Management of the Contralateral Neck in Unilateral Node-Positive Oral Squamous Cell Carcinoma
Abstract
:Simple Summary
Abstract
1. Introduction
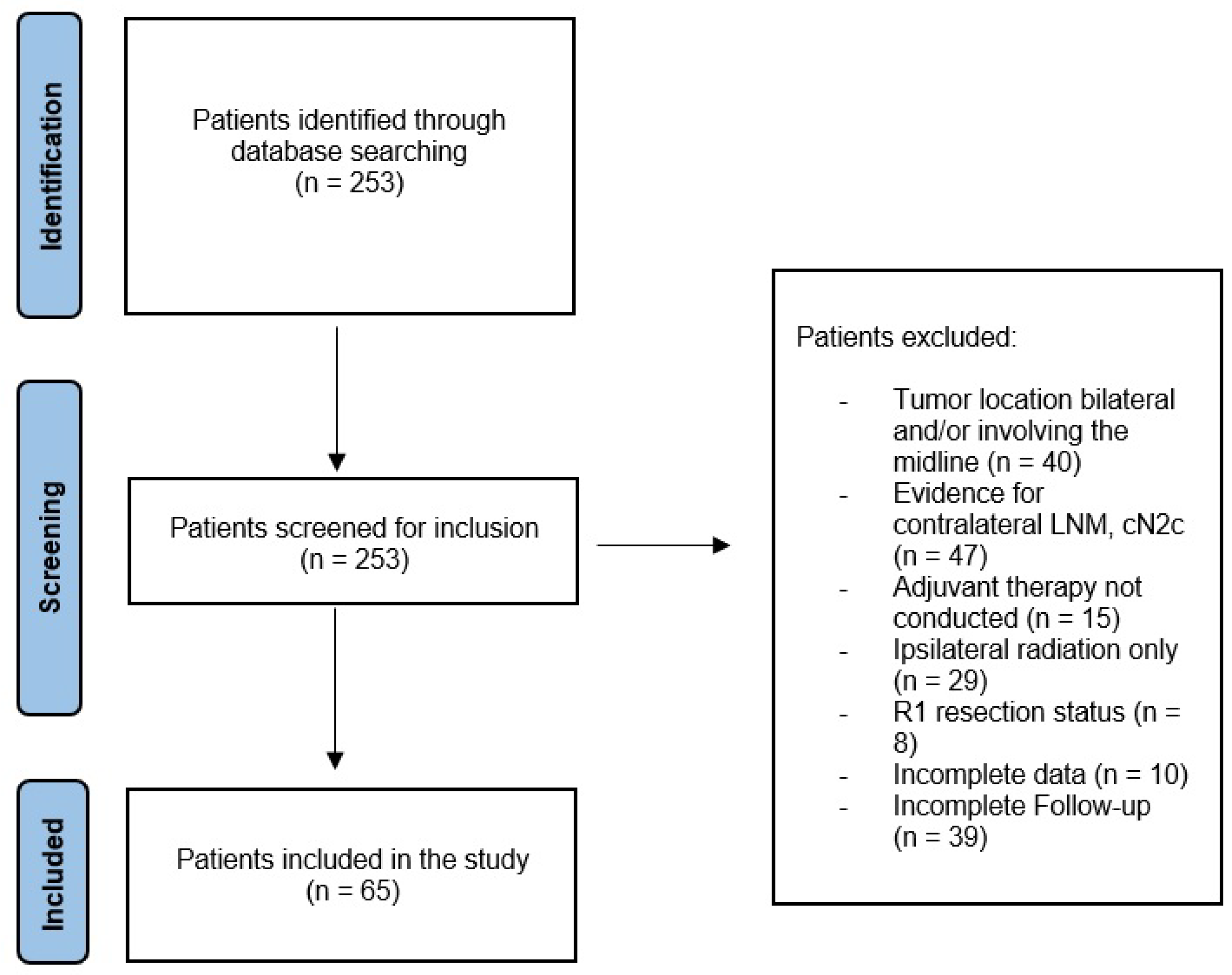
2. Material and Methods
2.1. Ethical Approval
2.2. Study Design
- I.
- Histologically proven OSCC, which was unilaterally located without reaching the midline. All patients underwent ipsilateral neck dissection (ND) and presented with histologically proven ipsilateral CLNM (≥pN1).
- II.
- The contralateral neck was determined to be clinically unsuspicious (cN0).
- III.
- Patients either receiving END for the contralateral neck (group I) or no surgical treatment for the contralateral neck (group II).
- IV.
- No history of previous OSCC or radiation therapy in the head and neck area.
- V.
- All patients underwent surgery with a curative intent and were treated with bilateral adjuvant radiotherapy (RT) with or without chemotherapy according to the national guidelines [4].
- VI.
- The minimum follow-up period was 6 months.
2.3. Statistical Analysis
3. Results
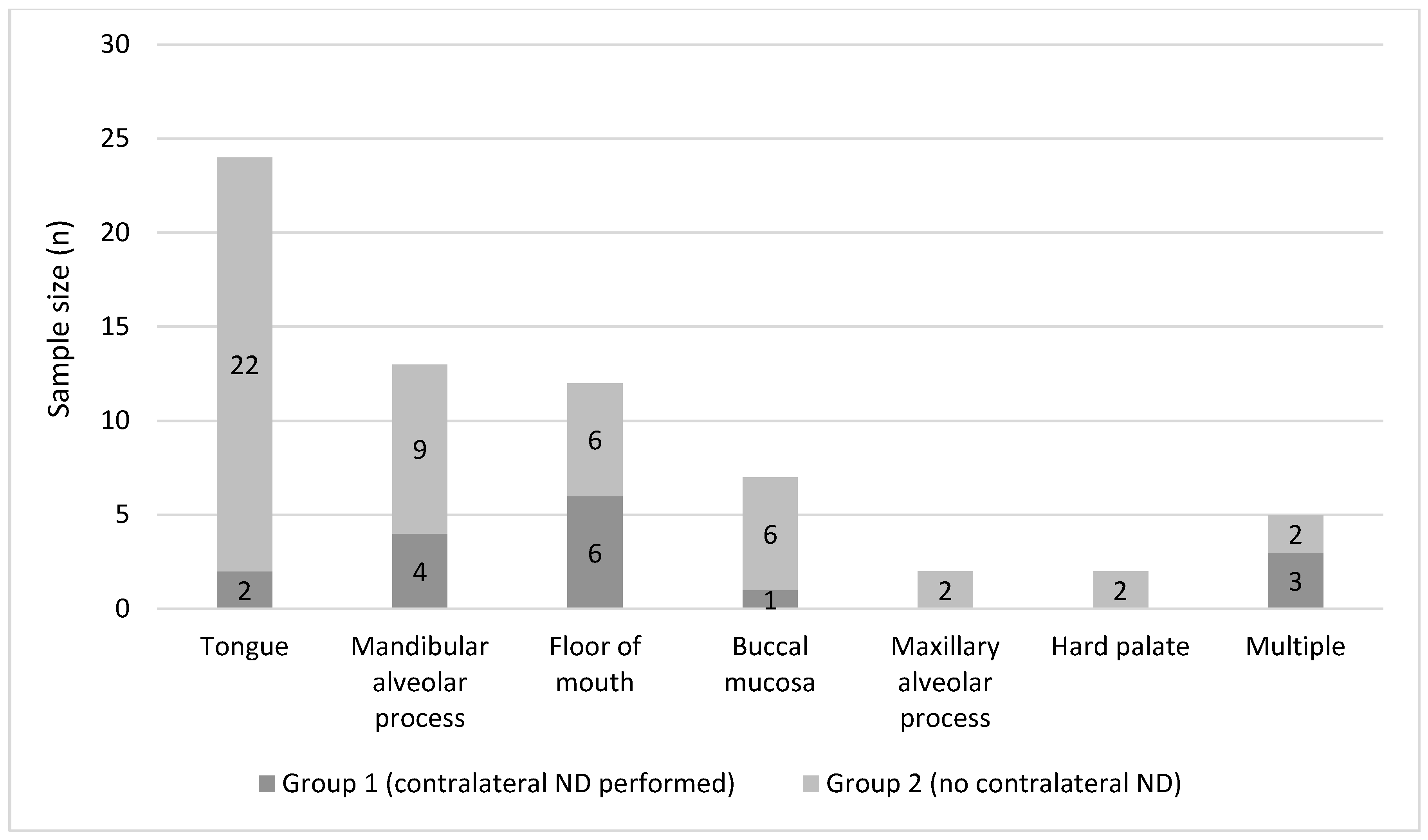
3.1. Patient Baseline Data
3.2. Lymph Node Management
3.3. Histopathological Examination
3.4. Follow-Up and Disease Recurrence
3.5. Occurrence of Contralateral CLNM in Ipsilateral N+ Necks during Follow-Up
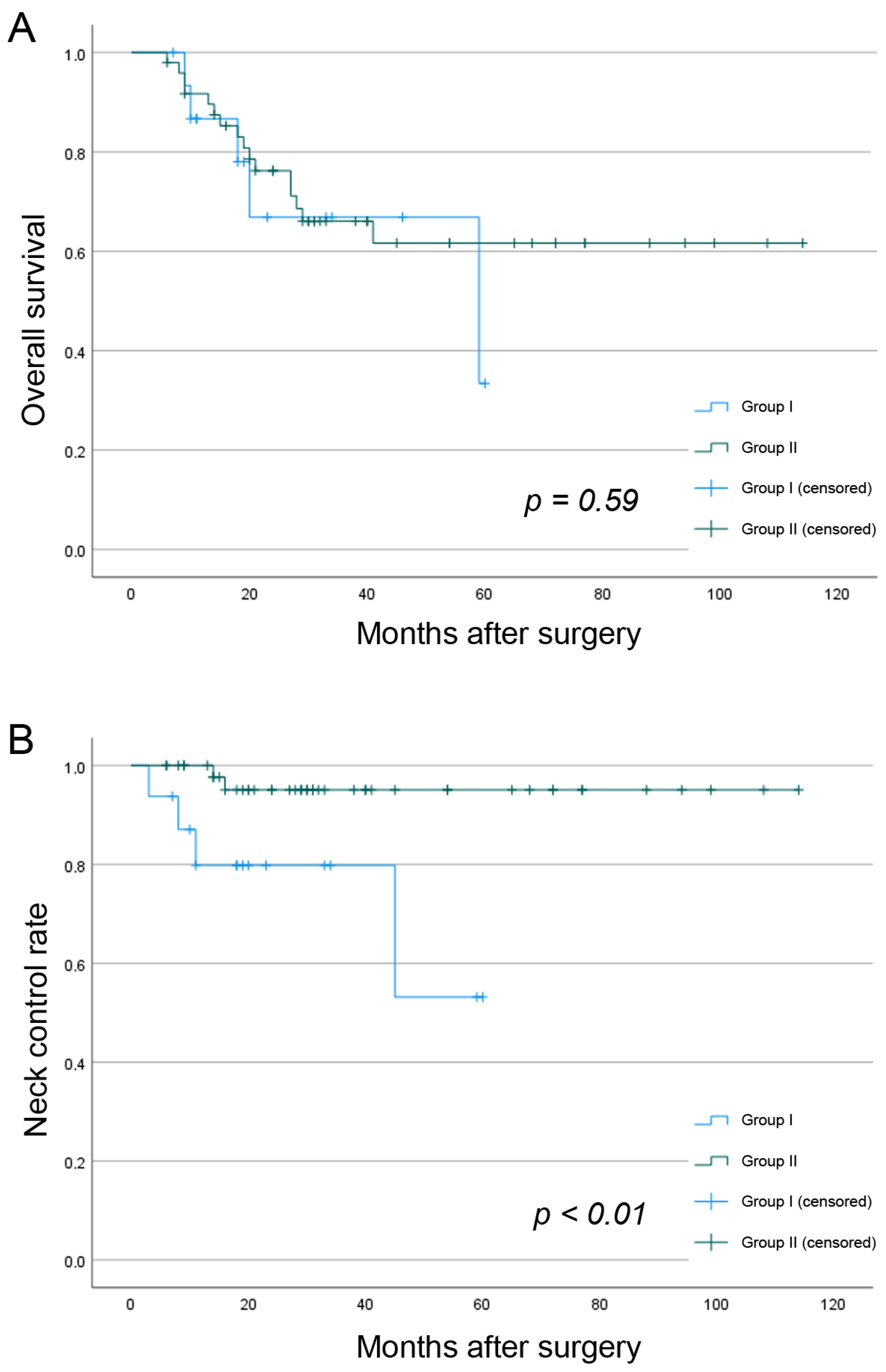
3.6. Survival Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shingaki, S.; Takada, M.; Sasai, K.; Bibi, R.; Kobayashi, T.; Nomura, T.; Saito, C. Impact of Lymph Node Metastasis on the Pattern of Failure and Survival in Oral Carcinomas. Am. J. Surg. 2003, 185, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.S.; El Naggar, A.K.; Mo, V.; Roberts, D.; Myers, J.N. Disparity in Pathologic and Clinical Lymph Node Staging in Oral Tongue Carcinoma: Implications for Therapeutic Decision Making. Cancer 2003, 98, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Zanoni, D.K.; Montero, P.H.; Migliacci, J.C.; Shah, J.P.; Wong, R.J.; Ganly, I.; Patel, S.G. Survival Outcomes after Treatment of Cancer of the Oral Cavity (1985–2015). Oral Oncol. 2019, 90, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Leitlinienprogramm; Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe). S3-Leitlinie Diagnostik und Therapie des Mundhöhlenkarzinoms. AWMF 2021, 3.01, 1–154. [Google Scholar]
- Koyfman, S.A.; Ismaila, N.; Crook, D.; D’Cruz, A.; Rodriguez, C.P.; Sher, D.J.; Silbermins, D.; Sturgis, E.M.; Tsue, T.T.; Weiss, J.; et al. Management of the Neck in Squamous Cell Carcinoma of the Oral Cavity and Oropharynx: ASCO Clinical Practice Guideline. J. Clin. Oncol. 2019, 37, 1753–1774. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network Head and Neck Cancers (Version 1.2023). Available online: https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf (accessed on 5 October 2022).
- Garrel, R.; Poissonnet, G.; Moyà Plana, A.; Fakhry, N.; Dolivet, G.; Lallemant, B.; Sarini, J.; Vergez, S.; Guelfucci, B.; Choussy, O.; et al. Equivalence Randomized Trial to Compare Treatment on the Basis of Sentinel Node Biopsy Versus Neck Node Dissection in Operable T1-T2N0 Oral and Oropharyngeal Cancer. J. Clin. Oncol. 2020, 38, 4010–4018. [Google Scholar] [CrossRef]
- Doll, C.; Steffen, C.; Amthauer, H.; Thieme, N.; Elgeti, T.; Huang, K.; Kreutzer, K.; Koerdt, S.; Heiland, M.; Beck-Broichsitter, B. Sentinel Lymph Node Biopsy in Early Stages of Oral Squamous Cell Carcinoma Using the Receptor-Targeted Radiotracer 99mTc-Tilmanocept. Diagnostics 2021, 11, 1231. [Google Scholar] [CrossRef]
- Agarwal, S.K.; Akali, N.R.; Sarin, D. Prospective Analysis of 231 Elective Neck Dissections in Oral Squamous Cell Carcinoma with Node Negative Neck—To Decide the Extent of Neck Dissection. Auris Nasus Larynx 2018, 45, 156–161. [Google Scholar] [CrossRef]
- D’Cruz, A.K.; Vaish, R.; Kapre, N.; Dandekar, M.; Gupta, S.; Hawaldar, R.; Agarwal, J.P.; Pantvaidya, G.; Chaukar, D.; Deshmukh, A.; et al. Elective versus Therapeutic Neck Dissection in Node-Negative Oral Cancer. N. Engl. J. Med. 2015, 373, 521–529. [Google Scholar] [CrossRef]
- Kurita, H.; Koike, T.; Narikawa, J.N.; Sakai, H.; Nakatsuka, A.; Uehara, S.; Kobayashi, H.; Kurashina, K. Clinical Predictors for Contralateral Neck Lymph Node Metastasis from Unilateral Squamous Cell Carcinoma in the Oral Cavity. Oral Oncol. 2004, 40, 898–903. [Google Scholar] [CrossRef]
- Kowalski, L.P.; Bagietto, R.; Lara, J.R.L.; Santos, R.L.; Tagawa, E.K.; Santos, I.R.B. Factors Influencing Contralateral Lymph Node Metastasis from Oral Carcinoma. Head Neck 1999, 21, 104–110. [Google Scholar] [CrossRef]
- Chow, T.L.; Chow, T.K.; Chan, T.T.F.; Yu, N.F.; Fung, S.C.; Lam, S.H. Contralateral Neck Recurrence of Squamous Cell Carcinoma of Oral Cavity and Oropharynx. J. Oral Maxillofac. Surg. 2004, 62, 1225–1228. [Google Scholar] [CrossRef]
- Koo, B.S.; Lim, Y.C.; Lee, J.S.; Choi, E.C. Management of Contralateral N0 Neck in Oral Cavity Squamous Cell Carcinoma. Head Neck 2006, 28, 896–901. [Google Scholar] [CrossRef]
- Capote-Moreno, A.; Naval, L.; Muñoz-Guerra, M.F.; Sastre, J.; Rodríguez-Campo, F.J. Prognostic Factors Influencing Contralateral Neck Lymph Node Metastases in Oral and Oropharyngeal Carcinoma. J. Oral Maxillofac. Surg. 2010, 68, 268–275. [Google Scholar] [CrossRef]
- Flörke, C.; Gülses, A.; Altmann, C.R.; Wiltfang, J.; Wieker, H.; Naujokat, H. Clinicopathological Risk Factors for Contralateral Lymph Node Metastases in Intraoral Squamous Cell Carcinoma: A Study of 331 Cases. Curr. Oncol. 2021, 28, 1886–1898. [Google Scholar] [CrossRef]
- Lloyd, S.; Yu, J.B.; Wilson, L.D.; Judson, B.L.; Decker, R.H. The Prognostic Importance of Midline Involvement in Oral Tongue Cancer. Am. J. Clin. Oncol. Cancer Clin. Trials 2012, 35, 468–473. [Google Scholar] [CrossRef]
- Koerdt, S.; Röckl, J.; Rommel, N.; Mücke, T.; Wolff, K.D.; Kesting, M.R. Lymph Node Management in the Treatment of Oral Cancer: Analysis of a Standardized Approach. J. Cranio-Maxillofac. Surg. 2016, 44, 1737–1742. [Google Scholar] [CrossRef]
- Paleri, V.; Urbano, T.G.; Mehanna, H.; Repanos, C.; Lancaster, J.; Roques, T.; Patel, M.; Sen, M. Management of Neck Metastases in Head and Neck Cancer: United Kingdom National Multidisciplinary Guidelines. J. Laryngol. Otol. 2016, 130, S161–S169. [Google Scholar] [CrossRef]
- AJCC. Cancer Staging Manual, 8th ed.; Amin, M.B., Edge, S.B., Greene, F.L., Byrd, D.R., Brookland, R.K., Washington, M.K., Gershenwald, J.E., Compton, C.C., Hess, K.R., Sullivan, D.C., et al., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; ISBN 978-3-319-40617-6. [Google Scholar]
- Schilling, C.; Stoeckli, S.J.; Haerle, S.K.; Broglie, M.A.; Huber, G.F.; Sorensen, J.A.; Bakholdt, V.; Krogdahl, A.; Von Buchwald, C.; Bilde, A.; et al. Sentinel European Node Trial (SENT): 3-Year Results of Sentinel Node Biopsy in Oral Cancer. Eur. J. Cancer 2015, 51, 2777–2784. [Google Scholar] [CrossRef]
- Mølstrøm, J.; Grønne, M.; Green, A.; Bakholdt, V.; Sørensen, J.A. Topographical Distribution of Sentinel Nodes and Metastases from T1–T2 Oral Squamous Cell Carcinomas. Eur. J. Cancer 2019, 107, 86–92. [Google Scholar] [CrossRef]
- Voss, J.O.; Freund, L.; Neumann, F.; Mrosk, F.; Rubarth, K.; Kreutzer, K.; Doll, C.; Heiland, M.; Koerdt, S. Prognostic Value of Lymph Node Involvement in Oral Squamous Cell Carcinoma. Clin. Oral Investig. 2022, 26, 6711–6720. [Google Scholar] [CrossRef] [PubMed]
- Sayed, S.I.; Sharma, S.; Rane, P.; Vaishampayan, S.; Talole, S.; Chaturvedi, P.; Chaukar, D.; Deshmukh, A.; Agarwal, J.P.; D’Cruz, A.K. Can Metastatic Lymph Node Ratio (LNR) Predict Survival in Oral Cavity Cancer Patients? J. Surg. Oncol. 2013, 108, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.G.; Amit, M.; Yen, T.C.; Liao, C.T.; Chaturvedi, P.; Agarwal, J.P.; Kowalski, L.P.; Ebrahimi, A.; Clark, J.R.; Cernea, C.R.; et al. Lymph Node Density in Oral Cavity Cancer: Results of the International Consortium for Outcomes Research. Br. J. Cancer 2013, 109, 2087–2095. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, P.C.; Soto, A.M.L.; de Aguiar, M.C.F.; Martins, C.C. Tumor Depth of Invasion and Prognosis of Early-Stage Oral Squamous Cell Carcinoma: A Meta-Analysis. Oral Dis. 2020, 26, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Tam, S.; Amit, M.; Zafereo, M.; Bell, D.; Weber, R.S. Depth of Invasion as a Predictor of Nodal Disease and Survival in Patients with Oral Tongue Squamous Cell Carcinoma. Head Neck 2019, 41, 177–184. [Google Scholar] [CrossRef]
- Mahieu, R.; den Toom, I.J.; Boeve, K.; Lobeek, D.; Bloemena, E.; Donswijk, M.L.; de Keizer, B.; Klop, W.M.C.; Leemans, C.R.; Willems, S.M.; et al. Contralateral Regional Recurrence in Lateralized or Paramedian Early-Stage Oral Cancer Undergoing Sentinel Lymph Node Biopsy—Comparison to a Historic Elective Neck Dissection Cohort. Front. Oncol. 2021, 11, 644306. [Google Scholar] [CrossRef]
- Liu, H.Y.; Tam, L.; Woody, N.M.; Caudell, J.; Reddy, C.A.; Ghanem, A.; Schymick, M.; Joshi, N.; Geiger, J.; Lamarre, E.; et al. Failure Rate in the Untreated Contralateral Node Negative Neck of Small Lateralized Oral Cavity Cancers: A Multi-Institutional Collaborative Study. Oral Oncol. 2021, 115, 105190. [Google Scholar] [CrossRef]
- Contrera, K.J.; Huang, A.T.; Shenson, J.A.; Tang, C.; Roberts, D.; Myers, J.N.; Weber, R.S.; Lai, S.Y.; Williams, M.; El-Hallal, M.; et al. Primary and Recurrent Regional Metastases for Lateralized Oral Cavity Squamous Cell Carcinoma. Surg. Oncol. 2022, 44, 101804. [Google Scholar] [CrossRef]
- Ganly, I.; Goldstein, D.; Carlson, D.L.; Patel, S.G.; O’Sullivan, B.; Lee, N.; Gullane, P.; Shah, J.P. Long-Term Regional Control and Survival in Patients with “Low-Risk,” Early Stage Oral Tongue Cancer Managed by Partial Glossectomy and Neck Dissection without Postoperative Radiation: The Importance of Tumor Thickness. Cancer 2013, 119, 1168–1176. [Google Scholar] [CrossRef]
- Habib, M.; Murgasen, J.; Gao, K.; Ashford, B.; Shannon, K.; Ebrahimi, A.; Clark, J.R. Contralateral Neck Failure in Lateralized Oral Squamous Cell Carcinoma. ANZ J. Surg. 2016, 86, 188–192. [Google Scholar] [CrossRef]
- Nobis, C.P.; Otto, S.; Grigorieva, T.; Alnaqbi, M.; Troeltzsch, M.; Schöpe, J.; Wagenpfeil, S.; Ehrenfeld, M.; Wolff, K.D.; Kesting, M.R. Elective Neck Dissection in Unilateral Carcinomas of the Tongue: Unilateral versus Bilateral Approach. J. Cranio-Maxillofacial Surg. 2017, 45, 579–584. [Google Scholar] [CrossRef]
- Lim, Y.C.; Lee, J.S.; Koo, B.S.; Kim, S.H.; Kim, Y.H.; Choi, E.C. Treatment of Contralateral N0 Neck in Early Squamous Cell Carcinoma of the Oral Tongue: Elective Neck Dissection versus Observation. Laryngoscope 2006, 116, 461–465. [Google Scholar] [CrossRef]
- Knopf, A.; Jacob, S.; Bier, H.; Scherer, E.Q. Bilateral versus Ipsilateral Neck Dissection in Oral and Oropharyngeal Cancer with Contralateral CN0 Neck. Eur. Arch. Oto-Rhino-Laryngol. 2020, 277, 3161–3168. [Google Scholar] [CrossRef]
- Singhavi, H.R.; Nair, S.; Mair, M.; Mathur, R.; Singh, A.; Pai, A.; Patil, A.; Nair, D.; Chaturvedi, P. Addressing the Contralateral Neck for Ipsilateral Disease Recurrence in Oral Cavity Cancers. Eur. J. Surg. Oncol. 2021, 47, 1384–1388. [Google Scholar] [CrossRef]
- Liang, L.; Zhang, T.; Kong, Q.; Liang, J.; Liao, G. A Meta-Analysis on Selective versus Comprehensive Neck Dissection in Oral Squamous Cell Carcinoma Patients with Clinically Node-Positive Neck. Oral Oncol. 2015, 51, 1076–1081. [Google Scholar] [CrossRef]
- Kao, Y.S.; Hsu, Y. Adjuvant Contralateral Neck Irradiation for Oral Cavity Cancer—A Systematic Review and Meta-Analysis. Am. J. Otolaryngol.—Head Neck Med. Surg. 2021, 42, 102885. [Google Scholar] [CrossRef]
- Al-Mamgani, A.; van Werkhoven, E.; Navran, A.; Karakullukcu, B.; Hamming-Vrieze, O.; Machiels, M.; van der Velden, L.A.; Vogel, W.V.; Klop, W.M. Contralateral Regional Recurrence after Elective Unilateral Neck Irradiation in Oropharyngeal Carcinoma: A Literature-Based Critical Review. Cancer Treat. Rev. 2017, 59, 102–108. [Google Scholar] [CrossRef]
| Group I (Contralateral ND; n = 16, 24.6%) | Group II (No Contralateral ND; n = 49, 75.4%) | p-Value | |
|---|---|---|---|
| Gender | 0.59 | ||
| Female | 5 (31.3%) | 19 (38.8%) | |
| Male | 11 (68.7%) | 30 (61.2%) | |
| Age in years (mean) | 62.9 (±7.0) | 62.4 (±12.3) | 0.89 |
| Alcohol & Tobacco | 0.12 | ||
| Yes | 12 (75.0%) | 26 (53.1%) | |
| No | 4 (25.0%) | 23 (46.9%) | |
| cT Stage | 0.15 | ||
| cT1–2 | 7 (43.8%) | 32 (65.3%) | |
| cT3–4 | 9 (56.2%) | 17 (34.7%) | |
| Ipsilateral cN Stage | 0.93 | ||
| cN0 | 7 (43.8%) | 18 (36.7%) | |
| cN1 | 4 (25.0%) | 12 (24.5%) | |
| cN2a | 2 (12.5%) | 6 (12.2%) | |
| cN2b | 3 (18.8%) | 13 (26.5%) |
| Group I (Contralateral ND; n = 16, 24.6%) | Group II (No Contralateral ND; n = 49, 75.4%) | p-Value | |
|---|---|---|---|
| pT Stage | 0.52 | ||
| pT1–2 | 7 (43.8%) | 26 (53.1%) | |
| pT3–4 | 9 (56.2%) | 23 (46.9%) | |
| pN Stage | |||
| pN1 | 9 (56.2%) | 12 (24.5%) | |
| pN2a | 0 | 0 | |
| pN2b | 2 (12.5%) | 22 (44.9%) | |
| pN2c | 0 | 0 | |
| pN3a | 0 | 0 | |
| pN3b | 5 (31.3%) | 15 (30.6%) | |
| ECS | 0.96 | ||
| Yes | 5 (31.3%) | 15 (30.6%) | |
| No | 11 (68.7%) | 34 (69.4%) | |
| Grade of differentiation | 0.46 | ||
| G1 | 0 | 2 (4.1%) | |
| G2 | 14 (87.5%) | 36 (73.5%) | |
| G3 | 2 (12.5%) | 11 (22.4%) | |
| Depth of invasion (mean) | 12.0 (±7.4) | 10.8 (±6.6) | 0.57 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doll, C.; Mrosk, F.; Freund, L.; Neumann, F.; Kreutzer, K.; Voss, J.; Raguse, J.-D.; Beck, M.; Böhmer, D.; Rubarth, K.; et al. Management of the Contralateral Neck in Unilateral Node-Positive Oral Squamous Cell Carcinoma. Cancers 2023, 15, 1088. https://doi.org/10.3390/cancers15041088
Doll C, Mrosk F, Freund L, Neumann F, Kreutzer K, Voss J, Raguse J-D, Beck M, Böhmer D, Rubarth K, et al. Management of the Contralateral Neck in Unilateral Node-Positive Oral Squamous Cell Carcinoma. Cancers. 2023; 15(4):1088. https://doi.org/10.3390/cancers15041088
Chicago/Turabian StyleDoll, Christian, Friedrich Mrosk, Lea Freund, Felix Neumann, Kilian Kreutzer, Jan Voss, Jan-Dirk Raguse, Marcus Beck, Dirk Böhmer, Kerstin Rubarth, and et al. 2023. "Management of the Contralateral Neck in Unilateral Node-Positive Oral Squamous Cell Carcinoma" Cancers 15, no. 4: 1088. https://doi.org/10.3390/cancers15041088
APA StyleDoll, C., Mrosk, F., Freund, L., Neumann, F., Kreutzer, K., Voss, J., Raguse, J.-D., Beck, M., Böhmer, D., Rubarth, K., Heiland, M., & Koerdt, S. (2023). Management of the Contralateral Neck in Unilateral Node-Positive Oral Squamous Cell Carcinoma. Cancers, 15(4), 1088. https://doi.org/10.3390/cancers15041088







