Perioperative Observations and Outcome in Surgical Treatment of Malignant Peripheral Nerve Sheath Tumors
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
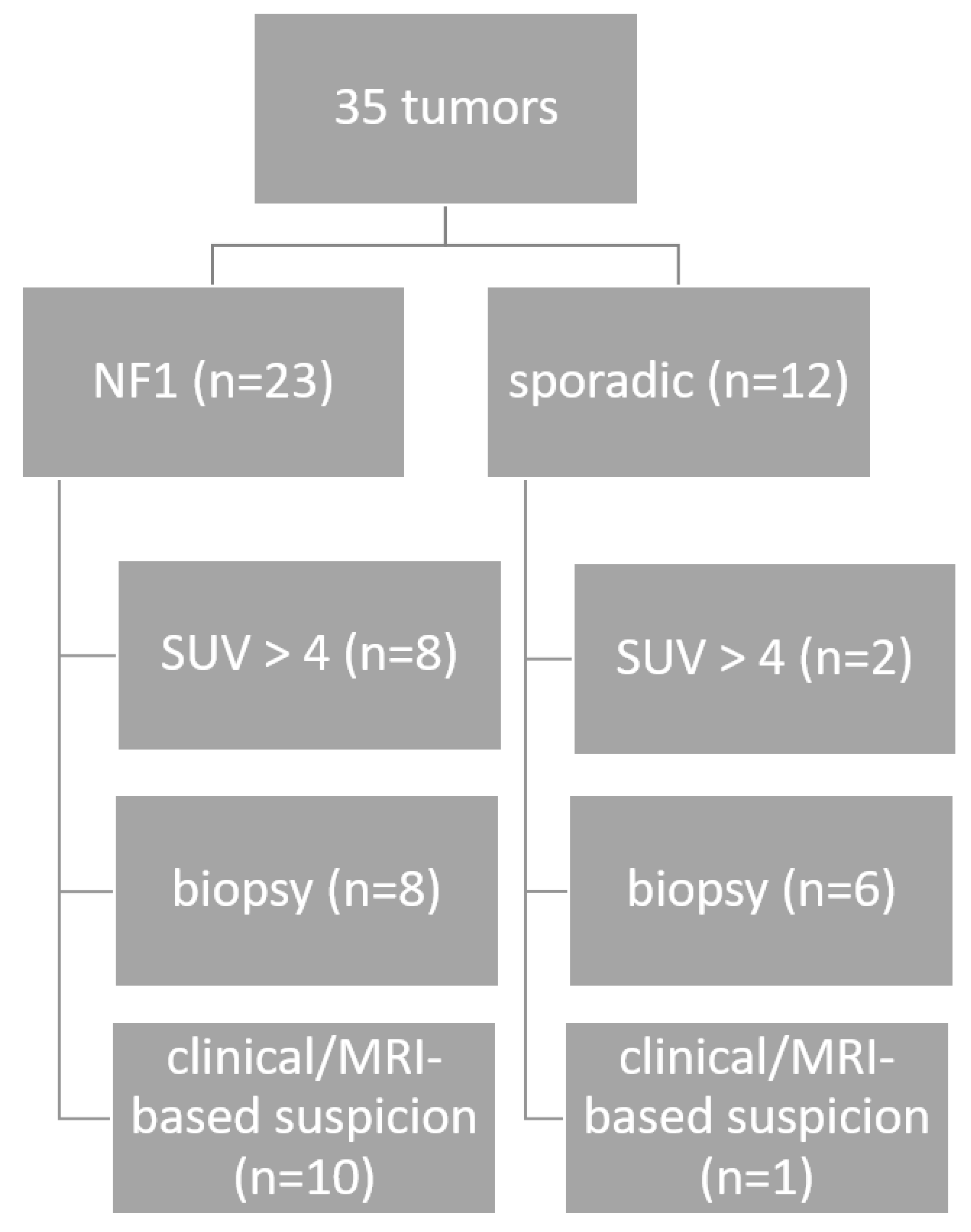
2.1. Patients and Clinical Data
- (1)
- Persisting and drug-resistant pain;
- (2)
- Tumor growth progression in known tumors (clinically in visible or palpable lesions or radiologically by ultrasound or MRI for deeper lesions);
- (3)
- Development or worsening of known focal neurological deficits;
- (4)
- Clinical and/or radiological suspicion of a malignant transformation as described beforehand, if a PET MRI/CT was available/performed;
- (5)
- Increased SUVs > 4 in FDG PET in known symptomatic or asymptomatic tumor lesions.
2.2. Data Evaluation
3. Results
3.1. Patients, Tumors, and Clinics
- (1)
- Progressive and palliative situation due to multiple metastases; thus, surgical intervention was required to stop/prevent focal neurological deficits (cases 2, 8, 16).
- (2)
- Initial partial resection under functional preservation criteria (cases 25, 27) with secondary total removal after histological MPNST confirmation.
- (3)
- Inoperability due to the local/surrounding infiltration of critical structures (e.g., vessels, bone; cases 8, 19).
3.2. Correlation Between Maximum Histopathological MIB-1 (Proliferation) Index (%), FDG PET SUV, and Preoperative Volume for All Tumors
3.3. Association Between Histopathological Grading (High- vs. Low-Grade), Disease Type (NF1-Associated vs. Sporadic), and Parameters
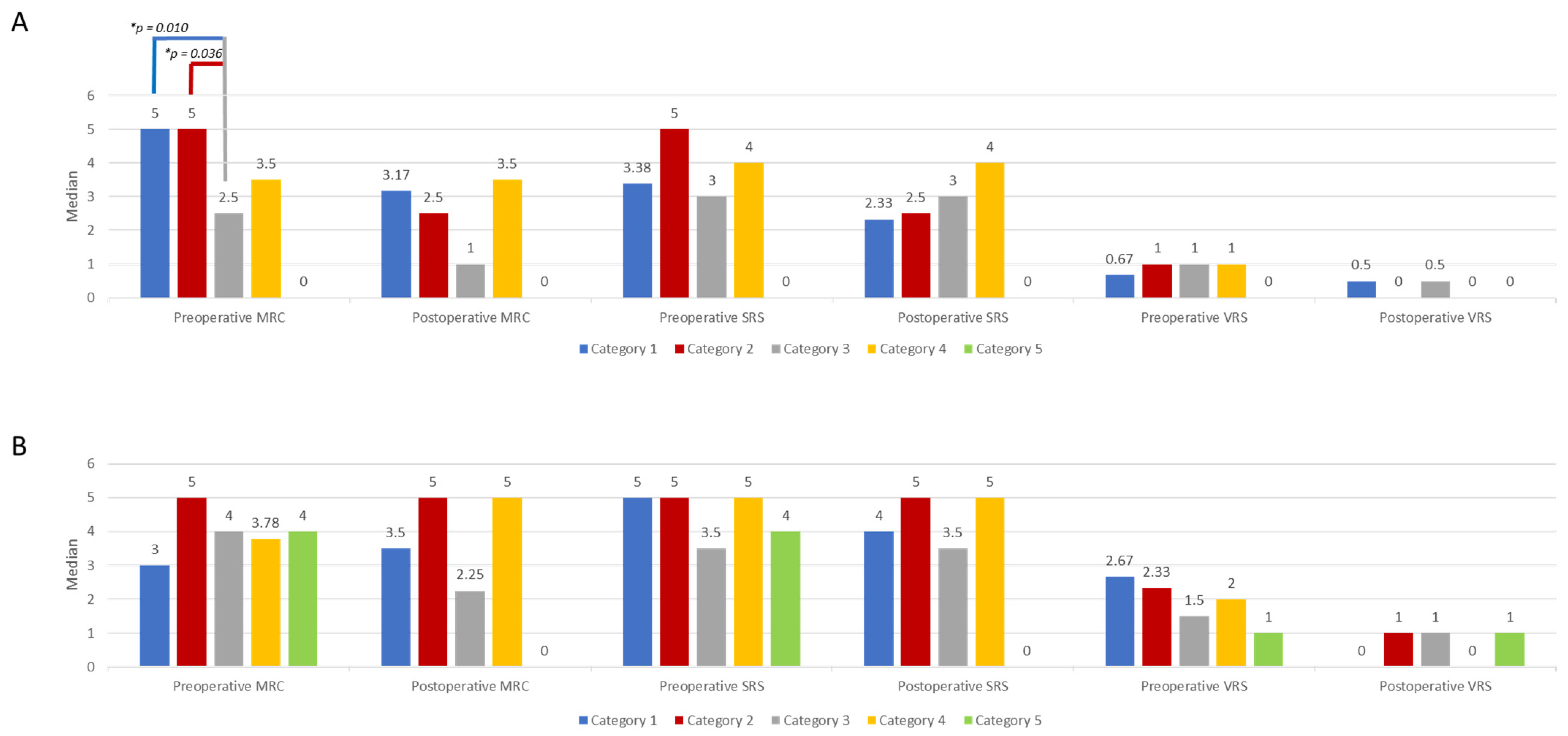
3.4. Location Category Distribution of NF1 and SPO-Related MPNSTs and Association with Parameters
3.5. Overall Survival Between Comparisons
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amirian, E.S.; Goodman, J.C.; New, P.; Scheurer, M.E. Pediatric and adult malignant peripheral nerve sheath tumors: An analysis of data from the surveillance, epidemiology, and end results program. J. Neurooncol. 2014, 116, 609–616. [Google Scholar] [CrossRef]
- Sharma, M.S.; Singh, R.; Kale, S.S.; Agrawal, D.; Sharma, B.S.; Mahapatra, A.K. Tumor control and hearing preservation after Gamma Knife radiosurgery for vestibular schwannomas in neurofibromatosis type 2. J. Neurooncol. 2010, 98, 265–270. [Google Scholar] [CrossRef]
- Stucky, C.C.; Johnson, K.N.; Gray, R.J.; Pockaj, B.A.; Ocal, I.T.; Rose, P.S.; Wasif, N. Malignant peripheral nerve sheath tumors (MPNST): The Mayo Clinic experience. Ann. Surg. Oncol. 2012, 19, 878–885. [Google Scholar] [CrossRef]
- Ducatman, B.S.; Scheithauer, B.W.; Piepgras, D.G.; Reiman, H.M.; Ilstrup, D.M. Malignant peripheral nerve sheath tumors. A clinicopathologic study of 120 cases. Cancer 1986, 57, 2006–2021. [Google Scholar] [CrossRef]
- Mowery, A.; Clayburgh, D. Malignant peripheral nerve sheath tumors: Analysis of the national cancer database. Oral Oncol. 2019, 98, 13–19. [Google Scholar] [CrossRef]
- Dombi, E.; Ardern-Holmes, S.L.; Babovic-Vuksanovic, D.; Barker, F.G.; Connor, S.; Evans, D.G.; Fisher, M.J.; Goutagny, S.; Harris, G.J.; Jaramillo, D.; et al. Recommendations for imaging tumor response in neurofibromatosis clinical trials. Neurology 2013, 81, S33–S40. [Google Scholar] [CrossRef]
- Watson, K.L.; Al Sannaa, G.A.; Kivlin, C.M.; Ingram, D.R.; Landers, S.M.; Roland, C.L.; Cormier, J.N.; Hunt, K.K.; Feig, B.W.; Ashleigh Guadagnolo, B.; et al. Patterns of recurrence and survival in sporadic, neurofibromatosis Type 1-associated, and radiation-associated malignant peripheral nerve sheath tumors. J. Neurosurg. 2017, 126, 319–329. [Google Scholar] [CrossRef]
- Chang, Y.W.; Liao, W.C. Survival analysis of malignant peripheral nerve sheath tumor: Experience of a tertiary center in Taiwan. J. Chin. Med. Assoc. 2023, 86, 584–588. [Google Scholar] [CrossRef]
- Kinoshita, I.; Yamada, Y.; Kohashi, K.; Yamamoto, H.; Iwasaki, T.; Ishihara, S.; Toda, Y.U.; Ito, Y.; Susuki, Y.; Kawaguchi, K.; et al. Frequent MN1 Gene Mutations in Malignant Peripheral Nerve Sheath Tumor. Anticancer. Res. 2020, 40, 6221–6228. [Google Scholar] [CrossRef]
- Reinert, C.P.; Schuhmann, M.U.; Bender, B.; Gugel, I.; la Fougere, C.; Schafer, J.; Gatidis, S. Comprehensive anatomical and functional imaging in patients with type I neurofibromatosis using simultaneous FDG-PET/MRI. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 776–787. [Google Scholar] [CrossRef]
- Hwang, I.K.; Hahn, S.M.; Kim, H.S.; Kim, S.K.; Kim, H.S.; Shin, K.H.; Suh, C.O.; Lyu, C.J.; Han, J.W. Outcomes of Treatment for Malignant Peripheral Nerve Sheath Tumors: Different Clinical Features Associated with Neurofibromatosis Type 1. Cancer Res. Treat. 2017, 49, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Schuch, L.F.; Kirschnick, L.B.; de Arruda, J.A.A.; Klein, I.P.; Silveira, F.M.; Vasconcelos, A.C.U.; Santos-Silva, A.R.; Lopes, M.A.; Carrard, V.C.; Vargas, P.A.; et al. Malignant peripheral nerve sheath tumour of the oral and maxillofacial region-A systematic review. Oral Dis. 2022, 28, 2072–2082. [Google Scholar] [CrossRef]
- Jensen, V.; Sorensen, F.B.; Bentzen, S.M.; Ladekarl, M.; Nielsen, O.S.; Keller, J.; Jensen, O.M. Proliferative activity (MIB-1 index) is an independent prognostic parameter in patients with high-grade soft tissue sarcomas of subtypes other than malignant fibrous histiocytomas: A retrospective immunohistological study including 216 soft tissue sarcomas. Histopathology 1998, 32, 536–546. [Google Scholar] [CrossRef]
- Mautner, V.F.; Asuagbor, F.A.; Dombi, E.; Funsterer, C.; Kluwe, L.; Wenzel, R.; Widemann, B.C.; Friedman, J.M. Assessment of benign tumor burden by whole-body MRI in patients with neurofibromatosis 1. Neuro-Oncology 2008, 10, 593–598. [Google Scholar] [CrossRef]
- Miettinen, M.M.; Antonescu, C.R.; Fletcher, C.D.M.; Kim, A.; Lazar, A.J.; Quezado, M.M.; Reilly, K.M.; Stemmer-Rachamimov, A.; Stewart, D.R.; Viskochil, D.; et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1—A consensus overview. Hum. Pathol. 2017, 67, 1–10. [Google Scholar] [CrossRef]
- Legius, E.; Messiaen, L.; Wolkenstein, P.; Pancza, P.; Avery, R.A.; Berman, Y.; Blakeley, J.; Babovic-Vuksanovic, D.; Cunha, K.S.; Ferner, R.; et al. Revised diagnostic criteria for neurofibromatosis type 1 and Legius syndrome: An international consensus recommendation. Genet. Med. 2021, 23, 1506–1513. [Google Scholar] [CrossRef]
- Zipfel, J.; Al-Hariri, M.; Gugel, I.; Haas-Lude, K.; Grimm, A.; Warmann, S.; Krimmel, M.; Mautner, V.F.; Tatagiba, M.; Schuhmann, M.U. Surgical management of peripheral nerve sheath tumours in children, with special consideration of neurofibromatoses. Childs Nerv. Syst. 2020, 36, 2433–2442. [Google Scholar] [CrossRef]
- Compston, A. Aids to the investigation of peripheral nerve injuries. Medical Research Council: Nerve Injuries Research Committee. His Majesty’s Stationery Office: 1942; pp. 48 (iii) and 74 figures and 7 diagrams; with aids to the examination of the peripheral nervous system. By Michael O’Brien for the Guarantors of Brain. Saunders Elsevier: 2010; pp. [8] 64 and 94 Figures. Brain 2010, 133, 2838–2844. [Google Scholar] [CrossRef]
- Zipfel, J.; Al-Hariri, M.; Gugel, I.; Grimm, A.; Steger, V.; Ladurner, R.; Krimmel, M.; Tatagiba, M.; Schuhmann, M.U. Surgical Management of Sporadic Peripheral Nerve Schwannomas in Adults: Indications and Outcome in a Single Center Cohort. Cancers 2021, 13, 1017. [Google Scholar] [CrossRef]
- Lee, H.J.; Cho, Y.; Joo, H.; Jeon, J.Y.; Jang, Y.E.; Kim, J.T. Comparative study of verbal rating scale and numerical rating scale to assess postoperative pain intensity in the post anesthesia care unit: A prospective observational cohort study. Medicine 2021, 100, e24314. [Google Scholar] [CrossRef]
- Hermanek, P.; Wittekind, C. The pathologist and the residual tumor (R) classification. Pathol. Res. Pract. 1994, 190, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Wittekind, C.; Compton, C.C.; Greene, F.L.; Sobin, L.H. TNM residual tumor classification revisited. Cancer 2002, 94, 2511–2516. [Google Scholar] [CrossRef] [PubMed]
- Valentin, T.; Le Cesne, A.; Ray-Coquard, I.; Italiano, A.; Decanter, G.; Bompas, E.; Isambert, N.; Thariat, J.; Linassier, C.; Bertucci, F.; et al. Management and prognosis of malignant peripheral nerve sheath tumors: The experience of the French Sarcoma Group (GSF-GETO). Eur. J. Cancer 2016, 56, 77–84. [Google Scholar] [CrossRef]
- Friedman, J.M. Neurofibromatosis 1. In GeneReviews(R); Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Mefford, H.C., Stephens, K., Amemiya, A., Ledbetter, N., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Warbey, V.S.; Ferner, R.E.; Dunn, J.T.; Calonje, E.; O’Doherty, M.J. [18F]FDG PET/CT in the diagnosis of malignant peripheral nerve sheath tumours in neurofibromatosis type-1. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 751–757. [Google Scholar] [CrossRef]
- Ferner, R.E.; Golding, J.F.; Smith, M.; Calonje, E.; Jan, W.; Sanjayanathan, V.; O’Doherty, M. [18F]2-fluoro-2-deoxy-D-glucose positron emission tomography (FDG PET) as a diagnostic tool for neurofibromatosis 1 (NF1) associated malignant peripheral nerve sheath tumours (MPNSTs): A long-term clinical study. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. ESMO 2008, 19, 390–394. [Google Scholar] [CrossRef]
- Yao, L.; Alahmari, M.; Temel, Y.; Hovinga, K. Therapy of Sporadic and NF2-Related Vestibular Schwannoma. Cancers 2020, 12, 835. [Google Scholar] [CrossRef] [PubMed]
- Kluwe, L.; Friedrich, R.E.; Peiper, M.; Friedman, J.; Mautner, V.F. Constitutional NF1 mutations in neurofibromatosis 1 patients with malignant peripheral nerve sheath tumors. Hum. Mutat. 2003, 22, 420. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, R.; Jett, K.; Harris, G.J.; Cai, W.; Friedman, J.M.; Mautner, V.F. Benign whole body tumor volume is a risk factor for malignant peripheral nerve sheath tumors in neurofibromatosis type 1. J. Neurooncol. 2014, 116, 307–313. [Google Scholar] [CrossRef]

| NF1 | SPO | |
|---|---|---|
| No. of patients/tumors | 21/23 | 12/12 |
| Sex (no. of females/males) | 7/14 | 5/7 |
| Total follow-up in months (mean ± SD, range) | 34 ± 42, 1–165 | 43 ± 39, 1–114 |
| Age at time of surgery in years (mean ± SD, range) | 29 ± 13, 8–54 | 45 ± 13, 24–67 |
| Family history of NF1 (yes/no) | 7/14 | 0/0 |
| Detected NF1-related gene mutation types | No of patients | |
| 2 | - |
| 2 | - |
| 2 | - |
| 1 | - |
| No mutation detected | 3 | 1 |
| Not examined | 11 | 11 |
| Location Categories of peripheral (1–4) and intraspinal (5) MPNSTs | %, n = number of tumors | |
| 26%, n = 6 | 50%, n = 6 |
| 13%, n = 3 | 17%, n = 2 |
| 17%, n = 4 | 17%, n = 2 |
| 39%, n = 9 | 17%, n = 2 |
| 4%, n = 1 | 0 |
| Indication for surgery | %, n = number of tumors | |
| Persisting/drug-resistant/progressive pain; | 26%, n = 6 | 25%, n = 3 |
| 22%, n = 5 | 33%, n = 4 |
| 13%, n = 3 | 25%, n = 3 |
| 39%, n = 9 | 17%, n = 2 |
| 9%, n = 2 | 8%, n = 1 |
| Preoperative tumor volume in cm3 (mean ± SD, range) p-value | 299.1 ± 500, 11.5–2294.3 0.048 | 17.8 ± 15.46, 1.2–44.9 |
| Preoperative SUV in FDG PET (mean ± SD, range) No of PET MRI/CT scans | 9.8 ± 6.45, 3.9–25.6 n = 5/5 | 6 ± 0.6, 5.4–6.6 n = 1/1 |
| Histopathological grade | %, n = number of tumors | |
| 61%, n = 14 | 64%, n = 7 |
| 39%, n = 9 | 36%, n = 4 |
| - | 1 |
| Maximum MIB-1 Proliferation Index | n = number of tumors | |
| <5/5/10/20/30/40/50/60/70/80 in % | 1/5/1/5/1/2/2/3/1/1 | 1/1/1/2/1/1/1/0/0 |
| Resection margins | %, n = number of tumors *** | |
| 70%, n = 16 | 58%, n = 7 |
| 13%, n = 3 | 17%, n = 2 |
| 17%, n = 4 | 25%, n = 3 |
| Radiation (neo-/adjuvant/both) (no. of tumors) | 0/6/1 | 0/2/0 |
| Chemotherapy (neo-/adjuvant/both) (no. of tumors) | 1/6/1 | 1/4/0 |
| Variable | U | z | p | Mean Rank/Median High-Grade | Mean Rank/Median Low-Grade |
| Preoperative MRC | 154 | −0.075 | 0.961 | 18.11/5, n = 19 | 17.88/5, n = 16 |
| Postoperative MRC | 169 | −0.604 | 0.567 | 18.92/4, n = 19 | 16.91/4, n = 16 |
| Preoperative SRS | 140 | −0.417 | 0.707 | 17.37/4, n = 19 | 18.75/4, n = 16 |
| Postoperative SRS | 153 | −0.017 | 0.987 | 17.97/3, n = 19 | 18.03/3, n = 16 |
| Preoperative VRS | 155 | −0.104 | 0.935 | 18.16/2, n = 19 | 17.81/2, n = 16 |
| Postoperative VRS | 171 | −1.098 | 0.365 | 19/0, n = 18 | 15.81/0, n = 16 |
| Max Proliferation Rate | 166 | −2.343 | 0.020 | 18.76/40, n = 17 | 11.23/20, n = 13 |
| Preoperative Volume | 149 | −0.812 | 0.433 | 17.76/62.3, n = 17 | 15.07/112.2, n = 15 |
| Variable | U | z | p | Mean Rank/Median NF1 | Mean Rank/Median Sporadic |
| Preoperative MRC | 107 | −1.220 | 0.294 | 16.65/5, n = 23 | 20.58/5, n = 12 |
| Postoperative MRC | 126 | −0.344 | 0.745 | 18.41/5, n = 23 | 17.21/3, n = 12 |
| Preoperative SRS | 109 | −1.040 | 0.327 | 16.76/4, n = 23 | 20.38/4.5, n = 12 |
| Postoperative SRS | 146 | −0.286 | 0.797 | 18.35/3, n = 23 | 17.33/3.5, n = 12 |
| Preoperative VRS | 223 | −3.082 | 0.002 | 21.70/2, n = 23 | 10.92/0.5, n = 12 |
| Postoperative VRS | 155 | −0.977 | 0.423 | 18.55/0, n = 22 | 15.58/0, n = 12 |
| Max. Proliferation Rate | 92 | −0.213 | 0.836 | 15.7/20, n = 22 | 14.94/27.5, n = 8 |
| Preoperative Volume | 195 | −3.456 | <0.001 | 20.36/112.2, n = 22 | 8/11.6, n = 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zipfel, J.; Tellermann, J.; Ferraris, K.P.; Grimm, F.; Bornemann, A.; Bender, B.; Dittmann, H.; Schäfer, J.; Nikolaou, K.; Ladurner, R.; et al. Perioperative Observations and Outcome in Surgical Treatment of Malignant Peripheral Nerve Sheath Tumors. Cancers 2024, 16, 3757. https://doi.org/10.3390/cancers16223757
Zipfel J, Tellermann J, Ferraris KP, Grimm F, Bornemann A, Bender B, Dittmann H, Schäfer J, Nikolaou K, Ladurner R, et al. Perioperative Observations and Outcome in Surgical Treatment of Malignant Peripheral Nerve Sheath Tumors. Cancers. 2024; 16(22):3757. https://doi.org/10.3390/cancers16223757
Chicago/Turabian StyleZipfel, Julian, Jonas Tellermann, Kevin Paul Ferraris, Florian Grimm, Antje Bornemann, Benjamin Bender, Helmut Dittmann, Jürgen Schäfer, Konstantin Nikolaou, Ruth Ladurner, and et al. 2024. "Perioperative Observations and Outcome in Surgical Treatment of Malignant Peripheral Nerve Sheath Tumors" Cancers 16, no. 22: 3757. https://doi.org/10.3390/cancers16223757
APA StyleZipfel, J., Tellermann, J., Ferraris, K. P., Grimm, F., Bornemann, A., Bender, B., Dittmann, H., Schäfer, J., Nikolaou, K., Ladurner, R., Steger, V., Tatagiba, M., Schuhmann, M. U., & Gugel, I. (2024). Perioperative Observations and Outcome in Surgical Treatment of Malignant Peripheral Nerve Sheath Tumors. Cancers, 16(22), 3757. https://doi.org/10.3390/cancers16223757







