Hodgkin Lymphoma in People Living with HIV
Abstract
Simple Summary
Abstract
1. Introduction
2. Epidemiology
3. Etiopathogenesis
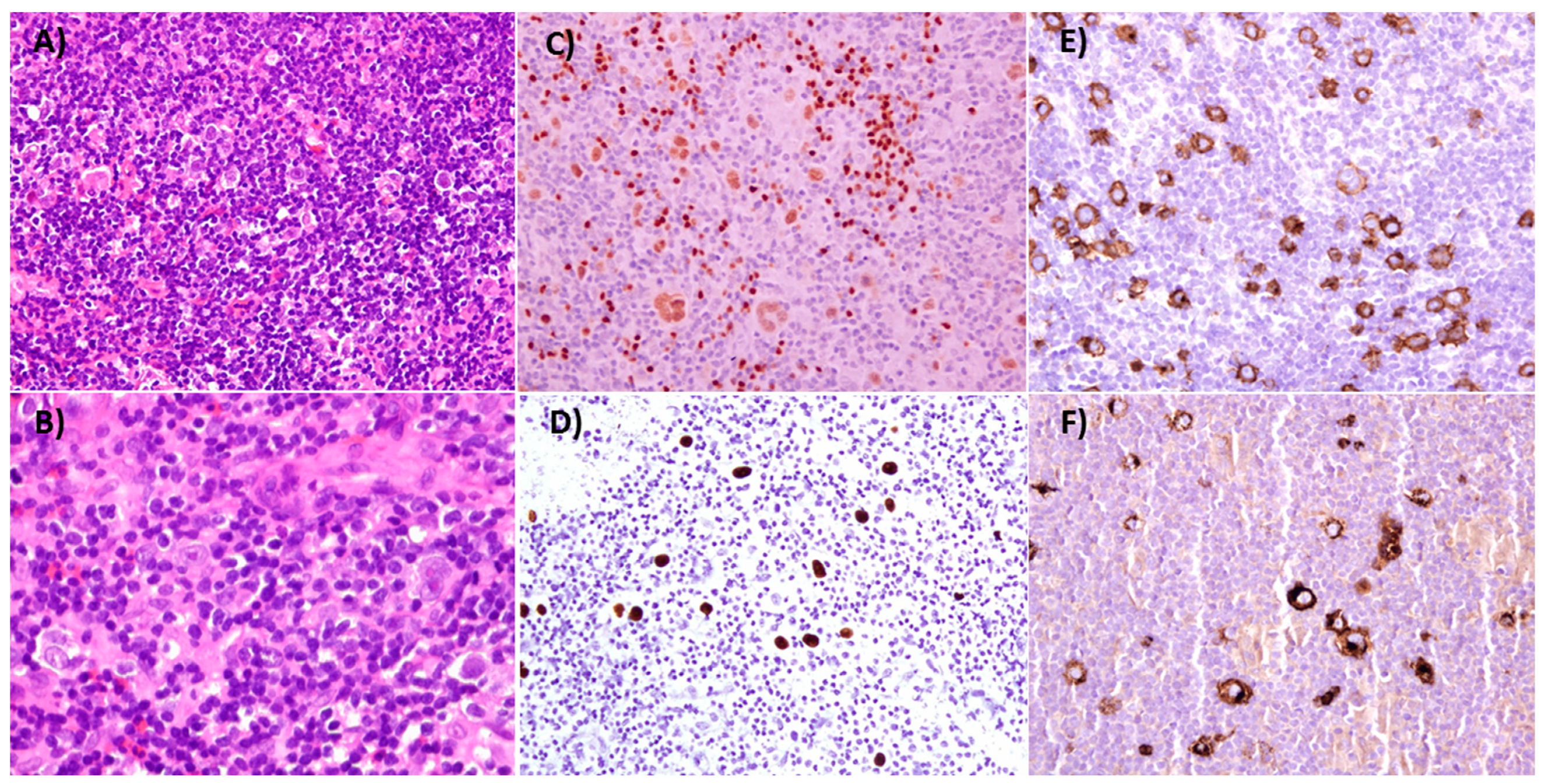
4. Pathological and Clinical Characteristics
5. Treatment and Prognosis
Additional Measures and Supportive Care
6. Management of cART in Patients with Classical Hodgkin Lymphoma
6.1. Initiation/Maintenance of cART
6.2. Drug Interactions between cART and Chemotherapy
6.3. Clinical Approach to Management of Patients on cART and Hodgkin Lymphoma
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shiels, M.S.; Pfeiffer, R.M.; Hall, H.I.; Li, J.; Goedert, J.J.; Morton, L.M.; Hartge, P.; Engels, E.A. Proportions of Kaposi Sarcoma, Selected Non-Hodgkin Lymphomas, and Cervical Cancer in the United States Occurring in Persons with AIDS, 1980–2007. JAMA 2011, 305, 1450–1459. [Google Scholar] [CrossRef]
- Kimani, S.M.; Painschab, M.S.; Horner, M.-J.; Muchengeti, M.; Fedoriw, Y.; Shiels, M.S.; Gopal, S. Epidemiology of Haematological Malignancies in People Living with HIV. Lancet HIV 2020, 7, e641–e651. [Google Scholar] [CrossRef]
- Horner, M.-J.; Shiels, M.S.; Pfeiffer, R.M.; Engels, E.A. Deaths Attributable to Cancer in the US Human Immunodeficiency Virus Population During 2001-2015. Clin. Infect. Dis. 2021, 72, e224–e231. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ramírez, R.U.; Shiels, M.S.; Dubrow, R.; Engels, E.A. Cancer Risk in HIV-Infected People in the USA from 1996 to 2012: A Population-Based, Registry-Linkage Study. Lancet HIV 2017, 4, e495–e504. [Google Scholar] [CrossRef]
- Robbins, H.A.; Shiels, M.S.; Pfeiffer, R.M.; Engels, E.A. Epidemiologic Contributions to Recent Cancer Trends among HIV-Infected People in the United States. AIDS 2014, 28, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Xicoy, B.; Ribera, J.-M.; Miralles, P.; Berenguer, J.; Rubio, R.; Mahillo, B.; Valencia, M.-E.; Abella, E.; López-Guillermo, A.; Sureda, A.; et al. Results of Treatment with Doxorubicin, Bleomycin, Vinblastine and Dacarbazine and Highly Active Antiretroviral Therapy in Advanced Stage, Human Immunodeficiency Virus-Related Hodgkin’s Lymphoma. Haematologica 2007, 92, 191–198. [Google Scholar] [CrossRef]
- Díez-Martín, J.L.; Balsalobre, P.; Re, A.; Michieli, M.; Ribera, J.M.; Canals, C.; Conde, E.; Rosselet, A.; Gabriel, I.; Varela, R.; et al. Comparable Survival between HIV+ and HIV- Non-Hodgkin and Hodgkin Lymphoma Patients Undergoing Autologous Peripheral Blood Stem Cell Transplantation. Blood 2009, 113, 6011–6014. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Parsons, C.; Cole, J. Characterization of HIV-Associated Hodgkin’s Lymphoma in HIV-Infected Patients: A Single-Center Experience. J. Int. Assoc. Physicians AIDS Care 2012, 11, 234–238. [Google Scholar] [CrossRef]
- Sorigué, M.; García, O.; Tapia, G.; Baptista, M.-J.; Moreno, M.; Mate, J.-L.; Sancho, J.M.; Feliu, E.; Ribera, J.-M.; Navarro, J.-T. HIV-Infection Has No Prognostic Impact on Advanced-Stage Hodgkin Lymphoma. AIDS 2017, 31, 1445–1449. [Google Scholar] [CrossRef]
- Mani, H.; Jaffe, E.S. Hodgkin Lymphoma: An Update on Its Biology with New Insights into Classification. Clin. Lymphoma Myeloma 2009, 9, 206–216. [Google Scholar] [CrossRef]
- Carbone, A.; Gloghini, A.; Caruso, A.; Paoli, P.D.; Dolcetti, R. The Impact of EBV and HIV Infection on the Microenvironmental Niche Underlying Hodgkin Lymphoma Pathogenesis. Int. J. Cancer 2017, 140, 1233–1245. [Google Scholar] [CrossRef]
- De Paoli, P.; Carbone, A. Microenvironmental Abnormalities Induced by Viral Cooperation: Impact on Lymphomagenesis. Semin. Cancer Biol. 2015, 34, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Chetaille, B.; Bertucci, F.; Finetti, P.; Esterni, B.; Stamatoullas, A.; Picquenot, J.M.; Copin, M.C.; Morschhauser, F.; Casasnovas, O.; Petrella, T.; et al. Molecular Profiling of Classical Hodgkin Lymphoma Tissues Uncovers Variations in the Tumor Microenvironment and Correlations with EBV Infection and Outcome. Blood 2009, 113, 2765–3775. [Google Scholar] [CrossRef]
- Montoto, S.; Shaw, K.; Okosun, J.; Gandhi, S.; Fields, P.; Wilson, A.; Shanyinde, M.; Cwynarski, K.; Marcus, R.; de Vos, J.; et al. HIV Status Does Not Influence Outcome in Patients with Classical Hodgkin Lymphoma Treated with Chemotherapy Using Doxorubicin, Bleomycin, Vinblastine, and Dacarbazine in the Highly Active Antiretroviral Therapy Era. J. Clin. Oncol. 2012, 30, 4111–4116. [Google Scholar] [CrossRef] [PubMed]
- Bower, M.; Palfreeman, A.; Alfa-Wali, M.; Bunker, C.; Burns, F.; Churchill, D.; Collins, S.; Cwynarski, K.; Edwards, S.; Fields, P.; et al. British HIV Association Guidelines for HIV-Associated Malignancies 2014. HIV Med. 2014, 15 (Suppl. S2), 1–92. [Google Scholar] [CrossRef] [PubMed]
- Hentrich, M.; Hoffmann, C.; Mosthaf, F.; Müller, M.; Siehl, J.; Wyen, C.; Hensel, M.; German Study Group of Physicians in Private Practice Treating HIV-Infected Patients (DAGNÄ); German AIDS Society (DAIG). Therapy of HIV-Associated Lymphoma-Recommendations of the Oncology Working Group of the German Study Group of Physicians in Private Practice Treating HIV-Infected Patients (DAGNÄ), in Cooperation with the German AIDS Society (DAIG). Ann. Hematol. 2014, 93, 913–921. [Google Scholar] [CrossRef]
- Miralles, P.; Navarro, J.T.; Berenguer, J.; Gómez Codina, J.; Kwon, M.; Serrano, D.; Díez-Martín, J.L.; Villà, S.; Rubio, R.; Menárguez, J.; et al. GESIDA/PETHEMA Recommendations on the Diagnosis and Treatment of Lymphomas in Patients Infected by the Human Immunodeficiency Virus. Med. Clin. 2018, 151, 39.e1–39.e17. [Google Scholar] [CrossRef]
- Bohlius, J.; Schmidlin, K.; Boué, F.; Fätkenheuer, G.; May, M.; Caro-Murillo, A.M.; Mocroft, A.; Bonnet, F.; Clifford, G.; Paparizos, V.; et al. HIV-1–Related Hodgkin Lymphoma in the Era of Combination Antiretroviral Therapy: Incidence and Evolution of CD4+ T-Cell Lymphocytes. Blood 2011, 117, 6100–6108. [Google Scholar] [CrossRef]
- Besson, C.; Lancar, R.; Prevot, S.; Brice, P.; Meyohas, M.-C.; Marchou, B.; Gabarre, J.; Bonnet, F.; Goujard, C.; Lambotte, O.; et al. High Risk Features Contrast With Favorable Outcomes in HIV-Associated Hodgkin Lymphoma in the Modern CART Era, ANRS CO16 LYMPHOVIR Cohort. Clin. Infect. Dis. 2015, 61, 1469–1475. [Google Scholar] [CrossRef]
- Calabresi, A.; Ferraresi, A.; Festa, A.; Scarcella, C.; Donato, F.; Vassallo, F.; Limina, R.; Castelli, F.; Quiros-Roldan, E.; Brescia HIV Cancer Study Group. Incidence of AIDS-Defining Cancers and Virus-Related and Non-Virus-Related Non-AIDS-Defining Cancers among HIV-Infected Patients Compared with the General Population in a Large Health District of Northern Italy, 1999–2009. HIV Med. 2013, 14, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Hleyhel, M.; Hleyhel, M.; Bouvier, A.M.; Belot, A.; Tattevin, P.; Pacanowski, J.; Genet, P.; De Castro, N.; Berger, J.-L.; Dupont, C.; et al. Risk of Non-AIDS-Defining Cancers among HIV-1-Infected Individuals in France between 1997 and 2009: Results from a French Cohort. AIDS 2014, 28, 2109–2118. [Google Scholar] [CrossRef] [PubMed]
- Re, A.; Cattaneo, C.; Rossi, G. Hiv and Lymphoma: From Epidemiology to Clinical Management. Mediterr. J. Hematol. Infect. Dis. 2019, 11, e2019004. [Google Scholar] [CrossRef] [PubMed]
- Biggar, R.J.; Jaffe, E.S.; Goedert, J.J.; Chaturvedi, A.; Pfeiffer, R.; Engels, E.A. Hodgkin Lymphoma and Immunodeficiency in Persons with HIV/AIDS. Blood 2006, 108, 3786–3791. [Google Scholar] [CrossRef]
- Bräuninger, A.; Schmitz, R.; Bechtel, D.; Renné, C.; Hansmann, M.-L.; Küppers, R. Molecular Biology of Hodgkin’s and Reed/Sternberg Cells in Hodgkin’s Lymphoma. Int. J. Cancer 2006, 118, 1853–1861. [Google Scholar] [CrossRef] [PubMed]
- van den Berg, A.; Visser, L.; Poppema, S. High Expression of the CC Chemokine TARC in Reed-Sternberg Cells: A Possible Explanation for the Characteristic T-Cell Infiltrate in Hodgkin’s Lymphoma. Am. J. Pathol. 1999, 154, 1685–1691. [Google Scholar] [CrossRef]
- Skinnider, B.F.; Mak, T.W. The Role of Cytokines in Classical Hodgkin Lymphoma. Blood 2002, 99, 4283–4297. [Google Scholar] [CrossRef]
- Gotti, D.; Danesi, M.; Calabresi, A.; Ferraresi, A.; Albini, L.; Donato, F.; Castelli, F.; Scalzini, A.; Quiros-Roldan, E.; Brescia HIV Cancer Study Group. Clinical Characteristics, Incidence, and Risk Factors of HIV-Related Hodgkin Lymphoma in the Era of Combination Antiretroviral Therapy. AIDS Patient Care STDs 2013, 27, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Lanoy, E.; Rosenberg, P.S.; Fily, F.; Lascaux, A.-S.; Martinez, V.; Partisani, M.; Poizot-Martin, I.; Rouveix, E.; Engels, E.A.; Costagliola, D.; et al. HIV-Associated Hodgkin Lymphoma during the First Months on Combination Antiretroviral Therapy. Blood 2011, 118, 44–49. [Google Scholar] [CrossRef]
- Kuppers, R.; Rajewsky, K.; Zhao, M.; Simons, G.; Laumann, R.; Fischer, R.; Hansmann, M.L. Hodgkin Disease: Hodgkin and Reed-Sternberg Cells Picked from Histological Sections Show Clonal Immunoglobulin Gene Rearrangements and Appear to Be Derived from B Cells at Various Stages of Development. Proc. Natl. Acad. Sci. USA 1994, 91, 10962–10966. [Google Scholar] [CrossRef]
- Weniger, M.A.; Küppers, R. Molecular Biology of Hodgkin Lymphoma. Leukemia 2021, 35, 968–981. [Google Scholar] [CrossRef]
- Braeuninger, A.; Küppers, R.; Strickler, J.G.; Wacker, H.-H.; Rajewsky, K.; Hansmann, M.-L. Hodgkin and Reed–Sternberg Cells in Lymphocyte Predominant Hodgkin Disease Represent Clonal Populations of Germinal Center-Derived Tumor B Cells. Proc. Natl. Acad. Sci. USA 1997, 94, 9337–9342. [Google Scholar] [CrossRef]
- Audouin, J.; Diebold, J.; Pallesen, G. Frequent Expression of Epstein-Barr Virus Latent Membrane Protein-1 in Tumour Cells of Hodgkin’s Disease in HIV-Positive Patients. J. Pathol. 1992, 167, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Carbone, A.; Gloghini, A.; Larocca, L.M.; Antinori, A.; Falini, B.; Tirelli, U.; Dalla-Favera, R.; Gaidano, G. Human Immunodeficiency Virus–Associated Hodgkin’s Disease Derives From Post–Germinal Center B Cells. Blood 1999, 93, 2319–2326. [Google Scholar] [CrossRef]
- Linke-Serinsöz, E.; Fend, F.; Quintanilla-Martinez, L. Human Immunodeficiency Virus (HIV) and Epstein-Barr Virus (EBV) Related Lymphomas, Pathology View Point. Semin. Diagn. Pathol. 2017, 34, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Deacon, E.M.; Pallesen, G.; Niedobitek, G.; Crocker, J.; Brooks, L.; Rickinson, A.B.; Young, L.S. Epstein-Barr Virus and Hodgkin’s Disease: Transcriptional Analysis of Virus Latency in the Malignant Cells. J. Exp. Med. 1993, 177, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Niedobitek, G.; Kremmer, E.; Herbst, H.; Whitehead, L.; Dawson, C.W.; Niedobitek, E.; von Ostau, C.; Rooney, N.; Grässer, F.A.; Young, L.S. Immunohistochemical Detection of the Epstein-Barr Virus-Encoded Latent Membrane Protein 2A in Hodgkin’s Disease and Infectious Mononucleosis. Blood 1997, 90, 1664–1672. [Google Scholar] [CrossRef] [PubMed]
- Young, L.S.; Murray, P.G. Epstein-Barr Virus and Oncogenesis: From Latent Genes to Tumours. Oncogene 2003, 22, 5108–5121. [Google Scholar] [CrossRef]
- Kapatai, G.; Murray, P. Contribution of the Epstein Barr Virus to the Molecular Pathogenesis of Hodgkin Lymphoma. J. Clin. Pathol. 2007, 60, 1342–1349. [Google Scholar] [CrossRef]
- Kieser, A.; Sterz, K.R. The Latent Membrane Protein 1 (LMP1). Curr. Top. Microbiol. Immunol. 2015, 391, 119–149. [Google Scholar] [CrossRef]
- Gires, O.; Kohlhuber, F.; Kilger, E.; Baumann, M.; Kieser, A.; Kaiser, C.; Zeidler, R.; Scheffer, B.; Ueffing, M.; Hammerschmidt, W. Latent Membrane Protein 1 of Epstein-Barr Virus Interacts with JAK3 and Activates STAT Proteins. EMBO J. 1999, 18, 3064–3073. [Google Scholar] [CrossRef]
- Roberts, M.L.; Cooper, N.R. Activation of a Ras-MAPK-Dependent Pathway by Epstein-Barr Virus Latent Membrane Protein 1 Is Essential for Cellular Transformation. Virology 1998, 240, 93–99. [Google Scholar] [CrossRef]
- Martin, G.; Roy, J.; Barat, C.; Ouellet, M.; Gilbert, C.; Tremblay, M.J. Human Immunodeficiency Virus Type 1-Associated CD40 Ligand Transactivates B Lymphocytes and Promotes Infection of CD4+ T Cells. J. Virol. 2007, 81, 5872–5881. [Google Scholar] [CrossRef]
- Imbeault, M.; Ouellet, M.; Giguère, K.; Bertin, J.; Bélanger, D.; Martin, G.; Tremblay, M.J. Acquisition of Host-Derived CD40L by HIV-1 IN VIVO and Its Functional Consequences in the B-Cell Compartment. J. Virol. 2011, 85, 2189–2200. [Google Scholar] [CrossRef] [PubMed]
- Aldinucci, D.; Gloghini, A.; Pinto, A.; Colombatti, A.; Carbone, A. The Role of CD40/CD40L and Interferon Regulatory Factor 4 in Hodgkin Lymphoma Microenvironment. Leuk. Lymphoma 2012, 53, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.L.; Burkhardt, A.L.; Lee, J.H.; Stealey, B.; Longnecker, R.; Bolen, J.B.; Kieff, E. Integral Membrane Protein 2 of Epstein—Barr Virus Regulates Reactivation from Latency through Dominant Negative Effects on Protein-Tyrosine Kinases. Immunity 1995, 2, 155–166. [Google Scholar] [CrossRef]
- Caldwell, R.G.; Wilson, J.B.; Anderson, S.J.; Longnecker, R. Epstein-Barr Virus LMP2A Drives B Cell Development and Survival in the Absence of Normal B Cell Receptor Signals. Immunity 1998, 9, 405–411. [Google Scholar] [CrossRef]
- Green, M.R.; Rodig, S.; Juszczynski, P.; Ouyang, J.; Sinha, P.; O’Donnell, E.; Neuberg, D.; Shipp, M.A. Constitutive AP-1 Activity and EBV Infection Induce PD-L1 in Hodgkin Lymphomas and Posttransplant Lymphoproliferative Disorders: Implications for Targeted Therapy. Clin. Cancer Res. 2012, 18, 1611–1618. [Google Scholar] [CrossRef]
- Frisan, T.; Sjöberg, J.; Dolcetti, R.; Boiocchi, M.; De Re, V.; Carbone, A.; Brautbar, C.; Battat, S.; Biberfeld, P.; Eckman, M. Local Suppression of Epstein-Barr Virus (EBV)-Specific Cytotoxicity in Biopsies of EBV-Positive Hodgkin’s Disease. Blood 1995, 86, 1493–1501. [Google Scholar] [CrossRef]
- Chapman, A.L.; Rickinson, A.B.; Thomas, W.A.; Jarrett, R.F.; Crocker, J.; Lee, S.P. Epstein-Barr Virus-Specific Cytotoxic T Lymphocyte Responses in the Blood and Tumor Site of Hodgkin’s Disease Patients: Implications for a T-Cell-Based Therapy. Cancer Res. 2001, 61, 6219–6226. [Google Scholar]
- Marshall, N.A.; Christie, L.E.; Munro, L.R.; Culligan, D.J.; Johnston, P.W.; Barker, R.N.; Vickers, M.A. Immunosuppressive Regulatory T Cells Are Abundant in the Reactive Lymphocytes of Hodgkin Lymphoma. Blood 2004, 103, 1755–1762. [Google Scholar] [CrossRef]
- Álvaro, T.; Lejeune, M.; Salvadó, M.T.; Bosch, R.; García, J.F.; Jaén, J.; Banham, A.H.; Roncador, G.; Montalbán, C.; Piris, M.A. Outcome in Hodgkin’s Lymphoma Can Be Predicted from the Presence of Accompanying Cytotoxic and Regulatory T Cells. Clin. Cancer Res. 2005, 11, 1467–1473. [Google Scholar] [CrossRef]
- Baráth, S.; Aleksza, M.; Keresztes, K.; Tóth, J.; Sipka, S.; Szegedi, G.; Illés, A. Immunoregulatory T Cells in the Peripheral Blood of Patients with Hodgkin’s Lymphoma. Acta Haematol. 2006, 116, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Baumforth, K.R.N.; Birgersdotter, A.; Reynolds, G.M.; Wei, W.; Kapatai, G.; Flavell, J.R.; Kalk, E.; Piper, K.; Lee, S.; Machado, L.; et al. Expression of the Epstein-Barr Virus-Encoded Epstein-Barr Virus Nuclear Antigen 1 in Hodgkin’s Lymphoma Cells Mediates Up-Regulation of CCL20 and the Migration of Regulatory T Cells. Am. J. Pathol. 2008, 173, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Marshall, N.A.; Culligan, D.J.; Tighe, J.; Johnston, P.W.; Barker, R.N.; Vickers, M.A. The Relationships between Epstein-Barr Virus Latent Membrane Protein 1 and Regulatory T Cells in Hodgkin’s Lymphoma. Exp. Hematol. 2007, 35, 596–604. [Google Scholar] [CrossRef]
- Kamper, P.; Bendix, K.; Hamilton-Dutoit, S.; Honoré, B.; Nyengaard, J.R.; d’Amore, F. Tumor-Infiltrating Macrophages Correlate with Adverse Prognosis and Epstein-Barr Virus Status in Classical Hodgkin’s Lymphoma. Haematologica 2011, 96, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.L.; Scott, D.W.; Hong, F.; Kahl, B.S.; Fisher, R.I.; Bartlett, N.L.; Advani, R.H.; Buckstein, R.; Rimsza, L.M.; Connors, J.M.; et al. Tumor-Associated Macrophages Predict Inferior Outcomes in Classic Hodgkin Lymphoma: A Correlative Study from the E2496 Intergroup Trial. Blood 2012, 120, 3280–3287. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D. Anatomy of a Discovery: M1 and M2 Macrophages. Front. Immunol. 2015, 6. [Google Scholar] [CrossRef]
- Barros, M.H.M.; Hauck, F.; Dreyer, J.H.; Kempkes, B.; Niedobitek, G. Macrophage Polarisation: An Immunohistochemical Approach for Identifying M1 and M2 Macrophages. PLoS ONE 2013, 8, e80908. [Google Scholar] [CrossRef]
- Hartmann, S.; Jakobus, C.; Rengstl, B.; Döring, C.; Newrzela, S.; Brodt, H.-R.; Wolf, T.; Hansmann, M.-L. Spindle-Shaped CD163+ Rosetting Macrophages Replace CD4+ T-Cells in HIV-Related Classical Hodgkin Lymphoma. Mod. Pathol. 2013, 26, 648–657. [Google Scholar] [CrossRef]
- Cassol, E.; Cassetta, L.; Rizzi, C.; Alfano, M.; Poli, G. M1 and M2a Polarization of Human Monocyte-Derived Macrophages Inhibits HIV-1 Replication by Distinct Mechanisms. J. Immunol. 2009, 182, 6237–6246. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.D.R.; Fisher, S.I.; Chu, W.S.; Nelson, A.; Abbondanzo, S.L. HIV-Associated Hodgkin Lymphoma: A Clinicopathologic and Immunophenotypic Study of 45 Cases. Am. J. Clin. Pathol. 2004, 121, 727–738. [Google Scholar] [CrossRef]
- Koulis, A.; Trivedi, P.; Ibrahim, H.; Bower, M.; Naresh, K.N. The Role of the Microenvironment in Human Immunodeficiency Virus-Associated Classical Hodgkin Lymphoma. Histopathology 2014, 65, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Scala, G.; Ruocco, M.R.; Ambrosino, C.; Mallardo, M.; Giordano, V.; Baldassarre, F.; Dragonetti, E.; Quinto, I.; Venuta, S. The Expression of the Interleukin 6 Gene Is Induced by the Human Immunodeficiency Virus 1 TAT Protein. J. Exp. Med. 1994, 179, 961–971. [Google Scholar] [CrossRef]
- Blazevic, V.; Heino, M.; Lagerstedt, A.; Ranki, A.; Krohn, K.J. Interleukin-10 Gene Expression Induced by HIV-1 Tat and Rev in the Cells of HIV-1 Infected Individuals. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1996, 13, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, E.; Hussain, S.K.; Breen, E.C.; Magpantay, L.I.; Widney, D.P.; Jacobson, L.P.; Variakojis, D.; Knowlton, E.R.; Bream, J.H.; Ambinder, R.F.; et al. Serum Levels of Cytokines and Biomarkers for Inflammation and Immune Activation, and HIV-Associated Non-Hodgkin B-Cell Lymphoma Risk. Cancer Epidemiol. Biomark. Prev. 2014, 23, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, M.; Kinoshita, K.; Fagarasan, S.; Yamada, S.; Shinkai, Y.; Honjo, T. Class Switch Recombination and Hypermutation Require Activation-Induced Cytidine Deaminase (AID), a Potential RNA Editing Enzyme. Cell 2000, 102, 553–563. [Google Scholar] [CrossRef]
- Robbiani, D.F.; Bunting, S.; Feldhahn, N.; Bothmer, A.; Camps, J.; Deroubaix, S.; McBride, K.M.; Klein, I.A.; Stone, G.; Eisenreich, T.R.; et al. AID Produces DNA Double-Strand Breaks in Non-Ig Genes and Mature B Cell Lymphomas with Reciprocal Chromosome Translocations. Mol. Cell 2009, 36, 631–641. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 Revision of the World Health Organization Classification of Lymphoid Neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [CrossRef]
- Hentrich, M.; Maretta, L.; Chow, K.U.; Bogner, J.R.; Schürmann, D.; Neuhoff, P.; Jäger, H.; Reichelt, D.; Vogel, M.; Ruhnke, M.; et al. Highly Active Antiretroviral Therapy (HAART) Improves Survival in HIV-Associated Hodgkin’s Disease: Results of a Multicenter Study. Ann. Oncol. 2006, 17, 914–919. [Google Scholar] [CrossRef]
- Corti, M.; Villafañe, M.; Minue, G.; Campitelli, A.; Narbaitz, M.; Gilardi, L. Clinical Features of AIDS Patients with Hodgkin’s Lymphoma with Isolated Bone Marrow Involvement: Report of 12 Cases at a Single Institution. Cancer Biol. Med. 2015, 12, 41–45. [Google Scholar] [CrossRef]
- Muthukrishnan, S.; Amudhan, A.; Rajendran, S. Primary Hodgkin’s Lymphoma of Liver in HIV—A Case Report and Review of Literature. AME Case Rep. 2018, 2. [Google Scholar] [CrossRef] [PubMed]
- Spina, M.; Gabarre, J.; Rossi, G.; Fasan, M.; Schiantarelli, C.; Nigra, E.; Mena, M.; Antinori, A.; Ammassari, A.; Talamini, R.; et al. Stanford V Regimen and Concomitant HAART in 59 Patients with Hodgkin Disease and HIV Infection. Blood 2002, 100, 1984–1988. [Google Scholar] [CrossRef][Green Version]
- Hartmann, P.; Rehwald, U.; Salzberger, B.; Franzen, C.; Sieber, M.; Wöhrmann, A.; Diehl, V. BEACOPP Therapeutic Regimen for Patients with Hodgkin’s Disease and HIV Infection. Ann. Oncol. 2003, 14, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- Hentrich, M.; Berger, M.; Wyen, C.; Siehl, J.; Rockstroh, J.K.; Müller, M.; Fätkenheuer, G.; Seidel, E.; Nickelsen, M.; Wolf, T.; et al. Stage-Adapted Treatment of HIV-Associated Hodgkin Lymphoma: Results of a Prospective Multicenter Study. J. Clin. Oncol. 2012, 30, 4117–4123. [Google Scholar] [CrossRef] [PubMed]
- Castillo, J.J.; Bower, M.; Brühlmann, J.; Novak, U.; Furrer, H.; Tanaka, P.Y.; Besson, C.; Montoto, S.; Cwynarski, K.; Abramson, J.S.; et al. Prognostic Factors for Advanced-Stage Human Immunodeficiency Virus-Associated Classical Hodgkin Lymphoma Treated with Doxorubicin, Bleomycin, Vinblastine, and Dacarbazine plus Combined Antiretroviral Therapy: A Multi-Institutional Retrospective Study. Cancer 2015, 121, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Yotsumoto, M.; Ito, Y.; Hagiwara, S.; Terui, Y.; Nagai, H.; Ota, Y.; Ajisawa, A.; Uehira, T.; Tanuma, J.; Ohyashiki, K.; et al. HIV Positivity May Not Have a Negative Impact on Survival in Epstein-Barr Virus-Positive Hodgkin Lymphoma: A Japanese Nationwide Retrospective Survey. Oncol. Lett. 2018, 16, 3923–3928. [Google Scholar] [CrossRef] [PubMed]
- Lawal, I.O.; Ankrah, A.O.; Popoola, G.O.; Nyakale, N.E.; Boshomane, T.G.; Reyneke, F.; Lengana, T.; Vorster, M.; Sathekge, M.M. 18F-FDG-PET Metabolic Metrics and International Prognostic Score for Risk Assessment in HIV-Infected Patients with Hodgkin Lymphoma. Nucl. Med. Commun. 2018, 39, 1005–1012. [Google Scholar] [CrossRef]
- Okosun, J.; Warbey, V.; Shaw, K.; Montoto, S.; Fields, P.; Marcus, R.; Virchis, A.; McNamara, C.; Bower, M.; Cwynarski, K. Interim Fluoro-2-Deoxy-D-Glucose-PET Predicts Response and Progression-Free Survival in Patients with Hodgkin Lymphoma and HIV Infection. AIDS 2012, 26, 861–865. [Google Scholar] [CrossRef]
- Danilov, A.V.; Li, H.; Press, O.W.; Shapira, I.; Swinnen, L.J.; Noy, A.; Reid, E.; Smith, S.M.; Friedberg, J.W. Feasibility of Interim Positron Emission Tomography (PET)-Adapted Therapy in HIV-Positive Patients with Advanced Hodgkin Lymphoma (HL): A Sub-Analysis of SWOG S0816 Phase 2 Trial. Leuk. Lymphoma 2017, 58, 461–465. [Google Scholar] [CrossRef]
- Rubinstein, P.G.; Moore, P.C.; Rudek, M.A.; Henry, D.H.; Ramos, J.C.; Ratner, L.; Reid, E.; Sharon, E.; Noy, A.; AIDS Malignancy Consortium (AMC). Brentuximab Vedotin with AVD Shows Safety, in the Absence of Strong CYP3A4 Inhibitors, in Newly Diagnosed HIV-Associated Hodgkin Lymphoma. AIDS 2018, 32, 605–611. [Google Scholar] [CrossRef]
- Krishnan, A.; Molina, A.; Zaia, J.; Smith, D.; Vasquez, D.; Kogut, N.; Falk, P.M.; Rosenthal, J.; Alvarnas, J.; Forman, S.J. Durable Remissions with Autologous Stem Cell Transplantation for High-Risk HIV-Associated Lymphomas. Blood 2005, 105, 874–878. [Google Scholar] [CrossRef]
- Spitzer, T.R.; Ambinder, R.F.; Lee, J.Y.; Kaplan, L.D.; Wachsman, W.; Straus, D.J.; Aboulafia, D.M.; Scadden, D.T. Dose-Reduced Busulfan, Cyclophosphamide, and Autologous Stem Cell Transplantation for Human Immunodeficiency Virus-Associated Lymphoma: AIDS Malignancy Consortium Study 020. Biol. Blood Marrow Transplant. 2008, 14, 59–66. [Google Scholar] [CrossRef][Green Version]
- Re, A.; Michieli, M.; Casari, S.; Allione, B.; Cattaneo, C.; Rupolo, M.; Spina, M.; Manuele, R.; Vaccher, E.; Mazzucato, M.; et al. High-Dose Therapy and Autologous Peripheral Blood Stem Cell Transplantation as Salvage Treatment for AIDS-Related Lymphoma: Long-Term Results of the Italian Cooperative Group on AIDS and Tumors (GICAT) Study with Analysis of Prognostic Factors. Blood 2009, 114, 1306–1313. [Google Scholar] [CrossRef]
- Durand, C.M.; Capoferri, A.A.; Redd, A.D.; Zahurak, M.; Rosenbloom, D.I.S.; Cash, A.; Avery, R.K.; Bolaños-Meade, J.; Bollard, C.M.; Bullen, C.K.; et al. Allogeneic Bone Marrow Transplantation with Post-Transplant Cyclophosphamide for Patients with HIV and Haematological Malignancies: A Feasibility Study. Lancet HIV 2020, 7, e602–e610. [Google Scholar] [CrossRef]
- Chang, E.; Rivero, G.; Patel, N.R.; Chiao, E.Y.; Lai, S.; Bajaj, K.; Mbue, J.E.; Yellapragada, S.V. HIV-Related Refractory Hodgkin Lymphoma: A Case Report of Complete Response to Nivolumab. Clin. Lymphoma Myeloma Leuk. 2018, 18, e143–e146. [Google Scholar] [CrossRef] [PubMed]
- Sandoval-Sus, J.D.; Mogollon-Duffo, F.; Patel, A.; Visweshwar, N.; Laber, D.A.; Kim, R.; Jagal, M.V. Nivolumab as Salvage Treatment in a Patient with HIV-Related Relapsed/Refractory Hodgkin Lymphoma and Liver Failure with Encephalopathy. J. Immunother. Cancer 2017, 5, 49. [Google Scholar] [CrossRef]
- Cullen, M.; Steven, N.; Billingham, L.; Gaunt, C.; Hastings, M.; Simmonds, P.; Stuart, N.; Rea, D.; Bower, M.; Fernando, I.; et al. Antibacterial Prophylaxis after Chemotherapy for Solid Tumors and Lymphomas. N. Engl. J. Med. 2005, 353, 988–998. [Google Scholar] [CrossRef] [PubMed]
- Re, A.; Cattaneo, C.; Montoto, S. Treatment Management of Haematological Malignancies in People Living with HIV. Lancet Haematol. 2020, 7, e679–e689. [Google Scholar] [CrossRef]
- Powles, T.; Imami, N.; Nelson, M.; Gazzard, B.G.; Bower, M. Effects of Combination Chemotherapy and Highly Active Antiretroviral Therapy on Immune Parameters in HIV-1 Associated Lymphoma. AIDS 2002, 16, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Esdaile, B.; Davis, M.; Portsmouth, S.; Sarker, D.; Nelson, M.; Gazzard, B.; Bower, M. The Immunological Effects of Concomitant Highly Active Antiretroviral Therapy and Liposomal Anthracycline Treatment of HIV-1-Associated Kaposi’s Sarcoma. AIDS 2002, 16, 2344–2347. [Google Scholar] [CrossRef]
- Alfa-Wali, M.; Allen-Mersh, T.; Antoniou, A.; Tait, D.; Newsom-Davis, T.; Gazzard, B.; Nelson, M.; Bower, M. Chemoradiotherapy for Anal Cancer in HIV Patients Causes Prolonged CD4 Cell Count Suppression. Ann. Oncol. 2012, 23, 141–147. [Google Scholar] [CrossRef]
- Uldrick, T.S.; Little, R.F. How I Treat Classical Hodgkin Lymphoma in Patients Infected with Human Immunodeficiency Virus. Blood 2015, 125, 1226–1235. [Google Scholar] [CrossRef]
- Little, R.F.; Dunleavy, K. Update on the Treatment of HIV-Associated Hematologic Malignancies. Hematol. Am. Soc. Hematol. Educ. Program. 2013, 2013, 382–388. [Google Scholar] [CrossRef]
- Navarro, J.-T.; Ribera, J.-M.; Oriol, A.; Romeu, J.; Sirera, G.; Mate, J.-L.; Batlle, M.; Xicoy, B.; Grau, J.; Millá, F.; et al. Favorable Impact of Virological Response to Highly Active Antiretroviral Therapy on Survival in Patients with AIDS-Related Lymphoma. Leuk. Lymphoma 2002, 43, 1837–1842. [Google Scholar] [CrossRef]
- Weiss, R.; Mitrou, P.; Arasteh, K.; Schuermann, D.; Hentrich, M.; Duehrsen, U.; Sudeck, H.; Schmidt-Wolf, I.G.H.; Anagnostopoulos, I.; Huhn, D. Acquired Immunodeficiency Syndrome-Related Lymphoma: Simultaneous Treatment with Combined Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone Chemotherapy and Highly Active Antiretroviral Therapy Is Safe and Improves Survival--Results of the German Multicenter Trial. Cancer 2006, 106, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- Mounier, N.; Spina, M.; Gabarre, J.; Raphael, M.; Rizzardini, G.; Golfier, J.-B.; Vaccher, E.; Carbone, A.; Coiffier, B.; Chichino, G.; et al. AIDS-Related Non-Hodgkin Lymphoma: Final Analysis of 485 Patients Treated with Risk-Adapted Intensive Chemotherapy. Blood 2006, 107, 3832–3840. [Google Scholar] [CrossRef]
- Gopal, S.; Patel, M.R.; Yanik, E.L.; Cole, S.R.; Achenbach, C.J.; Napravnik, S.; Burkholder, G.A.; Reid, E.G.; Rodriguez, B.; Deeks, S.G.; et al. Association of Early HIV Viremia with Mortality after HIV-Associated Lymphoma. AIDS 2013, 27, 2365–2373. [Google Scholar] [CrossRef] [PubMed]
- Vaccher, E.; Spina, M.; di Gennaro, G.; Talamini, R.; Nasti, G.; Schioppa, O.; Vultaggio, G.; Tirelli, U. Concomitant Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone Chemotherapy plus Highly Active Antiretroviral Therapy in Patients with Human Immunodeficiency Virus-Related, Non-Hodgkin Lymphoma. Cancer 2001, 91, 155–163. [Google Scholar] [CrossRef]
- Antinori, A.; Cingolani, A.; Alba, L.; Ammassari, A.; Serraino, D.; Ciancio, B.C.; Palmieri, F.; De Luca, A.; Larocca, L.M.; Ruco, L.; et al. Better Response to Chemotherapy and Prolonged Survival in AIDS-Related Lymphomas Responding to Highly Active Antiretroviral Therapy. AIDS 2001, 15, 1483–1491. [Google Scholar] [CrossRef] [PubMed]
- Strategies for Management of Antiretroviral Therapy (SMART) Study Group; El-Sadr, W.M.; Lundgren, J.D.; Neaton, J.D.; Gordin, F.; Abrams, D.; Arduino, R.C.; Babiker, A.; Burman, W.; Clumeck, N.; et al. CD4+ Count-Guided Interruption of Antiretroviral Treatment. N. Engl. J. Med. 2006, 355, 2283–2296. [Google Scholar] [CrossRef]
- Torres, H.A.; Mulanovich, V. Management of HIV Infection in Patients with Cancer Receiving Chemotherapy. Clin. Infect. Dis. 2014, 59, 106–114. [Google Scholar] [CrossRef]
- Department of Health and Human Services. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents; Department of Health and Human Services: Washington, DC, USA.
- King, J.R.; Wynn, H.; Brundage, R.; Acosta, E.P. Pharmacokinetic Enhancement of Protease Inhibitor Therapy. Clin. Pharmacokinet. 2004, 43, 291–310. [Google Scholar] [CrossRef]
- Shah, B.M.; Schafer, J.J.; Priano, J.; Squires, K.E. Cobicistat: A New Boost for the Treatment of Human Immunodeficiency Virus Infection. Pharmacotherapy 2013, 33, 1107–1116. [Google Scholar] [CrossRef]
- Sombogaard, F.; Franssen, E.J.F.; Terpstra, W.E.; Kerver, E.D.; van den Berk, G.E.L.; Crul, M. Outcome Effects of Antiretroviral Drug Combinations in HIV-Positive Patients with Chemotherapy for Lymphoma: A Retrospective Analysis. Int. J. Clin. Pharm. 2018, 40, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Focà, E.; Cavaglià, G.; Rusconi, S.; Cascavilla, A.; Cenderello, G.; Re, A.; Casari, S.; van den Bogaart, L.; Zinzani, P.L.; Caracciolo, D.; et al. Survival in HIV-Infected Patients with Lymphoma According to the Choice of Antiretroviral Treatment: An Observational Multicentre Study. HIV Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Torres, H.A.; Rallapalli, V.; Saxena, A.; Granwehr, B.P.; Viola, G.M.; Ariza-Heredia, E.; Adachi, J.A.; Chemaly, R.F.; Marfatia, R.; Jiang, Y.; et al. Efficacy and Safety of Antiretrovirals in HIV-Infected Patients with Cancer. Clin. Microbiol. Infect. 2014, 20, O672–O679. [Google Scholar] [CrossRef] [PubMed]
- Bower, M.; Powles, T.; Stebbing, J.; Thirlwell, C. Potential Antiretroviral Drug Interactions with Cyclophosphamide, Doxorubicin, and Etoposide. J. Clin. Oncol. 2005, 23, 1328–1329. [Google Scholar] [CrossRef]
- Levêque, D.; Santucci, R.; Pavillet, J.; Herbrecht, R.; Bergerat, J.P. Paralytic Ileus Possibly Associated with Interaction between Ritonavir/Lopinavir and Vincristine. Pharm. World Sci. PWS 2009, 31, 619–621. [Google Scholar] [CrossRef]
- Liedtke, M.D.; Tomlin, C.R.; Lockhart, S.M.; Miller, M.M.; Rathbun, R.C. Long-Term Efficacy and Safety of Raltegravir in the Management of HIV Infection. Infect. Drug Resist. 2014, 7, 73–84. [Google Scholar] [CrossRef]
- Cottrell, M.L.; Hadzic, T.; Kashuba, A.D.M. Clinical Pharmacokinetic, Pharmacodynamic and Drug-Interaction Profile of the Integrase Inhibitor Dolutegravir. Clin. Pharmacokinet. 2013, 52, 981–994. [Google Scholar] [CrossRef]
- Zeuli, J.; Rizza, S.; Bhatia, R.; Temesgen, Z. Bictegravir, a Novel Integrase Inhibitor for the Treatment of HIV Infection. Drugs Today Barc. 2019, 55, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Moltó, J.; Rajoli, R.; Back, D.; Valle, M.; Miranda, C.; Owen, A.; Clotet, B.; Siccardi, M. Use of a Physiologically Based Pharmacokinetic Model to Simulate Drug-Drug Interactions between Antineoplastic and Antiretroviral Drugs. J. Antimicrob. Chemother. 2017, 72, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Saravolatz, L.D. Rilpivirine: A New Non-Nucleoside Reverse Transcriptase Inhibitor. J. Antimicrob. Chemother. 2013, 68, 250–256. [Google Scholar] [CrossRef]
- Khalilieh, S.; Yee, K.L.; Sanchez, R.; Stoch, S.A.; Wenning, L.; Iwamoto, M. Clinical Pharmacokinetics of the Novel HIV-1 Non-Nucleoside Reverse Transcriptase Inhibitor Doravirine: An Assessment of the Effect of Patient Characteristics and Drug-Drug Interactions. Clin. Drug Investig. 2020, 40, 927–946. [Google Scholar] [CrossRef] [PubMed]
- Scherzer, R.; Estrella, M.; Li, Y.; Choi, A.I.; Deeks, S.G.; Grunfeld, C.; Shlipak, M.G. Association of Tenofovir Exposure with Kidney Disease Risk in HIV Infection. AIDS 2012, 26, 867–875. [Google Scholar] [CrossRef] [PubMed]
- James, C.W.; Szabo, S.; Kahal, D.; Goldstein, N.D. The Effect of Multivitamins and Polyvalent Cations on Virologic Suppression with Integrase Strand Transfer Inhibitors. AIDS 2020, 34, 487–489. [Google Scholar] [CrossRef]
- Rock, A.E.; DeMarais, P.L.; Vergara-Rodriguez, P.T.; Max, B.E. HIV-1 Virologic Rebound Due to Coadministration of Divalent Cations and Bictegravir. Infect. Dis. Ther. 2020, 9, 691–696. [Google Scholar] [CrossRef]
- Liverpool HIV Interactions. Available online: https://www.hiv-druginteractions.org/ (accessed on 30 May 2021).
- Immunodeficiency Clinic. Available online: https://hivclinic.ca/main/drugs_interact.html (accessed on 30 May 2021).
- Rubinstein, P.G.; Aboulafia, D.M.; Zloza, A. Malignancies in HIV/AIDS: From Epidemiology to Therapeutic Challenges. AIDS 2014, 28, 453–465. [Google Scholar] [CrossRef]
- Rudek, M.A.; Flexner, C.; Ambinder, R.F. Use of Antineoplastic Agents in Patients with Cancer Who Have HIV/AIDS. Lancet Oncol. 2011, 12, 905–912. [Google Scholar] [CrossRef]
| Author | Chemotherapy Regimen | N | Median Age * (Range) | Stage | CD4+ Count/µL * Median (Range) | CR (%) | Survival (%) | Overall Survival (%) |
|---|---|---|---|---|---|---|---|---|
| Spina et al. [72] | Stanford V | 59 | 38 (28–64) | I–IV | 238 (32–1038) | 81 | 68 (3-year DFS) | 51 (3-year) |
| Hartmann et al. [73] | BEACOPP | 12 | 33 (22–49) | III–IV | 205 (110–1020) | 100 | 70 (5-year DFS) | 70 (5-year) |
| Xicoy et al. [6] | ABVD | 51 | 37 (24–61) | II–IV | 129 (5–1209) | 87 | 95 (5-year EFS) | 76 (5-year) |
| Montoto et al. [14] | ABVD | 93 | 41 (26–73) | I–IV | NA | 74 | 59 (5-year EFS) | 81 (5-year) |
| Hentrich et al. [74] 1 | BEACOPP baseline or ABVD 2 Stage-adapted | 71/108 | 44 (27–70) 3 | III–IV | 240 (7–967) 3 | 86 1 | 87.5 (2-year PFS) 1 | 87 (2-year) 1 |
| Castillo et al. [75] | ABVD | 229 | NA | III–IV | NA | 83 | 69 (5-year PFS) | 78 (5-year) |
| Besson et al. [19] | ABVD (96%) | 68 | 44 (38–48) | I–IV | 387 (151–540) | NA | 89 (2-year PFS | 94 (2-year) |
| Sorigué et al. [9] | ABVD | 21 | 40 (18–56) | III–IV | NA | 89 | 70 (10-year DFS) | 73 (10-year) |
| DRV/r DRV/c | ATV/r ATV/c | LPV/r | NVP | EFV | ETR | RPV | DOR | RAL | EVG/c | DTG | BIC | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cyclophosphamide (CYC) | Monitor CYC toxicity | Monitor CYC toxicity | Monitor CYC toxicity | Monitor CYC efficacy/toxicity | Monitor CYC efficacy/toxicity | Monitor CYC efficacy/toxicity | No DDI expected | No DDI expected | No DDI expected | Monitor CYC toxicity | No DDI expected | No DDI expected |
| Doxorubicin (DOX) | No DDI expected | Monitor ECG ** | Monitor ECG ** | No DDI expected | No DDI expected | No DDI expected | Monitor ECG ** | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
| Vincristine/Vinblastine (VIN) | Increased VIN toxicity | Increased VIN toxicity | Increased VIN toxicity | Monitor VIN efficacy | Monitor VIN efficacy | Monitor VIN efficacy | No DDI expected | No DDI expected | No DDI expected | Increased VIN toxicity | No DDI expected | No DDI expected |
| Prednisone (PRE) | Monitor PRE toxicity | Monitor PRE toxicity | Monitor PRE toxicity | Monitor PRE efficacy | Monitor PRE efficacy | Monitor PRE efficacy | No DDI expected | No DDI expected | No DDI expected | Monitor PRE toxicity | No DDI expected | No DDI expected |
| Etoposide (ETO) | Monitor ETO toxicity | Monitor ETO toxicity | Monitor ETO toxicity | Monitor ETO efficacy | Monitor ETO efficacy | Monitor ETO efficacy | No DDI expected | No DDI expected | No DDI expected | Monitor ETO toxicity | No DDI expected | No DDI expected |
| Bleomycin (BLE) | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
| Brentuximab (BRE) | Monitor BRE toxicity | Monitor BRE toxicity | Monitor BRE toxicity | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | Monitor BRE toxicity | No DDI expected | No DDI expected |
| Dacarbazine (DAC) | Monitor DAC toxicity | Monitor DAC toxicity | Monitor DAC toxicity | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
| Nivolumab (NIV) | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
| Pembrolizumab (PEM) | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
| Procarbazine (PRO) | Monitor PRO efficacy | Monitor PRO efficacy | Monitor PRO efficacy | Monitor PRO efficacy | Monitor PRO efficacy | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
| Rituximab (RIT) | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected | No DDI expected |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro, J.-T.; Moltó, J.; Tapia, G.; Ribera, J.-M. Hodgkin Lymphoma in People Living with HIV. Cancers 2021, 13, 4366. https://doi.org/10.3390/cancers13174366
Navarro J-T, Moltó J, Tapia G, Ribera J-M. Hodgkin Lymphoma in People Living with HIV. Cancers. 2021; 13(17):4366. https://doi.org/10.3390/cancers13174366
Chicago/Turabian StyleNavarro, Jose-Tomas, José Moltó, Gustavo Tapia, and Josep-Maria Ribera. 2021. "Hodgkin Lymphoma in People Living with HIV" Cancers 13, no. 17: 4366. https://doi.org/10.3390/cancers13174366
APA StyleNavarro, J.-T., Moltó, J., Tapia, G., & Ribera, J.-M. (2021). Hodgkin Lymphoma in People Living with HIV. Cancers, 13(17), 4366. https://doi.org/10.3390/cancers13174366






