Anastomotic Leak in Ovarian Cancer Cytoreduction Surgery: A Systematic Review and Meta-Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Inclusion/Exclusion Criteria
2.2. Search Strategy
2.3. Data Extraction
2.4. Risk of Bias Assessment
2.5. Statistical Analysis
3. Results
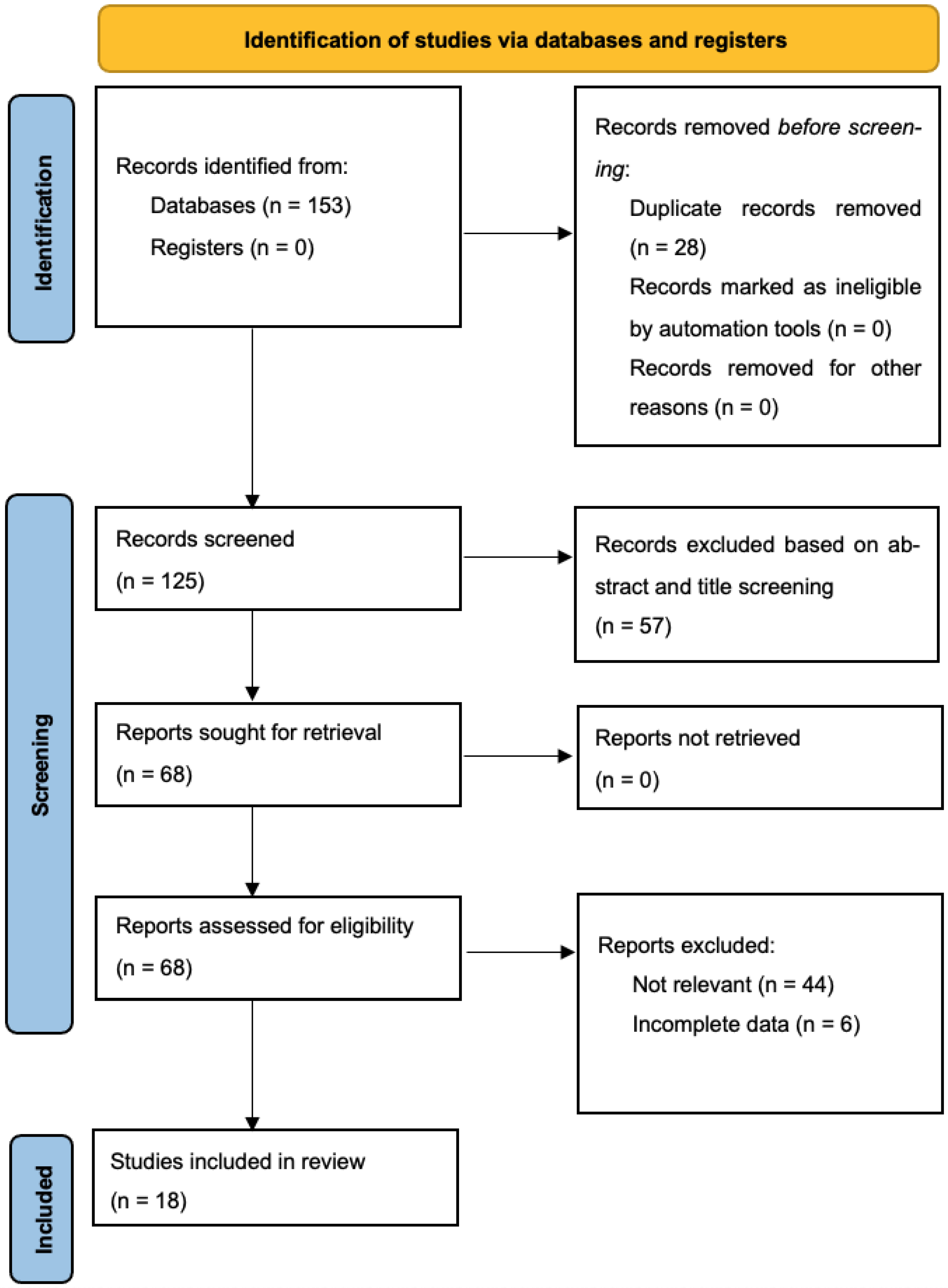
3.1. Study Selection, Study Characteristics
3.2. Patient Characteristics
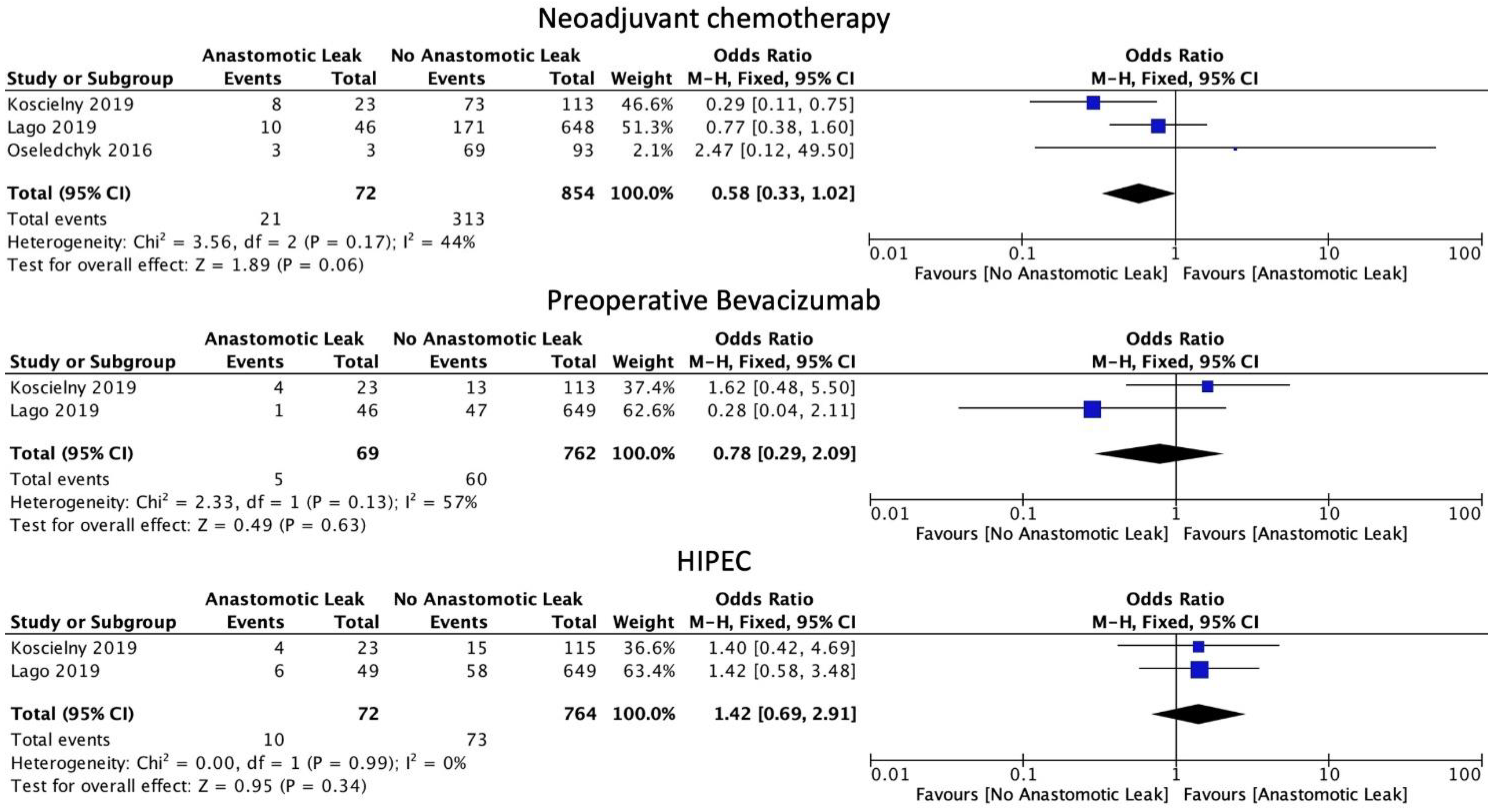
3.3. Neoadjuvant, Bevacizumab and HIPEC Therapy
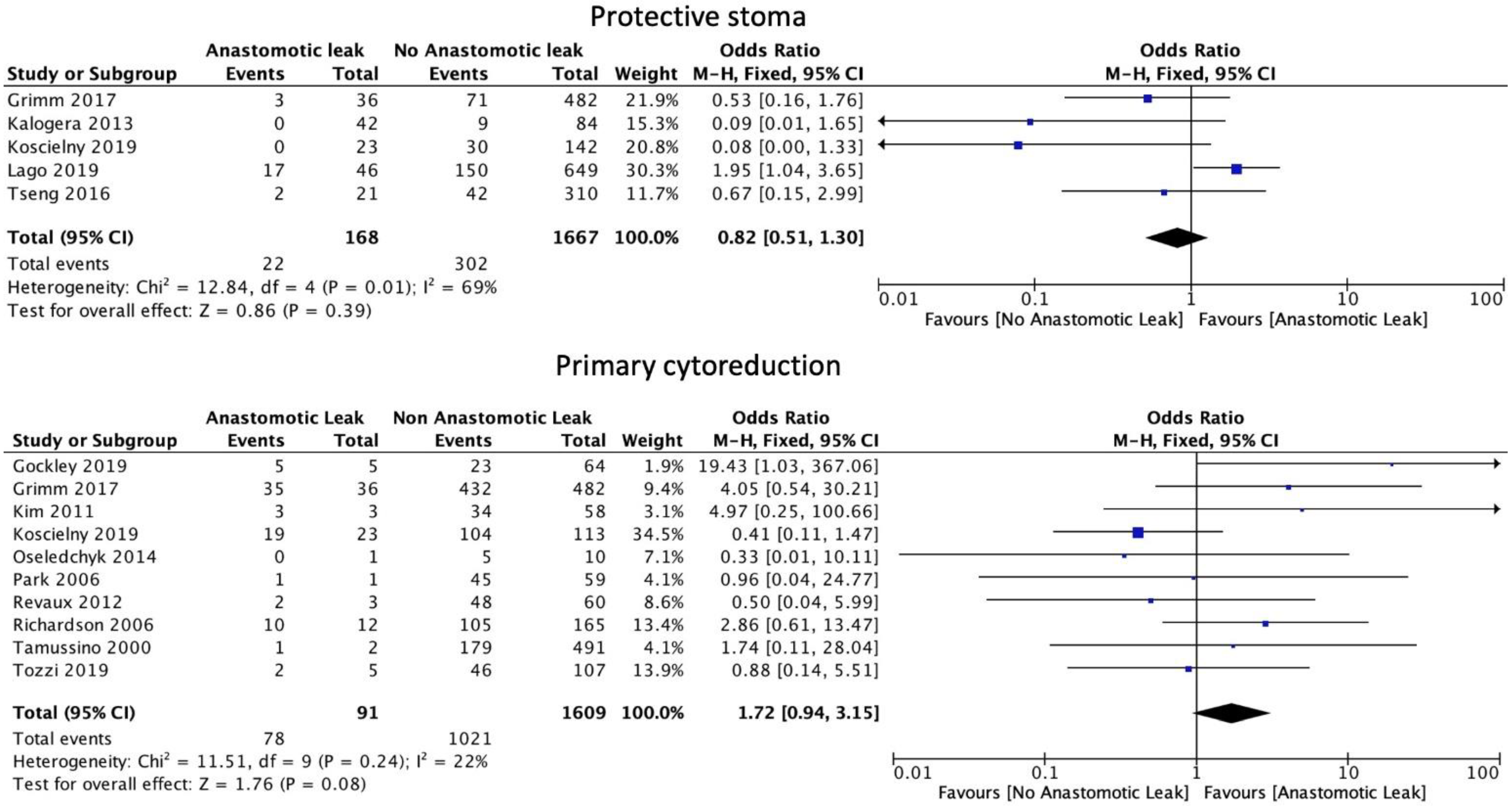
3.4. Intraoperative Considerations
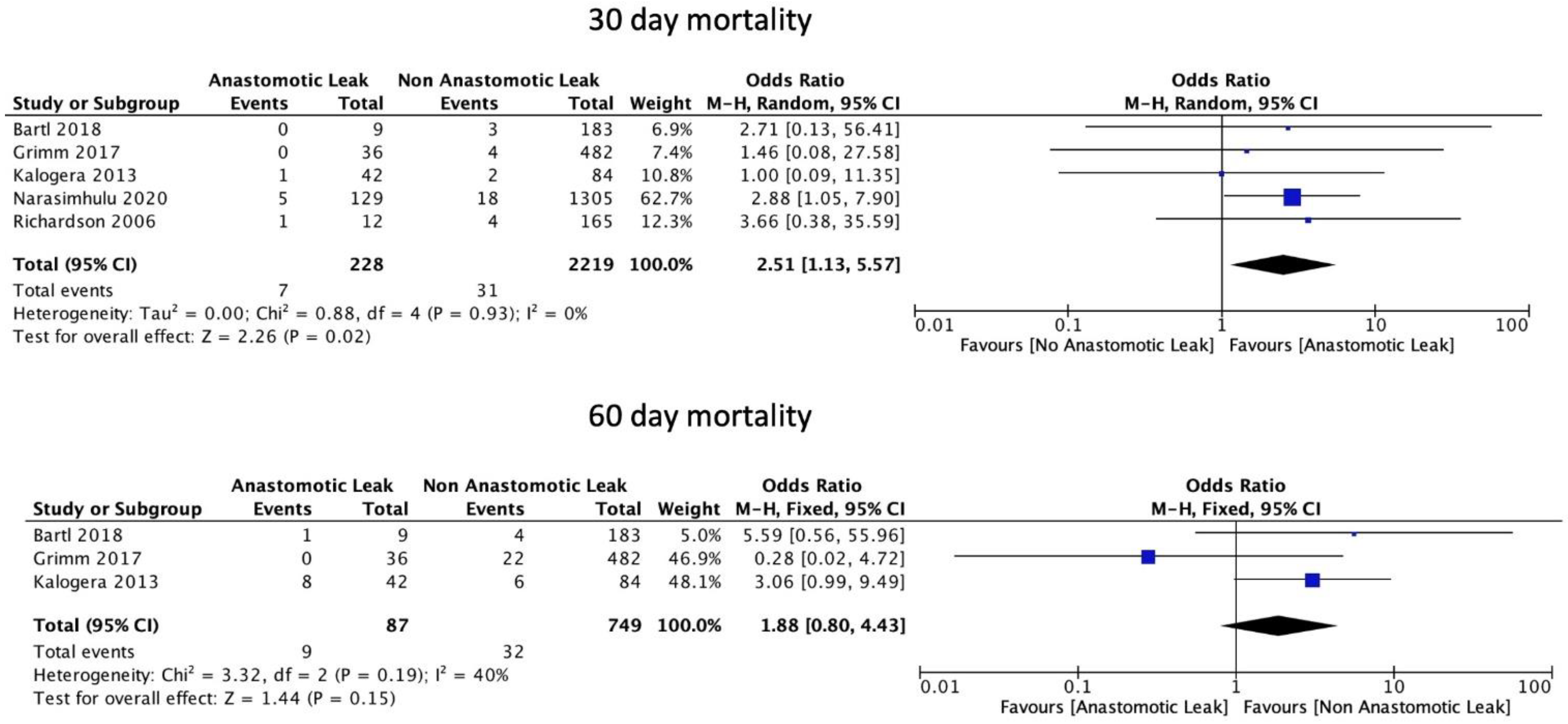
3.5. Mortality
3.6. Risk of Bias Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cancer Research UK. Ovarian Cancer Survival Statistics. Available online: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/ovarian-cancer/survival#heading-Zero (accessed on 29 September 2022).
- Aletti, G.D.; Dowdy, S.C.; Gostout, B.S.; Jones, M.B.; Stanhope, C.R.; Wilson, T.O.; Podratz, K.C.; Cliby, W.A. Aggressive surgical effort and improved survival in advanced-stage ovarian cancer. Obstet. Gynecol. 2006, 107, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, P.P. ASO Author Reflections: Ultra-Radical Resection in Ovarian Cancer: Where Are We and Where Are We Going? Ann. Surg. Oncol. 2021, 28, 231–232. [Google Scholar] [CrossRef] [PubMed]
- Bristow, R.E.; Del Carmen, M.G.; Kaufman, H.S.; Montz, F.J. Radical oophorectomy with primary stapled colorectal anastomosis for resection of locally advanced epithelial ovarian cancer. J. Am. Coll. Surg. 2003, 197, 565–574. [Google Scholar] [CrossRef]
- Peiretti, M.; Bristow, R.E.; Zapardiel, I.; Gerardi, M.; Zanagnolo, V.; Biffi, R.; Landoni, F.; Bocciolone, L.; Aletti, G.D.; Maggioni, A. Rectosigmoid resection at the time of primary cytoreduction for advanced ovarian cancer. A multi-center analysis of surgical and oncological outcomes. Gynecol. Oncol. 2012, 126, 220–223. [Google Scholar] [CrossRef]
- Maguire, B.; Clancy, C.; Connelly, T.M.; Mehigan, B.J.; McCormick, P.; Altomare, D.F.; Gosselink, M.P.; Larkin, J.O. Quality of life meta-analysis following coloanal anastomosis versus abdominoperineal resection for low rectal cancer. Colorectal Dis. 2022, 24, 811–820. [Google Scholar] [CrossRef]
- Gruner, M.; Chambers, L.M.; Yao, M.; Chichura, A.; Morton, M.; Costales, A.B.; Horowitz, M.; Rose, P.G.; Debernardo, R.; Michener, C.M. Anastomotic leak following interval debulking surgery with or without hyperthermic intraperitoneal chemotherapy in women with advanced epithelial ovarian Cancer. Gynecol. Oncol. 2021, 162, 645–651. [Google Scholar] [CrossRef]
- Koscielny, A.; Ko, A.; Egger, E.K.; Kuhn, W.; Kalff, J.C.; Keyver-Paik, M.D. Prevention of anastomotic leakage in ovarian cancer debulking surgery and its impact on overall survival. Anticancer Res. 2019, 39, 5209–5218. [Google Scholar] [CrossRef]
- Kingham, P.T.; Pachter, L.H. Colonic anastomotic leak: Risk factors, diagnosis, and treatment. J. Am. Coll. Surg. 2009, 208, 269–278. [Google Scholar] [CrossRef]
- Kwak, H.D.; Kim, S.H.; Kang, D.W.; Baek, S.J.; Kwak, J.M.; Kim, J. Risk factors and oncologic outcomes of anastomosis leakage after laparoscopic right colectomy. Surg. Laparosc. Endosc. Percutan. Tech. 2017, 27, 440–444. [Google Scholar] [CrossRef]
- Shimura, T.; Toiyama, Y.; Hiro, J.; Imaoka, H.; Fujikawa, H.; Kobayashi, M.; Ohi, M.; Inoue, Y.; Mohri, Y.; Kusunoki, M. Monitoring perioperative serum albumin can identify anastomotic leakage in colorectal cancer patients with curative intent. Asian J. Surg. 2018, 1, 30–38. [Google Scholar] [CrossRef]
- Choi, D.H.; Hwang, J.K.; Ko, Y.T.; Jang, H.J.; Shin, H.K.; Lee, Y.C.; Lim, C.H.; Jeong, S.K.; Yang, H.K. Risk factors for anastomotic leakage after laparoscopic rectal resection. J. Korean Soc. Coloproctol. 2010, 26, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Study Quality Assessment Tools, Quality Assessment of Case Control Studies. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 31 August 2022).
- The Cochrane Collaboration. Cochrane Handbook for Systematic Reviews of Interventions, 5.1.0. Available online: www.handbook.cochrane.org (accessed on 31 August 2022).
- Grimm, C.; Harter, P.; Alesina, P.F.; Prader, S.; Schneider, S.; Ataseven, B.; Meier, B.; Brunkhorst, V.; Hinrichs, J.; Kurzeder, C.; et al. The impact of type and number of bowel resections on anastomotic leakage risk in advanced ovarian cancer surgery. Gynecol. Oncol. 2017, 146, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Oseledchyk, A.; Abramian, A.; Kaiser, C.; Debald, M.; Domroese, C.; Kiefer, N.; Putensen, C.; Pantelis, D.; Kuhn, W.; Schäfer, N.; et al. Total or subtotal colectomy in patients undergoing surgery for primary or recurrent epithelial ovarian cancer. Oncol. Res. Treat. 2014, 37, 448–454. [Google Scholar] [CrossRef]
- Oseledchyk, A.; Hunold, L.E.; Mallmann, M.R.; Domröse, C.M.; Abramian, A.; Debald, M.; Kaiser, C.; Kiefer, N.; Putensen, C.; Pantelis, D.; et al. Impact of extended primary surgery on suboptimally operable patients with advanced ovarian cancer. Int. J. Gynecol. Cancer 2016, 26, 873–883. [Google Scholar] [CrossRef]
- Revaux, A.; Rouzier, R.; Ballester, M.; Selle, F.; Daraï, E.; Chéreau, E. Comparison of morbidity and survival between primary and interval cytoreductive surgery in patients after modified posterior pelvic exenteration for advanced ovarian cancer. Int. J. Gynecol. Cancer 2012, 22, 1349–1354. [Google Scholar] [CrossRef]
- Tozzi, R.; Casarin, J.; Baysal, A.; Valenti, G.; Kilic, Y.; Majd, H.S.; Morotti, M. Bowel resection rate but not bowel related morbidity is decreased after interval debulking surgery compared to primary surgery in patents with stage IIIC–IV ovarian cancer. J. Gynecol. Oncol. 2019, 30, e25. [Google Scholar] [CrossRef] [PubMed]
- Narasimhulu, D.M.; Bews, K.A.; Hanson, K.T.; Chang, Y.H.H.; Dowdy, S.C.; Cliby, W.A. Using evidence to direct quality improvement efforts: Defining the highest impact complications after complex cytoreductive surgery for ovarian cancer. Gynecol. Oncol. 2020, 156, 278–283. [Google Scholar] [CrossRef]
- Lago, V.; Fotopoulou, C.; Chiantera, V.; Minig, L.; Gil-Moreno, A.; Cascales-Campos, P.A.; Jurado, M.; Tejerizo, A.; Padilla-Iserte, P.; Malune, M.E.; et al. Risk factors for anastomotic leakage after colorectal resection in ovarian cancer surgery: A multi-centre study. Gynecol. Oncol. 2019, 153, 549–554. [Google Scholar] [CrossRef]
- Son, J.H.; Kong, T.W.; Paek, J.; Chang, S.J.; Ryu, H.S. Perioperative outcomes of extensive bowel resection during cytoreductive surgery in patients with advanced ovarian cancer. J. Surg. Oncol. 2019, 119, 1011–1015. [Google Scholar] [CrossRef]
- Bartl, T.; Schwameis, R.; Stift, A.; Bachleitner-Hofmann, T.; Reinthaller, A.; Grimm, C.; Polterauer, S. Predictive and prognostic implication of bowel resections during primary cytoreductive surgery in advanced epithelial ovarian cancer. Int. J. Gynecol. Cancer. 2018, 28, 1664–1671. [Google Scholar] [CrossRef]
- Tseng, J.H.; Suidan, R.S.; Zivanovic, O.; Gardner, G.J.; Sonoda, Y.; Levine, D.A.; Abu-Rustum, N.R.; Tew, W.P.; Chi, D.S.; Roche, K.L. Diverting ileostomy during primary debulking surgery for ovarian cancer: Associated factors and postoperative outcomes. Gynecol. Oncol. 2016, 142, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Richardson, D.L.; Mariani, A.; Cliby, W.A. Risk factors for anastomotic leak after recto-sigmoid resection for ovarian cancer. Gynecol. Oncol. 2006, 103, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Tamussino, K.F.; Lim, P.C.; Webb, M.J.; Lee, R.A.; Lesnick, T.G. Gastrointestinal surgery in patients with ovarian cancer. Gynecol. Oncol. 2001, 80, 79–84. [Google Scholar] [CrossRef]
- Park, J.Y.; Seo, S.S.; Kang, S.; Lee, K.B.; Lim, S.Y.; Choi, H.S.; Park, S.Y. The benefits of low anterior en bloc resection as part of cytoreductive surgery for advanced primary and recurrent epithelial ovarian cancer patients outweigh morbidity concerns. Gynecol. Oncol. 2006, 103, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, E.N.; Jeong, S.Y.; Chung, H.H.; Kim, Y.B.; Kim, J.W.; Park, K.J.; Park, N.H.; Song, Y.S.; Park, J.G.; et al. Comparison of the efficacy of low anterior resection with primary anastomosis and Hartmann’s procedure in advanced primary or recurrent epithelial ovarian cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 156, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Kalogera, E.; Dowdy, S.C.; Mariani, A.; Weaver, A.L.; Aletti, G.; Bakkum-Gamez, J.N.; Cliby, W.A. Multiple large bowel resections: Potential risk factor for anastomotic leak. Gynecol. Oncol. 2013, 130, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Estes, J.M.; Leath, C.A., III; Straughn, J.M., Jr.; Rocconi, R.P.; Kirby, T.O.; Huh, W.K.; Barnes, M.N., III. Bowel resection at the time of primary debulking for epithelial ovarian carcinoma: Outcomes in patients treated with platinum and taxane-based chemotherapy. J. Am. Coll. Surg. 2006, 203, 527–532. [Google Scholar] [CrossRef]
- Lago, V.; Flor, B.; Matute, L.; Padilla-Iserte, P.; García-Granero, A.; Bustamante, M.; Domingo, S. Ghost ileostomy in advanced ovarian cancer: A reliable option. Int. J. Gynecol. Cancer 2018, 28, 1418–1426. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Phillips, R.K.S.; Hittinger, R.; Blesovsky, L.; Fry, J.S.; Fielding, L.P. Local recurrence following ‘curative’ surgery for large bowel cancer: I. The overall picture. Br. J. Surg. 1984, 71, 12–16. [Google Scholar] [CrossRef]
- Sauven, P.; Playforth, M.J.; Evans, M.; Pollock, A.V. Early infective complications and late recurrent cancer in stapled colonic anastomoses. Dis. Colon Rectum 1989, 32, 33–35. [Google Scholar] [CrossRef] [PubMed]
- Guyton, K.L.; Hyman, N.H.; Alverdy, J.C. Prevention of perioperative anastomotic healing complications: Anastomotic stricture and anastomotic leak. Adv. Surg. 2016, 50, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Gessler, B.; Eriksson, O.; Angenete, E. Diagnosis, treatment, and consequences of anastomotic leakage in colorectal surgery. Int J Colorectal Dis. 2017, 32, 549–556. [Google Scholar] [CrossRef]
- Joseph, N.; Clark, R.M.; Dizon, D.S.; Lee, M.S.; Goodman, A.; Boruta, D., Jr.; Schorge, J.O.; Del Carmen, M.G.; Growdon, W.B. Delay in chemotherapy administration impacts survival in elderly patients with epithelial ovarian cancer. Gynecol. Oncol. 2015, 137, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Pölcher, M.; Mahner, S.; Ortmann, O.; Hilfrich, J.; Diedrich, K.; Breitbach, G.P.; Höss, C.; Leutner, C.; Braun, M.; Möbus, V.; et al. Neoadjuvant chemotherapy with carboplatin and docetaxel in advanced ovarian cancer—A prospective multicenter phase II trial (PRIMOVAR). Oncol. Rep. 2009, 22, 605–613. [Google Scholar] [CrossRef]
- Armstrong, D.K.; Alvarez, R.D.; Bakkum-Gamez, J.N.; Barroilhet, L.; Behbakht, K.; Berchuck, A.; Berek, J.S.; Chen, L.M.; Cristea, M.; DeRosa, M.; et al. NCCN guidelines insights: Ovarian cancer, version 1.2019: Featured updates to the NCCN guidelines. J. Natl. Compr. Cancer Netw. 2019, 17, 896–909. [Google Scholar] [CrossRef]
- Colombo, N.; Sessa, C.; du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO–ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann. Oncol. 2019, 30, 672–705. [Google Scholar] [CrossRef]
- Kehoe, S.; Hook, J.; Nankivell, M.; Jayson, G.C.; Kitchener, H.; Lopes, T.; Luesley, D.; Perren, T.; Bannoo, S.; Mascarenhas, M.; et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): An open-label, randomised, controlled, non-inferiority trial. Lancet 2015, 386, 249–257. [Google Scholar] [CrossRef]
- Vergote, I.; Tropé, C.G.; Amant, F.; Kristensen, G.B.; Ehlen, T.; Johnson, N.; Verheijen, R.H.; Van Der Burg, M.E.; Lacave, A.J.; Panici, P.B.; et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N. Engl. J. Med. 2010, 363, 943–953. [Google Scholar] [CrossRef]
- Bristow, R.E.; Chi, D.S. Platinum-based neoadjuvant chemotherapy and interval surgical cytoreduction for advanced ovarian cancer: A meta-analysis. Gynecol. Oncol. 2006, 103, 1070–1076. [Google Scholar] [CrossRef]
- Van Driel, W.J.; Koole, S.N.; Sikorska, K.; Schagen van Leeuwen, J.H.; Schreuder, H.W.; Hermans, R.H.; De Hingh, I.H.; Van Der Velden, J.; Arts, H.J.; Massuger, L.F.; et al. Hyperthermic intraperitoneal chemotherapy in ovarian cancer. N. Engl. J. Med. 2018, 378, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Cristian, D.A.; Grama, F.A.; Burcoş, T.; Bordea, A. Temporary protective loop ileostomy in open low rectal resection—An alternative technique. Chirurgia 2014, 109, 238–242. [Google Scholar]
- Hüser, N.; Michalski, C.W.; Erkan, M.; Schuster, T.; Rosenberg, R.; Kleeff, J.; Friess, H. Systematic review and meta-analysis of the role of defunctioning stoma in low rectal cancer surgery. Ann. Surg. 2008, 248, 52–60. [Google Scholar] [CrossRef]
- Santana, B.N.; Torralba, E.G.; Soriano, J.V.; Laseca, M.; Martinez, A.M. Protective ostomies in ovarian cancer surgery: A systematic review and meta-analysis. J. Gynecol. Oncol. 2022, 33, e21. [Google Scholar] [CrossRef] [PubMed]
- Lago, V.; Sanchez-Migallón, A.; Flor, B.; Padilla-Iserte, P.; Matute, L.; García-Granero, Á.; Bustamante, M.; Domingo, S. Comparative study of three different managements after colorectal anastomosis in ovarian cancer: Conservative management, diverting ileostomy, and ghost ileostomy. Int. J. Gynecol. Cancer 2019, 29, 1170–1176. [Google Scholar] [CrossRef] [PubMed]
- Harter, P.; Sehouli, J.; Reuss, A.; Hasenburg, A.; Scambia, G.; Cibula, D.; Mahner, S.; Vergote, I.; Reinthaller, A.; Burges, A.; et al. Prospective validation study of a predictive score for operability of recurrent ovarian cancer: The Multicenter Intergroup Study DESKTOP II. A project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. Int. J. Gynecol. Cancer 2011, 21, 289–295. [Google Scholar] [CrossRef]
- Harter, P.; Hahmann, M.; Lueck, H.J.; Poelcher, M.; Wimberger, P.; Ortmann, O.; Canzler, U.; Richter, B.; Wagner, U.; Hasenburg, A.; et al. Surgery for recurrent ovarian cancer: Role of peritoneal carcinomatosis: Exploratory analysis of the DESKTOP I Trial about risk factors, surgical implications, and prognostic value of peritoneal carcinomatosis. Ann. Surg. Oncol. 2009, 16, 1324–1330. [Google Scholar] [CrossRef]
- Bristow, R.E.; Puri, I.; Chi, D.S. Cytoreductive surgery for recurrent ovarian cancer: A meta-analysis. Gynecol. Oncol. 2009, 112, 265–274. [Google Scholar] [CrossRef]
- McDermott, F.D.; Heeney, A.; Kelly, M.E.; Steele, R.J.; Carlson, G.L.; Winter, D.C. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br. J. Surg. 2015, 102, 462–479. [Google Scholar] [CrossRef]
- Parthasarathy, M.; Greensmith, M.; Bowers, D.; Groot-Wassink, T. Risk factors for anastomotic leakage after colorectal resection: A retrospective analysis of 17 518 patients. Color. Dis. 2017, 19, 288–298. [Google Scholar] [CrossRef]
- Ionescu, D.; Tibrea, C.; Puia, C. Pre-operative hypoalbuminemia in colorectal cancer patients undergoing elective surgery—A major risk factor for postoperative outcome. Chirurgia 2013, 108, 822–828. [Google Scholar] [PubMed]
- Telem, D.A.; Chin, E.H.; Nguyen, S.Q.; Divino, C.M. Risk factors for anastomotic leak following colorectal surgery: A case-control study. Arch Surg. 2010, 145, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Valenti, G.; Vitagliano, A.; Morotti, M.; Giorda, G.; Sopracordevole, F.; Sapia, F.; Lo Presti, V.; Chiofalo, B.; Forte, S.; Lo Presti, L.; et al. Risks factors for anastomotic leakage in advanced ovarian cancer: A systematic review and meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 269, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.; Liu, Y.; Bi, D.S. Clinical risk factors for anastomotic leakage after laparoscopic anterior resection for rectal cancer: A systematic review and meta-analysis. Surg. Endosc. 2015, 29, 3608–3617. [Google Scholar] [CrossRef] [PubMed]



| Study (Year) | Country | Period | Sample Size (n) | Number of AL Patients (%) | Histology, n (type) | FIGO Staging, n (% of Sample Size) (Stage) | Number of CC-0 Resections (%) |
|---|---|---|---|---|---|---|---|
| Richardson et al. (2006) | USA | January 1999 to December 2004 | 177 | 12 (7%) | 167 (E) 7 (MMT) 3 (G) | 3 (1.7) (I) 11 (6.2) (II) 128 (72.3) (III) 26 (14.6) (IV) 9 (5.1) (Unknown) | 73 (41%) |
| Lago et al. (2019) | Multicentric | January 2010 to June 2018 | 695 | 46 (7%) | 572 (Serous) 49 (Endometrioid) 15 (Mucinous) 22 (Clear Cell) 36 (Other) | 29 (4.2) (II) 418 (60.1) (III) 114 (16.4) (IV) 134 (19.3) (Unknown) | |
| Tseng et al. (2016) | USA | January 2005 to January 2014 | 331 | 21 (6%) | 292 (Serous) 5 (Endometrioid) 4 (Clear cell) 30 (Other) | 11 (3.3) (II) 231 (69.8) (III) 89 (26.9) (IV) | |
| Bartl et al. (2018) | Austria | Between 2003 and 2017 | 192 | 9 (5%) | 193 (Epithelial ovarian cancer) | 154 (80.2) (IIB-IIIC) 38 (19.8) (IV) | 90 (47%) |
| Son et al. (2019) | South Korea | January 2006 to January 2018 | 172 | 5 (3%) | 146 (Serous) 21 (Non-serous) 5 (Non-epithelian ovarian cancer) | 128 (74.4) (IIIc or IV) | |
| Tozzi et al. (2019) | United Kingdom | January 2009 to March 2016 | 112 | 5 (5%) | 83 (Serous) 29 (Others) | 81 (72.3) (IIIc) 29 (27.7) (IV) | |
| Oseledchyk et al. (2016) | Germany | Between 2002 to 2013 | 96 | 3 (3%) | 91 (Serous) 2 (Mucinous) 1 (MMT) 1 (Clear cell) 1 (Intestinal) | 75 (78.1) (IIIc) 21 (21.9) (IV) | 0 (0%) |
| Oseledchyk et al. (2014) | Germany | January 2005 to September 2013 | 11 | 1 (9%) | 10 (Epithelial) 1 (MMT) | 9 (81.8) (III) 2 (18.1) (IV) | 7 (64%) |
| Estes et al. (2006) | USA | 1996 to 2001 | 48 | 1 (2%) | 24 (Papillary) 11 (Endometrioid) 8 (Mixed) 2 (Mucinous) 2 (Unkown) 1 (Clear cell) | 1 (2.1) (IIIa) 3 (6.3) (IIIb) 42 (87.5) ((IIIc) 2 (4.2) (IV) | 14 (29%) |
| Grimm et al. (2017) | Germany | January 1999 to December 2015 | 518 | 36 (7%) | 518 (Serous) | 262 (50.6) (III) 256 (49.4) (IV) | 469 (59%) |
| Kalogera et al. (2013) | USA | January 1994 to May 2011 | 126 (AL and 1:2 matched controls) | 42 (33%) | NG | 12 (9.5) (IIIa/b) 81 (64.3) (IIIc) 33 (26.2) (IV) | 11 (91%) |
| Kim et al. (2011) | South Korea | January 1998 to August 2008 | 61 | 3 (5%) | 61 (Epithelial, of which 25 are serous) | 51 (83.6) (III) 10 (16.4) (IV) | 35 (57%) |
| Koscielny et al. (2019) | Germany | 2010 to 2017 | 136 | 23 (17%) | 136 Epithelial | 104 (76.5) (I/II) 30 (22.1) (III/IV) | |
| Lago et al. (2018) | Spain | December 2016 to December 2017 | 26 | 2 (8%) | 22 (Serous) 2 (MMMT) 2 (Undifferentiated) | 7 (26.9)) (IIb) 1 (3.8) (IIIb) 12 (46.1) (IIIc) 4 (15.4) (IVa) 2 (7.7) (Not applicable—relapse) | |
| Park et al. (2006) | South Korea | April 2001 to May 2005 | 60 | 1 (2%) | 47 (Serous) 13 (Other) | 2 (3.3) (I) 1 (1.7) (II) 54 (90) (III) 3 (5) (IV) | 50 (83%) |
| Revaux et al. (2012) | France | 2001 to 2009 | 63 | 3 (5%) | 39 (Serous) 12 (Endometrioid) 2 (Mucinous) 4 (Papillary) 6 (Other) | 5 (7.9) (II) 52 (82.5) (III) 6 (9.5) (IV) | 51 (81%) |
| Tamussino et al. (2000) | USA | January 1983 to December 1992 | 364 | 2 (1%) | 306 (Serous) 17 (Mucinous) 39 (Endometrioid) 8 (Clear cell) 72 (Undifferentiated) 43 (Other) | 29 (8.0) (I) 31 (8.5) (II) 254 (69.8) ((III) 50 (13.7) (IV) | 216 (59%) |
| Narasimhulu et al. (2020) | USA | January 2012 to December 2016 | 1434 | 129 (9%) |
| Study (Year) | Inclusion Criteria | Exclusion Criteria | Anastomotic Leak Definition |
|---|---|---|---|
| Richardson et al. (2006) | Debulking surgery which included a rectosigmoid resection primary or recurrent ovarian or primary peritoneal cancer | Patient having end colostomy, diverting stoma, and inadequate postoperative follow-up after hospital discharge to assess bowel integrity | Evidence of fecal drainage from deep drains, wound or vagina, and/or evidence of either extravasation of contrast at the anastomotic site or evidence of communication between the rectum and pelvic abscess noted on radiographic imaging. |
| Lago et al. (2019) | Cytoreductive surgery for primary advanced or relapsed ovarian cancer with colorectal resection and anastomosis | Patients with end colostomy or end ileostomy, as well as those for whom relevant information was missing | |
| Tseng et al. (2016) | Stage II to IV ovarian, fallopian tube, or primary peritoneal carcinoma who underwent large bowel resection during PDS | Patients excluded if they had received neoadjuvant chemotherapy prior to attempted primary debulking surgery | |
| Bartl et al. (2018) | Patients with International Federation of Gynecology and Obstetrics (FIGO) advanced Epithelial ovarian carcinoma stage (IIB-IV) who underwent primary cytoreductive surgery | Patients with missing data about primary therapy, with missing informed consent, with recurrent disease, or with other concomitant malignancies were excluded from the study | |
| Son et al. (2018) | Patients with advanced ovarian cancer who underwent bowel resection as part of debulking surgery | patients who did not undergo full circumference bowel resection or those who underwent only appendectomy during the debulking procedure | Anastomotic leakage was defined as the drainage of fecal fluid or extravasations of anastomosis sites verified with computed tomography and the patients’ clinical symptoms |
| Tozzi et al. (2019) | Stage IIIC–IV ovarian cancer patients who had bowel surgery with rectosigmoid resection | ||
| Oseledchyk et al. (2016) | Patients with macroscopic tumor residuals after surgery for advanced-stage ovarian cancer were included | Patients who were operated on for borderline tumors, nonepithelial histology, or for recurrent disease were excluded | |
| Oseledchyk et al. (2014) | All patients who had undergone surgery including a total or subtotal colectomy as part of cytoreductive surgery for primary or recurrent ovarian cancer. | ||
| Estes et al. (2006) | Stage III or IV epithelial ovarian cancer who had bowel resection as part of primary cytoreductive surgery. | Patients diagnosed with ovarian cancer undergoing bowel resection to avoid obstruction | |
| Grimm et al. (2017) | Patients with primary advanced high-grade serous epithelial ovarian cancer (stage III-IV) undergoing primary or interval debulking surgery. | All patients who did not undergo full circumference bowel resection or received an appendectomy only | Feculent fluid from drains; vaginal vault or wound; extravasation from anastomotic site verified by computed tomography and/or leakage confirmed at revision surgery. |
| Kalogera et al. (2013) | All patients who underwent large bowel resection with primary anastomosis during cytoreductive surgery for primary or recurrent ovarian cancer. | Excluded from the analysis given the lack of appropriately matched control patients. | Feculent fluid from drains, wound or vagina; definitive radiographic evidence of extravasation at the anastomotic site or leakage found at reoperation. |
| Kim et al. (2011) | Advanced primary epithelial ovarian cancer with histological confirmation; stage III-IV disease; Eastern Co-operative Oncology Group performance status 0–2; primary cytoreductive surgery including low anterior resection and anastomosis or Hartmann’s procedure; adjuvant treatment with taxane and platinum-based chemotherapy after primary surgery and no neoadjuvant chemotherapy For patients with recurrent epithelial ovarian cancer inclusion criteria were: platinum sensitivity; up to three resectable metastatic tumors at preoperative evaluation; low anterior resection and anastomosis or Hartmann’s procedure for cytoreduction of recurrent tumors; no previous bowel surgery at rectosigmoid colon; up to three previous chemotherapeutic regimens and performance status 0–2 | ||
| Koscielny et al. (2019) | All patients who underwent any type of bowel resection and primary anastomosis during debulking surgery of confirmed ovarian cancer. | Patients without full circumferential bowel resection; Hartmann’s procedure or other discontinuity resection without anastomosis; later diagnosis of histology other than ovarian cancer; diffuse and deep infiltration of the small bowel mesentery root; diffuse carcinomatosis of the small bowel involving such large resections that resection would result in short bowel syndrome; diffuse involvement of stomach/duodenum without the possibility of limited resection, or of the head or middle part of the pancreas; tumor involvement of truncus coeliacus, hepatic arteries or left gastric artery; with central or multiple liver and pulmonary metastases. | Feculent secretion from drains, wound or vagina, extravasation from an anastomotic site verified by computed tomography, air exiting from drains during diagnostic rectoscopy or leakage confirmed at revision surgery. |
| Lago et al. (2018) | Patients who underwent bowel resection as part of modified pelvic exenteration in the treatment of ovarian cancer. | ||
| Park et al. (2006) | Patients with advanced (stage IIIb-IV) ovarian cancer undergoing low anterior en bloc resection as part of cytoreductive surgery, followed by adjuvant chemotherapy with taxane and platinum | ||
| Revaux et al. (2012) | Patients undergoing modified posterior pelvic exenteration for advanced ovarian cancer, either as part of primary cytoreductive surgery or interval cytoreductive surgery. | ||
| Tamussino et al. (2000) | Patients who underwent one or more operations for ovarian cancer, including a gastrointestinal procedure by gynecological surgeons. | Patients who underwent appendectomy. | |
| Narasimhulu et al. (2020) | Patients who underwent colon resection as part of complex cytoreductive surgery | Patients with ASA > 4; ventilator dependence; open wound; acute renal failure; undergoing dialysis; sepsis within 48 h prior to surgery and those undergoing emergent surgery. |
| First Author | Publication Year | Was the Study Question or Objective Clearly Stated? | Was the Study Population Clearly and Fully Described, Including a Case Definition? | Were The Cases Consecutive? | Were the Subjects Comparable? | Was the Intervention Clearly Described? | Were the Outcome Measures Clearly Defined, Valid, Reliable, and Implemented Consistently across All Study Participants? | Was the Length of Follow-Up Adequate? | Were the Statistical Methods Well-Described? | Were the Results Well-Described? | Total Score | Quality Rating |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C. Grimm | 2017 | N | Y | Y | Y | Y | Y | Y | Y | Y | 8 | High Quality |
| A. Koscielny | 2019 | Y | Y | Y | Y | Y | Y | Y | Y | Y | 9 | High Quality |
| V. Lago | 2019 | Y | N | Y | Y | N | Y | Y | Y | Y | 7 | High Quality |
| A. Oseledchyk | 2014 | Y | Y | Y | Y | Y | Y | Y | N | Y | 8 | High Quality |
| A. Oseledchyk | 2016 | Y | N | Y | Y | Y | Y | Y | Y | Y | 8 | High Quality |
| A. Revaux | 2012 | Y | N | Y | Y | Y | Y | Y | Y | Y | 8 | High Quality |
| D. Richardson | 2016 | Y | Y | Y | Y | Y | Y | Y | Y | Y | 9 | High Quality |
| J.H. Son | 2019 | Y | Y | Y | N | Y | Y | Y | N | Y | 7 | High Quality |
| R. Tozzi | 2019 | Y | N | Y | Y | Y | Y | Y | Y | Y | 8 | High Quality |
| J. Tseng | 2016 | Y | Y | Y | N | Y | Y | Y | Y | Y | 8 | High Quality |
| T. Bartl | 2018 | Y | Y | Y | Y | Y | Y | Y | Y | Y | 9 | High Quality |
| J. Estes | 2006 | Y | Y | Y | N | Y | Y | Y | Y | Y | 8 | High Quality |
| E. Kalogera | 2013 | Y | Y | Y | N | Y | Y | Y | Y | Y | 8 | High Quality |
| H. Kim | 2011 | Y | Y | Y | Y | Y | N | Y | Y | Y | 8 | High Quality |
| V. Lago | 2018 | Y | Y | Y | Y | Y | N | Y | Y | Y | 8 | High Quality |
| J.Y. Park | 2006 | Y | Y | Y | N | Y | Y | Y | Y | Y | 8 | High Quality |
| K. Tamussino | 2000 | Y | Y | Y | N | Y | Y | Y | Y | Y | 8 | High Quality |
| D. Narasimhulu | 2020 | Y | Y | Y | N | Y | N | Y | Y | Y | 7 | High Quality |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fornasiero, M.; Geropoulos, G.; Kechagias, K.S.; Psarras, K.; Katsikas Triantafyllidis, K.; Giannos, P.; Koimtzis, G.; Petrou, N.A.; Lucocq, J.; Kontovounisios, C.; et al. Anastomotic Leak in Ovarian Cancer Cytoreduction Surgery: A Systematic Review and Meta-Analysis. Cancers 2022, 14, 5464. https://doi.org/10.3390/cancers14215464
Fornasiero M, Geropoulos G, Kechagias KS, Psarras K, Katsikas Triantafyllidis K, Giannos P, Koimtzis G, Petrou NA, Lucocq J, Kontovounisios C, et al. Anastomotic Leak in Ovarian Cancer Cytoreduction Surgery: A Systematic Review and Meta-Analysis. Cancers. 2022; 14(21):5464. https://doi.org/10.3390/cancers14215464
Chicago/Turabian StyleFornasiero, Massimiliano, Georgios Geropoulos, Konstantinos S. Kechagias, Kyriakos Psarras, Konstantinos Katsikas Triantafyllidis, Panagiotis Giannos, Georgios Koimtzis, Nikoletta A. Petrou, James Lucocq, Christos Kontovounisios, and et al. 2022. "Anastomotic Leak in Ovarian Cancer Cytoreduction Surgery: A Systematic Review and Meta-Analysis" Cancers 14, no. 21: 5464. https://doi.org/10.3390/cancers14215464
APA StyleFornasiero, M., Geropoulos, G., Kechagias, K. S., Psarras, K., Katsikas Triantafyllidis, K., Giannos, P., Koimtzis, G., Petrou, N. A., Lucocq, J., Kontovounisios, C., & Giannis, D. (2022). Anastomotic Leak in Ovarian Cancer Cytoreduction Surgery: A Systematic Review and Meta-Analysis. Cancers, 14(21), 5464. https://doi.org/10.3390/cancers14215464









