The Emerging Role of PET/CT with PSMA-Targeting Radiopharmaceuticals in Clear Cell Renal Cancer: An Updated Systematic Review
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Literature Search Strategy and Information Sources
2.3. Eligibility Criteria
2.4. Selection Process
2.5. Data Collection Process and Data Extraction
2.6. Quality Assessment (Risk of Bias Assessment)
3. Results
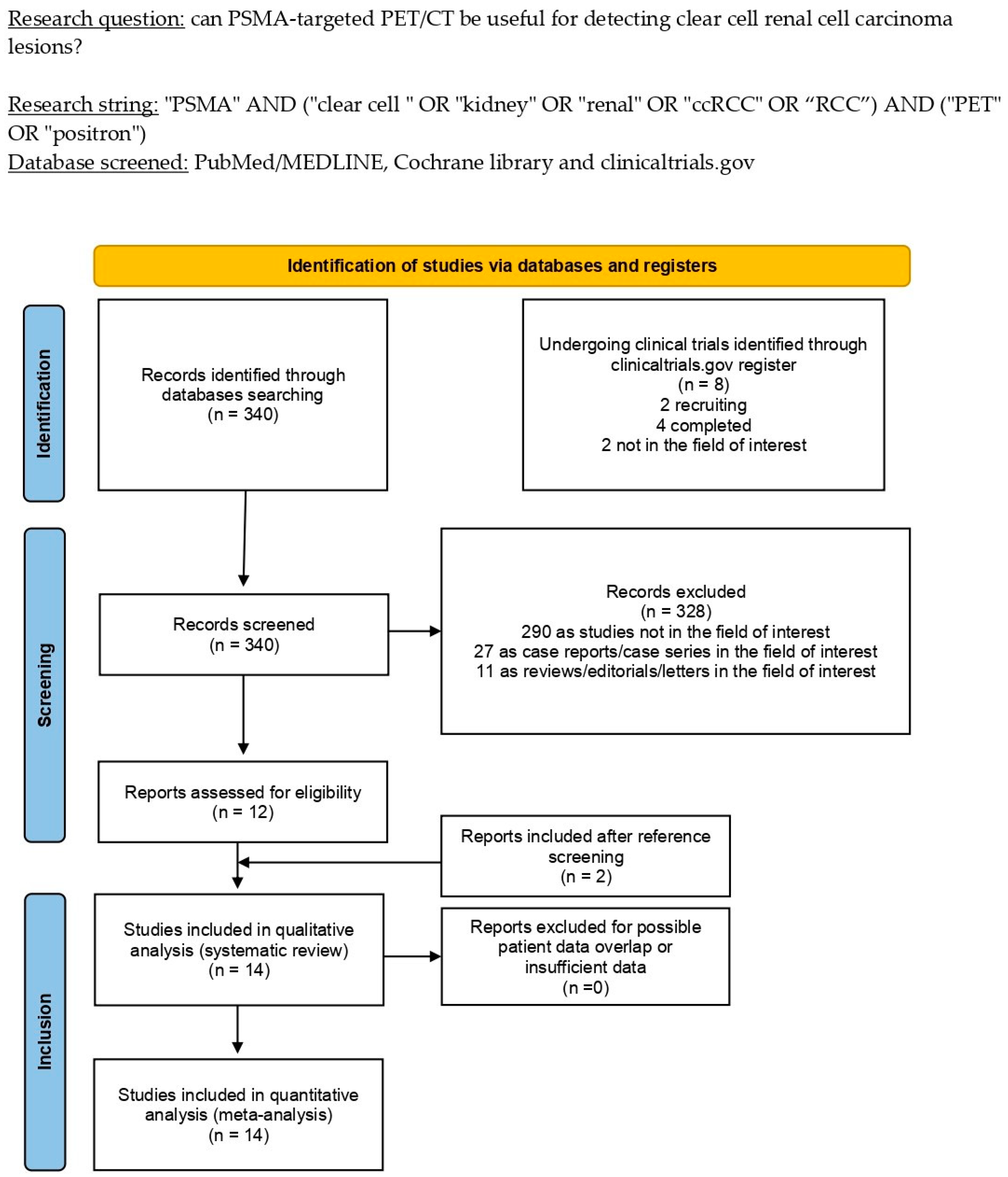
3.1. Literature Search and Study Selection
3.2. Study Characteristics
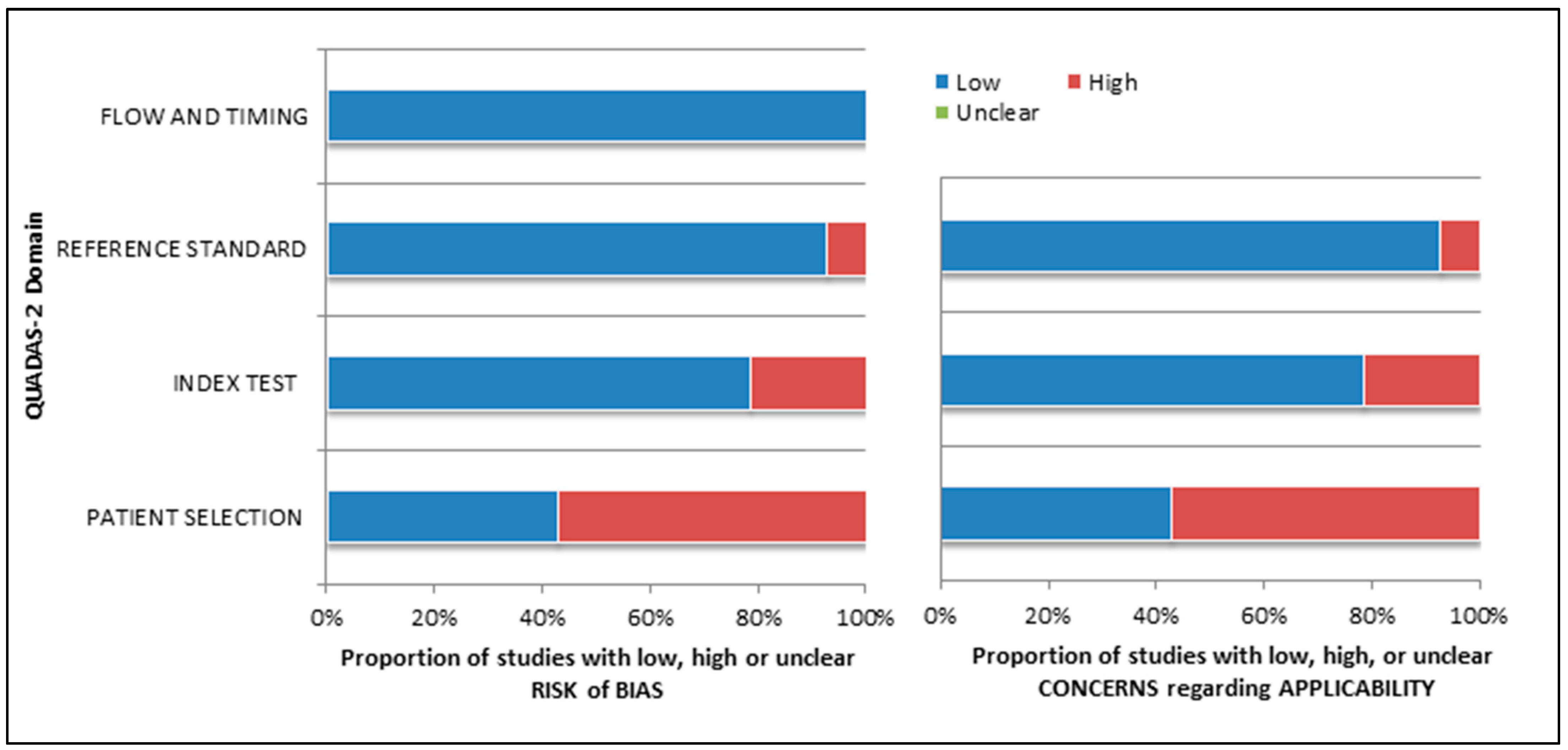
3.3. Risk of Bias and Applicability
3.4. Results of Individual Studies (Qualitative Synthesis)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA A Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, U.; Bensalah, K.; Bex, A.; Boorjian, S.A.; Bray, F.; Coleman, J.; Gore, J.L.; Sun, M.; Wood, C.; Russo, P. Epidemiology of Renal Cell Carcinoma. Eur. Urol. 2019, 75, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Heng, D.Y.; Xie, W.; Regan, M.M.; Warren, M.A.; Golshayan, A.R.; Sahi, C.; Eigl, B.J.; Ruether, J.D.; Cheng, T.; North, S.; et al. Prognostic Factors for Overall Survival in Patients with Metastatic Renal Cell Carcinoma Treated with Vascular Endothelial Growth Factor–Targeted Agents: Results from a Large, Multicenter Study. J. Clin. Oncol. 2009, 27, 5794–5799. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Ricketts, C.J.; Sourbier, C.; Linehan, W.M. New Strategies in Renal Cell Carcinoma: Targeting the Genetic and Metabolic Basis of Disease. Clin. Cancer Res. 2015, 21, 10–17. [Google Scholar] [CrossRef]
- Linehan, W.M.; Srinivasan, R.; Schmidt, L.S. The genetic basis of kidney cancer: A metabolic disease. Nat. Rev. Urol. 2010, 7, 277–285. [Google Scholar] [CrossRef]
- Linehan, W.M. Genetic basis of kidney cancer: Role of genomics for the development of disease-based therapeutics. Genome Res. 2012, 22, 2089–2100. [Google Scholar] [CrossRef]
- Maranchie, J.K.; Afonso, A.; Albert, P.S.; Kalyandrug, S.; Phillips, J.L.; Zhou, S.; Peterson, J.; Ghadimi, B.M.; Hurley, K.; Riss, J.; et al. Solid renal tumor severity in von Hippel Lindau disease is related to germline deletion length and location. Hum. Mutat. 2004, 23, 40–46. [Google Scholar] [CrossRef]
- Moore, L.E.; Nickerson, M.L.; Brennan, P.; Toro, J.R.; Jaeger, E.; Rinsky, J.; Han, S.S.; Zaridze, D.; Matveev, V.; Janout, V.; et al. Von Hippel-Lindau (VHL) Inactivation in Sporadic Clear Cell Renal Cancer: Associations with Germline VHL Polymorphisms and Etiologic Risk Factors. PLoS Genet. 2011, 7, e1002312. [Google Scholar] [CrossRef]
- Nickerson, M.L.; Jaeger, E.; Shi, Y.; Durocher, J.A.; Mahurkar, S.; Zaridze, D.; Matveev, V.; Janout, V.; Kollarova, H.; Bencko, V.; et al. Improved Identification of von Hippel-Lindau Gene Alterations in Clear Cell Renal Tumors. Clin. Cancer Res. 2008, 14, 4726–4734. [Google Scholar] [CrossRef]
- Hsieh, J.J.; Purdue, M.P.; Signoretti, S.; Swanton, C.; Albiges, L.; Schmidinger, M.; Heng, D.Y.; Larkin, J.; Ficarra, V. Renal cell carcinoma. Nat. Rev. Dis. Prim. 2017, 3, 17009. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ding, Y.; Zhou, M.; Rini, B.I.; Petillo, D.; Qian, C.-N.; Kahnoski, R.; Futreal, P.A.; Furge, K.A.; Teh, B.T. Interleukin-8 Mediates Resistance to Antiangiogenic Agent Sunitinib in Renal Cell Carcinoma. Cancer Res. 2010, 70, 1063–1071. [Google Scholar] [CrossRef]
- Low, G.; Huang, G.; Fu, W.; Moloo, Z.; Girgis, S. Review of renal cell carcinoma and its common subtypes in radiology. World J. Radiol. 2016, 8, 484–500. [Google Scholar] [CrossRef] [PubMed]
- Karlo, C.A.; Di Paolo, P.L.; Chaim, J.; Hakimi, A.A.; Ostrovnaya, I.; Russo, P.; Hricak, H.; Motzer, R.; Hsieh, J.J.; Akin, O. Radiogenomics of Clear Cell Renal Cell Carcinoma: Associations between CT Imaging Features and Mutations. Radiology 2014, 270, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Alessandrino, F.; Krajewski, K.M.; Shinagare, A.B. Update on Radiogenomics of Clear Cell Renal Cell Carcinoma. Eur. Urol. Focus 2016, 2, 572–573. [Google Scholar] [CrossRef]
- Lindenberg, L.; Mena, E.; Choyke, P.L.; Bouchelouche, K. PET imaging in renal cancer. Curr. Opin. Oncol. 2019, 31, 216–221. [Google Scholar] [CrossRef]
- Motzer, R.J.; Jonasch, E.; Agarwal, N.; Alva, A.; Baine, M.; Beckermann, K.; Carlo, M.I.; Choueiri, T.K.; Costello, B.A.; Derweesh, I.H.; et al. Kidney Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2022, 20, 71–90. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Porta, C.; Schmidinger, M.; Rioux-Leclercq, N.; Bex, A.; Khoo, V.; Grünwald, V.; Gillessen, S.; Horwich, A.; ESMO Guidelines Committee. Renal cell carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 706–720. [Google Scholar] [CrossRef]
- Ljungberg, B.; Albiges, L.; Abu-Ghanem, Y.; Bedke, J.; Capitanio, U.; Dabestani, S.; Fernández-Pello, S.; Giles, R.H.; Hofmann, F.; Hora, M.; et al. European Association of Urology Guidelines on Renal Cell Carcinoma: The 2022 Update. Eur. Urol. 2022, 82, 399–410. [Google Scholar] [CrossRef]
- Chen, R.; Zhou, X.; Huang, G.; Liu, J. Fructose 1,6-Bisphosphatase 1 Expression Reduces 18F-FDG Uptake in Clear Cell Renal Cell Carcinoma. Contrast Media Mol. Imaging 2019, 2019, 9463926. [Google Scholar] [CrossRef]
- Israeli, R.S.; Powell, C.T.; Fair, W.R.; Heston, W.D. Molecular cloning of a complementary DNA encoding a prostate-specific membrane antigen. Cancer Res. 1993, 53, 227–230. [Google Scholar]
- Horoszewicz, J.S.; Kawinski, E.; Murphy, G.P. Monoclonal antibodies to a new antigenic marker in epithelial prostatic cells and serum of prostatic cancer patients. Anticancer Res. 1987, 7, 927–935. [Google Scholar] [PubMed]
- Wester, H.-J.; Schottelius, M. PSMA-Targeted Radiopharmaceuticals for Imaging and Therapy. Semin. Nucl. Med. 2019, 49, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 2021, 385, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Jadvar, H.; Calais, J.; Fanti, S.; Feng, F.; Greene, K.L.; Gulley, J.L.; Hofman, M.; Koontz, B.F.; Lin, D.W.; Morris, M.J.; et al. Appropriate Use Criteria for Prostate-Specific Membrane Antigen PET Imaging. J. Nucl. Med. 2022, 63, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, R.; Treglia, G. Systematic reviews and meta-analyses of diagnostic studies: A practical guideline. Clin. Transl. Imaging 2017, 5, 83–87. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Whiting, P.F.; Rutjes, A.W.S.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.G.; Sterne, J.A.C.; Bossuyt, P.M.M.; QUADAS-2 Group. QUADAS-2: A Revised Tool for the Quality Assessment of Diagnostic Accuracy Studies. Ann. Intern. Med. 2011, 155, 529–536. Available online: https://www.acpjournals.org/doi/10.7326/0003-4819-155-8-201110180-00009 (accessed on 21 August 2022). [CrossRef]
- Rhee, H.; Blazak, J.; Tham, C.M.; Ng, K.L.; Shepherd, B.; Lawson, M.; Preston, J.; Vela, I.; Thomas, P.; Wood, S. Pilot study: Use of gallium-68 PSMA PET for detection of metastatic lesions in patients with renal tumour. EJNMMI Res. 2016, 6, 76. [Google Scholar] [CrossRef]
- Meyer, A.R.; Carducci, M.A.; Denmeade, S.R.; Markowski, M.C.; Pomper, M.G.; Pierorazio, P.M.; Allaf, M.E.; Rowe, S.P.; Gorin, M.A. Improved identification of patients with oligometastatic clear cell renal cell carcinoma with PSMA-targeted 18F-DCFPyL PET/CT. Ann. Nucl. Med. 2019, 33, 617–623. [Google Scholar] [CrossRef]
- Raveenthiran, S.; Esler, R.; Yaxley, J.; Kyle, S. The use of 68Ga-PET/CT PSMA in the staging of primary and suspected recurrent renal cell carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2280–2288. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, G.; Yu, H.; Wu, Y.; Lin, M.; Gao, J.; Xu, B. Comparison of 18F-DCFPyL and 18F-FDG PET/computed tomography for the restaging of clear cell renal cell carcinoma: Preliminary results of 15 patients. Nucl. Med. Commun. 2020, 41, 1299–1305. [Google Scholar] [CrossRef]
- Gao, J.; Xu, Q.; Fu, Y.; He, K.; Zhang, C.; Zhang, Q.; Shi, J.; Zhao, X.; Wang, F.; Guo, H. Comprehensive evaluation of 68Ga-PSMA-11 PET/CT parameters for discriminating pathological characteristics in primary clear-cell renal cell carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Golan, S.; Aviv, T.; Groshar, D.; Yakimov, M.; Zohar, Y.; Prokocimer, Y.; Nadu, A.; Baniel, J.; Domachevsky, L.; Bernstine, H. Dynamic 68Ga-PSMA-11 PET/CT for the Primary Evaluation of Localized Renal Mass: A Prospective Study. J. Nucl. Med. 2021, 62, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Gühne, F.; Seifert, P.; Theis, B.; Steinert, M.; Freesmeyer, M.; Drescher, R. PSMA-PET/CT in Patients with Recurrent Clear Cell Renal Cell Carcinoma: Histopathological Correlations of Imaging Findings. Diagnostics 2021, 11, 1142. [Google Scholar] [CrossRef]
- Gao, J.; Meng, L.; Xu, Q.; Zhao, X.; Deng, Y.; Fu, Y.; Guo, S.; He, K.; Shi, J.; Wang, F.; et al. 68Ga-PSMA-11 PET/CT Parameter Correlates with Pathological VEGFR-2/PDGFR-β Expression in Renal Cell Carcinoma Patients. Mol. Imaging Biol. 2022, 24, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Zhang, S.; Gao, J.; Xu, Q.; Fu, Y.; Zhou, Y.-H.; Wang, F.; Guo, H. [68Ga]Ga-PSMA-11 PET/CT has potential application in predicting tumor HIF-2α expression and therapeutic response to HIF-2α antagonists in patients with RCC. Eur. Radiol. 2022, 32, 6545–6553. [Google Scholar] [CrossRef] [PubMed]
- Tariq, A.; Kwok, M.; Pearce, A.; Rhee, H.; Kyle, S.; Marsh, P.; Raveenthiran, S.; Wong, D.; McBean, R.; Westera, J.; et al. The role of dual tracer PSMA and FDG PET/CT in renal cell carcinoma (RCC) compared to conventional imaging: A multi-institutional case series with intra-individual comparison. Urol. Oncol. Semin. Orig. Investig. 2022, 40, 66.e1–66.e9. [Google Scholar] [CrossRef] [PubMed]
- Tariq, A.; McGeorge, S.; Pearce, A.; Rhee, H.; Wood, S.; Kyle, S.; Marsh, P.; Raveenthiran, S.; Wong, D.; McBean, R.; et al. Characterization of tumor thrombus in renal cell carcinoma with prostate specific membrane antigen (PSMA) positron emission tomography (PET)/computed tomography (CT). Urol. Oncol. Semin. Orig. Investig. 2022, 40, 276.e1–276.e9. [Google Scholar] [CrossRef] [PubMed]
- Udovicich, C.; Callahan, J.; Bressel, M.; Ong, W.L.; Perera, M.; Tran, B.; Azad, A.; Haran, S.; Moon, D.; Chander, S.; et al. Impact of Prostate-specific Membrane Antigen Positron Emission Tomography/Computed Tomography in the Management of Oligometastatic Renal Cell Carcinoma. Eur. Urol. Open Sci. 2022, 44, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Mittlmeier, L.M.; Unterrainer, M.; Rodler, S.; Todica, A.; Albert, N.L.; Burgard, C.; Cyran, C.C.; Kunz, W.G.; Ricke, J.; Bartenstein, P.; et al. 18F-PSMA-1007 PET/CT for response assessment in patients with metastatic renal cell carcinoma undergoing tyrosine kinase or checkpoint inhibitor therapy: Preliminary results. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2031–2037. [Google Scholar] [CrossRef]
- Li, Y.; Zheng, R.; Zhang, Y.; Huang, C.; Tian, L.; Liu, R.; Liu, Y.; Zhang, Z.; Han, H.; Zhou, F.; et al. Special issue “The advance of solid tumor research in China”: 68Ga-PSMA-11 PET/CT for evaluating primary and metastatic lesions in different histological subtypes of renal cell carcinoma. Int. J. Cancer 2022, 152, 42–50. [Google Scholar] [CrossRef]
- Rizzo, A.; Dall’Armellina, S.; Pizzuto, D.A.; Perotti, G.; Zagaria, L.; Lanni, V.; Treglia, G.; Racca, M.; Annunziata, S. PSMA Radioligand Uptake as a Biomarker of Neoangiogenesis in Solid Tumours: Diagnostic or Theragnostic Factor? Cancers 2022, 14, 4039. [Google Scholar] [CrossRef] [PubMed]
- Conway, R.E.; Rojas, C.; Alt, J.; Nováková, Z.; Richardson, S.M.; Rodrick, T.C.; Fuentes, J.L.; Richardson, N.H.; Attalla, J.; Stewart, S.; et al. Prostate-specific membrane antigen (PSMA)-mediated laminin proteolysis generates a pro-angiogenic peptide. Angiogenesis 2016, 19, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Moch, H.; Cubilla, A.L.; Humphrey, P.A.; Reuter, V.E.; Ulbright, T.M. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs—Part A: Renal, Penile, and Testicular Tumours. Eur. Urol. 2016, 70, 93–105. [Google Scholar] [CrossRef]
- Sengupta, S.; Lohse, C.M.; Leibovich, B.C.; Frank, I.; Thompson, R.H.; Webster, W.S.; Zincke, H.; Blute, M.L.; Cheville, J.C.; Kwon, E.D. Histologic coagulative tumor necrosis as a prognostic indicator of renal cell carcinoma aggressiveness. Cancer 2005, 104, 511–520. [Google Scholar] [CrossRef]
- Khor, L.-Y.; Dhakal, H.P.; Jia, X.; Reynolds, J.P.; McKenney, J.K.; Rini, B.I.; Magi-Galluzzi, C.; Przybycin, C.G. Tumor Necrosis Adds Prognostically Significant Information to Grade in Clear Cell Renal Cell Carcinoma: A Study of 842 Consecutive Cases from a Single Institution. Urol. Oncol. Semin. Orig. Investig. 2016, 40, 1224–1231. [Google Scholar] [CrossRef]
- Pichler, R.; Compérat, E.; Klatte, T.; Pichler, M.; Loidl, W.; Lusuardi, L.; Schmidinger, M. Renal Cell Carcinoma with Sarcomatoid Features: Finally New Therapeutic Hope? Cancers 2019, 11, 422. [Google Scholar] [CrossRef]
- Przybycin, C.G.; McKenney, J.K.; Reynolds, J.P.; Campbell, S.; Zhou, M.; Karafa, M.T.; Magi-Galluzzi, C. Rhabdoid Differentiation Is Associated with Aggressive Behavior in Renal Cell Carcinoma: A Clinicopathologic Analysis of 76 Cases with Clinical Follow-Up. Am. J. Surg. Pathol. 2014, 38, 1260–1265. [Google Scholar] [CrossRef]
- Tyson, M.D.; Chang, S.S. Optimal Surveillance Strategies After Surgery for Renal Cell Carcinoma. J. Natl. Compr. Cancer Netw. 2017, 15, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ghesani, N.V.; Zuckier, L.S. Physiology and Pathophysiology of Incidental Findings Detected on FDG-PET Scintigraphy. Semin. Nucl. Med. 2010, 40, 294–315. [Google Scholar] [CrossRef] [PubMed]
- van Oostenbrugge, T.; Mulders, P. Targeted PET/CT imaging for clear cell renal cell carcinoma with radiolabeled antibodies: Recent developments using girentuximab. Curr. Opin. Urol. 2021, 31, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Andreucci, E.; Peppicelli, S.; Carta, F.; Brisotto, G.; Biscontin, E.; Ruzzolini, J.; Bianchini, F.; Biagioni, A.; Supuran, C.T.; Calorini, L. Carbonic anhydrase IX inhibition affects viability of cancer cells adapted to extracellular acidosis. J. Mol. Med. 2017, 95, 1341–1353. [Google Scholar] [CrossRef] [PubMed]
- Hekman, M.C.H.; Rijpkema, M.; Aarntzen, E.H.; Mulder, S.F.; Langenhuijsen, J.F.; Oosterwijk, E.; Boerman, O.C.; Oyen, W.J.G.; Mulders, P.F.A. Positron Emission Tomography/Computed Tomography with 89Zr-girentuximab Can Aid in Diagnostic Dilemmas of Clear Cell Renal Cell Carcinoma Suspicion. Eur. Urol. 2018, 74, 257–260. [Google Scholar] [CrossRef]
- Verhoeff, S.R.; van Es, S.C.; Boon, E.; van Helden, E.; Angus, L.; Elias, S.G.; Oosting, S.F.; Aarntzen, E.H.; Brouwers, A.H.; Kwee, T.C.; et al. Lesion detection by [89Zr]Zr-DFO-girentuximab and [18F]FDG-PET/CT in patients with newly diagnosed metastatic renal cell carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1931–1939. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lisok, A.; Chatterjee, S.; Wharram, B.; Pullambhatla, M.; Wang, Y.; Sgouros, G.; Mease, R.C.; Pomper, M.G. [18F]Fluoroethyl Triazole Substituted PSMA Inhibitor Exhibiting Rapid Normal Organ Clearance. Bioconjug. Chem. 2016, 27, 1655–1662. [Google Scholar] [CrossRef]
- Giesel, F.L.; Hadaschik, B.; Cardinale, J.; Radtke, J.; Vinsensia, M.; Lehnert, W.; Kesch, C.; Tolstov, Y.; Singer, S.; Grabe, N.; et al. F-18 labelled PSMA-1007: Biodistribution, radiation dosimetry and histopathological validation of tumor lesions in prostate cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 678–688. [Google Scholar] [CrossRef]
- Ling, S.W.; de Blois, E.; Hooijman, E.; van der Veldt, A.; Brabander, T. Advances in 177Lu-PSMA and 225Ac-PSMA Radionuclide Therapy for Metastatic Castration-Resistant Prostate Cancer. Pharmaceutics 2022, 14, 2166. [Google Scholar] [CrossRef]
- Yin, Y.; Campbell, S.P.; Markowski, M.C.; Pierorazio, P.M.; Pomper, M.G.; Allaf, M.E.; Rowe, S.P.; Gorin, M.A. Inconsistent Detection of Sites of Metastatic Non-Clear Cell Renal Cell Carcinoma with PSMA-Targeted [18F]DCFPyL PET/CT. Mol. Imaging Biol. 2019, 21, 567–573. [Google Scholar] [CrossRef]
- Vuijk, F.A.; Kleiburg, F.; Noortman, W.A.; Heijmen, L.; Shahbazi, S.F.; van Velden, F.H.P.; Baart, V.M.; Bhairosingh, S.S.; Windhorst, B.D.; Hawinkels, L.J.A.C.; et al. Prostate-Specific Membrane Antigen Targeted Pet/CT Imaging in Patients with Colon, Gastric and Pancreatic Cancer. Cancers 2022, 14, 6209. [Google Scholar] [CrossRef]
- Muselaers, S.; Erdem, S.; Bertolo, R.; Ingels, A.; Kara, Ö.; Pavan, N.; Roussel, E.; Pecoraro, A.; Marchioni, M.; Carbonara, U.; et al. PSMA PET/CT in Renal Cell Carcinoma: An Overview of Current Literature. J. Clin. Med. 2022, 11, 1829. [Google Scholar] [CrossRef] [PubMed]
- Urso, L.; Castello, A.; Rocca, G.C.; Lancia, F.; Panareo, S.; Cittanti, C.; Uccelli, L.; Florimonte, L.; Castellani, M.; Ippolito, C.; et al. Role of PSMA-ligands imaging in Renal Cell Carcinoma management: Current status and future perspectives. J. Cancer Res. Clin. Oncol. 2022, 148, 1299–1311. [Google Scholar] [CrossRef] [PubMed]
| Authors [Ref.] | Year | Country | Study Design/Number of Involved Centers | Funding Sources |
|---|---|---|---|---|
| Rhee et al. [29] | 2016 | Australia | Prospective/monocentric | None declared |
| Meyer et al. [30] | 2019 | U.S.A. | Prospective/monocentric | None declared |
| Raveenthiran [31] | 2019 | Australia | Retrospective/monocentric | None declared |
| Liu et al. [32] | 2020 | China | Retrospective/monocentric | None declared |
| Gao et al. [33] | 2020 | China | Retrospective/monocentric | None declared |
| Mittlmeier et al. [41] | 2020 | Germany | Retrospective/monocentric | None declared |
| Golan et al. [34] | 2021 | Israel | Prospective/monocentric | None declared |
| Gühne et al. [35] | 2021 | Germany | Retrospective/monocentric | None declared |
| Gao et al. [36] | 2022 | China | Retrospective/monocentric | None declared |
| Meng et al. [37] | 2022 | China | Retrospective/monocentric | None declared |
| Tariq et al. [38] | 2022 | Australia | Retrospective/multicentric | None declared |
| Tariq et al. [39] | 2022 | Australia | Retrospective/multicentric | None declared |
| Udovicich et al. [40] | 2022 | Australia | Retrospective/monocentric | None declared |
| Li et al. [42] | 2022 | China | Retrospective/monocentric | National Natural Science Foundation of China |
| Authors [Ref.] | Sample Size (No. ccRCC Patients/All Included Patients) | Mean/Median Age (Years) | Gender (Male %) | Clinical Setting (No. Patients) | WHO/ISUP Grade (No. Lesions) | PSMA Staining Analysis | Comparative Imaging |
|---|---|---|---|---|---|---|---|
| Rhee et al. [29] | 8/10 | Median: 57 | 100% | 10 staging | n.a. | n.a. | CT |
| Meyer et al. [30] | 14/14 | Median: 59 | 64.3% | 14 staging | n.a. | n.a. | CT or MRI of abdomen + CT of the chest |
| Raveenthiran et al. [31] | 28/38 | Median: 64 | 25.7% | 16 staging 22 restaging | n.a. | n.a. | CT |
| Liu et al. [32] | 15/15 | Mean: 57.5 | 86.6% | 15 restaging | n.a. | n.a. | [18F]FDG PET/CT |
| Gao et al. [33] | 36/36 | Median: 61 | 58.3% | 36 staging | 9 grade 1 12 grade 2 9 grade 3 6 grade 4 | Yes | No comparative exam |
| Mittlmeier et al. [41] | 8/11 | Mean: 59.6 | 72.7% | 11 staging and restaging after treatment | n.a. | n.a. | CT |
| Golan et al. [34] | 18/27 | Median: 66 | 70% | 27 staging | 14 grade 1–2 4 grade 3 | Yes | No comparative exam |
| Gühne et al. [35] | 9/9 | Range: 52–80 | 88% | 9 Restaging | 1 grade 1 6 grade 2 3 grade 3 1 grade 4 | Yes | CT |
| Gao et al. [36] | 37/48 | Median: 59 | 60.4% | 48 staging | 10 grade 1 13 Grade 2 11 grade 3 7 grade 4 | Yes | No comparative exam |
| Meng et al. [37] | 40/53 | Median: 56 | 60.4 | 53 staging | 10 grade 1 17 grade 2 15 grade 3 3 grade 4 | n.a. | No comparative exam |
| Tariq et al. [38] | 10/11 | Mean: 65.5 | 64% | 4 staging 7 restaging | n.a. | n.a. | [18F]FDG PET/CT; CT |
| Tariq et al. [39] | 14/14 | Median: 61 | 64.3% | 12 staging 2 restaging | 3 grade 2 5 grade 3 5 grade 4 1 ungraded | n.a. | MRI |
| Udovicich et al. [40] | 54/61 | Mean: 65 | 56% | 61 restaging | n.a. | n.a. | [18F]FDG PET/CT; CT |
| Li et al. [42] | 40/50 | Median age: 55 | 78% | 50 staging | n.a. | Yes | CT or MRI |
| Authors [Ref.] | Tracer | Hybrid Imaging | Tomograph | Administered Activity | Uptake Time (Minutes) | Image Analysis |
|---|---|---|---|---|---|---|
| Rhee et al. [29] | [68Ga]Ga-PSMA-11 | PET/CT | Biograph mCT FLOW (Siemens ®) | 150 MBq | 60 | Qualitative and semiquantative (SUVmax) |
| Meyer et al. [30] | [18F]F-DCFPyL | PET/CT | n.a. | 333 MBq | 60 | Qualitative and semiquantative (SUVmax) |
| Raveenthiran et al. [31] | [68Ga]Ga-PSMA-11 | PET/CT | Biograph mCT (Siemens ®) | 150 MBq | 45–60 | Qualitative and semiquantitative (SUVmax) |
| Liu et al. [32] | [18F]F-DCFPyL | PET/CT | Biograph 64 (Siemens ®) | 0.15 Ci/kg | 60 | Qualitative and semiquantitative (SUVmax, TBR) |
| Gao et al. [33] | [68Ga]Ga-PSMA-11 | PET/CT | uMI 780 (United Imaging Healthcare ®) | n.a. | 45 | Qualitative and semiquantitative (SUVmax, SUVmean, SUVpeak) |
| Mittlmeier et al. [41] | [18F]F-PSMA-1007 | PET/CT | Biograph mCT (Siemens ®); Biograph 64 (Siemens ®) | 217–268 | 60 | Qualitative and semiquantitative (SUVmean, SUL) |
| Golan et al. [34] | [68Ga]Ga-PSMA-11 | Dynamic PET/CT | Discovery 710 (GE healthcare ®) | 75–150 MBq | 0 | Qualitative, semiquantitative (SUVmax, SUVmean, TBR) and kinetic analysis |
| Gühne et al. [35] | [68Ga]Ga-PSMA-11 | PET/CT | Biograph mCT 40 (Siemens ®) | 221–272 MBq | 74–103 | Qualitative and semiquantitative (SUVmax, SUVmean, TBR) |
| Gao et al. [36] | [68Ga]Ga-PSMA-11 | PET/CT | uMI 780 (United Imaging Healthcare ®) | n.a. | 45 | Qualitative and semiquantitative (SUVmax) |
| Meng et al. [37] | [68Ga]Ga-PSMA-11 | PET/CT | uMI 780 (United Imaging Healthcare ®) | n.a. | 45 | Qualitative and semiquantitative (SUVmax) |
| Tariq et al. [38] | [68Ga]Ga-PSMA-11; [18F]F-PSMA-1007 | PET/CT | Biograph mCT (Siemens ®); Ingenuity TF (Philips ®); Discovery MI DR (GE ®) | 124–168 MBq for [68Ga]Ga-PSMA-11; 224–244 MBq for [18F]F-PSMA-1007 | 45–63 for [68Ga]Ga-PSMA-11; 120–130 for [18F]F-PSMA-1007 | Qualitative and semiquantitative (SUVmax) |
| Tariq et al. [39] | [68Ga]Ga-PSMA-11; [18F]F-PSMA-1007 | PET/CT | Biograph mCT (Siemens ®); Ingenuity TF (Philips ®); Discovery MI DR (GE ®) | 121–267 MBq for [68Ga]Ga-PSMA-11; 247–260 MBq for [18F]F-PSMA-1007 | 41–94 for [68Ga]Ga-PSMA-11; 117–142 for [18F]F-PSMA-1007 | Qualitative and semiquantitative (SUVmax) |
| Udovicich et al. [40] | [68Ga]Ga-PSMA-11; [18F]F-DCFPyL | PET/CT | Discovery 690 (GE ®); Discovery 710 (GE ®) | 2.6 MBq/kg for [68Ga]Ga-PSMA-11; 3.6 MBq/kg for [18F]F-DCFPyL | 60 for [68Ga]Ga-PSMA-11; 120 for [18F]F-DCFPyL | Qualitative and semiquantitative (SUVmax, MTV, TLP) |
| Li et al. [42] | [68Ga]Ga-PSMA-11 | PET/CT | Biograph mCT.X (Siemens ®) | 0.05 mCi/kg | 60–90 | Qualitative and semiquantitative (SUVmax, TBR) |
| Authors [Ref.] | Primitive Lesion SUVmax | Metastatic Lesions SUVmax | Correlation of Uptake with Histology or IHC | Detection Rate | Change of Management (No. Patients) |
|---|---|---|---|---|---|
| Rhee et al. [29] | Mean: 18.0 | Mean: 19.5 | n.a. | Per-lesion: 100% | 2 (20%) |
| Meyer et al. [30] | Median: 9.6 | Median: 2.7 | n.a. | Per-patient: 92.8% Per-lesion: 88.9% | 3 (21.4%) |
| Raveenthiran et al. [31] | n.a. | n.a. | n.a. | Per-lesion: 80.5% | Staging: 7 (43.8%) Restaging: 9 (40.9%) |
| Liu et al. [32] | n.a. | Soft tissue mean: 6.9 Bone mean: 8.2 | n.a. | Per-patient: 100% * Per-lesion: 100% * | n.a. |
| Gao et al. [33] | Mean: 17.78 | n.a. | SUVmax differentiates WHO/ISUP grade and adverse pathology | n.a. | n.a. |
| Mittlmeier et al. [41] | n.a. | n.a. | n.a. | n.a. | 9 (81.8%) |
| Golan et al. [34] | Median: 9.4 | n.a. | Cytoplasmatic PSMA staining was associated with the washout coefficient. | n.a. | n.a. |
| Gühne et al. [35] | n.a. | Median: 3.1 | No correlation between uptake and PSMA staining in IHC analysis | n.a. | n.a. |
| Gao et al. [36] | n.a. | n.a. | SUVmax differentiates VEGFR-2 expression, PDGFR-β expression, and VEGFR-2 and PDGFR-β coexpression | n.a. | n.a. |
| Meng et al. [37] | n.a. | n.a. | SUVmax correlates with high HIF-2α expression | n.a. | n.a. |
| Tariq et al. [38] | Median: 3.2 | Median: 8.0 | n.a. | n.a. | 3 (27%) |
| Tariq et al. [39] | Mean: 25.3 | n.a. | n.a. | n.a. | 3 (30%) |
| Udovicich et al. [40] | n.a. | 15.0 | n.a. | Per-patient: 84% | 30 (49%) |
| Li et al. [42] | Median: 18 | Median: 3.7–9.6 (based on location) | SUVmax values are related to pathologic subtypes and PSMA staining scores | Per-lesion: 93.6% | 4 (12.9%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rizzo, A.; Racca, M.; Dall’Armellina, S.; Rescigno, P.; Banna, G.L.; Albano, D.; Dondi, F.; Bertagna, F.; Annunziata, S.; Treglia, G. The Emerging Role of PET/CT with PSMA-Targeting Radiopharmaceuticals in Clear Cell Renal Cancer: An Updated Systematic Review. Cancers 2023, 15, 355. https://doi.org/10.3390/cancers15020355
Rizzo A, Racca M, Dall’Armellina S, Rescigno P, Banna GL, Albano D, Dondi F, Bertagna F, Annunziata S, Treglia G. The Emerging Role of PET/CT with PSMA-Targeting Radiopharmaceuticals in Clear Cell Renal Cancer: An Updated Systematic Review. Cancers. 2023; 15(2):355. https://doi.org/10.3390/cancers15020355
Chicago/Turabian StyleRizzo, Alessio, Manuela Racca, Sara Dall’Armellina, Pasquale Rescigno, Giuseppe Luigi Banna, Domenico Albano, Francesco Dondi, Francesco Bertagna, Salvatore Annunziata, and Giorgio Treglia. 2023. "The Emerging Role of PET/CT with PSMA-Targeting Radiopharmaceuticals in Clear Cell Renal Cancer: An Updated Systematic Review" Cancers 15, no. 2: 355. https://doi.org/10.3390/cancers15020355
APA StyleRizzo, A., Racca, M., Dall’Armellina, S., Rescigno, P., Banna, G. L., Albano, D., Dondi, F., Bertagna, F., Annunziata, S., & Treglia, G. (2023). The Emerging Role of PET/CT with PSMA-Targeting Radiopharmaceuticals in Clear Cell Renal Cancer: An Updated Systematic Review. Cancers, 15(2), 355. https://doi.org/10.3390/cancers15020355









