High Serum Elafin Prediction of Poor Prognosis of Locoregional Esophageal Squamous Cell Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Study
2.1.1. Study Subjects
2.1.2. Serum Elafin Levels by Enzyme-Linked Immunosorbent Assay (ELISA)
2.2. In Vitro Study
2.2.1. Cell Lines and Cell Culture
2.2.2. Elafin Knockdown by shRNA
2.2.3. Cell Motility and Invasion Assays
2.2.4. Immunoblot Analysis
2.3. Statistical Analysis
3. Results
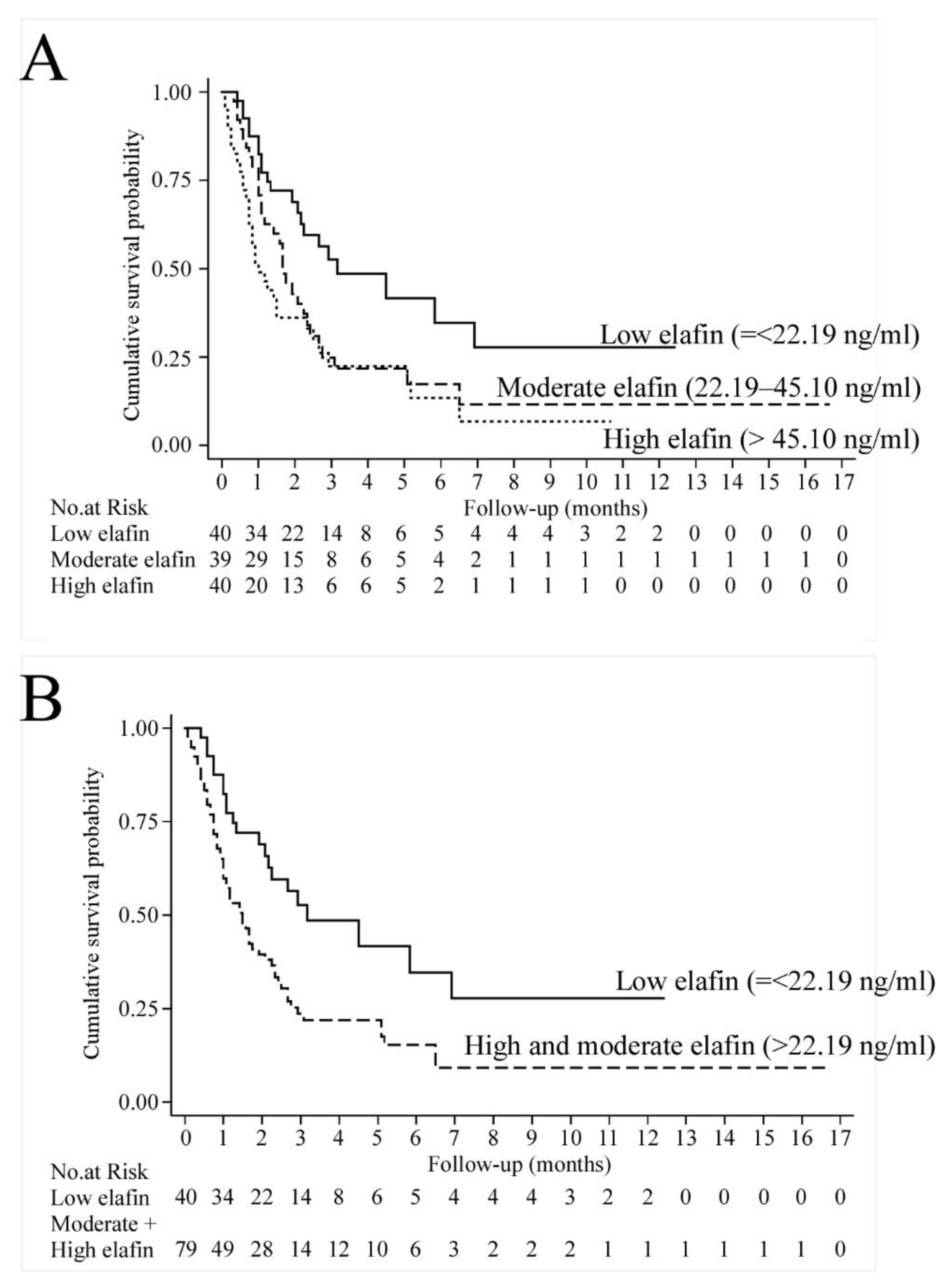
3.1. Human Study
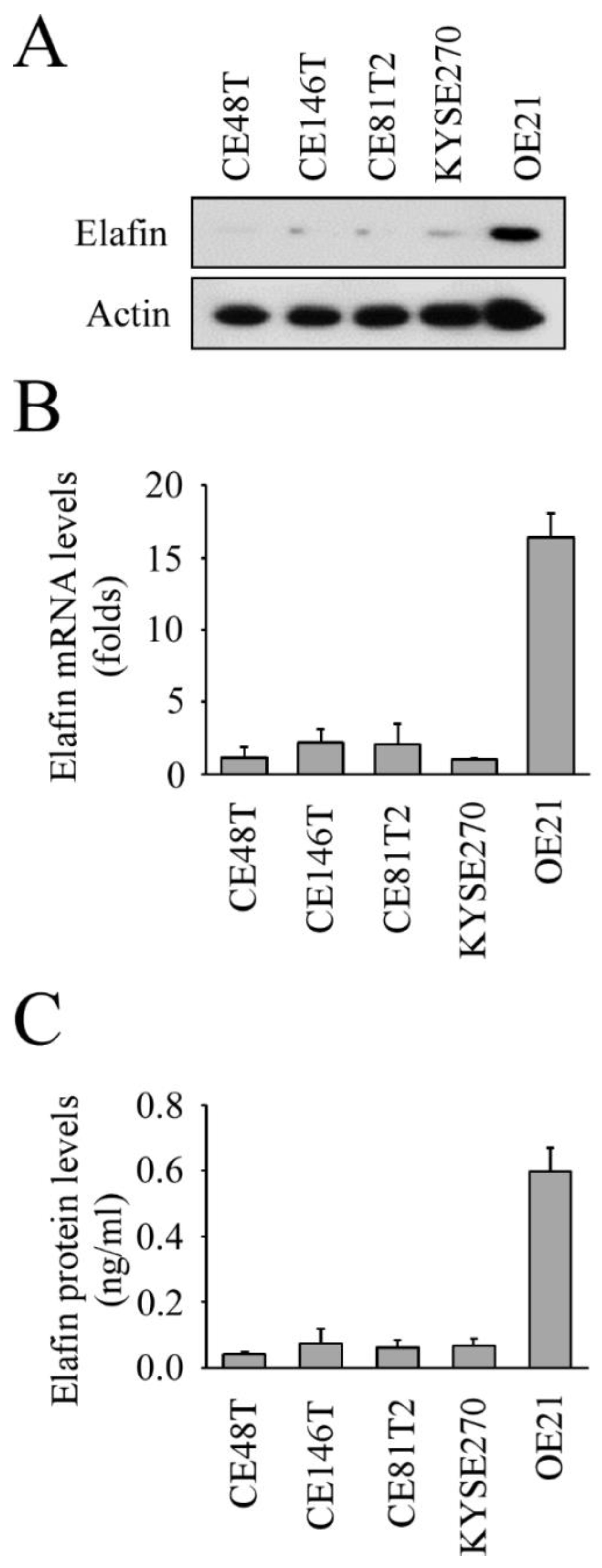
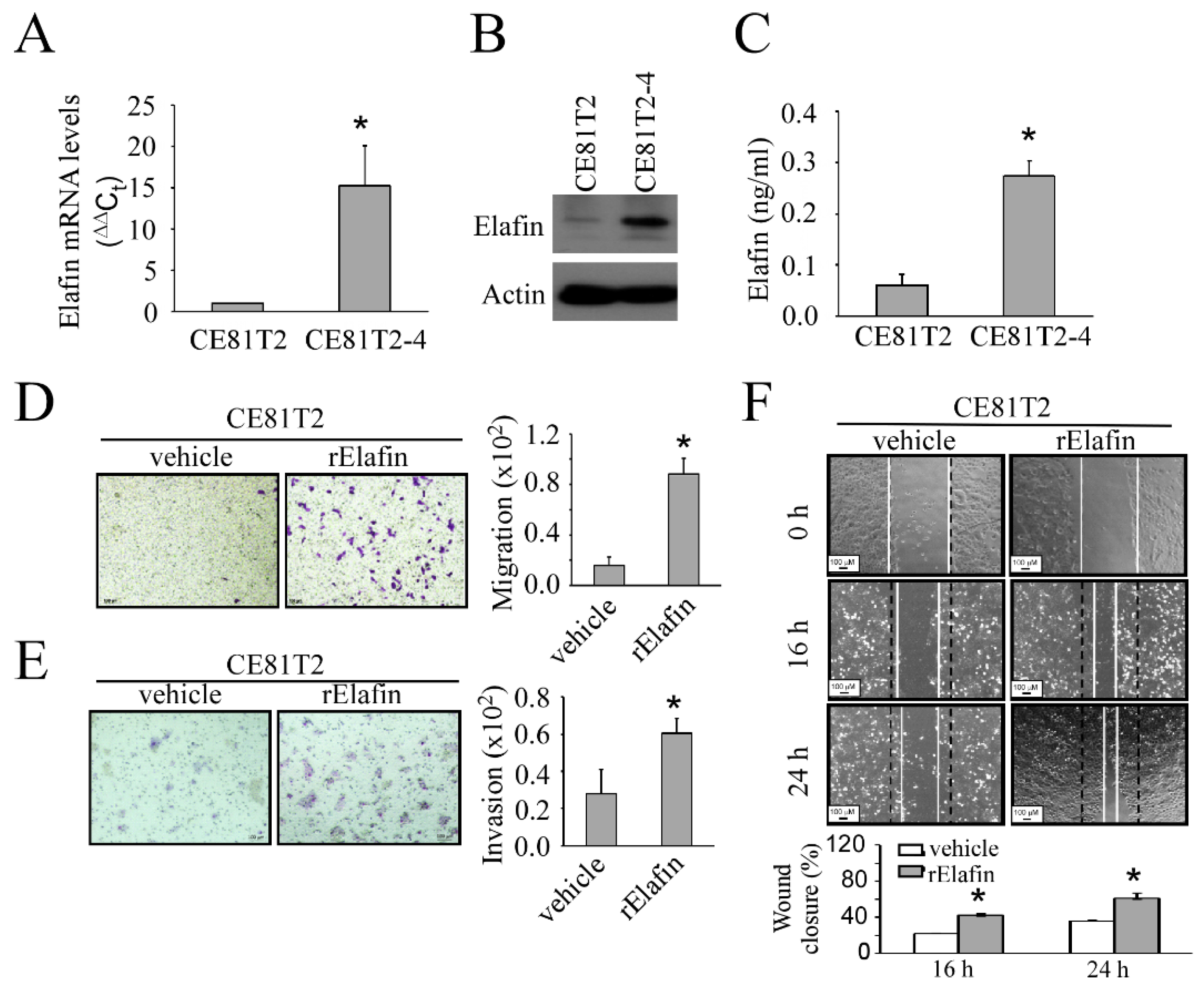
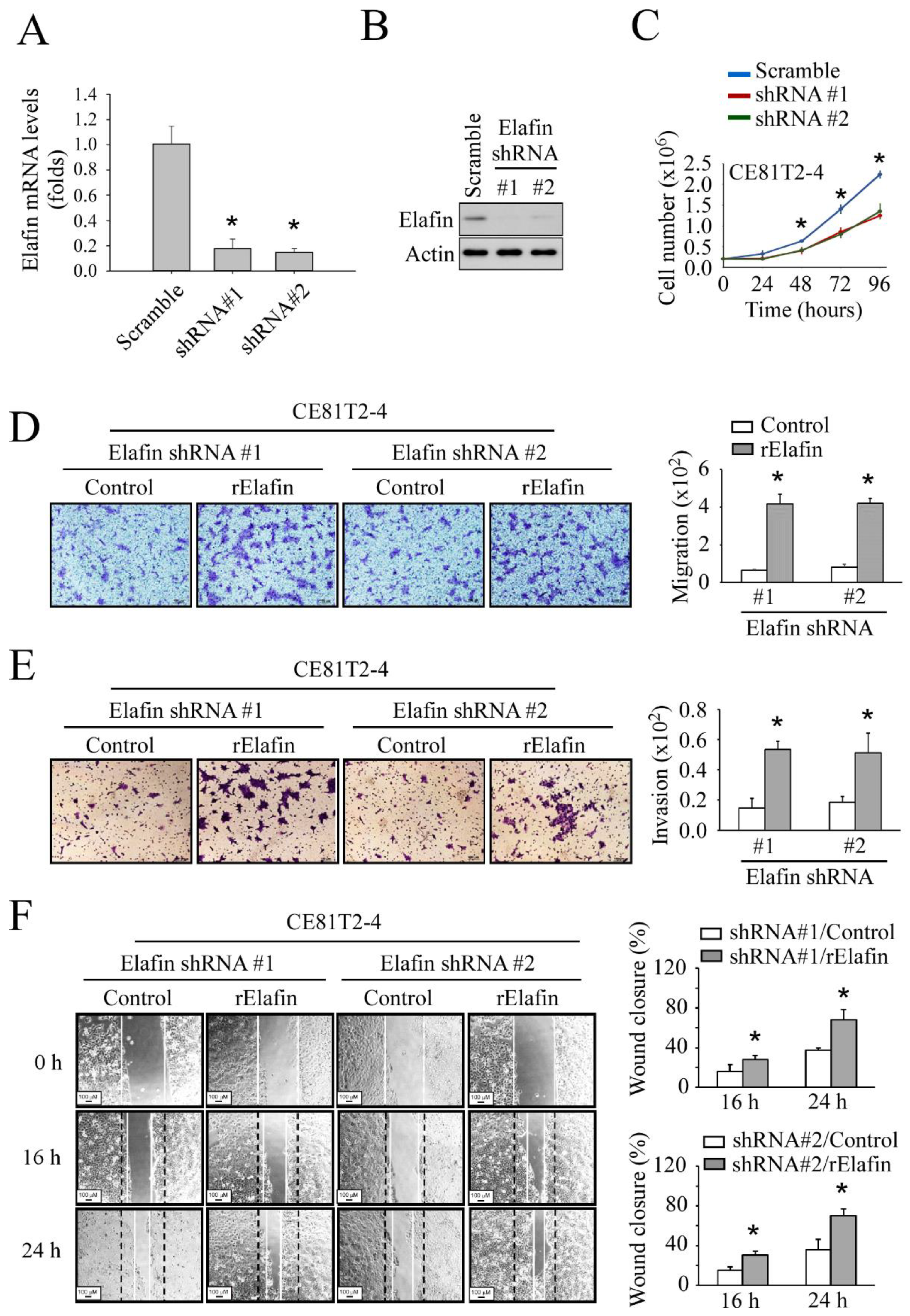
3.2. In Vitro Study
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abnet, C.C.; Arnold, M.; Wei, W.Q. Epidemiology of Esophageal Squamous Cell Carcinoma. Gastroenterology 2018, 154, 360–373. [Google Scholar] [CrossRef] [PubMed]
- Pennathur, A.; Gibson, M.K.; Jobe, B.A.; Luketich, J.D. Oesophageal carcinoma. Lancet 2013, 381, 400–412. [Google Scholar] [CrossRef]
- Singhi, A.D.; Seethala, R.R.; Nason, K.; Foxwell, T.J.; Roche, R.L.; McGrath, K.M.; Levy, R.M.; Luketich, J.D.; Davison, J.M. Undifferentiated carcinoma of the esophagus: A clinicopathological study of 16 cases. Hum. Pathol. 2015, 46, 366–375. [Google Scholar] [CrossRef]
- Wang, Q.L.; Xie, S.H.; Wahlin, K.; Lagergren, J. Global time trends in the incidence of esophageal squamous cell carcinoma. Clin. Epidemiol. 2018, 10, 717–728. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Egami, H.; Kurizaki, T.; Ohmachi, H.; Hayashi, N.; Okino, T.; Shibata, Y.; Schalkwijk, J.; Ogawa, M. Immunohistochemical expression of SKALP/elafin in squamous cell carcinoma of the oesophagus. Br. J. Cancer 1997, 76, 1081–1086. [Google Scholar] [CrossRef]
- Westin, U.; Nystrom, M.; Ljungcrantz, I.; Eriksson, B.; Ohlsson, K. The presence of elafin, SLPI, IL1-RA and STNFalpha RI in head and neck squamous cell carcinomas and their relation to the degree of tumour differentiation. Mediat. Inflamm. 2002, 11, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.; Weldon, S.; Taggart, C.C. SLPI and elafin: Multifunctional antiproteases of the WFDC family. Biochem. Soc. Trans. 2011, 39, 1437–1440. [Google Scholar] [CrossRef]
- Shaw, L.; Wiedow, O. Therapeutic potential of human elafin. Biochem. Soc. Trans. 2011, 39, 1450–1454. [Google Scholar] [CrossRef]
- Alam, S.R.; Newby, D.E.; Henriksen, P.A. Role of the endogenous elastase inhibitor, elafin, in cardiovascular injury: From epithelium to endothelium. Biochem. Pharmacol. 2012, 83, 695–704. [Google Scholar] [CrossRef]
- Yoshida, N.; Egami, H.; Yamashita, J.; Takai, E.; Tamori, Y.; Fujino, N.; Kitaoka, M.; Schalkwijk, J.; Ogawa, M. Immunohistochemical expression of SKALP/elafin in squamous cell carcinoma of human lung. Oncol. Rep. 2002, 9, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.K.; Chang, W.S.; Wu, I.C.; Li, L.H.; Yang, S.F.; Chen, J.Y.; Hsu, M.C.; Chen, S.H.; Wu, D.C.; Lee, J.M.; et al. Molecular characterization of invasive subpopulations from an esophageal squamous cell carcinoma cell line. Anticancer Res. 2010, 30, 727–736. [Google Scholar]
- Caruso, J.A.; Karakas, C.; Zhang, J.; Yi, M.; Albarracin, C.; Sahin, A.; Bondy, M.; Liu, J.; Hunt, K.K.; Keyomarsi, K. Elafin is downregulated during breast and ovarian tumorigenesis but its residual expression predicts recurrence. Breast Cancer Res. 2014, 16, 3417. [Google Scholar] [CrossRef]
- Labidi-Galy, S.I.; Clauss, A.; Ng, V.; Duraisamy, S.; Elias, K.M.; Piao, H.Y.; Bilal, E.; Davidowitz, R.A.; Lu, Y.; Badalian-Very, G.; et al. Elafin drives poor outcome in high-grade serous ovarian cancers and basal-like breast tumors. Oncogene 2015, 34, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Doberstein, K.; Drapkin, R. Elafin: A double agent in breast and ovarian cancer. Oncoscience 2015, 2, 793–794. [Google Scholar] [CrossRef] [PubMed]
- Sallenave, J.M.; Silva, A. Characterization and gene sequence of the precursor of elafin, an elastase-specific inhibitor in bronchial secretions. Am. J. Respir. Cell Mol. Biol. 1993, 8, 439–445. [Google Scholar] [CrossRef]
- Verrier, T.; Solhonne, B.; Sallenave, J.M.; Garcia-Verdugo, I. The WAP protein Trappin-2/Elafin: A handyman in the regulation of inflammatory and immune responses. Int. J. Biochem. Cell Biol. 2012, 44, 1377–1380. [Google Scholar] [CrossRef] [PubMed]
- O’Blenes, S.B.; Zaidi, S.H.; Cheah, A.Y.; McIntyre, B.; Kaneda, Y.; Rabinovitch, M. Gene transfer of the serine elastase inhibitor elafin protects against vein graft degeneration. Circulation 2000, 102, III289–III295. [Google Scholar] [CrossRef]
- Majchrzak-Gorecka, M.; Majewski, P.; Grygier, B.; Murzyn, K.; Cichy, J. Secretory leukocyte protease inhibitor (SLPI), a multifunctional protein in the host defense response. Cytokine Growth Factor Rev. 2016, 28, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Kramps, J.A.; Klasen, E.C. Characterization of a low molecular weight anti-elastase isolated from human bronchial secretion. Exp. Lung Res. 1985, 9, 151–165. [Google Scholar] [CrossRef]
- Mihaila, A.; Tremblay, G.M. Human alveolar macrophages express elafin and secretory leukocyte protease inhibitor. Zeitschrift Naturforschung C J. Biosci. 2001, 56, 291–297. [Google Scholar] [CrossRef]
- Bouchard, D.; Morisset, D.; Bourbonnais, Y.; Tremblay, G.M. Proteins with whey-acidic-protein motifs and cancer. Lancet Oncol. 2006, 7, 167–174. [Google Scholar] [CrossRef]
- Yokota, T.; Bui, T.; Liu, Y.; Yi, M.; Hunt, K.K.; Keyomarsi, K. Differential regulation of elafin in normal and tumor-derived mammary epithelial cells is mediated by CCAAT/enhancer binding protein beta. Cancer Res. 2007, 67, 11272–11283. [Google Scholar] [CrossRef][Green Version]
- Clauss, A.; Ng, V.; Liu, J.; Piao, H.; Russo, M.; Vena, N.; Sheng, Q.; Hirsch, M.S.; Bonome, T.; Matulonis, U.; et al. Overexpression of elafin in ovarian carcinoma is driven by genomic gains and activation of the nuclear factor kappaB pathway and is associated with poor overall survival. Neoplasia 2010, 12, 161–172. [Google Scholar] [CrossRef]
- Saidi, A.; Javerzat, S.; Bellahcene, A.; De Vos, J.; Bello, L.; Castronovo, V.; Deprez, M.; Loiseau, H.; Bikfalvi, A.; Hagedorn, M. Experimental anti-angiogenesis causes upregulation of genes associated with poor survival in glioblastoma. Int. J. Cancer 2008, 122, 2187–2198. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.S.; Lee, Y.; Kim, C.M.; Park, E.C.; Choi, J.; Lim, D.S.; Chung, Y.H.; Koh, S.S. The protease inhibitor, elafin, induces p53-dependent apoptosis in human melanoma cells. Int. J. Cancer 2010, 127, 1308–1320. [Google Scholar] [CrossRef]
- Alkemade, H.A.; Molhuizen, H.O.; van Vlijmen-Willems, I.M.; van Haelst, U.J.; Schalkwijk, J. Differential expression of SKALP/Elafin in human epidermal tumors. Am. J. Pathol 1993, 143, 1679–1687. [Google Scholar]
- Kunzmann, A.T.; McMenamin, U.C.; Spence, A.D.; Gray, R.T.; Murray, L.J.; Turkington, R.C.; Coleman, H.G. Blood biomarkers for early diagnosis of oesophageal cancer: A systematic review. Eur. J. Gastroenterol. Hepatol. 2018, 30, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Yazbeck, R.; Jaenisch, S.E.; Watson, D.I. From blood to breath: New horizons for esophageal cancer biomarkers. World J. Gastroenterol. 2016, 22, 10077–10083. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hunt, K.K.; Wingate, H.; Yokota, T.; Liu, Y.; Mills, G.B.; Zhang, F.; Fang, B.; Su, C.H.; Zhang, M.; Yi, M.; et al. Elafin, an inhibitor of elastase, is a prognostic indicator in breast cancer. Breast Cancer Res. 2013, 15, R3. [Google Scholar] [CrossRef] [PubMed]
- Caruso, J.A.; Akli, S.; Pageon, L.; Hunt, K.K.; Keyomarsi, K. The serine protease inhibitor elafin maintains normal growth control by opposing the mitogenic effects of neutrophil elastase. Oncogene 2015, 34, 3556–3567. [Google Scholar] [CrossRef]
- Wei, H.; Hellstrom, K.E.; Hellstrom, I. Elafin selectively regulates the sensitivity of ovarian cancer cells to genotoxic drug-induced apoptosis. Gynecol. Oncol. 2012, 125, 727–733. [Google Scholar] [CrossRef] [PubMed][Green Version]
| No. of Patients | n = 119 |
| Characteristics | Mean ± SD (range) or No. (%) |
| Gender | |
| Male | 111 (95.3) |
| Female | 8 (4.6) |
| Age (years) | 57.83 ± 10.49 (38–82) |
| Alcohol | |
| No | 16 (13.4) |
| Yes | 103 (86.6) |
| Smoke | |
| No | 11 (9.2) |
| Yes | 108 (90.8) |
| Betel nut | |
| No | 57 (47.9) |
| Yes | 62(52.1) |
| Stage | |
| I | 17 (14.3) |
| II | 85 (71.4) |
| IIIA | 17 (14.3) |
| Treatment | |
| CCRT + surgery | 48 (40.3) |
| CCRT only | 41 (59.6) |
| Serum elafin level (ng/mL) | 40.14 ± 40.29 (0.59–367.89) |
| Serum Elafin Levels (ng/mL) | Low Elafin (n = 40) 0.59–22.19 | Moderate Elafin (n = 39) 22.19–45.10 | High Elafin (n = 40) 45.10–367.89 | p * |
|---|---|---|---|---|
| Characteristics | Mean ± SD or No. (%) | |||
| Age, years | 54.64 ± 9.43 | 58.09 ± 10.81 | 60.78 ± 10.56 | 0.030 |
| Gender (%, men) | 37 (92.5) | 36 (92.3) | 38 (95.0) | 0.867 |
| Alcohol (%, yes) | 37 (92.5) | 31 (79.4) | 31 (77.5) | 0.232 |
| Smoke (%, yes) | 37 (92.5) | 33 (84.6) | 38 (95.0) | 0.252 |
| Betel nut (%, yes) | 22 (55.0) | 21 (53.8) | 19 (47.5) | 0.770 |
| Stage | 0.006 | |||
| I | 10 (25.0) | 3 (7.7) | 4 (10.0) | |
| II | 20 (50.0) | 31 (79.4) | 34 (85.0) | |
| IIIA | 10 (25.0) | 5 (12.8) | 2 (5.0) | |
| Treatment | <0.001 | |||
| CCRT + surgery | 26 (65.0) | 11 (28.2) | 11 (27.5) | |
| CCRT only | 14 (35.0) | 28 (71.8) | 29 (72.5) | |
| Cut-Off Point | n | Unadjusted | Risk-Adjusted * | Risk-Adjusted ** | |||
|---|---|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | ||
| Tertile | |||||||
| Low | 40 | 1 | 1 | 1 | |||
| Moderate | 39 | 1.88 (1.07, 3.28) | 0.028 | 1.96 * (1.10, 3.47) | 0.022 | 1.88 ** (1.04, 3.37) | 0.035 |
| High | 40 | 2.47 (1.42, 4.29) | 0.001 | 2.22 * (1.24, 3.97) | 0.007 | 2.13 ** (1.06, 3.88) | 0.013 |
| Dichotomize | |||||||
| Low | 40 | 1 | 1 | 1 | |||
| Moderate + High | 79 | 2.14 (1.30, 3.52) | 0.003 | 2.08 * (1.24, 3.47) | 0.005 | 1.99 ** (1.17, 3.38) | 0.011 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, I.-C.; Wang, Y.-K.; Chen, Y.-H.; Wu, C.-C.; Wu, M.-C.; Chen, W.-C.; Wang, W.-L.; Lin, H.-S.; Chen, C.-C.; Chou, S.-H.; et al. High Serum Elafin Prediction of Poor Prognosis of Locoregional Esophageal Squamous Cell Carcinoma. Cancers 2021, 13, 3082. https://doi.org/10.3390/cancers13123082
Wu I-C, Wang Y-K, Chen Y-H, Wu C-C, Wu M-C, Chen W-C, Wang W-L, Lin H-S, Chen C-C, Chou S-H, et al. High Serum Elafin Prediction of Poor Prognosis of Locoregional Esophageal Squamous Cell Carcinoma. Cancers. 2021; 13(12):3082. https://doi.org/10.3390/cancers13123082
Chicago/Turabian StyleWu, I-Chen, Yao-Kuang Wang, Yi-Hsun Chen, Chun-Chieh Wu, Meng-Chieh Wu, Wei-Chung Chen, Wen-Lun Wang, Hung-Shun Lin, Chou-Cheng Chen, Shah-Hwa Chou, and et al. 2021. "High Serum Elafin Prediction of Poor Prognosis of Locoregional Esophageal Squamous Cell Carcinoma" Cancers 13, no. 12: 3082. https://doi.org/10.3390/cancers13123082
APA StyleWu, I.-C., Wang, Y.-K., Chen, Y.-H., Wu, C.-C., Wu, M.-C., Chen, W.-C., Wang, W.-L., Lin, H.-S., Chen, C.-C., Chou, S.-H., Liu, Y.-P., & Wu, M.-T. (2021). High Serum Elafin Prediction of Poor Prognosis of Locoregional Esophageal Squamous Cell Carcinoma. Cancers, 13(12), 3082. https://doi.org/10.3390/cancers13123082







