Combined Fluorescence-Guided Resection and Intracavitary Thermotherapy with Superparamagnetic Iron-Oxide Nanoparticles for Recurrent High-Grade Glioma: Case Series with Emphasis on Complication Management
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
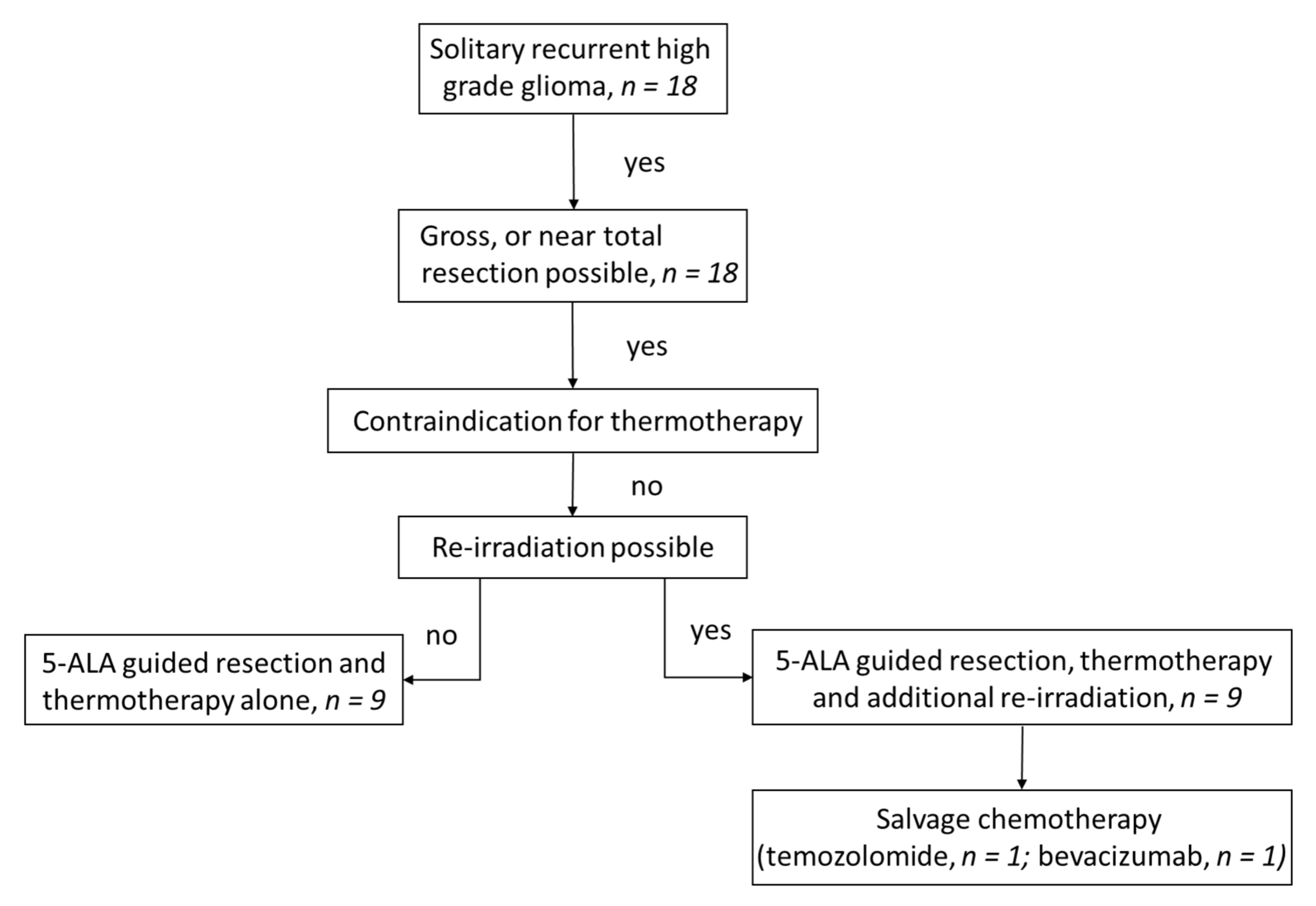
2.1. Patients
2.2. Nanoparticles
2.3. Fluorescence-Guided Resection and Nanoparticle Application
2.4. Thermotherapy
2.5. Follow Up
2.6. Data Analyses
3. Results
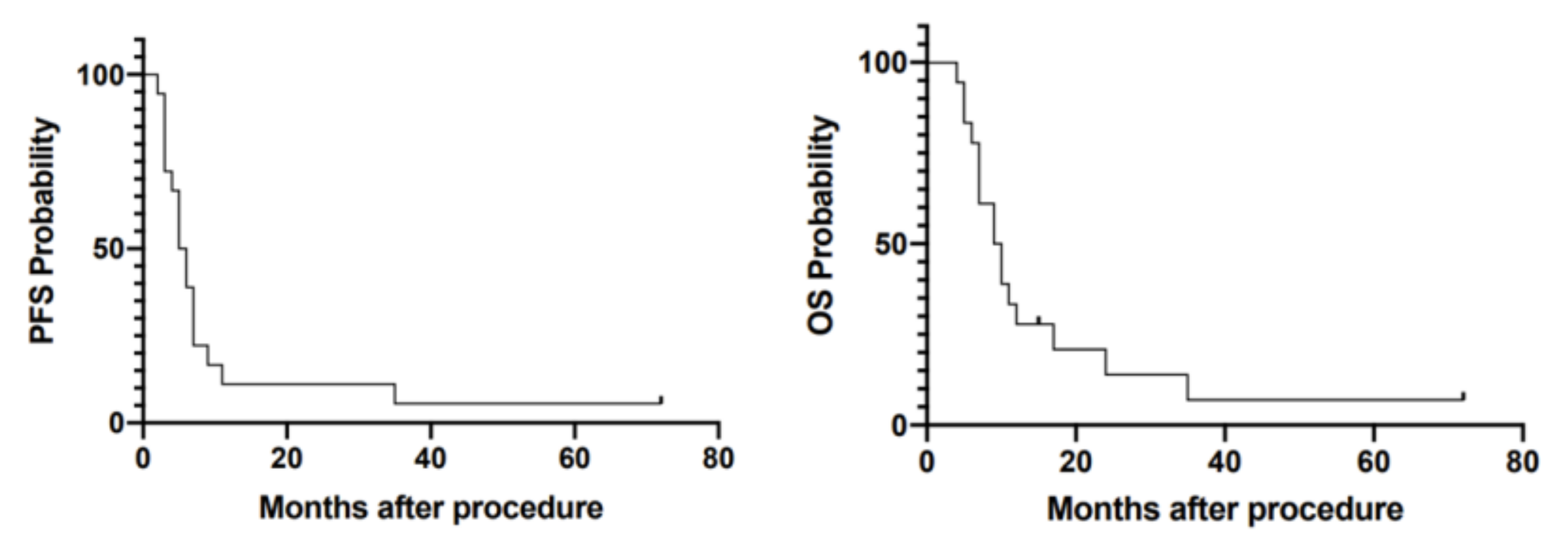
3.1. Survival Analysis
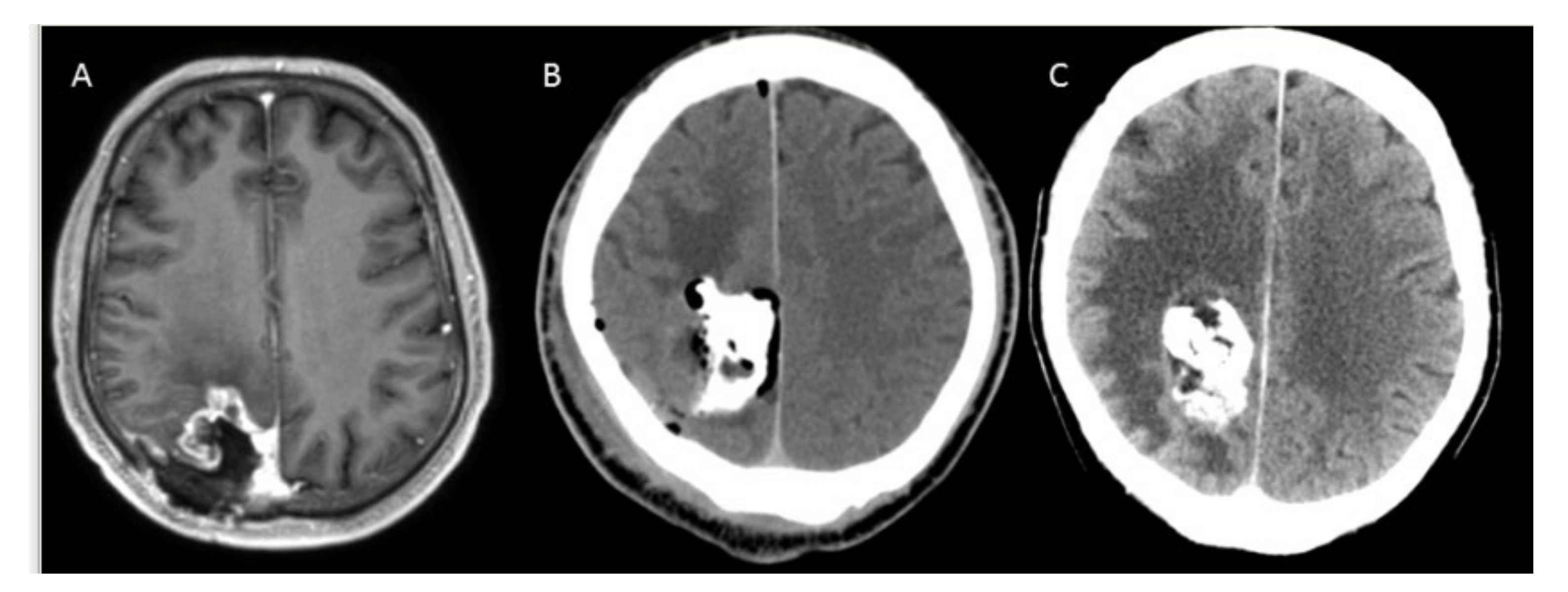
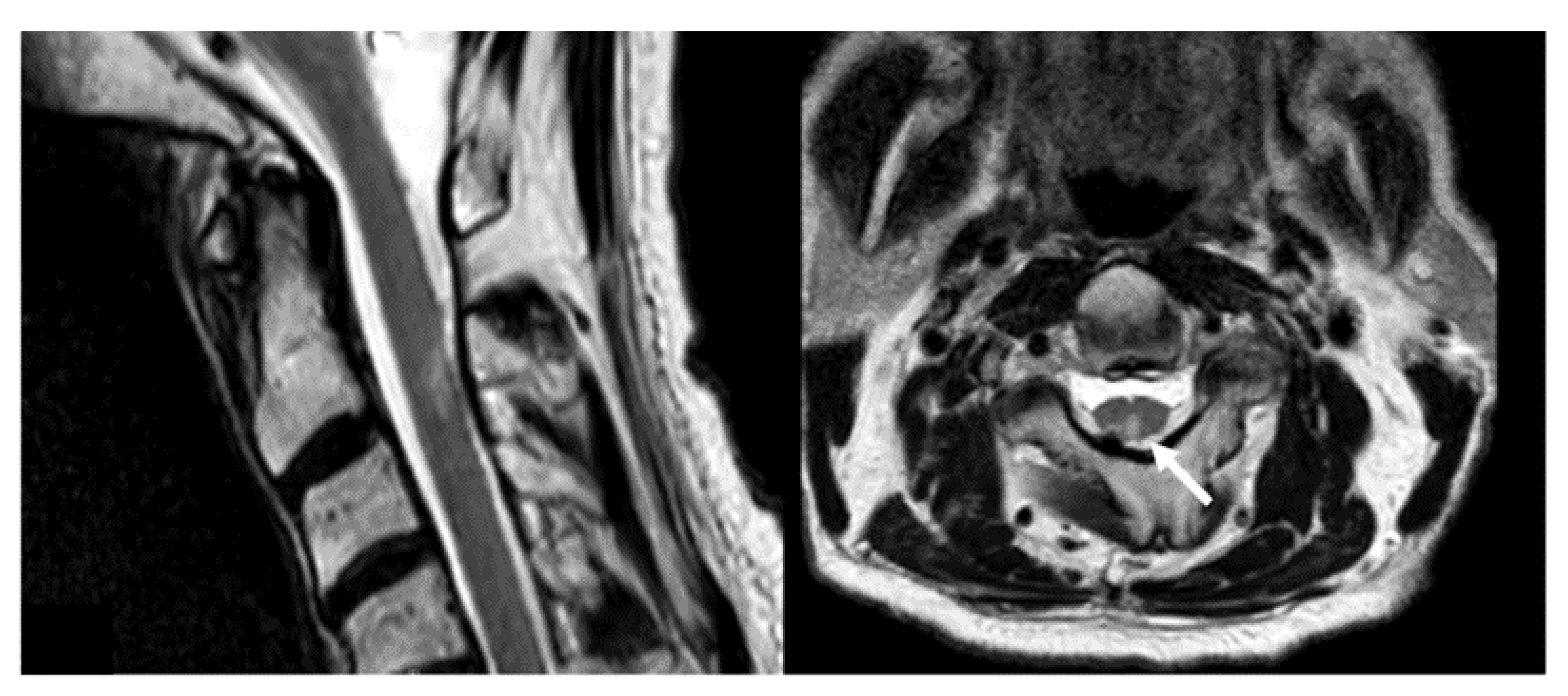
3.2. Safety and Complications
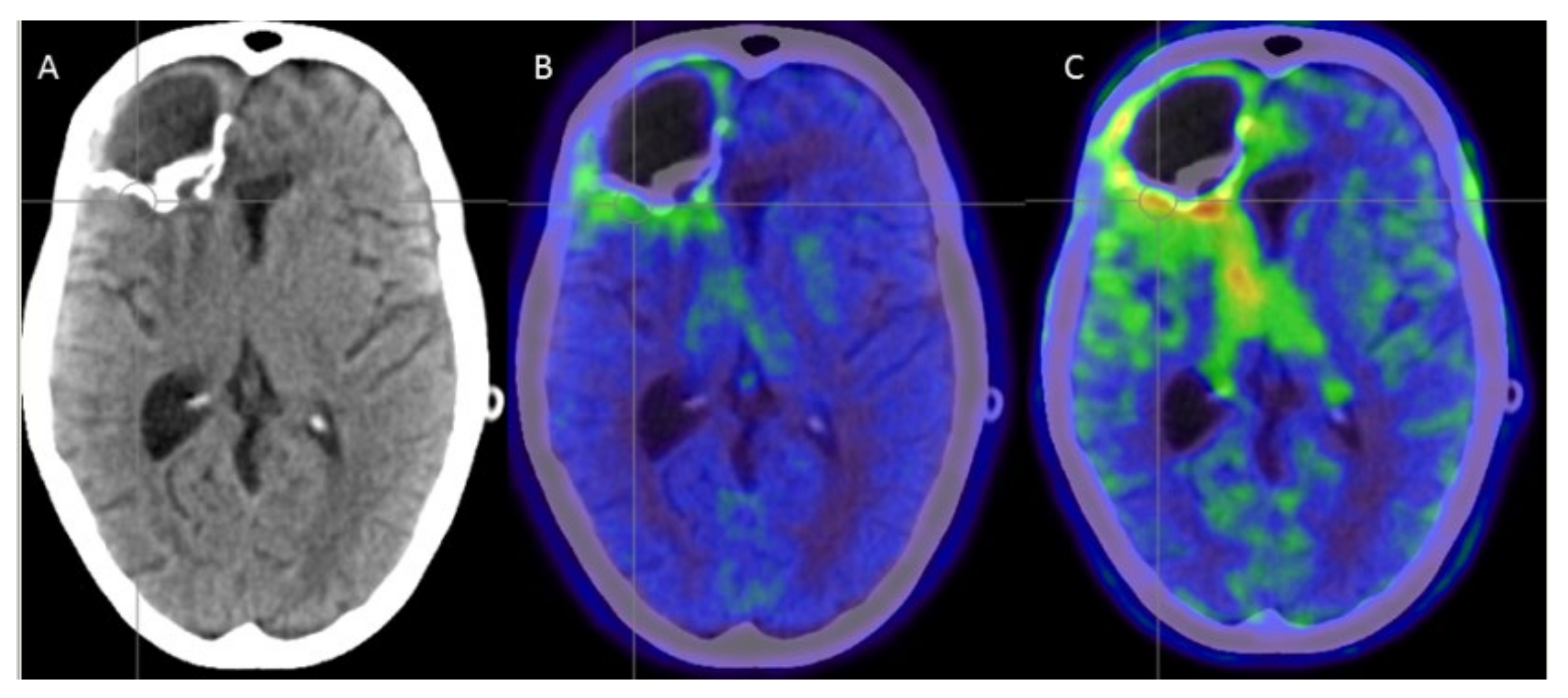
4. Discussion
4.1. Combining FGR with Application of Nanoparticles
4.2. Complication Management
4.3. Outlook
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Goldbrunner, R.; Minniti, G.; Preusser, M.; Jenkinson, M.D.; Sallabanda, K.; Houdart, E.; von Deimling, A.; Stavrinou, P.; Lefranc, F.; Lund-Johansen, M.; et al. EANO guidelines for the diagnosis and treatment of meningiomas. Lancet Oncol. 2016, 17, e383–e391. [Google Scholar] [CrossRef] [Green Version]
- Stummer, W.; Pichlmeier, U.; Meinel, T.; Wiestler, O.D.; Zanella, F.; Reulen, H.-J.; ALA-Glioma Study Group. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: A randomised controlled multicentre phase III trial. Lancet Oncol. 2006, 7, 392–401. [Google Scholar] [CrossRef]
- Stummer, W.; Novotny, A.; Stepp, H.; Goetz, C.; Bise, K.; Reulen, H.J. Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: A prospective study in 52 consecutive patients. J. Neurosurg. 2000, 93, 1003–1013. [Google Scholar] [CrossRef]
- Schucht, P.; Beck, J.; Abu-Isa, J.; Andereggen, L.; Murek, M.; Seidel, K.; Stieglitz, L.; Raabe, A. Gross Total Resection Rates in Contemporary Glioblastoma Surgery. Neurosurgery 2012, 71, 927–936. [Google Scholar] [CrossRef] [Green Version]
- Seidel, K.; Beck, J.; Stieglitz, L.; Schucht, P.; Raabe, A. The warning-sign hierarchy between quantitative subcortical motor mapping and continuous motor evoked potential monitoring during resection of supratentorial brain tumors; Clinical article. J. Neurosurg. 2013, 118, 287–296. [Google Scholar] [CrossRef] [Green Version]
- Hadani, M.; Spiegelman, R.; Feldman, Z.; Berkenstadt, H.; Ram, Z. Novel, compact, intraoperative magnetic resonance imaging-guided system for conventional neurosurgical operating rooms. Neurosurgery 2001, 48, 799–807; discussion 807–809. [Google Scholar]
- Eyüpoglu, I.Y.; Hore, N.; Savaskan, N.E.; Grummich, P.; Roessler, K.; Buchfelder, M.; Ganslandt, O. Improving the Extent of Malignant Glioma Resection by Dual Intraoperative Visualization Approach. PLoS ONE 2012, 7, e44885. [Google Scholar] [CrossRef]
- Stupp, R.; Taillibert, S.; Kanner, A.; Read, W.; Steinberg, D.M.; Lhermitte, B.; Toms, S.; Idbaih, A.; Ahluwalia, M.S.; Fink, K.; et al. Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma a randomized clinical trial. JAMA J. Am. Med. Assoc. 2017, 318, 2306–2316. [Google Scholar] [CrossRef] [Green Version]
- Díez Valle, R.; Slof, J.; Galván, J.; Arza, C.; Romariz, C.; Vidal, C. Observational, retrospective study of the effectiveness of 5-aminolevulinic acid in malignant glioma surgery in Spain (The VISIONA study). Neurologia 2014, 29, 131–138. [Google Scholar] [CrossRef]
- Aldave, G.; Tejada, S.; Pay, E.; Marigil, M.; Bejarano, B.; Idoate, M.A.; Díez-Valle, R. Prognostic value of residual fluorescent tissue in glioblastoma patients after gross total resection in 5-aminolevulinic Acid-guided surgery. Neurosurgery 2013, 72, 915–920; discussion 920–921. [Google Scholar] [CrossRef]
- Stummer, W.; Reulen, H.-J.J.; Meinel, T.; Pichlmeier, U.; Schumacher, W.; Tonn, J.-C.C.; Rohde, V.; Oppel, F.; Turowski, B.; Woiciechowsky, C.; et al. Extent of resection and survival in glioblastoma multiforme: Identification of and adjustment for bias. Neurosurgery 2008, 62, 564–576; discussion 564–576. [Google Scholar] [CrossRef]
- Valdés, P.A.; Samkoe, K.; O’Hara, J.A.; Roberts, D.W.; Paulsen, K.D.; Pogue, B.W. Deferoxamine iron chelation increases delta-aminolevulinic acid induced protoporphyrin IX in xenograft glioma model. Photochem. Photobiol. 2010, 86, 471–475. [Google Scholar] [CrossRef] [Green Version]
- Valdés, P.A.; Leblond, F.; Kim, A.; Harris, B.T.; Wilson, B.C.; Fan, X.; Tosteson, T.D.; Hartov, A.; Ji, S.; Erkmen, K.; et al. Quantitative fluorescence in intracranial tumor: Implications for ALA-induced PpIX as an intraoperative biomarker. J. Neurosurg. 2011, 115, 11–17. [Google Scholar] [CrossRef] [Green Version]
- Stummer, W.; Tonn, J.-C.; Goetz, C.; Ullrich, W.; Stepp, H.; Bink, A.; Pietsch, T.; Pichlmeier, U. 5-Aminolevulinic Acid-derived Tumor Fluorescence. Neurosurgery 2014, 74, 310–320. [Google Scholar] [CrossRef] [Green Version]
- Hefti, M.; von Campe, G.; Moschopulos, M.; Siegner, A.; Looser, H.; Landolt, H. 5-Aminolevulinic acid induced protoporphyrin IX fluorescence in high-grade glioma surgery: A one-year experience at a single institutuion. Swiss Med. Wkly. 2008, 138, 180–185. [Google Scholar] [CrossRef]
- Cage, T.A.; Pekmezci, M.; Prados, M.; Berger, M.S. Subependymal spread of recurrent glioblastoma detected with the intraoperative use of 5-aminolevulinic acid: Case report. J. Neurosurg. 2013, 118, 1220–1223. [Google Scholar] [CrossRef]
- Müther, M.; Koch, R.; Weckesser, M.; Sporns, P.; Schwindt, W.; Stummer, W. 5-Aminolevulinic Acid Fluorescence-Guided Resection of 18F-FET-PET Positive Tumor Beyond Gadolinium Enhancing Tumor Improves Survival in Glioblastoma. Neurosurgery 2019, 85, E1020–E1029. [Google Scholar] [CrossRef] [Green Version]
- Nabavi, A.; Thurm, H.; Zountsas, B.; Pietsch, T.; Lanfermann, H.; Pichlmeier, U.; Mehdorn, M.; 5-ALA Recurrent Glioma Study Group. Five-aminolevulinic acid for fluorescence-guided resection of recurrent malignant gliomas: A phase ii study. Neurosurgery 2009, 65, 1070–1076; discussion 1076–1077. [Google Scholar] [CrossRef]
- Weller, M.; van den Bent, M.; Tonn, J.C.; Stupp, R.; Preusser, M.; Cohen-Jonathan-Moyal, E.; Henriksson, R.; Le Rhun, E.; Balana, C.; Chinot, O.; et al. European Association for Neuro-Oncology (EANO) guideline on the diagnosis and treatment of adult astrocytic and oligodendroglial gliomas. Lancet Oncol. 2017, 18, e315–e329. [Google Scholar] [CrossRef] [Green Version]
- Desjardins, A.; Gromeier, M.; Herndon, J.E.; Beaubier, N.; Bolognesi, D.P.; Friedman, A.H.; Friedman, H.S.; McSherry, F.; Muscat, A.M.; Nair, S.; et al. Recurrent Glioblastoma Treated with Recombinant Poliovirus. N. Engl. J. Med. 2018, 379, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Wick, W.; Gorlia, T.; Bendszus, M.; Taphoorn, M.; Sahm, F.; Harting, I.; Brandes, A.A.; Taal, W.; Domont, J.; Idbaih, A.; et al. Lomustine and bevacizumab in progressive glioblastoma. N. Engl. J. Med. 2017, 377, 1954–1963. [Google Scholar] [CrossRef] [PubMed]
- Schipmann, S.; Müther, M.; Stögbauer, L.; Zimmer, S.; Brokinkel, B.; Holling, M.; Grauer, O.; Suero Molina, E.; Warneke, N.; Stummer, W. Combination of ALA-induced fluorescence-guided resection and intraoperative open photodynamic therapy for recurrent glioblastoma: Case series on a promising dual strategy for local tumor control. J. Neurosurg. 2020, 134, 426–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallner, K.E.; Galicich, J.H.; Krol, G.; Arbit, E.; Malkin, M.G. Patterns of failure following treatment for glioblastoma multiforme and anaplastic astrocytoma. Int. J. Radiat. Oncol. Biol. Phys. 1989, 16, 1405–1409. [Google Scholar] [CrossRef]
- Westphal, M.; Hilt, D.C.; Bortey, E.; Delavault, P.; Olivares, R.; Warnke, P.C.; Whittle, I.R.; Jääskeläinen, J.; Ram, Z. A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro. Oncol. 2003, 5, 79–88. [Google Scholar] [CrossRef]
- Stepp, H.; Beck, T.; Pongratz, T.; Meinel, T.; Kreth, F.-W.; Tonn, J.C.; Stummer, W. ALA and malignant glioma: Fluorescence-guided resection and photodynamic treatment. J. Environ. Pathol. Toxicol. Oncol. 2007, 26, 157–164. [Google Scholar] [CrossRef]
- Maier-Hauff, K.; Ulrich, F.; Nestler, D.; Niehoff, H.; Wust, P.; Thiesen, B.; Orawa, H.; Budach, V.; Jordan, A. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neurooncol. 2011, 103, 317–324. [Google Scholar] [CrossRef] [Green Version]
- Maier-Hauff, K.; Rothe, R.; Scholz, R.; Gneveckow, U.; Wust, P.; Thiesen, B.; Feussner, A.; Deimling, A.; Waldoefner, N.; Felix, R.; et al. Intracranial thermotherapy using magnetic nanoparticles combined with external beam radiotherapy: Results of a feasibility study on patients with glioblastoma multiforme. J. Neurooncol. 2007, 81, 53–60. [Google Scholar] [CrossRef]
- Yanase, M.; Shinkai, M.; Honda, H.; Wakabayashi, T.; Yoshida, J.; Kobayashi, T. Antitumor Immunity Induction by Intracellular Hyperthermia Using Magnetite Cationic Liposomes. Jpn. J. Cancer Res. 1998, 89, 775–782. [Google Scholar] [CrossRef]
- Ito, A.; Shinkai, M.; Honda, H.; Yoshikawa, K.; Saga, S.; Wakabayashi, T.; Yoshida, J.; Kobayashi, T. Heat shock protein 70 expression induces antitumor immunity during intracellular hyperthermia using magnetite nanoparticles. Cancer Immunol. Immunother. 2003, 52, 80–88. [Google Scholar] [CrossRef]
- Tanaka, K.; Ito, A.; Kobayashi, T.; Kawamura, T.; Shimada, S.; Matsumoto, K.; Saida, T.; Honda, H. Intratumoral injection of immature dendritic cells enhances antitumor effect of hyperthermia using magnetic nanoparticles. Int. J. Cancer 2005, 116, 624–633. [Google Scholar] [CrossRef]
- Grauer, O.; Jaber, M.; Hess, K.; Weckesser, M.; Schwindt, W.; Maring, S.; Wölfer, J.; Stummer, W. Combined intracavitary thermotherapy with iron oxide nanoparticles and radiotherapy as local treatment modality in recurrent glioblastoma patients. J. Neurooncol. 2019, 141, 83–94. [Google Scholar] [CrossRef] [Green Version]
- Molina, E.S.; Schipmann, S.; Mueller, I.; Wölfer, J.; Ewelt, C.; Maas, M.; Brokinkel, B.; Stummer, W. Conscious sedation with dexmedetomidine compared with asleep-awake-asleep craniotomies in glioma surgery: An analysis of 180 patients. J. Neurosurg. 2018, 129, 1223–1230. [Google Scholar] [CrossRef] [Green Version]
- Plotkin, M.; Gneveckow, U.; Meier-Hauff, K.; Amthauer, H.; Feußner, A.; Denecke, T.; Gutberlet, M.; Jordan, A.; Felix, R.; Wust, P. 18F-FET PET for planning of thermotherapy using magnetic nanoparticles in recurrent glioblastoma. Int. J. Hyperth. 2006, 22, 319–325. [Google Scholar] [CrossRef]
- Yoo, T.S.; Ackerman, M.J.; Lorensen, W.E.; Schroeder, W.; Chalana, V.; Aylward, S.; Metaxas, D.; Whitaker, R. Engineering and algorithm design for an image processing API: A technical report on ITK—The Insight Toolkit. Stud. Health Technol. Inform. 2002, 85, 586–592. [Google Scholar] [CrossRef]
- Gneveckow, U.; Jordan, A.; Scholz, R.; Brüss, V.; Waldöfner, N.; Ricke, J.; Feussner, A.; Hildebrandt, B.; Rau, B.; Wust, P. Description and characterization of the novel hyperthermia- and thermoablation-system MFH 300F for clinical magnetic fluid hyperthermia. Med. Phys. 2004, 31, 1444–1451. [Google Scholar] [CrossRef]
- Zinnhardt, B.; Müther, M.; Roll, W.; Backhaus, P.; Jeibmann, A.; Foray, C.; Barca, C.; Döring, C.; Tavitian, B.; Dollé, F.; et al. TSPO imaging-guided characterization of the immunosuppressive myeloid tumor microenvironment in patients with malignant glioma. Neuro. Oncol. 2020, 22, 1030–1043. [Google Scholar] [CrossRef]
- Wen, P.Y.; Macdonald, D.R.; Reardon, D.A.; Cloughesy, T.F.; Sorensen, A.G.; Galanis, E.; DeGroot, J.; Wick, W.; Gilbert, M.R.; Lassman, A.B.; et al. Updated response assessment criteria for high-grade gliomas: Response assessment in neuro-oncology working group. J. Clin. Oncol. 2010, 28, 1963–1972. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [Green Version]
- Issels, R.D.; Lindner, L.H.; Verweij, J.; Wust, P.; Reichardt, P.; Schem, B.C.; Abdel-Rahman, S.; Daugaard, S.; Salat, C.; Wendtner, C.M.; et al. Neo-adjuvant chemotherapy alone or with regional hyperthermia for localised high-risk soft-tissue sarcoma: A randomised phase 3 multicentre study. Lancet Oncol. 2010, 11, 561–570. [Google Scholar] [CrossRef] [Green Version]
- Thiesen, B.; Jordan, A. Clinical applications of magnetic nanoparticles for hyperthermia. Int. J. Hyperth. 2008, 24, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Van Der Zee, J.; González González, D.; Van Rhoon, G.C.; Van Dijk, J.D.P.; Van Putten, W.L.J.; Hart, A.A.M. Comparison of radiotherapy alone with radiotherapy plus hyperthermia in locally advanced pelvic tumours: A prospective, randomised, multicentre trial. Lancet 2000, 355, 1119–1125. [Google Scholar] [CrossRef]
- Ringel, F.; Pape, H.; Sabel, M.; Krex, D.; Bock, H.C.; Misch, M.; Weyerbrock, A.; Westermaier, T.; Senft, C.; Schucht, P.; et al. Clinical benefit from resection of recurrent glioblastomas: Results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro. Oncol. 2016, 18, 96–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaichana, K.L.; Zadnik, P.; Weingart, J.D.; Olivi, A.; Gallia, G.L.; Blakeley, J.; Lim, M.; Brem, H.; Quiñones-Hinojosa, A. Multiple resections for patients with glioblastoma: Prolonging survival. J. Neurosurg. 2013, 118, 812–820. [Google Scholar] [CrossRef] [Green Version]
- Helseth, R.; Helseth, E.; Johannesen, T.B.; Langberg, C.W.; Lote, K.; Rønning, P.; Scheie, D.; Vik, A.; Meling, T.R. Overall survival, prognostic factors, and repeated surgery in a consecutive series of 516 patients with glioblastoma multiforme. Acta Neurol. Scand. 2010, 122, 159–167. [Google Scholar] [CrossRef]
- Suchorska, B.; Weller, M.; Tabatabai, G.; Senft, C.; Hau, P.; Sabel, M.C.; Herrlinger, U.; Ketter, R.; Schlegel, U.; Marosi, C.; et al. Complete resection of contrast-enhancing tumor volume is associated with improved survival in recurrent glioblastoma-results from the DIRECTOR trial. Neuro. Oncol. 2016, 18, 549–556. [Google Scholar] [CrossRef]
- Shi, W.; Scannell Bryan, M.; Gilbert, M.R.; Mehta, M.P.; Blumenthal, D.T.; Brown, P.D.; Valeinis, E.; Hopkins, K.; Souhami, L.; Andrews, D.W.; et al. Investigating the Effect of Reirradiation or Systemic Therapy in Patients With Glioblastoma After Tumor Progression: A Secondary Analysis of NRG Oncology/Radiation Therapy Oncology Group Trial 0525. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 38–44. [Google Scholar] [CrossRef]
- Perry, J.R.; Bélanger, K.; Mason, W.P.; Fulton, D.; Kavan, P.; Easaw, J.; Shields, C.; Kirby, S.; Macdonald, D.R.; Eisenstat, D.D.; et al. Phase II trial of continuous dose-intense temozolomide in recurrent malignant glioma: RESCUE study. J. Clin. Oncol. 2010, 28, 2051–2057. [Google Scholar] [CrossRef]
- Weller, M.; Tabatabai, G.; Kästner, B.; Felsberg, J.; Steinbach, J.P.; Wick, A.; Schnell, O.; Hau, P.; Herrlinger, U.; Sabel, M.C.; et al. MGMT Promoter Methylation Is a Strong Prognostic Biomarker for Benefit from Dose-Intensified Temozolomide Rechallenge in Progressive Glioblastoma: The DIRECTOR Trial. Clin. Cancer Res. 2015, 21, 2057–2064. [Google Scholar] [CrossRef] [Green Version]
- Batchelor, T.T.; Mulholland, P.; Neyns, B.; Nabors, L.B.; Campone, M.; Wick, A.; Mason, W.; Mikkelsen, T.; Phuphanich, S.; Ashby, L.S.; et al. Phase III randomized trial comparing the efficacy of cediranib as monotherapy, and in combination with lomustine, versus lomustine alone in patients with recurrent glioblastoma. J. Clin. Oncol. 2013, 31, 3212–3218. [Google Scholar] [CrossRef] [Green Version]
- Friedman, H.S.; Prados, M.D.; Wen, P.Y.; Mikkelsen, T.; Schiff, D.; Abrey, L.E.; Yung, W.K.A.; Paleologos, N.; Nicholas, M.K.; Jensen, R.; et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J. Clin. Oncol. 2009, 27, 4733–4740. [Google Scholar] [CrossRef] [Green Version]
- Lombardi, G.; De Salvo, G.L.; Brandes, A.A.; Eoli, M.; Rudà, R.; Faedi, M.; Lolli, I.; Pace, A.; Daniele, B.; Pasqualetti, F.; et al. Regorafenib compared with lomustine in patients with relapsed glioblastoma (REGOMA): A multicentre, open-label, randomised, controlled, phase 2 trial. Lancet. Oncol. 2019, 20, 110–119. [Google Scholar] [CrossRef]
- Diaz, R.; Ivan, M.E.; Hanft, S.; Vanni, S.; Manzano, G.; Jagid, J.; Komotar, R.J. Laser Interstitial Thermal Therapy. Neurosurgery 2016, 79, S3–S7. [Google Scholar] [CrossRef] [Green Version]
- Munoz-Casabella, A.; Alvi, M.A.; Rahman, M.; Burns, T.C.; Brown, D.A. Laser Interstitial Thermal Therapy for Recurrent Glioblastoma: Pooled Analyses of Available Literature. World Neurosurg. 2021, 153, 91–97.e1. [Google Scholar] [CrossRef]
- Chohan, M.O.; Berger, M.S. 5-Aminolevulinic acid fluorescence guided surgery for recurrent high-grade gliomas. J. Neurooncol. 2019, 141, 517–522. [Google Scholar] [CrossRef]
- Levin, V.A.; Bidaut, L.; Hou, P.; Kumar, A.J.; Wefel, J.S.; Bekele, B.N.; Prabhu, S.; Loghin, M.; Gilbert, M.R.; Jackson, E.F. Randomized double-blind placebo-controlled trial of bevacizumab therapy for radiation necrosis of the central nervous system. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1487–1495. [Google Scholar] [CrossRef] [Green Version]
- Haider, A.S.; van den Bent, M.; Wen, P.Y.; Vogelbaum, M.A.; Chang, S.; Canoll, P.D.; Horbinski, C.M.; Huse, J.T. Toward a standard pathological and molecular characterization of recurrent glioma in adults: A Response Assessment in Neuro-Oncology effort. Neuro. Oncol. 2020, 22, 450–456. [Google Scholar] [CrossRef]
| ID | Age (Years) | Sex | Initial Diagnosis | Time Since Initial Diagnosis (Months) | Number of Recurrences | Current Diagnosis | MGMT Promotor | Location |
|---|---|---|---|---|---|---|---|---|
| 1 | 42 | F | Glioblastoma, IDH mutated | 6 | 1 | Glioblastoma, IDH mutated | methylated | LP |
| 2 | 60 | M | Glioblastoma, IDH wild type | 13 | 2 | Glioblastoma, IDH wild type | unmethylated | RP |
| 3 | 59 | M | Glioblastoma, IDH wild type | 15 | 1 | Glioblastoma, IDH wild type | unmethylated | LP |
| 4 | 65 | M | Glioblastoma, IDH wild type | 8 | 2 | Glioblastoma, IDH wild type | unmethylated | RP |
| 5 | 75 | F | Glioblastoma, IDH wild type | 4 | 1 | Glioblastoma, IDH wild type | unmethylated | RF |
| 6 | 42 | M | Glioblastoma, IDH wild type | 3 | 4 | Glioblastoma, IDH wild type | methylated | LF |
| 7 | 63 | F | Glioblastoma, IDH wild type | 13 | 1 | Glioblastoma, IDH wildtype | unmethylated | LP |
| 8 | 36 | F | Anaplastic astrocytoma, IDH mutated | 57 | 2 | Anaplastic astrocytoma, IDH mutated | unmethylated | RF |
| 9 | 38 | M | Glioblastoma, IDH wildtype | 42 | 2 | Glioblastoma, IDH wildtype | unmethylated | LO |
| 10 | 62 | M | Glioblastoma, IDH wildtype | 7 | 1 | Glioblastoma, IDH wildtype | unmethylated | LO |
| 11 | 42 | F | Glioblastoma, IDH wildtype | 33 | 1 | Glioblastoma, IDH wildtype | unmethylated | LO |
| 12 | 40 | F | Diffuse Astrocytoma, IDH mutated | 248 | 3 | Anaplastic Astrocytoma, IDH mutated | methylated | RF |
| 13 | 58 | M | Glioblastoma, IDH wildtype | 6 | 1 | Glioblastoma, IDH wildtype | unmethylated | RP |
| 14 | 44 | F | Glioblastoma, IDH wildtype | 8 | 1 | Glioblastoma, IDH wildtype | unmethylated | LF |
| 15 | 46 | M | Glioblastoma, IDH wildtype | 10 | 1 | Glioblastoma, IDH wildtype | unmethylated | RP |
| 16 | 57 | M | Diffuse Astrocytoma, IDH mutated | 167 | 3 | Anaplastic Oligodendroglioma, IDH mutated | not available | LT |
| 17 | 58 | F | Glioblastoma, IDH wildtype | 16 | 3 | Glioblastoma, IDH wildtype | unmethylated | LT |
| 18 | 45 | M | Glioblastoma, NOS | 133 | 3 | Glioblastoma, IDH mutated | methylated | LT |
| Patient ID | EOR | Volume of Nanoparticles (mL) | Peak Temperature (°C) | Re-Irradiation (Gy) | Salvage Chemotherapy | Time to Edema (Months) | Revision Due to Edema | PFS (Months) | OS (Months) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | STR | 2.8 | 59 | 39.6 | - | 3.67 | Yes | 72 * | 72 † |
| 2 | GTR | 3.2 | 60 | 39.6 | - | 0.33 | No | 7 | 7 |
| 3 | NTR | 2 | 56 | 39.6 | - | 1.97 | Yes | 11 | 24 |
| 4 | STR | 2.1 | 53 | 39.6 | - | Non | Yes | 6 | 9 |
| 5 | GTR | 5 | 53 | - | - | 4.43 | No | 5 | 7 |
| 6 | NTR | 3 | 54 | - | - | Non | No | 3 | 4 |
| 7 | NRT | 3.2 | 49 | - | - | Non | No | 3 | 5 |
| 8 | GTR | 4.3 | 44 | - | Temozolomide | 5.07 | No | 35 | 35 |
| 9 | GTR | 3.4 | 50 | - | - | 2.73 | No | 4 | 10 |
| 10 | GTR | 3.8 | 48 | 39.6 | Bevacizumab | 3.33 | No | 8 | 8 |
| 11 | NTR | 5 | 45 | 39.6 | - | 3.33 | No | 5 | 10 |
| 12 | NTR | 3 | 42 | - | Non | No | 7 | 17 | |
| 13 | NTR | 7 | 46 | 39.6 | - | 1.5 | Yes | 6 | 7 |
| 14 | GTR | 3 | 39 | 39.6 | - | 2.0 | No | 5 | 6 |
| 15 | GTR | 1.9 | 43.3 | 39.6 | - | 2.33 | Yes | 3 | 15 † |
| 16 | NTR | 5 | 45 | - | - | Non | No | 7 | 12 |
| 17 | STR | 5 | 44.6 | - | - | 0.77 | No | 3 | 5 |
| 18 | STR | 5 | 45 | - | - | 1.83 | Yes | 2 | 11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwake, M.; Müther, M.; Bruns, A.-K.; Zinnhardt, B.; Warneke, N.; Holling, M.; Schipmann, S.; Brokinkel, B.; Wölfer, J.; Stummer, W.; et al. Combined Fluorescence-Guided Resection and Intracavitary Thermotherapy with Superparamagnetic Iron-Oxide Nanoparticles for Recurrent High-Grade Glioma: Case Series with Emphasis on Complication Management. Cancers 2022, 14, 541. https://doi.org/10.3390/cancers14030541
Schwake M, Müther M, Bruns A-K, Zinnhardt B, Warneke N, Holling M, Schipmann S, Brokinkel B, Wölfer J, Stummer W, et al. Combined Fluorescence-Guided Resection and Intracavitary Thermotherapy with Superparamagnetic Iron-Oxide Nanoparticles for Recurrent High-Grade Glioma: Case Series with Emphasis on Complication Management. Cancers. 2022; 14(3):541. https://doi.org/10.3390/cancers14030541
Chicago/Turabian StyleSchwake, Michael, Michael Müther, Ann-Katrin Bruns, Bastian Zinnhardt, Nils Warneke, Markus Holling, Stephanie Schipmann, Benjamin Brokinkel, Johannes Wölfer, Walter Stummer, and et al. 2022. "Combined Fluorescence-Guided Resection and Intracavitary Thermotherapy with Superparamagnetic Iron-Oxide Nanoparticles for Recurrent High-Grade Glioma: Case Series with Emphasis on Complication Management" Cancers 14, no. 3: 541. https://doi.org/10.3390/cancers14030541
APA StyleSchwake, M., Müther, M., Bruns, A.-K., Zinnhardt, B., Warneke, N., Holling, M., Schipmann, S., Brokinkel, B., Wölfer, J., Stummer, W., & Grauer, O. (2022). Combined Fluorescence-Guided Resection and Intracavitary Thermotherapy with Superparamagnetic Iron-Oxide Nanoparticles for Recurrent High-Grade Glioma: Case Series with Emphasis on Complication Management. Cancers, 14(3), 541. https://doi.org/10.3390/cancers14030541







