Effect of Cancer-Related Cachexia and Associated Changes in Nutritional Status, Inflammatory Status, and Muscle Mass on Immunotherapy Efficacy and Survival in Patients with Advanced Non-Small Cell Lung Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria
2.3. Outcomes
2.4. Collection of Clinical and Laboratory Data
2.4.1. Assessment of Skeletal Muscle Mass and SMI
2.4.2. CT image Analysis
2.4.3. Assessment of Cachexia Using the miniCASCO
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.1.1. Distribution of Patients Based on BMI and the Presence of Weight Loss and Sarcopenia
3.1.2. Baseline Body Composition and Nutritional and Inflammatory Parameters
| Parameter (Normal Value) | Mean Value ± SD (Range) | N. (%) |
|---|---|---|
| BMI, kg/m2 | ||
| <18.5 | 4 (5.4) | |
| 18.5–25 | 38 (51.4) | |
| ≥25–30 | 24 (32.4) | |
| ≥30 | 8 (10.8) | |
| LBM, kg (NA) | 30.5 ± 9.3 (15–48.6) | |
| SMI, cm2/m2 | 43.9 ± 12.8 (24–69) | |
| Sarcopenia | 51 (68.9) | |
| SMI ≤39 cm2/m2 for women | ||
| SMI ≤55 cm2/m2 for men | ||
| CRP, mg/L (0–5) | 22.5 ± 16.7 (0.1–150.8) | |
| IL-6, pg/mL (0–7) | 12.8 ± 3.3 (1–85) | |
| Neutrophils, cells/microl (5000–10000) | 5700 ± 2578.7 (2000–12800) | |
| Lymphocytes, cells/microl (1200–4000) | 1703 ± 580.9 (600–3300) | |
| NLR (NA) | 3.8 ± 2.3 (1–12) | |
| Albumin, g/dL (>3.2) | 3.5 ± 0.4 (2.6–4.4) | |
| Hb, g/dL (≥12) | 12.2 ± 1.9 (7.7–16.3) | |
| mGPS | ||
| 0 | 20 (27) | |
| 1 | 20 (27) | |
| 2 | 34 (46) |
3.1.3. Analysis of the Findings of the miniCASCO Questionnaire and Its Subscales
3.2. Association between Classic Prognostic Factors (PD-L1 Expression and ECOG PS) and Clinical Outcomes (Objective Clinical Response, PFS, and OS)
3.3. Association between Cachexia Status, miniCASCO, Body Composition, Inflammatory Status, and Nutritional Status and the Clinical Response (Disease Control Rate)
3.4. Cox Regression Analysis of Variables Determinant of PFS and OS
| Variable | PFS | OS | ||
|---|---|---|---|---|
| Adjusted Exp(b) (95%CI) | p-Value | Adjusted Exp(b) (95%CI) | p-Value | |
| IL-6 | 1.0388 (1.0230–1.0548) | <0.0001 | 1.0404 (1.0221–1.0589) | <0.0001 |
| miniCASCO-based cachexia severity | 1.2587 (1.0850–1.4602) | 0.0024 | 2.3834 (1.1504–4.9378) | 0.0194 |
| Cachexia according to the Fearon criteria | - | - | 0.7774 (0.4107–1.4715) | 0.4392 |
| mGPS | 1.3171 (0.9881–1.5936 | 0.0056 | 1.3494 (0.8355–2.1796) | 0.2205 |
| miniCASCO | 0.9797 (0.9428–1.0180) | 0.2948 | 0.9884 (0.9479–1.0307) | 0.5855 |
| IMD subscale | 0.9760 (0.8908–1.0693) | 0.6018 | 1.0003 (0.9089–1.1009) | 0.9955 |
| QoL subscale | 1.4822 (0.7517–2.9226) | 0.2560 | - | - |
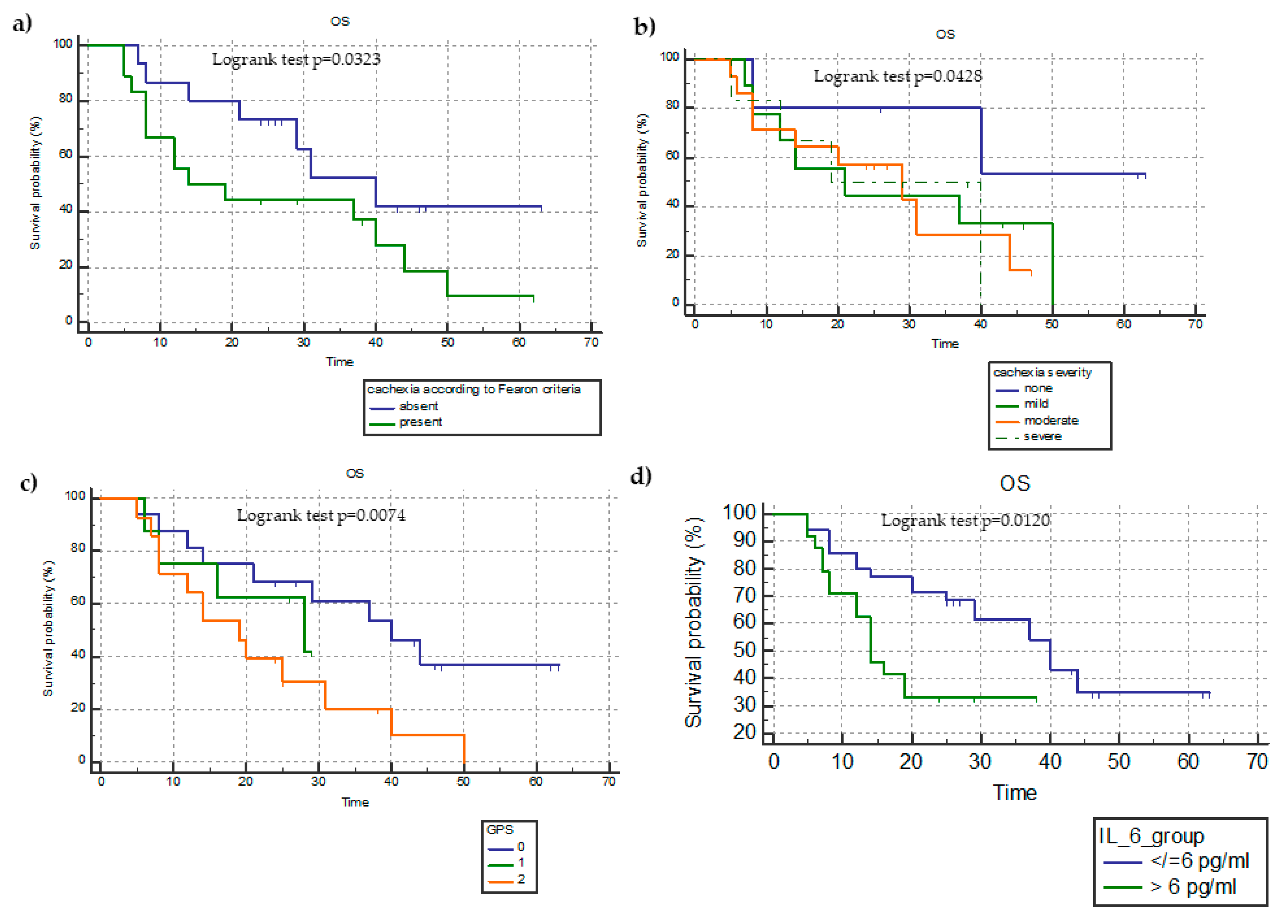
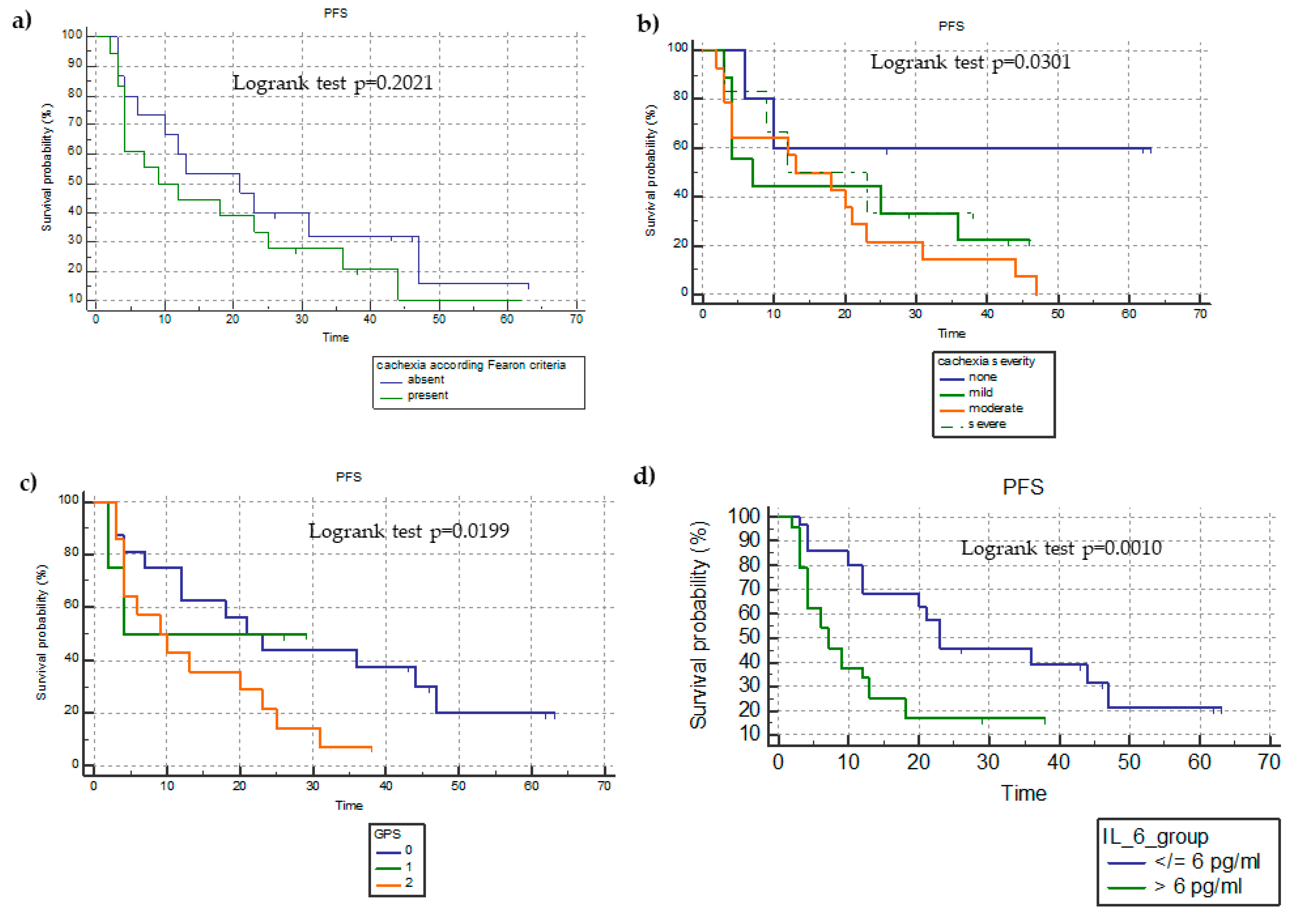
3.5. Kaplan–Meier Survival Analysis of OS and PFS in Terms of the Presence of Cachexia, miniCASCO-Based Cachexia Severity, Sarcopenia Status, GPS Categories, and IL-6 Values
3.6. Changes in Inflammatory Status, Nutritional Status, SMI, and miniCASCO Based on the Objective Clinical Response to Treatment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thai, A.A.; Solomon, B.J.; Sequist, L.V.; Gainor, J.F.; Heist, R.S. Lung cancer. Lancet 2021, 398, 535–554. [Google Scholar] [CrossRef] [PubMed]
- Garon, E.B.; Hellmann, M.D.; Rizvi, N.A.; Carcereny, E.; Leighl, N.B.; Ahn, M.J.; Eder, J.P.; Balmanoukian, A.S.; Aggarwal, C.; Horn, L.; et al. Five-year overall survival for patients with advanced non-small-cell lung cancer treated with pembrolizumab: Results from the phase I KEYNOTE-001 study. J. Clin. Oncol. 2019, 37, 2518–2527. [Google Scholar] [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Sosman, J.A.; Atkins, M.B.; Leming, P.D.; et al. Five year survival and correlates among patients with advanced melanoma, renal cell carcinoma, or non-small cell lung cancer treated with nivolumab. JAMA Oncol. 2019, 5, 1411. [Google Scholar] [CrossRef] [PubMed]
- Bagley, S.J.; Kothari, S.; Aggarwal, C.; Bauml, J.M.; Alley, E.W.; Evans, T.L.; Kosteva, J.A.; Ciunci, C.A.; Gabriel, P.E.; Thompson, J.C.; et al. Pretreatment neutrophil-to-lymphocyte ratio as a marker of outcomes in nivolumab-treated patients with advanced non-small-cell lung cancer. Lung Cancer 2017, 106, 1–7. [Google Scholar] [CrossRef]
- Jafri, S.H.; Shi, R.; Mills, G. Advance lung cancer inflammation index (ALI) at diagnosis is a prognostic marker in patients with metastatic non-small cell lung cancer (NSCLC): A retrospective review. BMC Cancer 2013, 13, 158. [Google Scholar] [CrossRef]
- Shiroyama, T.; Suzuki, H.; Tamiya, M.; Tamiya, A.; Tanaka, A.; Okamoto, N.; Nakahama, K.; Taniguchi, Y.; Isa, S.I.; Inoue, T.; et al. Pretreatment advanced lung cancer inflammation index (ALI) for predicting early progression in nivolumab-treated patients with advanced non-small cell lung cancer. Cancer Med. 2018, 7, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Araki, T.; Tateishi, K.; Sonehara, K.; Hirota, S.; Komatsu, M.; Yamamoto, M.; Kanda, S.; Kuraishi, H.; Hanaoka, M.; Koizumi, T. Clinical utility of the C-reactive protein:albumin ratio in non-small cell lung cancer patients treated with nivolumab. Thorac. Cancer 2021, 12, 603–612. [Google Scholar] [CrossRef]
- Matsubara, T.; Takamori, S.; Haratake, N.; Toyozawa, R.; Miura, N.; Shimokawa, M.; Yamaguchi, M.; Seto, T.; Takenoyama, M. The impact of immune-inflammation-nutritional parameters on the prognosis of non-small cell lung cancer patients treated with atezolizumab. J. Thorac. Dis. 2020, 12, 1520–1528. [Google Scholar] [CrossRef]
- Russo, A.; Franchina, T.; Ricciardi, G.R.R.; Battaglia, A.; Scimone, A.; Berenato, R.; Giordano, A.; Adamo, V. Baseline neutrophilia, derived neutrophil-to-lymphocyte ratio (dNLR), platelet-tolymphocyte ratio (PLR), and outcome in non-small cell lung cancer (NSCLC) treated with Nivolumab or Docetaxel. J. Cell. Physiol. 2018, 233, 6337–6343. [Google Scholar] [CrossRef]
- Jiang, T.; Bai, Y.; Zhou, F.; Li, W.; Gao, G.; Su, C.; Ren, S.; Chen, X.; Zhou, C. Clinical value of neutrophil-to lymphocyte ratio in patients with non-small-cell lung cancer treated with PD-1/PD-L1 inhibitors. Lung Cancer 2019, 130, 76–83. [Google Scholar] [CrossRef]
- Deng, H.Y.; Chen, Z.J.; Qiu, X.M.; Zhu, D.X.; Tang, X.J.; Zhou, Q. Sarcopenia and prognosis of advanced cancer patients receiving immune checkpoint inhibitors: A comprehensive systematic review and meta-analysis. Nutrition 2021, 90, 111345. [Google Scholar] [CrossRef] [PubMed]
- Bozzetti, F. Forcing the vicious circle: Sarcopenia increases toxicity, decreases response to chemotherapy and worsens with chemotherapy. Ann. Oncol. 2017, 28, 2107–2118. [Google Scholar] [CrossRef] [PubMed]
- Cortellini, A.; Palumbo, P.; Porzio, G.; Verna, L.; Giordano, A.V.; Masciocchi, C.; Parisi, A.; Cannita, K.; Ficorella, C.; Bozzetti, F. Single-institution study of correlations between skeletal muscle mass, its density, and clinical outcomes in nonsmall cell lung cancer patients treated with first-line chemotherapy. Thorac. Cancer 2018, 9, 1623–1630. [Google Scholar] [CrossRef]
- Shachar, S.S.; Williams, G.R.; Muss, H.B.; Nishijima, T.F. Prognostic value of sarcopenia in adults with solid tumours: A meta-analysis and systematic review. Eur. J. Cancer 2016, 57, 58–67. [Google Scholar] [CrossRef]
- Yang, M.; Shen, Y.; Tan, L.; Li, W. Prognostic value of sarcopenia in lung cancer: A systematic review and meta-analysis. Chest 2019, 156, 101–111. [Google Scholar] [CrossRef]
- Degens, J.; Sanders, K.J.C.; de Jong, E.E.C.; Groen, H.J.M.; Smit, E.F.; Aerts, J.G.; Schols, A.M.W.J.; Dingemans, A.M.C. The prognostic value of early onset, CT derived loss of muscle and adipose tissue during chemotherapy in metastatic non-small cell lung cancer. Lung Cancer 2019, 133, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Baracos, V.E.; Martin, L.; Korc, M.; Guttridge, D.C.; Fearon, K.C.H. Cancer-associated cachexia. Nat. Rev. Dis. Primers 2018, 4, 17105. [Google Scholar] [CrossRef]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Argilés, J.M.; Betancourt, A.; Guàrdia-Olmos, J.; Peró-Cebollero, M.; López-Soriano, F.J.; Madeddu, C.; Serpe, R.; Busquets, S. Validation of the CAchexia SCOre (CASCO). Staging Cancer Patients: The Use of miniCASCO as a Simplified Tool. Front. Physiol. 2017, 8, 92. [Google Scholar] [CrossRef]
- World Health Organization. Cancer. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 7 February 2022).
- Baracos, V.E.; Reiman, T.; Mourtzakis, M.; Gioulbasanis, I.; Antoun, S. Body composition in patients with non-small cell lung cancer: A contemporary view of cancer cachexia with the use of computed tomography image analysis. Am. J. Clin. Nutr. 2010, 91, 1133S–1137S. [Google Scholar] [CrossRef]
- Muscaritoli, M.; Lucia, S.; Farcomeni, A.; Lorusso, V.; Saracino, V.; Barone, C.; Plastino, F.; Gori, S.; Magarotto, R.; Carteni, G.; et al. Prevalence of malnutrition in patients at first medical oncology visit: The PreMiO study. Oncotarget 2017, 8, 79884–79896. [Google Scholar] [CrossRef] [PubMed]
- Maccio, A.; Sanna, E.; Neri, M.; Oppi, S.; Madeddu, C. Cachexia as Evidence of the Mechanisms of Resistance and Tolerance during the Evolution of Cancer Disease. Int. J. Mol. Sci. 2021, 22, 2890. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.; Strasser, F.; Gonella, S.; Solheim, T.S.; Madeddu, C.; Ravasco, P.; Buonaccorso, L.; de van der Schueren, M.A.E.; Baldwin, C.; Chasen, M.; et al. Cancer cachexia in adult patients: ESMO Clinical Practice Guidelines☆. ESMO Open 2021, 6, 100092. [Google Scholar] [CrossRef]
- Cuenca, A.G.; Cuenca, A.L.; Winfield, R.D.; Joiner, D.N.; Gentile, L.; Delano, M.J.; Kelly-Scumpia, K.M.; Scumpia, P.O.; Matheny, M.K.; Scarpace, P.J.; et al. Novel role for tumor-induced expansion of myeloid-derived cells in cancer cachexia. J. Immunol. 2014, 192, 6111–6119. [Google Scholar] [CrossRef]
- Flint, T.R.; Janowitz, T.; Connell, C.M.; Roberts, E.W.; Denton, A.E.; Coll, A.P.; Jodrell, D.I.; Fearon, D.T. Tumor-Induced IL-6 Reprograms Host Metabolism to Suppress Antitumor Immunity. Cell Metab. 2016, 24, 672–684. [Google Scholar] [CrossRef]
- Macciò, A.; Madeddu, C. Blocking inflammation to improve immunotherapy of advanced cancer. Immunology 2020, 159, 357–364. [Google Scholar] [CrossRef]
- Shiroyama, T.; Nagatomo, I.; Koyama, S.; Hirata, H.; Nishida, S.; Miyake, K.; Fukushima, K.; Shirai, Y.; Mitsui, Y.; Takata, S.; et al. Impact of sarcopenia in patients with advanced non-small cell lung cancer treated with PD-1 inhibitors: A preliminary retrospective study. Sci. Rep. 2019, 9, 2447. [Google Scholar] [CrossRef]
- Tsukagoshi, M.; Yokobori, T.; Yajima, T.; Maeno, T.; Shimizu, K.; Mogi, A.; Araki, K.; Harimoto, N.; Shirabe, K.; Kaira, K. Skeletal muscle mass predicts the outcome of nivolumab treatment for non-small cell lung cancer. Medicine 2020, 99, e19059. [Google Scholar] [CrossRef]
- Takada, K.; Yoneshima, Y.; Tanaka, K.; Okamoto, I.; Shimokawa, M.; Wakasu, S.; Takamori, S.; Toyokawa, G.; Oba, T.; Osoegawa, A.; et al. Clinical impact of skeletal muscle area in patients with non-small cell lung cancer treated with anti-PD-1 inhibitors. J. Cancer Res. Clin. Oncol. 2020, 146, 1217–1225. [Google Scholar] [CrossRef]
- Nishioka, N.; Uchino, J.; Hirai, S.; Katayama, Y.; Yoshimura, A.; Okura, N.; Tanimura, K.; Harita, S.; Imabayashi, T.; Chihara, Y.; et al. Association of Sarcopenia with and Efficacy of Anti-PD-1/PD-L1 Therapy in Non-Small-Cell Lung Cancer. J. Clin. Med. 2019, 8, 450. [Google Scholar] [CrossRef]
- Cortellini, A.; Verna, L.; Porzio, G.; Bozzetti, F.; Palumbo, P.; Masciocchi, C.; Cannita, K.; Parisi, A.; Brocco, D.; Tinari, N.; et al. Predictive value of skeletal muscle mass for immunotherapy with nivolumab in non-small cell lung cancer patients: A “hypothesis-generator” preliminary report. Thorac. Cancer 2019, 10, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Petrova, M.P.; Donev, I.S.; Radanova, M.A.; Eneva, M.I.; Dimitrova, E.G.; Valchev, G.N.; Minchev, V.T.; Taushanova, M.S.; Boneva, M.V.; Karanikolova, T.S.; et al. Sarcopenia and high NLR are associated with the development of hyperprogressive disease after second-line pembrolizumab in patients with non-small-cell lung cancer. Clin. Exp. Immunol. 2020, 202, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Roch, B.; Coffy, A.; Jean-Baptiste, S.; Palaysi, E.; Daures, J.P.; Pujol, J.L.; Bommart, S. Cachexia-sarcopenia as a determinant of disease control rate and survival in non-small lung cancer patients receiving immune-checkpoint inhibitors. Lung Cancer 2020, 143, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Mourtzakis, M.; Prado, C.M.M.; Lieffers, J.R.; Reiman, T.; McCargar, L.J.; Baracos, V.E. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl. Physiol. Nutr. Metab. 2008, 33, 997–1006. [Google Scholar] [CrossRef]
- Vokes, E.E.; Ready, N.; Felip, E.; Horn, L.; Burgio, M.A.; Antonia, S.J.; Frontera, O.A.; Gettinger, S.; Holgado, E.; Spigel, D.; et al. Nivolumab versus docetaxel in previously treated advanced non-small-cell lung cancer (CheckMate 017 and CheckMate 057): 3-year update and outcomes in patients with liver metastases. Ann. Oncol. 2018, 29, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodriguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Updated analysis of KEYNOTE-024: Pembrolizumab versus platinum based chemotherapy for advanced non-small-cell lung cancer with PD-L1 tumor proportion score of 50% or greater. J. Clin. Oncol. 2019, 37, 537–546. [Google Scholar] [CrossRef]
- Peters, S.; Reck, M.; Smit, E.F.; Mok, T.; Hellmann, M.D. How to make the best use of immunotherapy as first-line treatment of advanced/metastatic non-small-cell lung cancer. Ann. Oncol. 2019, 30, 884–896. [Google Scholar] [CrossRef]
- Rizvi, N.A.; Hellmann, M.D.; Snyder, A.; Kvistborg, P.; Makarov, V.; Havel, J.J.; Lee, W.; Yuan, J.; Wong, P.; Ho, T.S.; et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015, 348, 124–128. [Google Scholar] [CrossRef]
- Boudou-Rouquette, P.; Arrondeau, J.; Gervais, C.; Durand, J.; Fabre, E.; De Percin, S.; Villeminey, C.V.; Piketty, A.; Rassy, N.; Ulmann, G.; et al. Development and validation of a host-dependent, PDL1-independent, biomarker to predict 6-month progression-free survival in metastatic non-small cell lung cancer (mNSCLC) patients treated with anti-PD1 immune checkpoint inhibitors (ICI) in the CERTIM Cohort: The ELY study. EBiomedicine 2021, 73, 103630. [Google Scholar] [CrossRef]
- Meng, X.; Huang, Z.; Teng, F.; Xing, L.; Yu, J. Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat. Rev. 2015, 41, 868–876. [Google Scholar] [CrossRef]
- Yarchoan, M.; Hopkins, A.; Jaffee, E.M. Tumor mutational burden and response rate to PD-1 Inhibition. N. Engl. J. Med. 2017, 377, 2500–2501. [Google Scholar] [CrossRef] [PubMed]
- Mouw, K.W.; Goldberg, M.S.; Konstantinopoulos, P.A.; D’Andrea, A.D. DNA Damage and Repair Biomarkers of Immunotherapy Response. Cancer Discov. 2017, 7, 675–693. [Google Scholar] [CrossRef] [PubMed]
- Gettinger, S.N.; Choi, J.; Mani, N.; Sanmamed, M.F.; Datar, I.; Sowell, R.; Du, V.Y.; Kaftan, E.; Goldberg, S.; Dong, W.; et al. A dormant TIL phenotype defines non-small cell lung carcinomas sensitive to immune checkpoint blockers. Nat. Commun. 2018, 9, 3196. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. KEYNOTE-024 Investigators. Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crinò, L.; Eberhardt, W.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef]
- Lutz, C.T.; Quinn, L.S. Sarcopenia, obesity, and natural killer cell immune senescence in aging: Altered cytokine levels as a common mechanism. Aging 2012, 4, 535–546. [Google Scholar] [CrossRef]
- Miyawaki, T.; Naito, T.; Kodama, A.; Nishioka, N.; Miyawaki, E.; Mamesaya, N.; Kawamura, T.; Kobayashi, H.; Omori, S.; Wakuda, K.; et al. Desensitizing effect of cancer cachexia on immune checkpoint inhibitors in patients with advanced NSCLC. JTO Clin. Res. Rep. 2020, 1, 100020. [Google Scholar] [CrossRef]
- Miyawaki, T.; Naito, T.; Yabe, M.; Kodama, H.; Nishioka, N.; Miyawaki, E.; Mamesaya, N.; Kobayashi, H.; Omori, S.; Wakuda, K.; et al. Impact of weight loss on treatment with PD-1/PD-L1 inhibitors plus chemotherapy in advanced non-small-cell lung cancer. Support Care Cancer 2022, 30, 1633–1641. [Google Scholar] [CrossRef]
- Morimoto, K.; Uchino, J.; Yokoi, T.; Kijima, T.; Goto, Y.; Nakao, A.; Hibino, M.; Takeda, T.; Yamaguchi, H.; Takumi, C.; et al. Impact of cancer cachexia on the therapeutic outcome of combined chemoimmunotherapy in patients with non-small cell lung cancer: A retrospective study. Oncoimmunology 2021, 10, 1950411. [Google Scholar] [CrossRef]
- Magri, V.; Gottfried, T.; Di Segni, M.; Urban, D.; Peled, M.; Daher, S.; Stoff, R.; Bar, J.; Onn, A. Correlation of body composition by computerized tomography and metabolic parameters with survival of nivolumab-treated lung cancer patients. Cancer Manag. Res. 2019, 11, 8201–8207. [Google Scholar] [CrossRef]
- Rounis, K.; Makrakis, D.; Tsigkas, A.; Georgiou, A.; Galanakis, N.; Papadaki, C.; Monastirioti, A.; Vamvakas, L.; Kalbakis, K.; Vardakis, N.; et al. Cancer cachexia syndrome and clinical outcome in patients with metastatic non-small cell lung cancer treated with PD-1/PD-L1 inhibitors: Results from a prospective, observational study. Transl. Lung Cancer Res. 2021, 10, 3538–3549. [Google Scholar] [CrossRef] [PubMed]
- Turcott, J.G.; Martinez-Samano, J.E.; Cardona, A.F.; Bassarmal, S.S.; Ramírez-Tirado, L.A.; Zatarain-Barrón, Z.L.; Barrón, F.; Corrales, L.; Martín, C.; Barragán-Castillo, P.A.; et al. The Role of a Cachexia Grading System in Patients with Non-Small Cell Lung Cancer Treated with Immunotherapy: Implications for Survival. Nutr. Cancer 2021, 73, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, T.; Naito, T.; Doshita, K.; Kodama, H.; Mori, M.; Nishioka, N.; Iida, Y.; Miyawaki, E.; Mamesaya, N.; Kobayashi, H.; et al. Predicting the efficacy of first-line immunotherapy by combining cancer cachexia and tumor burden in advanced non-small cell lung cancer. Thorac Cancer 2022, 13, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, P.; Huang, J.; Liu, M.; Peng, D.; Li, Z.; Chen, T.; Hong, S.; Zhou, Y. Assessment of sarcopenia as a predictor of poor overall survival for advanced non-small-cell lung cancer patients receiving salvage anti-PD-1 immunotherapy. Ann. Transl. Med. 2021, 9, 1801. [Google Scholar] [CrossRef]
- Tenuta, M.; Gelibter, A.; Pandozzi, C.; Sirgiovanni, G.; Campolo, F.; Venneri, M.A.; Caponnetto, S.; Cortesi, E.; Marchetti, P.; Isidori, A.; et al. Impact of Sarcopenia and Inflammation on Patients with Advanced Non-Small Cell Lung Cancer (NCSCL) Treated with Immune Checkpoint Inhibitors (ICIs): A Prospective Study. Cancers 2021, 13, 6355. [Google Scholar] [CrossRef]
- Wang, J.; Cao, L.; Xu, S. Sarcopenia affects clinical efficacy of immune checkpoint inhibitors in non-small cell lung cancer patients: A systematic review and meta-analysis. Int. Immunopharmacol. 2020, 88, 106907. [Google Scholar] [CrossRef]
- Dercle, L.; Ammari, S.; Champiat, S.; Massard, C.; Ferté, C.; Taihi, L.; Seban, R.; Aspeslagh, S.; Mahjoubi, L.; Kamsu-Kom, N.; et al. Rapid and objective CT scan prognostic scoring identifies metastatic patients with long-term clinical benefit on anti-PD-1/-L1 therapy. Eur. J. Cancer 2016, 65, 33–42. [Google Scholar] [CrossRef]
- Cortellini, A.; Bozzetti, F.; Palumbo, P.; Brocco, D.; Di Marino, P.; Tinari, N.; De Tursi, M.; Agostinelli, V.; Patruno, L.; Valdesi, C.; et al. Weighing the role of skeletal muscle mass and muscle density in cancer patients receiving PD-1/PD-L1 checkpoint inhibitors: A multicenter real-life study. Sci. Rep. 2020, 10, 1456. [Google Scholar] [CrossRef]
- Baldessari, C.; Guaitoli, G.; Valoriani, F.; Bonacini, R.; Marcheselli, R.; Reverberi, L.; Pecchi, A.; Menozzi, R.; Torricelli, P.; Bertolini, F.; et al. Impact of body composition, nutritional and inflammatory status on outcome of non-small cell lung cancer patients treated with immunotherapy. Clin. Nutr. ESPEN 2021, 43, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Macciò, A.; Oppi, S.; Madeddu, C. COVID-19 and cytokine storm syndrome: Can what we know about interleukin-6 in ovarian cancer be applied? J. Ovarian Res. 2021, 14, 28. [Google Scholar] [CrossRef]
- Madeddu, C.; Mantovani, G.; Gramignano, G.; Astara, G.; Macciò, A. Muscle wasting as main evidence of energy impairment in cancer cachexia: Future therapeutic approaches. Future Oncol. 2015, 11, 2697–2710. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Cánoves, P.; Scheele, C.; Pedersen, B.K.; Serrano, A.L. Interleukin-6 myokine signaling in skeletal muscle: A double-edged sword? FEBS J. 2013, 280, 4131–4148. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, P.C.; Castell, J.V.; Andus, T. Interleukin-6 and the acute phase response. Biochem. J. 1990, 265, 621–636. [Google Scholar] [CrossRef] [PubMed]
- Macciò, A.; Lai, P.; Santona, M.C.; Pagliara, L.; Melis, G.B.; Mantovani, G. High serum levels of soluble IL-2 receptor, cytokines, and C reactive protein correlate with impairment of T cell response in patients with advanced epithelial ovarian cancer. Gynecol. Oncol. 1998, 69, 248–252. [Google Scholar] [CrossRef]
- Bano, G.; Trevisan, C.; Carraro, S.; Solmi, M.; Luchini, C.; Stubbs, B.; Manzato, E.; Sergi, G.; Veronese, N. Inflammation and sarcopenia: A systematic review and meta-analysis. Maturitas 2017, 96, 10–15. [Google Scholar] [CrossRef]
- Öztürk, Z.A.; Kul, S.; Türkbeyler, İ.H.; Sayıner, Z.A.; Abiyev, A. Is increased neutrophil lymphocyte ratio remarking the inflammation in sarcopenia? Exp. Gerontol. 2018, 110, 223–229. [Google Scholar] [CrossRef]
- Chambard, L.; Girard, N.; Ollier, E.; Rousseau, J.C.; Duboeuf, F.; Carlier, M.C.; Brevet, M.; Szulc, P.; Pialat, J.; Wegrzyn, J.; et al. Bone, muscle, and metabolic parameters predict survival in patients with synchronous bone metastases from lung cancers. Bone 2018, 108, 202–209. [Google Scholar] [CrossRef]
- McMillan, D.C. The systemic inflammation-based Glasgow Prognostic Score: A decade of experience in patients with cancer. Cancer Treat. Rev. 2013, 39, 534–540. [Google Scholar] [CrossRef]
- Simmons, C.P.; Koinis, F.; Fallon, M.T.; Fearon, K.C.; Bowden, J.; Solheim, T.S.; Gronberg, B.H.; McMillan, D.C.; Gioulbasanis, I.; Laird, B.J. Prognosis in advanced lung cancer–a prospective study examining key clinicopathological factors. Lung Cancer 2015, 88, 304–309. [Google Scholar] [CrossRef]
- Laird, B.J.; Kaasa, S.; McMillan, D.C.; Fallon, M.T.; Hjermstad, M.J.; Fayers, P.; Klepstad, P. Prognostic factors in patients with advanced cancer: A comparison of clinicopathological factors and the development of an inflammation-based prognostic system. Clin. Cancer Res. 2013, 19, 5456–5464. [Google Scholar] [CrossRef]
- Srdic, D.; Plestina, S.; Sverko-Peternac, A.; Nikolac, N.; Simundic, A.M.; Samarzija, M. Cancer cachexia, sarcopenia and biochemical markers in patients with advanced non-small cell lung cancer-chemotherapy toxicity and prognostic value. Support Care Canc. 2016, 24, 4495–4502. [Google Scholar] [CrossRef]
- Wang, W.; Huang, Z.; Yu, Z.; Zhuang, W.; Zheng, W.; Cai, Z.; Shi, L.; Yu, X.; Lou, G.; Hong, W.; et al. Prognostic Value of the Lung Immune Prognostic Index May Differ in Patients Treated With Immune Checkpoint Inhibitor Monotherapy or Combined With Chemotherapy for Non-small Cell Lung Cancer. Front. Oncol. 2020, 10, 572853. [Google Scholar] [CrossRef] [PubMed]
- Mezquita, L.; Auclin, E.; Ferrara, R.; Charrier, M.; Remon, J.; Planchard, D.; Ponce, S.; Ares, L.P.; Leroy, L.; Audigier-Valette, C.; et al. Association of the Lung Immune Prognostic Index with Immune Checkpoint Inhibitor Outcomes in Patients with Advanced Non–Small Cell Lung Cancer. JAMA Oncol. 2018, 4, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Shoji, F.; Takeoka, H.; Kozuma, Y.; Toyokawa, G.; Yamazaki, K.; Ichiki, M.; Takeo, S. Pretreatment prognostic nutritional index as a novel biomarker in non-small cell lung cancer patients treated with immune checkpoint inhibitors. Lung Canc. 2019, 136, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Yu, J.; Li, W.; Luo, J.; Deng, Q.; Chen, B.; He, Y.; Zhang, J.; Zhou, C. Prognostic value of inflammatory and nutritional indexes among advanced NSCLC patients receiving PD-1 inhibitor therapy. Clin. Exp. Pharm. Physiol. 2022, 50, 178–190. [Google Scholar] [CrossRef]
- Jin, J.; Yang, L.; Liu, D.; Li, W. Association of the neutrophil to lymphocyte ratio and clinical outcomes in patients with lung cancer receiving immunotherapy: A meta-analysis. BMJ Open 2020, 10, e035031. [Google Scholar] [CrossRef]
- Petrova, M.P.; Eneva, M.I.; Arabadjiev, J.I.; Conev, N.V.; Dimitrova, E.G.; Koynov, K.D.; Karanikolova, T.S.; Valev, S.S.; Gencheva, R.B.; Zhbantov, G.A.; et al. Neutrophil to lymphocyte ratio as a potential predictive marker for treatment with pembrolizumab as a second line treatment in patients with non-small cell lung cancer. BioSci. Trends 2020, 14, 48–55. [Google Scholar] [CrossRef]
- Teramukai, S.; Kitano, T.; Kishida, Y.; Kawahara, M.; Kubota, K.; Komuta, K.; Minato, K.; Mio, T.; Fujita, Y.; Yonei, T.; et al. Pretreatment neutrophil count as an independent prognostic factor in advanced non-small-cell lung cancer: An analysis of Japan Multinational Trial Organisation LC00-03. Eur. J. Cancer 2009, 45, 1950–1958. [Google Scholar] [CrossRef]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Five-Year Outcomes With Pembrolizumab Versus Chemotherapy for Metastatic Non-Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score ≥ 50. J. Clin. Oncol. 2021, 39, 2339–2349. [Google Scholar] [CrossRef]
- Madeddu, C.; Sanna, E.; Nemolato, S.; Mulas, O.; Oppi, S.; Scartozzi, M.; La Nasa, G.; Maccio, A. Pathogenic and Prognostic Roles of Paraneoplastic Leukocytosis in Cervical Cancer: Can Genomic-Based Targeted Therapies Have a Role? A Literature Review and an Emblematic Case Report. Diagnostics 2022, 12, 1910. [Google Scholar] [CrossRef]
- Dumitru, C.A.; Lang, S.; Brandau, S. Modulation of neutrophil granulocytes in the tumor microenvironment: Mechanisms and consequences for tumor progression. Semin. Cancer Biol. 2013, 23, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Gooden, M.J.; de Bock, G.H.; Leffers, N.; Daemen, T.; Nijman, H.W. The prognostic influence of tumour-infiltrating lymphocytes in cancer: A systematic review with meta-analysis. Br. J. Cancer 2011, 105, 93–103. [Google Scholar] [CrossRef]
- Mantovani, G.; Macciò, A.; Pisano, M.; Versace, R.; Lai, P.; Esu, S.; Massa, E.; Ghiani, M.; Dessì, D.; Melis, G.B.; et al. Tumor-associated lympho-monocytes from neoplastic effusions are immunologically defective in comparison with patient autologous PBMCs but are capable of releasing high amounts of various cytokines. Int. J. Cancer 1997, 71, 724–731. [Google Scholar]
- Mantovani, G.; Macciò, A.; Versace, R.; Lai, P.; Esu, S.; Massa, W.; Ghiani, M.; Dessì, D.; Melis, G.B.; Del Giacco, G.S. Tumor-associated lymphocytes (TAL) are competent to produce higher levels of cytokines in neoplastic pleural and peritoneal effusions than those found in sera and are able to release into culture higher levels of IL-2 and IL-6 than those released by PBMC. J. Mol. Med. 1995, 73, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Mengheri, E.; Nobili, F.; Crocchioni, G.; Lewis, J.A. Protein starvation impairs the ability of activated lymphocytes to produce interferon-gamma. J. Interf. Res. 1992, 12, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Alwarawrah, Y.; Kiernan, K.; MacIver, N.J. Changes in nutritional status impact immune cell metabolism and function. Front. Immunol. 2018, 9, 1055. [Google Scholar] [CrossRef]
- Chhetri, J.K.; de Souto Barreto, P.; Fougere, B.; Rolland, Y.; Vellas, B.; Cesari, M. Chronic inflammation and sarcopenia: A regenerative cell therapy perspective. Exp. Gerontol. 2018, 103, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Crusz, S.M.; Balkwill, F.R. Inflammation and cancer: Advances and new agents. Nat. Rev. Clin. Oncol. 2015, 12, 584–596. [Google Scholar] [CrossRef]
- Greten, F.R.; Grivennikov, S.I. Inflammation and cancer: Triggers, mechanisms, and consequences. Immunity 2019, 51, 27–41. [Google Scholar] [CrossRef]
- Wherry, E.J.; Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 2015, 15, 486–499. [Google Scholar] [CrossRef]
- Li, J.; Xu, J.; Yan, X.; Jin, K.; Li, W.; Zhang, R. Targeting Interleukin-6 (IL-6) Sensitizes Anti-PD-L1 Treatment in a Colorectal Cancer Preclinical Model. Med. Sci. Monit. 2018, 24, 5501–5508. [Google Scholar] [CrossRef] [PubMed]
- Flint, T.R.; Fearon, D.T.; Janowitz, T. Connecting the Metabolic and Immune Responses to Cancer. Trends Mol. Med. 2017, 23, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, F.; Montfort, A.; Marcheteau, E.; Imbert, C.; Gilhodes, J.; Filleron, T.; Rochaix, P.; Andrieu-Abadie, N.; Levade, T.; Meyer, N.; et al. TNF-alpha blockade overcomes resistance to anti- PD-1 in experimental melanoma. Nat. Commun. 2017, 8, 2256. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zeng, D.; Ou, Q.; Liu, S.; Li, A.; Chen, Y.; Lin, D.; Gao, Q.; Zhou, H.; Liao, W.; et al. Association of Survival and Immune-Related Biomarkers with Immuno-Therapy in patients with non-small cell lung cancer: A meta-analysis and individual patient-level analysis. JAMA Netw. Open 2019, 2, e196879. [Google Scholar] [CrossRef]
- Macciò, A.; Madeddu, C. Inflammation and ovarian cancer. Cytokine 2012, 58, 133–147. [Google Scholar] [CrossRef]
- Tormo, A.J.; Letellier, M.; Sharma, M.; Elson, G.; Crabé, S.; Gauchat, J. IL-6 activates STAT5 in T cells. Cytokine 2012, 60, 575–582. [Google Scholar] [CrossRef]
- Wang, D.; DuBois, R.N. Immunosuppression associated with chronic inflammation in the tumor microenvironment. Carcinogenesis 2015, 36, 1085–1093. [Google Scholar] [CrossRef]
- Ostrand-Rosenberg, S. Myeloid-derived suppressor cells: More mechanisms for inhibiting antitumor immunity. Cancer Immunol. Immunother. 2010, 59, 1593–1600. [Google Scholar] [CrossRef]
- Almand, B.; Clark, J.I.; Nikitina, E.; van Beynen, B.J.; English, N.R.; Knight, S.C.; Carbone, D.P.; Gabrilovich, D.I. Increased production of immature myeloid cells in cancer patients: A mechanism of immunosuppression in cancer. J. Immunol. 2001, 166, 678–689. [Google Scholar] [CrossRef]
- Kumar, R.; Collins, D.; Dolly, S.; McDonald, F.; O’Brien, M.E.; Yap, T.A. Targeting the PD-1/PD-L1 axis in non-small cell lung cancer. Curr. Probl. Cancer 2017, 41, 111–124. [Google Scholar] [CrossRef]
- Turner, D.; Kondic, A.G.; Anderson, K.M.; Robinson, A.G.; Garon, E.B.; Riess, J.W.; Jain, L.; Mayawala, K.; Kang, J.; Ebbinghaus, S.W.; et al. Pembrolizumab exposure-response assessments challenged by association of cancer cachexia and catabolic clearance. Clin. Cancer Res. 2018, 24, 5841–5849. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.; Priyadharshini, B.; Turka, L.A. Immunometabolism of regulatory T cells. Nat. Immunol. 2016, 17, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.J.; Hammerman, P.S.; Thompson, C.B. Fuel feeds function: Energy metabolism and the T-cell response. Nat. Rev. Immunol. 2005, 5, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Danzaki, K.; MacIver, N.J. Nutritional effects on T-cell immunometabolism. Eur. J. Immunol. 2017, 47, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Buck, M.D.; Sowell, R.T.; Kaech, S.M.; Pearce, E.L. Metabolic instruction of immunity. Cell 2017, 169, 570–586. [Google Scholar] [CrossRef]
- White, J.P.; Puppa, M.J.; Gao, S.; Sato, S.; Welle, S.L.; Carson, J.A. Muscle mTORC1 suppression by IL-6 during cancer cachexia: A role for AMPK. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E1042–E1052. [Google Scholar] [CrossRef]
- Shijubou, N.; Sumi, T.; Yamada, Y.; Nakata, H.; Mori, Y.; Hirofumi Chiba, H. Immunological and nutritional predictive factors in patients receiving pembrolizumab for the first-line treatment of non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 2022, 148, 1893–1901. [Google Scholar] [CrossRef]
- Degens, J.H.R.J.; Dingemans, A.C.; Willemsen, A.C.H.; Gietema, H.A.; Hurkmans, D.P.; Aerts, J.G.; Hendriks, L.E.L.; Schols, A.M.W.J. The prognostic value of weight and body composition changes in patients with non-small-cell lung cancer treated with nivolumab. J. Cachexia Sarcopenia Muscle 2021, 12, 657–664. [Google Scholar] [CrossRef]
- Macciò, A.; Madeddu, C.; Gramignano, G.; Mulas, C.; Tanca, L.; Cherchi, M.C.; Floris, C.; Omoto, I.; Barracca, A.; Ganz, T. The role of inflammation, iron, and nutritional status in cancer-related anemia: Results of a large, prospective, observational study. Haematologica 2015, 100, 124–132. [Google Scholar] [CrossRef]
- Mantovani, G.; Macciò, A.; Lai, P.; Massa, E.; Massa, D.; Mulas, C.; Succu, G.; Mudu, M.C.; Manca, G.; Versace, R.; et al. Results of a dose-intense phase 1 study of a combination chemotherapy regimen with cisplatin and epidoxorubicin including medroxyprogesterone acetate and recombinant interleukin-2 in patients with inoperable primary lung cancer. J. Immunother. 2000, 23, 267–274. [Google Scholar] [CrossRef]
| Parameters | Mean ± SD (Range) | No. (%) |
|---|---|---|
| Enrolled patients | 74 (100) | |
| Males/females | 54/20 (73/27) | |
| Age (years) | 69.3 ± 11.3 (47–88) | |
| Weight (kg): | 68.9 ± 12.4 (46–95) | |
| Height (cm): | 164.1 ± 8.7 (151–185) | |
| Stage | ||
| IV | 74 (100) | |
| Histology | ||
| Adenocarcinoma | 57 (77) | |
| Squamocellular | 17 (23) | |
| PD-L1 expression | ||
| <1% | 10 (13.5) | |
| 1–50% | 22 (29.8) | |
| >50% | 32 (43.2) | |
| N.V. | 10 (13.5) | |
| ECOG–PS | ||
| 0 | 10 (13.5) | |
| 1 | 39 (52.7) | |
| 2 | 25 (33.8) | |
| Treatment | ||
| Nivolumab | 16 (43.2) | |
| Pembrolizumab | 21 (56.8) | |
| Previous line | 32 (43) |
| Parameters | Regression Coefficient | OR | 95%CI | p-Value |
|---|---|---|---|---|
| Baseline weigh loss % | −0.0346 | 0.966 | 0.8588–1.0866 | 0.5646 |
| Cachexia according to the standard criteria [24] | −0.7885 | 0.4545 | 0.1602–1.2893 | 0.1383 |
| LBM | −0.0158 | 0.9843 | 0.9326–1.0389 | 0.5658 |
| SMI | 0.0129 | 0.9871 | 0.9493–1.0265 | 0.5154 |
| C-reactive protein | −0.672 | 0.9971 | 0.8164–0.9848 | 0.0227 |
| IL-6 | −0.0799 | 0.9232 | 0.8762–0.9728 | 0.0027 |
| Neutrophil count | −0.00022 | 0.9998 | 0.9996–1.0000 | 0.0403 |
| Lymphocyte count | −0.008 | 0.9992 | 0.9983–1.0001 | 0.0757 |
| NLR | −0.0819 | 0.9213 | 0.7460–1.1378 | 0.4467 |
| mGPS | −0.4 | 0.6701 | 0.3906–1.1498 | 0.1462 |
| Albumin | −0.2878 | 0.7499 | 0.2340–2.4034 | 0.6282 |
| Hemoglobin | 0.1545 | 1.1671 | 0.8953–1.5214 | 0.2533 |
| MiniCASCO score | −0.1651 | 0.9035 | 0.6766–0.9845 | 0.0186 |
| Anorexia subscale | 0.1607 | 1.1743 | 1.0056–1.3713 | 0.0423 |
| IMD subscale | −0.0466 | 0.9545 | 0.8565–0.9988 | 0.0469 |
| BWC subscale | −0.0187 | 0.9814 | 0.9318–1.0337 | 0.4789 |
| PHP subscale | −0.0164 | 0.9659 | 0.8847–1.002 | 0.2228 |
| QoL subscale | −0.1015 | 0.9035 | 0.6766–1.2065 | 0.4917 |
| Cachexia severity | −0.1511 | 0.8598 | 0.4957–1.4914 | 0.5908 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madeddu, C.; Busquets, S.; Donisi, C.; Lai, E.; Pretta, A.; López-Soriano, F.J.; Argilés, J.M.; Scartozzi, M.; Macciò, A. Effect of Cancer-Related Cachexia and Associated Changes in Nutritional Status, Inflammatory Status, and Muscle Mass on Immunotherapy Efficacy and Survival in Patients with Advanced Non-Small Cell Lung Cancer. Cancers 2023, 15, 1076. https://doi.org/10.3390/cancers15041076
Madeddu C, Busquets S, Donisi C, Lai E, Pretta A, López-Soriano FJ, Argilés JM, Scartozzi M, Macciò A. Effect of Cancer-Related Cachexia and Associated Changes in Nutritional Status, Inflammatory Status, and Muscle Mass on Immunotherapy Efficacy and Survival in Patients with Advanced Non-Small Cell Lung Cancer. Cancers. 2023; 15(4):1076. https://doi.org/10.3390/cancers15041076
Chicago/Turabian StyleMadeddu, Clelia, Silvia Busquets, Clelia Donisi, Eleonora Lai, Andrea Pretta, Francisco Javier López-Soriano, Josep Maria Argilés, Mario Scartozzi, and Antonio Macciò. 2023. "Effect of Cancer-Related Cachexia and Associated Changes in Nutritional Status, Inflammatory Status, and Muscle Mass on Immunotherapy Efficacy and Survival in Patients with Advanced Non-Small Cell Lung Cancer" Cancers 15, no. 4: 1076. https://doi.org/10.3390/cancers15041076
APA StyleMadeddu, C., Busquets, S., Donisi, C., Lai, E., Pretta, A., López-Soriano, F. J., Argilés, J. M., Scartozzi, M., & Macciò, A. (2023). Effect of Cancer-Related Cachexia and Associated Changes in Nutritional Status, Inflammatory Status, and Muscle Mass on Immunotherapy Efficacy and Survival in Patients with Advanced Non-Small Cell Lung Cancer. Cancers, 15(4), 1076. https://doi.org/10.3390/cancers15041076









