Defining the Criteria for Reflex Testing for BRAF Mutations in Cutaneous Melanoma Patients
Simple Summary
Abstract
1. Introduction
2. Pathophysiology
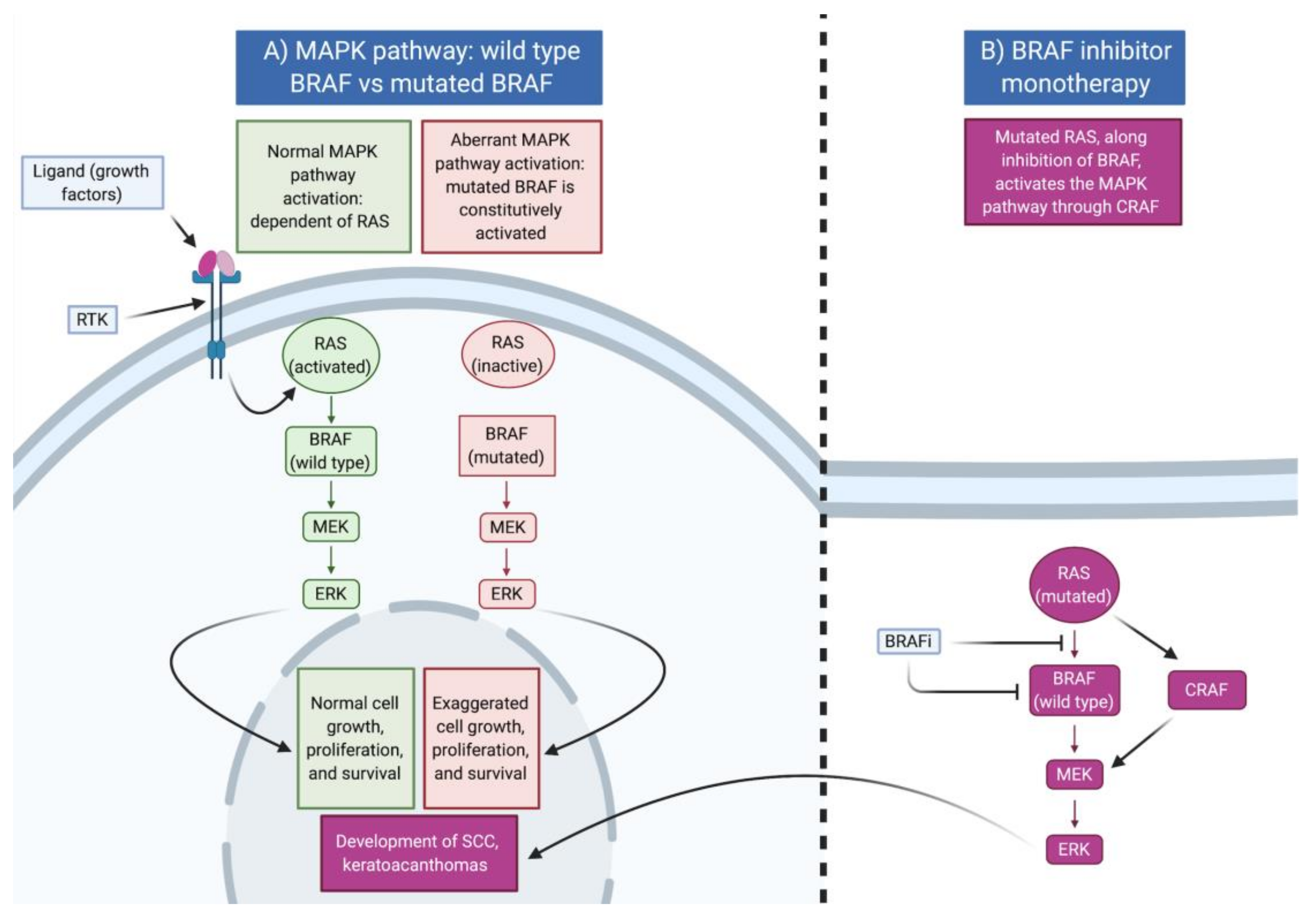
3. MAP Kinase Pathway
4. Clinical Presentation of BRAF-Mutated Melanomas and the Use of BRAF as a Prognostic or Treatment Response Predictive Marker
5. BRAF Testing at the Time of Diagnosis
5.1. Overview of Diagnosis
| Patient Features | Primary Melanoma | Metastatic Melanoma |
|---|---|---|
| BRAF mutation prevalence | Primary melanoma: 33–47% [6] | Metastatic melanoma: 41–55% [6] |
| Recurrent melanoma found to have higher frequency of BRAF mutation [14] | - | |
| Patient Features | ||
| Age of diagnosis | <50 [6,30] | Younger individuals [6] |
| UV exposure | High estimated lifetime exposure [28] and early-life exposure [28,32] | - |
| Total body nevus count | Patients with high number of nevi on back (>14) [28] more likely to harbor a BRAF mutation | - |
| Chronic sun-damaged skin | Fewer signs of chronic sun damage [30], such as lentigines [32] and solar elastosis [14,28] | Less chronic sun damage [6] |
| Melanoma Features | ||
| Number of primary lesions | - | Occult or 1 lesion [6] |
| Location of primary melanoma | Truncal location [6,30,41] | Truncal location [6] |
| Melanoma subtype | Superficial spreading [30] or nodular [14] | - |
| Pigmentation | Presence of pigmentation on pathology and as detected by patient [32] | - |
| Breslow thickness (of primary) | BRAF mutation associated with increased tumor thickness compared to wildtype [42,43] | - |
| Ulceration (of primary) | BRAF mutation associated with the presence of ulceration [14,41,44,45] | No association [6] |
| Dermoscopy features | Irregular peripheral streaks [31], blue-white veil [33], and “peppering” [34] | - |
| Outcomes | ||
| Stage at presentation | Presentation at a more advanced stage is associated with BRAF mutation [30,35] | |
| Response to chemotherapy | - | No association [6] |
| Response to BRAF/MEK inhibitor | - | Highly predictive of response to therapy [37] |
| Disease-free interval (primary diagnosis to first distant metastasis) | - | No association [6] |
| Outcome (survival) | No association [6] | Further investigation necessary |
5.2. Methods of BRAF Mutation Testing
5.3. Immunohistochemistry (IHC)
5.4. Real-Time PCR
| Features | IHC | RT-PCR | HRM | Sanger | Pyrosequencing | NGS | |
|---|---|---|---|---|---|---|---|
| Cobas® | THxID® | ||||||
| Detection of mutations [50,52] | VE1 antibody for V600E | V600E | V600E V600K | Indirectly detects mutations | Whole exon, detects rare mutations | Optimized for V600 mutations | Whole exon, detects rare mutations |
| Sensitivity | Up to 98.6% [51] | 95% [53] | >96% (V600E) >92% (V600K) [50] | 99% [52] | 92.5% (for V600E) [53] | 90 to 100% [52,54] | 99% [55] |
| Specificity | 97.7% [51] | 98% [50] | 100% [50] | 100% [52] | 100% [52] | 95 to 100% [52,54] | 100% [55] |
| Limit of detection (i.e., proportion of cells that are positive) | Few cells [56] | 7% [52] | 5% [50] | 6.6% [50] | 6.6% [57] | 5.0% [58] | 2% [52] |
| Turnaround time [52] | <1 day | 1 day | 1 day | Up to 3 days | 2 days | Up to 5 days | |
| Cost [52] | Low | Medium | Low | Medium | High | Very high | |
5.5. High-Resolution Melt
5.6. Sanger Sequencing
5.7. Pyrosequencing
5.8. Next-Generation Sequencing (NGS)
6. Implementation of Reflex Testing
7. Treatment
| Malignancy | Biomarker | Predictive Value | Patient Population | Reflex Testing Used | Outcomes Observed |
|---|---|---|---|---|---|
| Breast cancer | Oncotype Dx multigene assay | Predictive of chemotherapy benefit | Stage I, II ER+/PR+/HER2− | Reflex testing criteria developed for surgeons to order the test immediately after post-operative pathology results are available [71] | Incorporation of Oncotype DX testing reduces unwarranted chemotherapy use, improves life expectancy, and is cost-effective [72]. The introduction of reflex criteria testing for surgeons to implement reduced time from surgery to initiation of chemotherapy by 6.4 days [73] |
| Colon cancer | KRAS | Predictive for resistance to anti-EGFR therapy | Patients evaluated for metastatic disease, whenever anti-EGFR therapy is considered | Reflex KRAS testing is requested in metastatic cases of colon cancer starting second-line therapy [74] | Reflex testing offers maximal lead time to identify patients suitable for third-line anti-EGFR therapy [74] |
| Lung cancer (NSCLC) | EGFR ALK | Positive predictor of treatment with EGFR tyrosine kinase inhibitors or ALK tyrosine kinase inhibitors, respectively | Patients with advanced lung cancer who are candidates for targeted therapy. The NCCN recommends molecular profiling for all patients with metastatic NSCLC. | Reflex testing of ALK and EGFR by pathologists at the time of diagnosis of NSCLC [63] | Reduces the median time to treatment using systemic therapy by 10 days [63] |
| EGFR, ALK, ROS1, MET, RET, KRAS BRAF, PDL1, HER2 | EGFR and ALK have the greatest evidence supporting targeted therapy for mutations. Rearrangements in other genes have lower-level evidence to direct management | Patients with newly diagnosed lung adenocarcinoma of any pathologic stage | Retrospective examination of the effect of reflex testing of molecular biomarkers at the time of pathologic diagnosis of lung adenocarcinoma [44] | Reduces the average turnaround time of testing by 26 days and almost doubles the rate of variants that are detected [44] | |
| Ovarian cancer | BRCA1 BRCA 2 | Predictive of response to PARP inhibitor and eligibility for genetic counseling | Women with high-grade serous carcinoma are eligible for BRCA mutation testing | Reflex tumor testing of all high-grade serous carcinoma at initial diagnosis [45] | Reflex testing identifies more BRCA mutations, reduces the time to critical treatment decision, and helps to determine other BRCA mutation carriers that may benefit from preventative treatment [45] |
| Prostate cancer (castration resistant) | BRCA 1 BRCA 2 ATM | Predictive of response with PARP and other DDR enzyme inhibitors | Men with metastatic prostate cancer | Suggestion to examine whether men with earlier-stage disease may benefit from reflex testing strategies [75] | Yet to be tested |
| Malignancy | Biomarker | Purpose | When to Test | Utility of Reflex Testing |
|---|---|---|---|---|
| Anal squamous cell carcinoma | HPV | Screening test for anal squamous cell carcinoma (SCC) | Annual rectal exam in high-risk groups such as MSM | Reflex testing of HPV for high-risk patients (HIV+ and other immunocompromised individuals) to screen for anal squamous cell carcinoma |
| Chronic myeloid leukemia (CML) | BCR-ABL | Establish initial patient baseline level and assess response to therapy in follow-up samples | As part of workup for CML or acute lymphoblastic leukemia (ALL) | Following a positive BCR-ABL1 RT-PCR result, a reflex test is performed to provide a quantitative measurement of BCR/ABL1 mRNA transcript to be recorded as the baseline level [76] |
| Chronic myeloid leukemia (CML) Acute myeloid leukemia with myelodysplasia-related changes (AML-MRC) | KIT | Identifying the co-occurrence of systemic mastocytosis | Patients diagnosed with CML or AML-MRC, with an identified D816V mutation of KIT | Identifying systemic mastocytosis with associated hematologic malignancy allows for appropriate treatment of the systemic mastocytosis component [77] |
| Colon cancer | Mismatch repair genes | Genetic counseling to identify patients with Lynch syndrome and also predictive of response to immune-checkpoint inhibitors | As detailed in the Bethesda testing guidelines for Lynch syndrome | Ontario is performing reflex IHC in colorectal cases presenting before the age of 40 [78] to identify Lynch syndrome patients |
| Endometrial cancer | Mismatch repair genes | Detection of Lynch syndrome | As detailed in the Bethesda testing guidelines for Lynch syndrome | Implementation of reflex testing of all newly diagnosed endometrial cancers with IHC is suggested to identify patients, who are at high risk and could benefit from prevention strategies [79] |
| Head and neck squamous cell carcinoma | HPV | Positive prognostic and predictive marker of response to treatment | Patients with newly diagnosed oropharyngeal squamous cell carcinoma | Reflex testing of oropharyngeal primary tumors with p16 IHC [80] |
| Pancreatic cancer | BRCA1 BRCA2 | Genetic counseling to identify other potential carriers of founder mutations. Predictive of response to PARP inhibitors | All patients with pancreatic cancer (NCCN guidelines) | Reflex testing of founder mutations recommended for patients with pancreatic adenocarcinoma with French Canadian or Ashkenazi Jewish ancestry [81] |
8. Recommendation on BRAF Reflex Testing
| Stage | BRAF Status | Tumor | Recommended Systemic Treatment Options |
|---|---|---|---|
| I | Any | Resected | None |
| II | Any | Resected | None |
| III | Wildtype (adjuvant) | Resected | Nivolumab Pembrolizumab Ipilimumab #,* |
| Wildtype (therapeutic) | Unresected | Nivolumab Pembrolizumab Ipilimumab * Nivolumab/ipilimumab | |
| BRAF-mutated (adjuvant) | Resected | Nivolumab Pembrolizumab Ipilimumab Dabrafenib/trametinib | |
| BRAF-mutated (therapeutic) | Unresected | Nivolumab Pembrolizumab Ipilimumab * Dabrafenib/trametinib Nivolumab/ipilimumab | |
| IV | Any (adjuvant) | Resected | Nivolumab Pembrolizumab Ipilimumab * Nivolumab/ipilimumab |
| BRAF-mutated (therapeutic) | Unresected | Nivolumab Pembrolizumab Ipilimumab * Dabrafenib/trametinib Vemurafenib/cobimetinib Encorafenib/binimetinib Nivolumab/ipilimumab |
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ghazawi, F.M.; Cyr, J.; Darwich, R.; Le, M.; Rahme, E.; Moreau, L.; Netchiporouk, E.; Zubarev, A.; Roshdy, O.; Glassman, S.J.; et al. Cutaneous malignant melanoma incidence and mortality trends in Canada: A comprehensive population-based study. J. Am. Acad. Dermatol. 2019, 80, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Ghazawi, F.M.; Le, M.; Lagacé, F.; Cyr, J.; AlGhazawi, N.; Zubarev, A.; Roy, S.F.; Rahme, E.; Netchiporouk, E.; Roshdy, O.; et al. Incidence, Mortality, and Spatiotemporal Distribution of Cutaneous Malignant Melanoma Cases Across Canada. J. Cutan. Med. Surg. 2019, 23, 394–412. [Google Scholar] [CrossRef] [PubMed]
- Muntyanu, A.; Savin, E.; Ghazawi, F.M.; Alakel, A.; Zubarev, A.; Litvinov, I.V. Geographic Variations in Cutaneous Melanoma Distribution in the Russian Federation. Dermatology 2020, 236, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ghazawi, F.M.; Le, M.; Alghazawi, N.; Rahme, E.; Moreau, L.; Netchiporouk, E.; Zubarev, A.; Roshdy, O.; Glassman, S.J.; Sasseville, D.; et al. Trends in incidence of cutaneous malignant melanoma in Canada: 1992-2010 versus 2011-2015. J. Am. Acad. Dermatol. 2019, 80, 1157–1159. [Google Scholar] [CrossRef]
- Petrella, T.; Ernst, S.; Spatz, A.; Claveau, J. Canadian perspective on the clinical management of metastatic melanoma. Lung 2012, 70, 87. [Google Scholar]
- Long, G.V.; Menzies, A.M.; Nagrial, A.M.; Haydu, L.E.; Hamilton, A.L.; Mann, G.J.; Hughes, T.M.; Thompson, J.F.; Scolyer, R.A.; Kefford, R.F. Prognostic and Clinicopathologic Associations of Oncogenic BRAF in Metastatic Melanoma. J. Clin. Oncol. 2011, 29, 1239–1246. [Google Scholar] [CrossRef]
- Lawrence, M.S.; Stojanov, P.; Polak, P.; Kryukov, G.V.; Cibulskis, K.; Sivachenko, A.; Carter, S.L.; Stewart, C.; Mermel, C.H.; Roberts, S.A.; et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nat. Cell Biol. 2013, 499, 214–218. [Google Scholar] [CrossRef]
- Eddy, K.; Shah, R.; Chen, S. Decoding Melanoma Development and Progression: Identification of Therapeutic Vulnerabilities. Front. Oncol. 2021, 10, 626129. [Google Scholar] [CrossRef]
- Ito, T.; Tanaka, Y.; Murata, M.; Kaku-Ito, Y.; Furue, K.; Furue, M. BRAF Heterogeneity in Melanoma. Curr. Treat. Options Oncol. 2021, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Newton-Bishop, J.; Bishop, D.; Harland, M. Melanoma Genomics. Acta Derm. Venereol. 2020, 100, adv00138. [Google Scholar] [CrossRef]
- McCain, J. The MAPK (ERK) Pathway: Investigational Combinations for the Treatment Of BRAF-Mutated Metastatic Melanoma. Pharm. Ther. 2013, 38, 96–108. [Google Scholar]
- Shain, A.H.; Bastian, B.C. From melanocytes to melanomas. Nat. Rev. Cancer 2016, 16, 345–358. [Google Scholar] [CrossRef]
- Burotto, M.; Chiou, V.L.; Lee, J.-M.; Kohn, E.C. The MAPK pathway across different malignancies: A new perspective. Cancer 2014, 120, 3446–3456. [Google Scholar] [CrossRef] [PubMed]
- Spathis, A.; Katoulis, A.C.; Damaskou, V.; Liakou, A.I.; Kottaridi, C.; Leventakou, D.; Sgouros, D.; Mamantopoulos, A.; Rigopoulos, D.; Karakitsos, P.; et al. BRAF mutation status in primary, recurrent, and metastatic malignant melanoma and its relation to histopathological parameters. Dermatol. Pract. Concept. 2019, 9, 54–62. [Google Scholar] [CrossRef]
- Bradish, J.R.; Cheng, L. Molecular pathology of malignant melanoma: Changing the clinical practice paradigm toward a personalized approach. Hum. Pathol. 2014, 45, 1315–1326. [Google Scholar] [CrossRef] [PubMed]
- Wan, P.T.; Garnett, M.J.; Roe, S.M.; Lee, S.; Niculescu-Duvaz, D.; Good, V.M.; Jones, C.M.; Marshall, C.J.; Springer, C.J.; Cancer Genome Project; et al. Mechanical acts RAF-Erk signal pathway by oncological mutations B-Raf. Cell 2004, 116, 855–867. [Google Scholar] [CrossRef]
- Ascierto, P.A.; McArthur, G.A.; Dréno, B.; Atkinson, V.; Liszkay, G.; Di Giacomo, A.M.; Mandalà, M.; Demidov, L.; Stroyakovskiy, D.; Thomas, L.; et al. Cobimetinib combined with vemurafenib in advanced BRAFV600-mutant melanoma (coBRIM): Updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016, 17, 1248–1260. [Google Scholar] [CrossRef]
- Long, G.V.; Flaherty, K.T.; Stroyakovskiy, D.; Gogas, H.; Levchenko, E.; de Braud, F.; Larkin, J.; Garbe, C.; Jouary, T.; Hauschild, A.; et al. Dabrafenib plus trametinib versus dabrafenib monotherapy in patients with metastatic BRAF V600E/K-mutant melanoma: Long-term survival and safety analysis of a phase 3 study. Ann. Oncol. 2017, 28, 1631–1639. [Google Scholar] [CrossRef]
- Robert, C.; Karaszewska, B.; Schachter, J.; Rutkowski, P.; Mackiewicz, A.; Stroiakovski, D.; Lichinitser, M.; Dummer, R.; Grange, F.; Mortier, L.; et al. Improved Overall Survival in Melanoma with Combined Dabrafenib and Trametinib. N. Engl. J. Med. 2015, 372, 30–39. [Google Scholar] [CrossRef]
- Shi, H.; Hugo, W.; Kong, X.; Hong, A.; Koya, R.C.; Moriceau, G.; Chodon, T.; Guo, R.; Johnson, D.B.; Dahlman, K.B.; et al. Acquired Resistance and Clonal Evolution in Melanoma during BRAF Inhibitor Therapy. Cancer Discov. 2014, 4, 80–93. [Google Scholar] [CrossRef]
- Robert, C.; Grob, J.J.; Stroyakovskiy, D.; Karaszewska, B.; Hauschild, A.; Levchenko, E.; Sileni, V.C.; Schachter, J.; Garbe, C.; Bondarenko, I.; et al. Five-Year Outcomes with Dabrafenib plus Trametinib in Metastatic Melanoma. N. Engl. J. Med. 2019, 381, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Gutzmer, R.; Stroyakovskiy, D.; Gogas, H.; Robert, C.; Lewis, K.; Protsenko, S.; Pereira, R.P.; Eigentler, T.; Rutkowski, P.; Demidov, L.; et al. Atezolizumab, vemurafenib, and cobimetinib as first-line treatment for unresectable advanced BRAFV600 mutation-positive melanoma (IMspire150): Primary analysis of the randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2020, 395, 1835–1844. [Google Scholar] [CrossRef]
- Heidorn, S.J.; Milagre, C.; Whittaker, S.; Nourry, A.; Niculescu-Duvas, I.; Dhomen, N.; Hussain, J.; Reis-Filho, J.S.; Springer, C.J.; Pritchard, C.; et al. Kinase-Dead BRAF and Oncogenic RAS Cooperate to Drive Tumor Progression through CRAF. Cell 2010, 140, 209–221. [Google Scholar] [CrossRef]
- Su, F.; Viros, A.; Milagre, C.; Trunzer, K.; Bollag, G.; Spleiss, O.; Reis-Filho, J.S.; Kong, X.; Koya, R.C.; Flaherty, K.T.; et al. RASMutations in Cutaneous Squamous-Cell Carcinomas in Patients Treated with BRAF Inhibitors. N. Engl. J. Med. 2012, 366, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Montagut, C.; Sharma, S.V.; Shioda, T.; McDermott, U.; Ulman, M.; Ulkus, L.E.; Dias-Santagata, D.; Stubbs, H.; Lee, D.Y.; Singh, A.; et al. Elevated CRAF as a Potential Mechanism of Acquired Resistance to BRAF Inhibition in Melanoma. Cancer Res. 2008, 68, 4853–4861. [Google Scholar] [CrossRef]
- Arozarena, I.; Wellbrock, C. Overcoming resistance to BRAF inhibitors. Ann. Transl. Med. 2017, 5, 387. [Google Scholar] [CrossRef] [PubMed]
- Raaijmakers, M.I.G.; Widmer, D.S.; Narechania, A.; Eichhoff, O.; Freiberger, S.N.; Wenzina, J.; Cheng, P.F.; Mihic-Probst, D.; DeSalle, R.; Dummer, R.; et al. Co-existence of BRAF and NRAS driver mutations in the same melanoma cells results in heterogeneity of targeted therapy resistance. Oncotarget 2016, 7, 77163–77174. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.E.; Edmiston, S.N.; Alexander, A.; Millikan, R.C.; Groben, P.A.; Hao, H.; Tolbert, D.; Berwick, M.; Busam, K.; Begg, C.B.; et al. Number of Nevi and Early-Life Ambient UV Exposure Are Associated with BRAF-Mutant Melanoma. Cancer Epidemiol. Biomark. Prev. 2007, 16, 991–997. [Google Scholar] [CrossRef]
- Network, N.C.C. Cutaneous Melanoma. Available online: https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma_blocks.pdf (accessed on 2 March 2020).
- Kim, S.Y.; Hahn, H.J.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J.; Kim, S.N. Metaanalysis of BRAF mutations and clinicopathologic characteristics in primary melanoma. J. Am. Acad. Dermatol. 2015, 72, 1036–1046.e2. [Google Scholar] [CrossRef]
- Bombonato, C.; Ribero, S.; Pozzobon, F.; Puig-Butille, J.; Badenas, C.; Carrera, C.; Malvehy, J.; Moscarella, E.; Lallas, A.; Piana, S.; et al. Association between dermoscopic and reflectance confocal microscopy features of cutaneous melanoma with BRAF mutational status. J. Eur. Acad. Dermatol. Venereol. 2016, 31, 643–649. [Google Scholar] [CrossRef]
- Liu, W.; Kelly, J.W.; Trivett, M.; Murray, W.K.; Dowling, J.P.; Wolfe, R.; Mason, G.; Magee, J.; Angel, C.; Dobrovic, A.; et al. Distinct Clinical and Pathological Features Are Associated with the BRAFT1799A(V600E) Mutation in Primary Melanoma. J. Investig. Dermatol. 2007, 127, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Armengot-Carbó, M.; Nagore, E.; García-Casado, Z.; Botella-Estrada, R. The association between dermoscopic features and BRAF mutational status in cutaneous melanoma: Significance of the blue-white veil. J. Am. Acad. Dermatol. 2018, 78, 920–926.e4. [Google Scholar] [CrossRef] [PubMed]
- Pozzobon, F.; Puig-Butillé, J.; González-Alvarez, T.; Carrera, C.; Aguilera, P.; Alos, L.; Badenas, C.; Grichnik, J.; Malvehy, J.; Puig, S. Dermoscopic criteria associated with BRAF and NRAS mutation status in primary cutaneous melanoma. Br. J. Dermatol. 2014, 171, 754–759. [Google Scholar] [CrossRef]
- Mann, G.J.; Pupo, G.M.; Campain, A.E.; Carter, C.D.; Schramm, S.-J.; Pianova, S.; Gerega, S.K.; De Silva, C.; Lai, K.; Wilmott, J.S.; et al. BRAF Mutation, NRAS Mutation, and the Absence of an Immune-Related Expressed Gene Profile Predict Poor Outcome in Patients with Stage III Melanoma. J. Investig. Dermatol. 2013, 133, 509–517. [Google Scholar] [CrossRef]
- Hugdahl, E.; Kalvenes, M.B.; Puntervoll, H.E.; Ladstein, R.G.; Akslen, L.A. BRAF-V600E expression in primary nodular melanoma is associated with aggressive tumour features and reduced survival. Br. J. Cancer 2016, 114, 801–808. [Google Scholar] [CrossRef]
- Tarhini, A.; Kudchadkar, R.R. Predictive and on-treatment monitoring biomarkers in advanced melanoma: Moving toward personalized medicine. Cancer Treat. Rev. 2018, 71, 8–18. [Google Scholar] [CrossRef]
- Ascierto, P.A.; Dummer, R.; Gogas, H.J.; Flaherty, K.T.; Arance, A.; Mandala, M.; Liszkay, G.; Garbe, C.; Schadendorf, D.; Krajsova, I.; et al. Update on tolerability and overall survival in COLUMBUS: Landmark analysis of a randomised phase 3 trial of encorafenib plus binimetinib vs vemurafenib or encorafenib in patients with BRAF V600–mutant melanoma. Eur. J. Cancer 2020, 126, 33–44. [Google Scholar] [CrossRef]
- Coit, D.G.; Thompson, J.A.; Albertini, M.R.; Barker, C.; Carson, W.E.; Contreras, C.; Daniels, G.A.; DiMaio, D.; Fields, R.C.; Fleming, M.D.; et al. Cutaneous Melanoma, Version 2.2019, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2019, 17, 367–402. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, P.; Friedlander, P.; Zakaria, E.A.; Kandil, E. Impact of BRAF mutation status in the prognosis of cutaneous melanoma: An area of ongoing research. Ann. Transl. Med. 2015, 3, 24. [Google Scholar]
- Gershenwald, J.E.; Scolyer, R.A.; Hess, K.R.; Sondak, V.K.; Long, G.V.; Ross, M.I.; Lazar, A.J.; Faries, M.B.; Kirkwood, J.M.; McArthur, G.A.; et al. Melanoma staging: Evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J. Clin. 2017, 67, 472–492. [Google Scholar] [CrossRef] [PubMed]
- Ellerhorst, J.A.; Greene, V.R.; Ekmekcioglu, S.; Warneke, C.L.; Johnson, M.M.; Cooke, C.P.; Wang, L.-E.; Prieto, V.G.; Gershenwald, J.E.; Wei, Q.; et al. Clinical Correlates of NRAS and BRAF Mutations in Primary Human Melanoma. Clin. Cancer Res. 2011, 17, 229–235. [Google Scholar] [CrossRef] [PubMed]
- García-Casado, Z.; Traves, V.; Banuls, J.; Niveiro, M.; Gimeno-Carpio, E.; Jimenez-Sanchez, A.; Moragon, M.; Onrubia, J.; Oliver, V.; Kumar, R. BRAF, NRAS and MC 1R status in a prospective series of primary cutaneous melanoma. Br. J. Dermatol. 2015, 172, 1128–1131. [Google Scholar] [CrossRef] [PubMed]
- Anand, K.; Phung, T.L.; Bernicker, E.H.; Cagle, P.T.; Olsen, R.J.; Thomas, J.S. Clinical Utility of Reflex Ordered Testing for Molecular Biomarkers in Lung Adenocarcinoma. Clin. Lung Cancer 2020, 21, 437–442. [Google Scholar] [CrossRef] [PubMed]
- The Society of Gynecologic Oncology of Canada. Why is Tumour Testing in Ovarian Cancer Needed in Canada? An Opinion Statement Developed by the National BRCA Collaborative. Available online: http://g-o-c.org/wp-content/uploads/2020/02/20BRCACollaborative_TumourTestinginCanada_FINAL_Jan30.pdf (accessed on 2 March 2021).
- Michielin, O.; Van Akkooi, A.C.J.; Ascierto, P.A.; Dummer, R.; Keilholz, U.; ESMO Guidelines Committee. Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 1884–1901. [Google Scholar] [CrossRef]
- Miller, R.; Walker, S.; Shui, I.; Brandtmüller, A.; Cadwell, K.; Scherrer, E. Epidemiology and survival outcomes in stages II and III cutaneous melanoma: A systematic review. Melanoma Manag. 2020, 7, 39–53. [Google Scholar] [CrossRef]
- Tan, S.Y.; Najita, J.; Li, X.; Strazzulla, L.C.; Dunbar, H.; Lee, M.-Y.; Seery, V.J.; Buchbinder, E.I.; Tawa, N.E.; McDermott, D.F.; et al. Clinicopathologic features correlated with paradoxical outcomes in stage IIC versus IIIA melanoma patients. Melanoma Res. 2019, 29, 70–76. [Google Scholar] [CrossRef]
- Normanno, N. BRAF in Melanoma: ESMO Biomarker Factsheet. Available online: https://oncologypro.esmo.org/education-library/factsheets-on-biomarkers/braf-in-melanoma (accessed on 23 April 2021).
- Cheng, L.; Lopez-Beltran, A.; Massari, F.; MacLennan, G.T.; Montironi, R. Molecular testing for BRAF mutations to inform melanoma treatment decisions: A move toward precision medicine. Mod. Pathol. 2018, 31, 24–38. [Google Scholar] [CrossRef]
- Manfredi, L.; Meyer, N.; Tournier, E.; Grand, D.; Uro-Coste, E.; Rochaix, P.; Brousset, P.; Lamant, L. Highly Concordant Results Between Immunohistochemistry and Molecular Testing of Mutated V600E BRAF in Primary and Metastatic Melanoma. Acta Derm. Venereol. 2016, 96, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Ihle, M.A.; Fassunke, J.; König, K.; Grünewald, I.; Schlaak, M.; Kreuzberg, N.; Tietze, L.; Schildhaus, H.-U.; Büttner, R.; Merkelbach-Bruse, S. Comparison of high resolution melting analysis, pyrosequencing, next generation sequencing and immunohistochemistry to conventional Sanger sequencing for the detection of p.V600E and non-p.V600E BRAFmutations. BMC Cancer 2014, 14, 13. [Google Scholar] [CrossRef]
- Anderson, S.; Bloom, K.J.; Vallera, D.U.; Rueschoff, J.; Meldrum, C.; Schilling, R.; Kovach, B.; Lee, J.R.-J.; Ochoa, P.; Langland, R.; et al. Multisite Analytic Performance Studies of a Real-Time Polymerase Chain Reaction Assay for the Detection of BRAF V600E Mutations in Formalin-Fixed, Paraffin-Embedded Tissue Specimens of Malignant Melanoma. Arch. Pathol. Lab. Med. 2012, 136, 1385–1391. [Google Scholar] [CrossRef] [PubMed]
- Colomba, E.; Hélias-Rodzewicz, Z.; Von Deimling, A.; Marin, C.; Terrones, N.; Pechaud, D.; Surel, S.; Côté, J.-F.; Peschaud, F.; Capper, D.; et al. Detection of BRAF p.V600E Mutations in Melanomas. J. Mol. Diagn. 2013, 15, 94–100. [Google Scholar] [CrossRef]
- Mancini, I.; Simi, L.; Salvianti, F.; Castiglione, F.; Sonnati, G.; Pinzani, P. Simi Analytical Evaluation of an NGS Testing Method for Routine Molecular Diagnostics on Melanoma Formalin-Fixed, Paraffin-Embedded Tumor-Derived DNA. Diagnostics 2019, 9, 117. [Google Scholar] [CrossRef]
- Bisschop, C.; ter Elst, A.; Bosman, L.J.; Platteel, I.; Jalving, M.; Berg, A.V.D.; Diepstra, A.; van Hemel, B.; Diercks, G.F.; Hospers, G.A.; et al. Rapid BRAF mutation tests in patients with advanced melanoma: Comparison of immunohistochemistry, Droplet Digital PCR, and the Idylla Mutation Platform. Melanoma Res. 2018, 28, 96–104. [Google Scholar] [CrossRef]
- A Monzon, F.; Ogino, S.; Hammond, M.E.H.; Halling, K.C.; Bloom, K.J.; Nikiforova, M.N. The role of KRAS mutation testing in the management of patients with metastatic colorectal cancer. Arch. Pathol. Lab. Med. 2009, 133, 1600–1606. [Google Scholar] [CrossRef]
- Tsiatis, A.C.; Norris-Kirby, A.; Rich, R.G.; Hafez, M.J.; Gocke, C.D.; Eshleman, J.R.; Murphy, K.M. Comparison of Sanger Sequencing, Pyrosequencing, and Melting Curve Analysis for the Detection of KRAS Mutations: Diagnostic and Clinical Implications. J. Mol. Diagn. 2010, 12, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Melchior, L.; Grauslund, M.; Bellosillo, B.; Montagut, C.; Torres, E.; Moragón, E.; Micalessi, I.; Frans, J.; Noten, V.; Bourgain, C.; et al. Multi-center evaluation of the novel fully-automated PCR-based Idylla™ BRAF Mutation Test on formalin-fixed paraffin-embedded tissue of malignant melanoma. Exp. Mol. Pathol. 2015, 99, 485–491. [Google Scholar] [CrossRef]
- Chen, N.; Wang, Y.-Y.; Chuai, Z.-R.; Huang, J.-F.; Wang, Y.-X.; Liu, K.; Zhang, L.-Q.; Yang, Z.; Shi, D.-C.; Liu, Q.; et al. High-Resolution Melting Analysis for accurate detection of BRAF mutations: A systematic review and meta-analysis. Sci. Rep. 2015, 4, 4168. [Google Scholar] [CrossRef] [PubMed]
- Vanni, I.; Tanda, E.T.; Spagnolo, F.; Andreotti, V.; Bruno, W.; Ghiorzo, P. The Current State of Molecular Testing in the BRAF-Mutated Melanoma Landscape. Front. Mol. Biosci. 2020, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Qu, K.; Pan, Q.; Zhang, X.; Rodriguez, L.; Zhang, K.; Li, H.; Ho, A.; Sanders, H.; Sferruzza, A.; Cheng, S.-M.; et al. Detection of BRAF V600 Mutations in Metastatic Melanoma. J. Mol. Diagn. 2013, 15, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Cheema, P.K.; Raphael, S.; El-Maraghi, R.; Li, J.; McClure, R.; Zibdawi, L.; Chan, A.; Victor, J.; Dolley, A.; Dziarmaga, A. Rate of Egfr Mutation Testing for Patients with Nonsquamous Non-Small-Cell Lung Cancer with Implementation of Reflex Testing by Pathologists. Curr. Oncol. 2017, 24, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Barth, A.; Wanek, L.A.; Morton, D.L. Prognostic factors in 1521 melanoma patients with distant metastases. J. Am. Coll. Surg. 1995, 181, 193–201. [Google Scholar]
- Ascierto, P.A.; Ferrucci, P.F.; Fisher, R.; Del Vecchio, M.; Atkinson, V.; Schmidt, H.; Schachter, J.; Queirolo, P.; Long, G.V.; Di Giacomo, A.M.; et al. Dabrafenib, trametinib and pembrolizumab or placebo in BRAF-mutant melanoma. Nat. Med. 2019, 25, 941–946. [Google Scholar] [CrossRef]
- Grob, J.J.; Garbe, C.; Ascierto, P.; Larkin, J.; Dummer, R.; Schadendorf, D. Adjuvant melanoma therapy with new drugs: Should physicians continue to focus on metastatic disease or use it earlier in primary melanoma? Lancet Oncol. 2018, 19, e720–e725. [Google Scholar] [CrossRef]
- Long, G.V.; Stroyakovskiy, D.; Gogas, H.; Levchenko, E.; de Braud, F.; Larkin, J.; Garbe, C.; Jouary, T.; Hauschild, A.; Grob, J.-J.; et al. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: A multicentre, double-blind, phase 3 randomised controlled trial. Lancet 2015, 386, 444–451. [Google Scholar] [CrossRef]
- Larkin, J.; Ascierto, P.A.; Dréno, B.; Atkinson, V.; Liszkay, G.; Maio, M.; Mandalà, M.; Demidov, L.; Stroyakovskiy, D.; Thomas, L.; et al. Combined Vemurafenib and Cobimetinib in BRAF-Mutated Melanoma. N. Engl. J. Med. 2014, 371, 1867–1876. [Google Scholar] [CrossRef]
- Long, G.V.; Eroglu, Z.; Infante, J.; Patel, S.; Daud, A.; Johnson, D.B.; Gonzalez, R.; Kefford, R.; Hamid, O.; Schuchter, L.; et al. Long-Term Outcomes in Patients With BRAF V600–Mutant Metastatic Melanoma Who Received Dabrafenib Combined With Trametinib. J. Clin. Oncol. 2018, 36, 667–673. [Google Scholar] [CrossRef]
- El-Deiry, W.S.; Goldberg, R.M.; Lenz, H.; Shields, A.F.; Gibney, G.T.; Tan, A.R.; Brown, J.; Eisenberg, B.; Heath, E.I.; Phuphanich, S.; et al. The current state of molecular testing in the treatment of patients with solid tumors, 2019. CA Cancer J. Clin. 2019, 69, 305–343. [Google Scholar] [CrossRef] [PubMed]
- Natsuhara, K.H.; Losk, K.; King, T.A.; Lin, N.U.; Camuso, K.; Golshan, M.; Pochebit, S.; Brock, J.E.; Bunnell, C.A.; Freedman, R.A. Impact of Genomic Assay Testing and Clinical Factors on Chemotherapy Use After Implementation of Standardized Testing Criteria. Oncologist 2018, 24, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Katz, G.; Romano, O.; Foa, C.; Vataire, A.-L.; Chantelard, J.-V.; Hervé, R.; Barletta, H.; Durieux, A.; Martin, J.-P.; Salmon, R. Economic Impact of Gene Expression Profiling in Patients with Early-Stage Breast Cancer in France. PLoS ONE 2015, 10, e0128880. [Google Scholar] [CrossRef]
- Losk, K.; Freedman, R.A.; Lin, N.U.; Golshan, M.; Pochebit, S.M.; Lester, S.C.; Natsuhara, K.; Camuso, K.; King, T.A.; Bunnell, C.A. Implementation of Surgeon-Initiated Gene Expression Profile Testing (Oncotype DX) Among Patients With Early-Stage Breast Cancer to Reduce Delays in Chemotherapy Initiation. J. Oncol. Pract. 2017, 13, e815–e820. [Google Scholar] [CrossRef] [PubMed]
- Aubin, F.; Gill, S.; Burkes, R.; Colwell, B.; Kamel–Reid, S.; Koski, S.; Pollett, A.; Samson, B.; Tehfe, M.; Wong, R.; et al. Canadian Expert Group Consensus Recommendations: KRAS Testing in Colorectal Cancer. Curr. Oncol. 2011, 18, 180–184. [Google Scholar] [CrossRef]
- Giri, V. New Recommendations Offer Guidance for Clinicians and Patients on Genetic Testing for Prostate Cancer. Available online: https://ascopost.com/issues/july-10-2020/new-recommendations-offer-guidance-for-clinicians-and-patients-on-genetic-testing-for-prostate-cancer/ (accessed on 2 March 2021).
- Laboratories, M.C. BCR/ABL1 Qualitative Diagnostic Assay with Reflex to BCR/ABL1 p190 Quantitative Assay or BCR/ABL1 p210 Quantitative Assay, Varies. Available online: https://www.mayocliniclabs.com/test-catalog/Overview/65248 (accessed on 1 September 2020).
- Craig, J.W.; Hasserjian, R.P.; Kim, A.S.; Aster, J.C.; Pinkus, G.S.; Hornick, J.L.; Steensma, D.P.; Lindsley, R.C.; DeAngelo, D.J.; Morgan, E.A. Detection of the KITD816V mutation in myelodysplastic and/or myeloproliferative neoplasms and acute myeloid leukemia with myelodysplasia-related changes predicts concurrent systemic mastocytosis. Mod. Pathol. 2020, 33, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Assasi, N.; Blackhouse, G.; Campbell, K.; Weeks, L.; Levine, M. Mismatch Repair Deficiency Testing for Patients with Colorectal Cancer: A Clinical and Cost-Effectiveness Evaluation; CADTH: Ottawa, ON, Canada, 2015. [Google Scholar]
- Mills, A.M.; Liou, S.; Ford, J.M.; Berek, J.S.; Pai, R.K.; Longacre, T.A. Lynch Syndrome Screening Should Be Considered for All Patients With Newly Diagnosed Endometrial Cancer. Am. J. Surg. Pathol. 2014, 38, 1501–1509. [Google Scholar] [CrossRef]
- Lewis, J.S.; Beadle, B.; Bishop, J.A.; Chernock, R.D.; Colasacco, C.; Lacchetti, C.; Moncur, J.T.; Rocco, J.W.; Schwartz, M.R.; Seethala, R.R.; et al. Human Papillomavirus Testing in Head and Neck Carcinomas: Guideline From the College of American Pathologists. Arch. Pathol. Lab. Med. 2017, 142, 559–597. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.L.; Wong, C.; Cuggia, A.; Borgida, A.; Holter, S.; Hall, A.; Connor, A.A.; Bascuñana, C.; Asselah, J.; Bouganim, N.; et al. Reflex Testing for Germline BRCA1, BRCA2, PALB2, and ATM Mutations in Pancreatic Cancer: Mutation Prevalence and Clinical Outcomes From Two Canadian Research Registries. JCO Precis. Oncol. 2018, 1–16. [Google Scholar] [CrossRef]
- Dummer, R.; Hauschild, A.; Lindenblatt, N.; Pentheroudakis, G.; Keilholz, U. Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26, v126–v132. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, S.; Sikorski, D.; Xu, H.; Zubarev, A.; Chergui, M.; Lagacé, F.; Miller, W.H., Jr.; Redpath, M.; Ghazal, S.; Butler, M.O.; et al. Defining the Criteria for Reflex Testing for BRAF Mutations in Cutaneous Melanoma Patients. Cancers 2021, 13, 2282. https://doi.org/10.3390/cancers13092282
Zhou S, Sikorski D, Xu H, Zubarev A, Chergui M, Lagacé F, Miller WH Jr., Redpath M, Ghazal S, Butler MO, et al. Defining the Criteria for Reflex Testing for BRAF Mutations in Cutaneous Melanoma Patients. Cancers. 2021; 13(9):2282. https://doi.org/10.3390/cancers13092282
Chicago/Turabian StyleZhou, Sarah, Daniel Sikorski, Honghao Xu, Andrei Zubarev, May Chergui, François Lagacé, Wilson H. Miller, Jr., Margaret Redpath, Stephanie Ghazal, Marcus O. Butler, and et al. 2021. "Defining the Criteria for Reflex Testing for BRAF Mutations in Cutaneous Melanoma Patients" Cancers 13, no. 9: 2282. https://doi.org/10.3390/cancers13092282
APA StyleZhou, S., Sikorski, D., Xu, H., Zubarev, A., Chergui, M., Lagacé, F., Miller, W. H., Jr., Redpath, M., Ghazal, S., Butler, M. O., Petrella, T. M., Claveau, J., Nessim, C., Salopek, T. G., Gniadecki, R., & Litvinov, I. V. (2021). Defining the Criteria for Reflex Testing for BRAF Mutations in Cutaneous Melanoma Patients. Cancers, 13(9), 2282. https://doi.org/10.3390/cancers13092282









