Changing Epidemiological Trends of Hepatobiliary Carcinomas in Austria 2010–2018
Abstract
:Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Data Retrieval
2.2. Statistics
3. Results
3.1. Patients
3.2. Hepatocellular Carcinoma (HCC, ICD-10 code C22.0)
3.3. Intrahepatic Cholangiocarcinoma (iCC, ICD-10 code C22.1)
3.4. Extrahepatic Cholangiocarcinoma (eCC, ICD-10 code C24.0)
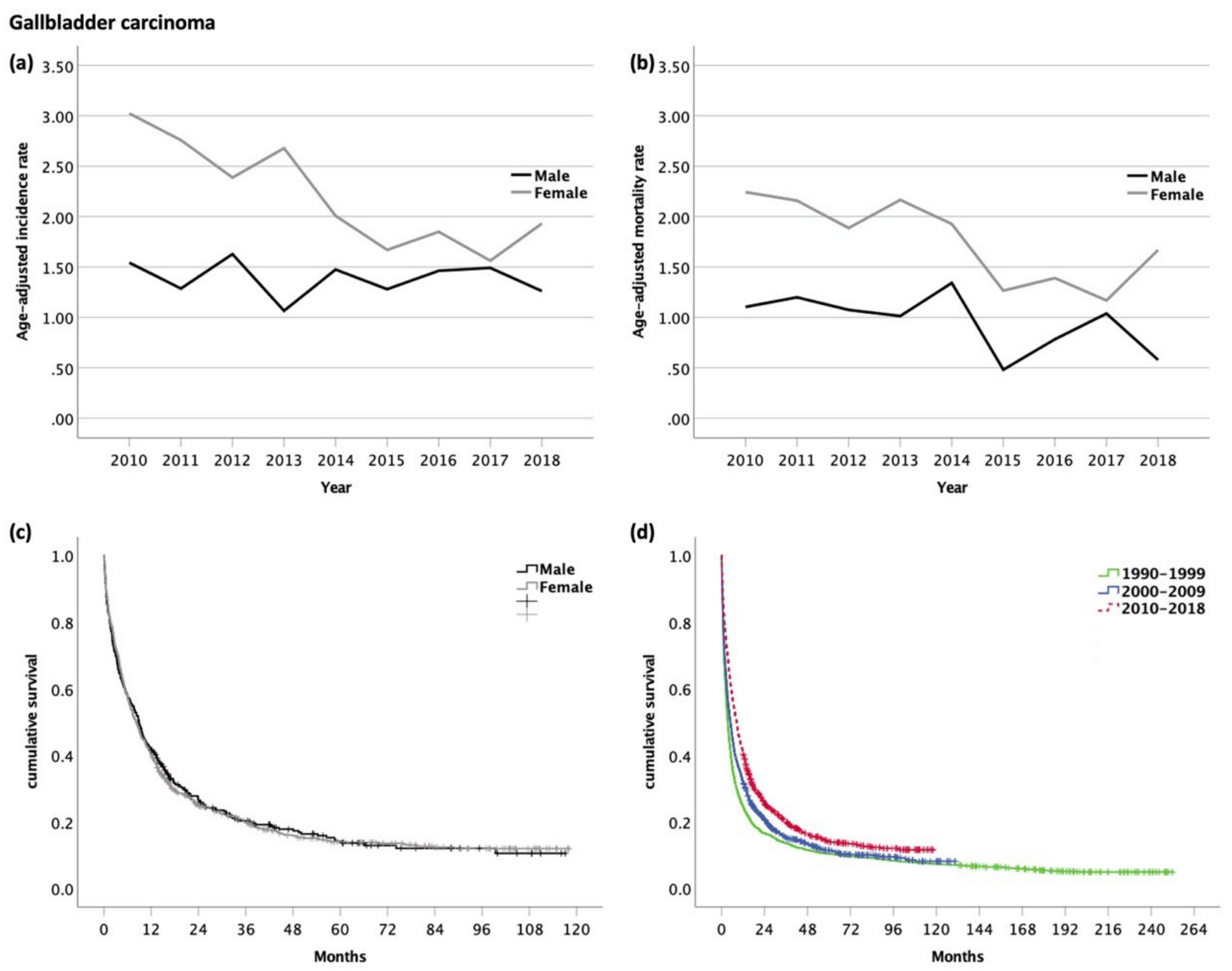
3.5. Gallbladder Carcinoma (GBC, ICD-10 code C23.0)
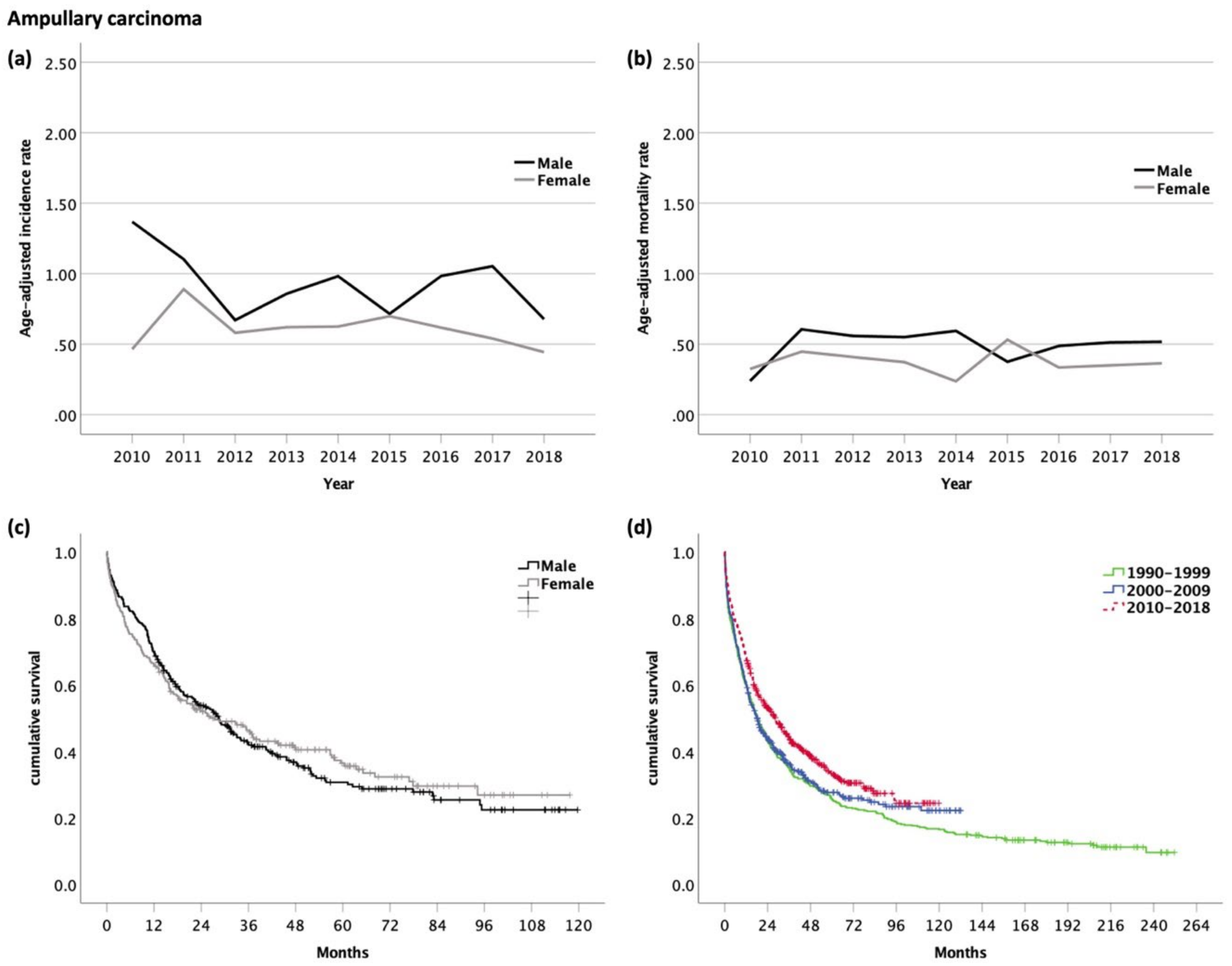
3.6. Carcinoma of the Ampulla of Vater (AC, ICD-10 code C24.1)
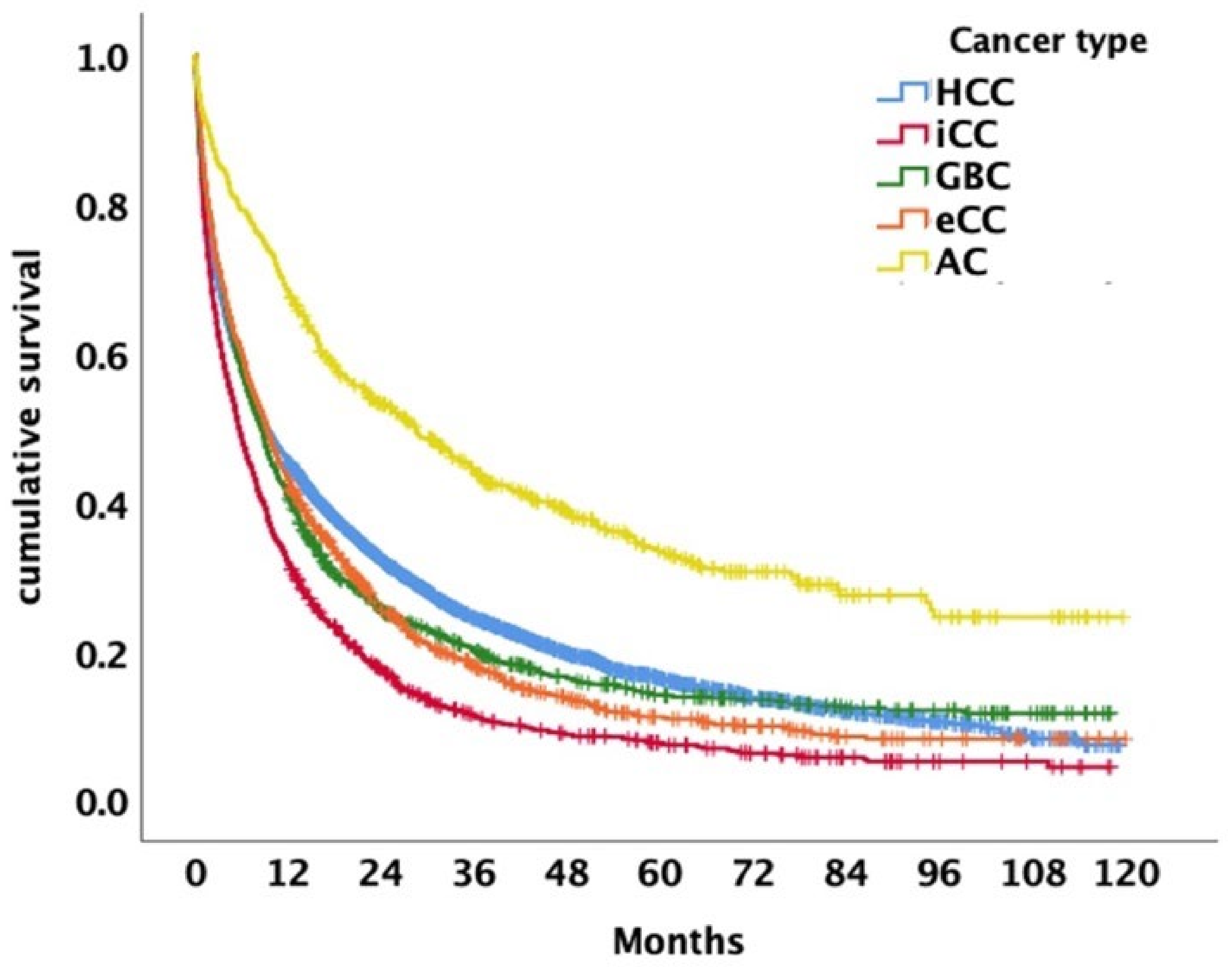
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valle, J.W.; Kelley, R.K.; Nervi, B.; Oh, D.Y.; Zhu, A.X. Biliary tract cancer. Lancet 2021, 397, 428–444. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Weaver, A.J.; Stafford, R.; Hale, J.; Denning, D.; Sanabria, J.R.; Collaborators, G.B.D. Geographical and Temporal Variation in the Incidence and Mortality of Hepato-Pancreato-Biliary Primary Malignancies:1990–2017. J. Surg. Res. 2020, 245, 89–98. [Google Scholar] [CrossRef] [Green Version]
- Clements, O.; Eliahoo, J.; Kim, J.U.; Taylor-Robinson, S.D.; Khan, S.A. Risk factors for intrahepatic and extrahepatic cholangiocarcinoma: A systematic review and meta-analysis. J. Hepatol. 2020, 72, 95–103. [Google Scholar] [CrossRef]
- McGlynn, K.A.; Petrick, J.L.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma. Hepatology 2021, 73, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Banales, J.M.; Cardinale, V.; Carpino, G.; Marzioni, M.; Andersen, J.B.; Invernizzi, P.; Lind, G.E.; Folseraas, T.; Forbes, S.J.; Fouassier, L.; et al. Expert consensus document: Cholangiocarcinoma: Current knowledge and future perspectives consensus statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 261–280. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- Pinter, M.; Peck-Radosavljevic, M. Review article: Systemic treatment of hepatocellular carcinoma. Aliment. Pharmacol. Ther. 2018, 48, 598–609. [Google Scholar] [CrossRef] [Green Version]
- European Association for the Study of the Liver. Electronic address, e.e.e.; European Association for the Study of the, L. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [Green Version]
- Pinter, M.; Hucke, F.; Zielonke, N.; Trauner, M.; Sieghart, W.; Peck-Radosavljevic, M. Epidemiological trends of hepatocellular carcinoma in Austria. Dig. Dis. 2014, 32, 664–669. [Google Scholar] [CrossRef]
- Pinter, M.; Hucke, F.; Zielonke, N.; Waldhor, T.; Trauner, M.; Peck-Radosavljevic, M.; Sieghart, W. Incidence and mortality trends for biliary tract cancers in Austria. Liver. Int. 2014, 34, 1102–1108. [Google Scholar] [CrossRef]
- Petrick, J.L.; Florio, A.A.; Znaor, A.; Ruggieri, D.; Laversanne, M.; Alvarez, C.S.; Ferlay, J.; Valery, P.C.; Bray, F.; McGlynn, K.A. International trends in hepatocellular carcinoma incidence, 1978–2012. Int. J. Cancer 2020, 147, 317–330. [Google Scholar] [CrossRef]
- Valery, P.C.; Laversanne, M.; Clark, P.J.; Petrick, J.L.; McGlynn, K.A.; Bray, F. Projections of primary liver cancer to 2030 in 30 countries worldwide. Hepatology 2018, 67, 600–611. [Google Scholar] [CrossRef] [Green Version]
- Bertuccio, P.; Turati, F.; Carioli, G.; Rodriguez, T.; La Vecchia, C.; Malvezzi, M.; Negri, E. Global trends and predictions in hepatocellular carcinoma mortality. J. Hepatol. 2017, 67, 302–309. [Google Scholar] [CrossRef]
- Anzenberger, J.B.M.; Gaiswinkler, S.; Grabenhofer-Eggerth, A.; Klein, C.; Schmutterer, I.; Schwarz, T.; Strizek, J. Epidemiologiebericht Sucht 2020. Illegale Drogen, Alkohol und Tabak. Gesundh. Osterr. Wien 2020. [Google Scholar]
- European Association for the Study of the Liver. Electronic address, e.e.e.; Clinical Practice Guidelines Panel, C.; representative, E.G.B.; Panel, m. EASL recommendations on treatment of hepatitis C: Final update of the series. J. Hepatol. 2020, 73, 1170–1218. [Google Scholar] [CrossRef]
- Waziry, R.; Hajarizadeh, B.; Grebely, J.; Amin, J.; Law, M.; Danta, M.; George, J.; Dore, G.J. Hepatocellular carcinoma risk following direct-acting antiviral HCV therapy: A systematic review, meta-analyses, and meta-regression. J. Hepatol. 2017, 67, 1204–1212. [Google Scholar] [CrossRef]
- Kanwal, F.; Kramer, J.R.; Asch, S.M.; Cao, Y.; Li, L.; El-Serag, H.B. Long-Term Risk of Hepatocellular Carcinoma in HCV Patients Treated With Direct Acting Antiviral Agents. Hepatology 2020, 71, 44–55. [Google Scholar] [CrossRef]
- WHO. Global Health Sector Strategy on Viral Hepatitis 2016–2021; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Schaefer, B.; Viveiros, A.; Al-Zoairy, R.; Blach, S.; Brandon, S.; Razavi, H.; Dorn, L.; Finkenstedt, A.; Effenberger, M.; Graziadei, I.; et al. Disease burden of hepatitis C in the Austrian state of Tyrol—Epidemiological data and model analysis to achieve elimination by 2030. PLoS ONE 2018, 13, e0200750. [Google Scholar] [CrossRef]
- Blach, S.; Kondili, L.A.; Aghemo, A.; Cai, Z.; Dugan, E.; Estes, C.; Gamkrelidze, I.; Ma, S.; Pawlotsky, J.M.; Razavi-Shearer, D.; et al. Impact of COVID-19 on global HCV elimination efforts. J. Hepatol. 2021, 74, 31–36. [Google Scholar] [CrossRef]
- Anstee, Q.M.; Reeves, H.L.; Kotsiliti, E.; Govaere, O.; Heikenwalder, M. From NASH to HCC: Current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 411–428. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, C.S.; Graubard, B.I.; Thistle, J.E.; Petrick, J.L.; McGlynn, K.A. Attributable Fractions of Nonalcoholic Fatty Liver Disease for Mortality in the United States: Results From the Third National Health and Nutrition Examination Survey With 27 Years of Follow-up. Hepatology 2020, 72, 430–440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanwal, F.; Kramer, J.R.; Mapakshi, S.; Natarajan, Y.; Chayanupatkul, M.; Richardson, P.A.; Li, L.; Desiderio, R.; Thrift, A.P.; Asch, S.M.; et al. Risk of Hepatocellular Cancer in Patients With Non-Alcoholic Fatty Liver Disease. Gastroenterology 2018, 155, 1828–1837.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hucke, F.; Sieghart, W.; Schoniger-Hekele, M.; Peck-Radosavljevic, M.; Muller, C. Clinical characteristics of patients with hepatocellular carcinoma in Austria - is there a need for a structured screening program? Wien Klin Wochenschr 2011, 123, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Bru, C.; Bruix, J. Prognosis of hepatocellular carcinoma: The BCLC staging classification. Semin. Liver. Dis. 1999, 19, 329–338. [Google Scholar] [CrossRef]
- Heimbach, J.K.; Kulik, L.M.; Finn, R.S.; Sirlin, C.B.; Abecassis, M.M.; Roberts, L.R.; Zhu, A.X.; Murad, M.H.; Marrero, J.A. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018, 67, 358–380. [Google Scholar] [CrossRef] [Green Version]
- Reig, M.; Forner, A.; Rimola, J.; Ferrer-Fabrega, J.; Burrel, M.; Garcia-Criado, A.; Kelley, R.K.; Galle, P.R.; Mazzaferro, V.; Salem, R.; et al. BCLC strategy for prognosis prediction and treatment recommendation Barcelona Clinic Liver Cancer (BCLC) staging system. The 2022 update. J. Hepatol. 2021, 76, 681–693. [Google Scholar] [CrossRef]
- Singal, A.G.; Pillai, A.; Tiro, J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: A meta-analysis. PLoS Med. 2014, 11, e1001624. [Google Scholar] [CrossRef]
- Zhao, C.; Jin, M.; Le, R.H.; Le, M.H.; Chen, V.L.; Jin, M.; Wong, G.L.; Wong, V.W.; Lim, Y.S.; Chuang, W.L.; et al. Poor adherence to hepatocellular carcinoma surveillance: A systematic review and meta-analysis of a complex issue. Liver. Int. 2018, 38, 503–514. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [Green Version]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef] [Green Version]
- Chau, I.; Peck-Radosavljevic, M.; Borg, C.; Malfertheiner, P.; Seitz, J.F.; Park, J.O.; Ryoo, B.Y.; Yen, C.J.; Kudo, M.; Poon, R.; et al. Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib: Patient-focused outcome results from the randomised phase III REACH study. Eur. J. Cancer 2017, 81, 17–25. [Google Scholar] [CrossRef]
- Finn, R.S.; Cheng, A.L. Atezolizumab and Bevacizumab in Hepatocellular Carcinoma. Reply. N. Engl. J. Med. 2020, 383, 695. [Google Scholar] [CrossRef]
- Bruix, J.; Chan, S.L.; Galle, P.R.; Rimassa, L.; Sangro, B. Systemic treatment of hepatocellular carcinoma: An EASL position paper. J. Hepatol. 2021, 75, 960–974. [Google Scholar] [CrossRef]
- Rutherford, M.J.; Arnold, M.; Bardot, A.; Ferlay, J.; De, P.; Tervonen, H.; Little, A.; Bucher, O.; St Jacques, N.; Gavin, A.; et al. Comparison of liver cancer incidence and survival by subtypes across seven high-income countries. Int. J. Cancer 2021, 149, 2020–2031. [Google Scholar] [CrossRef]
- Saha, S.K.; Zhu, A.X.; Fuchs, C.S.; Brooks, G.A. Forty-Year Trends in Cholangiocarcinoma Incidence in the U.S.: Intrahepatic Disease on the Rise. Oncologist 2016, 21, 594–599. [Google Scholar] [CrossRef] [Green Version]
- Khan, J.; Ullah, A.; Matolo, N.; Waheed, A.; Nama, N.; Khan, T.; Tareen, B.; Khan, Z.; Singh, S.G.; Cason, F.D. Klatskin Tumor in the Light of ICD-O-3: A Population-Based Clinical Outcome Study Involving 1,144 Patients from the Surveillance, Epidemiology, and End Result (SEER) Database (2001–2012). Cureus 2021, 13, e18941. [Google Scholar] [CrossRef]
- Khan, S.A.; Emadossadaty, S.; Ladep, N.G.; Thomas, H.C.; Elliott, P.; Taylor-Robinson, S.D.; Toledano, M.B. Rising trends in cholangiocarcinoma: Is the ICD classification system misleading us? J. Hepatol. 2012, 56, 848–854. [Google Scholar] [CrossRef]
- Hainsworth, J.D.; Rubin, M.S.; Spigel, D.R.; Boccia, R.V.; Raby, S.; Quinn, R.; Greco, F.A. Molecular gene expression profiling to predict the tissue of origin and direct site-specific therapy in patients with carcinoma of unknown primary site: A prospective trial of the Sarah Cannon research institute. J. Clin. Oncol. 2013, 31, 217–223. [Google Scholar] [CrossRef]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef] [Green Version]
- Manne, A.; Woods, E.; Tsung, A.; Mittra, A. Biliary Tract Cancers: Treatment Updates and Future Directions in the Era of Precision Medicine and Immuno-Oncology. Front. Oncol. 2021, 11, 768009. [Google Scholar] [CrossRef]
- Valle, J.W.; Borbath, I.; Khan, S.A.; Huguet, F.; Gruenberger, T.; Arnold, D.; Committee, E.G. Biliary cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, v28–v37. [Google Scholar] [CrossRef]
- Oh, D.-Y.; He, A.R.; Qin, S.; Chen, L.-T.; Okusaka, T.; Vogel, A.; Kim, J.W.; Suksombooncharoen, T.; Lee, M.A.; Kitano, M.; et al. A phase 3 randomized, double-blind, placebo-controlled study of durvalumab in combination with gemcitabine plus cisplatin (GemCis) in patients (pts) with advanced biliary tract cancer (BTC): TOPAZ-1. J. Clin. Oncol. 2022, 40, 378. [Google Scholar] [CrossRef]
- Hsing, A.W.; Gao, Y.T.; Han, T.Q.; Rashid, A.; Sakoda, L.C.; Wang, B.S.; Shen, M.C.; Zhang, B.H.; Niwa, S.; Chen, J.; et al. Gallstones and the risk of biliary tract cancer: A population-based study in China. Br. J. Cancer 2007, 97, 1577–1582. [Google Scholar] [CrossRef]
- Gunasekaran, G.; Bekki, Y.; Lourdusamy, V.; Schwartz, M. Surgical Treatments of Hepatobiliary Cancers. Hepatology 2021, 73 (Suppl. S1), 128–136. [Google Scholar] [CrossRef]
- Rizzo, A.; Dadduzio, V.; Lombardi, L.; Ricci, A.D.; Gadaleta-Caldarola, G. Ampullary Carcinoma: An Overview of a Rare Entity and Discussion of Current and Future Therapeutic Challenges. Curr. Oncol. 2021, 28, 293. [Google Scholar] [CrossRef]
- Rumgay, H.; Ferlay, J.; de Martel, C.; Georges, D.; Ibrahim, A.S.; Zheng, R.; Wei, W.; Lemmens, V.E.P.P.; Soerjomataram, I. Global, regional and national burden of primary liver cancer by subtype. Eur. J. Cancer 2022, 161, 108–118. [Google Scholar] [CrossRef]
- Hackl, M.I.P. Einfluss der neuen Europäischen Standardbevölkerung 2013 auf die Trendentwicklung der österreichischen Krebsinzidenzraten 1983–2014. Stat. Nachr. 2017, 2, 106–114. [Google Scholar]
- Johnson, P.; Zhou, Q.; Dao, D.Y.; Lo, Y. Circulating biomarkers in the diagnosis and management of hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.M.; Vogel, A.; Arrese, M.; Balderramo, D.C.; Valle, J.W.; Banales, J.M. Next-Generation Biomarkers for Cholangiocarcinoma. Cancers 2021, 13, 3222. [Google Scholar] [CrossRef] [PubMed]
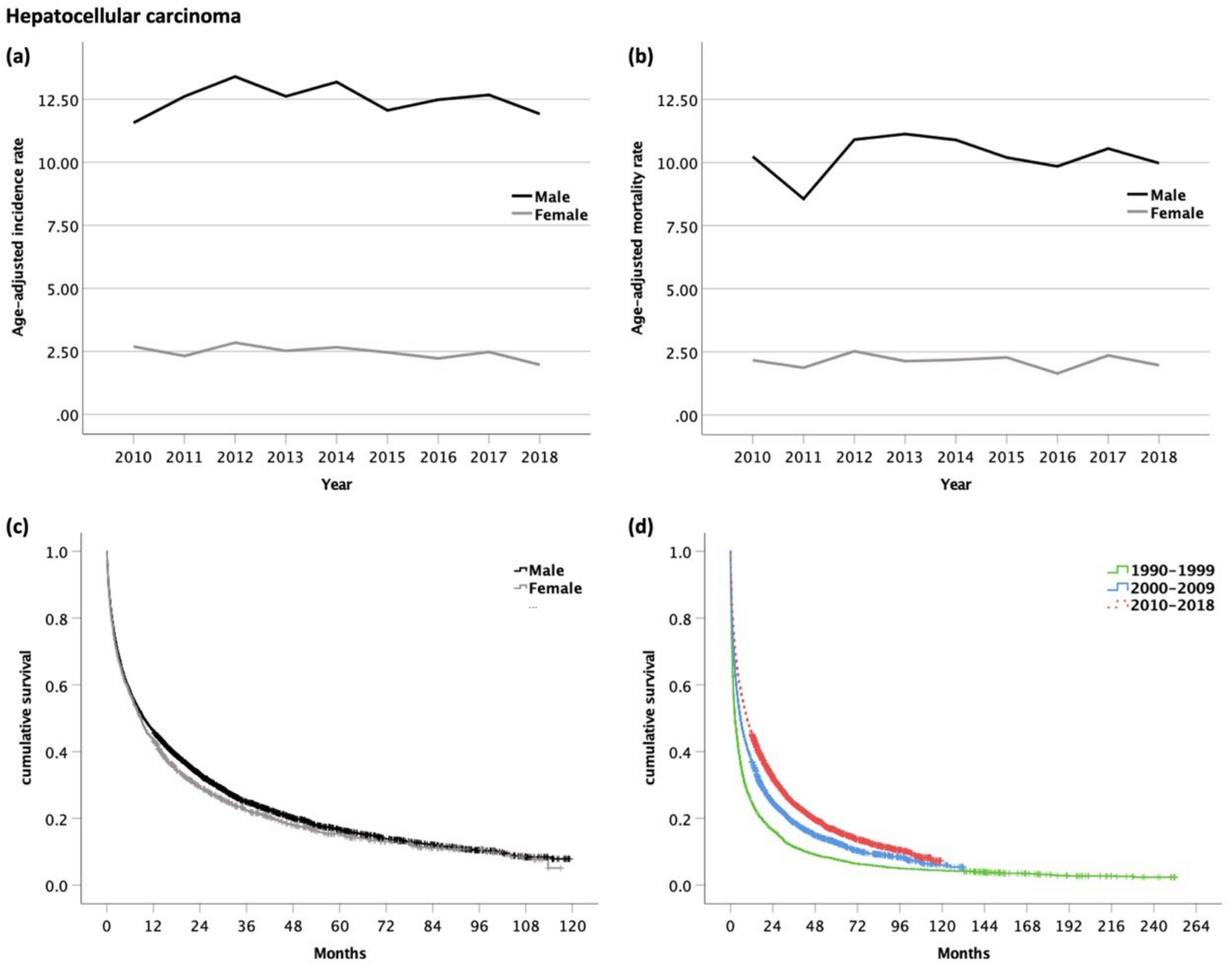
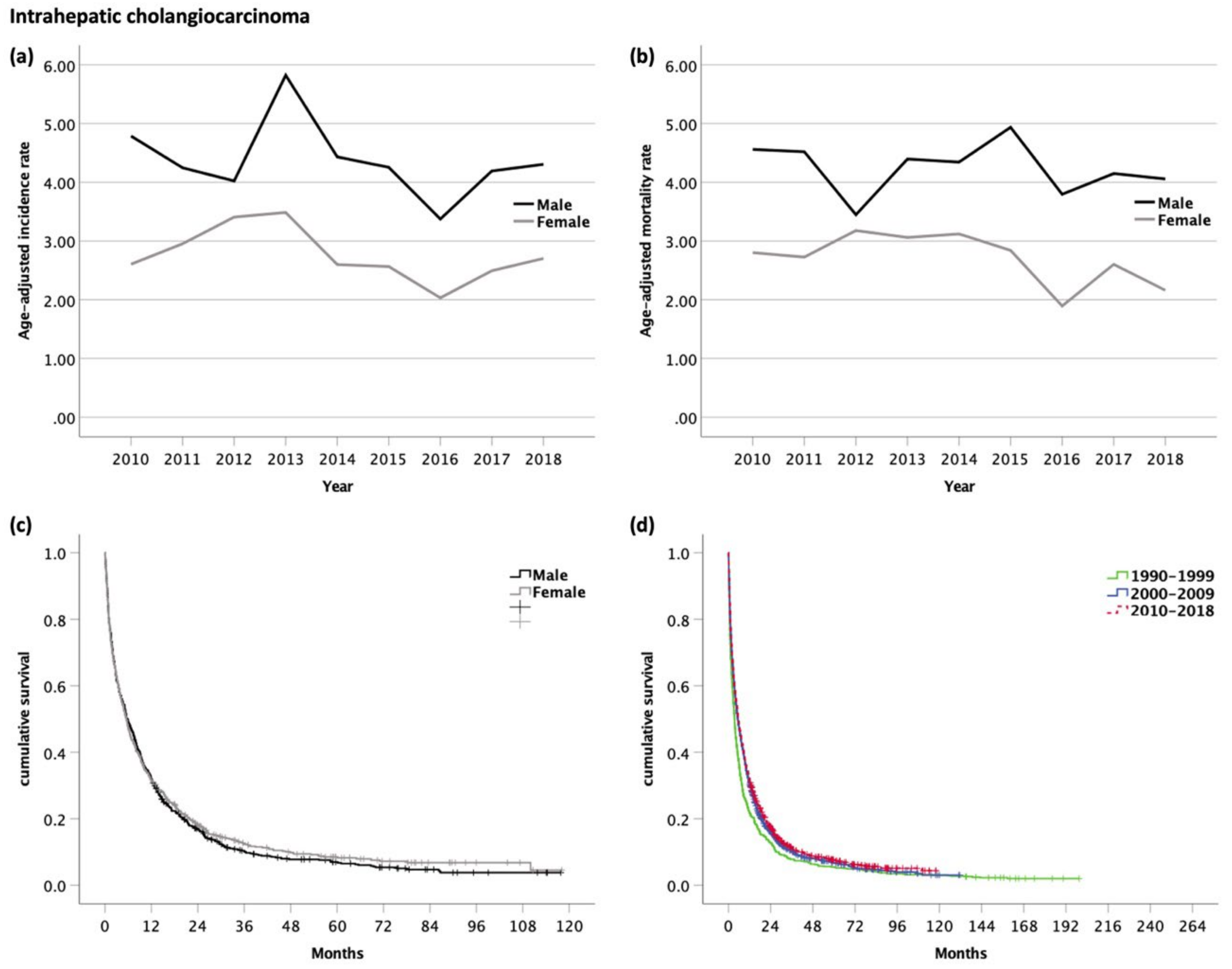
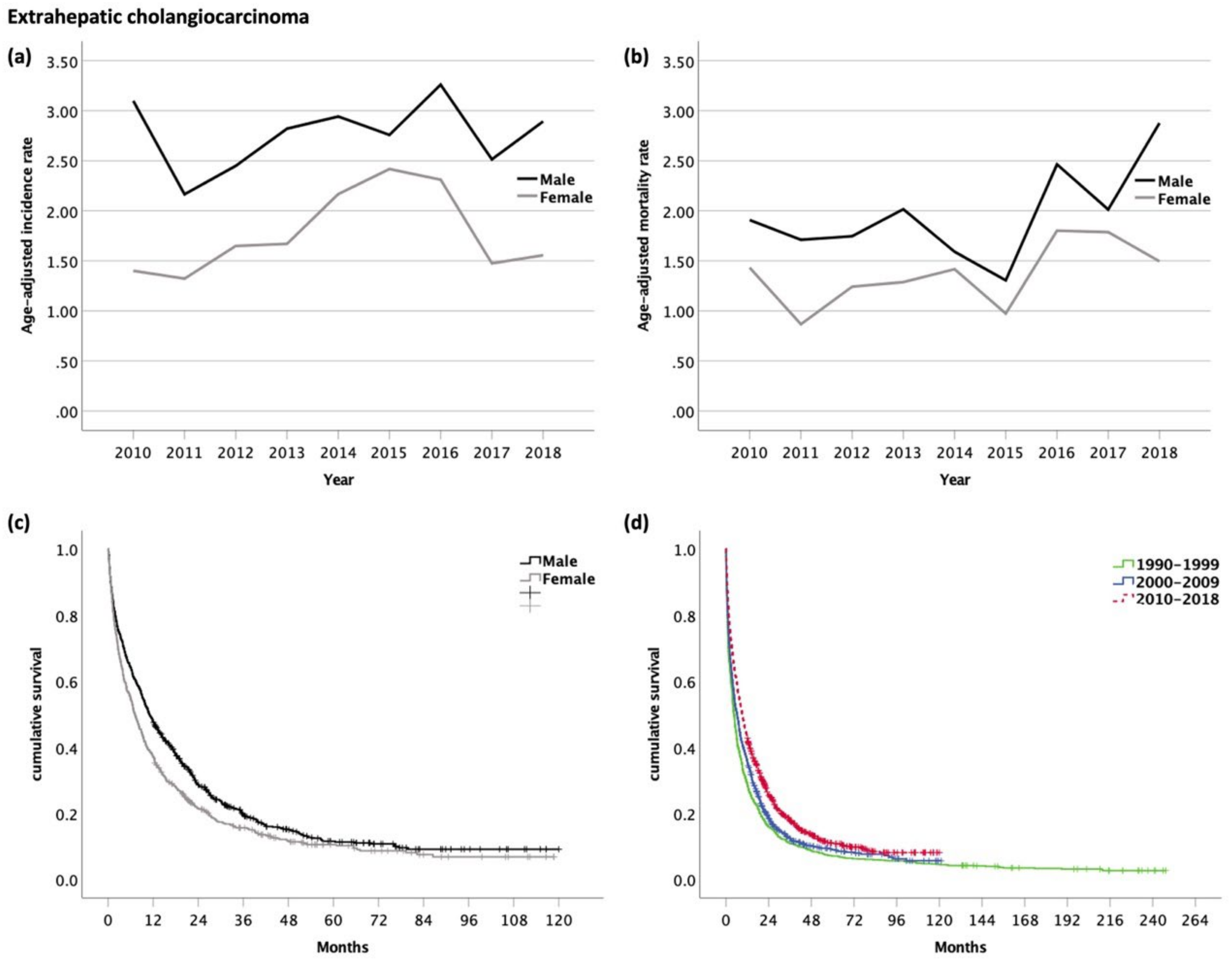
| All Analyzed Patients (n = 12,577) | HCC (n = 7146) | iCC (n = 1858) | eCC (n = 1649) | GBC (n = 1365) | AC (n = 559) | ||
|---|---|---|---|---|---|---|---|
| Variable | n (%) | n (%) | n (%) | n (%) | n (%) | ||
| Age | Mean ± SD | 71 ± 11.7 | 70 ± 11.5 | 71 ± 12.0 | 73 ± 11.4 | 71 ± 11.2 | 71 ± 12.2 |
| Sex | Male | 7959 (63) | 5342 (75) | 997 (54) | 884 (54) | 437 (32) | 299 (54) |
| (n (%)) | Female | 4618 (37) | 1804 (25) | 861 (46) | 765 (46) | 928 (68) | 260 (46) |
| Metastases (n (%)) | Lymph node 1 | 1190 (10) | 310 (4) | 167(9) | 289 (18) | 255 (19) | 169 (30) |
| Distant 2 | 1489 (12) | 574 (8) | 412 (22) | 214 (13) | 343 (25) | 43 (8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hucke, F.; Pinter, M.; Hucke, M.; Bota, S.; Bolf, D.; Hackl, M.; Peck-Radosavljevic, M. Changing Epidemiological Trends of Hepatobiliary Carcinomas in Austria 2010–2018. Cancers 2022, 14, 3093. https://doi.org/10.3390/cancers14133093
Hucke F, Pinter M, Hucke M, Bota S, Bolf D, Hackl M, Peck-Radosavljevic M. Changing Epidemiological Trends of Hepatobiliary Carcinomas in Austria 2010–2018. Cancers. 2022; 14(13):3093. https://doi.org/10.3390/cancers14133093
Chicago/Turabian StyleHucke, Florian, Matthias Pinter, Miriam Hucke, Simona Bota, Dajana Bolf, Monika Hackl, and Markus Peck-Radosavljevic. 2022. "Changing Epidemiological Trends of Hepatobiliary Carcinomas in Austria 2010–2018" Cancers 14, no. 13: 3093. https://doi.org/10.3390/cancers14133093
APA StyleHucke, F., Pinter, M., Hucke, M., Bota, S., Bolf, D., Hackl, M., & Peck-Radosavljevic, M. (2022). Changing Epidemiological Trends of Hepatobiliary Carcinomas in Austria 2010–2018. Cancers, 14(13), 3093. https://doi.org/10.3390/cancers14133093






