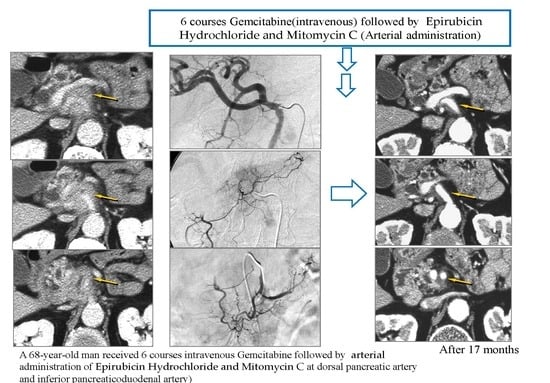
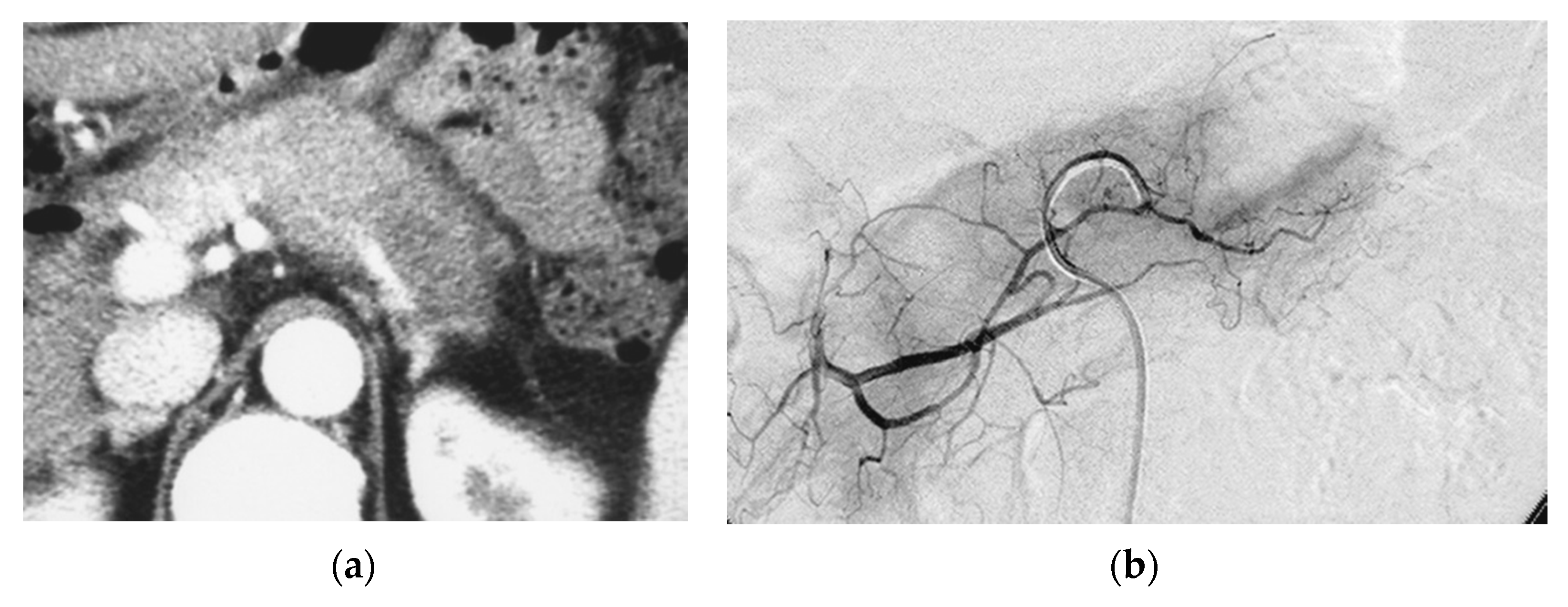
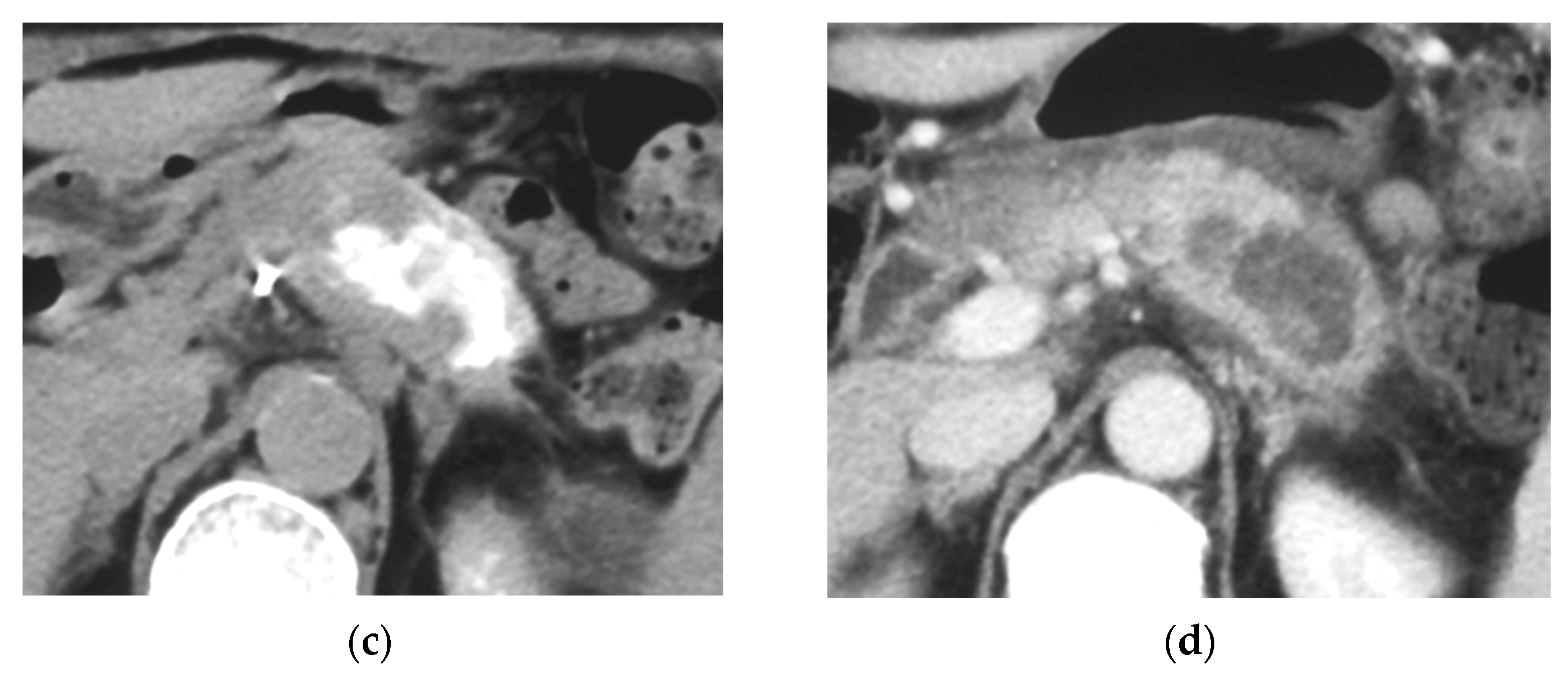
Arterial Administration of DNA Crosslinking Agents with Restraint of Homologous Recombination Repair by Intravenous Low-Dose Gemcitabine Is Effective for Locally Advanced Pancreatic Cancer
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Pilot Study
2.2. Study Design
2.3. Patients
2.4. Procedure and Treatment
2.4.1. Treatment Course
2.4.2. Systemic Chemotherapy by Intravenous Low-Dose Gemcitabine
2.4.3. Selective Arterial Administration of Agents Which Cause DNA Double-Strand Break or DNA Crosslink into Pancreatic Arteries and Adrenal Arteries
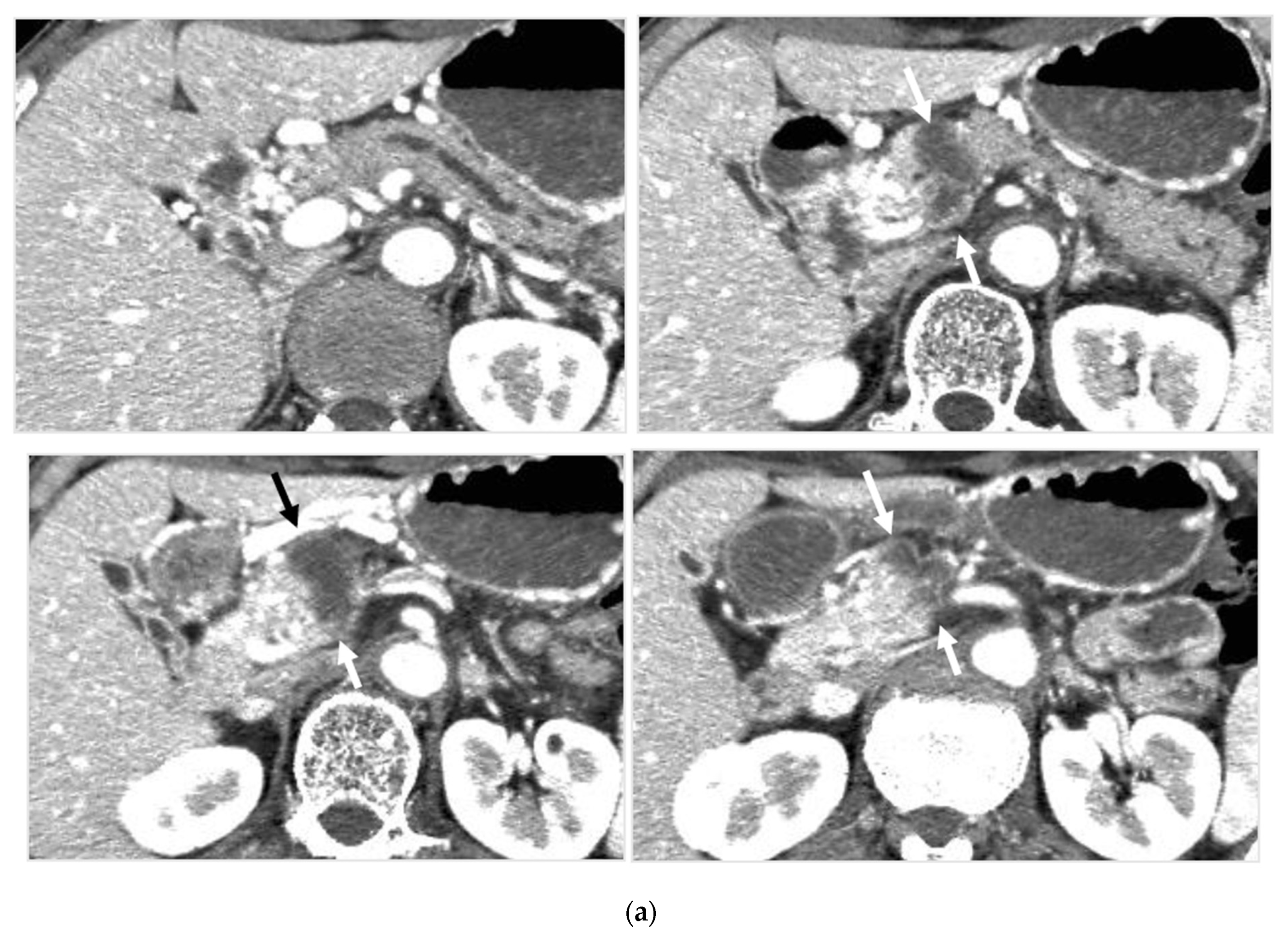
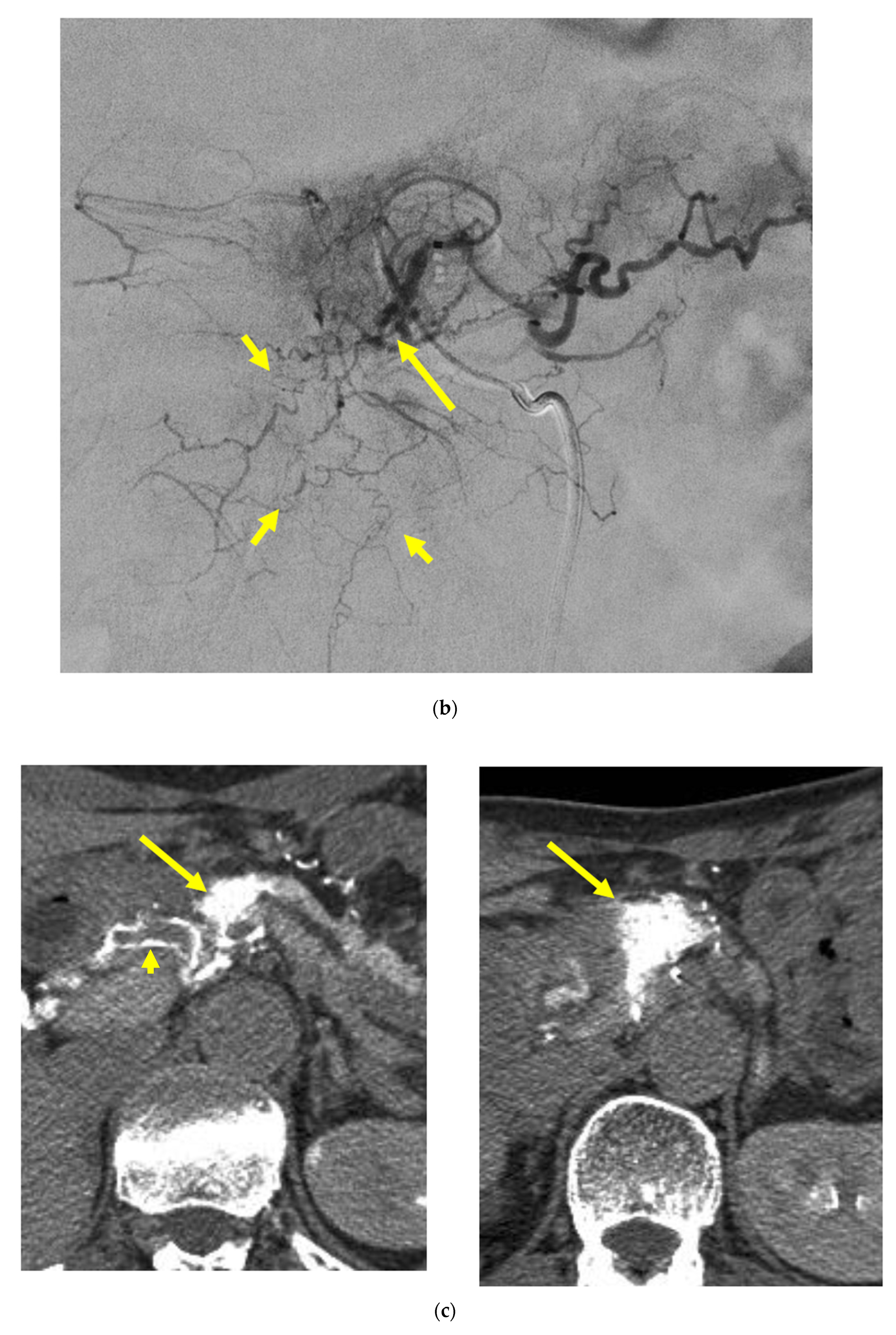
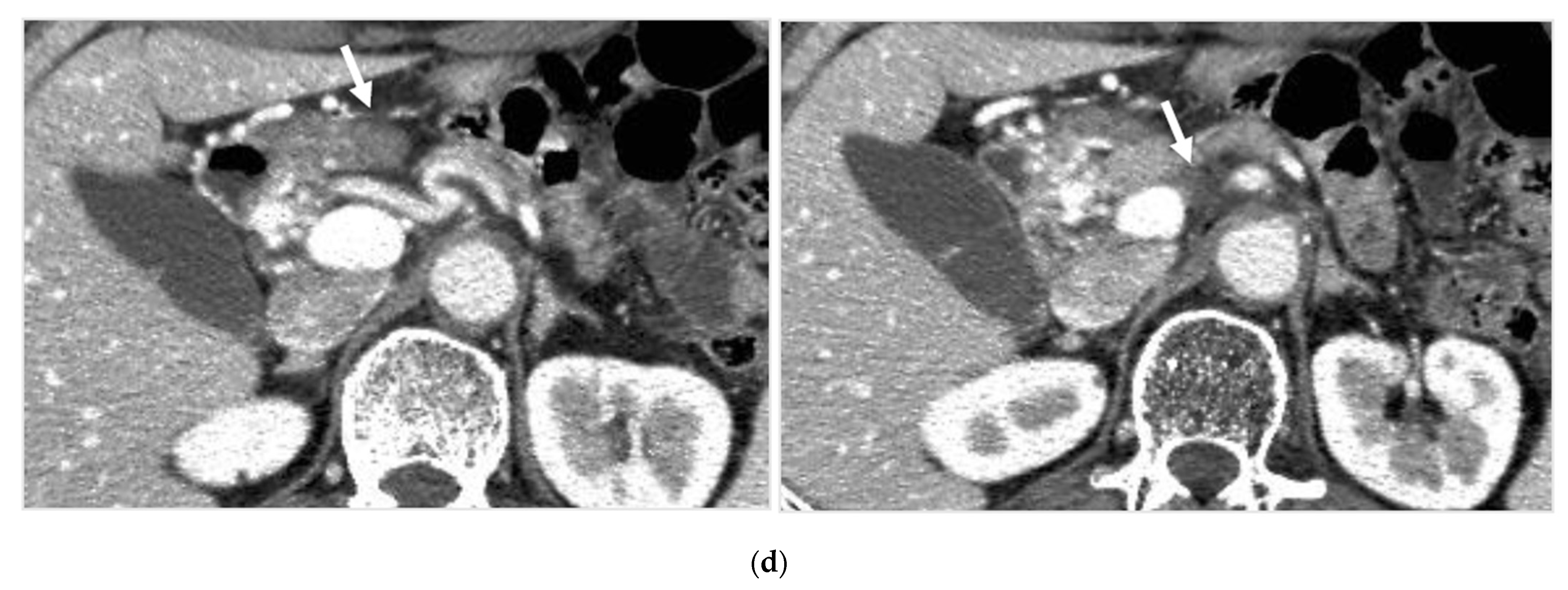
Identifying Supplying Arteries to Pancreatic Cancer
Method of Arterial Administration of Anticancer Agents
2.4.4. Maintenance of Systemic Chemotherapy
2.5. Assessments
2.5.1. Judging Completeness or Incompleteness of Arterial Administration of Chemotherapeutic Agents
2.5.2. Survival Times
2.5.3. Tumor Response
2.5.4. Safety Assessment
2.5.5. Statistical Analysis
3. Results
3.1. Patient’s Characteristics
3.2. Treatment Compliance
3.3. Patient Subgrouping According to Treatment Compliance
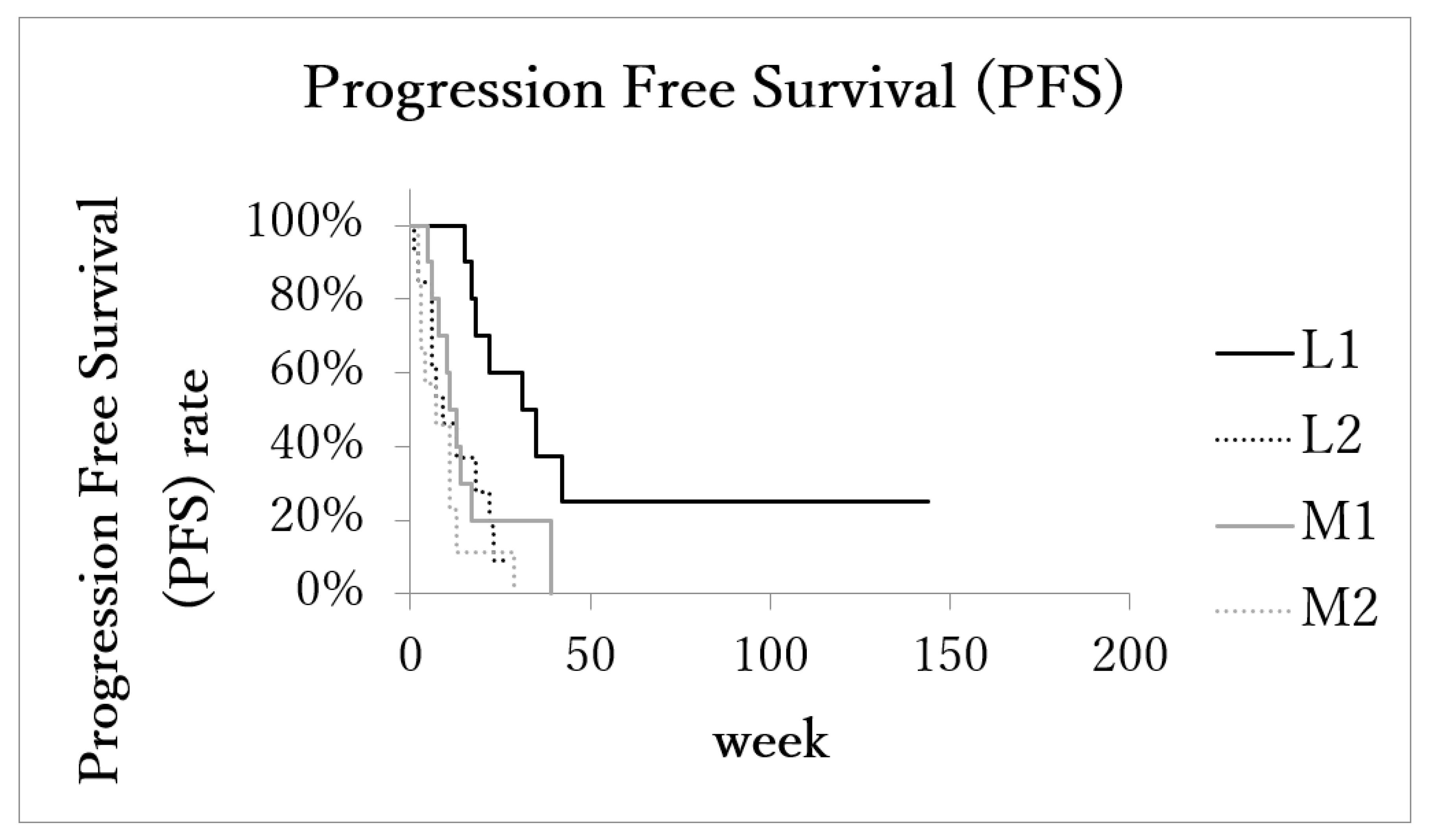
3.4. Survival and Disease Progression
3.5. Tumor Response
3.6. Treatment Toxicity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Kamisawa, T.; Wood, L.D.; Itoi, T.; Takaori, K. Pancreatic cancer. Lancet 2016, 388, 73–85. [Google Scholar] [CrossRef]
- Malvezzi, M.; Bertuccio, P.; Levi, F.; La Vecchia, C.; Negri, E. European cancer mortality predictions for the year 2014. Ann. Oncol. 2014, 25, 1650–1656. [Google Scholar] [CrossRef]
- Arnold, M.; Abnet, C.C.; Neale, R.E.; Vignat, J.; Giovannuci, E.L.; McGlynn, K.A.; Bray, F. Global burden of 5 major types of gastrointestinal cancer. Gastroenterology. 2020, 159, 335–349. [Google Scholar] [CrossRef]
- Mazur, P.; Siveke, J.T. Genetically engineered mouse models of pancreatic cancer: Unravelling tumour biology and progressing translational oncology. Gut 2012, 61, 1488–1500. [Google Scholar] [CrossRef]
- Yachida, S.; Jones, S.; Božić, I.; Antal, T.; Leary, R.; Fu, B.; Kamiyama, M.; Hruban, R.H.; Eshleman, J.R.; Nowak, M.A.; et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 2010, 467, 1114–1117. [Google Scholar] [CrossRef]
- Mohammed, A.; Janakiram, N.B.; Lightfoot, S.; Gali, H.; Vibhudutta, A.; Rao, C.V. Early detection and prevention of pancreatic cancer: Use of genetically engineered mouse models and advanced imaging technologies. Curr. Med. Chem. 2012, 19, 3701–3713. [Google Scholar] [CrossRef]
- Burris, H.A., III; Moore, M.J.; Anderson, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef]
- Matano, E.; Tagliaferri, P.; Libroia, A.; Damiano, V.; Fabbrocini, A.; De Lorenzo, S.; Bianco, A.R. Gemcitabine combined with continuous infusion 5-fluorouracil in advanced and symptomatic pancreatic cancer: A clinical benefit-oriented phase II study. Br. J. Cancer 2000, 82, 1772–1775. [Google Scholar] [CrossRef]
- Vaccaro, V.; Sperduti, I.; Milella, M. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 365, 768–769. [Google Scholar] [CrossRef]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.-L.; Gourgou-Bourgade, S.; De La Fouchardière, C.; et al. FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [PubMed]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, G.E.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Stein, S.M.; James, E.S.; Deng, Y.; Cong, C.; Kortmansky, J.S.; Li, J.; Staugaard, C.; Indukala, D.; Boustani, A.M.; Patel, V. Final analysis of a phase II study of modified FOLFIRINOX in locally advanced and met-astatic pancreatic cancer. Br. J. Cancer 2016, 114, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Reni, M.; Cereda, S.; Balzano, G.; Passoni, P.; Rognone, A.; Zerbi, A.; Nicoletti, R.; Mazza, E.; Arcidiacono, P.G.; Di Carlo, V.; et al. Outcome of upfront combination chemotherapy followed by chemoradiation for locally advanced pancreatic adenocarcinoma. Cancer Chemother. Pharmacol. 2009, 64, 1253–1259. [Google Scholar] [CrossRef]
- Van Zweeden, A.A.; van der Vliet, H.J.; Wilmink, J.W.; Meijerink, M.R.; Meijer, O.W.M.; Bruynzeel, A.M.E.; van Tienhoven, G.; Giovannetti, E.; Kazemier, G.; Jacobs, M.A.J.M.; et al. Phase I Clinical Trial to Determine the Feasibility and Maximum Tol-erated Dose of Panitumumab to Standard Gemcitabine-Based Chemoradiation in Locally Advanced Pancreatic Cancer. Clin. Cancer Res. 2015, 21, 4569–4575. [Google Scholar] [CrossRef]
- Dholakia, A.S.; Chaudhry, M.; Leal, J.P.; Chang, D.T.; Raman, S.P.; Hacker-Prietz, A.; Su, Z.; Pai, J.; Oteiza, K.E.; Griffith, M.E.; et al. Baseline metabolic tumor volume and total lesion glycolysis are associated with sur-vival outcomes in patients with locally advanced pancreatic cancer receiving stereotactic body radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 539–546. [Google Scholar] [CrossRef]
- Kamada, T.; Tsujii, H.; Blakely, E.A.; Debus, J.; De Neve, W.; Durante, M.; Jäkel, O.; Mayer, R.; Orecchia, R.; Pötter, R.; et al. Carbon ion radiotherapy in Japan: An assessment of 20 years of clinical experience. Lancet Oncol. 2015, 16, e93–e100. [Google Scholar] [CrossRef]
- Homma, H.; Doi, T.; Mezawa, S.; Takada, K.; Kukitsu, T.; Oku, T.; Akiyama, T.; Kusakabe, T.; Miyanishi, K.; Niitsu, Y. A novel arterial infusion chemotherapy for the treatment of patients with advanced pancreatic carcinoma after vascular supply distribution via superselective embolization. Cancer 2000, 89, 303–313. [Google Scholar] [CrossRef]
- Ikeda, O.; Kusunoki, S.; Kudoh, K.; Takamori, H.; Tsuji, T.; Kanemitsu, K.; Yamashita, Y. Evaluation of the efficacy of combained continuous arterial infusion and systemic chemotherapy for the treatment of advanced pancreatic carcinoma. Cardiovasc. Intervent. Radiol. 2006, 29, 362–370. [Google Scholar] [CrossRef]
- Sasada, T.; Denno, R.; Tanaka, T.; Kanai, M.; Mizukami, Y.; Kohno, S.; Takabayashi, A. Intra-arterial infusion chemotherapy with 5-fluorouracil and cisplatin in advanced pan-creatic cancer: A feasibility study. Am J. Clin. Oncol. 2008, 31, 71–78. [Google Scholar] [CrossRef]
- Tanaka, T.; Sho, M.; Nishiofuku, H.; Sakaguchi, H.; Inaba, Y.; Nakajima, Y.; Kichikaiwa, K. Unresectable Pancreatic Cancer: Arterial Embolization to Achieve a Single Blood Supply for Intraarterial Infusion of 5-Fluorouracil and Full-Dose IV Gemcitabine. Am. J. Roentgenol. 2012, 198, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.L.; Pandalai, P.; Ripley, R.T.; Langan, R.C.; Steinberg, S.M.; Walker, M.; Toomey, M.A.; Levy, E.; Avital, I. Regional Chemotherapy in Locally Advanced Pancreatic Cancer: RECLAP Trial. Trials 2011, 12, 129. [Google Scholar] [CrossRef] [PubMed]
- Maacke, H.; Jost, K.; Opitz, S.; Miska, S.; Yuan, Y.; Hasselbach, L.; Lüttges, J.; Kalthoff, H.; Stürzbecher, H.W. DNA repair and recombination factor Rad51 is overexpressed in human pancreatic adeno-carcinoma. Oncogene 2000, 19, 2791–2795. [Google Scholar] [CrossRef] [PubMed]
- Nagathihalli, N.S.; Nagaraju, G. RAD51 as a potential biomarker and therapeutic target for pancreatic cancer. Biochim. Biophys. Acta Bioenerg. 2011, 1816, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Wachters, F.M.; van Putten, J.W.; Maring, J.G.; Zdzienicka, M.Z.; Groen, H.J.; Kampinga, H.H. Selective targeting of homologous DNA recombination repair by gemcitabine. Int. J. Radiat. Oncol. 2003, 57, 553–562. [Google Scholar] [CrossRef]
- Okahara, M.; Mori, H.; Kiyosue, H.; Yamada, Y.; Sagara, Y.; Matsumoto, S. Arterial supply to the pancreas; variations and cross-sectional anatomy. Abdom. Imaging 2010, 35, 134–142. [Google Scholar] [CrossRef]
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; Van Glabbeke, M.; Van Oosterom, A.T.; Christian, M.C.; et al. New Guidelines to Evaluate the Response to Treatment in Solid Tumors. J. Natl. Cancer Inst. 2000, 92, 205–216. [Google Scholar] [CrossRef]
- National Cancer Institute: Common Terminology Criteria for Adverse Events v.4.0 (CTCAE-NCI v.4.0). Available online: http://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm (accessed on 14 June 2010).
- Görich, J.; Rilinger, N.; Sokiranski, R.; Vogel, J.; Wikström, M.; Krämer, S.; Merkle, E.; Rieber, A.; Brambs, H.J. CT-guided intraarterial chemotherapy in locally advanced tumors. Radiology 1996, 199, 567–570. [Google Scholar] [CrossRef]
- Ishii, R.; Mori, H.; Matsumura, K.; Hongo, N.; Kiyosue, H.; Matsumoto, S.; Yoshimi, T.; Ujiie, S. Molecular interactions between anticancer drugs and iodinated contrast media: An in vitro spectroscopic study. J. Biomed. Sci. Eng. 2012, 5, 24–33. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tu-moritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar]
- Maeda, H.; Nakamura, H.; Fang, J. The EPR effect for macromolecular drug delivery to solid tumors: Improvement of tumor uptake, lowering of systemic toxicity, and distinct tumor imaging in vivo. Adv. Drug Deliv. Rev. 2013, 65, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, K. Clinical relevance of the homologous recombination machinery in cancer therapy. Cancer Sci. 2007, 99, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Meike, S.; Yamamori, T.; Yasui, H.; Eitaki, M.; Matsuda, A.; Morimatsu, M.; Fukushima, M.; Yamasaki, Y.; Inanami, O. A nucleoside anticancer drug, 1-(3-C-ethynyl-β-D-ribo-pentofuranosyl)cytosine (TAS106), sensitizes cells to radiation by suppressing BRCA2 expression. Mol. Cancer 2011, 10, 92. [Google Scholar] [CrossRef] [PubMed]
- Takagi, M.; Sakata, K.-I.; Someya, M.; Tauchi, H.; Iijima, K.; Matsumoto, Y.; Torigoe, T.; Takahashi, A.; Hareyama, M.; Fukushima, M. Gimeracil sensitizes cells to radiation via inhibition of homologous recombination. Radiother. Oncol. 2010, 96, 259–266. [Google Scholar] [CrossRef]
- Asaad, N.A.; Zeng, Z.C.; Guan, J.; Thacker, J.; Iliakis, G. Homologous recombination as a potential target for caffeine radiosensitization in mamma-lian cells: Reduced caffeine radiosensitization in XRCC2 and XRCC3 mutants. Oncogene 2000, 19, 5788–5800. [Google Scholar] [CrossRef] [PubMed]
- Chinnaiyan, P.; Huang, S.; Vallabhaneni, G.; Armstrong, E.; Varambally, S.; Tomlins, S.A.; Chinnaiyan, A.M.; Harari, P.M. Mechanisms of enhanced radiation response following epidermal growth fac-tor receptor signaling inhibition by erlotinib (Tarceva). Cancer Res. 2005, 65, 3328–3335. [Google Scholar] [CrossRef]
- Li, L.; Wang, H.; Yang, E.S.; Arteaga, C.L.; Xia, F. Erlotinib Attenuates Homologous Recombinational Repair of Chromosomal Breaks in Human Breast Cancer Cells. Cancer Res. 2008, 68, 9141–9146. [Google Scholar] [CrossRef]
- Choudhury, A.; Zhao, H.; Jalali, F.; Al Rashid, S.; Ran, J.; Supiot, S.; Kiltie, A.; Bristow, R. Targeting homologous recombination using imatinib results in enhanced tumor cell chemosensitivity and radiosensitivity. Mol. Cancer Ther. 2009, 8, 203–213. [Google Scholar] [CrossRef]
- Kobashigawa, S.; Morikawa, K.; Mori, H.; Kashino, G. Gemcitabine Induces Radiosensitization Through Inhibition of RAD51-dependent Repair for DNA Double-strand Breaks. Anticancer. Res. 2015, 35, 2731–2737. [Google Scholar]
- Leone, F.; Gatti, M.; Massucco, P.; Colombi, F.; Sperti, E.; Campanella, D.; Regge, D.; Gabriele, P.; Capussotti, L.; Aglietta, M. Induction gemcitabine and oxaliplatin therapy followed by a twice-weekly infusion of gemcitabine and concurrent external-beam radiation for neoadjuvant treatment of locally advanced pancreatic cancer: A sin-gle-institutional experience. Cancer 2013, 119, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Satoi, S.; Yanagimoto, H.; Toyokawa, H.; Takahashi, K.; Matsui, Y.; Kitade, H.; Mergental, H.; Tanigawa, N.; Takai, S.; Kwon, A.-H. Surgical results after preoperative chemoradiation therapy for patients with pan-creatic cancer. Pancreas 2009, 38, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Mizuno, S.; Murata, Y.; Kishiwada, M.; Usui, M.; Sakurai, H.; Tabata, M.; Ii, N.; Yamakado, K.; Inoue, H.; et al. Gemcitabine-based chemoradiotherapy followed by surgery for borderline resectable and locally unresectable pancreatic ductal adenocarcinoma: Significance of the CA19-9 reduction rate and intratumoral hu-man equilibrative nucleoside transporter 1 expression. Pancreas 2014, 43, 350–360. [Google Scholar] [CrossRef]
- Kamachi, H.; Tsuruga, Y.; Orimo, T.; Wakayama, K.; Shimada, S.; Nagatsu, A.; Yokoo, H.; Kamiyama, T.; Katoh, N.; Taketomi, A. R0 Resection for Locally Advanced Pancreatic Cancer with Low-dose Gemcitabine with Wide Irradiation Area as Neoadjuvant Chemoradiotherapy. Vivo 2018, 32, 1183–1191. [Google Scholar] [CrossRef]
- Flores, J.P.E.; Diasio, R.B.; Saif, M.W. Drug metabolism and pancreatic cancer. Ann. Gastroenterol. 2016, 30, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Overdevest, J.B.; Theodorescu, D.; Lee, J.K. Utilizing the Molecular Gateway: The Path to Personalized Cancer Management. Clin Chem. 2009, 55, 684–697. [Google Scholar] [CrossRef]
- Yadav, D.K.; Bai, X.; Yadav, R.K.; Singh, A.; Li, G.; Ma, T.; Chen, W.; Liang, T. Liquid biopsy in pancreatic cancer: The beginning of a new era. Oncotarget 2018, 9, 26900–26933. [Google Scholar] [CrossRef]
- Lewis, A.R. Pancreatic cancer: Are “liquid biopsies” ready for prime-time. World J. Gastroenterol. 2016, 22, 7175–7185. [Google Scholar] [CrossRef]
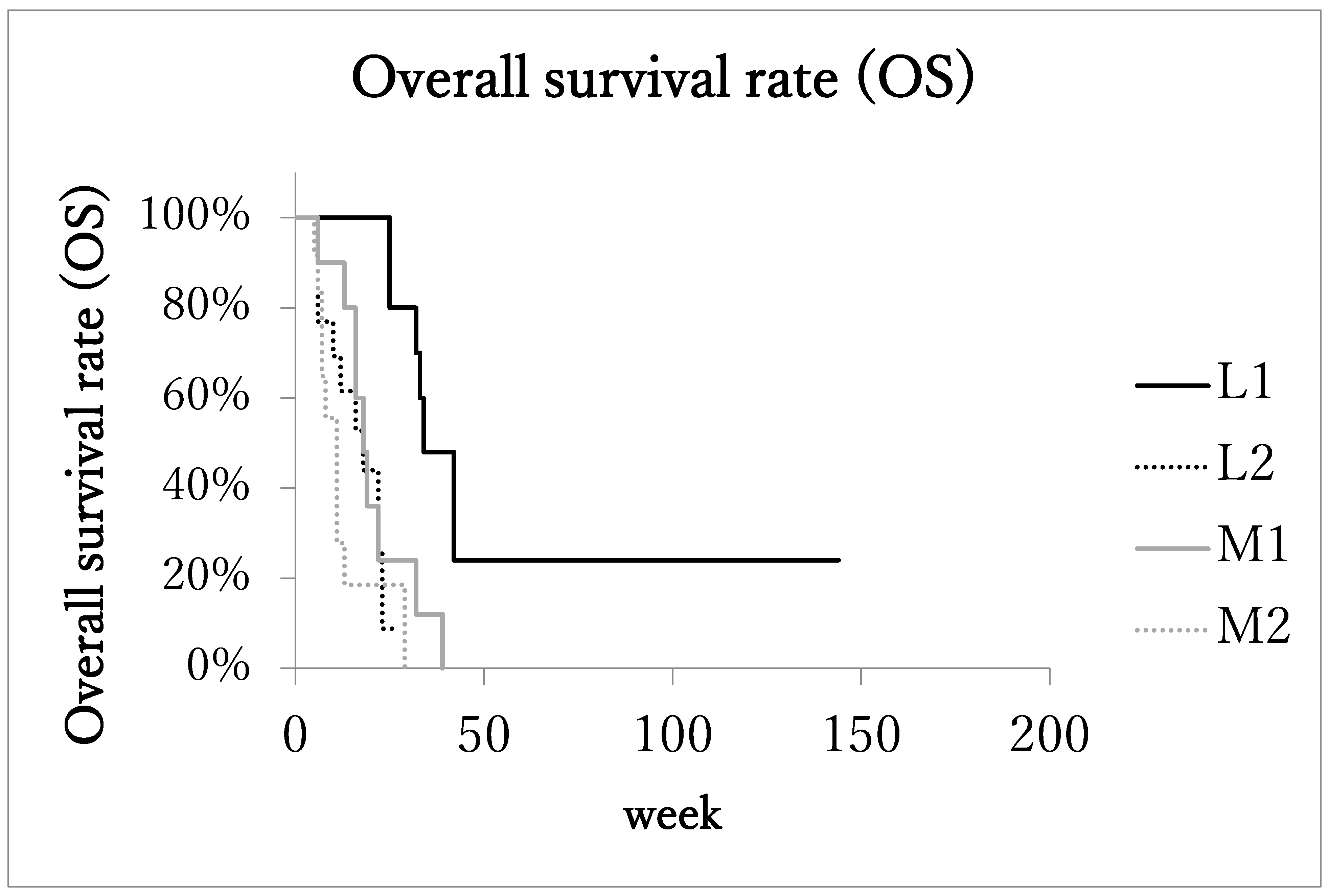
| Groups of PCA | Locally Advanced PCA | Metastatic Advanced PCA | ||
|---|---|---|---|---|
| Subgroups of PCA | L1 | L2 | M1 | M2 |
| Characteristics | n = 10 | n = 13 | n = 10 | n = 12 |
| Age (years) | 66.4 (43–82) | 75.0 (42–89) | 63.5 (36–87) | 67.5 (56–76) |
| Gender (Male/Female) | 8:2 | 5:8 | 6:4 | 7:5 |
| Location of mass (head:body:tail) | 7:3:0 | 9:3:1 | 3:7:0 | 5:3:4 |
| Maximum tumor size in average cm | 3.6 (3.0–4.2) | 4.2 (3.0–5.5) | 4.7 (3.5–6.0) | 4.9 (4.0–5.8) |
| Performance status 0 | 5 | 2 | 2 | 2 |
| Performance status 1 | 4 | 7 | 4 | 4 |
| Performance status 2 | 1 | 4 | 4 | 4 |
| Number of treatment courses | 4.9 (2–9) | 2.8 (1–8) | 3.6 (2–9) | 1.7 (1–4) |
| Time of uninterrupted systemic chemotherapy after the last treatment course (months) | 11 (5–24) | 5.2 (1–12) | 8.3 (4–17) | 7.5 (2–28) |
| Tumor response (RECIST) CR/PR/SD/PD | 4/6/0 | 0/12/1 | 0/9/1 | 0/6/6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mori, H.; Tanoue, S.; Takaji, R.; Ueda, S.; Okahara, M.; Ueda, S.S. Arterial Administration of DNA Crosslinking Agents with Restraint of Homologous Recombination Repair by Intravenous Low-Dose Gemcitabine Is Effective for Locally Advanced Pancreatic Cancer. Cancers 2022, 14, 220. https://doi.org/10.3390/cancers14010220
Mori H, Tanoue S, Takaji R, Ueda S, Okahara M, Ueda SS. Arterial Administration of DNA Crosslinking Agents with Restraint of Homologous Recombination Repair by Intravenous Low-Dose Gemcitabine Is Effective for Locally Advanced Pancreatic Cancer. Cancers. 2022; 14(1):220. https://doi.org/10.3390/cancers14010220
Chicago/Turabian StyleMori, Hiromu, Shuichi Tanoue, Ryo Takaji, Shinya Ueda, Mika Okahara, and Saori Sugi Ueda. 2022. "Arterial Administration of DNA Crosslinking Agents with Restraint of Homologous Recombination Repair by Intravenous Low-Dose Gemcitabine Is Effective for Locally Advanced Pancreatic Cancer" Cancers 14, no. 1: 220. https://doi.org/10.3390/cancers14010220
APA StyleMori, H., Tanoue, S., Takaji, R., Ueda, S., Okahara, M., & Ueda, S. S. (2022). Arterial Administration of DNA Crosslinking Agents with Restraint of Homologous Recombination Repair by Intravenous Low-Dose Gemcitabine Is Effective for Locally Advanced Pancreatic Cancer. Cancers, 14(1), 220. https://doi.org/10.3390/cancers14010220