Current Evidence on Immunotherapy for Gestational Trophoblastic Neoplasia (GTN)
Abstract
Simple Summary
Abstract
1. Introduction
1.1. Gestational Trophoblastic Disease
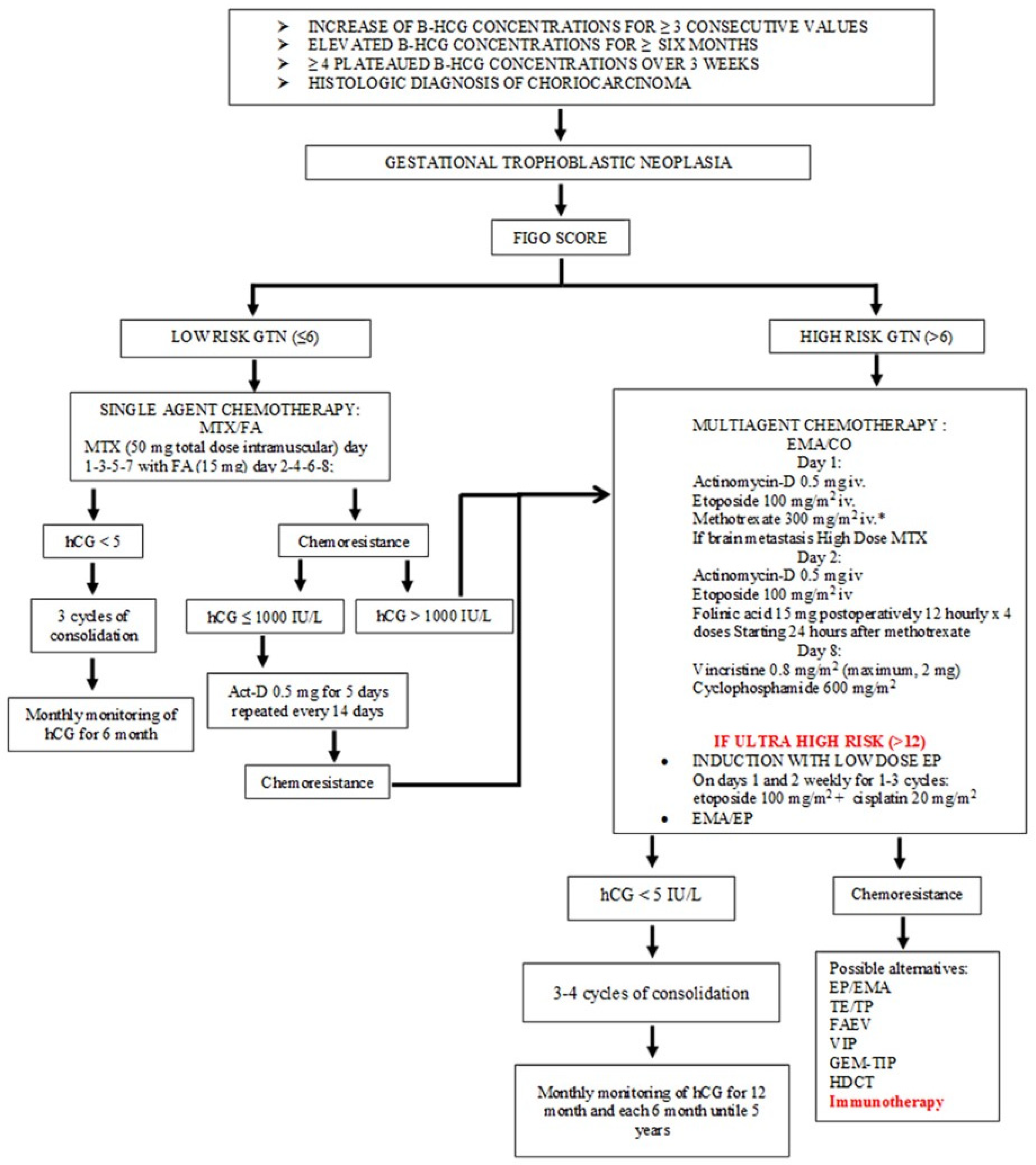
1.2. Standard Treatment in Gestational Trophoblastic Neoplasia
2. Materials and Methods
3. Results
Immunotherapy and GTN
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Seckl, M.J.; Sebire, N.J.; Berkowitz, R.S. Gestational trophoblastic disease. Lancet 2010, 376, 717–729. [Google Scholar] [CrossRef]
- Altieri, A.; Franceschi, S.; Ferlay, J.; Smith, J.; La Vecchia, C. Epidemiology and aetiology of gestational trophoblastic diseases. Lancet Oncol. 2003, 4, 670–678. [Google Scholar] [CrossRef]
- Mangili, G.; Lorusso, D.; Brown, J.; Pfisterer, J.; Massuger, L.; Vaughan, M.; Ngan, H.Y.; Golfier, F.; Sekharan, P.K.; Charry, R.C.; et al. Trophoblastic disease review for diagnosis and management: A joint report from the International Society for the Study of Trophoblastic Disease, European Organisation for the Treatment of Trophoblastic Disease, and the Gynecologic Cancer InterGroup. Int. J. Gynecol. Cancer 2014, 24 (Suppl. S3), S109–S116. [Google Scholar] [CrossRef] [PubMed]
- Hui, P.; Buza, N.; Murphy, K.M.; Ronnett, B.M. Hydatidiform Moles: Genetic Basis and Precision Diagnosis. Annu. Rev. Pathol. 2017, 12, 449–485. [Google Scholar] [CrossRef]
- Lok, C.; van Trommel, N.; Massuger, L.; Golfier, F.; Seckl, M. Practical clinical guidelines of the EOTTD for treatment and referral of gestational trophoblastic disease. Eur. J. Cancer 2020, 130, 228–240. [Google Scholar] [CrossRef]
- Elias, K.M.; Berkowitz, R.S.; Horowitz, N.S. State-of-the-Art Workup and Initial Management of Newly Diagnosed Molar Pregnancy and Postmolar Gestational Trophoblastic Neoplasia. J. Natl. Compr. Cancer Netw. 2019, 17, 1396–1401. [Google Scholar] [CrossRef]
- Ngan, H.Y.; Bender, H.; Benedet, J.L.; Jones, H.; Montruccoli, G.C.; Pecorelli, S. FIGO Committee on Gynecologic Oncology. Gestational trophoblastic neoplasia, FIGO 2000 staging and classification. Int. J. Gynaecol. Obstet. 2003, 83 (Suppl. S1), 175–177, Erratum in Int. J. Gynaecol. Obstet. 2021, 155, 563. [Google Scholar] [CrossRef]
- Abu-Rustum, N.R.; Yashar, C.M.; Bradley, K.; Brooks, R.; Campos, S.M.; Chino, J.; Chon, H.S.; Cohn, D.; Chu, C.; Cohn, D.; et al. Gestational Trophoblastic Neoplasia, Version 1.2022, NCCN Clinical Practice Guidelines in Oncology. Online to NCCN.org. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1489 (accessed on 20 May 2022).
- Ning, F.; Hou, H.; Morse, A.N.; Lash, G.E. Understanding and management of gestational trophoblastic disease. F1000Research 2019, 8, 428. [Google Scholar] [CrossRef]
- Horowitz, N.S.; Goldstein, D.P.; Berkowitz, R.S. Placental site trophoblastic tumors and epithelioid trophoblastic tumors: Biology, natural history, and treatment modalities. Gynecol. Oncol. 2017, 144, 208–214. [Google Scholar] [CrossRef]
- Palmer, J.E.; Macdonald, M.; Wells, M.; Hancock, B.W.; Tidy, J.A. Epithelioid trophoblastic tumor: A review of the literature. J. Reprod. Med. 2008, 53, 465–475. [Google Scholar]
- Gadducci, A.; Carinelli, S.; Guerrieri, M.E.; Aletti, G.D. Placental site trophoblastic tumor and epithelioid trophoblastic tumor: Clinical and pathological features, prognostic variables and treatment strategy. Gynecol. Oncol. 2019, 153, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Frega, G.; Kepp, O.; Turchetti, D.; Rizzo, A.; Pantaleo, M.A.; Brandi, G. Hypothesis on the possible relevance of the immunogenic cell death in the treatment of gestational trophoblastic neoplasms. Transl. Oncol. 2021, 12, 101224. [Google Scholar] [CrossRef] [PubMed]
- King, J.R.; Wilson, M.L.; Hetey, S.; Kiraly, P.; Matsuo, K.; Castaneda, A.V.; Toth, E.; Krenacs, T.; Hupuczi, P.; Mhawech-Fauceglia, P.; et al. Dysregulation of Placental Functions and Immune Pathways in Complete Hydatidiform Moles. Int. J. Mol. Sci. 2019, 20, 4999. [Google Scholar] [CrossRef] [PubMed]
- Szabolcsi, Z.; Demeter, A.; Kiraly, P.; Balogh, A.; Wilson, M.L.; King, J.R.; Hetey, S.; Gelencser, Z.; Matsuo, K.; Hargitai, B.; et al. Epigenetic Dysregulation of Trophoblastic Gene Expression in Gestational Trophoblastic Disease. Biomedicines 2021, 9, 1935. [Google Scholar] [CrossRef] [PubMed]
- Gomez, S.; Tabernacki, T.; Kobyra, J.; Roberts, P.; Chiappinelli, K.B. Combining epigenetic and immune therapy to overcome cancer resistance. Semin. Cancer Biol. 2020, 65, 99–113. [Google Scholar] [CrossRef]
- Mangili, G.; Cioffi, R.; Bergamini, A.; Sabetta, G.; Vasta, F.; Candotti, G.; Rabaiotti, E.; Petrone, M.; Taccagni, G.; Bocciolone, L.; et al. Gestational trophoblastic disease: An update on pathology, diagnosis and state of-the-art management. J. Gynaecol. Obstet. 2021, 33, 90–101. [Google Scholar] [CrossRef]
- Ngan, H.Y.S.; Seckl, M.J.; Berkowitz, R.S.; Xiang, Y.; Golfier, F.; Sekharan, P.K.; Lurain, J.R.; Massuger, L. Diagnosis and management of gestational trophoblastic disease: 2021 update. Int. J. Gynaecol. Obstet. 2021, 1, 86–93. [Google Scholar] [CrossRef]
- Braga, A.; Mora, P.; de Melo, A.C.; Nogueira-Rodrigues, A.; Amim-Junior, J.; Rezende-Filho, J.; Seckl, M.J. Challenges in the diagnosis and treatment of gestational trophoblastic neoplasia worldwide. World J. Clin. Oncol. 2019, 10, 28–37. [Google Scholar] [CrossRef]
- Clark, J.; Slater, S.; Seckl, M.J. Treatment of Gestational Trophoblastic Disease in the 2020s. Curr. Opin. Obstet. Gynecol. 2021, 33, 7–12. [Google Scholar] [CrossRef]
- Poli, J.G.; Paiva, G.; Freitas, F.; Mora, P.; Velarde, L.G.C.; Junior, J.A.; Filho, J.R.; Elias, K.M.; Horowitz, N.S.; Braga, A.; et al. Folinic acid rescue during methotrexate treatment for low-risk gestational trophoblastic neoplasia—How much is just right? Gynecol. Oncol. 2021, 162, 638–644. [Google Scholar] [CrossRef]
- Maestá, I.; Nitecki, R.; Horowitz, N.S.; Goldstein, D.P.; de Freitas Segalla Moreira, M.; Elias, K.M.; Berkowitz, R.S. Effectiveness and toxicity of first-line methotrexate chemotherapy in low-risk postmolar gestational trophoblastic neoplasia: The New England Trophoblastic Disease Center experience. Gynecol. Oncol. 2018, 148, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Santaballa, A.; García, Y.; Herrero, A.; Laínez, N.; Fuentes, J.; De Juan, A.; Rodriguez Freixinós, V.; Aparicio, J.; Casado, A.; García-Martinez, E. SEOM clinical guidelines in gestational trophoblastic disease (2017). Clin. Transl. Oncol. 2018, 20, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Maestá, I.; Nitecki, R.; Desmarais, C.C.F.; Horowitz, N.S.; Goldstein, D.P.; Elias, K.M.; Berkowitz, R.S. Effectiveness and toxicity of second-line actinomycin D in patients with methotrexate-resistant postmolar low-risk gestational trophoblastic neoplasia. Gynecol. Oncol. 2020, 157, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Gillett, S.; Ireson, J.; Hills, A.; Tidy, J.A.; Coleman, R.E.; Hancock, B.W.; Winter, M.C. M-EA (methotrexate, etoposide, dactinomycin) and EMA-CO (methotrexate, etoposide, dactinomycin/cyclophosphamide, vincristine) regimens as first-line treatment of high-risk gestational trophoblastic neoplasia. Int. J. Cancer 2021, 148, 2335–2344. [Google Scholar] [CrossRef] [PubMed]
- Alifrangis, C.; Agarwal, R.; Short, D.; Fisher, R.A.; Sebire, N.J.; Harvey, R.; Savage, P.M.; Seckl, M.J. EMA/CO for high-risk gestational trophoblastic neoplasia: Good outcomes with induction low-dose etoposide-cisplatin and genetic analysis. J. Clin. Oncol. 2013, 31, 280–286. [Google Scholar] [CrossRef]
- Braga, A.; Elias, K.M.; Horowitz, N.S.; Berkowitz, R.S. Treatment of high-risk gestational trophoblastic neoplasia and chemoresistance/relapsed disease. Best Pract. Res. Clin. Obstet. Gynaecol. 2021, 74, 81–96. [Google Scholar] [CrossRef]
- Di Mattei, V.E.; Carnelli, L.; Mazzetti, M.; Bernardi, M.; Di Pierro, R.; Bergamini, A.; Mangili, G.; Candiani, M.; Sarno, L. Mental Representations of Illness in Patients with Gestational Trophoblastic Disease: How Do Patients Perceive Their Condition? PLoS ONE 2016, 11, e0153869. [Google Scholar] [CrossRef]
- Benigno, B.B. High-dose chemotherapy with autologous stem cell support as salvage therapy in recurrent gestational trophoblastic disease. Int. J. Gynecol. Cancer 2013, 23, 1331–1333. [Google Scholar] [CrossRef]
- Yamamoto, E.; Niimi, K.; Fujikake, K.; Nishida, T.; Murata, M.; Mitsuma, A.; Ando, Y.; Kikkawa, F. High-dose chemotherapy with autologous peripheral blood stem cell transplantation for choriocarcinoma: A case report and literature review. Mol. Clin. Oncol. 2016, 5, 660–664. [Google Scholar] [CrossRef][Green Version]
- El-Helw, L.M.; Seckl, M.J.; Haynes, R.; Evans, L.S.; Lorigan, P.C.; Long, J.; Kanfer, E.J.; Newlands, E.S.; Hancock, B.W. High-dose chemotherapy and peripheral blood stem cell support in refractory gestational trophoblastic neoplasia. Br. J. Cancer 2005, 93, 620–621. [Google Scholar] [CrossRef]
- Frijstein, M.M.; Lok, C.A.R.; Short, D.; Singh, K.; Fisher, R.A.; Hancock, B.W.; Tidy, J.A.; Sarwar, N.; Kanfer, E.; Winter, M.C.; et al. The results of treatment with high-dose chemotherapy and peripheral blood stem cell support for gestational trophoblastic neoplasia. Eur. J. Cancer 2019, 109, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Bolze, P.A.; Patrier, S.; Massardier, J.; Hajri, T.; Abbas, F.; Schott, A.M.; Allias, F.; Devouassoux-Shisheboran, M.; Freyer, G.; Golfier, F.; et al. PD-L1 Expression in Premalignant and Malignant Trophoblasts from Gestational Trophoblastic Diseases Is Ubiquitous and Independent of Clinical Outcomes. Int. J. Gynecol. Cancer 2017, 27, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Veras, E.; Kurman, R.J.; Wang, T.L.; Shih, I.M. PD-L1 Expression in Human Placentas and Gestational Trophoblastic Diseases. Int. J. Gynecol. Pathol. 2017, 36, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Inaguma, S.; Wang, Z.; Lasota, J.; Sarlomo-Rikala, M.; McCue, P.A.; Ikeda, H.; Miettinen, M. Comprehensive Immunohistochemical Study of Programmed Cell Death Ligand 1 (PD-L1): Analysis in 5536 Cases Revealed Consistent Expression in Trophoblastic Tumors. Am. J. Surg. Pathol. 2016, 40, 1133–1142. [Google Scholar] [CrossRef]
- Keir, M.E.; Butte, M.J.; Freeman, G.J.; Sharpe, A.H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 2008, 26, 677–704. [Google Scholar] [CrossRef] [PubMed]
- Tumeh, P.C.; Harview, C.L.; Yearley, J.H.; Shintaku, I.P.; Taylor, E.J.; Robert, L.; Chmielowski, B.; Spasic, M.; Henry, G.; Ciobanu, V.; et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 2014, 515, 568–571. [Google Scholar] [CrossRef] [PubMed]
- Apetoh, L.; Smyth, M.J.; Drake, C.G.; Abastado, J.P.; Apte, R.N.; Ayyoub, M.; Blay, J.Y.; Bonneville, M.; Butterfield, L.H.; Caignard, A.; et al. Consensus nomenclature for CD8(+) T cell phenotypes in cancer. Oncoimmunology 2015, 4, e998538. [Google Scholar] [CrossRef] [PubMed]
- Gatalica, Z.; Snyder, C.; Maney, T.; Ghazalpour, A.; Holterman, D.A.; Xiao, N.; Overberg, P.; Rose, I.; Basu, G.D.; Vranic, S.; et al. Programmed cell death 1 (PD-1) and its ligand (PD-L1) in common cancers and their correlation with molecular cancer type. Cancer Epidemiol. Biomark. Prev. 2014, 23, 2965–2970. [Google Scholar] [CrossRef]
- Grywalska, E.; Sobstyl, M.; Putowski, L.; Roliński, J. Current Possibilities of Gynecologic Cancer Treatment with the Use of Immune Checkpoint Inhibitors. Int. J. Mol. Sci. 2019, 20, 4705. [Google Scholar] [CrossRef]
- Herbst, R.S.; Baas, P.; Kim, D.W.; Felip, E.; Pérez-Gracia, J.L.; Han, J.Y.; Molina, J.; Kim, J.H.; Arvis, C.D.; Ahn, M.J.; et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Lipson, E.J.; Forde, P.M.; Hammers, H.J.; Emens, L.A.; Taube, J.M.; Topalian, S.L. Antagonists of PD-1 and PD-L1 in Cancer Treatment. Semin. Oncol. 2015, 42, 587–600. [Google Scholar] [CrossRef] [PubMed]
- Kwok, G.; Yau, T.C.; Chiu, J.W.; Tse, E.; Kwong, Y.L. Pembrolizumab (Keytruda). Hum. Vaccin. Immunother. 2016, 12, 2777–2789. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Chaudhary, N.; Garg, M.; Floudas, C.S.; Soni, P.; Chandra, A.B. Current Diagnosis and Management of Immune Related Adverse Events (irAEs) Induced by Immune Checkpoint Inhibitor Therapy. Front. Pharmacol. 2017, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Pinto, A.; Castillo, R.P.; Slomovitz, B.M. Complete Serologic Response to Pembrolizumab in a Woman With Chemoresistant Metastatic Choriocarcinoma. Clin. Oncol. 2017, 35, 3172–3174. [Google Scholar] [CrossRef]
- Ghorani, E.; Kaur, B.; Fisher, R.A.; Short, D.; Joneborg, U.; Carlson, J.W.; Akarca, A.; Marafioti, T.; Quezada, S.A.; Sarwar, N.; et al. Pembrolizumab is effective for drug-resistant gestational trophoblastic neoplasia. Lancet 2017, 390, 2343–2345. [Google Scholar] [CrossRef]
- Chul Choi, M.; Oh, J.; Lee, C. Effective anti-programmed cell death 1 treatment for chemoresistant gestational trophoblastic neoplasia. Eur. J. Cancer 2019, 121, 94–97. [Google Scholar] [CrossRef]
- Goldfarb, J.A.; Dinoi, G.; Mariani, A.; Langstraat, C.L. A case of multiagent drug resistant choriocarcinoma treated with Pembrolizumab. Gynecol. Oncol. Rep. 2020, 32, 100574. [Google Scholar] [CrossRef]
- Clair, K.H.; Gallegos, N.; Bristow, R.E. Successful treatment of metastatic refractory gestational choriocarcinoma with pembrolizumab: A case for immune checkpoint salvage therapy in trophoblastic tumors. Gynecol. Oncol. Rep. 2020, 34, 100625. [Google Scholar] [CrossRef]
- Pisani, D.; Calleja-Agius, J.; Di Fiore, R.; O’Leary, J.J.; Beirne, J.P.; O’Toole, S.A.; Felix, A.; Said-Huntingford, I. Epithelioid Trophoblastic Tumour: A Case with Genetic Linkage to a Child Born over Seventeen Years Prior, Successfully Treated with Surgery and Pembrolizumab. Curr. Oncol. 2021, 28, 5346–5355. [Google Scholar] [CrossRef]
- Bell, S.G.; Uppal, S.; Sakala, M.D.; Sciallis, A.P.; Rolston, A. An extrauterine extensively metastatic epithelioid trophoblastic tumor responsive to pembrolizumab. Gynecol. Oncol. Rep. 2021, 37, 100819. [Google Scholar] [CrossRef]
- Paspalj, V.; Polterauer, S.; Poetsch, N.; Reinthaller, A.; Grimm, C.; Bartl, T. Long-term survival in multiresistant metastatic choriocarcinoma after pembrolizumab treatment: A case report. Gynecol. Oncol. Rep. 2021, 37, 100817. [Google Scholar] [CrossRef] [PubMed]
- You, B.; Bolze, P.A.; Lotz, J.P.; Massardier, J.; Gladieff, L.; Joly, F.; Hajri, T.; Maucort-Boulch, D.; Bin, S.; Rousset, P.; et al. Avelumab in Patients with Gestational Trophoblastic Tumors with Resistance to Single-Agent Chemotherapy: Cohort A of the TROPHIMMUN Phase II Trial. J. Clin. Oncol. 2020, 38, 3129–3137. [Google Scholar] [CrossRef] [PubMed]
- Lurain, J.R.; Schink, J.C. Immunotherapy Versus Chemotherapy for Methotrexate-Resistant Low-Risk Gestational Trophoblastic Neoplasia. J. Clin. Oncol. 2020, 38, 4349–4350. [Google Scholar] [CrossRef]
- ClinicalTrials.gov Identifier: NCT04396223. Available online: https://clinicaltrials.gov/ct2/show/NCT04396223 (accessed on 20 May 2022).
- Cheng, H.; Zong, L.; Kong, Y.; Wang, X.; Gu, Y.; Cang, W.; Zhao, J.; Wan, X.; Yang, J.; Xiang, Y. Camrelizumab plus apatinib in patients with high-risk chemorefractory or relapsed gestational trophoblastic neoplasia (CAP 01): A single-arm, open-label, phase 2 trial. Lancet Oncol. 2021, 22, 1609–1617. [Google Scholar] [CrossRef]
| FIGO STAGING | |
|---|---|
| Stage I | GTN confined to the uterus |
| Stage II | GTN extends to the other genital structures |
| Stage III | GTN extends to the lungs, with or without genital tract involvement |
| Stage IV | All other distant metastases |
| Prognostic Factors | Score 1 | Score 2 | Score 3 | Score 4 |
|---|---|---|---|---|
| Age | <40 | > or =40 | ||
| Antecedent gestation | Mole | Abortion | Term | |
| Interval in months prior to end of antecedent pregnancy and start of treatment | <4 | 4–6 | 7–12 | >12 |
| Largest tumor size | <3 | 3–4 | > or =5 | |
| Site of metastases | Lung | Spleen, kidney | Gastrointestinal tract | Brain, liver |
| Number of metastases | 1–4 | 5–8 | >8 | |
| Pretreatment serum hCG (IU/L) | <103 | 103–104 | 104–105 | >105 |
| Previously failed chemotherapy | Single drug | Two or more drugs |
| References | Tumor Type | PD-L1 Expression | Pembrolizumab Cycles to hCG Normalization | Pembrolizumab Cycles as Consolidation | Response |
|---|---|---|---|---|---|
| Huang et al., 2017 [45] | Choriocarcinoma | Strong | 2 | 4 | CR |
| Ghorani et al., 2017 [46] | Choriocarcinoma | 100% | 4 | 5 | CR |
| PSTT/ETT | >90% | 5 | 0 | PD | |
| PSTT | >90% | 8 | 5 | CR | |
| Choriocarcinoma | 100% | 2 | 5 | CR | |
| Chul Choi et al., 2019 [47] | PSTT | 100% | 1 | 13 | CR |
| ETT | 50% | 11 | 4 | PR | |
| Goldfarb et al., 2020 [48] | Choriocarcinoma | 100% | 3 | 3 | CR |
| Clair et al., 2020 [49] | Choriocarcinoma | Strong | 10 | 0 | CR |
| Pisani et al., 2021 [50] | ETT | Not evaluated | Undeclared | Undeclared | CR |
| Bell et al., 2021 [51] | ETT | >5% | Ongoing | Ongoing | PR (Cut-off of 29 cycles) |
| Paspalj et al., 2021 [52] | Choriocarcinoma | >90% | 4 | 7 | CR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mangili, G.; Sabetta, G.; Cioffi, R.; Rabaiotti, E.; Candotti, G.; Pella, F.; Candiani, M.; Bergamini, A. Current Evidence on Immunotherapy for Gestational Trophoblastic Neoplasia (GTN). Cancers 2022, 14, 2782. https://doi.org/10.3390/cancers14112782
Mangili G, Sabetta G, Cioffi R, Rabaiotti E, Candotti G, Pella F, Candiani M, Bergamini A. Current Evidence on Immunotherapy for Gestational Trophoblastic Neoplasia (GTN). Cancers. 2022; 14(11):2782. https://doi.org/10.3390/cancers14112782
Chicago/Turabian StyleMangili, Giorgia, Giulia Sabetta, Raffaella Cioffi, Emanuela Rabaiotti, Giorgio Candotti, Francesca Pella, Massimo Candiani, and Alice Bergamini. 2022. "Current Evidence on Immunotherapy for Gestational Trophoblastic Neoplasia (GTN)" Cancers 14, no. 11: 2782. https://doi.org/10.3390/cancers14112782
APA StyleMangili, G., Sabetta, G., Cioffi, R., Rabaiotti, E., Candotti, G., Pella, F., Candiani, M., & Bergamini, A. (2022). Current Evidence on Immunotherapy for Gestational Trophoblastic Neoplasia (GTN). Cancers, 14(11), 2782. https://doi.org/10.3390/cancers14112782









