Small High-Risk Uveal Melanomas Have a Lower Mortality Rate
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
3. Discussion
4. Methods
4.1. Study Design
4.2. Clinical Methods
4.3. Tumor Sampling
4.4. Genetic Analysis
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Lead Time Bias Calculations with Range of Lead Time Estimates
| Relative Fatality Rate | Mixing Fraction | Large Tumours Metastases Detection Probability | Large Tumours Fatality Rate | True RR | Approximate HR | |
| 0.5 | 0.5 | 0.1333 | 0.6707 | 0.7761 | 0.6617 | |
| 0.6 | 0.5 | 0.1500 | 0.6363 | 0.7271 | 0.6141 | |
| 0.7 | 0.5 | 0.1647 | 0.6035 | 0.6981 | 0.5913 | |
| 0.4 | 0.5 | 0.1143 | 0.7061 | 0.8599 | 0.7631 | |
| 0.3 | 0.5 | 0.0923 | 0.7416 | 1.0138 | 1.0298 | |
| 0.5 | 0.6 | 0.1429 | 0.6294 | 0.7719 | 0.6701 | |
| 0.6 | 0.6 | 0.1579 | 0.6064 | 0.7249 | 0.6210 | |
| 0.7 | 0.6 | 0.1707 | 0.5831 | 0.6971 | 0.5962 | |
| 0.4 | 0.6 | 0.1250 | 0.6513 | 0.8524 | 0.7689 | |
| 0.3 | 0.6 | 0.1034 | 0.6708 | 1.0001 | 1.0001 | |
| 0.5 | 0.7 | 0.1538 | 0.5941 | 0.7593 | 0.6652 | |
| 0.6 | 0.7 | 0.1667 | 0.5798 | 0.7182 | 0.6211 | |
| 0.7 | 0.7 | 0.1772 | 0.5644 | 0.6938 | 0.5980 | |
| 0.4 | 0.7 | 0.1379 | 0.6064 | 0.8297 | 0.7502 | |
| 0.3 | 0.7 | 0.1176 | 0.6152 | 0.9589 | 0.9334 | |
| 0.5 | 0.4 | 0.1250 | 0.7193 | 0.7719 | 0.6377 | |
| 0.6 | 0.4 | 0.1429 | 0.6702 | 0.7249 | 0.5996 | |
| 0.7 | 0.4 | 0.1591 | 0.6258 | 0.6971 | 0.5830 | |
| 0.4 | 0.4 | 0.1053 | 0.7735 | 0.8524 | 0.7252 | |
| 0.3 | 0.4 | 0.0833 | 0.8328 | 1.0001 | 1.0002 | |
| 0.5 | 0.3 | 0.1176 | 0.7769 | 0.7593 | 0.5941 | |
| 0.6 | 0.3 | 0.1364 | 0.7087 | 0.7182 | 0.5767 | |
| 0.7 | 0.3 | 0.1538 | 0.6502 | 0.6938 | 0.5711 | |
| 0.4 | 0.3 | 0.0976 | 0.8573 | 0.8297 | 0.6382 | |
| 0.3 | 0.3 | 0.0759 | 0.9529 | 0.9589 | 0.8022 | |
| Parameter | Large | Small | RR | 95% CI or Range | HR | 95% CI or Range |
| No. of Cancers | 332 | 44 | ||||
| Number with fup > 5 years | 147 | 33 | ||||
| Number of deaths ≤ 5 years | 165 | 10 | ||||
| 5-year fatality rate | 0.52 | 0.23 | ||||
| 5-year survival | 0.48 | 0.77 | 0.45 | 0.26–0.8 | 0.44 | 0.3–0.58 |
| after Lead Time Bias correction | ||||||
| 5-year fatality rate | 0.52 | 0.35 | ||||
| 5-year survival | 0.48 | 0.65 | 0.69 | 0.45–1.05 | 0.67 | 0.53–0.81 |
| after Length Time Bias correction | ||||||
| 5-year fatality rate | 0.52 | 0.40 | ||||
| 5-year survival | 0.48 | 0.60 | 0.78 | 0.69–1.01 | 0.64 | 0.57–1.03 |
References
- De Potter, P.; Shields, C.L.; Shields, J.A.; Cater, J.R.; Tardio, D.J. Impact of enucleation versus plaque radiotherapy in the management of juxtapapillary choroidal melanoma on patient survival. Br. J. Ophthalmol. 1994, 78, 109–114. [Google Scholar] [CrossRef]
- Collaborative Ocular Melanoma Study Group. The COMS randomized trial of iodine 125 brachytherapy for choroidal melanoma: V. Twelve-year mortality rates and prognostic factors: COMS report No. 28. Arch. Ophthalmol. 2006, 124, 1684–1693. [Google Scholar] [CrossRef] [PubMed]
- Finger, P.T. Radiation therapy for choroidal melanoma. Surv. Ophthalmol. 1997, 42, 215–232. [Google Scholar] [CrossRef]
- Chang, M.Y.; McCannel, T.A. Local treatment failure after globe-conserving therapy for choroidal melanoma. Br. J. Ophthalmol. 2013, 97, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Damato, B.; Kacperek, A.; Chopra, M.; Campbell, I.R.; Errington, R.D. Proton beam radiotherapy of choroidal melanoma: The Liverpool-Clatterbridge experience. Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 1405–1411. [Google Scholar] [CrossRef]
- Russo, A.; Laguardia, M.; Damato, B. Eccentric ruthenium plaque radiotherapy of posterior choroidal melanoma. Graefes Arch. Clin. Exp. Ophthalmol. 2012, 250, 1533–1540. [Google Scholar] [CrossRef] [PubMed]
- Jmor, F.; Hussain, R.N.; Damato, B.E.; Heimann, H. Photodynamic therapy as initial treatment for small choroidal melanomas. Photodiagn. Photodyn. 2017, 20, 175–181. [Google Scholar] [CrossRef]
- Journee-de Korver, J.G.; Oosterhuis, J.A.; de Wolff-Rouendaal, D.; Kemme, H. Histopathological findings in human choroidal melanomas after transpupillary thermotherapy. Br. J. Ophthalmol. 1997, 81, 234–239. [Google Scholar] [CrossRef]
- Mashayekhi, A.; Shields, C.L.; Rishi, P.; Atalay, H.T.; Pellegrini, M.; McLaughlin, J.P.; Patrick, K.A.; Morton, S.J.; Remmer, M.H.; Parendo, A.; et al. Primary transpupillary thermotherapy for choroidal melanoma in 391 cases: Importance of risk factors in tumor control. Ophthalmology 2015, 122, 600–609. [Google Scholar] [CrossRef]
- Oosterhuis, J.A.; Journée-de Korver, H.G.; Keunen, J.E. Transpupillary thermotherapy: Results in 50 patients with choroidal melanoma. Arch. Ophthalmol. 1998, 116, 157–162. [Google Scholar] [CrossRef]
- Horgan, N.; Shields, C.L.; Mashayekhi, A.; Shields, J.A. Classification and treatment of radiation maculopathy. Curr. Opin. Ophthalmol. 2010, 21, 233–238. [Google Scholar] [CrossRef]
- Damato, B.E.; Dukes, J.; Goodall, H.; Carvajal, R.D. Tebentafusp: T Cell Redirection for the Treatment of Metastatic Uveal Melanoma. Cancers 2019, 11, 971. [Google Scholar] [CrossRef] [PubMed]
- Homsi, J.; Bedikian, A.Y.; Papadopoulos, N.E.; Kim, K.B.; Hwu, W.J.; Mahoney, S.L.; Hwu, P. Phase 2 open-label study of weekly docosahexaenoic acid-paclitaxel in patients with metastatic uveal melanoma. Melanoma Res. 2010, 20, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Schmittel, A.; Schmidt-Hieber, M.; Martus, P.; Bechrakis, N.E.; Schuster, R.; Siehl, J.M.; Foerster, M.H.; Thiel, E.; Keilholz, U. A randomized phase II trial of gemcitabine plus treosulfan versus treosulfan alone in patients with metastatic uveal melanoma. Ann. Oncol. 2006, 17, 1826–1829. [Google Scholar] [CrossRef] [PubMed]
- Spagnolo, F.; Grosso, M.; Picasso, V.; Tornari, E.; Pesce, M.; Queirolo, P. Treatment of metastatic uveal melanoma with intravenous fotemustine. Melanoma Res. 2013, 23, 196–198. [Google Scholar] [CrossRef]
- Spagnolo, F.; Caltabiano, G.; Queirolo, P. Uveal melanoma. Cancer Treat. Rev. 2012, 38, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, R.D.; Sosman, J.A.; Quevedo, J.F.; Milhem, M.M.; Joshua, A.M.; Kudchadkar, R.R.; Linette, G.P.; Gajewski, T.F.; Lutzky, J.; Lawson, D.H.; et al. Effect of selumetinib vs chemotherapy on progression-free survival in uveal melanoma: A randomized clinical trial. JAMA 2014, 311, 2397–2405. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Moon, J.; Margolin, K.A.; Weber, J.S.; Lao, C.D.; Othus, M.; Aparicio, A.M.; Ribas, A.; Sondak, V.K. Phase II trial of sorafenib in combination with carboplatin and paclitaxel in patients with metastatic uveal melanoma: SWOG S0512. PLoS ONE 2012, 7, e48787. [Google Scholar] [CrossRef]
- Gomez, D.; Wetherill, C.; Cheong, J.; Jones, L.; Marshall, E.; Damato, B.; Coupland, S.E.; Ghaneh, P.; Poston, G.J.; Malik, H.Z.; et al. The Liverpool uveal melanoma liver metastases pathway: Outcome following liver resection. J. Surg. Oncol. 2014, 109, 542–547. [Google Scholar] [CrossRef]
- Marshall, E.; Romaniuk, C.; Ghaneh, P.; Wong, H.; McKay, M.; Chopra, M.; Coupland, S.E.; Damato, B.E. MRI in the detection of hepatic metastases from high-risk uveal melanoma: A prospective study in 188 patients. Br. J. Ophthalmol. 2013, 97, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Karydis, I.; Chan, P.Y.; Wheater, M.; Arriola, E.; Szlosarek, P.W.; Ottensmeier, C.H. Clinical activity and safety of Pembrolizumab in Ipilimumab pre-treated patients with uveal melanoma. Oncoimmunology 2016, 5, e1143997. [Google Scholar] [CrossRef]
- Maio, M.; Danielli, R.; Chiarion-Sileni, V.; Pigozzo, J.; Parmiani, G.; Ridolfi, R.; De Rosa, F.; Del Vecchio, M.; Di Guardo, L.; Queirolo, P.; et al. Efficacy and safety of ipilimumab in patients with pre-treated, uveal melanoma. Ann. Oncol. 2013, 24, 2911–2915. [Google Scholar] [CrossRef] [PubMed]
- Luke, J.J.; Callahan, M.K.; Postow, M.A.; Romano, E.; Ramaiya, N.; Bluth, M.; Giobbie-Hurder, A.; Lawrence, D.P.; Ibrahim, N.; Ott, P.A.; et al. Clinical activity of ipilimumab for metastatic uveal melanoma: A retrospective review of the Dana-Farber Cancer Institute, Massachusetts General Hospital, Memorial Sloan-Kettering Cancer Center, and University Hospital of Lausanne experience. Cancer 2013, 119, 3687–3695. [Google Scholar] [CrossRef] [PubMed]
- Khattak, M.A.; Fisher, R.; Hughes, P.; Gore, M.; Larkin, J. Ipilimumab activity in advanced uveal melanoma. Melanoma Res. 2013, 23, 79–81. [Google Scholar] [CrossRef]
- Kelderman, S.; van der Kooij, M.K.; van den Eertwegh, A.J.; Soetekouw, P.M.; Jansen, R.L.; van den Brom, R.R.; Hospers, G.A.; Haanen, J.B.; Kapiteijn, E.; Blank, C.U. Ipilimumab in pretreated metastastic uveal melanoma patients. Results of the Dutch Working group on Immunotherapy of Oncology (WIN-O). Acta Oncol. 2013, 52, 1786–1788. [Google Scholar] [CrossRef] [PubMed]
- Sacco, J.J.; Kalirai, H.; Kenyani, J.; Figueiredo, C.R.; Coulson, J.M.; Coupland, S.E. Recent breakthroughs in metastatic uveal melanoma: A cause for optimism? Future Oncol. 2018, 14, 1335–1338. [Google Scholar] [CrossRef]
- Diener-West, M.; Hawkins, B.S.; Markowitz, J.A.; Schachat, A.P. A review of mortality from choroidal melanoma. II. A meta-analysis of 5-year mortality rates following enucleation, 1966 through 1988. Arch. Ophthalmol. 1992, 110, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Kujala, E.; Makitie, T.; Kivela, T. Very long-term prognosis of patients with malignant uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4651–4659. [Google Scholar] [CrossRef] [PubMed]
- Burr, J.M.; Mitry, E.; Rachet, B.; Coleman, M.P. Survival from uveal melanoma in England and Wales 1986 to 2001. Ophthalmic Epidemiol. 2007, 14, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Diener-West, M.; Reynolds, S.M.; Agugliaro, D.J.; Caldwell, R.; Cumming, K.; Earle, J.D.; Hawkins, B.S.; Hayman, J.A.; Jaiyesimi, I.; Jampol, L.M.; et al. Development of metastatic disease after enrollment in the COMS trials for treatment of choroidal melanoma: Collaborative Ocular Melanoma Study Group Report No. 26. Arch. Ophthalmol. 2005, 123, 1639–1643. [Google Scholar] [CrossRef]
- Rietschel, P.; Panageas, K.S.; Hanlon, C.; Patel, A.; Abramson, D.H.; Chapman, P.B. Variates of survival in metastatic uveal melanoma. J. Clin. Oncol. 2005, 23, 8076–8080. [Google Scholar] [CrossRef]
- Damato, B.; Duke, C.; Coupland, S.E.; Hiscott, P.; Smith, P.A.; Campbell, I.; Douglas, A.; Howard, P. Cytogenetics of uveal melanoma: A 7-year clinical experience. Ophthalmology 2007, 114, 1925–1931. [Google Scholar] [CrossRef]
- Eleuteri, A.; Taktak, A.F.G.; Coupland, S.E.; Heimann, H.; Kalirai, H.; Damato, B. Prognostication of metastatic death in uveal melanoma patients: A Markov multi-state model. Comput. Biol. Med. 2018, 102, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Kivela, T.; Kujala, E. Prognostication in eye cancer: The latest tumor, node, metastasis classification and beyond. Eye 2013, 27, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Royer-Bertrand, B.; Torsello, M.; Rimoldi, D.; El Zaoui, I.; Cisarova, K.; Pescini-Gobert, R.; Raynaud, F.; Zografos, L.; Schalenbourg, A.; Speiser, D.; et al. Comprehensive Genetic Landscape of Uveal Melanoma by Whole-Genome Sequencing. Am. J. Hum. Genet. 2016, 99, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Decatur, C.L.; Ong, E.; Garg, N.; Anbunathan, H.; Bowcock, A.M.; Field, M.G.; Harbour, J.W. Driver Mutations in Uveal Melanoma: Associations with Gene Expression Profile and Patient Outcomes. JAMA Ophthalmol. 2016, 134, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Landreville, S.; Agapova, O.A.; Harbour, J.W. Emerging insights into the molecular pathogenesis of uveal melanoma. Future Oncol. 2008, 4, 629–636. [Google Scholar] [CrossRef] [PubMed]
- White, V.A.; Chambers, J.D.; Courtright, P.D.; Chang, W.Y.; Horsman, D.E. Correlation of cytogenetic abnormalities with the outcome of patients with uveal melanoma. Cancer 1998, 83, 354–359. [Google Scholar] [CrossRef]
- Scholes, A.G.; Damato, B.E.; Nunn, J.; Hiscott, P.; Grierson, I.; Field, J.K. Monosomy 3 in uveal melanoma: Correlation with clinical and histologic predictors of survival. Invest. Ophthalmol. Vis. Sci. 2003, 44, 1008–1011. [Google Scholar] [CrossRef]
- Damato, B.; Dopierala, J.; Klaasen, A.; van Dijk, M.; Sibbring, J.; Coupland, S.E. Multiplex ligation-dependent probe amplification of uveal melanoma: Correlation with metastatic death. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3048–3055. [Google Scholar] [CrossRef]
- Shields, C.L.; Ganguly, A.; Bianciotto, C.G.; Turaka, K.; Tavallali, A.; Shields, J.A. Prognosis of uveal melanoma in 500 cases using genetic testing of fine-needle aspiration biopsy specimens. Ophthalmology 2011, 118, 396–401. [Google Scholar] [CrossRef]
- Rospond-Kubiak, I.; Wróblewska-Zierhoffer, M.; Twardosz-Pawlik, H.; Kociecki, J. The Liverpool Uveal Melanoma Prognosticator Online (LUMPO) for prognosing metastasis free survival in the absence of cytogenetic data after ruthenium brachytherapy for uveal melanoma. Acta Ophthalmol. 2015, 93. [Google Scholar] [CrossRef]
- DeParis, S.W.; Taktak, A.; Eleuteri, A.; Enanoria, W.; Heimann, H.; Coupland, S.E.; Damato, B. External Validation of the Liverpool Uveal Melanoma Prognosticator Online. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6116–6122. [Google Scholar] [CrossRef] [PubMed]
- Rola, A.C.; Taktak, A.; Eleuteri, A.; Kalirai, H.; Heimann, H.; Hussain, R.; Bonnett, L.J.; Hill, C.J.; Traynor, M.; Jager, M.J.; et al. Multicenter External Validation of the Liverpool Uveal Melanoma Prognosticator Online: An OOG Collaborative Study. Cancers 2020, 12, 477. [Google Scholar] [CrossRef]
- Damato, E.M.; Damato, B.E. Detection and time to treatment of uveal melanoma in the United Kingdom: An evaluation of 2384 patients. Ophthalmology 2012, 119, 1582–1589. [Google Scholar] [CrossRef] [PubMed]
- Damato, B. Time to treatment of uveal melanoma in the United Kingdom. Eye 2001, 15, 155–158. [Google Scholar] [CrossRef]
- Shields, C.L.; Furuta, M.; Thangappan, A.; Nagori, S.; Mashayekhi, A.; Lally, D.R.; Kelly, C.C.; Rudich, D.S.; Nagori, A.V.; Wakade, O.A.; et al. Metastasis of uveal melanoma millimeter-by-millimeter in 8033 consecutive eyes. Arch. Ophthalmol. 2009, 127, 989–998. [Google Scholar] [CrossRef]
- Shields, C.L.; Sioufi, K.; Srinivasan, A.; Di Nicola, M.; Masoomian, B.; Barna, L.E.; Bekerman, V.P.; Say, E.A.T.; Mashayekhi, A.; Emrich, J.; et al. Visual Outcome and Millimeter Incremental Risk of Metastasis in 1780 Patients with Small Choroidal Melanoma Managed by Plaque Radiotherapy. JAMA Ophthalmol. 2018, 136, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Straatsma, B.R.; Diener-West, M.; Caldwell, R.; Engstrom, R.E.; Collaborative Ocular Melanoma Study, G. Mortality after deferral of treatment or no treatment for choroidal melanoma. Am. J. Ophthalmol. 2003, 136, 47–54. [Google Scholar] [CrossRef]
- Damato, B. Ocular treatment of choroidal melanoma in relation to the prevention of metastatic death—A personal view. Prog. Retin. Eye Res. 2018, 66, 187–199. [Google Scholar] [CrossRef]
- Damato, B. Choroidal melanoma endoresection, dandelions and allegory-based medicine. Br. J. Ophthalmol. 2008, 92, 1013–1014. [Google Scholar] [CrossRef] [PubMed]
- Hussain, R.; Czanner, G.; Taktak, A.; Damato, B.; Praidou, A.; Heimann, H. Mortality of Patients with Uveal Melanoma Detected by Diabetic Retinopathy Screening. Retina 2020, 40, 2198–2206. [Google Scholar] [CrossRef] [PubMed]
- Damato, B.E.; Heimann, H.; Kalirai, H.; Coupland, S.E. Age, survival predictors, and metastatic death in patients with choroidal melanoma: Tentative evidence of a therapeutic effect on survival. JAMA Ophthalmol. 2014, 132, 605–613. [Google Scholar] [CrossRef]
- Shields, C.L.; Shields, J.A.; Kiratli, H.; De Potter, P.; Cater, J.R. Risk factors for growth and metastasis of small choroidal melanocytic lesions. Ophthalmology 1995, 102, 1351–1361. [Google Scholar] [CrossRef]
- Kujala, E.; Damato, B.; Coupland, S.E.; Desjardins, L.; Bechrakis, N.E.; Grange, J.D.; Kivela, T. Staging of ciliary body and choroidal melanomas based on anatomic extent. J. Clin. Oncol. 2013, 31, 2825–2831. [Google Scholar] [CrossRef] [PubMed]
- McLean, I.W.; Zimmerman, L.E.; Foster, W.D. Survival rates after enucleation of eyes with malignant melanoma. Am. J. Ophthalmol. 1979, 88, 794–797. [Google Scholar] [CrossRef]
- Zimmerman, L.E.; McLean, I.W.; Foster, W.D. Does enucleation of the eye containing a malignant melanoma prevent or accelerate the dissemination of tumour cells. Br. J. Ophthalmol. 1978, 62, 420–425. [Google Scholar] [CrossRef] [PubMed]
- COMS. The Collaborative Ocular Melanoma Study (COMS) randomized trial of pre-enucleation radiation of large choroidal melanoma III: Local complications and observations following enucleation COMS report no. 11. Am. J. Ophthalmol. 1998, 126, 362–372. [Google Scholar] [CrossRef]
- Diener-West, M.; Earle, J.D.; Fine, S.L.; Hawkins, B.S.; Moy, C.S.; Reynolds, S.M.; Schachat, A.P.; Straatsma, B.R.; Collaborative Ocular Melanoma Study Group. The COMS randomized trial of iodine 125 brachytherapy for choroidal melanoma, II: Characteristics of patients enrolled and not enrolled. COMS Report No. 17. Arch. Ophthalmol. 2001, 119, 951–965. [Google Scholar] [CrossRef]
- Zimmerman, L.E.; McLean, I.W. Do growth and onset of symptoms of uveal melanomas indicate subclinical metastasis? Ophthalmology 1984, 91, 685–691. [Google Scholar] [CrossRef]
- Singh, A.D. Uveal melanoma: Implications of tumor doubling time. Ophthalmology 2001, 108, 829–831. [Google Scholar] [CrossRef]
- Eskelin, S.; Pyrhonen, S.; Summanen, P.; Hahka-Kemppinen, M.; Kivela, T. Tumor doubling times in metastatic malignant melanoma of the uvea: Tumor progression before and after treatment. Ophthalmology 2000, 107, 1443–1449. [Google Scholar] [CrossRef]
- Tura, A.; Merz, H.; Reinsberg, M.; Luke, M.; Jager, M.J.; Grisanti, S.; Luke, J. Analysis of monosomy-3 in immunomagnetically isolated circulating melanoma cells in uveal melanoma patients. Pigment. Cell Melanoma Res. 2016, 29, 583–589. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, J. Invited commentary. Uveal melanoma: Implications of tumor doubling time. Ophthalmology 2001, 108, 831–832. [Google Scholar] [CrossRef]
- Jouhi, S.; Jager, M.J.; de Geus, S.J.R.; Desjardins, L.; Eide, N.A.; Grange, J.D.; Kiilgaard, J.F.; Seregard, S.; Midena, E.; Parrozzani, R.; et al. The Small Fatal Choroidal Melanoma Study. A Survey by the European Ophthalmic Oncology Group. Am. J. Ophthalmol. 2019, 202, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Boldt, H.C.; Binkley, E. Treating Small Choroidal Melanoma: Smaller Is Better. JAMA Ophthalmol. 2018, 136, 1333–1334. [Google Scholar] [CrossRef] [PubMed]
- Dopierala, J.; Damato, B.E.; Lake, S.L.; Taktak, A.F.; Coupland, S.E. Genetic heterogeneity in uveal melanoma assessed by multiplex ligation-dependent probe amplification. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4898–4905. [Google Scholar] [CrossRef] [PubMed]
- Murray, T.G.; Sobrin, L. The case for observational management of suspected small choroidal melanoma. Arch. Ophthalmol. 2006, 124, 1342–1344. [Google Scholar] [CrossRef]
- Curtin, V.T. Natural course of small malignant melanomas of choroid and ciliary body. Trans. Ophthalmol. Soc. NZ 1978, 30, 61. [Google Scholar]
- Char, D.H. The management of small choroidal melanomas. Surv. Ophthalmol. 1978, 22, 377–386. [Google Scholar] [CrossRef]
- Lane, A.M.; Egan, K.M.; Kim, I.K.; Gragoudas, E.S. Mortality after diagnosis of small melanocytic lesions of the choroid. Arch. Ophthalmol. 2010, 128, 996–1000. [Google Scholar] [CrossRef]
- Butler, P.; Char, D.H.; Zarbin, M.; Kroll, S. Natural history of indeterminate pigmented choroidal tumors. Ophthalmology 1994, 101, 710–716, discussion 717. [Google Scholar] [CrossRef]
- Weis, E.; Roelofs, K.; Larocque, M.; Murtha, A. Gene Expression Profiling as an Adjunctive Measure to Guide the Management of Indeterminate, High-Risk Choroidal Melanocytic Lesions: A Pilot Study. Ocul. Oncol. Pathol. 2019, 5, 102–109. [Google Scholar] [CrossRef]
- Miller, A.K.; Benage, M.J.; Wilson, D.J.; Skalet, A.H. Uveal Melanoma with Histopathologic Intratumoral Heterogeneity Associated with Gene Expression Profile Discordance. Ocul. Oncol. Pathol. 2017, 3, 156–160. [Google Scholar] [CrossRef]
- Callejo, S.A.; Dopierala, J.; Coupland, S.E.; Damato, B. Sudden growth of a choroidal melanoma and multiplex ligation-dependent probe amplification findings suggesting late transformation to monosomy 3 type. Arch. Ophthalmol. 2011, 129, 958–960. [Google Scholar] [CrossRef]
- Field, M.G.; Durante, M.A.; Anbunathan, H.; Cai, L.Z.; Decatur, C.L.; Bowcock, A.M.; Kurtenbach, S.; Harbour, J.W. Punctuated evolution of canonical genomic aberrations in uveal melanoma. Nat. Commun. 2018, 9, 116. [Google Scholar] [CrossRef]
- White, V.A.; McNeil, B.K.; Thiberville, L.; Horsman, D.E. Acquired homozygosity (isodisomy) of chromosome 3 during clonal evolution of a uveal melanoma: Association with morphologic heterogeneity. Genes Chromosomes Cancer 1996, 15, 138–143. [Google Scholar] [CrossRef]
- Afshar, A.R.; Deiner, M.; Allen, G.; Damato, B.E. The Patient’s Experience of Ocular Melanoma in the US: A Survey of the Ocular Melanoma Foundation. Ocul. Oncol. Pathol. 2018, 4, 280–290. [Google Scholar] [CrossRef]
- Hopkins, J.; Keane, P.A.; Balaskas, K. Delivering personalized medicine in retinal care: From artificial intelligence algorithms to clinical application. Curr. Opin. Ophthalmol. 2020, 31, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Hussain, R.N.; Kalirai, H.; Groenewald, C.; Kacperek, A.; Errington, R.D.; Coupland, S.E.; Heimann, H.; Damato, B. Prognostic Biopsy of Choroidal Melanoma after Proton Beam Radiation Therapy. Ophthalmology 2016, 123, 2264–2265. [Google Scholar] [CrossRef] [PubMed]
- Angi, M.; Kalirai, H.; Taktak, A.; Hussain, R.; Groenewald, C.; Damato, B.E.; Heimann, H.; Coupland, S.E. Prognostic biopsy of choroidal melanoma: An optimised surgical and laboratory approach. Br. J. Ophthalmol. 2017, 101, 1143–1146. [Google Scholar] [CrossRef]
- Duffy, S.W.; Nagtegaal, I.D.; Wallis, M.; Cafferty, F.H.; Houssami, N.; Warwick, J.; Allgood, P.C.; Kearins, O.; Tappenden, N.; O’Sullivan, E.; et al. Correcting for lead time and length bias in estimating the effect of screen detection on cancer survival. Am. J. Epidemiol. 2008, 168, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Damato, B.; Eleuteri, A.; Hussain, R.; Kalirai, H.; Thornton, S.; Taktak, A.; Heimann, H.; Coupland, S.E. Parsimonious Models for Predicting Mortality from Choroidal Melanoma. Investig. Ophthalmol. Vis. Sci. 2020, 61, 35. [Google Scholar] [CrossRef]
- Parrella, P.; Sidransky, D.; Merbs, S.L. Allelotype of posterior uveal melanoma: Implications for a bifurcated tumor progression pathway. Cancer Res. 1999, 59, 3032–3037. [Google Scholar]
- Field, M.G.; Harbour, J.W. Recent developments in prognostic and predictive testing in uveal melanoma. Curr. Opin. Ophthalmol. 2014, 25, 234–239. [Google Scholar] [CrossRef]
- Onken, M.D.; Worley, L.A.; Ehlers, J.P.; Harbour, J.W. Gene expression profiling in uveal melanoma reveals two molecular classes and predicts metastatic death. Cancer Res. 2004, 64, 7205–7209. [Google Scholar] [CrossRef] [PubMed]
- Onken, M.D.; Ehlers, J.P.; Worley, L.A.; Makita, J.; Yokota, Y.; Harbour, J.W. Functional gene expression analysis uncovers phenotypic switch in aggressive uveal melanomas. Cancer Res. 2006, 66, 4602–4609. [Google Scholar] [CrossRef] [PubMed]
- Harbour, J.W. The genetics of uveal melanoma: An emerging framework for targeted therapy. Pigment. Cell Melanoma Res. 2012, 25, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Harbour, J.W.; Onken, M.D.; Roberson, E.D.; Duan, S.; Cao, L.; Worley, L.A.; Council, M.L.; Matatall, K.A.; Helms, C.; Bowcock, A.M. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 2010, 330, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.G.; Shih, J.; Yau, C.; Gibb, E.A.; Oba, J.; Mungall, K.L.; Hess, J.M.; Uzunangelov, V.; Walter, V.; Danilova, L.; et al. Integrative Analysis Identifies Four Molecular and Clinical Subsets in Uveal Melanoma. Cancer Cell 2017, 32, 204–220.e215. [Google Scholar] [CrossRef]
- Smit, K.N.; van Poppelen, N.M.; Vaarwater, J.; Verdijk, R.; van Marion, R.; Kalirai, H.; Coupland, S.E.; Thornton, S.; Farquhar, N.; Dubbink, H.J.; et al. Combined mutation and copy-number variation detection by targeted next-generation sequencing in uveal melanoma. Mod. Pathol. 2018, 31, 763–771. [Google Scholar] [CrossRef]
- Thornton, S.; Coupland, S.E.; Olohan, L.; Sibbring, J.S.; Kenny, J.G.; Hertz-Fowler, C.; Liu, X.; Haldenby, S.; Heimann, H.; Hussain, R.; et al. Targeted Next-Generation Sequencing of 117 Routine Clinical Samples Provides Further Insights into the Molecular Landscape of Uveal Melanoma. Cancers 2020, 12, 1039. [Google Scholar] [CrossRef] [PubMed]
- Grixti, A.; Angi, M.; Damato, B.E.; Jmor, F.; Konstantinidis, L.; Groenewald, C.; Heimann, H. Vitreoretinal surgery for complications of choroidal tumor biopsy. Ophthalmology 2014, 121, 2482–2488. [Google Scholar] [CrossRef] [PubMed]
- Sen, J.; Groenewald, C.; Hiscott, P.S.; Smith, P.A.; Damato, B.E. Transretinal choroidal tumor biopsy with a 25-gauge vitrector. Ophthalmology 2006, 113, 1028–1031. [Google Scholar] [CrossRef] [PubMed]
- Coupland, S.E.; Kalirai, H.; Ho, V.; Thornton, S.; Damato, B.E.; Heimann, H. Concordant chromosome 3 results in paired choroidal melanoma biopsies and subsequent tumour resection specimens. Br. J. Ophthalmol. 2015, 99, 1444–1450. [Google Scholar] [CrossRef] [PubMed]
- Thornton, S.; Coupland, S.E.; Heimann, H.; Hussain, R.; Groenewald, C.; Kacperek, A.; Damato, B.; Taktak, A.; Eleuteri, A.; Kalirai, H. Effects of plaque brachytherapy and proton beam radiotherapy on prognostic testing: A comparison of uveal melanoma genotyped by microsatellite analysis. Br. J. Ophthalmol. 2020, 104, 1462–1466. [Google Scholar] [CrossRef]
- Tschentscher, F.; Prescher, G.; Zeschnigk, M.; Horsthemke, B.; Lohmann, D.R. Identification of chromosomes 3, 6, and 8 aberrations in uveal melanoma by microsatellite analysis in comparison to comparative genomic hybridization. Cancer Genet. Cytogenet. 2000, 122, 13–17. [Google Scholar] [CrossRef]
- Thomas, S.; Pütter, C.; Weber, S.; Bornfeld, N.; Lohmann, D.R.; Zeschnigk, M. Prognostic significance of chromosome 3 alterations determined by microsatellite analysis in uveal melanoma: A long-term follow-up study. Br. J. Cancer 2012, 106, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Lake, S.L.; Kalirai, H.; Dopierala, J.; Damato, B.E.; Coupland, S.E. Comparison of formalin-fixed and snap-frozen samples analyzed by multiplex ligation-dependent probe amplification for prognostic testing in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2647–2652. [Google Scholar] [CrossRef]
- Gray, R.J. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann. Stat. 1988, 16, 1141–1154. [Google Scholar] [CrossRef]
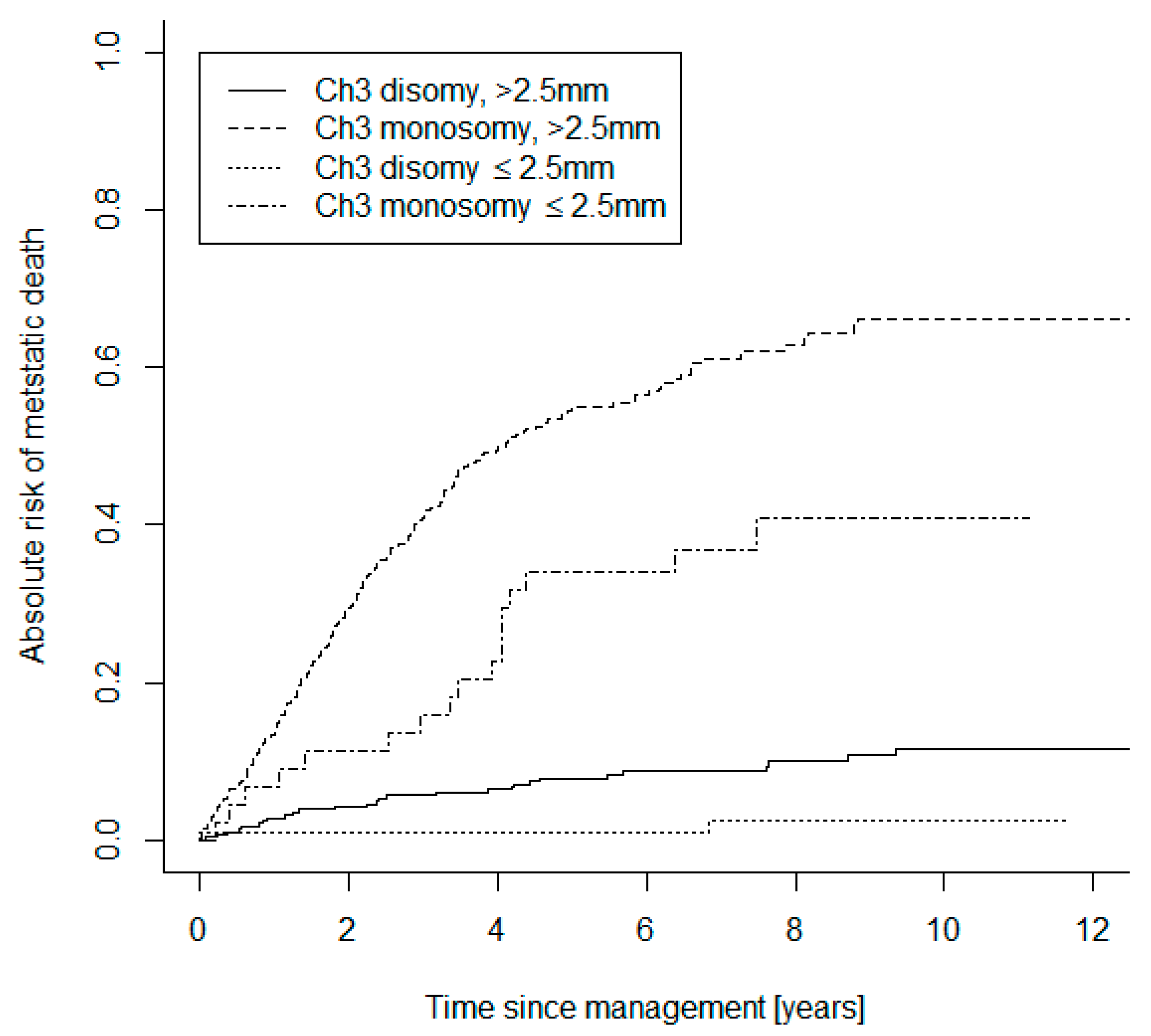
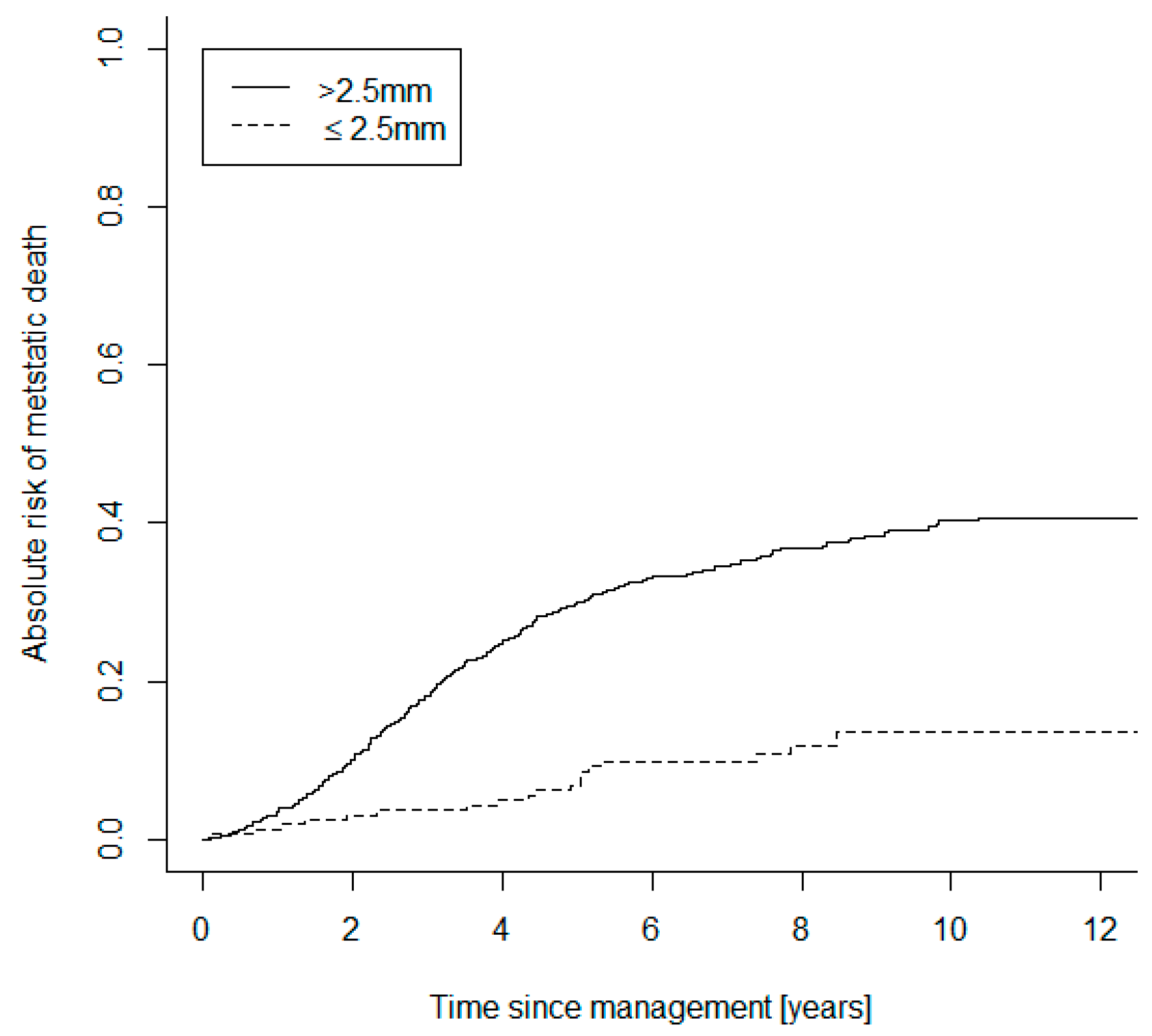
| Baseline Characteristics and Treatments | Small UM Group (n = 196) | Large UM Group (n = 744) | |
|---|---|---|---|
| Baseline characteristics | Age at diagnosis (years) | 59 (22–89) | 62 (21–94) |
| Longitudinal base diameter (mm) | 8.5 (1.8–19.8) | 14.2 (2.3–26) | |
| Tumor thickness (mm) | 1.8 (0.7–2.5) | 6.8 (2.6–18.3) | |
| Ciliary body involvement | 17 (8.7%) | 169 (22.7%) | |
| Primary treatment | Ruthenium-106 plaque brachytherapy | 77 (39.2%) | 145 (19.5%) |
| Proton beam radiotherapy | 65 (33.2%) | 132 (17.7%) | |
| Trans scleral tumor resection | 8 (4.1%) | 60 (8.1%) | |
| Endoresection | 9 (4.6%) | 33 (4.4%) | |
| Primary enucleation | 35 (17.9%) | 372 (50.0%) | |
| - | PDT | 1 (0.5%) | 1 (0.1%) |
| Risk Ratio and Hazard Rates | RR (95% CI) | HR (95% CI) |
|---|---|---|
| Uncorrected | 0.45 (0.26–0.80) | 0.44 (0.30–0.58) |
| After lead time bias correction | 0.69 (0.45–1.05) | 0.67 (0.53–0.81) |
| After length time bias correction after 5 years | 0.78 (0.69–1.01) | 0.64 (0.5–1.03) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, R.N.; Coupland, S.E.; Kalirai, H.; Taktak, A.F.G.; Eleuteri, A.; Damato, B.E.; Groenewald, C.; Heimann, H. Small High-Risk Uveal Melanomas Have a Lower Mortality Rate. Cancers 2021, 13, 2267. https://doi.org/10.3390/cancers13092267
Hussain RN, Coupland SE, Kalirai H, Taktak AFG, Eleuteri A, Damato BE, Groenewald C, Heimann H. Small High-Risk Uveal Melanomas Have a Lower Mortality Rate. Cancers. 2021; 13(9):2267. https://doi.org/10.3390/cancers13092267
Chicago/Turabian StyleHussain, Rumana N., Sarah E. Coupland, Helen Kalirai, Azzam F. G. Taktak, Antonio Eleuteri, Bertil E. Damato, Carl Groenewald, and Heinrich Heimann. 2021. "Small High-Risk Uveal Melanomas Have a Lower Mortality Rate" Cancers 13, no. 9: 2267. https://doi.org/10.3390/cancers13092267
APA StyleHussain, R. N., Coupland, S. E., Kalirai, H., Taktak, A. F. G., Eleuteri, A., Damato, B. E., Groenewald, C., & Heimann, H. (2021). Small High-Risk Uveal Melanomas Have a Lower Mortality Rate. Cancers, 13(9), 2267. https://doi.org/10.3390/cancers13092267







