aromatic amines [
5].
The most common histological type of bladder cancer is urothelial carcinoma (UC), which originates from the transitional epithelium lining the urinary tract (accounting for approximately 95% of cases). Among urothelial cancers, 15–25% include subtypes (e.g., nested, micropapillary, plasmacytoid, sarcomatoid, or poorly differentiated urothelial carcinoma), as well as tumors with divergent differentiation (e.g., squamous, glandular, or trophoblastic differentiation) [
6].
Due to the uncommon nature of these histopathological variants, there are no distinct guidelines for their systemic or local treatments. Nevertheless, the progression and response to oncological therapies of rare urothelial cancer variants may vary from those of classical urothelial carcinoma.
Most bladder cancers (approximately 75%) are diagnosed at a noninvasive stage (limited to the mucosa or submucosal connective tissue without muscle layer invasion) [
7]. In the case of non-muscle invasive urothelial cancer (NMIBC), the treatment of choice is local therapy: transurethral resection of bladder tumor (TURBT) complemented, if indicated, by intravesical therapy (BCG immunotherapy or chemotherapy).
The primary treatment for muscle-invasive bladder cancer (MIBC) is radical cystectomy. Given the unsatisfactory outcomes of surgical treatment alone, attempts have been made to improve patients’ outcomes with the use of perioperative systemic therapy. The current standard approach in this context is preoperative chemotherapy. The GETUG-AFU V05 VESPER (VESPER) phase III trial (NCT01812369) has defined the reference treatment protocol for neoadjuvant chemotherapy in MIBC [
8]. This study compared the effectiveness of the cisplatin and gemcitabine combination (PG) regimen with ddMVAC (dose-dense MVAC: methotrexate, vinblastine, doxorubicin, cisplatin), demonstrating the significant superiority of the ddMVAC regimen. Results of a recently published phase III study NIAGARA (NCT03732677) demonstrated that the addition of immunotherapy (durvalumab) increases the activity of neoadjuvant chemotherapy, but the chemotherapy backbone was based on the suboptimal (PG) regimen [
9]. Therefore, the results of NIAGARA, albeit intriguing, have not dethroned ddMVAC as the neoadjuvant regimen of choice in MIBC. However, the ddMVAC regimen poses a particular logistical challenge because this regimen must be administered over two consecutive days (methotrexate on day 1; vinblastine, doxorubicin, and cisplatin on day 2), which often complicates patient compliance, requiring either overnight hospitalization or outpatient visits on two consecutive days.
Our analysis sought to assess the effectiveness and safety of a one-day ddMVAC modification, known as the accelerated MVAC (aMVAC) regimen. This approach offers a distinct opportunity to reduce the strain on patients, staff, and healthcare facilities while maintaining the activity of a classical two-day ddMVAC regimen by allowing patients to receive complete chemotherapy on day 1 within 5 h without a need for a next-day visit.
2. Materials and Methods
The retrospective analysis included 119 patients diagnosed with urothelial MIBC (diagnosed based on TURBT) who underwent preoperative chemotherapy with the aMVAC regimen. The patients were treated between 2016 and 2024 at the Clinical Department of Oncology of the University Hospital in Krakow.
The preoperative chemotherapy regimen (aMVAC) consisted of methotrexate at 30 mg/m2, vinblastine at 3 mg/m2, doxorubicin at 30 mg/m2, and cisplatin at 70 mg/m2 administered on day one of the biweekly cycles. Due to the >20% risk of febrile neutropenia, treatment was conducted with pegfilgrastim support as primary prophylaxis, administered subcutaneously at a dose of 6 mg 24–48 h after the completion of chemotherapy.
The planned treatment included 4–6 cycles of preoperative chemotherapy. In the case of intolerance or urgent clinical conditions requiring expedited surgery (e.g., bladder bleeding), systemic treatment was shortened. Following the completion of systemic therapy, patients underwent surgical treatment (radical cystectomy) at the Clinical Department of Urology and Oncological Urology of the University Hospital in Krakow.
Before the initiation of treatment, all patients underwent staging, including a computed tomography (CT) scan of the chest, abdomen, and pelvis with intravenous contrast. Due to the use of doxorubicin, baseline echocardiography was performed for all patients.
Neoadjuvant chemotherapy was given to patients diagnosed with MIBC cT2-cT4a and cN0-cN3 without distant metastases. Patients who qualified for therapy had appropriate renal function with a GFR > 60 mL/min and a left ventricular ejection fraction of at least 50% [
Table 1].
A dedicated, experienced pathologist at the Department of Pathomorphology of the University Hospital in Krakow conducted the histopathological examination of surgical specimens. The analysis of the degree of histopathological response to treatment was based on the three-grade TRG (tumor regression grade) classification developed initially by Fleischmann A et al. [
10]. In the Fleischmann scale, TRG1 represents a complete response: no tumor cells and extensive fibrosis in the tumor bed; TRG2 represents a strong (partial) response: dominant fibrosis in the tumor bed, with residual tumor tissue comprising <50% of this area; TRG3 represents a poor response or no response: dominant tumor tissue outweighing fibrosis in the tumor bed (≥50% of the area occupied by tumor cells) or no regressive changes (indicating lack of tumor regression).
At the Department of Pathomorphology of the University Hospital in Krakow, this scale was applied with a modification: changes not exceeding the basement membrane of the mucosa (pTa and pTis) were classified as TRG1.
The chi-square test was used to analyze categorical variables, and a one-way ANOVA was performed to analyze continuous variables. Unconditional maximum likelihood (Wald) was used to calculate risk ratios for categorical variables.
The analyses were performed in R software (The R Foundation for Statistical Computing, version 4.3.1). All tests were two-sided, and statistical significance was defined as p < 0.05. No correction was applied for multiple statistical tests due to this study’s exploratory nature.
4. Discussion
Neoadjuvant chemotherapy represents a standard approach in patients with urothelial MIBC. However, there is still some discussion about whether the neoadjuvant approach is indeed better than adjuvant systemic treatment. Although some analyses indicate that cancer-specific survival is similar between neoadjuvant and adjuvant approaches, the overall survival data still support the preoperative strategies [
11,
12]. Adjuvant systemic treatment in large prospective studies could not significantly improve overall survival despite the significant improvement of event-free survival [
13]. Still, a recent meta-analysis demonstrated that adjuvant chemotherapy may indeed be associated with improved overall survival [
14]. All current guidelines recommend administering dose-intensive chemotherapy regimens like ddMVAC, ddGC (dose-dense GC: gemcitabine, cisplatin), or aMVAC [
15,
16,
17]. However, the most robust data support ddMVAC, representing the only regimen to be evaluated within a phase III clinical study (GETUG-AFU-VESPER) [
8]. The accelerated MVAC regimen, which is much more feasible from logistical and patient preference points of view, is supported by much weaker data. A phase II study evaluated accelerated MVAC (aMVAC) in 44 patients with urothelial MIBC [
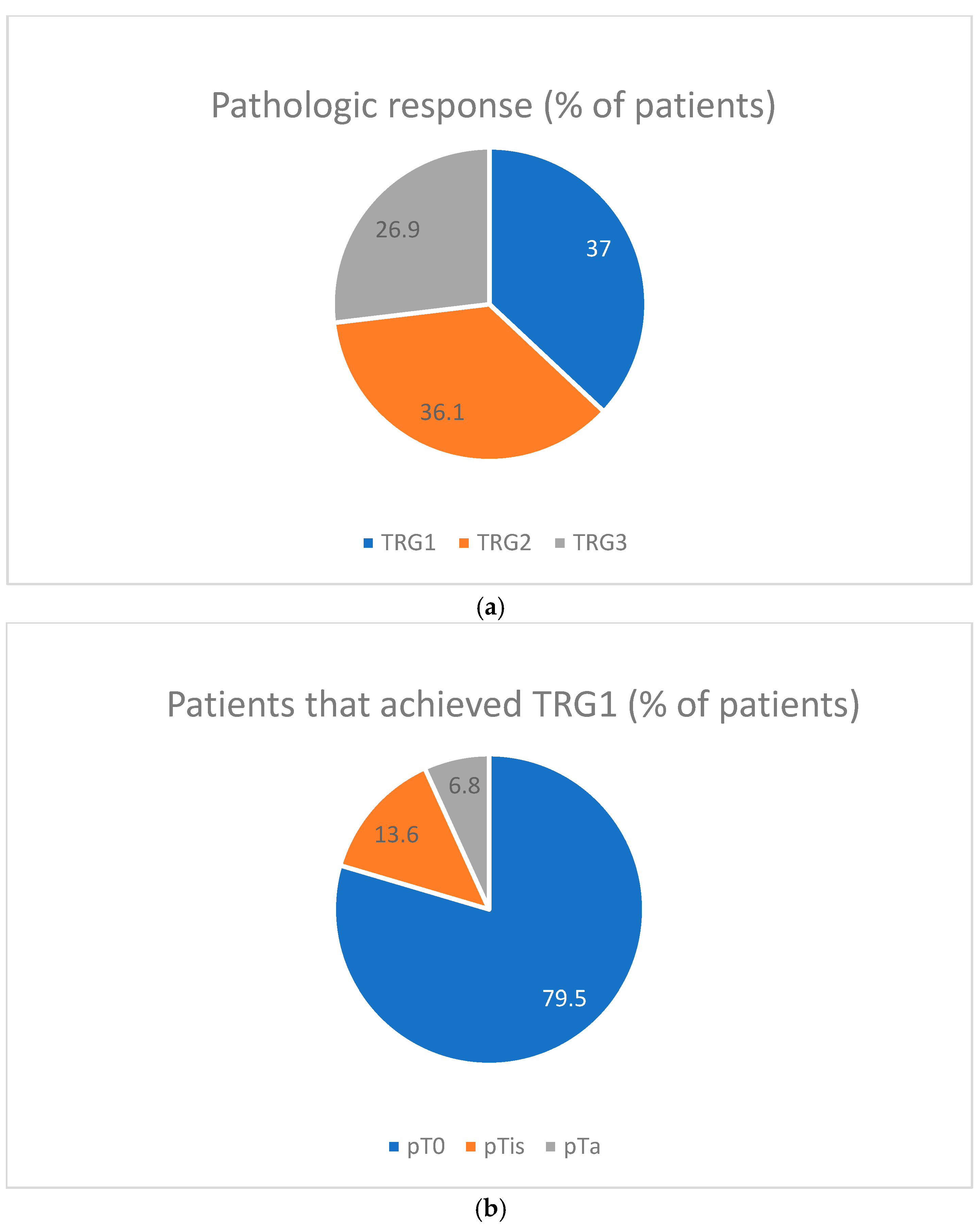
18]. Complete pathological response (pT0) to three cycles of aMVAC was observed in 38% of patients, and another 8% achieved pTis or pTa. Overall, 54% of patients who received aMVAC demonstrated downstaging to non-muscle-invasive disease <ypT2. The treatment was well tolerated, with 82% of patients experiencing only grade 1–2 treatment-related adverse events and no grade 3–4 renal toxicities. Our analysis confirms the high activity of aMVAC, albeit with a lower rate of ypT0 (29%) and <ypT2 downstaging (49%). The slightly lower rate of response to aMVAC in our study may be associated with a trial non-selected patient population reflecting a real-life clinical practice and with slight differences in the initial clinical stage of the disease as follows: cT2 40% vs. 36%, cT3 50% vs. 43%, cT4 9% vs. 14%, and N+ 14% vs. 7% for our and Plimack’s analyses, respectively. Compared to Plimack’s study, the GETUG-AFU-VESPER study, which evaluated six cycles of neoadjuvant ddMVAC, demonstrated even higher response rates (ypT0–42%, <ypT2–63%). However, the percentage of patients with initially muscle-confined MIBC (cT2) in the VESPER study was 90% compared to only 40% in our analysis.
Complete pathological response (pCR) is a strong prognostic factor for long-term outcomes in various cancers, including MIBC [
19,
20]. It provides early confirmation of the activity of neoadjuvant chemotherapy and allows for the optimization of postoperative systemic treatment. The VESPER study demonstrated that the pCR rate depends on the activity of the chemotherapy regimen (ddMVAC was superior to GC) [
8]. Cisplatin is a critically important agent used for the treatment of urothelial cancers both in curative and palliative settings [
21,
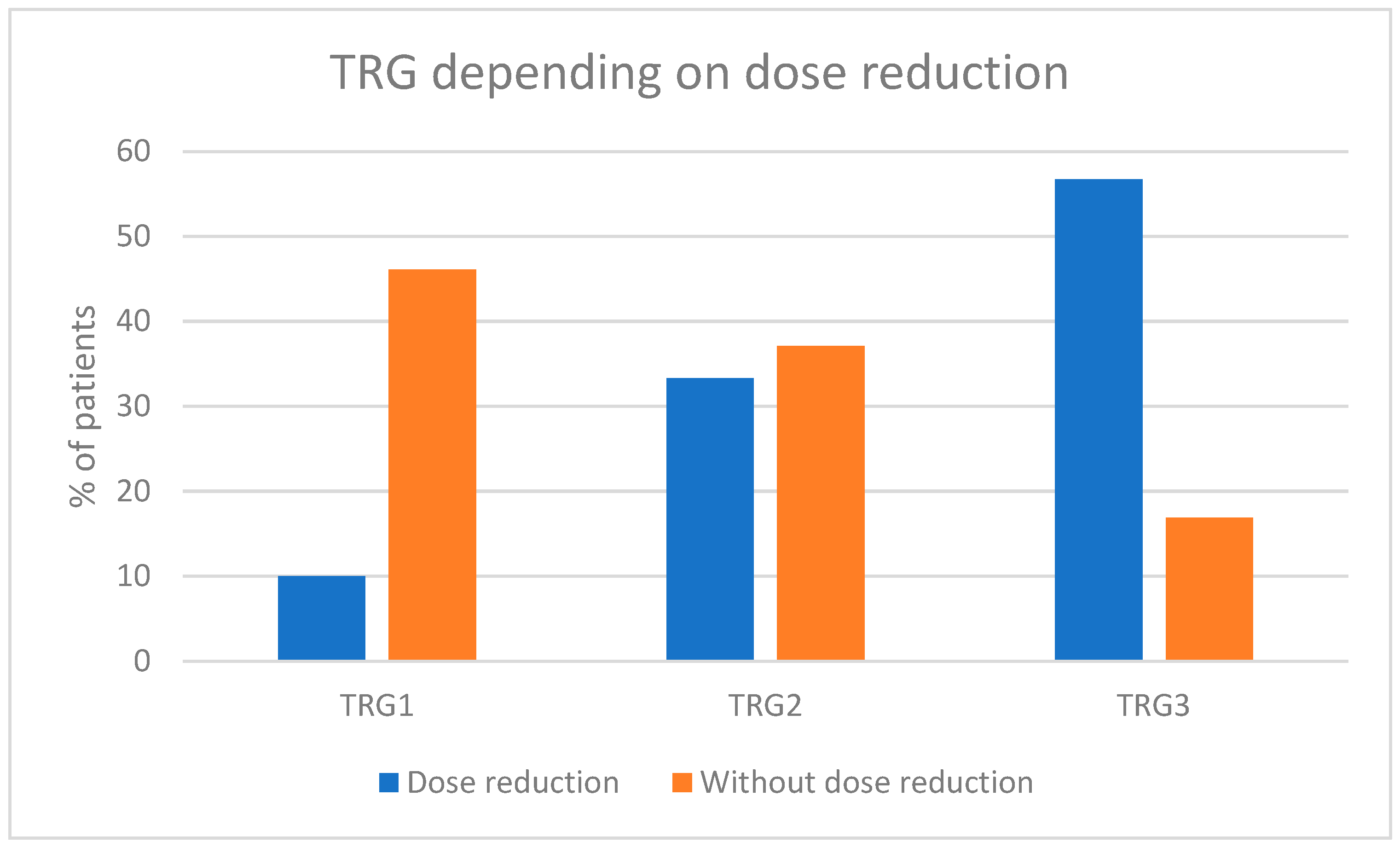
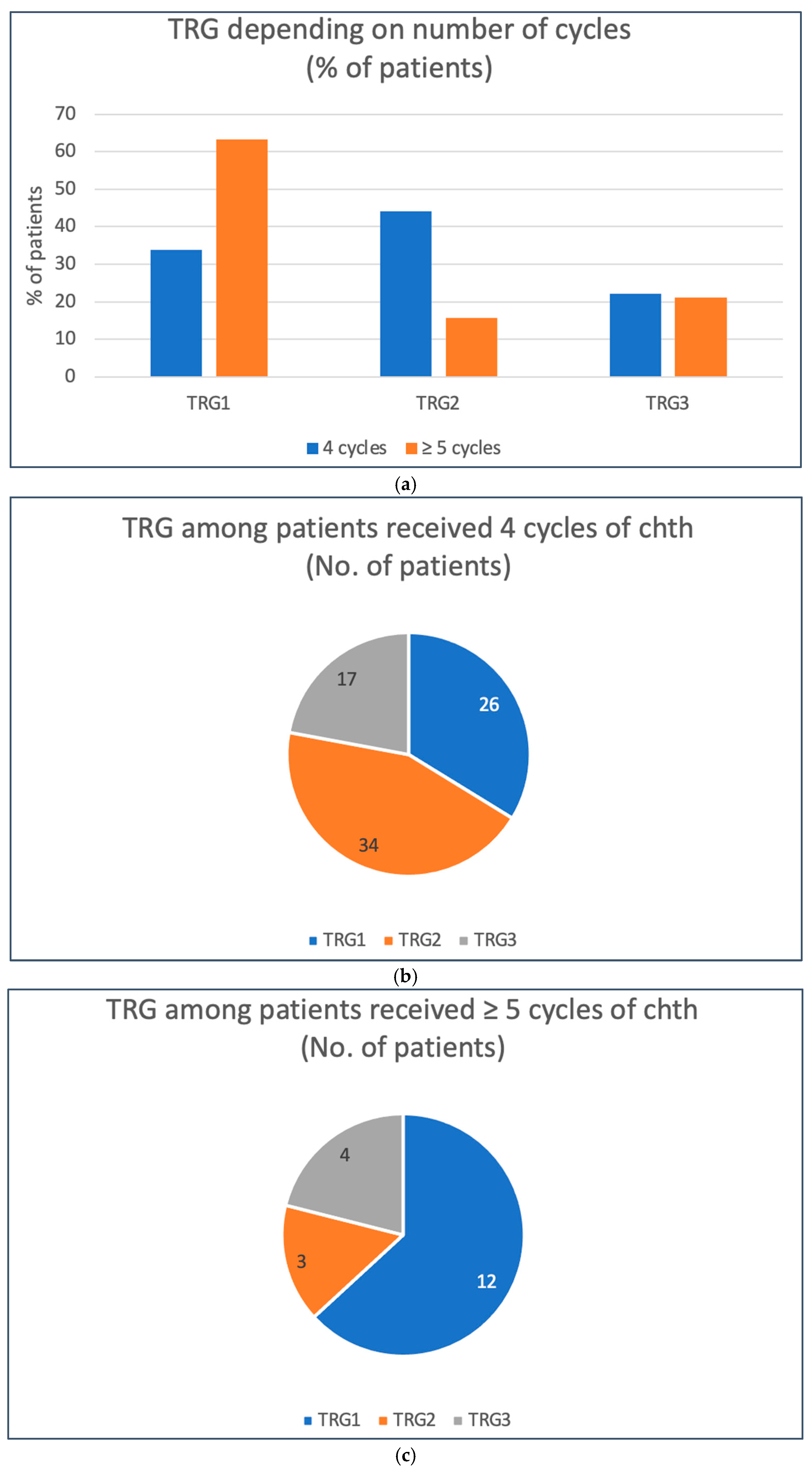
22]. Our analysis revealed that any reduction in the cisplatin dose was linked to a lower likelihood of achieving a pathological complete response (pCR). This finding suggests that maintaining the optimal cisplatin dosage should be prioritized, even if adjustments are made to other components of the aMVAC regimen. Our analysis additionally shows that the pCR rate is associated with administering a particular number of aMVAC cycles. At least five cycles of aMVAC were associated with the highest probability of pCR and thus with the lowest risk of disease regrowth upon delayed surgery.
One of the important drawbacks of our analysis is the lack of comprehensive survival data of our patients. Many MIBC patients who were referred to our center for surgery and underwent neoadjuvant chemotherapy were lost to follow-up at our hospital due to being taken care of by their primary urologists. Therefore, we were unable to present data regarding event-free or cancer-specific survival. However, we continue to collect data on overall survival, which, as for now, are not mature and do not provide any reliable information on patients’ outcomes.
The optimal number of neoadjuvant chemotherapy cycles in MIBC is not well established. On the one hand, older age, diminished organ reserves, and comorbidities characteristic of patients with MIBC represent risk factors for increased toxicity. On the other hand, even though the median age of patients in the VESPER trial was 63 years (58–68), the majority of them (61%) were capable of receiving six cycles of neoadjuvant chemotherapy. There were three (1%) treatment-related deaths in the VESPER study, and grade 3–4 toxicities (mainly hematological) occurred in 52% of patients. A significant cardiovascular or renal impairment incidence was relatively low (each occurring in approximately 6% of patients) [
23]. In our analysis, the median number of cycles was 4, with 81% of patients receiving ≥4 cycles and 16% ≥5 cycles of aMVAC. The treatment was also well tolerated in our real-life population, with G3 toxicities occurring in three patients and G4 in one patient.
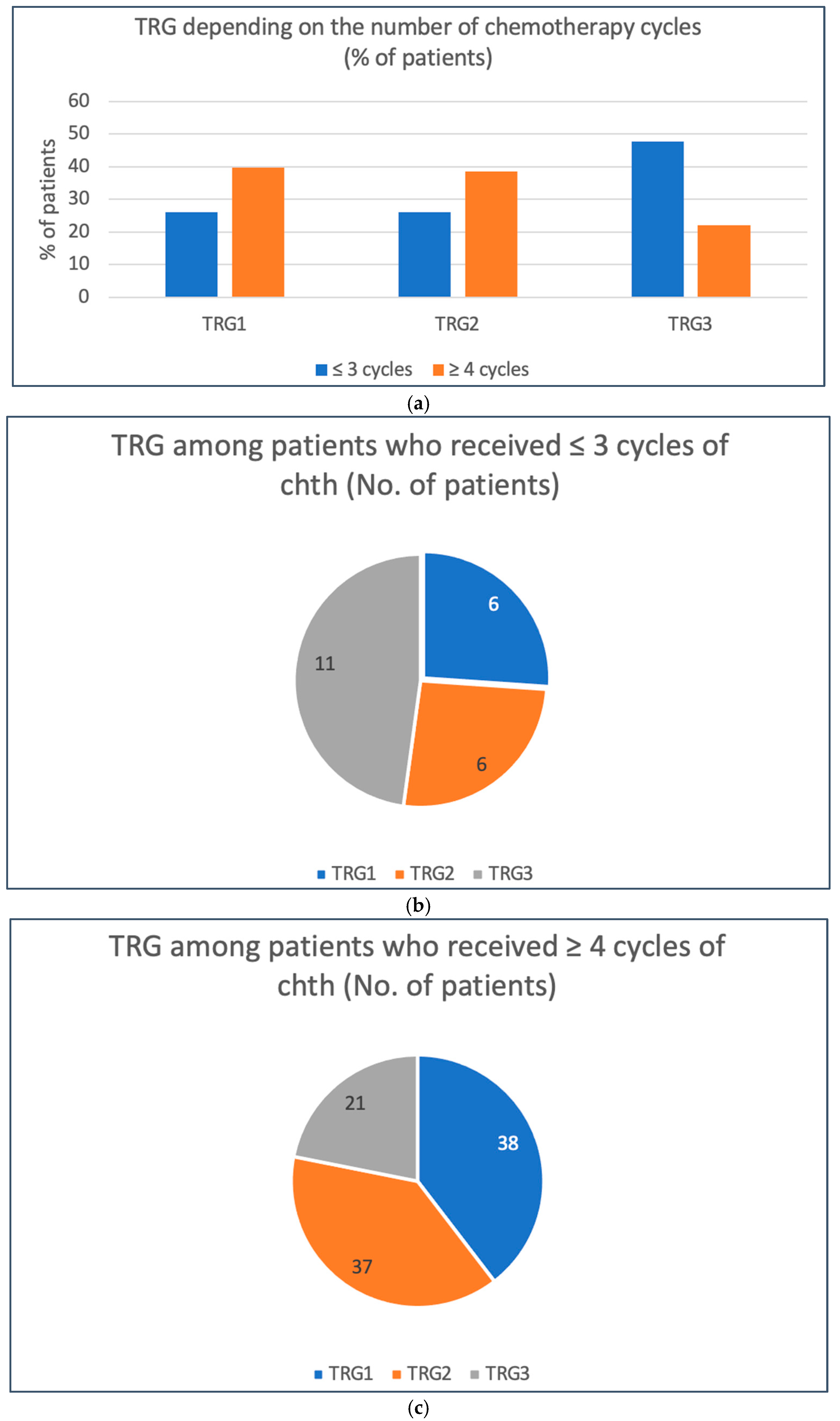
Our analysis demonstrated that the number of chemotherapy cycles is an important predictive factor for the benefit of neoadjuvant therapy. While receiving less than four cycles was significantly correlated with an increased risk of neoadjuvant chemotherapy failure (TRG3), receiving less than five cycles significantly diminished the chance for a complete pathological response. These results clearly show that the minimal number of chemotherapy cycles that benefit patients is four and that fragile patients in suboptimal performance status who are at risk of not completing at least four cycles of chemotherapy should be considered for upfront cystectomy.
Prolonged treatment-related toxicity and logistical reasons may delay the moment of cystectomy in patients who have completed neoadjuvant chemotherapy. There is no consensus on the optimal duration of the post-chemotherapy period, which would allow patients to recover from chemotherapy sequelae and to prepare for the surgical procedure optimally. The median delay of surgery from the last course of chemotherapy in our analysis was 48 (17 to 122) days, which was virtually the same as in the VESPER study—a median of 48 days (95% range—27 to 97 days). While delaying surgery had no impact on the probability of the complete pathological response, it significantly increased the risk of detection of chemotherapy unresponsiveness (TRG3). This observation indicates that delayed surgery negatively impacts patients who are not achieving pCR, in whom the delay allows for cancer cell recovery, regrowth, and possibly progression, which could worsen patient outcomes.
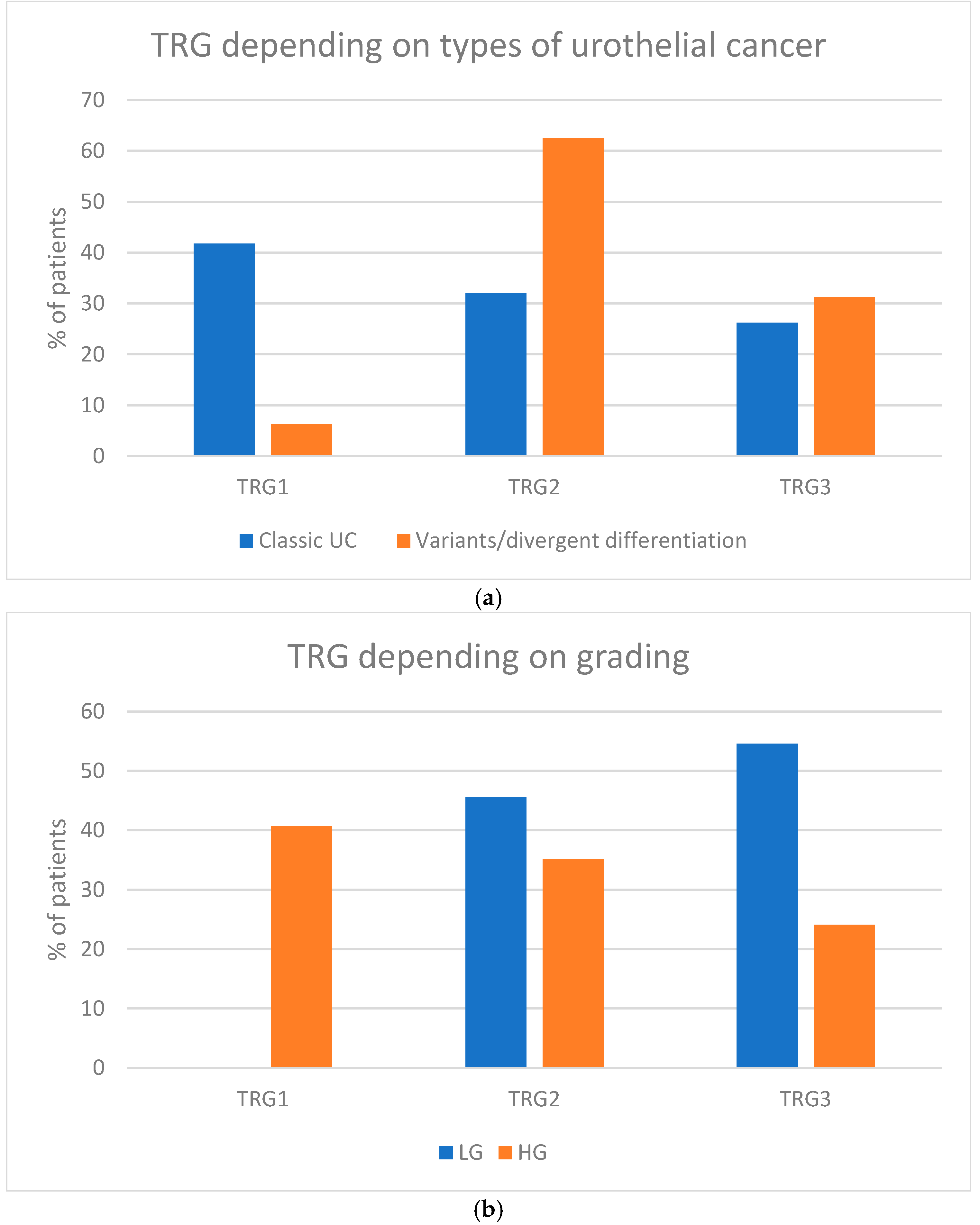
Our analysis demonstrated that patients with organ-confined disease had a higher chance of complete response (TRG1) than those without. However, available data suggest that even though neoadjuvant chemotherapy is associated with a lower probability of complete response in patients with non-organ-confined disease, the scale of improvement of long-term outcomes with the use of preoperative treatment is larger in non-organ-confined than in organ-confined MIBC patients [
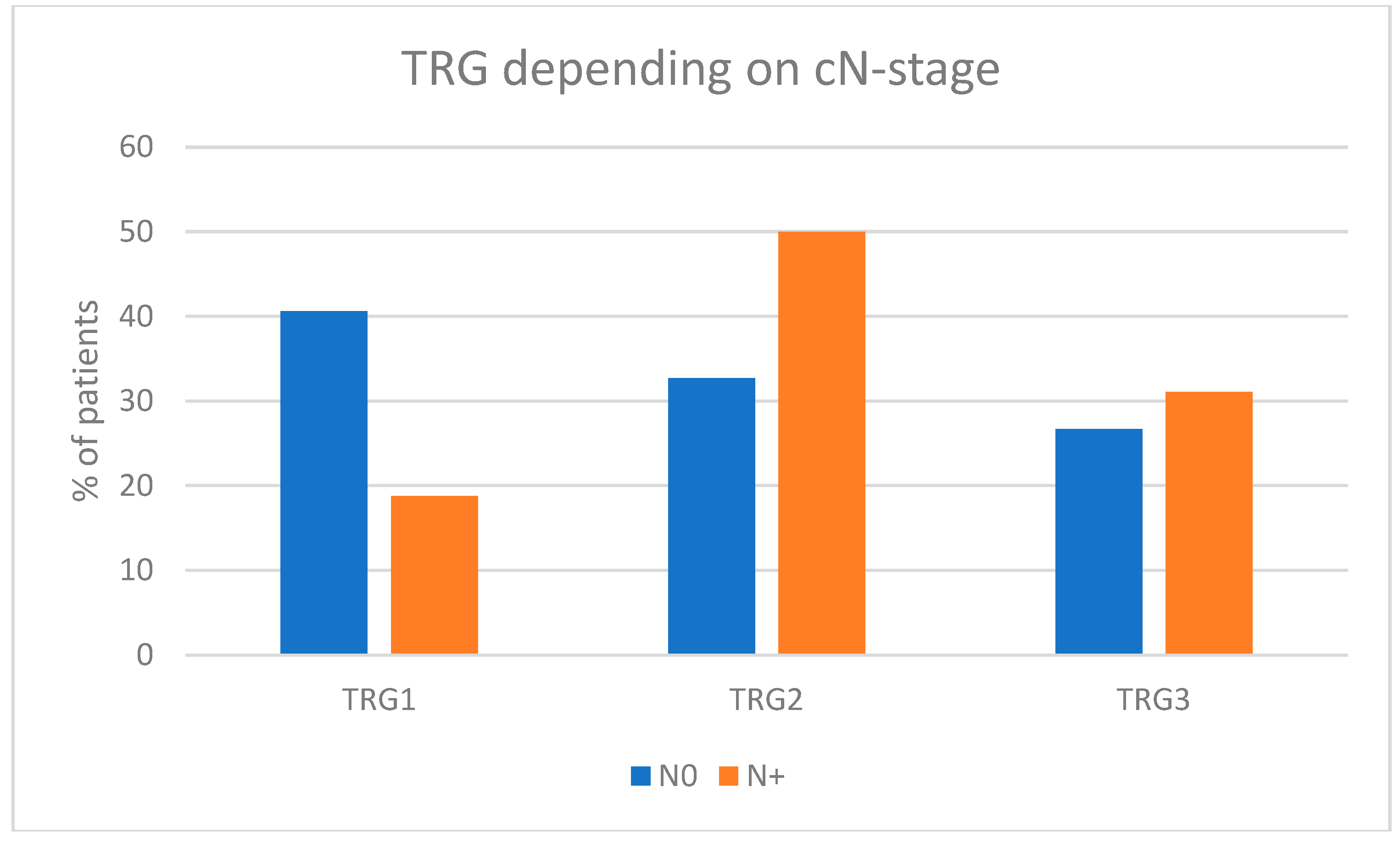
24]. However, the involvement of locoregional lymph nodes represents a detrimental risk factor associated with significantly worse overall survival and decreased benefit from neoadjuvant chemotherapy than in patients with clinically negative lymph nodes [
25].
The population of patients in our analysis was very homogenous for race and ethnicity since 100% of patients were Caucasian. Available retrospective studies have demonstrated that Caucasian MIBC patients are most likely to benefit from various radical approaches (neoadjuvant chemotherapy before radical cystectomy or trimodal therapy) compared especially to African Americans [
26,
27].
Our analysis has not involved patients with UTUC, because previous guidelines on the treatment of urothelial cancer have not supported the use of neoadjuvant chemotherapy in such patients and endorsed primary nephroureterectomy followed by adjuvant chemotherapy. The use of adjuvant treatment was supported by a phase III clinical trial (POUT, NCT01993979), which demonstrated significant improvement in event-free survival following gemcitabine and cisplatin/carboplatin combination [
28]. However, some recent guidelines endorse the use of neoadjuvant chemotherapy in UTUC patients with N+ disease. These guidelines are based on retrospective but robust data demonstrating significantly better outcomes in UTUC N+ patients who received standard (as in MIBC) neoadjuvant chemotherapy [
29,
30,
31].