Sorafenib Versus Lenvatinib-Based Sequential Systemic Therapy for Advanced Hepatocellular Carcinoma: A Real-World Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Sequences and Durations of Systemic Therapies
3.3. Sequential Treatment after First-Line Therapy with Sorafenib
3.4. Sequential Treatment after First-Line Therapy with Lenvatinib
3.5. Deterioration of Liver Function during Systemic Therapy
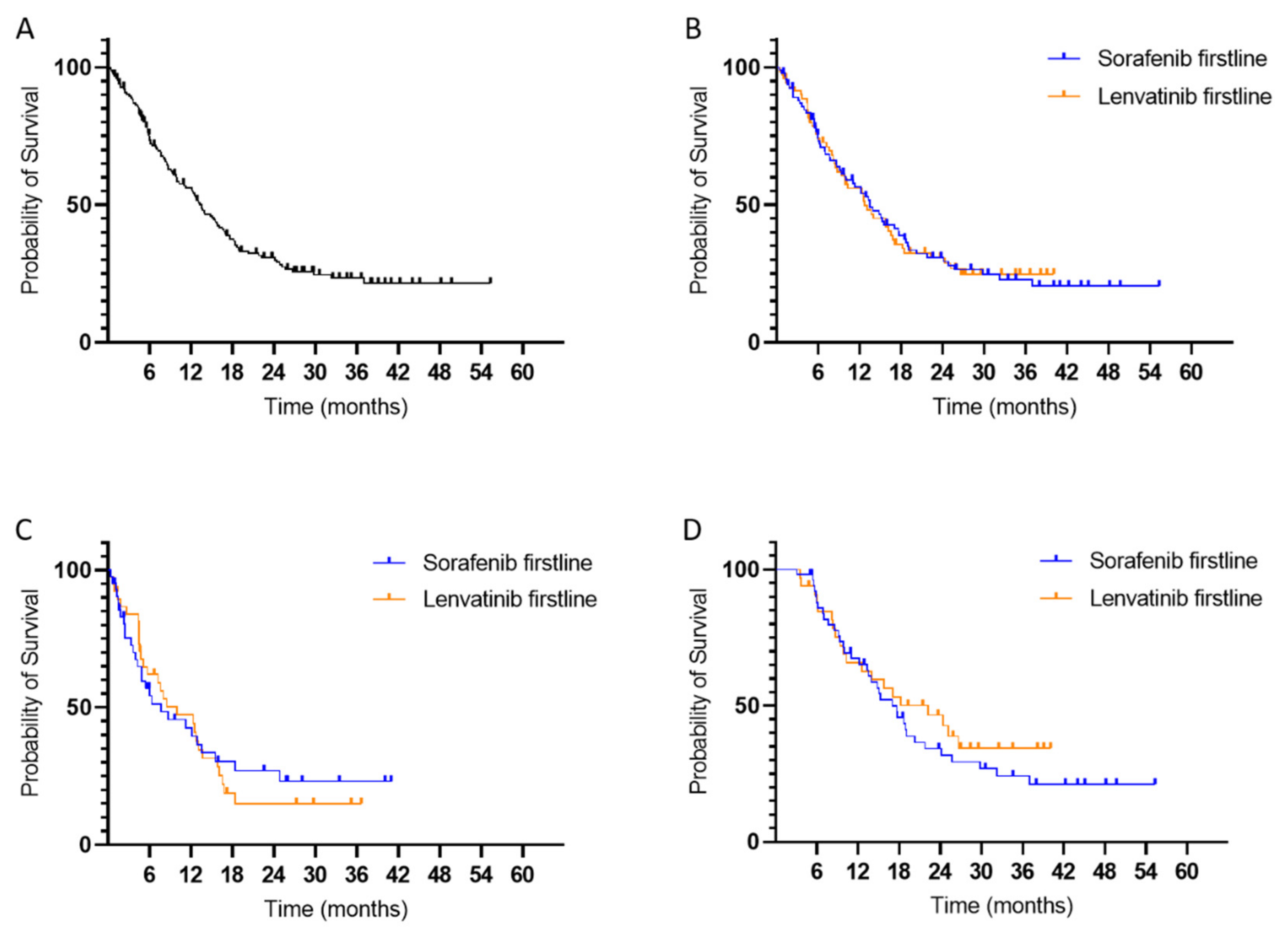
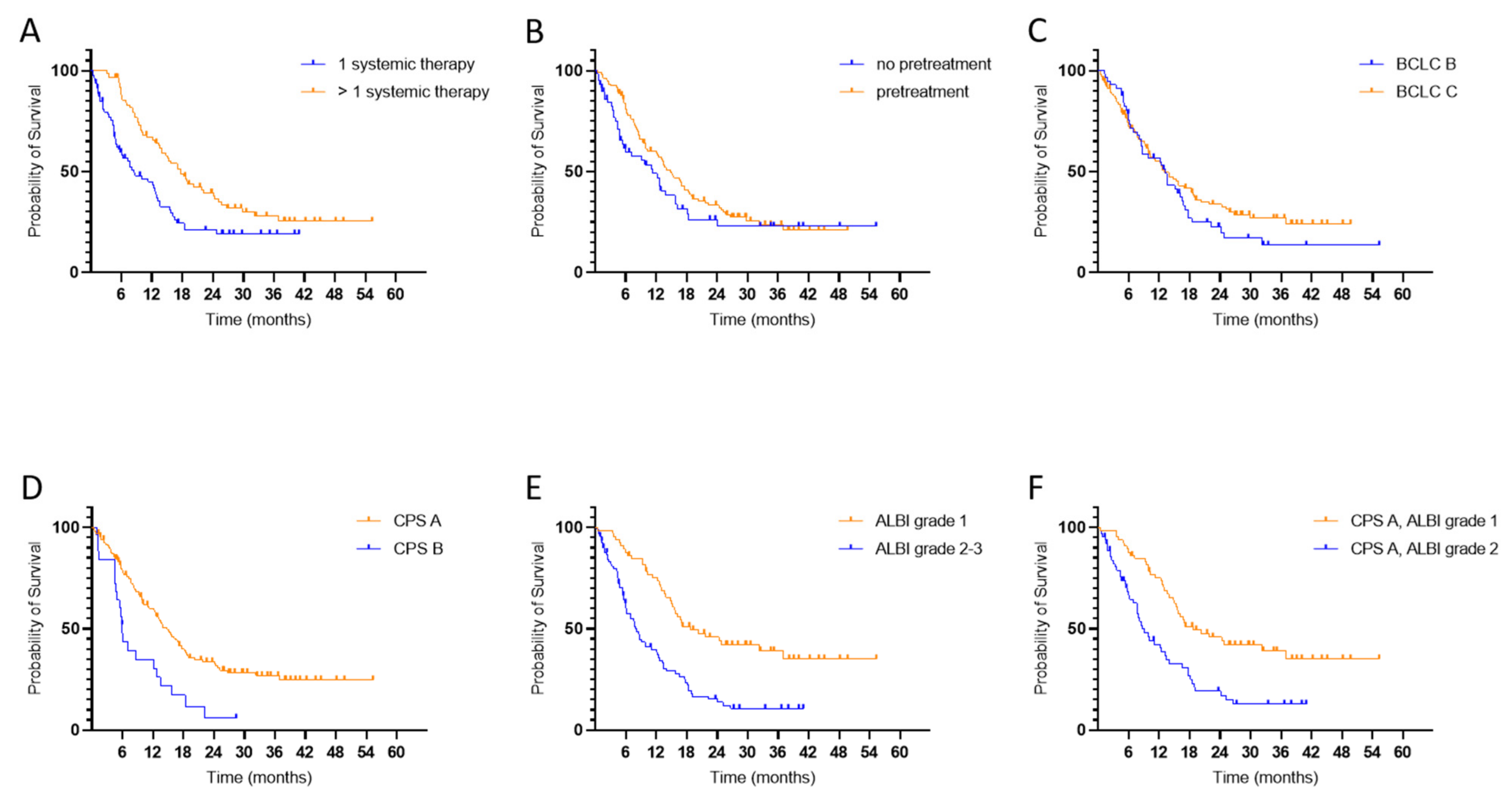
3.6. Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McGlynn, K.A.; Petrick, J.L.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma. Hepatology 2021, 73, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.; Brú, C.; Bruix, J. Prognosis of Hepatocellular Carcinoma: The BCLC Staging Classification. Semin. Liver Dis. 1999, 19, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Vogel, A.; Martinelli, E.; Vogel, A.; Cervantes, A.; Chau, I.; Daniele, B.; Llovet, J.M.; Meyer, T.; Nault, J.-C.; Neumann, U.; et al. Updated Treatment Recommendations for Hepatocellular Carcinoma (HCC) from the ESMO Clinical Practice Guidelines. Ann. Oncol. 2021, 32, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Reig, M.; Forner, A.; Rimola, J.; Ferrer-Fábrega, J.; Burrel, M.; Garcia-Criado, A.; Kelley, R.K.; Galle, P.R.; Mazzaferro, V.; Salem, R.; et al. BCLC Strategy for Prognosis Prediction and Treatment Recommendation Barcelona Clinic Liver Cancer (BCLC) Staging System. The 2022 Update. J. Hepatol. 2022, 76, 681–693. [Google Scholar] [CrossRef]
- Llovet, J.M.; Hilgard, P.; de Oliveira, A.C.; Forner, A.; Zeuzem, S.; Galle, P.R.; Häussinger, D.; Moscovici, M. Sorafenib in Advanced Hepatocellular Carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [Green Version]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.-H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.-W.; Han, G.; Jassem, J.; et al. Lenvatinib versus Sorafenib in First-Line Treatment of Patients with Unresectable Hepatocellular Carcinoma: A Randomised Phase 3 Non-Inferiority Trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef] [Green Version]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.-H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for Patients with Hepatocellular Carcinoma Who Progressed on Sorafenib Treatment (RESORCE): A Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.-L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.-Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.-W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef]
- Zhu, A.X.; Kang, Y.-K.; Yen, C.-J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after Sorafenib in Patients with Advanced Hepatocellular Carcinoma and Increased α-Fetoprotein Concentrations (REACH-2): A Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Cheng, A.-L.; Qin, S.; Ikeda, M.; Galle, P.; Ducreux, M.; Zhu, A.; Kim, T.-Y.; Kudo, M.; Breder, V.; Merle, P.; et al. IMbrave150: Efficacy and Safety Results from a Ph III Study Evaluating Atezolizumab (Atezo) + Bevacizumab (Bev) vs Sorafenib (Sor) as First Treatment (Tx) for Patients (Pts) with Unresectable Hepatocellular Carcinoma (HCC). Ann. Oncol. 2019, 30, ix186–ix187. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.-Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. JCO 2020, 38, 193–202. [Google Scholar] [CrossRef]
- Yau, T.; Park, J.W.; Finn, R.S.; Cheng, A.-L.; Mathurin, P.; Edeline, J.; Kudo, M.; Han, K.-H.; Harding, J.J.; Merle, P.; et al. CheckMate 459: A Randomized, Multi-Center Phase III Study of Nivolumab (NIVO) vs Sorafenib (SOR) as First-Line (1L) Treatment in Patients (Pts) with Advanced Hepatocellular Carcinoma (AHCC). Ann. Oncol. 2019, 30, v874–v875. [Google Scholar] [CrossRef]
- Finkelmeier, F.; Scheiner, B.; Leyh, C.; Best, J.; Fründt, T.W.; Czauderna, C.; Beutel, A.; Bettinger, D.; Weiß, J.; Meischl, T.; et al. Cabozantinib in Advanced Hepatocellular Carcinoma: Efficacy and Safety Data from an International Multicenter Real-Life Cohort. Liver Cancer 2021, 10, 360–369. [Google Scholar] [CrossRef]
- Welland, S.; Leyh, C.; Finkelmeier, F.; Jefremow, A.; Shmanko, K.; Gonzalez-Carmona, M.A.; Kandulski, A.; Jeliazkova, P.; Best, J.; Fründt, T.; et al. Real-World Data for Lenvatinib in Hepatocellular Carcinoma (ELEVATOR): A Retrospective Multicenter Study. Liver Cancer 2022. [Google Scholar] [CrossRef]
- Hatanaka, T.; Kakizaki, S.; Nagashima, T.; Namikawa, M.; Ueno, T.; Tojima, H.; Takizawa, D.; Naganuma, A.; Arai, H.; Sato, K.; et al. Liver Function Changes in Patients with Hepatocellular Carcinoma Treated with Lenvatinib: Predictive Factors of Progression to Child-Pugh Class B, the Formation of Ascites and the Candidates for the Post-Progression Treatment. Cancers 2020, 12, 2906. [Google Scholar] [CrossRef]
- Hatanaka, T.; Naganuma, A.; Kakizaki, S. Lenvatinib for Hepatocellular Carcinoma: A Literature Review. Pharmaceuticals 2021, 14, 36. [Google Scholar] [CrossRef]
- Yoo, C.; Kim, J.H.; Ryu, M.-H.; Park, S.R.; Lee, D.; Kim, K.M.; Shim, J.H.; Lim, Y.-S.; Lee, H.C.; Lee, J.; et al. Clinical Outcomes with Multikinase Inhibitors after Progression on First-Line Atezolizumab plus Bevacizumab in Patients with Advanced Hepatocellular Carcinoma: A Multinational Multicenter Retrospective Study. Liver Cancer 2021, 10, 107–114. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Atsukawa, M.; Hirooka, M.; Tsuji, K.; Ishikawa, T.; Takaguchi, K.; Kariyama, K.; Itobayashi, E.; Tajiri, K.; et al. Important Clinical Factors in Sequential Therapy Including Lenvatinib against Unresectable Hepatocellular Carcinoma. Oncology 2019, 97, 277–285. [Google Scholar] [CrossRef]
- Kudo, M. Impact of Multi-Drug Sequential Therapy on Survival in Patients with Unresectable Hepatocellular Carcinoma. Liver Cancer 2021, 10, 1–9. [Google Scholar] [CrossRef]
- Ricke, J.; Schinner, R.; Seidensticker, M.; Gasbarrini, A.; van Delden, O.M.; Amthauer, H.; Peynircioglu, B.; Bargellini, I.; Iezzi, R.; De Toni, E.N.; et al. Liver Function after Combined Selective Internal Radiation Therapy or Sorafenib Monotherapy in Advanced Hepatocellular Carcinoma. J. Hepatol. 2021, 75, 1387–1396. [Google Scholar] [CrossRef]
- Vogel, A.; Frenette, C.; Sung, M.; Daniele, B.; Baron, A.; Chan, S.L.; Blanc, J.F.; Tamai, T.; Ren, M.; Lim, H.J.; et al. Baseline Liver Function and Subsequent Outcomes in the Phase 3 REFLECT Study of Patients with Unresectable Hepatocellular Carcinoma. Liver Cancer 2021, 10, 510–521. [Google Scholar] [CrossRef]
- Tovoli, F.; Dadduzio, V.; De Lorenzo, S.; Rimassa, L.; Masi, G.; Iavarone, M.; Marra, F.; Garajova, I.; Brizzi, M.P.; Daniele, B.; et al. Real-Life Clinical Data of Cabozantinib for Unresectable Hepatocellular Carcinoma. Liver Cancer 2021, 10, 370–379. [Google Scholar] [CrossRef]
- Maruta, S.; Ogasawara, S.; Ooka, Y.; Obu, M.; Inoue, M.; Itokawa, N.; Haga, Y.; Seki, A.; Okabe, S.; Azemoto, R.; et al. Potential of Lenvatinib for an Expanded Indication from the REFLECT Trial in Patients with Advanced Hepatocellular Carcinoma. Liver Cancer 2020, 9, 382–396. [Google Scholar] [CrossRef]
- Kim, H.; Bang, Y.; Lee, M.A.; Kim, J.W.; Kim, J.H.; Chon, H.J.; Kang, B.; Kang, M.J.; Kim, I.; Cheon, J.; et al. Regorafenib in Patients with Advanced Child-Pugh B Hepatocellular Carcinoma: A Multicentre Retrospective Study. Liver Int. 2020, 40, 2544–2552. [Google Scholar] [CrossRef]

| All Patients (n = 164) | Sorafenib as 1st Line (n = 93) | Lenvatinib as 1st Line (n = 71) | p Value (SOR vs. LEN) | |
|---|---|---|---|---|
| Age (years), median (IQR) | 68 (60.25–73) | 68 (62–72) | 66 (57–74) | 0.1 |
| Male gender, n (%) | 140 (85) | 81 (87) | 59 (83) | 0.4 |
| ECOG PS a | 0.04 | |||
| 0, n (%) | 92 (57) | 44 (48) | 48 (69) | |
| 1, n (%) | 64 (40) | 44 (48) | 20 (29) | |
| 2, n (%) | 4 (2) | 3 (3) | 1 (1) | |
| 3, n (%) | 1 (1) | 0 (0) | 1 (1) | |
| Liver cirrhosis, n (%) | 123 (75) | 78 (84) | 45 (63) | 0.003 |
| Underlying liver disease b | 0.0003 | |||
| ASH, n (%) | 45 (29) | 36 (40) | 9 (13) | |
| NASH, n (%) | 32 (20) | 17 (19) | 15 (22) | |
| HCV, n (%) | 22 (14) | 14 (16) | 8 (12) | |
| HBV, n (%) | 24 (15) | 10 (11) | 14 (21) | |
| Autoimmune, n (%) | 3 (2) | 2 (2) | 1 (1) | |
| Other or cryptogenic, n (%) | 32 (20) | 11 (12) | 21 (31) | |
| Child-Pugh score b | 0.8 | |||
| CPS A, n (%) | 132 (83.5) | 72 (83) | 60 (84.5) | |
| CPS B, n (%) | 26 (16.5) | 15 (17) | 11 (15.5) | |
| CPS C, n (%) | 0 | 0 | 0 | |
| ALBI grade c | 0.1 | |||
| 1, n (%) | 65 (42) | 33 (39) | 32 (46) | |
| 2, n (%) | 85 (55) | 47 (55) | 38 (54) | |
| 3, n (%) | 5 (3) | 5 (6) | 0 (0) | |
| ALBI score, median (IQR) c | −2.47 (−2.91 to −2.06) | −2.40 (−2.87 to −1.93) | −2.54 (−3.00 to −2.14) | 0.46 |
| Albumin (g/dl), median (IQR) c | 3.9 (3.5–4.2) | 3.8 (3.4–4.2) | 4.0 (3.6–4.3) | 0.08 |
| Bilirubin (mg/dl), median (IQR) b | 1.0 (0.6–1.6) | 1.0 (0.6–1.7) | 0.9 (0.6–1.5) | 0.5 |
| Ascites, n (%) d | 40 (25) | 22 (24) | 18 (25) | 0.8 |
| Refractory ascites, n (%) | 7 (4) | 3 (3) | 4 (6) | 1.0 |
| Esophageal varices, n (%) a | 59 (37) | 35 (39) | 24 (34) | 0.4 |
| Tumor stage e | 0.4 | |||
| BCLC A, n (%) | 1 (1) | 0 (0) | 1 (1) | |
| BCLC B, n (%) | 57 (35) | 35 (38) | 22 (32) | |
| BCLC C, n (%) | 104 (63) | 57 (61) | 47 (67) | |
| BCLC D, n (%) | 1 (1) | 1 (1) | 0 | |
| Macrovascular invasion, n (%) | 49 (30) | 23 (25) | 26 (37) | 0.1 |
| Extrahepatic tumor manifestations, n (%) | 80 (49) | 43 (46) | 37 (52) | 0.5 |
| Prior locoregional therapy, n (%) | 106 (65) | 60 (65) | 46 (65) | 0.9 |
| Resection, n (%) | 29 (18) | 16 (17) | 13 (18) | 0.9 |
| TACE, n (%) | 55 (34) | 30 (32) | 25 (35) | 0.7 |
| TARE, n (%) | 55 (34) | 32 (34) | 23 (32) | 0.8 |
| Sorafenib (n = 88) | Lenvatinib (n = 66) | |
|---|---|---|
| 1 therapy line | n = 40 | n = 32 |
| Progress, n (%) | 9 (22.5) | 3 (9) |
| Intolerance/Toxicity, n (%) | 18 (45) | 16 (50) |
| Of that, liver function deterioration, n (%) | 13 (72) | 8 (50) |
| Of that, other adverse events, n (%) | 5 (28) | 8 (50) |
| Progress + intolerance, n (%) | 5 (12.5) | 3 (9) |
| Death, n (%) | 6 (15) | 9 (28) |
| Other, n (%) | 2 (5) | 1 (3) |
| >1 therapy line | n = 48 | n = 34 |
| Progress, n (%) | 25 (52) | 20 (59) |
| Intolerance/Toxicity, n (%) | 17 (35) | 9 (26) |
| Of that, liver function deterioration, n (%) | 6 (35) | 3 (33) |
| Of that, other adverse events, n (%) | 11 (65) | 6 (67) |
| Progress + intolerance, n (%) | 5 (10) | 4 (12) |
| Other, n (%) | 1 (2) | 1 (3) |
| Sorafenib as 1st-Line (n = 93) | Lenvatinib as 1st-Line (n = 71) | p Value | |
|---|---|---|---|
| First-line, days, median (IQR) | 99.5 (47–199.5), n = 88 | 120 (44–202.5), n = 65 | 0.3 |
| Second-line, days, median (IQR) | 112 (57–175), n = 43 | 92 (37–201.5), n = 29 | 0.3 |
| Third-line, days, median (IQR) | 87 (26–143.8), n = 12 | 109 (27.25–192.8), n = 12 | 0.4 |
| Entire therapy, whole cohort, median (IQR) | 175.5 (80.5–333), n = 74 | 155 (61.5–395.5), n = 57 | 0.8 |
| Entire therapy, patients with 2 or more systemic therapies, median (IQR) | 262 (182–434), n = 39 | 303 (195.5–581.5), n = 26 | 0.7 |
| Liver Function at Beginning of First-Line Therapy with Sorafenib | Liver Function at the End of First-Line Therapy with Sorafenib | Liver Function at the End of the Entire Sequential Therapy If the Sequence Started with Sorafenib | ||||
|---|---|---|---|---|---|---|
| Child-Pugh Score | ||||||
| n= 87 | n = 77 | n = 62 | ||||
| A5, n (%) | 47 (54) | 72 (83) | 30 (39) | 37 (48) | 12 (19) | 16 (26) |
| A6, n (%) | 25 (29) | 7 (9) | 4 (7) | |||
| B7, n (%) | 9 (10) | 15 (17) | 13 (17) | 32 (42) | 9 (15) | 32 (52) |
| B8, n (%) | 4 (5) | 9 (12) | 8 (13) | |||
| B9, n (%) | 2 (2) | 10 (13) | 15 (24) | |||
| C, n (%) | 0 | 0 | 8 (10) | 8 (10) | 14 (23) | 14 (23) |
| ALBI Grade/ALBI Score | ||||||
| n = 87 | n = 77 | n = 62 | ||||
| 1, n (%) | 35 (40) | 14 (18) | 4 (6.5) | |||
| 2, n (%) | 47 (54) | 35 (46) | 23 (37) | |||
| 3, n (%) | 5 (6) | 28 (36) | 35 (56.5) | |||
| ALBI score, median (IQR) | −2.40 (−2.87 to −1.93) | −1.67 (−2.48 to −1.15) | −1.33 (−1.85 to −0.71) | |||
| Liver Function at the Beginning of First-Line Therapy with Lenvatinib | Liver Function at the End of First-Line Therapy with Lenvatinib | Liver Function at the End of the Entire Sequential Therapy if the Sequence Started with Lenvatinib | ||||
| Child-Pugh score | ||||||
| n = 71 | n = 56 | n = 35 | ||||
| A5, n (%) | 45 (63) | 60 (85) | 23 (41) | 34 (61) | 5 (14) | 11 (31) |
| A6, n (%) | 15 (21) | 11 (20) | 6 (17) | |||
| B7, n (%) | 6 (9) | 11 (15) | 6 (11) | 16 (29) | 7 (20) | 19 (54) |
| B8, n (%) | 4 (6) | 6 (11) | 8 (23) | |||
| B9, n (%) | 1 (1) | 4 (7) | 4 (11) | |||
| C, n (%) | 0 | 0 | 6 (11) | 6 (11) | 5 (14) | 5 (14) |
| ALBI grade/ALBI score | ||||||
| n = 71 | n = 56 | n = 38 | ||||
| 1, n (%) | 33 (46.5) | 12 (21) | 4 (10.5) | |||
| 2, n (%) | 38 (56.3) | 27 (48) | 19 (50) | |||
| 3, n (%) | 0 | 17 (30) | 15 (39.5) | |||
| ALBI score, median (IQR) | −2.54 (−3.00 to −2.14) | −1.89 (−2.46 to −1.21) | −1.74 (−2–23 to −0.88) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leyh, C.; Ehmer, U.; Roessler, D.; Philipp, A.B.; Reiter, F.P.; Jeliazkova, P.; Jochheim, L.S.; Jeschke, M.; Hammig, J.; Ludwig, J.M.; et al. Sorafenib Versus Lenvatinib-Based Sequential Systemic Therapy for Advanced Hepatocellular Carcinoma: A Real-World Analysis. Cancers 2022, 14, 1975. https://doi.org/10.3390/cancers14081975
Leyh C, Ehmer U, Roessler D, Philipp AB, Reiter FP, Jeliazkova P, Jochheim LS, Jeschke M, Hammig J, Ludwig JM, et al. Sorafenib Versus Lenvatinib-Based Sequential Systemic Therapy for Advanced Hepatocellular Carcinoma: A Real-World Analysis. Cancers. 2022; 14(8):1975. https://doi.org/10.3390/cancers14081975
Chicago/Turabian StyleLeyh, Catherine, Ursula Ehmer, Daniel Roessler, Alexander B. Philipp, Florian P. Reiter, Petia Jeliazkova, Leonie S. Jochheim, Matthias Jeschke, Janina Hammig, Johannes M. Ludwig, and et al. 2022. "Sorafenib Versus Lenvatinib-Based Sequential Systemic Therapy for Advanced Hepatocellular Carcinoma: A Real-World Analysis" Cancers 14, no. 8: 1975. https://doi.org/10.3390/cancers14081975
APA StyleLeyh, C., Ehmer, U., Roessler, D., Philipp, A. B., Reiter, F. P., Jeliazkova, P., Jochheim, L. S., Jeschke, M., Hammig, J., Ludwig, J. M., Theysohn, J. M., Geier, A., & Lange, C. M. (2022). Sorafenib Versus Lenvatinib-Based Sequential Systemic Therapy for Advanced Hepatocellular Carcinoma: A Real-World Analysis. Cancers, 14(8), 1975. https://doi.org/10.3390/cancers14081975






