Combined Application of Patient-Derived Cells and Biomaterials as 3D In Vitro Tumor Models
Abstract
:Simple Summary
Abstract
1. Introduction
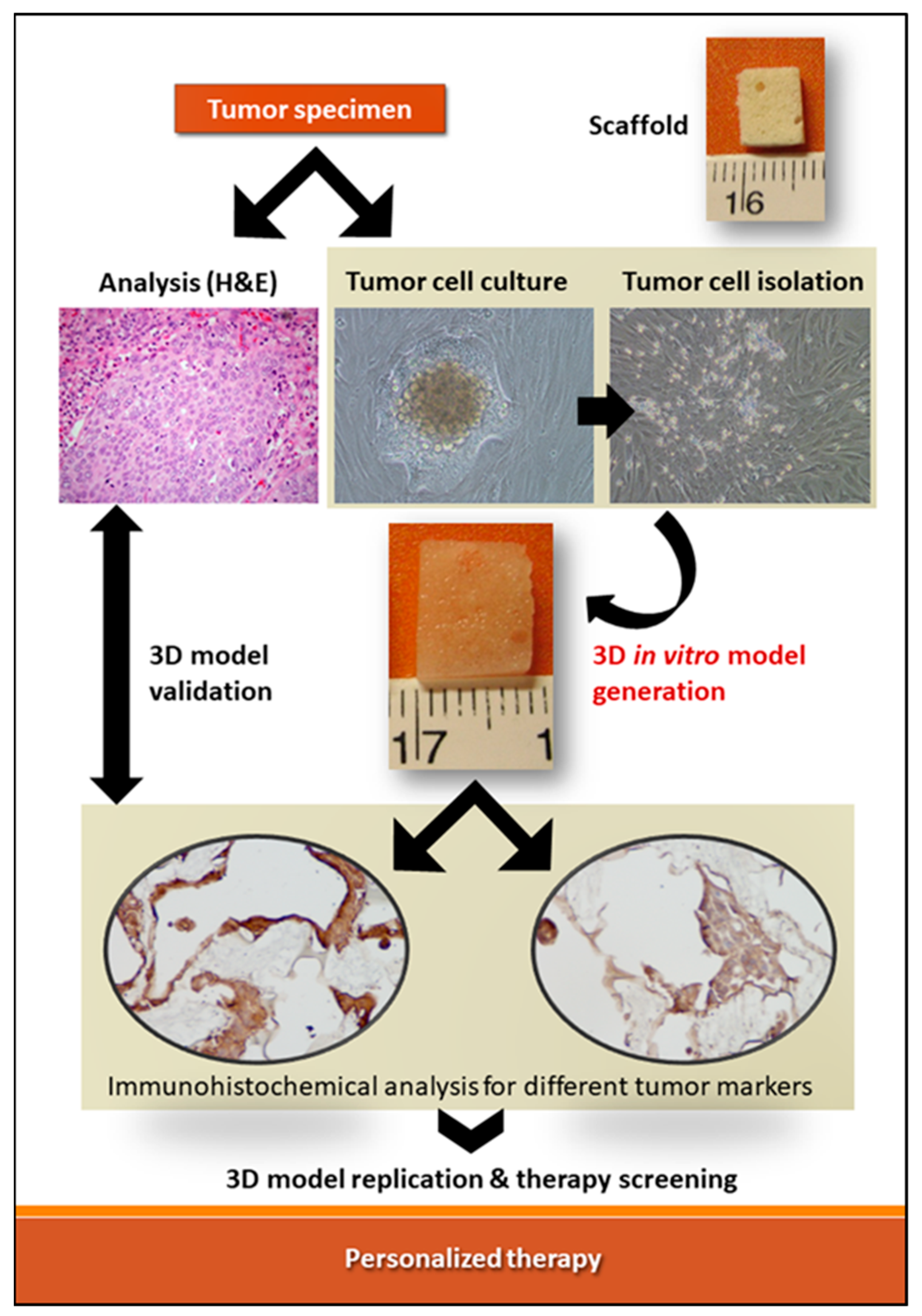
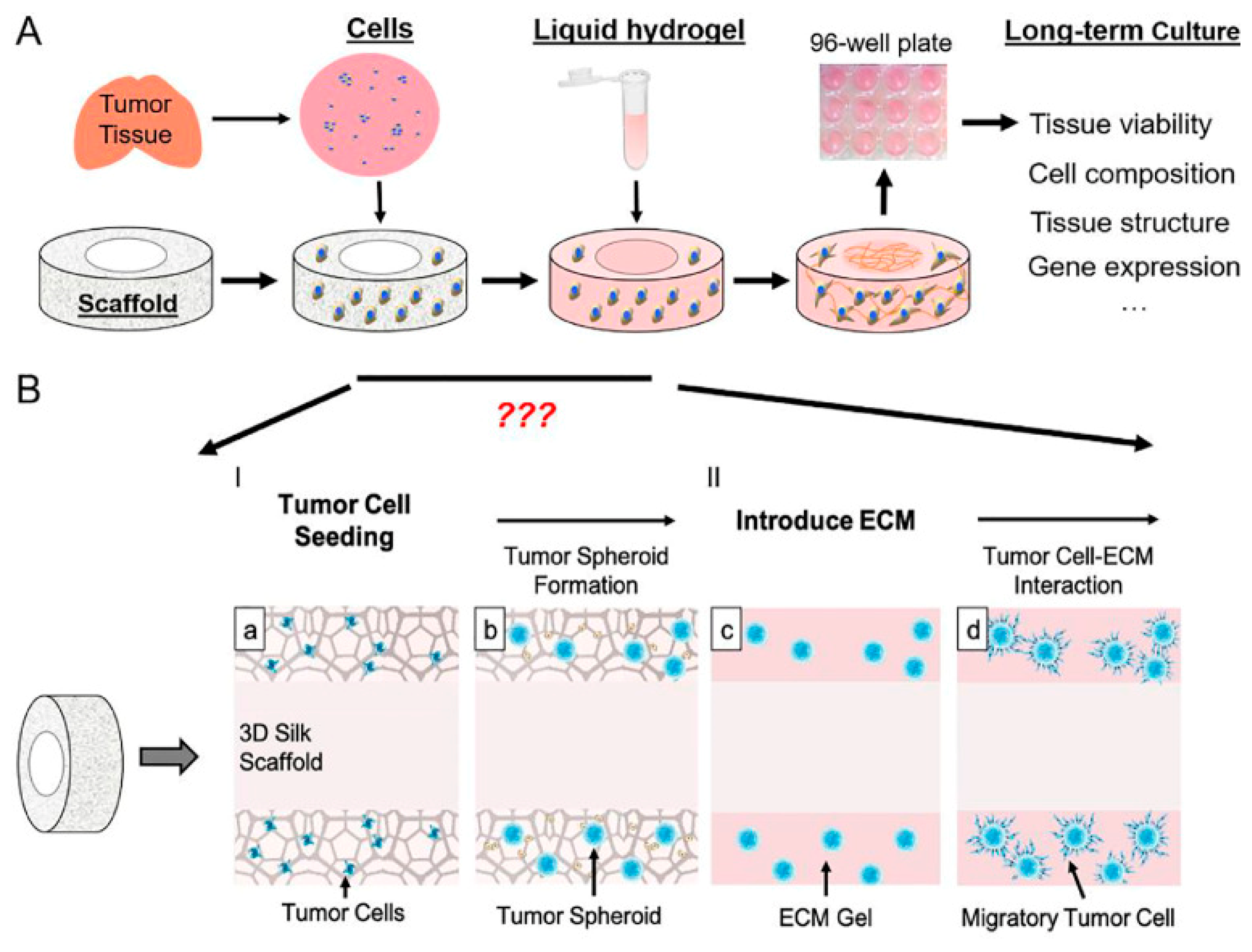
2. 3D In Vitro Models for Therapy Screening: The Role of Patient-Derived Cancer Cells and Biomaterials
3. Combined Application of Biomaterials and Patient-Derived Cancer Cells
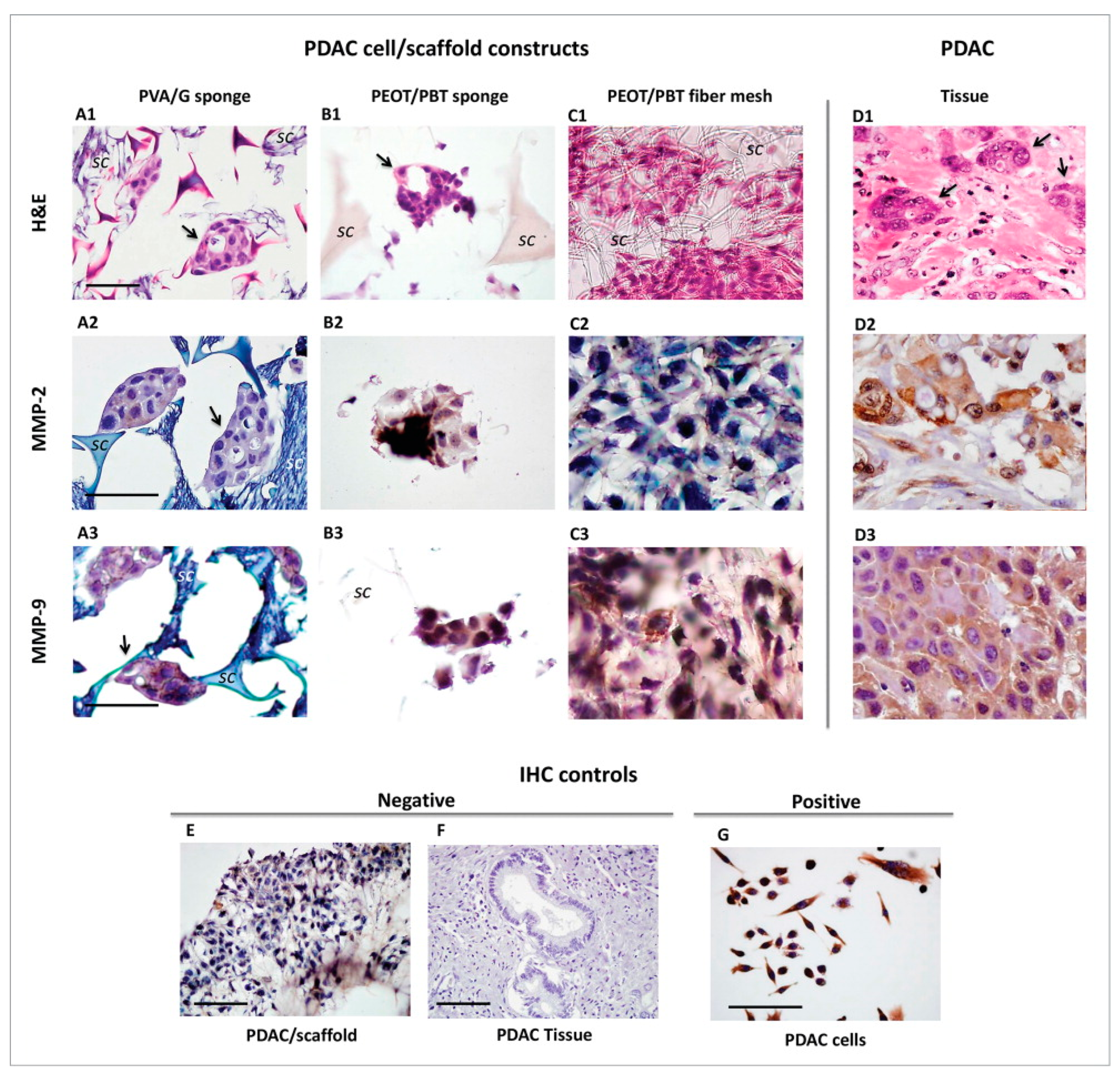
3.1. Polymeric Scaffolds and Primary Pancreatic Ductal Adenocarcinoma Cells
3.2. Bioprinting of Patient-Derived Intrahepatic Cholangiocarcinoma
3.3. Porous Scaffold Composed from Bombyxmoricocoons Silk Was Infused with Liquid ECM Gel for Pediatric Brain Tumors
3.4. Co-Assembly of Peptide Amphiphiles (PAs) with Custom ECM Components (PA-ECM) with Pancreatic Ductal Adenocarcinoma (PDAC)
3.5. Patient-Derived Malignant Hematopoietic Stem Cells in HA Scaffold Developed with a Bioreactor System for Acute Myeloid Leukemia and Myeloproliferative Neoplasms
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, F.B.; Cisewski, J.A.; Minino, A.; Anderson, R.N. Provisional Mortality Data—United States, 2020. Morb. Mortal. Wkly. Rep. 2021, 70, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Cancer statistics for the year 2020: An overview. Int. J. Cancer 2021, 70, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Islami, F.; Ward, E.M.; Sung, H.; Cronin, K.A.; Tangka, F.K.L.; Sherman, R.L.; Zhao, J.; Anderson, R.N.; Henley, S.J.; Yabroff, K.R.; et al. Annual Report to the Nation on the Status of Cancer, Part 1: National Cancer Statistics. J. Natl. Cancer Inst. 2021, 113, 1648–1669. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.; Heinrich, M.A.; Teixeira, L.M.; Prakash, J. 3D In Vitro Model (R)evolution: Unveiling Tumor-Stroma Interactions. Trends Cancer 2021, 7, 249–264. [Google Scholar] [CrossRef]
- King, S.M.; Presnell, S.C.; Nguyen, D.G. Abstract 2034: Development of 3D bioprinted human breast cancer for in vitro drug screening. Cancer Res. 2014, 74, 2034. [Google Scholar] [CrossRef]
- Augustine, R.; Kalva, S.N.; Ahmad, R.; Zahid, A.A.; Hasan, S.; Nayeem, A.; McClements, L.; Hasan, A. 3D Bioprinted cancer models: Revolutionizing personalized cancer therapy. Transl. Oncol. 2021, 14, 101015. [Google Scholar] [CrossRef]
- Mehrotra, S.; Moses, J.C.; Bandyopadhyay, A.; Mandal, B.B. 3D Printing/Bioprinting Based Tailoring of in Vitro Tissue Models: Recent Advances and Challenges. ACS Appl. Bio Mater. 2019, 2, 1385–1405. [Google Scholar] [CrossRef]
- Goodspeed, A.; Heiser, L.M.; Gray, J.W.; Costello, J.C. Tumor-Derived Cell Lines as Molecular Models of Cancer Pharmacogenomics. Mol. Cancer Res. 2016, 14, 3–13. [Google Scholar] [CrossRef] [Green Version]
- Pan, C.; Kumar, C.; Bohl, S.; Klingmueller, U.; Mann, M. Comparative proteomic phenotyping of cell lines and primary cells to assess preservation of cell type-specific functions. Mol. Cell Proteom. 2009, 8, 443–450. [Google Scholar] [CrossRef] [Green Version]
- Santoro, M.; Lamhamedi-Cherradi, S.E.; Menegaz, B.A.; Ludwig, J.A.; Mikos, A.G. Flow perfusion effects on three-dimensional culture and drug sensitivity of Ewing sarcoma. Proc. Natl. Acad. Sci. USA 2015, 112, 10304–10309. [Google Scholar] [CrossRef] [Green Version]
- Abbott, A.; Bond, K.; Chiba, T.; Sims-Lucas, S.; Oxburgh, L.; Coburn, J.M. Development of a mechanically matched silk scaffolded 3D clear cell renal cell carcinoma model. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 126, 112141. [Google Scholar] [CrossRef] [PubMed]
- Persson, E.; Gregersson, P.; Gustafsson, A.; Fitzpatrick, P.; Rhost, S.; Stahlberg, A.; Landberg, G. Patient-derived scaffolds influence secretion profiles in cancer cells mirroring clinical features and breast cancer subtypes. Cell Commun. Signal. 2021, 19, 66. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yang, H.; Wang, Y.; Zhang, X.; Jin, B.; Xie, F.; Jin, Y.; Pang, Y.; Zhao, H.; Lu, X.; et al. Application of a 3D Bioprinted Hepatocellular Carcinoma Cell Model in Antitumor Drug Research. Front. Oncol. 2020, 10, 878. [Google Scholar] [CrossRef] [PubMed]
- Domcke, S.; Sinha, R.; Levine, D.A.; Sander, C.; Schultz, N. Evaluating cell lines as tumour models by comparison of genomic profiles. Nat. Commun. 2013, 4, 2126. [Google Scholar] [CrossRef]
- Li, H.; Wawrose, J.S.; Gooding, W.E.; Garraway, L.A.; Lui, V.W.; Peyser, N.D.; Grandis, J.R. Genomic analysis of head and neck squamous cell carcinoma cell lines and human tumors: A rational approach to preclinical model selection. Mol. Cancer Res. 2014, 12, 571–582. [Google Scholar] [CrossRef] [Green Version]
- Neve, R.M.; Chin, K.; Fridlyand, J.; Yeh, J.; Baehner, F.L.; Fevr, T.; Clark, L.; Bayani, N.; Coppe, J.P.; Tong, F.; et al. A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 2006, 10, 515–527. [Google Scholar] [CrossRef] [Green Version]
- Czekanska, E.M.; Stoddart, M.J.; Ralphs, J.R.; Richards, R.G.; Hayes, J.S. A phenotypic comparison of osteoblast cell lines versus human primary osteoblasts for biomaterials testing. J. Biomed. Mater. Res. A 2014, 102, 2636–2643. [Google Scholar] [CrossRef]
- Gillet, J.P.; Calcagno, A.M.; Varma, S.; Marino, M.; Green, L.J.; Vora, M.I.; Patel, C.; Orina, J.N.; Eliseeva, T.A.; Singal, V.; et al. Redefining the relevance of established cancer cell lines to the study of mechanisms of clinical anti-cancer drug resistance. Proc. Natl. Acad. Sci. USA 2011, 108, 18708–18713. [Google Scholar] [CrossRef] [Green Version]
- Yi, H.G.; Jeong, Y.H.; Kim, Y.; Choi, Y.J.; Moon, H.E.; Park, S.H.; Kang, K.S.; Bae, M.; Jang, J.; Youn, H.; et al. A bioprinted human-glioblastoma-on-a-chip for the identification of patient-specific responses to chemoradiotherapy. Nat. Biomed. Eng. 2019, 3, 509–519. [Google Scholar] [CrossRef]
- Crystal, A.S.; Shaw, A.T.; Sequist, L.V.; Friboulet, L.; Niederst, M.J.; Lockerman, E.L.; Frias, R.L.; Gainor, J.F.; Amzallag, A.; Greninger, P.; et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science 2014, 346, 1480–1486. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Ory, V.; Chapman, S.; Yuan, H.; Albanese, C.; Kallakury, B.; Timofeeva, O.A.; Nealon, C.; Dakic, A.; Simic, V.; et al. ROCK inhibitor and feeder cells induce the conditional reprogramming of epithelial cells. Am. J. Pathol. 2012, 180, 599–607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brower, M.; Carney, D.N.; Oie, H.K.; Gazdar, A.F.; Minna, J.D. Growth of cell lines and clinical specimens of human non-small cell lung cancer in a serum-free defined medium. Cancer Res. 1986, 46, 798–806. [Google Scholar] [PubMed]
- Ricci, C.; Moroni, L.; Danti, S. Cancer tissue engineering—New perspectives in understanding the biology of solid tumors—A critical review. OA Tissue Eng. 2013, 1, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Weaver, V.M.; Petersen, O.W.; Larabell, C.A.; Dedhar, S.; Briand, P.; Lupu, R.; Bissell, M.J. Reciprocal interactions between beta1-integrin and epidermal growth factor receptor in three-dimensional basement membrane breast cultures: A different perspective in epithelial biology. Proc. Natl. Acad. Sci. USA 1998, 95, 14821–14826. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Mooney, D.J. Biomaterials and emerging anticancer therapeutics: Engineering the microenvironment. Nat. Rev. Cancer 2016, 16, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Rao, S.S.; Bushnell, G.G.; Azarin, S.M.; Spicer, G.; Aguado, B.A.; Stoehr, J.R.; Jiang, E.J.; Backman, V.; Shea, L.D.; Jeruss, J.S. Enhanced Survival with Implantable Scaffolds That Capture Metastatic Breast Cancer Cells In Vivo. Cancer Res. 2016, 76, 5209–5218. [Google Scholar] [CrossRef] [Green Version]
- Nii, T.; Makino, K.; Tabata, Y. Three-Dimensional Culture System of Cancer Cells Combined with Biomaterials for Drug Screening. Cancers 2020, 12, 2754. [Google Scholar] [CrossRef]
- DelNero, P.; Lane, M.; Verbridge, S.S.; Kwee, B.; Kermani, P.; Hempstead, B.; Stroock, A.; Fischbach, C. 3D culture broadly regulates tumor cell hypoxia response and angiogenesis via pro-inflammatory pathways. Biomaterials 2015, 55, 110–118. [Google Scholar] [CrossRef] [Green Version]
- Healy, K.E.; Thomas, C.H.; Rezania, A.; Kim, J.E.; McKeown, P.J.; Lom, B.; Hockberger, P.E. Kinetics of bone cell organization and mineralization on materials with patterned surface chemistry. Biomaterials 1996, 17, 195–208. [Google Scholar] [CrossRef]
- Minuth, W.W.; Sittinger, M.; Kloth, S. Tissue engineering: Generation of differentiated artificial tissues for biomedical applications. Cell Tissue Res. 1998, 291, 1–11. [Google Scholar] [CrossRef]
- Dhiman, H.K.; Ray, A.R.; Panda, A.K. Characterization and evaluation of chitosan matrix for in vitro growth of MCF-7 breast cancer cell lines. Biomaterials 2004, 25, 5147–5154. [Google Scholar] [CrossRef] [PubMed]
- Gelse, K.; Poschl, E.; Aigner, T. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duarte Campos, D.F.; Bonnin Marquez, A.; O’Seanain, C.; Fischer, H.; Blaeser, A.; Vogt, M.; Corallo, D.; Aveic, S. Exploring Cancer Cell Behavior In Vitro in Three-Dimensional Multicellular Bioprintable Collagen-Based Hydrogels. Cancers 2019, 11, 180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engel, B.J.; Constantinou, P.E.; Sablatura, L.K.; Doty, N.J.; Carson, D.D.; Farach-Carson, M.C.; Harrington, D.A.; Zarembinski, T.I. Multilayered, Hyaluronic Acid-Based Hydrogel Formulations Suitable for Automated 3D High Throughput Drug Screening of Cancer-Stromal Cell Cocultures. Adv. Healthc. Mater. 2015, 4, 1664–1674. [Google Scholar] [CrossRef]
- Luo, H.L.; Zhang, Y.; Gan, D.Q.; Yang, Z.W.; Ao, H.Y.; Zhang, Q.C.; Yao, F.L.; Wan, Y.Z. Incorporation of hydroxyapatite into nanofibrous PLGA scaffold towards improved breast cancer cell behavior. Mater. Chem. Phys. 2019, 226, 177–183. [Google Scholar] [CrossRef]
- Chen, H.; Cheng, Y.; Wang, X.; Wang, J.; Shi, X.; Li, X.; Tan, W.; Tan, Z. 3D printed in vitro tumor tissue model of colorectal cancer. Theranostics 2020, 10, 12127–12143. [Google Scholar] [CrossRef]
- Velliou, E.; Gupta, P.; Ricci, C.; Danti, S. Chapter 11—Biomaterial-based in vitro models for pancreatic cancer. In Biomaterials for 3D Tumor Modeling; Kundu, S.C., Reis, R.L., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 235–249. [Google Scholar] [CrossRef]
- Parkinson, G.T.; Salerno, S.; Ranji, P.; Hakansson, J.; Bogestal, Y.; Wettergren, Y.; Stahlberg, A.; Lindskog, E.B.; Landberg, G. Patient-derived scaffolds as a model of colorectal cancer. Cancer Med. 2021, 10, 867–882. [Google Scholar] [CrossRef]
- Gustafsson, A.; Garre, E.; Leiva, M.C.; Salerno, S.; Stahlberg, A.; Landberg, G. Patient-derived scaffolds as a drug-testing platform for endocrine therapies in breast cancer. Sci. Rep. 2021, 11, 13334. [Google Scholar] [CrossRef]
- Boucherit, N.; Gorvel, L.; Olive, D. 3D Tumor Models and Their Use for the Testing of Immunotherapies. Front. Immunol. 2020, 11, 603640. [Google Scholar] [CrossRef]
- Kenerson, H.L.; Sullivan, K.M.; Seo, Y.D.; Stadeli, K.M.; Ussakli, C.; Yan, X.W.; Lausted, C.; Pillarisetty, V.G.; Park, J.O.; Riehle, K.J.; et al. Tumor slice culture as a biologic surrogate of human cancer. Ann. Transl. Med. 2020, 8, 114. [Google Scholar] [CrossRef]
- Weiswald, L.B.; Bellet, D.; Dangles-Marie, V. Spherical cancer models in tumor biology. Neoplasia 2015, 17, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ricci, C.; Mota, C.; Moscato, S.; D’Alessandro, D.; Ugel, S.; Sartoris, S.; Bronte, V.; Boggi, U.; Campani, D.; Funel, N.; et al. Interfacing polymeric scaffolds with primary pancreatic ductal adenocarcinoma cells to develop 3D cancer models. Biomatter 2014, 4, e955386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, S.; He, J.; Zhao, Y.; Liu, T.; Xie, F.; Yang, H.; Mao, Y.; Pang, Y.; Sun, W. Bioprinting of patient-derived in vitro intrahepatic cholangiocarcinoma tumor model: Establishment, evaluation and anti-cancer drug testing. Biofabrication 2020, 12, 045014. [Google Scholar] [CrossRef] [PubMed]
- Tang-Schomer, M.D.; Chandok, H.; Wu, W.B.; Lau, C.C.; Bookland, M.J.; George, J. 3D patient-derived tumor models to recapitulate pediatric brain tumors In Vitro. Transl. Oncol. 2022, 20, 101407. [Google Scholar] [CrossRef]
- Osuna de la Pena, D.; Trabulo, S.M.D.; Collin, E.; Liu, Y.; Sharma, S.; Tatari, M.; Behrens, D.; Erkan, M.; Lawlor, R.T.; Scarpa, A.; et al. Bioengineered 3D models of human pancreatic cancer recapitulate in vivo tumour biology. Nat. Commun. 2021, 12, 5623. [Google Scholar] [CrossRef]
- Garcia-Garcia, A.; Klein, T.; Born, G.; Hilpert, M.; Scherberich, A.; Lengerke, C.; Skoda, R.C.; Bourgine, P.E.; Martin, I. Culturing patient-derived malignant hematopoietic stem cells in engineered and fully humanized 3D niches. Proc. Natl. Acad. Sci. USA 2021, 118, e2114227118. [Google Scholar] [CrossRef]
- Coussens, L.M.; Fingleton, B.; Matrisian, L.M. Matrix metalloproteinase inhibitors and cancer: Trials and tribulations. Science 2002, 295, 2387–2392. [Google Scholar] [CrossRef]
- Wiercinska, E.; Naber, H.P.; Pardali, E.; van der Pluijm, G.; van Dam, H.; ten Dijke, P. The TGF-beta/Smad pathway induces breast cancer cell invasion through the up-regulation of matrix metalloproteinase 2 and 9 in a spheroid invasion model system. Breast Cancer Res. Treat. 2011, 128, 657–666. [Google Scholar] [CrossRef] [Green Version]
- Bera, A.; Zhao, S.; Cao, L.; Chiao, P.J.; Freeman, J.W. Oncogenic K-Ras and loss of Smad4 mediate invasion by activating an EGFR/NF-kappaB Axis that induces expression of MMP9 and uPA in human pancreas progenitor cells. PLoS ONE 2013, 8, e82282. [Google Scholar] [CrossRef] [Green Version]
- Feula, M.; Milazzo, M.; Giannone, G.; Azimi, B.; Trombi, L.; Cacopardo, L.; Moscato, S.; Lazzeri, A.; Ahluwalia, A.; Berrettini, S.; et al. Bioartificial Sponges for Auricular Cartilage Engineering. In Proceedings of the Advances in Bionanomaterials II, Cham, Switzerland, 29 September–3 October 2020; pp. 191–209. [Google Scholar]
- D’Alessandro, D.; Moscato, S.; Fusco, A.; De la Ossa, J.G.; D’Acunto, M.; Trombi, L.; Feula, M.; Serino, L.P.; Donnarumma, G.; Petrini, M.; et al. Poly(vinyl alcohol)/Gelatin Scaffolds Allow Regeneration of Nasal Tissues. Appl. Sci. 2021, 11, 3651. [Google Scholar] [CrossRef]
- Sezgin, O.; Yaras, S.; Ozdogan, O. The course and prognostic value of increased pancreas stiffness detected by ultrasound elastography during acute pancreatitis. Pancreatology 2021, 21, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Ricci, C.; Danti, S. 3D Models of Pancreatic Ductal Adenocarcinoma via Tissue Engineering. Methods Mol. Biol. 2019, 1882, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Joshi, M.B.; Ivanov, D.; Feder-Mengus, C.; Spagnoli, G.C.; Martin, I.; Erne, P.; Resink, T.J. Use of multicellular tumor spheroids to dissect endothelial cell-tumor cell interactions: A role for T-cadherin in tumor angiogenesis. FEBS Lett. 2007, 581, 4523–4528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Chadalavada, K.; Wilshire, J.; Kowalik, U.; Hovinga, K.E.; Geber, A.; Fligelman, B.; Leversha, M.; Brennan, C.; Tabar, V. Glioblastoma stem-like cells give rise to tumour endothelium. Nature 2010, 468, 829–833. [Google Scholar] [CrossRef]
- Dominiak, A.; Chelstowska, B.; Olejarz, W.; Nowicka, G. Communication in the Cancer Microenvironment as a Target for Therapeutic Interventions. Cancers 2020, 12, 1232. [Google Scholar] [CrossRef]
- Dumas, J.F.; Brisson, L. Interaction between adipose tissue and cancer cells: Role for cancer progression. Cancer Metastasis Rev. 2021, 40, 31–46. [Google Scholar] [CrossRef]
- Calorini, L.; Bianchini, F. Environmental control of invasiveness and metastatic dissemination of tumor cells: The role of tumor cell-host cell interactions. Cell Commun. Signal. 2010, 8, 24. [Google Scholar] [CrossRef] [Green Version]
- Contessi Negrini, N.; Ricci, C.; Bongiorni, F.; Trombi, L.; D’Alessandro, D.; Danti, S.; Farè, S. An Osteosarcoma Model by 3D Printed Polyurethane Scaffold and In Vitro Generated Bone Extracellular Matrix. Cancers 2022, 14, 2003. [Google Scholar] [CrossRef]
| Cell Lines | Patient-Derived Cells | |
|---|---|---|
| Accessibility | Easily accessible | Difficult to access, limited availability of cells/tissues |
| Cost | Low cost to obtain and culture | Increased cost to obtain and culture |
| Proliferation | Proliferates rapidly and indefinitely | More difficult to proliferate due to tumor cell senescence, limited amount of pasages |
| Ease in culturing | Robust and easy to work with and maintain | More fragile and difficult to work with and maintain |
| Reproducibility | Pure population of cells therefore reproducible data can be obtained | Heterogenous population of cells therefore data can differ between cell populations |
| Ability to mimic TME and clinical response | Lack of complexity to mimic tumor environment and clinical response | Can better mimic the TME and clinical response |
| Ethical issues and research compliance | No ethical concerns and no need for institutional review board approval to obtain and use | Need to obtain institutional review board approval to obtain and use |
| Cancer Type | Type of Cells | Biomaterial/Scaffold | Main Outcome | Ref. |
|---|---|---|---|---|
| Pancreatic ductal adenocarcinoma (PDAC) | Cells obtained from PDAC pieces/explants | (PVA/G) blend sponges; (PEOT/PBT) copolymer compression molded scaffolds and electrospun fibers meshes. | PDAC cells demonstrated various behaviors when exposed to different scaffold types. Sponge-like pores allowed for cellular clustering resembling the native cancer morphostructure. In PVA/G sponges the active MMP-2 enzyme was the highest. | [43] |
| Intrahepatic cholangiocarcinoma (ICC) | Cells obtained from a male patient diagnosed with ICC | Gelatin-alginate-Matrigel™ hydrogel bioink | Printed ICC cells showed colony forming capacity, high survival rate, active proliferation, invasive and metastatic phenotype and other characteristics of ICC cells, e.g., expression levels of tumor markers and cancer stem cell markers. | [44] |
| Pediatric brain tumors | Cells obtained from 11 pediatric tumor cases, consisting of three medulloblastoma (MB) patients, three ependymoma (EPN) patients, one glioblastoma (GBM) patient, and four juvenile pilocytic astrocytoma (Ast) patients. | Bombyxmoricocoons silk porous scaffold infused with liquid ECM gel | The 3D scaffold silk base structure had a vital role in supporting tumor spheroids, giving structural stability to gels, and maintaining tumor stem cells in 3D. | [45] |
| PDAC | Cells obtained from PDAC patient | Co-assembly of PAs with custom ECM components (PA-ECM) | This model of PDAC was able to sustain patient-specific transcriptional profiles and demonstrated high cancer stem cell functionality. | [46] |
| Acute myeloid leukemia (AML) and myeloproliferative neoplasms (MPN) | Cells obtained from malignant human malignant hematopoietic stem and progenitor cells (HSPCs) | HA scaffold with perfusion bioreactor | The 3D model provided an environment that could sustain CD34+ cells from acute myeloid leukemia (AML) and myeloproliferative neoplasm patients for up to 3 weeks. | [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasbum, A.; Karabulut, O.; Reyes, R.E.; Ricci, C.; Franchi, A.; Danti, S.; Chew, S.A. Combined Application of Patient-Derived Cells and Biomaterials as 3D In Vitro Tumor Models. Cancers 2022, 14, 2503. https://doi.org/10.3390/cancers14102503
Hasbum A, Karabulut O, Reyes RE, Ricci C, Franchi A, Danti S, Chew SA. Combined Application of Patient-Derived Cells and Biomaterials as 3D In Vitro Tumor Models. Cancers. 2022; 14(10):2503. https://doi.org/10.3390/cancers14102503
Chicago/Turabian StyleHasbum, Asbiel, Ozan Karabulut, Ruben Edgar Reyes, Claudio Ricci, Alessandro Franchi, Serena Danti, and Sue Anne Chew. 2022. "Combined Application of Patient-Derived Cells and Biomaterials as 3D In Vitro Tumor Models" Cancers 14, no. 10: 2503. https://doi.org/10.3390/cancers14102503
APA StyleHasbum, A., Karabulut, O., Reyes, R. E., Ricci, C., Franchi, A., Danti, S., & Chew, S. A. (2022). Combined Application of Patient-Derived Cells and Biomaterials as 3D In Vitro Tumor Models. Cancers, 14(10), 2503. https://doi.org/10.3390/cancers14102503









