The Diagnostic and Therapeutic Role of snoRNA and lincRNA in Bladder Cancer
Abstract
Simple Summary
Abstract
1. Introduction
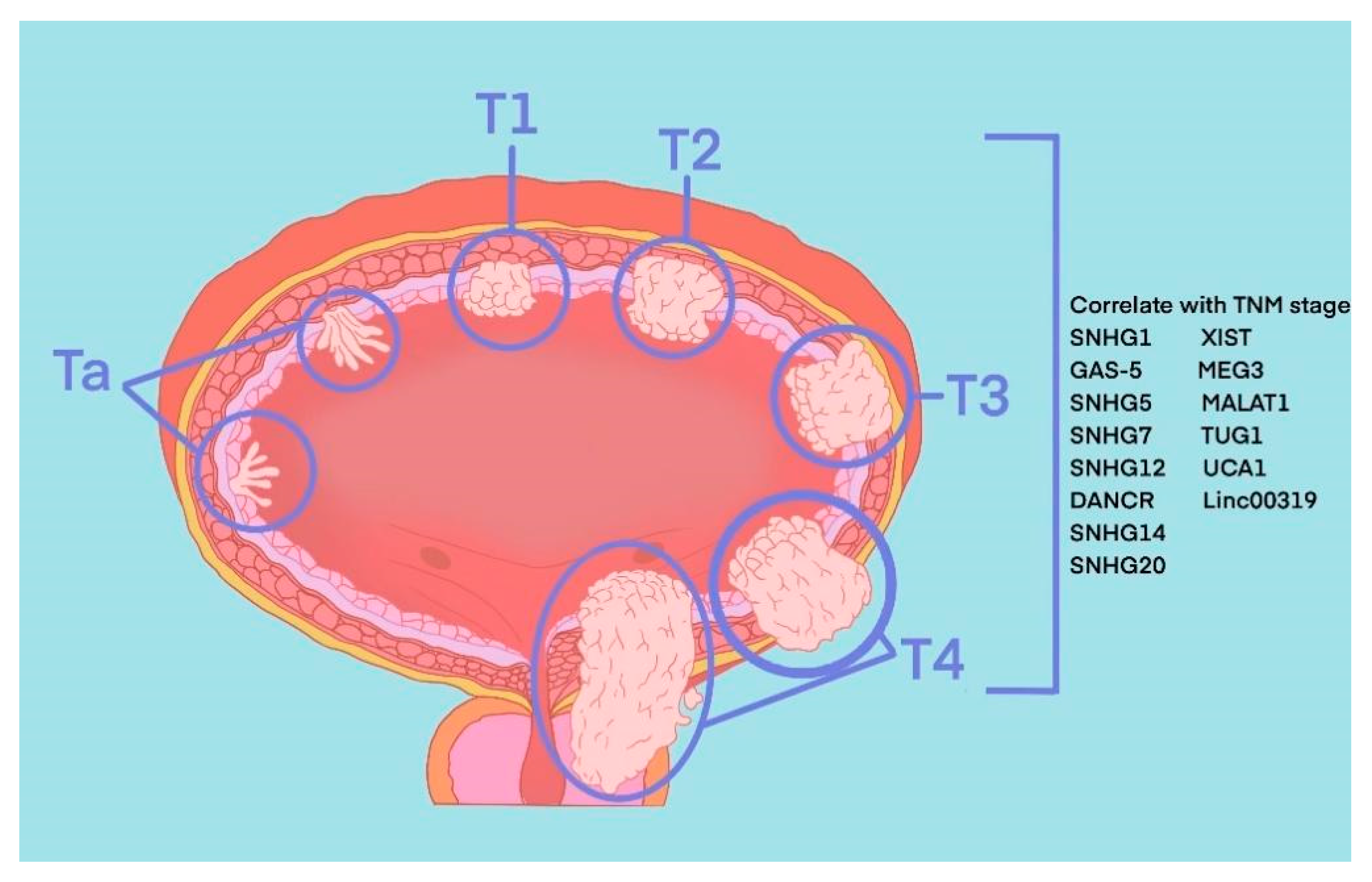
2. Pathophysiology of BLCA
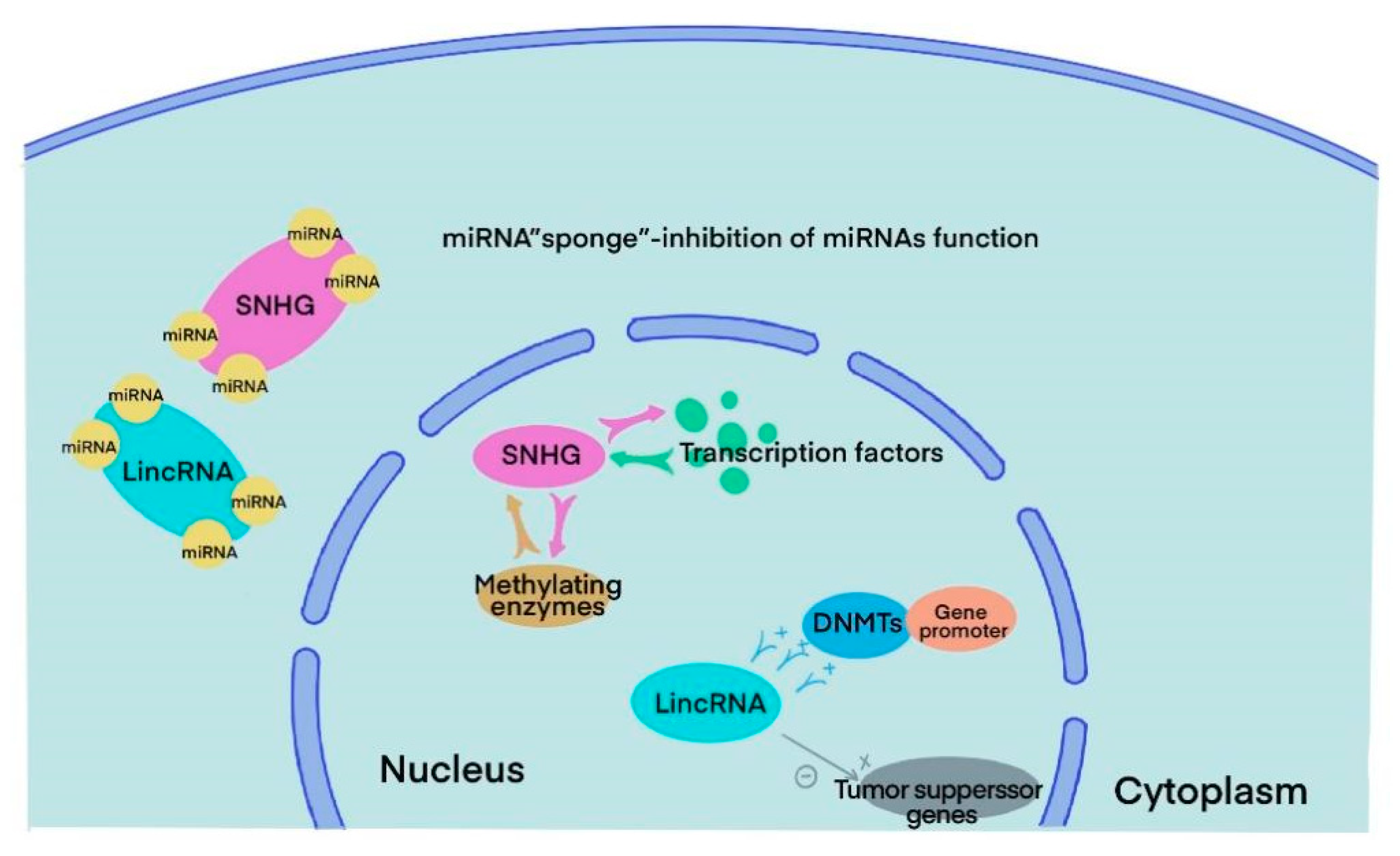
3. SnoRNA and lincRNA
4. The Role of snoRNA in BLCA
| Name | Expression | Related miRNAs, Proteins or Pathways | Related Tumour Cell Biology | Relevant Clinical Features | Reference |
|---|---|---|---|---|---|
| SNHG1 | Upregulated | miR-34a, miR-129-2-5p, miR-143-3p, miR-137-3p, miR-9-3p | Proliferation, migration, invasion, apoptosis, autophagy, EMT and stemness | TNM stage, LN invasion, metastasis, recurrence-free survival and prognosis | [21,22,23,24,25] |
| SNHG2 (GAS-5) | Downregulated | miR-101, miR-21, CDK6 | Proliferation, migration, apoptosis and doxorubicin resistance | Age, tumour size, tumour stage, LN invasion and prognosis | [26,27,28,29] |
| SNHG3 | Upregulated | miR-515-5p, c-MYC | Proliferation, migration, invasion, EMT and angiogenesis | TNM stage and prognosis | [30,31,32] |
| SNHG5 | Upregulated | p27 | Migration and apoptosis | Stage, tumour size, LN invasion, metastasis and prognosis | [35] |
| SNHG6 | Upregulated | hsa-miR-125b | Invasion and migration | - | [34] |
| SNHG7 | Upregulated | miR-2682-5p, Wnt/β-catenin | Proliferation, invasion and migration | Stage, tumour size, LN invasion and prognosis | [37,38,39] |
| SNHG12 | Upregulated | - | Proliferation | - | [44] |
| SNHG13 (DANCR) | Upregulated | miR-335, miR-149, IL-11 | Proliferation, invasion, migration and lymphatic metastasis | TNM stage, histological grade, LN invasion and prognosis | [41,42,43] |
| SNHG14 | Upregulated | miR-150-5p, miR-211-3p | Proliferation, invasion, migration and apoptosis | TNM stage, LN invasion and prognosis | [45,46] |
| SNHG15 | Upregulated | - | Proliferation and invasion | - | [47] |
| SNHG16 | Upregulated | EMT | Invasion and migration | - | [48] |
| SNHG20 | Upregulated | Wnt/β-catenin | Proliferation, invasion, migration and apoptosis | Stage, LN invasion and prognosis | [37] |
5. The Role of lincRNA in BLCA
| Name | Expression | Related miRNAs, Proteins or Pathways | Related Tumour Cell Biology | Relevant Clinical Features | Reference |
|---|---|---|---|---|---|
| Linc00001 (XIST) | Upregulated | miR-200c, miR-355, miR-133a, TET1 | Proliferation, migration, invasion, apoptosis and EMT | Tumour stage, metastasis, and prognosis | [51,52,53,54] |
| Linc00023 (MEG3) | Downregulated | miR-96, miR-27a, miR-494, p53 | Proliferation, migration, invasion, apoptosis and chemosensitivity | Tumour stage, invasion and metastasis | [57,58,59,60] |
| Linc00047 (MALAT1) | Upregulated | miR-384, miR-124, miR-101-3p | Proliferation, migration, invasion and chemosensitivity | Tumour stage, invasion and prognosis | [63,64,65] |
| Linc00080 (TUG1) | Upregulated | miR-140-3p, miR-194-5p, miR-142, miR-145, HMGB1 | Proliferation, migration, invasion, apoptosis, EMT, radiosensitivity and chemosensitivity | Tumour stage, invasion, LN metastasis and OS | [67,68,69,70,71] |
| Linc00178 (UCA1) | Upregulated | miR-195, miR-582-5p, miR-143 | Proliferation, migration, invasion, autophagy, EMT, mitochondrial function and drug resistance | Tumour stage | [73,74,75,76] |
| Linc00319 | Upregulated | miR-3127, miR-4492 | Proliferation, migration and invasion | Tumour stage and RFS | [81,82] |
| Linc00355 | Upregulated | miR-424-5p, miR-34b-5p, miR-15a-5p | Proliferation, migration, invasion, EMT and cisplatin resistance | Prognosis | [86,88,89] |
| Linc00641 | Downregulated | miR-197-3p | Proliferation, migration and invasion | Prognosis | [93] |
| Linc00649 | Upregulated | miR-15a-5p, miR-16-5p | Proliferation, migration and invasion | Muscle invasion and OS | [98,99] |
| Linc00958 | Upregulated | miR-490-3p, miR-625-5p, miR-378a-3p, MTDH, SAPK | Proliferation, migration, invasion, apoptosis, autophagy and angiogenesis | OS | [101,102,103,104,105] |
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Nogueira, L.; Devasia, T.; Mariotto, A.B.; Yabroff, K.R.; Jemal, A.; Kramer, J.; Siegel, R.L. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 2022, 72, 409–436. [Google Scholar] [CrossRef] [PubMed]
- Compérat, E.; Amin, M.B.; Cathomas, R.; Choudhury, A.; De Santis, M.; Kamat, A.; Stenzl, A.; Thoeny, H.C.; Witjes, J.A. Current best practice for bladder cancer: A narrative review of diagnostics and treatments. Lancet 2022, 400, 1712–1721. [Google Scholar] [CrossRef] [PubMed]
- Lenis, A.T.; Lec, P.M.; Chamie, K.; Mshs, M.D. Bladder Cancer: A Review. JAMA 2020, 324, 1980–1991. [Google Scholar] [CrossRef]
- Tran, L.; Xiao, J.-F.; Agarwal, N.; Duex, J.E.; Theodorescu, D. Advances in bladder cancer biology and therapy. Nat. Rev. Cancer 2021, 21, 104–121. [Google Scholar] [CrossRef]
- Kopp, F.; Mendell, J.T. Functional Classification and Experimental Dissection of Long Noncoding RNAs. Cell 2018, 172, 393–407. [Google Scholar] [CrossRef]
- Palazzo, A.F.; Koonin, E.V. Functional Long Non-coding RNAs Evolve from Junk Transcripts. Cell 2020, 183, 1151–1161. [Google Scholar] [CrossRef]
- Bridges, M.C.; Daulagala, A.C.; Kourtidis, A. LNCcation: LncRNA localization and function. J. Cell Biol. 2021, 220, e20200904. [Google Scholar] [CrossRef]
- Dsouza, V.L.; Adiga, D.; Sriharikrishnaa, S.; Suresh, P.S.; Chatterjee, A.; Kabekkodu, S.P. Small nucleolar RNA and its potential role in breast cancer—A comprehensive review. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188501. [Google Scholar] [CrossRef]
- Williams, G.T.; Farzaneh, F. Are snoRNAs and snoRNA host genes new players in cancer? Nat. Rev. Cancer 2012, 12, 84–88. [Google Scholar] [CrossRef]
- Ding, Y.; Sun, Z.; Zhang, S.; Li, Y.; Han, X.; Xu, Q.; Zhou, L.; Xu, H.; Bai, Y.; Xu, C.; et al. Downregulation of snoRNA SNORA52 and Its Clinical Significance in Hepatocellular Carcinoma. Biomed. Res. Int. 2021, 2021, 7020637. [Google Scholar] [CrossRef]
- Bao, H.-J.; Chen, X.; Liu, X.; Wu, W.; Li, Q.-H.; Xian, J.-Y.; Zhao, Y.; Chen, S. Box C/D snoRNA SNORD89 influences the occurrence and development of endometrial cancer through 2′-O-methylation modification of Bim. Cell Death Discov. 2022, 8, 309. [Google Scholar] [CrossRef]
- Zimta, A.-A.; Tigu, A.B.; Braicu, C.; Stefan, C.; Ionescu, C.; Berindan-Neagoe, I. An Emerging Class of Long Non-coding RNA With Oncogenic Role Arises From the snoRNA Host Genes. Front. Oncol. 2020, 10, 389. [Google Scholar] [CrossRef]
- Rinn, J.L.; Chang, H.Y. Long Noncoding RNAs: Molecular Modalities to Organismal Functions. Annu. Rev. Biochem. 2020, 89, 283–308. [Google Scholar] [CrossRef]
- Martens-Uzunova, E.S.; Böttcher, R.; Croce, C.M.; Jenster, G.; Visakorpi, T.; Calin, G.A. Long noncoding RNA in prostate, bladder, and kidney cancer. Eur. Urol. 2014, 65, 1140–1151. [Google Scholar] [CrossRef]
- Cabili, M.N.; Trapnell, C.; Goff, L.; Koziol, M.; Tazon-Vega, B.; Regev, A.; Rinn, J.L. Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev. 2011, 25, 1915–1927. [Google Scholar] [CrossRef]
- Pasieka, R.; Zasoński, G.; Raczyńska, K.D. Role of Long Intergenic Noncoding RNAs in Cancers with an Overview of MicroRNA Binding. Mol. Diagn. Ther. 2022, 27, 29–47. [Google Scholar] [CrossRef]
- Reichow, S.L.; Hamma, T.; Ferré-D'Amaré, A.R.; Varani, G. The structure and function of small nucleolar ribonucleoproteins. Nucleic Acids Res. 2007, 35, 1452–1464. [Google Scholar] [CrossRef] [PubMed]
- Matera, A.G.; Terns, R.M.; Terns, M.P. Non-coding RNAs: Lessons from the small nuclear and small nucleolar RNAs. Nat. Rev. Mol. Cell Biol. 2007, 8, 209–220. [Google Scholar] [CrossRef]
- Ransohoff, J.D.; Wei, Y.; Khavari, P.A. The functions and unique features of long intergenic non-coding RNA. Nat. Rev. Mol. Cell Biol. 2018, 19, 143–157. [Google Scholar] [CrossRef]
- Xu, J.; Yang, R.; Hua, X.; Huang, M.; Tian, Z.; Li, J.; Lam, H.Y.; Jiang, G.; Cohen, M.; Huang, C. lncRNA SNHG1 Promotes Basal Bladder Cancer Invasion via Interaction with PP2A Catalytic Subunit and Induction of Autophagy. Mol. Ther. Nucleic Acids 2020, 21, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yang, R.; Li, J.; Wang, L.; Cohen, M.; Simeone, D.M.; Costa, M.; Wu, X.-R. DNMT3A//Rac1 Is an Effector Pathway for to Drive Stem-Cell-like and Invasive Behaviors of Advanced Bladder Cancer Cells. Cancers 2022, 14, 4159. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.; Lyu, L.; Huang, T.; Zheng, F.; Yuan, J.; Zhang, C.; Jiang, G. The long non-coding RNA SNHG1 promotes bladder cancer progression by interacting with miR-143-3p and EZH2. J. Cell. Mol. Med. 2020, 24, 11858–11873. [Google Scholar] [CrossRef]
- Min, J.; Ma, J.; Wang, Q.; Yu, D. Long non-coding RNA SNHG1 promotes bladder cancer progression by upregulating EZH2 and repressing KLF2 transcription. Clinics 2022, 77, 100081. [Google Scholar] [CrossRef]
- Cai, H.; Xu, H.; Lu, H.; Xu, W.; Liu, H.; Wang, X.; Zhou, G.; Yang, X. LncRNA SNHG1 Facilitates Tumor Proliferation and Represses Apoptosis by Regulating PPARγ Ubiquitination in Bladder Cancer. Cancers 2022, 14, 4740. [Google Scholar] [CrossRef]
- Wang, M.; Guo, C.; Wang, L.; Luo, G.; Huang, C.; Li, Y.; Liu, D.; Zeng, F.; Jiang, G.; Xiao, X. Long noncoding RNA GAS5 promotes bladder cancer cells apoptosis through inhibiting EZH2 transcription. Cell Death Dis. 2018, 9, 238. [Google Scholar] [CrossRef]
- Chen, D.; Guo, Y.; Chen, Y.; Guo, Q.; Chen, J.; Li, Y.; Zheng, Q.; Jiang, M.; Xi, M.; Cheng, L. LncRNA growth arrest-specific transcript 5 targets miR-21 gene and regulates bladder cancer cell proliferation and apoptosis through PTEN. Cancer Med. 2020, 9, 2846–2858. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, W.; Jiang, J.; Bao, E.; Xu, D.; Zeng, Y.; Tao, L.; Qiu, J. Downregulation of GAS5 promotes bladder cancer cell proliferation, partly by regulating CDK6. PLoS ONE 2013, 8, e73991. [Google Scholar] [CrossRef]
- Zhang, H.; Guo, Y.; Song, Y.; Shang, C. Long noncoding RNA GAS5 inhibits malignant proliferation and chemotherapy resistance to doxorubicin in bladder transitional cell carcinoma. Cancer Chemother. Pharmacol. 2017, 79, 49–55. [Google Scholar] [CrossRef]
- Cao, Y.; Hu, Q.; Zhang, R.; Li, L.; Guo, M.; Wei, H.; Zhang, L.; Wang, J.; Li, C. Knockdown of Long Non-coding RNA SNGH3 by CRISPR-dCas9 Inhibits the Progression of Bladder Cancer. Front. Mol. Biosci. 2021, 8, 657145. [Google Scholar] [CrossRef]
- Xie, J.; Ni, J.; Shi, H.; Wang, K.; Ma, X.; Li, W.; Peng, B. LncRNA SNHG3 enhances BMI1 mRNA stability by binding and regulating c-MYC: Implications for the carcinogenic role of SNHG3 in bladder cancer. Cancer Med. 2022. [Google Scholar] [CrossRef] [PubMed]
- Dai, G.; Huang, C.; Yang, J.; Jin, L.; Fu, K.; Yuan, F.; Zhu, J.; Xue, B. LncRNA SNHG3 promotes bladder cancer proliferation and metastasis through miR-515-5p/GINS2 axis. J. Cell. Mol. Med. 2020, 24, 9231–9243. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-H.; Hu, Y.-Q.; Wang, S.-C.; Li, Y.; Chen, D.-M. LncRNA SNHG5: A new budding star in human cancers. Gene 2020, 749, 144724. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Tao, W.; Ni, S.; Chen, Q. Upregulation of lncRNA snoRNA host gene 6 regulates NUAK family SnF1-like kinase-1 expression by competitively binding microRNA-125b and interacting with Snail1/2 in bladder cancer. J. Cell. Biochem. 2019, 120, 357–367. [Google Scholar] [CrossRef]
- Ma, Z.; Xue, S.; Zeng, B.; Qiu, D. lncRNA is associated with poor prognosis of bladder cancer and promotes bladder cancer cell proliferation through targeting. Oncol. Lett. 2018, 15, 1924–1930. [Google Scholar] [CrossRef]
- Zhou, Y.; Tian, B.; Tang, J.; Wu, J.; Wang, H.; Wu, Z.; Li, X.; Yang, D.; Zhang, B.; Xiao, Y.; et al. SNHG7: A novel vital oncogenic lncRNA in human cancers. Biomed. Pharmacother. 2020, 124, 109921. [Google Scholar] [CrossRef]
- Zhao, Q.; Gao, S.; Du, Q.; Liu, Y. Long non-coding RNA SNHG20 promotes bladder cancer via activating the Wnt/β-catenin signalling pathway. Int. J. Mol. Med. 2018, 42, 2839–2848. [Google Scholar] [CrossRef]
- Wang, W.; Chen, S.; Song, X.; Gui, J.; Li, Y.; Li, M. ELK1/lncRNA-SNHG7/miR-2682-5p feedback loop enhances bladder cancer cell growth. Life Sci. 2020, 262, 118386. [Google Scholar] [CrossRef]
- Chen, Y.; Peng, Y.; Xu, Z.; Ge, B.; Xiang, X.; Zhang, T.; Gao, L.; Shi, H.; Wang, C.; Huang, J. Knockdown of lncRNA SNHG7 inhibited cell proliferation and migration in bladder cancer through activating Wnt/β-catenin pathway. Pathol. Res. Pract. 2019, 215, 302–307. [Google Scholar] [CrossRef]
- Kretz, M.; Webster, D.E.; Flockhart, R.J.; Lee, C.S.; Zehnder, A.; Lopez-Pajares, V.; Qu, K.; Zheng, G.X.Y.; Chow, J.; Kim, G.E.; et al. Suppression of progenitor differentiation requires the long noncoding RNA ANCR. Genes Dev. 2012, 26, 338–343. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, X.; Xie, R.; Huang, M.; Dong, W.; Han, J.; Zhang, J.; Zhou, Q.; Li, H.; Huang, J.; et al. DANCR Promotes Metastasis and Proliferation in Bladder Cancer Cells by Enhancing IL-11-STAT3 Signaling and CCND1 Expression. Mol. Ther. 2019, 27, 326–341. [Google Scholar] [CrossRef]
- Ping, Q.; Shi, Y.; Yang, M.; Li, H.; Zhong, Y.; Li, J.; Bi, X.; Wang, C. LncRNA DANCR regulates lymphatic metastasis of bladder cancer via the miR-335/VEGF-C axis. Transl. Androl. Urol. 2021, 10, 1743–1753. [Google Scholar] [CrossRef]
- Zhan, Y.; Chen, Z.; Li, Y.; He, A.; He, S.; Gong, Y.; Li, X.; Zhou, L. Long non-coding RNA DANCR promotes malignant phenotypes of bladder cancer cells by modulating the miR-149/MSI2 axis as a ceRNA. J. Exp. Clin. Cancer Res. 2018, 37, 273. [Google Scholar] [CrossRef]
- Jiang, B.; Hailong, S.; Yuan, J.; Zhao, H.; Xia, W.; Zha, Z.; Bin, W.; Liu, Z. Identification of oncogenic long noncoding RNA SNHG12 and DUXAP8 in human bladder cancer through a comprehensive profiling analysis. Biomed. Pharmacother. 2018, 108, 500–507. [Google Scholar] [CrossRef]
- Li, J.; Wang, A.S.; Wang, S.; Wang, C.Y.; Xue, S.; Guan, H.; Li, W.Y.; Ma, T.T.; Shan, Y.X. LncSNHG14 promotes the development and progression of bladder cancer by targeting miRNA-150-5p. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1022–1029. [Google Scholar]
- Feng, R.; Li, Z.; Wang, X.; Ge, G.; Jia, Y.; Wu, D.; Ji, Y.; Wang, C. Silenced lncRNA SNHG14 restrains the biological behaviors of bladder cancer cells via regulating microRNA-211-3p/ESM1 axis. Cancer Cell Int. 2021, 21, 67. [Google Scholar] [CrossRef]
- Mokhtar, A.; Kong, C.; Zhang, Z.; Du, Y. Down-regulation LncRNA-SNHG15 contributes to proliferation and invasion of bladder cancer cells. BMC Urol. 2021, 21, 83. [Google Scholar] [CrossRef]
- Chen, W.; Jiang, T.; Mao, H.; Gao, R.; Zhang, H.; He, Y.; Liu, C.; Chen, Q. SNHG16 regulates invasion and migration of bladder cancer through induction of epithelial-to-mesenchymal transition. Hum. Cell 2020, 33, 737–749. [Google Scholar] [CrossRef]
- Cao, X.; Xu, J.; Yue, D. LncRNA-SNHG16 predicts poor prognosis and promotes tumor proliferation through epigenetically silencing p21 in bladder cancer. Cancer Gene Ther. 2018, 25, 10–17. [Google Scholar] [CrossRef]
- Wang, W.; Min, L.; Qiu, X.; Wu, X.; Liu, C.; Ma, J.; Zhang, D.; Zhu, L. Biological Function of Long Non-coding RNA (LncRNA) Xist. Front. Cell Dev. Biol. 2021, 9, 645647. [Google Scholar] [CrossRef]
- Chen, D.; Chen, T.; Guo, Y.; Wang, C.; Dong, L.; Lu, C. Platycodin D (PD) regulates LncRNA-XIST/miR-335 axis to slow down bladder cancer progression in vitro and in vivo. Exp. Cell Res. 2020, 396, 112281. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Shi, G.; Li, Q.; Li, W.; Zhou, H. Long noncoding RNA XIST participates in bladder cancer by downregulating p53 via binding to TET1. J. Cell. Biochem. 2019, 120, 6330–6338. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Zhu, X.; Chen, F.; Huang, C.; Ai, K.; Wu, H.; Zhang, L.; Zhao, X. LncRNA XIST/miR-200c regulates the stemness properties and tumourigenicity of human bladder cancer stem cell-like cells. Cancer Cell Int. 2018, 18, 41. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Yang, J.; Li, X.; Chen, W. Long non-coding RNA XIST promotes cell proliferation and migration through targeting miR-133a in bladder cancer. Exp. Ther. Med. 2019, 18, 3475–3483. [Google Scholar] [CrossRef]
- Al-Rugeebah, A.; Alanazi, M.; Parine, N.R. MEG3: An Oncogenic Long Non-coding RNA in Different Cancers. Pathol. Oncol. Res. 2019, 25, 859–874. [Google Scholar] [CrossRef]
- Hsieh, P.-F.; Yu, C.-C.; Chu, P.-M.; Hsieh, P.-L. Long Non-Coding RNA MEG3 in Cellular Stemness. Int. J. Mol. Sci. 2021, 22, 5348. [Google Scholar] [CrossRef]
- Shan, G.; Tang, T.; Xia, Y.; Qian, H.-J. MEG3 interacted with miR-494 to repress bladder cancer progression through targeting PTEN. J. Cell. Physiol. 2020, 235, 1120–1128. [Google Scholar] [CrossRef]
- Liu, G.; Zhao, X.; Zhou, J.; Cheng, X.; Ye, Z.; Ji, Z. Long non-coding RNA MEG3 suppresses the development of bladder urothelial carcinoma by regulating miR-96 and TPM1. Cancer Biol. Ther. 2018, 19, 1039–1056. [Google Scholar] [CrossRef]
- Feng, S.Q.; Zhang, X.Y.; Fan, H.T.; Sun, Q.J.; Zhang, M. Up-regulation of LncRNA MEG3 inhibits cell migration and invasion and enhances cisplatin chemosensitivity in bladder cancer cells. Neoplasma 2018, 65, 925–932. [Google Scholar] [CrossRef]
- Huang, C.; Liao, X.; Jin, H.; Xie, F.; Zheng, F.; Li, J.; Zhou, C.; Jiang, G.; Wu, X.-R.; Huang, C. MEG3, as a Competing Endogenous RNA, Binds with miR-27a to Promote PHLPP2 Protein Translation and Impairs Bladder Cancer Invasion. Mol. Ther. Nucleic Acids 2019, 16, 51–62. [Google Scholar] [CrossRef]
- Li, Z.-X.; Zhu, Q.-N.; Zhang, H.-B.; Hu, Y.; Wang, G.; Zhu, Y.-S. MALAT1: A potential biomarker in cancer. Cancer Manag. Res. 2018, 10, 6757–6768. [Google Scholar] [CrossRef]
- Goyal, B.; Yadav, S.R.M.; Awasthee, N.; Gupta, S.; Kunnumakkara, A.B.; Gupta, S.C. Diagnostic, prognostic, and therapeutic significance of long non-coding RNA MALAT1 in cancer. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188502. [Google Scholar] [CrossRef]
- Jiao, D.; Li, Z.; Zhu, M.; Wang, Y.; Wu, G.; Han, X. LncRNA MALAT1 promotes tumor growth and metastasis by targeting miR-124/foxq1 in bladder transitional cell carcinoma (BTCC). Am. J. Cancer Res. 2018, 8, 748–760. [Google Scholar]
- Liu, P.; Li, X.; Cui, Y.; Chen, J.; Li, C.; Li, Q.; Li, H.; Zhang, X.; Zu, X. LncRNA-MALAT1 mediates cisplatin resistance via miR-101-3p/VEGF-C pathway in bladder cancer. Acta Biochim. Biophys. Sin. 2019, 51, 1148–1157. [Google Scholar] [CrossRef]
- Tao, L.; Mu, X.; Chen, H.; Jin, D.; Zhang, R.; Zhao, Y.; Fan, J.; Cao, M.; Zhou, Z. FTO modifies the m6A level of MALAT and promotes bladder cancer progression. Clin. Transl. Med. 2021, 11, e310. [Google Scholar] [CrossRef]
- Li, Z.; Shen, J.; Chan, M.T.V.; Wu, W.K.K. TUG1: A pivotal oncogenic long non-coding RNA of human cancers. Cell Prolif. 2016, 49, 471–475. [Google Scholar] [CrossRef]
- Tan, J.; Qiu, K.; Li, M.; Liang, Y. Double-negative feedback loop between long non-coding RNA TUG1 and miR-145 promotes epithelial to mesenchymal transition and radioresistance in human bladder cancer cells. FEBS Lett. 2015, 589 Pt B, 3175–3181. [Google Scholar] [CrossRef]
- Jiang, H.; Hu, X.; Zhang, H.; Li, W. Down-regulation of LncRNA TUG1 enhances radiosensitivity in bladder cancer via suppressing HMGB1 expression. Radiat. Oncol. 2017, 12, 65. [Google Scholar] [CrossRef]
- Yuan, J.-B.; Gu, L.; Chen, L.; Yin, Y.; Fan, B.-Y. Annexin A8 regulated by lncRNA-TUG1/miR-140-3p axis promotes bladder cancer progression and metastasis. Mol. Ther. Oncolytics 2021, 22, 36–51. [Google Scholar] [CrossRef]
- Yu, G.; Zhou, H.; Yao, W.; Meng, L.; Lang, B. lncRNA TUG1 Promotes Cisplatin Resistance by Regulating CCND2 via Epigenetically Silencing miR-194-5p in Bladder Cancer. Mol. Ther. Nucleic Acids 2019, 16, 257–271. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, H.; Cheng, H.; Li, Y.; Li, X.; Zhu, C. Downregulation of long noncoding RNA TUG1 inhibits proliferation and induces apoptosis through the TUG1/miR-142/ZEB2 axis in bladder cancer cells. Onco Targets Ther. 2017, 10, 2461–2471. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Fard, S.; Taheri, M. UCA1 long non-coding RNA: An update on its roles in malignant behavior of cancers. Biomed. Pharmacother. 2019, 120, 109459. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-J.; Sun, X.-M.; Li, Z.-K.; Yin, Q.-W.; Pang, H.; Pan, J.-J.; Li, X.; Chen, W. LncRNA UCA1 Promotes Mitochondrial Function of Bladder Cancer via the MiR-195/ARL2 Signaling Pathway. Cell Physiol. Biochem. 2017, 43, 2548–2561. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, W.; Ning, J.; Yu, W.; Rao, T.; Cheng, F. Long noncoding RNA UCA1 targets miR-582-5p and contributes to the progression and drug resistance of bladder cancer cells through ATG7-mediated autophagy inhibition. Onco Targets Ther. 2019, 12, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Chen, J.; Li, H.; Yang, Y.; Yun, H.; Yang, S.; Mao, X. LncRNA UCA1 promotes the invasion and EMT of bladder cancer cells by regulating the miR-143/HMGB1 pathway. Oncol. Lett. 2017, 14, 5556–5562. [Google Scholar] [CrossRef]
- Xue, M.; Chen, W.; Xiang, A.; Wang, R.; Chen, H.; Pan, J.; Pang, H.; An, H.; Wang, X.; Hou, H.; et al. Hypoxic exosomes facilitate bladder tumor growth and development through transferring long non-coding RNA-UCA1. Mol. Cancer 2017, 16, 143. [Google Scholar] [CrossRef]
- Sun, F.; Yu, Z.; Wu, B.; Zhang, H.; Ruan, J. LINC00319 promotes osteosarcoma progression by regulating the miR-455-3p/NFIB axis. J. Gene Med. 2020, 22, e3248. [Google Scholar] [CrossRef]
- Ma, Z.; Cai, Y.; Zhang, L.; Tian, C.; Lyu, L. LINC00319 Promotes Cervical Cancer Progression Via Targeting miR-147a/IGF1R Pathway. Cancer Biother. Radiopharm. 2020. [Google Scholar] [CrossRef]
- Zou, J.; Wu, K.; Lin, C.; Jie, Z.-G. LINC00319 acts as a sponge to accelerate tumor growth and metastasis in gastric cancer by upregulating. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 318, G10–G22. [Google Scholar] [CrossRef]
- Yuan, L.; Tian, X.; Zhang, Y.; Huang, X.; Li, Q.; Li, W.; Li, S. LINC00319 promotes cancer stem cell-like properties in laryngeal squamous cell carcinoma via E2F1-mediated upregulation of HMGB3. Exp. Mol. Med. 2021, 53, 1218–1228. [Google Scholar] [CrossRef]
- Wang, X.; Meng, R.; Hu, Q.-M. LINC00319-Mediated miR-3127 Repression Enhances Bladder Cancer Progression Through Upregulation of RAP2A. Front. Genet. 2020, 11, 180. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, F.; Huang, H.; Xie, Z.; Huang, W.; Xie, H.; Wang, F. Long noncoding RNA LINC00319 regulates ROMO1 expression and promotes bladder cancer progression via miR-4492/ROMO1 axis. J. Cell. Physiol. 2020, 235, 3768–3775. [Google Scholar] [CrossRef]
- Luo, X.; Abudureyimu, M.; Yang, G.; Yan, Z.; Fu, X.; Lu, P.; Zhang, D.; Zhang, S.; Ding, Z. LINC00355 triggers malignant progression of hepatocellular carcinoma via the sponge effect on miR-217-5p with the involvement of the Wnt/β-catenin signaling. J. BUON 2021, 26, 1964–1969. [Google Scholar]
- Qi, Z.-Y.; Wang, L.-L.; Qu, X.-L. lncRNA LINC00355 Acts as a Novel Biomarker and Promotes Glioma Biological Activities via the Regulation of miR-1225/FNDC3B. Dis. Markers 2021, 2021, 1683129. [Google Scholar] [CrossRef]
- Sun, X.; Wang, G.; Ding, P.; Li, S. LINC00355 promoted the progression of lung squamous cell carcinoma through regulating the miR-466/LYAR axis. Braz. J. Med. Biol. Res. 2020, 53, e9317. [Google Scholar] [CrossRef]
- Li, W.-J.; Li, G.; Liu, Z.-W.; Chen, Z.-Y.; Pu, R. LncRNA LINC00355 promotes EMT and metastasis of bladder cancer cells through the miR-424-5p/HMGA2 axis. Neoplasma 2021, 68, 1225–1235. [Google Scholar] [CrossRef]
- Yan, L.; Wang, P.; Fang, W.; Liang, C. Cancer-associated fibroblasts-derived exosomes-mediated transfer of LINC00355 regulates bladder cancer cell proliferation and invasion. Cell Biochem. Funct. 2020, 38, 257–265. [Google Scholar] [CrossRef]
- Luo, G.; Zhang, Y.; Wu, Z.; Zhang, L.; Liang, C.; Chen, X. Exosomal LINC00355 derived from cancer-associated fibroblasts promotes bladder cancer cell resistance to cisplatin by regulating miR-34b-5p/ABCB1 axis. Acta Biochim. Biophys. Sin. 2021, 53, 558–566. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, G.; You, S.; Zhang, L.; Liang, C.; Chen, X. Exosomal LINC00355 derived from cancer-associated fibroblasts promotes bladder cancer cell proliferation and invasion by regulating miR-15a-5p/HMGA2 axis. Acta Biochim. Biophys. Sin. 2021, 53, 673–682. [Google Scholar] [CrossRef]
- Han, X.; Zhang, S. Role of Long Non-Coding RNA LINC00641 in Cancer. Front. Oncol. 2021, 11, 829137. [Google Scholar] [CrossRef]
- Liu, W.H.; Lu, J.J.; Yu, R.K.; Zhou, L.; Yu, Q.; Li, D.F.; Zhu, Q.H. LINC00641 regulates prostate cancer cell growth and apoptosis via the miR-365a-3p/VGLL4 axis. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 108–115. [Google Scholar] [PubMed]
- Zhang, J.; Jin, S.; Xiao, W.; Zhu, X.; Jia, C.; Lin, Z. Long noncoding RNA LINC00641 promotes renal cell carcinoma progression via sponging microRNA-340-5p. Cancer Cell Int. 2021, 21, 210. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hong, S.; Liu, Z. LncRNA LINC00641 predicts prognosis and inhibits bladder cancer progression through miR-197-3p/KLF10/PTEN/PI3K/AKT cascade. Biochem. Biophys. Res. Commun. 2018, 503, 1825–1829. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Liu, Q.; Yang, X.; Ding, C.; Wang, Q.; Xiong, Y. LncRNA LINC00649 recruits TAF15 and enhances MAPK6 expression to promote the development of lung squamous cell carcinoma via activating MAPK signaling pathway. Cancer Gene Ther. 2022, 29, 1285–1295. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Du, C.; Zhang, L.; Wang, Y.; Zhang, Y.; Li, J. LncRNA LINC00649 promotes the growth and metastasis of triple-negative breast cancer by maintaining the stability of HIF-1α through the NF90/NF45 complex. Cell Cycle 2022, 21, 1034–1047. [Google Scholar] [CrossRef]
- Wang, H.; Di, X.; Bi, Y.; Sun, S.; Wang, T. Long non-coding RNA LINC00649 regulates YES-associated protein 1 (YAP1)/Hippo pathway to accelerate gastric cancer (GC) progression via sequestering miR-16-5p. Bioengineered 2021, 12, 1791–1802. [Google Scholar] [CrossRef]
- Feng, L.; Yang, J.; Zhang, W.; Wang, X.; Li, L.; Peng, M.; Luo, P. Prognostic significance and identification of basement membrane-associated lncRNA in bladder cancer. Front. Oncol. 2022, 12, 994703. [Google Scholar] [CrossRef]
- Chen, X.; Chen, S. LINC00649 promotes bladder cancer malignant progression by regulating the miR-15a-5p/HMGA1 axis. Oncol. Rep. 2021, 45, 8. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, X.; Guo, L.; Luo, N. LINC00649 Facilitates the Cellular Process of Bladder Cancer Cells via Signaling Axis miR-16-5p/JARID2. Urol. Int. 2022, 106, 304–312. [Google Scholar] [CrossRef]
- Li, H.; Gao, J.; Liu, L.; Zhang, S. LINC00958: A promising long non-coding RNA related to cancer. Biomed. Pharmacother. 2022, 151, 113087. [Google Scholar] [CrossRef]
- Seitz, A.K.; Christensen, L.L.; Christensen, E.; Faarkrog, K.; Ostenfeld, M.S.; Hedegaard, J.; Nordentoft, I.; Nielsen, M.M.; Palmfeldt, J.; Thomson, M.; et al. Profiling of long non-coding RNAs identifies LINC00958 and LINC01296 as candidate oncogenes in bladder cancer. Sci. Rep. 2017, 7, 395. [Google Scholar] [CrossRef]
- Xiao, Y.; He, L.; Dong, Y.; Huang, Y.; Ma, L.; Li, W. Highly Expressed Modulates the Growth and Epithelial-Mesenchymal Transition of Bladder Cancer Cells Through Signaling Pathway. Cancer Biother. Radiopharm. 2022. [Google Scholar] [CrossRef]
- Zhen, H.; Du, P.; Yi, Q.; Tang, X.; Wang, T. LINC00958 promotes bladder cancer carcinogenesis by targeting miR-490-3p and AURKA. BMC Cancer 2021, 21, 1145. [Google Scholar] [CrossRef]
- Xiao, Y.; Wang, T.; Cheng, X.; Liu, F.; Wu, Y.; Ma, L.; Li, W. LINC00958 Inhibits Autophagy of Bladder Cancer Cells via Sponge Adsorption of miR-625-5p to Promote Tumor Angiogenesis and Oxidative Stress. Oxid. Med. Cell Longev. 2022, 2022, 2435114. [Google Scholar] [CrossRef]
- Cui, Y.; Xie, M.; Zhang, Z. LINC00958 Involves in Bladder Cancer Through Sponging miR-378a-3p to Elevate IGF1R. Cancer Biother. Radiopharm. 2020, 35, 776–788. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Feng, Y.; Zheng, X.; Xu, X. The Diagnostic and Therapeutic Role of snoRNA and lincRNA in Bladder Cancer. Cancers 2023, 15, 1007. https://doi.org/10.3390/cancers15041007
Wang H, Feng Y, Zheng X, Xu X. The Diagnostic and Therapeutic Role of snoRNA and lincRNA in Bladder Cancer. Cancers. 2023; 15(4):1007. https://doi.org/10.3390/cancers15041007
Chicago/Turabian StyleWang, Hao, Yanfei Feng, Xiangyi Zheng, and Xin Xu. 2023. "The Diagnostic and Therapeutic Role of snoRNA and lincRNA in Bladder Cancer" Cancers 15, no. 4: 1007. https://doi.org/10.3390/cancers15041007
APA StyleWang, H., Feng, Y., Zheng, X., & Xu, X. (2023). The Diagnostic and Therapeutic Role of snoRNA and lincRNA in Bladder Cancer. Cancers, 15(4), 1007. https://doi.org/10.3390/cancers15041007






