Cerebellar High-Grade Glioma: A Translationally Oriented Review of the Literature
Abstract
Simple Summary
Abstract
1. Introduction
1.1. Clinical Epidemiology
1.2. Molecular Characterization and Features
- IDH mutation is uncommon
- EGFR amplification is rare
- Substantive HGAP subpopulation (~1 in 3)
- Substantive DMG H3K27 subpopulation (~1 in 10)
- More frequent RAS mutations, ATRX alteration, PDGFRA amplification, CDK2A/B loss, and CDK4 amplification than sHGG
- Less frequent TERT promotor mutations than sHGG
- Methylation classes predominantly:
- o
- (1) high-grade astrocytoma with piloid features (HGAP),
- o
- (2) GBM IDH-wildtype subclass midline (GBM-MID),
- o
- (3) GBM IDH-wildtype subclass RTK I,
- o
- (4) diffuse midline glioma H3K27-altered (DMG H3K27).
- Scant expression of telencephalic marker (FOX1)
- Widespread expression of cerebellar marker (PAX3)
- Widespread expression of oligodendrocyte progenitor marker (CSPG4)
2. Therapeutic Approaches
2.1. Classic Therapy
2.2. Emerging Molecular and Cellular Therapies
- cHGG is likely comprised of different proportions of distinct molecular subclasses compared to sHGG (GBM IDH-wt, HGAP, and DMG).
- Distinct approaches are engaged for major subclasses:
- o
- HGAP: MAPK kinase inhibitors, PI3K/mTOR inhibitors, cyclin-dependant kinase inhibitors, ATR inhibitors
- o
- DMG: GD2-CAR T cell therapy
- o
- GBM IDH-wt: PDGFRA inhibitors, cyclin-dependant kinase inhibitors, combination immunotherapies
- Unfavourable GBM IDH-wt immunological environment hampers many immunotherapies
- Oncolytic viruses are likely cerebellum-safe and promote favourable immunological environment, opening a gate for additional therapies.
- Oncolytic virus-based multi-modal immunotherapy is an attractive strategy for cerebellar GBM IDH-wt.
- Greater knowledge of cerebellar glioblastoma immunology is needed, particularly HGAP and GBM IDH-wt.
2.2.1. High-Grade Astrocytoma with Piloid Features
2.2.2. Diffuse Midline Glioma, H3K27M
2.2.3. Glioblastoma, IDH-wt
2.2.4. Immunotherapies
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adams, H.; Chaichana, K.L.; Avendaño, J.; Liu, B.; Raza, S.M.; Quiñones-Hinojosa, A. Adult Cerebellar Glioblastoma: Understanding Survival and Prognostic Factors Using a Population-Based Database from 1973 to 2009. World Neurosurg. 2013, 80, e237–e243. [Google Scholar] [CrossRef] [PubMed]
- von Bartheld, C.S.; Bahney, J.; Herculano-Houzel, S. The search for true numbers of neurons and glial cells in the human brain: A review of 150 years of cell counting: Quantifying neurons and glia in human brain. J. Comp. Neurol. 2016, 524, 3865–3895. [Google Scholar] [CrossRef] [PubMed]
- Zülch, K.J. Brain Tumors: Their Biology and Pathology; Springer Pub. Co.: New York, NY, USA, 1957; pp. 62–198. [Google Scholar]
- Jeswani, S.; Nuño, M.; Folkerts, V.; Mukherjee, D.; Black, K.L.; Patil, C.G. Comparison of Survival Between Cerebellar and Supratentorial Glioblastoma Patients: Surveillance, Epidemiology, and End Results (SEER) Analysis. Neurosurgery 2013, 73, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Picart, T.; Barritault, M.; Berthillier, J.; Meyronet, D.; Vasiljevic, A.; Frappaz, D.; Honnorat, J.; Jouanneau, E.; Poncet, D.; Ducray, F.; et al. Characteristics of cerebellar glioblastomas in adults. J. Neurooncol. 2018, 136, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.C.; Miller, R.C.; Villà, S.; Hanssens, P.; Baumert, B.G.; Castadot, P.; Varlet, P.; Abacioglu, U.; Igdem, S.; Szutowicz, E.; et al. Outcome and prognostic factors in cerebellar glioblastoma multiforme in adults: A retrospective study from the Rare Cancer Network. Int. J. Radiat. Oncol. 2006, 66, 179–186. [Google Scholar] [CrossRef]
- Djalilian, H.R.; Hall, W.A. Malignant gliomas of the cerebellum: An analytic review. J. Neurooncol. 1998, 36, 247–257. [Google Scholar] [CrossRef]
- Yang, S.; Liu, J.; Wang, T.; Li, X.; You, C. Cerebellar glioblastoma multiforme: A retrospective study of 28 patients at a single institution. Int. J. Neurosci. 2013, 123, 691–697. [Google Scholar] [CrossRef]
- Babu, R.; Sharma, R.; Karikari, I.O.; Owens, T.R.; Friedman, A.H.; Adamson, C. Outcome and prognostic factors in adult cerebellar glioblastoma. J. Clin. Neurosci. 2013, 20, 1117–1121. [Google Scholar] [CrossRef]
- Cho, H.J.; Zhao, J.; Jung, S.W.; Ladewig, E.; Kong, D.-S.; Suh, Y.-L.; Lee, Y.; Kim, D.; Ahn, S.H.; Bordyuh, M.; et al. Distinct genomic profile and specific targeted drug responses in adult cerebellar glioblastoma. Neuro-Oncology 2019, 21, 47–58. [Google Scholar] [CrossRef]
- Kast, R.E. Why cerebellar glioblastoma is rare and how that indicates adjunctive use of the FDA-approved anti-emetic aprepitant might retard cerebral glioblastoma growth: A new hypothesis to an old question. Clin. Transl. Oncol. 2009, 11, 408–410. [Google Scholar] [CrossRef]
- Hong, B.; Banan, R.; Christians, A.; Nakamura, M.; Lalk, M.; Lehmann, U.; Hartmann, C.; Krauss, J.K. Cerebellar glioblastoma: A clinical series with contemporary molecular analysis. Acta Neurochir. 2018, 160, 2237–2248. [Google Scholar] [CrossRef] [PubMed]
- Nobusawa, S.; Watanabe, T.; Kleihues, P.; Ohgaki, H. IDH1 Mutations as Molecular Signature and Predictive Factor of Secondary Glioblastomas. Clin. Cancer Res. 2009, 15, 6002–6007. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Tachibana, O.; Sato, K.; Yonekawa, Y.; Kleihues, P.; Ohgaki, H. Overexpression of the EGF Receptor and p53 Mutations are Mutually Exclusive in the Evolution of Primary and Secondary Glioblastomas. Brain Pathol. 1996, 6, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; McLendon, R.; Kos, I.; Riggins, G.J.; Reardon, D.; Velculescu, V.E.; Bigner, D.D. IDH1 and IDH2 Mutations in Gliomas. N. Engl. J. Med. 2009, 360, 765–773. [Google Scholar] [CrossRef]
- Milinkovic, V.P.; Skender Gazibara, M.K.; Manojlovic Gacic, E.M.; Gazibara, T.M.; Tanic, N.T. The impact of TP53 and RAS mutations on cerebellar glioblastomas. Exp. Mol. Pathol. 2014, 97, 202–207. [Google Scholar] [CrossRef]
- Verhaak, R.G.W.; Hoadley, K.A.; Purdom, E.; Wang, V.; Qi, Y.; Wilkerson, M.D.; Miller, C.R.; Ding, L.; Golub, T.; Mesirov, J.P.; et al. Integrated Genomic Analysis Identifies Clinically Relevant Subtypes of Glioblastoma Characterized by Abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 2010, 17, 98–110. [Google Scholar] [CrossRef]
- Brennan, C.W.; Verhaak, R.G.W.; McKenna, A.; Campos, B.; Noushmehr, H.; Salama, S.R.; Zheng, S.; Chakravarty, D.; Sanborn, J.Z.; Berman, S.H.; et al. The Somatic Genomic Landscape of Glioblastoma. Cell 2013, 155, 462–477. [Google Scholar] [CrossRef]
- Utsuki, S.; Oka, H.; Miyajima, Y.; Kijima, C.; Yasui, Y.; Fujii, K. Adult cerebellar glioblastoma cases have different characteristics from supratentorial glioblastoma. Brain Tumor Pathol. 2012, 29, 87–95. [Google Scholar] [CrossRef]
- Saito, T.; Hama, S.; Kajiwara, Y.; Sugiyama, K.; Yamasaki, F.; Arifin, M.-T.; Arita, K.; Kurisu, K. Prognosis of Cerebellar Glioblastomas: Correlation Between Prognosis and Immunoreactivity for Epidermal Growth Factor Receptor Compared with Supratentorial Glioblastomas. Anticancer Res. 2006, 26, 1351–1357. [Google Scholar]
- Takahashi, Y.; Makino, K.; Nakamura, H.; Hide, T.; Yano, S.; Kamada, H.; Kuratsu, J.-I. Clinical characteristics and pathogenesis of cerebellar glioblastoma. Mol. Med. Rep. 2014, 10, 2383–2388. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro-Oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Sturm, D.; Witt, H.; Hovestadt, V.; Khuong-Quang, D.-A.; Jones, D.T.W.; Konermann, C.; Pfaff, E.; Tönjes, M.; Sill, M.; Bender, S.; et al. Hotspot Mutations in H3F3A and IDH1 Define Distinct Epigenetic and Biological Subgroups of Glioblastoma. Cancer Cell 2012, 22, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Schulte, J.D.; Buerki, R.A.; Lapointe, S.; Molinaro, A.M.; Zhang, Y.; Villanueva-Meyer, J.E.; Perry, A.; Phillips, J.J.; Tihan, T.; Bollen, A.W.; et al. Clinical, radiologic, and genetic characteristics of histone H3 K27M-mutant diffuse midline gliomas in adults. Neuro-Oncol. Adv. 2020, 2, vdaa142. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, A.; Stichel, D.; Schrimpf, D.; Koelsche, C.; Wefers, A.K.; Ebrahimi, A.; Sievers, P.; Huang, K.; Casalini, M.B.; Fernández-Klett, F.; et al. Tumors diagnosed as cerebellar glioblastoma comprise distinct molecular entities. Acta Neuropathol. Commun. 2019, 7, 163. [Google Scholar] [CrossRef] [PubMed]
- Nomura, M.; Mukasa, A.; Nagae, G.; Yamamoto, S.; Tatsuno, K.; Ueda, H.; Fukuda, S.; Umeda, T.; Suzuki, T.; Otani, R.; et al. Distinct molecular profile of diffuse cerebellar gliomas. Acta Neuropathol. 2017, 134, 941–956. [Google Scholar] [CrossRef]
- Bender, K.; Perez, E.; Chirica, M.; Onken, J.; Kahn, J.; Brenner, W.; Ehret, F.; Euskirchen, P.; Koch, A.; Capper, D.; et al. High-grade astrocytoma with piloid features (HGAP): The Charité experience with a new central nervous system tumor entity. J. Neurooncol. 2021, 153, 109–120. [Google Scholar] [CrossRef]
- Reinhardt, A.; Stichel, D.; Schrimpf, D.; Sahm, F.; Korshunov, A.; Reuss, D.E.; Koelsche, C.; Huang, K.; Wefers, A.K.; Hovestadt, V.; et al. Anaplastic astrocytoma with piloid features, a novel molecular class of IDH wildtype glioma with recurrent MAPK pathway, CDKN2A/B and ATRX alterations. Acta Neuropathol. 2018, 136, 273–291. [Google Scholar] [CrossRef]
- Tsung, A.J.; Prabhu, S.S.; Lei, X.; Chern, J.J.; Benjamin Bekele, N.; Shonka, N.A. Cerebellar glioblastoma: A retrospective review of 21 patients at a single institution. J. Neurooncol. 2011, 105, 555–562. [Google Scholar] [CrossRef]
- Salazar, O.M. Primary malignant cerebellar astrocytomas in children: A signal for postoperative craniospinal irradiation. Int. J. Radiat. Oncol. 1981, 7, 1661–1665. [Google Scholar] [CrossRef]
- Barker, F.G.; Simmons, M.L.; Chang, S.M.; Prados, M.D.; Larson, D.A.; Sneed, P.K.; Wara, W.M.; Berger, M.S.; Chen, P.; Israel, M.A.; et al. EGFR overexpression and radiation response in glioblastoma multiforme. Int. J. Radiat. Oncol. 2001, 51, 410–418. [Google Scholar] [CrossRef]
- Zhu, A.; Shaeffer, J.; Leslie, S.; Kolm, P.; El-Mahdi, A.M. Epidermal growth factor receptor: An independent predictor of survival in astrocytic tumors given definitive irradiation. Int. J. Radiat. Oncol. 1996, 34, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.T.W.; Hutter, B.; Jäger, N.; Korshunov, A.; Kool, M.; Warnatz, H.-J.; Zichner, T.; Lambert, S.R.; Ryzhova, M.; Quang, D.A.K.; et al. Recurrent somatic alterations of FGFR1 and NTRK2 in pilocytic astrocytoma. Nat. Genet. 2013, 45, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, C.; Ellis, H.P.; Shaw, A.; Penman, C.; Palmer, A.; Wragg, C.; Greenslade, M.; Haynes, H.R.; Williams, H.; Lowis, S.; et al. BRAF Fusion Analysis in Pilocytic Astrocytomas: KIAA1549-BRAF 15-9 Fusions Are More Frequent in the Midline Than Within the Cerebellum. J. Neuropathol. Exp. Neurol. 2015, 74, 867–872. [Google Scholar] [CrossRef]
- Mistry, M.; Zhukova, N.; Merico, D.; Rakopoulos, P.; Krishnatry, R.; Shago, M.; Stavropoulos, J.; Alon, N.; Pole, J.D.; Ray, P.N.; et al. BRAF Mutation and CDKN2A Deletion Define a Clinically Distinct Subgroup of Childhood Secondary High-Grade Glioma. J. Clin. Oncol. 2015, 33, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Olar, A.; Tran, D.; Mehta, V.P.; Reinhardt, A.; Manekia, J.H.; Garnovskaya, M.; Ellezam, B.; Luthra, R.; Sulman, E.P.; Mohila, C.A.; et al. ATRX protein loss and deregulation of PI3K/AKT pathway is frequent in pilocytic astrocytoma with anaplastic features. Clin. Neuropathol. 2019, 38, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Sievert, A.J.; Lang, S.-S.; Boucher, K.L.; Madsen, P.J.; Slaunwhite, E.; Choudhari, N.; Kellet, M.; Storm, P.B.; Resnick, A.C. Paradoxical activation and RAF inhibitor resistance of BRAF protein kinase fusions characterizing pediatric astrocytomas. Proc. Natl. Acad. Sci. USA 2013, 110, 5957–5962. [Google Scholar] [CrossRef]
- Jain, P.; Silva, A.; Han, H.J.; Lang, S.-S.; Zhu, Y.; Boucher, K.; Smith, T.E.; Vakil, A.; Diviney, P.; Choudhari, N.; et al. Overcoming resistance to single-agent therapy for oncogenic BRAF gene fusions via combinatorial targeting of MAPK and PI3K/mTOR signaling pathways. Oncotarget 2017, 8, 84697–84713. [Google Scholar] [CrossRef]
- McNeill, R.S.; Canoutas, D.A.; Stuhlmiller, T.J.; Dhruv, H.D.; Irvin, D.M.; Bash, R.E.; Angus, S.P.; Herring, L.E.; Simon, J.M.; Skinner, K.R.; et al. Combination therapy with potent PI3K and MAPK inhibitors overcomes adaptive kinome resistance to single agents in preclinical models of glioblastoma. Neuro-Oncology 2017, 19, 1469–1480. [Google Scholar] [CrossRef]
- Loilome, W.; Joshi, A.D.; ap Rhys, C.M.J.; Piccirillo, S.; Angelo, V.L.; Gallia, G.L.; Riggins, G.J. Glioblastoma cell growth is suppressed by disruption of fibroblast growth factor pathway signaling. J. Neurooncol. 2009, 94, 359–366. [Google Scholar] [CrossRef]
- Koschmann, C.; Calinescu, A.-A.; Nunez, F.J.; Mackay, A.; Fazal-Salom, J.; Thomas, D.; Mendez, F.; Kamran, N.; Dzaman, M.; Mulpuri, L.; et al. ATRX loss promotes tumor growth and impairs nonhomologous end joining DNA repair in glioma. Sci. Transl. Med. 2016, 8, 328ra28. [Google Scholar] [CrossRef]
- Flynn, R.L.; Cox, K.E.; Jeitany, M.; Wakimoto, H.; Bryll, A.R.; Ganem, N.J.; Bersani, F.; Pineda, J.R.; Suvà, M.L.; Benes, C.H.; et al. Alternative lengthening of telomeres renders cancer cells hypersensitive to ATR inhibitors. Science 2015, 347, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Xiao, A.; Floyd, D.; Olmez, I.; Lee, J.; Godlewski, J.; Bronisz, A.; Bhat, K.P.L.; Sulman, E.P.; Nakano, I.; et al. CDK4/6 inhibition is more active against the glioblastoma proneural subtype. Oncotarget 2017, 8, 55319–55331. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.W.; Parikh, M.; Phillips, J.J.; James, C.D.; Molinaro, A.M.; Butowski, N.A.; Clarke, J.L.; Oberheim-Bush, N.A.; Chang, S.M.; Berger, M.S.; et al. Phase-2 trial of palbociclib in adult patients with recurrent RB1-positive glioblastoma. J. Neurooncol. 2018, 140, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, S.; Madani, D.; Joshi, S.; Chung, S.A.; Johns, T.; Day, B.; Khasraw, M.; McDonald, K.L. Combination of palbociclib and radiotherapy for glioblastoma. Cell Death Discov. 2017, 3, 17033. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, R.; Zhang, A.; Mueller, S.; Prados, M.D.; Lulla, R.R.; Goldman, S.; Saratsis, A.M.; Mazar, A.P.; Stegh, A.H.; Cheng, S.-Y.; et al. Inhibition of DNA damage repair by the CDK4/6 inhibitor palbociclib delays irradiated intracranial atypical teratoid rhabdoid tumor and glioblastoma xenograft regrowth. Neuro-Oncology 2016, 18, 1519–1528. [Google Scholar] [CrossRef]
- Grasso, C.S.; Tang, Y.; Truffaux, N.; Berlow, N.E.; Liu, L.; Debily, M.-A.; Quist, M.J.; Davis, L.E.; Huang, E.C.; Woo, P.J.; et al. Functionally defined therapeutic targets in diffuse intrinsic pontine glioma. Nat. Med. 2015, 21, 555–559. [Google Scholar] [CrossRef]
- Ochs, K.; Ott, M.; Bunse, T.; Sahm, F.; Bunse, L.; Deumelandt, K.; Sonner, J.K.; Keil, M.; von Deimling, A.; Wick, W.; et al. K27M-mutant histone-3 as a novel target for glioma immunotherapy. OncoImmunology 2017, 6, e1328340. [Google Scholar] [CrossRef]
- Majzner, R.G.; Ramakrishna, S.; Yeom, K.W.; Patel, S.; Chinnasamy, H.; Schultz, L.M.; Richards, R.M.; Jiang, L.; Barsan, V.; Mancusi, R.; et al. GD2-CAR T cell therapy for H3K27M-mutated diffuse midline gliomas. Nature 2022, 603, 934–941. [Google Scholar] [CrossRef]
- Mount, C.W.; Majzner, R.G.; Sundaresh, S.; Arnold, E.P.; Kadapakkam, M.; Haile, S.; Labanieh, L.; Hulleman, E.; Woo, P.J.; Rietberg, S.P.; et al. Potent antitumor efficacy of anti-GD2 CAR T cells in H3-K27M+ diffuse midline gliomas. Nat. Med. 2018, 24, 572–579. [Google Scholar] [CrossRef]
- Zhang, L.; Nesvick, C.L.; Day, C.A.; Choi, J.; Lu, V.M.; Peterson, T.; Power, E.A.; Anderson, J.B.; Hamdan, F.H.; Decker, P.A.; et al. STAT3 is a biologically relevant therapeutic target in H3K27M-mutant diffuse midline glioma. Neuro-Oncology 2022, 24, 1700–1711. [Google Scholar] [CrossRef]
- Pearson, J.R.D.; Regad, T. Targeting cellular pathways in glioblastoma multiforme. Signal Transduct. Target. Ther. 2017, 2, e17040. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.W.; Quail, D.F. Immunotherapy for Glioblastoma: Current Progress and Challenges. Front. Immunol. 2021, 12, 676301. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.; Xia, Y.; Bettegowda, C.; Weller, M. Current state of immunotherapy for glioblastoma. Nat. Rev. Clin. Oncol. 2018, 15, 422–442. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.P.; Tirosh, I.; Trombetta, J.J.; Shalek, A.K.; Gillespie, S.M.; Wakimoto, H.; Cahill, D.P.; Nahed, B.V.; Curry, W.T.; Martuza, R.L.; et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 2014, 344, 1396–1401. [Google Scholar] [CrossRef] [PubMed]
- Sottoriva, A.; Spiteri, I.; Piccirillo, S.G.M.; Touloumis, A.; Collins, V.P.; Marioni, J.C.; Curtis, C.; Watts, C.; Tavaré, S. Intratumor heterogeneity in human glioblastoma reflects cancer evolutionary dynamics. Proc. Natl. Acad. Sci. USA 2013, 110, 4009–4014. [Google Scholar] [CrossRef]
- Vinci, M.; Burford, A.; Molinari, V.; Kessler, K.; Popov, S.; Clarke, M.; Taylor, K.R.; Pemberton, H.N.; Lord, C.J.; Gutteridge, A.; et al. Functional diversity and cooperativity between subclonal populations of pediatric glioblastoma and diffuse intrinsic pontine glioma cells. Nat. Med. 2018, 24, 1204–1215. [Google Scholar] [CrossRef]
- Pearson, J.R.D.; Cuzzubbo, S.; McArthur, S.; Durrant, L.G.; Adhikaree, J.; Tinsley, C.J.; Pockley, A.G.; McArdle, S.E.B. Immune Escape in Glioblastoma Multiforme and the Adaptation of Immunotherapies for Treatment. Front. Immunol. 2020, 11, 582106. [Google Scholar] [CrossRef]
- Bernstock, J.D.; Hoffman, S.E.; Chen, J.A.; Gupta, S.; Kappel, A.D.; Smith, T.R.; Chiocca, E.A. The Current Landscape of Oncolytic Herpes Simplex Viruses as Novel Therapies for Brain Malignancies. Viruses 2021, 13, 1158. [Google Scholar] [CrossRef]
- Uche, I.K.; Kousoulas, K.G.; Rider, P.J.F. The Effect of Herpes Simplex Virus-Type-1 (HSV-1) Oncolytic Immunotherapy on the Tumor Microenvironment. Viruses 2021, 13, 1200. [Google Scholar] [CrossRef]
- Bernstock, J.D.; Vicario, N.; Li, R.; Nan, L.; Totsch, S.K.; Schlappi, C.; Gessler, F.; Han, X.; Parenti, R.; Beierle, E.A.; et al. Safety and efficacy of oncolytic HSV-1 G207 inoculated into the cerebellum of mice. Cancer Gene Ther. 2020, 27, 246–255. [Google Scholar] [CrossRef]
- Friedman, G.K.; Johnston, J.M.; Bag, A.K.; Bernstock, J.D.; Li, R.; Aban, I.; Kachurak, K.; Nan, L.; Kang, K.-D.; Totsch, S.; et al. Oncolytic HSV-1 G207 Immunovirotherapy for Pediatric High-Grade Gliomas. N. Engl. J. Med. 2021, 384, 1613–1622. [Google Scholar] [CrossRef] [PubMed]
- Bernstock, J.D.; Bag, A.K.; Fiveash, J.; Kachurak, K.; Elsayed, G.; Chagoya, G.; Gessler, F.; Valdes, P.A.; Madan-Swain, A.; Whitley, R.; et al. Design and Rationale for First-in-Human Phase 1 Immunovirotherapy Clinical Trial of Oncolytic HSV G207 to Treat Malignant Pediatric Cerebellar Brain Tumors. Hum. Gene Ther. 2020, 31, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Reardon, D.A.; Brandes, A.A.; Omuro, A.; Mulholland, P.; Lim, M.; Wick, A.; Baehring, J.; Ahluwalia, M.S.; Roth, P.; Bähr, O.; et al. Effect of Nivolumab vs Bevacizumab in Patients with Recurrent Glioblastoma: The CheckMate 143 Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020, 6, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Mende, A.L.; Schulte, J.D.; Okada, H.; Clarke, J.L. Current Advances in Immunotherapy for Glioblastoma. Curr. Oncol. Rep. 2021, 23, 21. [Google Scholar] [CrossRef]
- Cloughesy, T.F.; Mochizuki, A.Y.; Orpilla, J.R.; Hugo, W.; Lee, A.H.; Davidson, T.B.; Wang, A.C.; Ellingson, B.M.; Rytlewski, J.A.; Sanders, C.M.; et al. Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat. Med. 2019, 25, 477–486. [Google Scholar] [CrossRef]
- Darvin, P.; Toor, S.M.; Sasidharan Nair, V.; Elkord, E. Immune checkpoint inhibitors: Recent progress and potential biomarkers. Exp. Mol. Med. 2018, 50, 1–11. [Google Scholar] [CrossRef]
- Filley, A.C.; Henriquez, M.; Dey, M. Recurrent glioma clinical trial, CheckMate-143: The game is not over yet. Oncotarget 2017, 8, 91779–91794. [Google Scholar] [CrossRef]
- Hardcastle, J.; Mills, L.; Malo, C.S.; Jin, F.; Kurokawa, C.; Geekiyanage, H.; Schroeder, M.; Sarkaria, J.; Johnson, A.J.; Galanis, E. Immunovirotherapy with measles virus strains in combination with anti–PD-1 antibody blockade enhances antitumor activity in glioblastoma treatment. Neuro-Oncology 2016, 19, 493–502. [Google Scholar] [CrossRef]
- Saha, D.; Martuza, R.L.; Rabkin, S.D. Macrophage Polarization Contributes to Glioblastoma Eradication by Combination Immunovirotherapy and Immune Checkpoint Blockade. Cancer Cell 2017, 32, 253–267.e5. [Google Scholar] [CrossRef]
- Liau, L.M.; Ashkan, K.; Tran, D.D.; Campian, J.L.; Trusheim, J.E.; Cobbs, C.S.; Heth, J.A.; Salacz, M.; Taylor, S.; D’Andre, S.D.; et al. First results on survival from a large Phase 3 clinical trial of an autologous dendritic cell vaccine in newly diagnosed glioblastoma. J. Transl. Med. 2018, 16, 142. [Google Scholar] [CrossRef]
- Morantz, R.A.; Clark, M. Macrophages in experimental and human brain tumors. J. Neurosurg. 1979, 50, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Hambardzumyan, D.; Gutmann, D.H.; Kettenmann, H. The role of microglia and macrophages in glioma maintenance and progression. Nat. Neurosci. 2016, 19, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Andersen, B.M.; Faust Akl, C.; Wheeler, M.A.; Chiocca, E.A.; Reardon, D.A.; Quintana, F.J. Glial and myeloid heterogeneity in the brain tumour microenvironment. Nat. Rev. Cancer 2021, 21, 786–802. [Google Scholar] [CrossRef] [PubMed]
- Pyonteck, S.M.; Akkari, L.; Schuhmacher, A.J.; Bowman, R.L.; Sevenich, L.; Quail, D.F.; Olson, O.C.; Quick, M.L.; Huse, J.T.; Teijeiro, V.; et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat. Med. 2013, 19, 1264–1272. [Google Scholar] [CrossRef]
- Quail, D.F.; Bowman, R.L.; Akkari, L.; Quick, M.L.; Schuhmacher, A.J.; Huse, J.T.; Holland, E.C.; Sutton, J.C.; Joyce, J.A. The tumor microenvironment underlies acquired resistance to CSF-1R inhibition in gliomas. Science 2016, 352, aad3018. [Google Scholar] [CrossRef]
- Butowski, N.; Colman, H.; De Groot, J.F.; Omuro, A.M.; Nayak, L.; Wen, P.Y.; Cloughesy, T.F.; Marimuthu, A.; Haidar, S.; Perry, A.; et al. Orally administered colony stimulating factor 1 receptor inhibitor PLX3397 in recurrent glioblastoma: An Ivy Foundation Early Phase Clinical Trials Consortium phase II study. Neuro-Oncology 2016, 18, 557–564. [Google Scholar] [CrossRef]
- Price, G.; Bouras, A.; Hambardzumyan, D.; Hadjipanayis, C.G. Current knowledge on the immune microenvironment and emerging immunotherapies in diffuse midline glioma. eBioMedicine 2021, 69, 103453. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, B.; Hu, X.; Kim, H.; Squatrito, M.; Scarpace, L.; deCarvalho, A.C.; Lyu, S.; Li, P.; Li, Y.; et al. Tumor Evolution of Glioma-Intrinsic Gene Expression Subtypes Associates with Immunological Changes in the Microenvironment. Cancer Cell 2017, 32, 42–56.e6. [Google Scholar] [CrossRef]
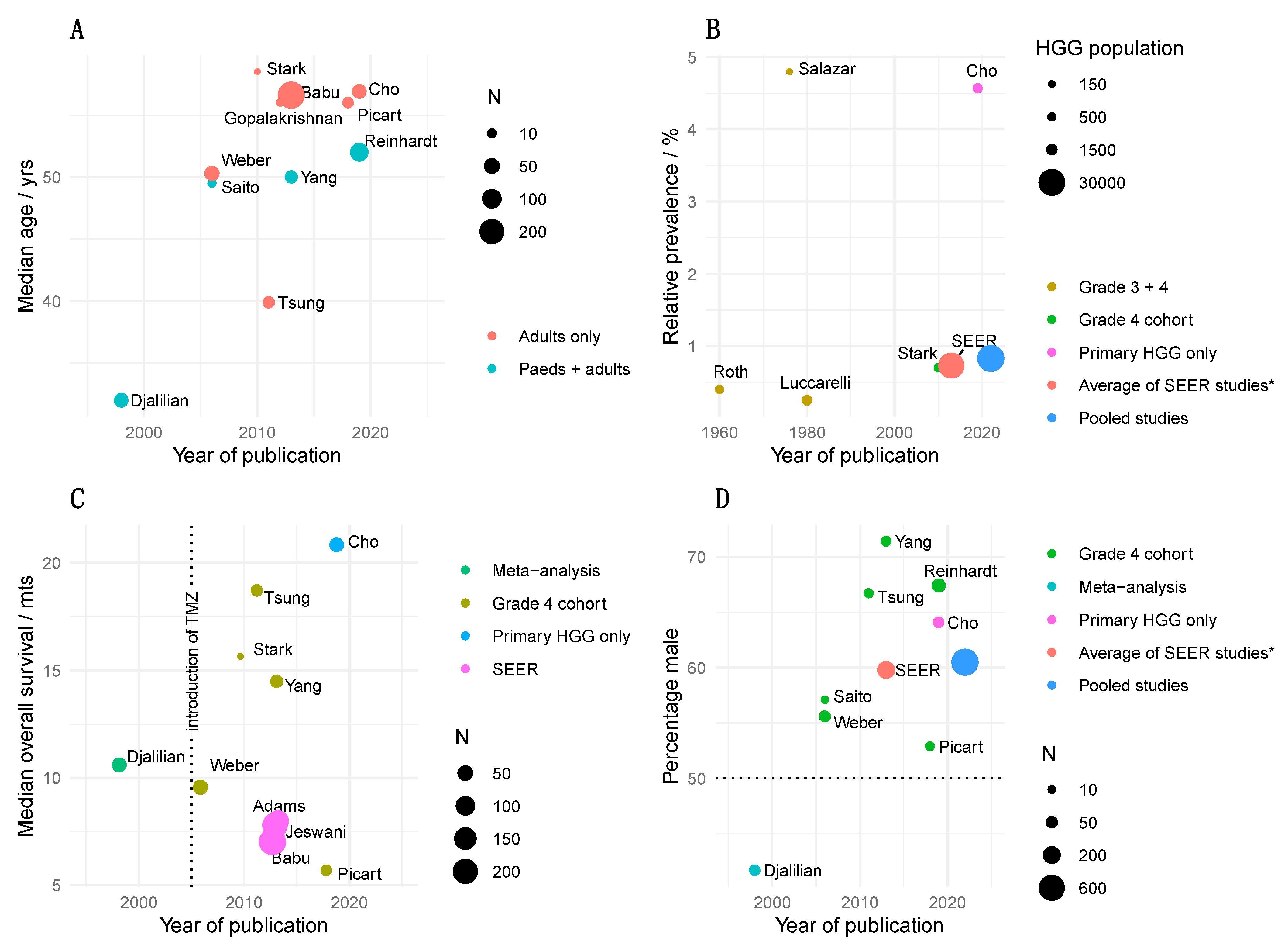
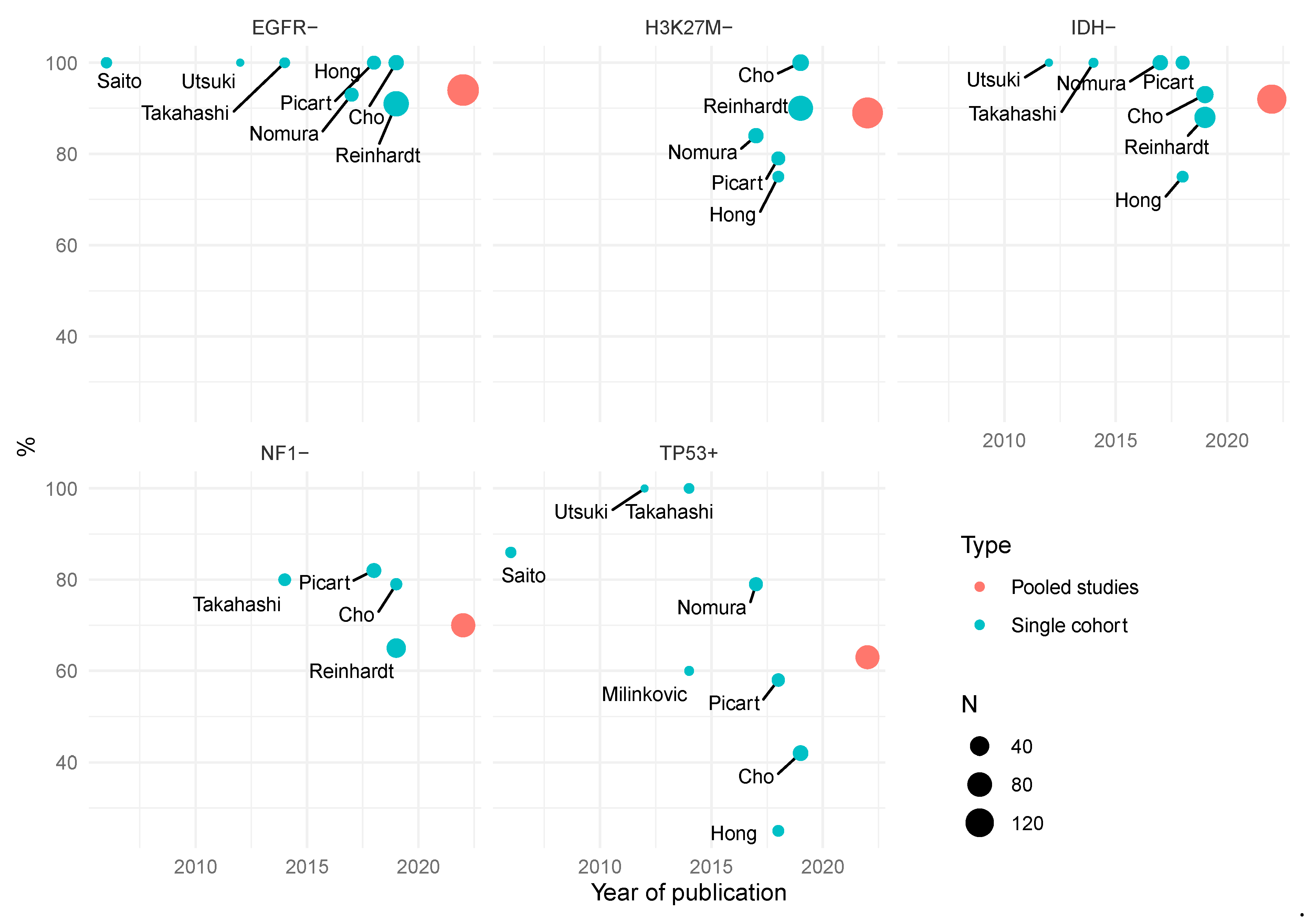
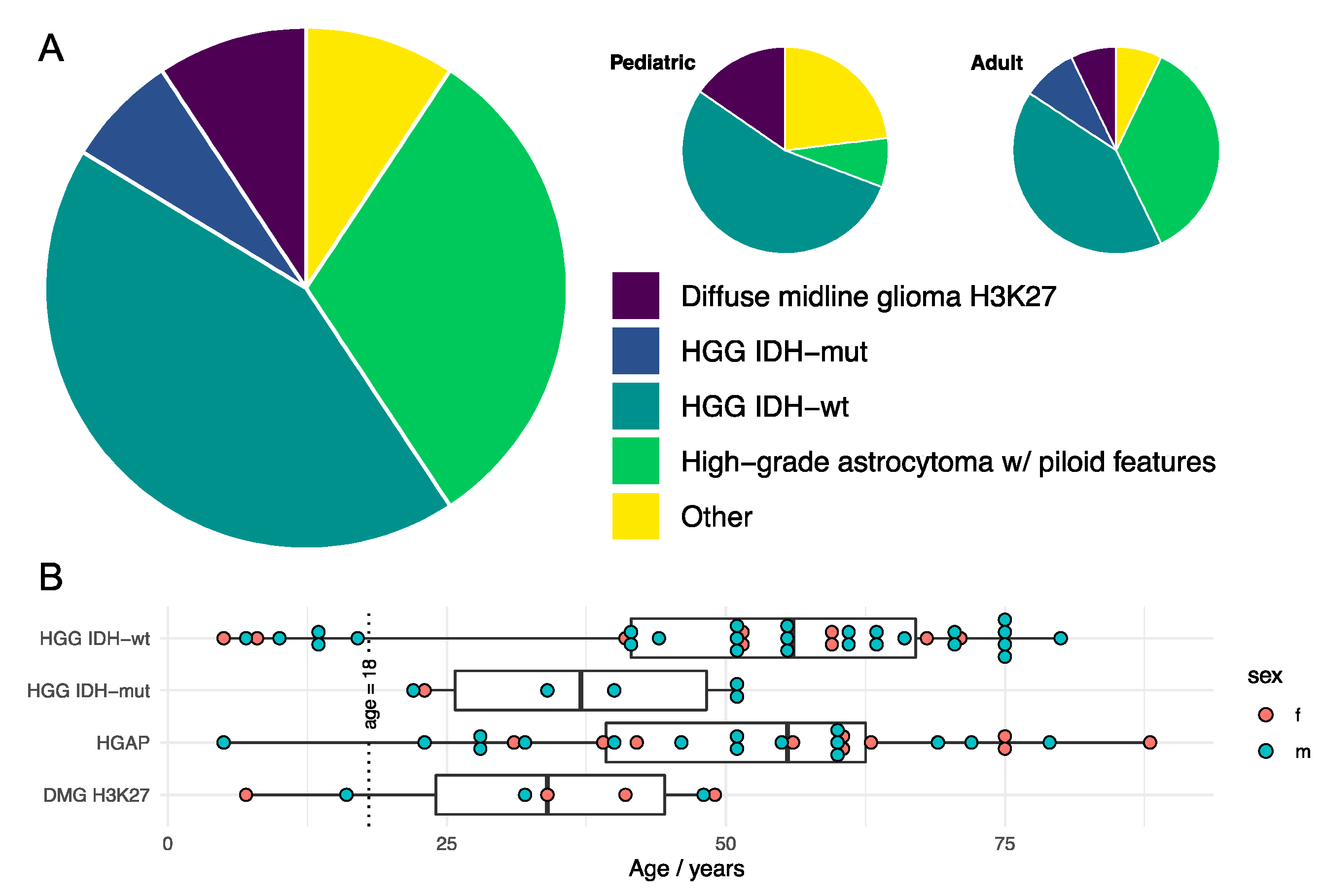
| Author | Study | Location | Male/% | Age/Years | MOS/Months |
|---|---|---|---|---|---|
| Babu et al. [9] | SEER database analysis | Supratentorial | 59.5 | μ = 61.8 * | 7 |
| Cerebellar | 58.1 | μ = 56.6 * | 8 | ||
| Adams et al. [1] | Supratentorial | n.s. p = 0.87 | μ (σ) = 61 (13) * | 8 † | |
| Cerebellar | μ (σ) = 58 (16) * | 9 † | |||
| Jeswani et al. [4] | Supratentorial | n.r. | 7.4% < 40 * | 8 † | |
| Cerebellar | 62 | 23.5% < 40 * | 7 † | ||
| Cho et al. [10] | Single cohort | Supratentorial | 54.5 | η = 55.3 | 16 |
| Cerebellar | 64.1 | η = 56.9 | 21 | ||
| Picart et al. [5] | Single cohort | Supratentorial | 60 * | μ (σ) = 63.2 (13.3) * | 14 * |
| Cerebellar | 52.9 * | μ (σ) = 53.4 (15.7) * | 6 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raghu, A.L.B.; Chen, J.A.; Valdes, P.A.; Essayed, W.I.; Claus, E.; Arnaout, O.; Smith, T.R.; Chiocca, E.A.; Peruzzi, P.P.; Bernstock, J.D. Cerebellar High-Grade Glioma: A Translationally Oriented Review of the Literature. Cancers 2023, 15, 174. https://doi.org/10.3390/cancers15010174
Raghu ALB, Chen JA, Valdes PA, Essayed WI, Claus E, Arnaout O, Smith TR, Chiocca EA, Peruzzi PP, Bernstock JD. Cerebellar High-Grade Glioma: A Translationally Oriented Review of the Literature. Cancers. 2023; 15(1):174. https://doi.org/10.3390/cancers15010174
Chicago/Turabian StyleRaghu, Ashley L. B., Jason A. Chen, Pablo A. Valdes, Walid Ibn Essayed, Elizabeth Claus, Omar Arnaout, Timothy R. Smith, E. Antonio Chiocca, Pier Paolo Peruzzi, and Joshua D. Bernstock. 2023. "Cerebellar High-Grade Glioma: A Translationally Oriented Review of the Literature" Cancers 15, no. 1: 174. https://doi.org/10.3390/cancers15010174
APA StyleRaghu, A. L. B., Chen, J. A., Valdes, P. A., Essayed, W. I., Claus, E., Arnaout, O., Smith, T. R., Chiocca, E. A., Peruzzi, P. P., & Bernstock, J. D. (2023). Cerebellar High-Grade Glioma: A Translationally Oriented Review of the Literature. Cancers, 15(1), 174. https://doi.org/10.3390/cancers15010174






