The Biological and Biomechanical Role of Transglutaminase-2 in the Tumour Microenvironment
Abstract
Simple Summary
Abstract
1. Introduction
2. TG2 and the Hallmarks of Cancer
2.1. Cellular Proliferative Signalling
2.2. Evading Growth Suppressors
2.3. Resisting Cell Death
2.4. Chemoresistance
2.5. Enabling Unlimited Replicative Immortality
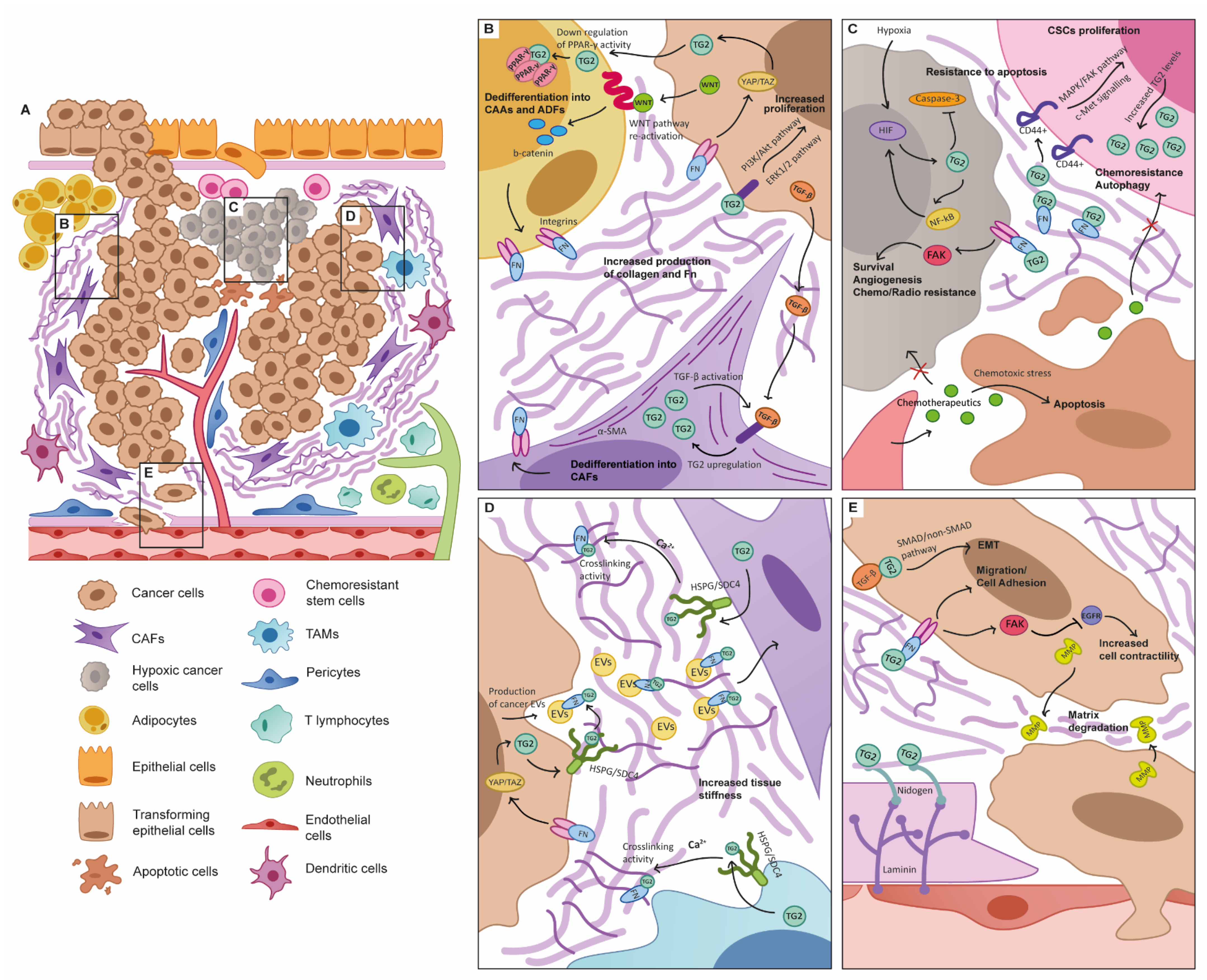
3. TG2 as a Key Functional Player in the TME
3.1. The Tumour Microenvironment
3.2. TG2 and the Cancer-Associated Fibroblast
3.3. TG2 and Adipocyte Function
3.4. TG2 and the Immune System in Cancer
3.5. The Hypoxic TME
3.6. Induction of Angiogenesis
4. TG2 Is Key to the Biomechanical Progression of Cancer
4.1. The Mechanics of Cancer Progression
4.2. Mechanical Forces and Invasion Initiation
4.3. Interaction and Invasion through the Basement Membrane
4.4. Role of TG2 in Interstitial Extracellular Matrix Remodelling
4.5. Role of TG2 in Biomechanical Signalling of TME
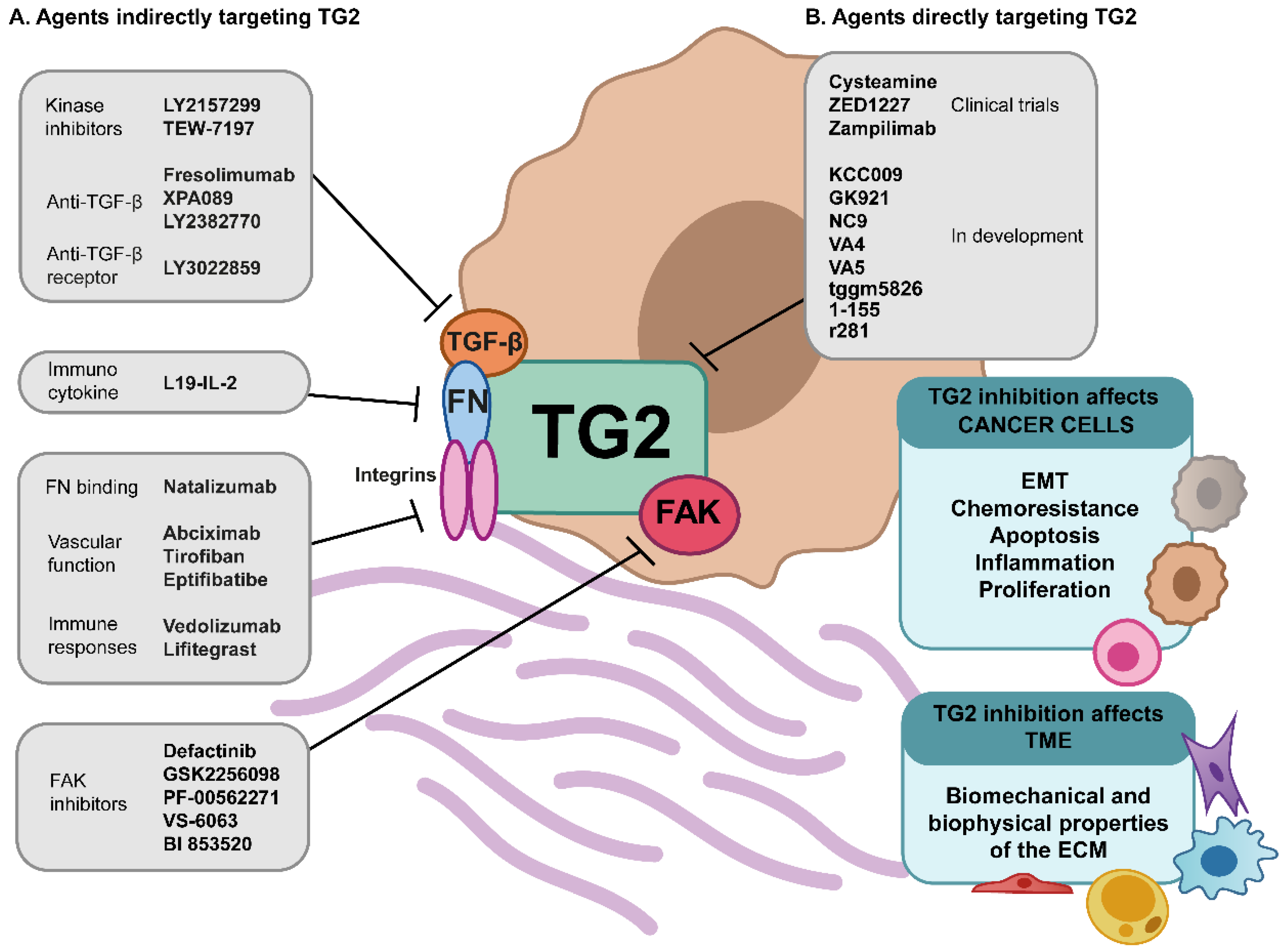
5. TG2—A Stage-Specific Cancer Target
5.1. TG2 and Tumour Metastasis
5.2. Targeting Mechanobiology of TME
5.3. Current State of TG2 Inhibitors
5.4. Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Murthy, S.N.P.; Iismaa, S.; Begg, G.; Freymann, D.M.; Graham, R.M.; Lorand, L. Conserved Tryptophan in the Core Domain of Transglutaminase is Essential for Catalytic Activity. Proc. Natl. Acad. Sci. USA 2002, 99, 2738–2742. [Google Scholar] [CrossRef]
- Király, R.; Demény, M.; Fésüs, L. Protein Transamidation by Transglutaminase 2 in Cells: A Disputed Ca2+-Dependent Action of a Multifunctional Protein. FEBS J. 2011, 278, 4717–4739. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.C.; Gray, C.D.; Sivasubramanian, N.; Im, M.J. Interaction Site of GTP Binding Gh(Transglutaminase II) with Phospholipase C. J. Biol. Chem. 1995, 270, 27058–27062. [Google Scholar] [CrossRef]
- Cerione, R.A.; Liu, S.; Clardy, J. Structural Basis for the Guanine Nucleotide-Binding Activity of Tissue Transglutaminase and its Regulation of Transamidation Activity. Proc. Natl. Acad. Sci. USA 2004, 99, 2743–2747. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. The Hallmarks of Cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Eckert, R.L. Transglutaminase 2 Takes Center Stage as a Cancer Cell Survival Factor and Therapy Target. Mol. Carcinog. 2019, 58, 837–853. [Google Scholar] [CrossRef] [PubMed]
- Pierce, A.; Whetton, A.D.; Meyer, S.; Ravandi-Kashani, F.; Borthakur, G.; Coombes, K.R.; Zhang, N.; Kornblau, S. Transglutaminase 2 Expression in Acute Myeloid Leukemia: Association with Adhesion Molecule Expression and Leukemic Blast Motility. Proteomics 2013, 13, 2216–2224. [Google Scholar] [CrossRef] [PubMed]
- Han, A.L.; Kumar, S.; Fok, J.Y.; Tyagi, A.K.; Mehta, K. Tissue Transglutaminase Expression Promotes Castration-Resistant Phenotype and Transcriptional Repression of Androgen Receptor. Eur. J. Cancer 2014, 50, 1685–1696. [Google Scholar] [CrossRef]
- Huang, L.; Xu, A.; Liu, W. Transglutaminase 2 in Cancer. Am. J. Cancer Res. 2015, 5, 2756–2776. [Google Scholar] [PubMed]
- Park, M.J.; Baek, H.W.; Rhee, Y.; Lee, C.; Park, J.W.; Kim, H.W.; Moon, K.C. Transglutaminase 2 Expression and its Prognostic Significance in Clear Cell Renal Cell Carcinoma. J. Pathol. Transl. Med. 2015, 49, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Huang, C.; Chen, W.; Tsai, C.; Chao, Y.; Liu, S.; Chen, J.; Wu, Y.; Lee, Y. Transglutaminase 2 Promotes Migration and Invasion of Lung Cancer Cells. Oncol. Res. 2018, 26, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Satpathy, M.; Cao, L.; Pincheira, R.; Emerson, R.; Bigsby, R.; Nakshatri, H.; Matei, D. Enhanced Peritoneal Ovarian Tumor Dissemination by Tissue Transglutaminase. Cancer Res. 2007, 67, 7194–7202. [Google Scholar] [CrossRef] [PubMed]
- Ayinde, O.; Wang, Z.; Pinton, G.; Moro, L.; Griffin, M. Transglutaminase 2 Maintains a Colorectal Cancer Stem Phenotype by Regulating Epithelial-Mesenchymal Transition. Oncotarget 2019, 10, 4556–4569. [Google Scholar] [CrossRef]
- Fisher, M.L.; Adhikary, G.; Xu, W.; Kerr, C.; Keillor, J.W.; Eckert, R.L. Type II Transglutaminase Stimulates Epidermal Cancer Stem Cell Epithelial-Mesenchymal Transition. Oncotarget 2015, 6, 20525–20539. [Google Scholar] [CrossRef] [PubMed]
- Zonca, S.; Pinton, G.; Wang, Z.; Soluri, M.F.; Tavian, D.; Griffin, M.; Sblattero, D.; Moro, L. Tissue Transglutaminase (TG2) Enables Survival of Human Malignant Pleural Mesothelioma Cells in Hypoxia. Cell Death Dis. 2017, 8, e2592. [Google Scholar] [CrossRef]
- Verma, A.; Guha, S.; Diagaradjane, P.; Kunnumakkara, A.B.; Sanguino, A.M.; Lopez-Berestein, G.; Sood, A.K.; Aggarwal, B.B.; Krishnan, S.; Gelovani, J.G.; et al. Therapeutic Significance of Elevated Tissue Transglutaminase Expression in Pancreatic Cancer. Clin. Cancer Res. 2008, 14, 2476–2483. [Google Scholar] [CrossRef] [PubMed]
- Gundemir, S.; Monteagudo, A.; Akbar, A.; Keillor, J.W.; Johnson, G.V.W. The Complex Role of Transglutaminase 2 in Glioblastoma Proliferation. Neuro Oncol. 2017, 19, 208–218. [Google Scholar] [CrossRef]
- Tabolacci, C.; de Martino, A.; Mischiati, C.; Feriotto, G.; Beninati, S. The Role of Tissue Transglutaminase in Cancer Cell Initiation, Survival and Progression. Med. Sci. 2019, 7, 19. [Google Scholar] [CrossRef]
- Chhabra, A.; Verma, A.; Mehta, K. Tissue Transglutaminase Promotes or Suppresses Tumors Depending on Cell Context. Anticancer Res. 2009, 29, 1909–1920. [Google Scholar] [PubMed]
- Cao, L.; Shao, M.; Schilder, J.; Guise, T.; Mohammad, K.S.; Matei, D. Tissue Transglutaminase Links TGF-Β, Epithelial to Mesenchymal Transition and a Stem Cell Phenotype in Ovarian Cancer. Oncogene 2011, 31, 2521–2534. [Google Scholar] [CrossRef]
- Fu, J.; Yang, Q.; Sai, K.; Chen, F.; Pang, J.C.S.; Ng, H.; Kwan, A.; Chen, Z. TGM2 Inhibition Attenuates ID1 Expression in CD44-High Glioma-Initiating Cells. Neuro Oncol. 2013, 15, 1353–1365. [Google Scholar] [CrossRef] [PubMed]
- Condello, S.; Cao, L.; Matei, D. Tissue Transglutaminase Regulates Β-catenin Signaling through a c-Src-dependent Mechanism. FASEB J. 2013, 27, 3100–3112. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yu, Z.; Zhou, Q.; Wu, X.; Chen, X.; Li, J.; Zhu, Z.; Liu, B.; Su, L. Tissue Transglutaminase-2 Promotes Gastric Cancer Progression Via the ERK1/2 Pathway. Oncotarget 2016, 7, 7066–7079. [Google Scholar] [CrossRef] [PubMed]
- Boehm, J.E.; Singh, U.; Combs, C.; Antonyak, M.A.; Cerione, R.A. Tissue Transglutaminase Protects Against Apoptosis by Modifying the Tumor Suppressor Protein p110 Rb. J. Biol. Chem. 2002, 277, 20127–20130. [Google Scholar] [CrossRef] [PubMed]
- Oliverio, S.; Amendola, A.; Di Sano, F.; Farrace, M.G.; Fesus, L.; Nemes, Z.; Piredda, L.; Spinedi, A.; Piacentini, M. Tissue Transglutaminase-Dependent Posttranslational Modification of the Retinoblastoma Gene Product in Promonocytic Cells Undergoing Apoptosis. Mol. Cell. Biol. 1997, 17, 6040–6048. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Murphy, L.J. The p53 Oncoprotein is a Substrate for Tissue Transglutaminase Kinase Activity. Biochem. Biophys. Res. Commun. 2006, 339, 726–730. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Wang, H.-G. Tissue Transglutaminase Serves as an Inhibitor of Apoptosis by Cross-Linking Caspase 3 in Thapsigargin-Treated Cells. Mol. Cell. Biol. 2005, 26, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Lee, J.; Bae, H.; Jeong, E.M.; Jang, G.; Kim, C.; Shin, D.; Jeon, J.; Kim, I. Transglutaminase 2 Inhibits Apoptosis Induced by Calcium-Overload through Down-Regulation of Bax. Exp. Mol. Med. 2010, 42, 639–650. [Google Scholar] [CrossRef]
- Jang, G.; Jeon, J.; Cho, S.; Shin, D.; Kim, C.; Jeong, E.M.; Bae, H.C.; Kim, T.W.; Lee, S.; Choi, Y.; et al. Transglutaminase 2 Suppresses Apoptosis by Modulating Caspase 3 and NF-kappaB Activity in Hypoxic Tumor Cells. Oncogene 2010, 29, 356–367. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.; Lim, Y.; Kim, Y.; Ha, K. Transglutaminase 2 Promotes both Caspase-Dependent and Caspase-Independent Apoptotic Cell Death Via the Calpain/Bax Protein Signaling Pathway. J. Biol. Chem. 2012, 287, 14377–14388. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xu, X.; Bai, L.; Chen, W.; Lin, Y. Epidermal Growth Factor Receptor-Mediated Tissue Transglutaminase Overexpression Couples Acquired Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand Resistance and Migration through C-FLIP and MMP-9 Proteins in Lung Cancer Cells. J. Biol. Chem. 2011, 286, 21164–21172. [Google Scholar] [CrossRef]
- Frese-Schaper, M.; Schardt, J.A.; Sakai, T.; Carboni, G.L.; Schmid, R.A.; Frese, S. Inhibition of Tissue Transglutaminase Sensitizes TRAIL-Resistant Lung Cancer Cells through Upregulation of Death Receptor 5. FEBS Lett. 2010, 584, 2867–2871. [Google Scholar] [CrossRef]
- Eckert, R.L.; Fisher, M.L.; Grun, D.; Adhikary, G.; Xu, W.; Kerr, C. Transglutaminase is a Tumor Cell and Cancer Stem Cell Survival Factor. Mol. Carcinog. 2015, 54, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, N.; Kumar, S.; Mehta, K. Tissue Transglutaminase as a Central Mediator in Inflammation-Induced Progression of Breast Cancer. Breast Cancer Res. 2013, 15, 202. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Xu, J.; Brady, S.; Gao, H.; Yu, D.; Reuben, J.; Mehta, K. Tissue Transglutaminase Promotes Drug Resistance and Invasion by Inducing Mesenchymal Transition in Mammary Epithelial Cells. PLoS ONE 2010, 5, e13390. [Google Scholar] [CrossRef]
- Kumar, A.; Gao, H.; Xu, J.; Reuben, J.; Yu, D.; Mehta, K. Evidence that Aberrant Expression of Tissue Transglutaminase Promotes Stem Cell Characteristics in Mammary Epithelial Cells. PLoS ONE 2011, 6, e20701. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.L.; Kerr, C.; Adhikary, G.; Grun, D.; Xu, W.; Keillor, J.W.; Eckert, R.L. Transglutaminase Interaction with α6/β4-Integrin Stimulates YAP1-Dependent ΔNp63α Stabilization and Leads to Enhanced Cancer Stem Cell Survival and Tumor Formation. Cancer Res. 2016, 76, 7265–7276. [Google Scholar] [CrossRef]
- Fisher, M.L.; Adhikary, G.; Kerr, C.; Grun, D.; Eckert, R.L. Transglutaminase 2 Is a Direct Target Gene of YAP-TAZ-Response. Cancer Res. 2017, 77, 4736. [Google Scholar] [CrossRef]
- Wang, Z.; Perez, M.; Caja, S.; Melino, G.; Johnson, T.S.; Lindfors, K.; Griffin, M. A Novel Extracellular Role for Tissue Transglutaminase in Matrix-Bound VEGF-Mediated Angiogenesis. Cell Death Dis. 2013, 4, e808. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kang, J.H.; Ha, J.S.; Lee, J.; Oh, S.; Choi, H.; Song, J.; Kim, S. Transglutaminase 2-Mediated p53 Depletion Promotes Angiogenesis by Increasing HIF-1α-p300 Binding in Renal Cell Carcinoma. Int. J. Mol. Sci. 2020, 21, 5042. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Chai, N.; Tian, M.; Zhang, Y.; Wang, G.; Liu, J.; Tian, Z.; Yi, X.; Chen, D.; Li, X.; et al. Novel Peptide GX1 Inhibits Angiogenesis by Specifically Binding to Transglutaminase-2 in the Tumorous Endothelial Cells of Gastric Cancer. Cell Death Dis. 2018, 9, 579. [Google Scholar] [CrossRef] [PubMed]
- Steppan, J.; Bergman, Y.; Viegas, K.; Armstrong, D.; Tan, S.; Wang, H.; Melucci, S.; Hori, D.; Park, S.Y.; Barreto, S.F.; et al. Tissue Transglutaminase Modulates Vascular Stiffness and Function through Crosslinking-Dependent and Crosslinking-Independent Functions. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Antonyak, M.A.; Li, B.; Regan, A.D.; Feng, Q.; Dusaban, S.S.; Cerione, R.A. Tissue Transglutaminase is an Essential Participant in the Epidermal Growth Factor-Stimulated Signaling Pathway Leading to Cancer Cell Migration and Invasion. J. Biol. Chem. 2009, 284, 17914–17925. [Google Scholar] [CrossRef]
- Bagatur, Y.; Ilter Akulke, A.Z.; Bihorac, A.; Erdem, M.; Telci, D. Tissue Transglutaminase Expression is Necessary for Adhesion, Metastatic Potential and Cancer Stemness of Renal Cell Carcinoma. Cell Adhes. Migr. 2017, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tabolacci, C.; Lentini, A.; Mattioli, P.; Provenzano, B.; Oliverio, S.; Carlomosti, F.; Beninati, S. Antitumor Properties of Aloe-Emodin and Induction of Transglutaminase 2 Activity in B16–F10 Melanoma Cells. Life Sci. 2010, 87, 316–324. [Google Scholar] [CrossRef]
- Cellura, D.; Pickard, K.; Quaratino, S.; Parker, H.; Strefford, J.C.; Thomas, G.J.; Mitter, R.; Mirnezami, A.H.; Peake, N.J. miR-19-Mediated Inhibition of Transglutaminase-2 Leads to Enhanced Invasion and Metastasis in Colorectal Cancer. Mol. Cancer Res. 2015, 13, 1095–1105. [Google Scholar] [CrossRef] [PubMed]
- Feitelson, M.A.; Arzumanyan, A.; Kulathinal, R.J.; Blain, S.W.; Holcombe, R.F.; Mahajna, J.; Marino, M.; Martinez-Chantar, M.L.; Nawroth, R.; Sanchez-Garcia, I.; et al. Sustained Proliferation in Cancer: Mechanisms and Novel Therapeutic Targets. Semin. Cancer Biol. 2015, 35, S25–S54. [Google Scholar] [CrossRef] [PubMed]
- Ikushima, H.; Miyazono, K. TGFβ Signalling: A Complex Web in Cancer Progression. Nat. Rev. Cancer 2010, 10, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Gutschner, T.; Diederichs, S. The Hallmarks of Cancer: A Long Non-Coding RNA Point of View. RNA Biol. 2012, 9, 703–719. [Google Scholar] [CrossRef]
- Lowe, S.W.; Evan, G.; Cepero, E. Intrinsic Tumour Suppression. Nature 2004, 432, 307–315. [Google Scholar] [CrossRef]
- Zhu, L.; Lu, Z.; Zhao, H. Antitumor Mechanisms when pRb and p53 are Genetically Inactivated. Oncogene 2014, 34, 4547–4557. [Google Scholar] [CrossRef]
- Sherr, C.J. Principles of Tumor Suppression. Cell 2004, 116, 235–246. [Google Scholar] [CrossRef]
- Giacinti, C.; Giordano, A. RB and Cell Cycle Progression. Oncogene 2006, 25, 5220–5227. [Google Scholar] [CrossRef] [PubMed]
- Budanov, A.V. The role of tumor suppressor p53 in the antioxidant defense and metabolism. Subcell. Biochem. 2014, 85, 337–358. [Google Scholar] [CrossRef] [PubMed]
- Burkhart, D.L.; Sage, J. Cellular Mechanisms of Tumour Suppression by the Retinoblastoma Gene. Nat. Rev. Cancer 2008, 8, 671–682. [Google Scholar] [CrossRef]
- Shi, D.; Gu, W. Dual Roles of MDM2 in the Regulation of p53: Ubiquitination Dependent and Ubiquitination Independent Mechanisms of MDM2 Repression of p53 Activity. Genes Cancer 2012, 3, 240–248. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as Anticancer Mechanism: Function and Dysfunction of its Modulators and Targeted Therapeutic Strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef]
- Roché, H.; Vahdat, L.T. Treatment of Metastatic Breast Cancer: Second Line and Beyond. Ann. Oncol. 2011, 22, 1000–1010. [Google Scholar] [CrossRef]
- Cummings, M. Apoptosis of Epithelial Cells in Vivo Involves Tissue Transglutaminase Upregulation. J. Pathol. 1996, 179, 288–293. [Google Scholar] [CrossRef]
- Piacentini, M.; D’Eletto, M.; Falasca, L.; Farrace, M.G.; Rodolfo, C. Transglutaminase 2 at the Crossroads between Cell Death and Survival. Adv. Enzymol. Relat. Subj. 2011, 78, 197–246. [Google Scholar]
- Fok, J.Y.; Mehta, K. Tissue Transglutaminase Induces the Release of Apoptosis Inducing Factor and Results in Apoptotic Death of Pancreatic Cancer Cells. Apoptosis 2007, 12, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Oliverio, S.; Amendola, A.; Rodolfo, C.; Spinedi, A.; Piacentini, M. Inhibition of “Tissue” Transglutaminase Increases Cell Survival by Preventing Apoptosis. J. Biol. Chem. 1999, 274, 34123–34128. [Google Scholar] [CrossRef][Green Version]
- Tatsukawa, H.; Fukaya, Y.; Frampton, G.; Martinez–Fuentes, A.; Suzuki, K.; Kuo, T.; Nagatsuma, K.; Shimokado, K.; Okuno, M.; Wu, J.; et al. Role of Transglutaminase 2 in Liver Injury Via Cross-Linking and Silencing of Transcription Factor Sp1. Gastroenterology 2009, 136, 1783–1795. [Google Scholar] [CrossRef]
- Antonyak, M.A.; Jansen, J.M.; Miller, A.M.; Ly, T.K.; Endo, M.; Cerione, R.A. Two Isoforms of Tissue Transglutaminase Mediate Opposing Cellular Fates. Proc. Natl. Acad. Sci. USA 2006, 103, 18609–18614. [Google Scholar] [CrossRef]
- Milakovic, T.; Tucholski, J.; McCoy, E.; Johnson, G.V.W. Intracellular Localization and Activity State of Tissue Transglutaminase Differentially Impacts Cell Death. J. Biol. Chem. 2004, 279, 8715–8722. [Google Scholar] [CrossRef] [PubMed]
- Fabbi, M.; Marimpietri, D.; Martini, S.; Brancolini, C.; Amoresano, A.; Scaloni, A.; Bargellesi, A.; Cosulich, E. Tissue Transglutaminase is a Caspase Substrate during Apoptosis. Cleavage Causes Loss of Transamidating Function and is a Biochemical Marker of Caspase 3 Activation. Cell Death Differ. 1999, 6, 992–1001. [Google Scholar] [CrossRef] [PubMed]
- Lai, T.; Liu, Y.; Li, W.; Greenberg, C.S. Identification of two GTP-independent alternatively spliced forms of tissue transglutaminase in human leukocytes, vascular smooth muscle, and endothelial cells. FASEB J. 2007, 21, 4131–4143. [Google Scholar] [CrossRef]
- Eligini, S.; Fiorelli, S.; Tremoli, E.; Colli, S. Inhibition of Transglutaminase 2 Reduces Efferocytosis in Human Macrophages: Role of CD14 and SR-AI Receptors. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Yen, J.; Yang, D.; Chen, M.; Yi-Ying, W.; Hsieh, Y.; Cheng, Y.; Huang, W.; Szondy, Z.; Tsay, G.J. Corrigendum to “Daidzein Enhances Efferocytosis Via Transglutaminase 2 and Augmentation of Rac1 Activity” [Mol. Immunol. 2014, 60, 135–142]. Mol. Immunol. 2015, 63, 605. [Google Scholar] [CrossRef]
- Nicholas, B.; Smethurst, P.; Verderio, E.; Jones, R.; Griffin, M. Cross-Linking of Cellular Proteins by Tissue Transglutaminase during Necrotic Cell Death: A Mechanism for Maintaining Tissue Integrity. Biochem. J. 2003, 371, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Abdulghani, J.; El-Deiry, W.S. TRAIL Receptor Signaling and Therapeutics. Expert Opin. Ther. Targets 2010, 14, 1091–1108. [Google Scholar] [CrossRef] [PubMed]
- Mehta, K. High Levels of Transglutaminase Expression in Doxorubicin-resistant Human Breast Carcinoma Cells. Int. J. Cancer 1994, 58, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Boroughs, L.K.; Antonyak, M.A.; Cerione, R.A. A novel Mechanism by which Tissue Transglutaminase Activates Signaling Events that Promote Cell Survival. J. Biol. Chem. 2014, 289, 10115–10125. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Huang, W. Compensatory Increase of Transglutaminase 2 is Responsible for Resistance to mTOR Inhibitor Treatment. PLoS ONE 2016, 11, e0149388. [Google Scholar] [CrossRef] [PubMed]
- Carbone, C.; Carbone, C.; Di Gennaro, E.; di Gennaro, E.; Piro, G.; Piro, G.; Milone, M.; Milone, M.; Pucci, B.; Pucci, B.; et al. Tissue Transglutaminase (TG2) is Involved in the Resistance of Cancer Cells to the Histone Deacetylase (HDAC) Inhibitor Vorinostat. Amino Acids 2017, 49, 517–528. [Google Scholar] [CrossRef]
- Glick, D.; Barth, S.; Macleod, K.F. Autophagy: Cellular and Molecular Mechanisms. J. Pathol. 2010, 221, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.; Lee, S. The Roles of Autophagy in Cancer. Int. J. Mol. Sci. 2018, 19, 3466. [Google Scholar] [CrossRef]
- Jung, H.J.; Chen, Z.; Wang, M.; Fayad, L.; Romaguera, J.; Kwak, L.W.; McCarty, N. Calcium Blockers Decrease the Bortezomib Resistance in Mantle Cell Lymphoma Via Manipulation of Tissue Transglutaminase Activities. Blood 2012, 119, 2568–2578. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, Z.; Miranda, R.N.; Medeiros, L.J.; McCarty, N. TG2 and NF-κB Signaling Coordinates the Survival of Mantle Cell Lymphoma Cells Via IL6-Mediated Autophagy. Cancer Res. 2016, 76, 6410–6423. [Google Scholar] [CrossRef]
- Luciani, A.; Villella, V.R.; Esposito, S.; Brunetti-Pierri, N.; Medina, D.; Settembre, C.; Gavina, M.; Pulze, L.; Giardino, I.; Pettoello-Mantovani, M.; et al. Defective CFTR Induces Aggresome Formation and Lung Inflammation in Cystic Fibrosis through ROS-Mediated Autophagy Inhibition. Nat. Cell Biol. 2010, 12, 863–875. [Google Scholar] [CrossRef]
- Chaudhari, N.; Talwar, P.; Parimisetty, A.; d’Hellencourt, L.C.; Ravanan, P. A Molecular Web: Endoplasmic Reticulum Stress, Inflammation, and Oxidative Stress. Front. Cell. Neurosci. 2014, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K. Reactive Oxygen Species (ROS) and Cancer: Role of Antioxidative Nutraceuticals. Cancer Lett. 2017, 387, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Hayflick, L.; Moorhead, P.S. The Serial Cultivation of Human Diploid Cell Strains. Exp. Cell Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Gilmore, A.P. Anoikis. Cell Death Differ. 2005, 12, 1473–1477. [Google Scholar] [CrossRef] [PubMed]
- Frisch, S.M.; Schaller, M.; Cieply, B. Mechanisms that Link the Oncogenic Epithelial-Mesenchymal Transition to Suppression of Anoikis. J. Cell Sci. 2013, 126, 21–29. [Google Scholar] [CrossRef]
- Verderio, E.A.M.; Telci, D.; Okoye, A.; Melino, G.; Griffin, M. A Novel RGD-Independent Cell Adhesion Pathway Mediated by Fibronectin-Bound Tissue Transglutaminase Rescues Cells from Anoikis. J. Biol. Chem. 2003, 278, 42604–42614. [Google Scholar]
- Thapa, R.; Wilson, G.D. The Importance of CD44 as a Stem Cell Biomarker and Therapeutic Target in Cancer. Stem Cells Int. 2016, 2016, 1–15. [Google Scholar] [CrossRef]
- Chen, C.; Zhao, S.; Karnad, A.; Freeman, J.W. The Biology and Role of CD44 in Cancer Progression: Therapeutic Implications. J. Hematol. Oncol. 2018, 11, 64. [Google Scholar] [CrossRef]
- Oh, K.; Lee, O.; Park, Y.; Seo, M.W.; Lee, D. IL-1β Induces IL-6 Production and Increases Invasiveness and Estrogen-Independent Growth in a TG2-Dependent Manner in Human Breast Cancer Cells. BMC Cancer 2016, 16, 724. [Google Scholar] [CrossRef]
- Condello, S.; Sima, L.; Ivan, C.; Cardenas, H.; Schiltz, G.; Mishra, R.K.; Matei, D. Tissue Tranglutaminase Regulates Interactions between Ovarian Cancer Stem Cells and the Tumor Niche. Cancer Res. 2018, 78, 2990–3001. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Bian, S.; Zhou, X.; Cui, Y.; Wang, W.; Wen, L.; Guo, L.; Fu, W.; Tang, F. Single-Cell Multiomics Sequencing Reveals Prevalent Genomic Alterations in Tumor Stromal Cells of Human Colorectal Cancer. Cancer Cell 2020, 38, 818–828. [Google Scholar] [CrossRef]
- Eng, C.; Leone, G.; Orloff, M.S.; Ostrowski, M.C. Genomic Alterations in Tumor Stroma. Cancer Res. 2009, 69, 6759–6764. [Google Scholar] [CrossRef]
- Jakubek, Y.A.; Chang, K.; Sivakumar, S.; Yu, Y.; Giordano, M.R.; Fowler, J.; Huff, C.D.; Kadara, H.; Vilar, E.; Scheet, P. Large-Scale Analysis of Acquired Chromosomal Alterations in Non-Tumor Samples from Patients with Cancer. Nat. Biotechnol. 2020, 38, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A.; Pollard, J.W. Microenvironmental Regulation of Metastasis. Nat. Rev. Cancer 2009, 9, 239–252. [Google Scholar] [CrossRef]
- Delaine-Smith, R.; Wright, N.; Hanley, C.; Hanwell, R.; Bhome, R.; Bullock, M.; Drifka, C.; Eliceiri, K.; Thomas, G.; Knight, M.; et al. Transglutaminase-2 Mediates the Biomechanical Properties of the Colorectal Cancer Tissue Microenvironment that Contribute to Disease Progression. Cancers 2019, 11, 701. [Google Scholar] [CrossRef] [PubMed]
- Assi, J.; Srivastava, G.; Matta, A.; Chang, M.C.; Walfish, P.G.; Ralhan, R. Transglutaminase 2 Overexpression in Tumor Stroma Identifies Invasive Ductal Carcinomas of Breast at High Risk of Recurrence. PLoS ONE 2013, 8, e74437. [Google Scholar] [CrossRef]
- Fernández-Aceñero, M.J.; Torres, S.; Garcia-Palmero, I.; Díaz Del Arco, C.; Casal, J.I. Prognostic role of tissue transglutaminase 2 in colon carcinoma. Virchows Arch. 2016, 469, 611–619. [Google Scholar] [CrossRef]
- Wu, J.; Xu, C.; Guan, X.; Ni, D.; Yang, X.; Yang, Z.; Wang, M. Comprehensive analysis of tumor microenvironment and identification of an immune signature to predict the prognosis and immunotherapeutic response in lung squamous cell carcinoma. Ann. Transl. Med. 2021, 9, 569. [Google Scholar] [CrossRef]
- Stephens, P.; Grenard, P.; Aeschlimann, P.; Langley, M.; Blain, E.; Errington, R.; Kipling, D.; Thomas, D.; Aeschlimann, D. Crosslinking and G-Protein Functions of Transglutaminase 2 Contribute Differentially to Fibroblast Wound Healing Responses. J. Cell Sci. 2004, 117, 3389–3403. [Google Scholar] [CrossRef] [PubMed]
- Upchurch, H.F.; Conway, E.; Patterson, M.K., Jr.; Maxwell, M.D. Localization of cellular transglutaminase on the extracellular matrix after wounding: Characteristics of the matrix bound enzyme. J. Cell. Physiol. 1991, 149, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Haroon, Z.A.; Hettasch, J.M.; Lai, T.S.; Dewhirst, M.W.; Greenberg, C.S. Tissue transglutaminase is expressed, active, and directly involved in rat dermal wound healing and angiogenesis. FASEB J. 1999, 13, 1787–1795. [Google Scholar] [CrossRef]
- Fell, S.; Wang, Z.; Blanchard, A.; Nanthakumar, C.; Griffin, M. Transglutaminase 2: A novel therapeutic target for idiopathic pulmonary fibrosis using selective small molecule inhibitors. Amino Acids 2021, 53, 205–217. [Google Scholar] [CrossRef]
- Torres, S.; Garcia-Palmero, I.; Herrera, M.; Bartolomé, R.A.; Peña, C.; Fernandez-Aceñero, M.J.; Padilla, G.; Peláez-García, A.; Lopez-Lucendo, M.; Rodriguez-Merlo, R.; et al. LOXL2 is Highly Expressed in Cancer-Associated Fibroblasts and Associates to Poor Colon Cancer Survival. Clin. Cancer Res. 2015, 21, 4892–4902. [Google Scholar] [CrossRef] [PubMed]
- Hanley, C.J.; Waise, S.; Parker, R.; Lopez, M.A.; Taylor, J.; Kimbley, L.M.; West, J.; Ottensmeier, C.H.; Rose-Zerilli, M.J.J.; Thomas, G.J. Spatially Discrete Signalling Niches Regulate Fibroblast Heterogeneity in Human Lung Cancer. bioRxiv 2020, 134270. [Google Scholar] [CrossRef]
- Elyada, E.; Bolisetty, M.; Laise, P.; Flynn, W.F.; Courtois, E.T.; Burkhart, R.A.; Teinor, J.A.; Belleau, P.; Biffi, G.; Lucito, M.S.; et al. Cross-Species Single-Cell Analysis of Pancreatic Ductal Adenocarcinoma Reveals Antigen-Presenting Cancer-Associated Fibroblasts. Cancer Discov. 2019, 9, 1102–1123. [Google Scholar] [CrossRef]
- Öhlund, D.; Handly-Santana, A.; Biffi, G.; Elyada, E.; Almeida, A.S.; Ponz-Sarvise, M.; Corbo, V.; Oni, T.E.; Hearn, S.A.; Lee, E.J.; et al. Distinct Populations of Inflammatory Fibroblasts and Myofibroblasts in Pancreatic Cancer. J. Exp. Med. 2017, 214, 579–596. [Google Scholar] [CrossRef] [PubMed]
- Mellone, M.; Hanley, C.J.; Thirdborough, S.; Mellows, T.; Garcia, E.; Woo, J.; Tod, J.; Frampton, S.; Jenei, V.; Moutasim, K.A.; et al. Induction of Fibroblast Senescence Generates a Non-Fibrogenic Myofibroblast Phenotype that Differentially Impacts on Cancer Prognosis. Aging 2016, 9, 114–132. [Google Scholar] [CrossRef]
- Hariton, F.; Xue, M.; Rabbani, N.; Fowler, M.; Thornalley, P.J. Sulforaphane Delays Fibroblast Senescence by Curbing Cellular Glucose Uptake, Increased Glycolysis, and Oxidative Damage. Oxidative Med. Cell. Longev. 2018, 2018, 5642148. [Google Scholar] [CrossRef]
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.; Hingorani, S.R.; Hunter, T.; et al. A Framework for Advancing our Understanding of Cancer-Associated Fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186. [Google Scholar] [CrossRef]
- Lewis, M.P.; Lygoe, K.A.; Nystrom, M.L.; Anderson, W.P.; Speight, P.M.; Marshall, J.F.; Thomas, G.J. Tumour-Derived TGF-Beta1 Modulates Myofibroblast Differentiation and Promotes HGF/SF-Dependent Invasion of Squamous Carcinoma Cells. Br. J. Cancer 2004, 90, 822–832. [Google Scholar] [CrossRef] [PubMed]
- Zeisberg, E.M.; Tarnavski, O.; Zeisberg, M.; Dorfman, A.L.; McMullen, J.R.; Gustafsson, E.; Chandraker, A.; Yuan, X.; Pu, W.T.; Roberts, A.B.; et al. Endothelial-to-Mesenchymal Transition Contributes to Cardiac Fibrosis. Nat. Med. 2007, 13, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Cirri, P.; Chiarugi, P. Cancer Associated Fibroblasts: The Dark Side of the Coin. Am. J. Cancer Res. 2011, 1, 482–497. [Google Scholar]
- Grauel, A.L.; Nguyen, B.; Ruddy, D.; Laszewski, T.; Schwartz, S.; Chang, J.; Chen, J.; Piquet, M.; Pelletier, M.; Yan, Z.; et al. TGFβ-Blockade Uncovers Stromal Plasticity in Tumors by Revealing the Existence of a Subset of Interferon-Licensed Fibroblasts. Nat. Commun. 2020, 11, 6315. [Google Scholar] [CrossRef]
- George, M.D.; Vollberg, T.M.; Floyd, E.E.; Stein, J.P.; Jetten, A.M. Regulation of Transglutaminase Type II by Transforming Growth Factor-Beta 1 in Normal and Transformed Human Epidermal Keratinocytes. J. Biol. Chem. 1990, 265, 11098–11104. [Google Scholar] [CrossRef]
- Verderio, E.; Gaudry, C.; Gross, S.; Smith, C.; Downes, S.; Griffin, M. Regulation of Cell Surface Tissue Transglutaminase: Effects on Matrix Storage of Latent Transforming Growth Factor-Β Binding Protein-1. J. Histochem. Cytochem. 1999, 47, 1417–1432. [Google Scholar] [CrossRef]
- Nunes, I.; Gleizes, P.; Metz, C.N.; Rifkin, D.B. Latent Transforming Growth Factor-Β Binding Protein Domains Involved in Activation and Transglutaminase-Dependent Cross-Linking of Latent Transforming Growth Factor-B. J. Cell Biol. 1997, 136, 1151–1163. [Google Scholar] [CrossRef]
- Jia, C.; Wang, G.; Wang, T.; Fu, B.; Zhang, Y.; Huang, L.; Deng, Y.; Chen, G.; Wu, X.; Chen, J.; et al. Cancer-Associated Fibroblasts Induce Epithelial-Mesenchymal Transition Via the Transglutaminase 2-Dependent IL-6/IL6R/STAT3 Axis in Hepatocellular Carcinoma. Int. J. Biol. Sci. 2020, 16, 2542–2558. [Google Scholar] [CrossRef] [PubMed]
- Saghy, T.; Koroskenyi, K.; Hegedus, K.; Antal, M.; Banko, C.; Bacso, Z.; Papp, A.; Stienstra, R.; Szondy, Z. Loss of Transglutaminase 2 Sensitizes for Diet-Induced Obesity-Related Inflammation and Insulin Resistance due to Enhanced Macrophage C-Src Signaling. Cell Death Dis. 2019, 10, 439. [Google Scholar] [CrossRef] [PubMed]
- Penumatsa, K.C.; Falcão-Pires, I.; Leite, S.; Leite-Moreira, A.; Bhedi, C.D.; Nasirova, S.; Ma, J.; Sutliff, R.L.; Fanburg, B.L. Increased Transglutaminase 2 Expression and Activity in Rodent Models of Obesity/Metabolic Syndrome and Aging. Front. Physiol. 2020, 11, 560019. [Google Scholar] [CrossRef]
- Zhao, C.; Wu, M.; Zeng, N.; Xiong, M.; Hu, W.; Lv, W.; Yi, Y.; Zhang, Q.; Wu, Y. Cancer-Associated Adipocytes: Emerging Supporters in Breast Cancer. J. Exp. Clin. Cancer Res. 2020, 39, 1–156. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Liang, Y.; Xing, C.; Wang, H.; Hu, P.; Li, J.; Huang, H.; Wang, W.; Jiang, C. Cancer-associated Adipocytes Exhibit Distinct Phenotypes and Facilitate Tumor Progression in Pancreatic Cancer. Oncol. Rep. 2019, 42, 2537–2549. [Google Scholar] [CrossRef]
- Wen, Y.; Xing, X.; Harris, J.W.; Zaytseva, Y.Y.; Mitov, M.I.; Napier, D.L.; Weiss, H.L.; Mark Evers, B.; Gao, T. Adipocytes Activate Mitochondrial Fatty Acid Oxidation and Autophagy to Promote Tumor Growth in Colon Cancer. Cell Death Dis. 2017, 8, e2593. [Google Scholar] [CrossRef]
- Bochet, L.; Lehuede, C.; Dauvillier, S.; Wang, Y.Y.; Dirat, B.; Laurent, V.; Dray, C.; Guiet, R.; Maridonneau-Parini, I.; Le Gonidec, S.; et al. Adipocyte-Derived Fibroblasts Promote Tumor Progression and Contribute to the Desmoplastic Reaction in Breast Cancer. Cancer Res. 2013, 73, 5657–5668. [Google Scholar] [CrossRef]
- Myneni, V.D.; Melino, G.; Kaartinen, M.T. Transglutaminase 2-a Novel Inhibitor of Adipogenesis. Cell Death Dis. 2015, 6, e1868. [Google Scholar] [CrossRef]
- Maiuri, L.; Luciani, A.; Giardino, I.; Raia, V.; Villella, V.R.; D’Apolito, M.; Pettoello-Mantovani, M.; Guido, S.; Ciacci, C.; Cimmino, M.; et al. Tissue Transglutaminase Activation Modulates Inflammation in Cystic Fibrosis Via PPARgamma Down-Regulation. J. Immunol. 2008, 180, 7697–7705. [Google Scholar] [CrossRef] [PubMed]
- Klöck, C.; DiRaimondo, T.; Khosla, C. Role of Transglutaminase 2 in Celiac Disease Pathogenesis. Semin. Immunopathol. 2012, 34, 513–522. [Google Scholar] [CrossRef]
- Johnson, T.S.; Fisher, M.; Haylor, J.L.; Hau, Z.; Skill, N.J.; Jones, R.; Saint, R.; Coutts, I.; Vickers, M.E.; El Nahas, A.M.; et al. Transglutaminase Inhibition Reduces Fibrosis and Preserves Function in Experimental Chronic Kidney Disease. J. Am. Soc. Nephrol. 2007, 18, 3078–3088. [Google Scholar] [CrossRef] [PubMed]
- Su, T.; Qin, X.; Furutani, Y. Transglutaminase 2 as a Marker for Inflammation and Therapeutic Target in Sepsis. Int. J. Mol. Sci. 2021, 22, 1897. [Google Scholar] [CrossRef] [PubMed]
- Bijli, K.; Kanter, B.; Minhajuddin, M.; Leonard, A.; Xu, L.; Fazal, F.; Rahman, A. Regulation of Endothelial Cell Inflammation and Lung Polymorphonuclear Lymphocyte Infiltration by Transglutaminase 2. Shock 2014, 42, 562–569. [Google Scholar] [CrossRef]
- Lee, J.; Kim, Y.; Choi, D.; Bang, M.S.; Han, T.R.; Joh, T.H.; Kim, S. Transglutaminase 2 Induces Nuclear Factor-kappaB Activation Via a Novel Pathway in BV-2 Microglia. J. Biol. Chem. 2004, 279, 53725–53735. [Google Scholar] [CrossRef] [PubMed]
- Mirza, A.; Liu, S.L.; Frizell, E.; Zhu, J.; Maddukuri, S.; Martinez, J.; Davies, P.; Schwarting, R.; Norton, P.; Zern, M.A. A Role for Tissue Transglutaminase in Hepatic Injury and Fibrogenesis, and its Regulation by NF-kappaB. Am. J. Physiol. Gastrointest. Liver Physiol. 1997, 272, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Masjedi, A.; Hashemi, V.; Hojjat-Farsangi, M.; Ghalamfarsa, G.; Azizi, G.; Yousefi, M.; Jadidi-Niaragh, F. The Significant Role of Interleukin-6 and its Signaling Pathway in the Immunopathogenesis and Treatment of Breast Cancer. Biomed. Pharmacother. 2018, 108, 1415–1424. [Google Scholar] [CrossRef]
- Oh, K.; Moon, H.; Lee, D.; Yoo, Y. Tissue Transglutaminase-Interleukin-6 Axis Facilitates Peritoneal Tumor Spreading and Metastasis of Human Ovarian Cancer Cells. Lab. Anim. Res. 2015, 31, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Bharti, R.; Dey, G.; Mandal, M. Cancer Development, Chemoresistance, Epithelial to Mesenchymal Transition and Stem Cells: A Snapshot of IL-6 Mediated Involvement. Cancer Lett. 2016, 375, 51–61. [Google Scholar] [CrossRef]
- Gonzalez, H.; Hagerling, C.; Werb, Z. Roles of the Immune System in Cancer: From Tumor Initiation to Metastatic Progression. Genes Dev. 2018, 32, 1267–1284. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.R.; Schmid, M.C. Macrophages as Key Drivers of Cancer Progression and Metastasis. Mediat. Inflamm. 2017, 2017, 9624760. [Google Scholar] [CrossRef] [PubMed]
- Van Strien, M.E.; de Vries, H.E.; Chrobok, N.L.; Bol, J.G.J.M.; Breve, J.J.P.; van der Pol, S.M.P.; Kooij, G.; van Buul, J.D.; Karpuj, M.; Steinman, L.; et al. Tissue Transglutaminase Contributes to Experimental Multiple Sclerosis Pathogenesis and Clinical Outcome by Promoting Macrophage Migration. Brain Behav. Immun. 2015, 50, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Hashemi Goradel, N.; Farhood, B.; Salehi, E.; Nashtaei, M.S.; Khanlarkhani, N.; Khezri, Z.; Majidpoor, J.; Abouzaripour, M.; Habibi, M.; et al. Macrophage Polarity in Cancer: A Review. J. Cell. Biochem. 2019, 120, 2756–2765. [Google Scholar] [CrossRef]
- Sun, H.; Kaartinen, M.T. Transglutaminases in Monocytes and Macrophages. Med. Sci. 2018, 6, 115. [Google Scholar] [CrossRef]
- Sestito, C.; Brevé, J.J.P.; Bol, J.G.J.M.; Wilhelmus, M.M.M.; Drukarch, B.; van Dam, A. Tissue Transglutaminase Contributes to Myelin Phagocytosis in Interleukin-4-Treated Human Monocyte-Derived Macrophages. Cytokine 2020, 128, 155024. [Google Scholar] [CrossRef]
- Nadella, V.; Wang, Z.; Johnson, T.S.; Griffin, M.; Devitt, A. Transglutaminase 2 Interacts with Syndecan-4 and CD44 at the Surface of Human Macrophages to Promote Removal of Apoptotic Cells. Biochim. Biophys. Acta Mol. Cell Res. 2015, 1853, 201–212. [Google Scholar]
- Scarpellini, A.; Huang, L.; Burhan, I.; Schroeder, N.; Funck, M.; Johnson, T.S.; Verderio, E.A.M. Syndecan-4 Knockout Leads to Reduced Extracellular Transglutaminase-2 and Protects Against Tubulointerstitial Fibrosis. J. Am. Soc. Nephrol. 2014, 25, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Benn, M.C.; Weber, W.; Klotzsch, E.; Vogel, V.; Pot, S.A. Tissue Transglutaminase in Fibrosis—More than an Extracellular Matrix Cross-Linker. Curr. Opin. Biomed. Eng. 2019, 10, 156–164. [Google Scholar] [CrossRef]
- Afik, R.; Zigmond, E.; Vugman, M.; Klepfish, M.; Shimshoni, E.; Pasmanik-Chor, M.; Shenoy, A.; Bassat, E.; Halpern, Z.; Geiger, T.; et al. Tumor Macrophages are Pivotal Constructors of Tumor Collagenous Matrix. J. Exp. Med. 2016, 213, 2315–2331. [Google Scholar] [CrossRef]
- Liguori, M.; Solinas, G.; Germano, G.; Mantovani, A.; Allavena, P. Tumor-Associated Macrophages as Incessant Builders and Destroyers of the Cancer Stroma. Cancers 2011, 3, 3740–3761. [Google Scholar] [CrossRef] [PubMed]
- Quaranta, V.; Rainer, C.; Nielsen, S.R.; Raymant, M.L.; Ahmed, M.S.; Engle, D.D.; Taylor, A.; Murray, T.; Campbell, F.; Palmer, D.H.; et al. Macrophage-Derived Granulin Drives Resistance to Immune Checkpoint Inhibition in Metastatic Pancreatic Cancer. Cancer Res. 2018, 78, 4253–4269. [Google Scholar] [CrossRef]
- Hodrea, J.; Demény, M.Á.; Majai, G.; Sarang, Z.; Korponay-Szabó, I.R.; Fésüs, L. Transglutaminase 2 is Expressed and Active on the Surface of Human Monocyte-Derived Dendritic Cells and Macrophages. Immunol. Lett. 2010, 130, 74–81. [Google Scholar] [CrossRef]
- Kim, J.; Jeong, E.M.; Jeong, Y.; Lee, W.J.; Kang, J.S.; Kim, I.; Hwang, Y. Transglutaminase 2 on the Surface of Dendritic Cells is Proposed to be Involved in Dendritic cell–T Cell Interaction. Cell. Immunol. 2014, 289, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Ráki, M.; Schjetne, K.W.; Stamnaes, J.; Molberg, Ø.; Jahnsen, F.L.; Issekutz, T.B.; Bogen, B.; Sollid, L.M. Surface Expression of Transglutaminase 2 by Dendritic Cells and its Potential Role for Uptake and Presentation of Gluten Peptides to T Cells. Scand. J. Immunol. 2007, 65, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Ghoneim, H.E.; Zamora, A.E.; Thomas, P.G.; Youngblood, B.A. Cell-Intrinsic Barriers of T Cell-Based Immunotherapy. Trends Mol. Med. 2016, 22, 1000–1011. [Google Scholar] [CrossRef]
- Ando, M.; Ito, M.; Srirat, T.; Kondo, T.; Yoshimura, A. Memory T Cell, Exhaustion, and Tumor Immunity. Immunol. Med. 2020, 43, 1–9. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Q.; Zhang, X.; Cui, M.; Li, T.; Zhang, Y.; Liao, Q. Immune Subtyping for Pancreatic Cancer with Implication in Clinical Outcomes and Improving Immunotherapy. Cancer Cell Int. 2021, 21, 137. [Google Scholar] [CrossRef]
- Yadav, L. Tumour Angiogenesis and Angiogenic Inhibitors: A Review. J. Clin. Diagn. Res. 2015, 9, XE01–XE05. [Google Scholar] [CrossRef]
- Lunt, S.Y.; Vander Heiden, M.G. Aerobic Glycolysis: Meeting the Metabolic Requirements of Cell Proliferation. Annu. Rev. Cell Dev. Biol. 2011, 27, 441–464. [Google Scholar] [CrossRef]
- Schwartz, L.; Supuran, C.T.; Alfarouk, O.K. The Warburg Effect and the Hallmarks of Cancer. Anti-Cancer Agents Med. Chem. 2017, 17, 164–170. [Google Scholar] [CrossRef]
- Mastroberardino, P.G.; Farrace, M.G.; Viti, I.; Pavone, F.; Fimia, G.M.; Melino, G.; Rodolfo, C.; Piacentini, M. “Tissue” transglutaminase contributes to the formation of disulphide bridges in proteins of mitochondrial respiratory complexes. Biochim. Biophys. Acta 2006, 1757, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Rossin, F.; D’Eletto, M.; Falasca, L.; Sepe, S.; Cocco, S.; Fimia, G.M.; Campanella, M.; Mastroberardino, P.G.; Farrace, M.G.; Piacentini, M. Transglutaminase 2 ablation leads to mitophagy impairment associated with a metabolic shift towards aerobic glycolysis. Cell Death Differ. 2015, 22, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Bhedi, C.D.; Nasirova, S.; Toksoz, D.; Warburton, R.R.; Morine, K.J.; Kapur, N.K.; Galper, J.B.; Preston, I.R.; Hill, N.S.; Fanburg, B.L.; et al. Glycolysis regulated transglutaminase 2 activation in cardiopulmonary fibrogenic remodeling. FASEB J. 2020, 34, 930–944. [Google Scholar] [CrossRef]
- Katt, W.P.; Antonyak, M.A.; Cerione, R.A. Simultaneously Targeting Tissue Transglutaminase and Kidney Type Glutaminase Sensitizes Cancer Cells to Acid Toxicity and Offers New Opportunities for Therapeutic Intervention. Mol. Pharm. 2015, 12, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.A.; Lomax-Browne, H.J.; Carter, T.M.; Kinch, C.E.; Hall, D.M.S. Molecular Interactions in Cancer Cell Metastasis. Acta Histochem. 2010, 112, 3–25. [Google Scholar] [CrossRef]
- Greijer, A.E.; van der Wall, E. The Role of Hypoxia Inducible Factor 1 (HIF-1) in Hypoxia Induced Apoptosis. J. Clin. Pathol. 2004, 57, 1009–1014. [Google Scholar] [CrossRef]
- Lossi, L.; Castagna, C.; Merighi, A. Caspase-3 Mediated Cell Death in the Normal Development of the Mammalian Cerebellum. Int. J. Mol. Sci. 2018, 19, 3999. [Google Scholar] [CrossRef] [PubMed]
- Baeriswyl, V.; Christofori, G. The Angiogenic Switch in Carcinogenesis. Semin. Cancer Biol. 2009, 19, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Jain, R.K. Molecular Mechanisms and Clinical Applications of Angiogenesis. Nature 2011, 473, 298–307. [Google Scholar] [CrossRef]
- Cooke, V.; LeBleu, V.; Keskin, D.; Khan, Z.; O’Connell, J.; Teng, Y.; Duncan, M.; Xie, L.; Maeda, G.; Vong, S.; et al. Pericyte Depletion Results in Hypoxia-Associated Epithelial-to-Mesenchymal Transition and Metastasis Mediated by Met Signaling Pathway. Cancer Cell 2012, 21, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.A.; Kotsakis, P.; Johnson, T.S.; Chau, D.Y.S.; Ali, S.; Melino, G.; Griffin, M. Matrix Changes Induced by Transglutaminase 2 Lead to Inhibition of Angiogenesis and Tumor Growth. Cell Death Differ. 2006, 13, 1442–1453. [Google Scholar] [CrossRef] [PubMed]
- Stylianopoulos, T.; Martin, J.D.; Chauhan, V.P.; Jain, S.R.; Diop-Frimpong, B.; Bardeesy, N.; Smith, B.L.; Ferrone, C.R.; Hornicek, F.J.; Boucher, Y.; et al. Causes, Consequences, and Remedies for Growth-Induced Solid Stress in Murine and Human Tumors. Proc. Natl. Acad. Sci. USA 2012, 109, 15101–15108. [Google Scholar] [CrossRef] [PubMed]
- Spurlin, T.A.; Bhadriraju, K.; Chung, K.; Tona, A.; Plant, A.L. Vascular Smooth Muscle Cell Response to Transglutaminase 2 Cross-Linked Collagen Fibril Thin Films. Biophys. J. 2009, 96, 297a. [Google Scholar] [CrossRef][Green Version]
- Chauhan, V.P.; Martin, J.D.; Liu, H.; Lacorre, D.A.; Jain, S.R.; Kozin, S.V.; Stylianopoulos, T.; Mousa, A.S.; Han, X.; Adstamongkonkul, P.; et al. Angiotensin Inhibition Enhances Drug Delivery and Potentiates Chemotherapy by Decompressing Tumor Blood Vessels. Nat. Commun. 2013, 4, 2516. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, V.P.; Chen, I.X.; Tong, R.; Ng, M.R.; Martin, J.D.; Naxerova, K.; Wu, M.W.; Huang, P.; Boucher, Y.; Kohane, D.S.; et al. Reprogramming the Microenvironment with Tumor-Selective Angiotensin Blockers Enhances Cancer Immunotherapy. Proc. Natl. Acad. Sci. USA 2019, 116, 10674–10680. [Google Scholar] [CrossRef]
- Paszek, M.J.; Zahir, N.; Johnson, K.R.; Lakins, J.N.; Rozenberg, G.I.; Gefen, A.; Reinhart-King, C.A.; Margulies, S.S.; Dembo, M.; Boettiger, D.; et al. Tensional Homeostasis and the Malignant Phenotype. Cancer Cell 2005, 8, 241–254. [Google Scholar] [CrossRef]
- Pickup, M.W.; Mouw, J.K.; Weaver, V.M. The Extracellular Matrix Modulates the Hallmarks of Cancer. EMBO Rep. 2014, 15, 1243–1253. [Google Scholar] [CrossRef]
- Yue, B. Biology of the Extracellular Matrix: An Overview. J. Glaucoma 2014, 23, S20–S23. [Google Scholar] [CrossRef] [PubMed]
- Nia, H.T.; Munn, L.L.; Jain, R.K. Physical Traits of Cancer. Science 2020, 370, 546. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.K.; Martin, J.D.; Stylianopoulos, T. The Role of Mechanical Forces in Tumor Growth and Therapy. Annu. Rev. Biomed. Eng. 2014, 16, 321–346. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Weaver, V. Mechanics, Malignancy, and Metastasis: The Force Journey of a Tumor Cell. Cancer Metastasis Rev. 2009, 28, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Kalli, M.; Papageorgis, P.; Gkretsi, V.; Stylianopoulos, T. Solid Stress Facilitates Fibroblasts Activation to Promote Pancreatic Cancer Cell Migration. Ann. Biomed. Eng. 2018, 46, 657–669. [Google Scholar] [CrossRef] [PubMed]
- Wipff, P.; Rifkin, D.B.; Meister, J.; Hinz, B. Myofibroblast Contraction Activates Latent TGF-Β1 from the Extracellular Matrix. J. Cell Biol. 2007, 179, 1311–1323. [Google Scholar] [CrossRef]
- Semkova, M.E.; Hsuan, J.J. TGFβ-1 Induced Cross-Linking of the Extracellular Matrix of Primary Human Dermal Fibroblasts. Int. J. Mol. Sci. 2021, 22, 984. [Google Scholar] [CrossRef]
- Zhang, R.; Ma, M.; Dong, G.; Yao, R.; Li, J.; Zheng, Q.; Dong, Y.; Ma, H.; Gao, D.; Cui, J.; et al. Increased Matrix Stiffness Promotes Tumor Progression of Residual Hepatocellular Carcinoma After Insufficient Heat Treatment. Cancer Sci. 2017, 108, 1778–1786. [Google Scholar] [CrossRef]
- Zhao, D.; Xue, C.; Li, Q.; Liu, M.; Ma, W.; Zhou, T.; Lin, Y. Substrate Stiffness Regulated Migration and Angiogenesis Potential of A549 Cells and HUVECs. J. Cell. Physiol. 2018, 233, 3407–3417. [Google Scholar] [CrossRef]
- Berger, A.J.; Renner, C.M.; Hale, I.; Yang, X.; Ponik, S.M.; Weisman, P.S.; Masters, K.S.; Kreeger, P.K. Scaffold Stiffness Influences Breast Cancer Cell Invasion Via EGFR-Linked Mena Upregulation and Matrix Remodeling. Matrix Biol. 2020, 85–86, 80–93. [Google Scholar] [CrossRef]
- Reid, S.E.; Kay, E.J.; Neilson, L.J.; Henze, A.; Serneels, J.; McGhee, E.J.; Dhayade, S.; Nixon, C.; Mackey, J.B.; Santi, A.; et al. Tumor Matrix Stiffness Promotes Metastatic Cancer Cell Interaction with the Endothelium. EMBO J. 2017, 36, 2373–2389. [Google Scholar] [CrossRef] [PubMed]
- Saatci, O.; Kaymak, A.; Raza, U.; Ersan, P.G.; Akbulut, O.; Banister, C.E.; Sikirzhytski, V.; Tokat, U.M.; Aykut, G.; Ansari, S.A.; et al. Targeting Lysyl Oxidase (LOX) Overcomes Chemotherapy Resistance in Triple Negative Breast Cancer. Nat. Commun. 2020, 11, 2416. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Condello, S.; Yakubov, B.; Emerson, R.; Caperell-Grant, A.; Hitomi, K.; Xie, J.; Matei, D. Tissue Transglutaminase Mediated Tumor-Stroma Interaction Promotes Pancreatic Cancer Progression. Clinical Cancer Res. 2015, 21, 4482–4493. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, H.; Sahai, E. Mechanisms and Impact of Altered Tumour Mechanics. Nat. Cell Biol. 2018, 20, 766–774. [Google Scholar] [CrossRef]
- Pankova, D.; Chen, Y.; Terajima, M.; Schliekelman, M.J.; Baird, B.N.; Fahrenholtz, M.; Sun, L.; Gill, B.J.; Vadakkan, T.J.; Kim, M.P.; et al. Cancer-Associated Fibroblasts Induce a Collagen Cross-Link Switch in Tumor Stroma. Mol. Cancer Res. 2016, 14, 287–295. [Google Scholar] [CrossRef]
- Wirtz, D.; Konstantopoulos, K.; Searson, P.C. The Physics of Cancer: The Role of Physical Interactions and Mechanical Forces in Metastasis. Nat. Rev. Cancer 2011, 11, 512. [Google Scholar] [CrossRef]
- Friedl, P.H.A.; Gilmour, D. Collective Cell Migration in Morphogenesis, Regeneration and Cancer. Nat. Rev. Mol. Cell Biol. 2009, 10, 445–457. [Google Scholar] [CrossRef]
- Tse, J.M.; Cheng, G.; Tyrrell, J.A.; Wilcox-Adelman, S.A.; Boucher, Y.; Jain, R.K.; Munn, L.L. Mechanical Compression Drives Cancer Cells Toward Invasive Phenotype. Proc. Natl. Acad. Sci. USA 2011, 109, 911–916. [Google Scholar] [CrossRef]
- Tsai, J.H.; Yang, J. Epithelial-Mesenchymal Plasticity in Carcinoma Metastasis. Genes Dev. 2013, 27, 2192–2206. [Google Scholar] [CrossRef] [PubMed]
- Mierke, C.T. The Matrix Environmental and Cell Mechanical Properties Regulate Cell Migration and Contribute to the Invasive Phenotype of Cancer Cells. Rep. Prog. Phys. 2019, 82, 64602. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Lin, C.; Lee, M.; Lee, L.; Chang, G.; Lee, P.; Hung, C.; Ko, W.; Tsai, P.; Schally, A.V.; et al. Up-Regulation of Fibronectin and Tissue Transglutaminase Promotes Cell Invasion Involving Increased Association with Integrin and MMP Expression in A431 Cells. Anticancer Res. 2010, 30, 4177–4186. [Google Scholar] [PubMed]
- Bordeleau, F.; Wang, W.; Simmons, A.; Antonyak, M.A.; Cerione, R.A.; Reinhart-King, C.A. Tissue Transglutaminase 2 Regulates Tumor Cell Tensional Homeostasis by Increasing Contractility. J. Cell Sci. 2020, 133, jcs231134. [Google Scholar] [CrossRef]
- Yurchenco, P.D.; Ruben, G.C. Basement Membrane Structure in Situ: Evidence for Lateral Associations in the Type IV Collagen Network. J. Cell Biol. 1987, 105, 2559–2568. [Google Scholar] [CrossRef]
- LeBleu, V.S.; MacDonald, B.; Kalluri, R. Structure and Function of Basement Membranes. Exp. Biol. Med. 2007, 232, 1121–1129. [Google Scholar] [CrossRef]
- Iozzo, R.V.; Zoeller, J.J.; Nyström, A. Basement Membrane Proteoglycans: Modulators Par Excellence of Cancer Growth and Angiogenesis. Mol. Cells 2009, 27, 503–513. [Google Scholar] [CrossRef]
- Wisdom, K.M.; Indana, D.; Chou, P.; Desai, R.; Kim, T.; Chaudhuri, O. Covalent Cross-Linking of Basement Membrane-Like Matrices Physically Restricts Invasive Protrusions in Breast Cancer Cells. Matrix Biol. 2020, 85–86, 94–111. [Google Scholar] [CrossRef]
- Hotary, K.; Li, X.; Allen, E.; Stevens, S.L.; Weiss, S.J. A Cancer Cell Metalloprotease Triad Regulates the Basement Membrane Transmigration Program. Genes Dev. 2006, 20, 2673–2686. [Google Scholar] [CrossRef]
- Chang, J.; Chaudhuri, O. Beyond Proteases: Basement Membrane Mechanics and Cancer Invasion. J. Cell Biol. 2019, 218, 2456–2469. [Google Scholar] [CrossRef]
- Strzyz, P. Forcing through Barriers. Nat. Rev. Mol. Cell Biol. 2019, 20, 136. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R. Basement Membranes: Structure, Assembly and Role in Tumour Angiogenesis. Nat. Rev. Cancer 2003, 3, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Philp, C.J.; Siebeke, I.; Clements, D.; Miller, S.; Habgood, A.; John, A.E.; Navaratnam, V.; Hubbard, R.B.; Jenkins, G.; Johnson, S.R. Extracellular Matrix Cross-Linking Enhances Fibroblast Growth and Protects Against Matrix Proteolysis in Lung Fibrosis. Am. J. Respir. Cell Mol. Biol. 2018, 58, 594–603. [Google Scholar] [CrossRef]
- Aeschlimann, D.; Paulsson, M. Cross-Linking of Laminin-Nidogen Complexes by Tissue Transglutaminase. A Novel Mechanism for Basement Membrane Stabilization. J. Biol. Chem. 1991, 266, 15308–15317. [Google Scholar] [CrossRef]
- Dziadek, M. Role of Laminin-Nidogen Complexes in Basement Membrane Formation during Embryonic Development. Experientia 1995, 51, 901–913. [Google Scholar] [CrossRef]
- Haroon, Z.A.; Lai, T.; Hettasch, J.M.; Lindberg, R.A.; Dewhirst, M.W.; Greenberg, C.S. Tissue Transglutaminase is Expressed as a Host Response to Tumor Invasion and Inhibits Tumor Growth. Lab. Investig. 1999, 79, 1679–1686. [Google Scholar]
- Mangala, L.S.; Arun, B.; Sahin, A.A.; Mehta, K. Tissue Transglutaminase-Induced Alterations in Extracellular Matrix Inhibit Tumor Invasion. Mol. Cancer 2005, 4, 33. [Google Scholar] [CrossRef]
- Di Giacomo, G.; Di Giacomo, G.; Lentini, A.; Lentini, A.; Beninati, S.; Beninati, S.; Piacentini, M.; Piacentini, M.; Rodolfo, C.; Rodolfo, C. In Vivo Evaluation of Type 2 Transglutaminase Contribution to the Metastasis Formation in Melanoma. Amino Acids 2009, 36, 717–724. [Google Scholar] [CrossRef]
- Facchiano, F.; Facchiano, F.; D’Arcangelo, D.; D’Arcangelo, D.; Lentini, A.; Lentini, A.; Rossi, S.; Rossi, S.; Senatore, C.; Senatore, C.; et al. Tissue Transglutaminase Activity Protects from Cutaneous Melanoma Metastatic Dissemination: An in Vivo Study. Amino Acids 2013, 44, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Levental, K.R.; Yu, H.; Kass, L.; Lakins, J.N.; Egeblad, M.; Erler, J.T.; Fong, S.F.T.; Csiszar, K.; Giaccia, A.; Weninger, W.; et al. Matrix Crosslinking Forces Tumor Progression by Enhancing Integrin Signaling. Cell 2009, 139, 891–906. [Google Scholar] [CrossRef] [PubMed]
- Satpathy, M.; Shao, M.; Emerson, R.; Donner, D.B.; Matei, D. Tissue Transglutaminase Regulates Matrix Metalloproteinase-2 in Ovarian Cancer by Modulating cAMP-Response Element-Binding Protein Activity. J. Biol. Chem. 2009, 284, 15390–15399. [Google Scholar] [CrossRef]
- Coulson-Thomas, V.; Coulson-Thomas, Y.; Gesteira, T.; de Paula, C.; Mader, A.; Waisberg, J.; Pinhal, M.; Friedl, A.; Toma, L.; Nader, H. Colorectal Cancer Desmoplastic Reaction Up-Regulates Collagen Synthesis and Restricts Cancer Cell Invasion. Cell Tissue Res. 2011, 346, 223–236. [Google Scholar] [CrossRef]
- Chau, D.Y.S.; Collighan, R.J.; Verderio, E.A.M.; Addy, V.L.; Griffin, M. The Cellular Response to Transglutaminase-Cross-Linked Collagen. Biomaterials 2005, 26, 6518–6529. [Google Scholar] [CrossRef]
- Belkin, A.M.; Zemskov, E.A.; Hang, J.; Akimov, S.S.; Sikora, S.; Strongin, A.Y. Cell-Surface-Associated Tissue Transglutaminase is a Target of MMP-2 Proteolysis. Biochemistry 2004, 43, 11760–11769. [Google Scholar] [CrossRef] [PubMed]
- Belkin, A.M.; Akimov, S.S.; Zaritskaya, L.S.; Ratnikov, B.I.; Deryugina, E.I.; Strongin, A.Y. Matrix-Dependent Proteolysis of Surface Transglutaminase by Membrane-Type Metalloproteinase Regulates Cancer Cell Adhesion and Locomotion. J. Biol. Chem. 2001, 276, 18415–18422. [Google Scholar] [CrossRef] [PubMed]
- Birckbichler, P.J.; Bonner, R.B.; Hurst, R.E.; Bane, B.L.; Pitha, J.V.; Hemstreet, G.P., 3rd. Loss of tissue transglutaminase as a biomarker for prostate adenocarcinoma. Cancer 2000, 89, 412–423. [Google Scholar]
- Hager, H.; Jensen, P.H.; Hamilton-Dutoit, S.; Neilsen, M.S.; Birckbichler, P.; Gliemann, J. Expression of tissue transglutaminase in human bladder carcinoma. J. Pathol. 1997, 183, 398–403. [Google Scholar] [CrossRef]
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor Microenvironment Complexity and Therapeutic Implications at a Glance. Cell Commun. Signal. 2020, 18, 59. [Google Scholar] [CrossRef] [PubMed]
- Naba, A.; Clauser, K.R.; Ding, H.; Whittaker, C.A.; Carr, S.A.; Hynes, R.O. The Extracellular Matrix: Tools and Insights for the “omics” Era. Matrix Biol. 2016, 49, 10–24. [Google Scholar] [CrossRef]
- Campbell, I.D.; Humphries, M.J. Integrin Structure, Activation, and Interactions. Cold Spring Harb. Perspect. Biol. 2011, 3, a004994. [Google Scholar] [CrossRef]
- Fullár, A.; Dudás, J.; Oláh, L.; Hollósi, P.; Papp, Z.; Sobel, G.; Karászi, K.; Paku, S.; Baghy, K.; Kovalszky, I. Remodeling of Extracellular Matrix by Normal and Tumor-Associated Fibroblasts Promotes Cervical Cancer Progression. BMC Cancer 2015, 15, 256. [Google Scholar] [CrossRef] [PubMed]
- Deville, S.S.; Cordes, N. The Extracellular, Cellular, and Nuclear Stiffness, a Trinity in the Cancer Resistome—A Review. Front. Oncol. 2019, 9, 1376. [Google Scholar] [CrossRef] [PubMed]
- Cox, T.R.; Erler, J.T. Remodeling and Homeostasis of the Extracellular Matrix: Implications for Fibrotic Diseases and Cancer. Dis. Models Mech. 2011, 4, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Huijbers, I.J.; Iravani, M.; Popov, S.; Robertson, D.; Al-Sarraj, S.; Jones, C.; Isacke, C.M. A Role for Fibrillar Collagen Deposition and the Collagen Internalization Receptor Endo180 in Glioma Invasion. PLoS ONE 2010, 5, e9808. [Google Scholar] [CrossRef]
- Distler, J.H.W.; Györfi, A.; Ramanujam, M.; Whitfield, M.L.; Königshoff, M.; Lafyatis, R. Shared and Distinct Mechanisms of Fibrosis. Nat. Rev. Rheumatol. 2019, 15, 705–730. [Google Scholar] [CrossRef]
- Hinz, B.; Phan, S.H.; Thannickal, V.J.; Prunotto, M.; Desmouliere, A.; Varga, J.; de Wever, O.; Mareel, M.; Gabbiani, G. Recent Developments in Myofibroblast Biology: Paradigms for Connective Tissue Remodeling. Am. J. Pathol. 2012, 180, 1340–1355. [Google Scholar] [CrossRef]
- Tschumperlin, D.J.; Lagares, D. Mechano-Therapeutics: Targeting Mechanical Signaling in Fibrosis and Tumor Stroma. Pharmacol. Ther. 2020, 212, 107575. [Google Scholar] [CrossRef]
- Kauppila, S.; Stenbäck, F.; Risteli, J.; Jukkola, A.; Risteli, L. Aberrant Type I and Type III Collagen Gene Expression in Human Breast Cancer in Vivo. J. Pathol. 1998, 186, 262–268. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the Extracellular Matrix in Development and Disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef]
- Wells, R.G.; Discher, D.E. Matrix Elasticity, Cytoskeletal Tension, and TGF-Beta: The Insoluble and Soluble Meet. Sci. Signal. 2008, 1, pe13. [Google Scholar] [CrossRef]
- Mickle, M.; Adhikary, G.; Shrestha, S.; Xu, W.; Eckert, R.L. VGLL4 inhibits YAP1/TEAD signaling to suppress the epidermal squamous cell carcinoma cancer phenotype. Mol. Carcinog. 2021. [Google Scholar] [CrossRef]
- Herman, J.F.; Mangala, L.S.; Mehta, K. Implications of Increased Tissue Transglutaminase (TG2) Expression in Drug-Resistant Breast Cancer (MCF-7) Cells. Oncogene 2006, 25, 3049–3058. [Google Scholar] [CrossRef]
- Akimov, S.S.; Krylov, D.; Fleischman, L.F.; Belkin, A.M. Tissue Transglutaminase is an Integrin-Binding Adhesion Coreceptor for Fibronectin. J. Cell Biol. 2000, 148, 825–838. [Google Scholar] [CrossRef]
- Kotsakis, P.; Kotsakis, P.; Wang, Z.; Wang, Z.; Collighan, R.; Collighan, R.; Griffin, M.; Griffin, M. The Role of Tissue Transglutaminase (TG2) in Regulating the Tumour Progression of the Mouse Colon Carcinoma CT26. Amino Acids 2011, 41, 909–921. [Google Scholar] [CrossRef][Green Version]
- Libring, S.; Shinde, A.; Chanda, M.K.; Nuru, M.; George, H.; Saleh, A.M.; Abdullah, A.; Kinzer-Ursem, T.L.; Calve, S.; Wendt, M.K.; et al. The Dynamic Relationship of Breast Cancer Cells and Fibroblasts in Fibronectin Accumulation at Primary and Metastatic Tumor Sites. Cancers 2020, 12, 1270. [Google Scholar] [CrossRef]
- Khanna, M.; Chelladurai, B.; Gavini, A.; Li, L.; Shao, M.; Courtney, D.; Turchi, J.J.; Matei, D.; Meroueh, S. Targeting Ovarian Tumor Cell Adhesion Mediated by Tissue Transglutaminase. Mol. Cancer Ther. 2011, 10, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Mangala, L.S.; Fok, J.Y.; Zorrilla-Calancha, I.R.; Verma, A.; Mehta, K. Tissue Transglutaminase Expression Promotes Cell Attachment, Invasion and Survival in Breast Cancer Cells. Oncogene 2007, 26, 2459–2470. [Google Scholar] [CrossRef] [PubMed]
- Parri, M.; Chiarugi, P. Rac and Rho GTPases in Cancer Cell Motility Control. Cell Commun. Signal. 2010, 8, 23. [Google Scholar] [CrossRef]
- Dillekås, H.; Rogers, M.S.; Straume, O. Are 90% of Deaths from Cancer Caused by Metastases? Cancer Med. 2019, 8, 5574–5576. [Google Scholar] [CrossRef]
- Erdem, S.; Erdem, S.; Yegen, G.; Yegen, G.; Telci, D.; Telci, D.; Yildiz, I.; Yildiz, I.; Tefik, T.; Tefik, T.; et al. The Increased Transglutaminase 2 Expression Levels during Initial Tumorigenesis Predict Increased Risk of Metastasis and Decreased Disease-Free and Cancer-Specific Survivals in Renal Cell Carcinoma. World J. Urol. 2015, 33, 1553–1560. [Google Scholar] [CrossRef]
- Erdem, M.; Erdem, S.; Sanli, O.; Sak, H.; Kilicaslan, I.; Sahin, F.; Telci, D. Up-Regulation of TGM2 with ITGB1 and SDC4 is Important in the Development and Metastasis of Renal Cell Carcinoma. Urol. Oncol. 2014, 32, 25.e13–25.e20. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Moon, Y.; Choi, J.; Yoon, S.; Jung, K.H.; Cheon, J.; Kim, W.; Kim, D.; Lee, C.H.; Kim, S.; et al. The GTP Binding Activity of Transglutaminase 2 Promotes Bone Metastasis of Breast Cancer Cells by Downregulating microRNA-205. Am. J. Cancer Res. 2019, 9, 597–607. [Google Scholar]
- Fok, J.Y.; Ekmekcioglu, S.; Mehta, K. Implications of Tissue Transglutaminase Expression in Malignant Melanoma. Mol. Cancer Ther. 2006, 5, 1493–1503. [Google Scholar] [CrossRef] [PubMed]
- Antonyak, M.A.; Li, B.; Boroughs, L.K.; Johnson, J.L.; Druso, J.E.; Bryant, K.L.; Holowka, D.A.; Cerione, R.A. Cancer Cell-Derived Microvesicles Induce Transformation by Transferring Tissue Transglutaminase and Fibronectin to Recipient Cells. Proc. Natl. Acad. Sci. USA 2011, 108, 4852–4857. [Google Scholar] [CrossRef]
- Van den Akker, J.; van Weert, A.; Afink, G.; Bakker, E.N.T.P.; van der Pol, E.; Böing, A.N.; Nieuwland, R.; VanBavel, E. Transglutaminase 2 is Secreted from Smooth Muscle Cells by Transamidation-Dependent Microparticle Formation. Amino Acids 2012, 42, 961–973. [Google Scholar] [CrossRef][Green Version]
- Bianchi, N.; Beninati, S.; Bergamini, C.M. Spotlight on the Transglutaminase 2 Gene: A Focus on Genomic and Transcriptional Aspects. Biochem. J. 2018, 475, 1643–1667. [Google Scholar] [CrossRef]
- Liu, Y.; Cao, X. Characteristics and Significance of the Pre-Metastatic Niche. Cancer Cell 2016, 30, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, A.; Costa-Silva, B.; Shen, T.; Rodrigues, G.; Hashimoto, A.; Tesic Mark, M.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour Exosome Integrins Determine Organotropic Metastasis. Nature 2015, 527, 329–335. [Google Scholar] [CrossRef]
- Shinde, A.; Paez, J.S.; Libring, S.; Hopkins, K.; Solorio, L.; Wendt, M.K. Transglutaminase-2 Facilitates Extracellular Vesicle-Mediated Establishment of the Metastatic Niche. Oncogenesis 2020, 9, 16. [Google Scholar] [CrossRef]
- Shafiq, A.; Suwakulsiri, W.; Rai, A.; Chen, M.; Greening, D.W.; Zhu, H.J.; Xu, R.; Simpson, R.J. Transglutaminase-2, RNA-binding proteins and mitochondrial proteins selectively traffic to MDCK cell-derived microvesicles following H-Ras-induced epithelial-mesenchymal transition. Proteomics 2021, 27, e2000221. [Google Scholar] [CrossRef] [PubMed]
- Brill-Karniely, Y.; Dror, D.; Duanis-Assaf, T.; Goldstein, Y.; Schwob, O.; Millo, T.; Orehov, N.; Stern, T.; Jaber, M.; Loyfer, N.; et al. Triangular Correlation (TrC) between Cancer Aggressiveness, Cell Uptake Capability, and Cell Deformability. Sci. Adv. 2020, 6, eaax2861. [Google Scholar] [CrossRef]
- Eligula, L.; Chuang, L.; Phillips, M.L.; Motoki, M.; Seguro, K.; Muhlrad, A. Transglutaminase-Induced Cross-Linking between Subdomain 2 of G-Actin and the 636–642 Lysine-Rich Loop of Myosin Subfragment 1. Biophys. J. 1998, 74, 953–963. [Google Scholar] [CrossRef]
- Dolge, L.; Dolge, L.; Aufenvenne, K.; Aufenvenne, K.; Traupe, H.; Traupe, H.; Baumgartner, W.; Baumgartner, W. Beta-Actin is a Target for Transglutaminase Activity at Synaptic Endings in Chicken Telencephalic Cell Cultures. J. Mol. Neurosci. 2012, 46, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Kirkpatrick, L.; Schilling, A.; Helseth, D.; Chabot, N.; Keillor, J.; Johnson, G.W.; Brady, S. Transglutaminase and Polyamination of Tubulin: Posttranslational Modification for Stabilizing Axonal Microtubules. Neuron 2013, 78, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Schwager, S.C.; Bordeleau, F.; Zhang, J.; Antonyak, M.A.; Cerione, R.A.; Reinhart-King, C.A. Matrix Stiffness Regulates Microvesicle-Induced Fibroblast Activation. Am. J. Physiol. Cell Physiol. 2019, 317, C82–C92. [Google Scholar] [CrossRef]
- Adamczyk, M.; Griffiths, R.; Dewitt, S.; Knäuper, V.; Aeschlimann, D. P2X7 Receptor Activation Regulates Rapid Unconventional Export of Transglutaminase-2. J. Cell Sci. 2015, 128, 4615–4628. [Google Scholar] [CrossRef] [PubMed]
- Arnaud-Sampaio, V.F.; Rabelo, I.L.A.; Ulrich, H.; Lameu, C. The P2X7 Receptor in the Maintenance of Cancer Stem Cells, Chemoresistance and Metastasis. Stem Cell Rev. Rep. 2020, 16, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Wang-Gillam, A.; Lockhart, A.C.; Tan, B.R.; Suresh, R.; Lim, K.; Ratner, L.; DeNardo, D.G. Phase I Study of Defactinib Combined with Pembrolizumab and Gemcitabine in Patients with Advanced Cancer. J. Clin. Oncol. 2018, 36, 2561. [Google Scholar] [CrossRef]
- Mohanty, A.; Pharaon, R.R.; Nam, A.; Salgia, S.; Kulkarni, P.; Massarelli, E. FAK-Targeted and Combination Therapies for the Treatment of Cancer: An Overview of Phase I and II Clinical Trials. Expert Opin. Investig. Drugs 2020, 29, 399–409. [Google Scholar] [CrossRef]
- Akhurst, R.J. Targeting TGF-Beta Signaling for Therapeutic Gain. Cold Spring Harb. Perspect. Biol. 2017, 9, a022301. [Google Scholar] [CrossRef] [PubMed]
- Raab-Westphal, S.; Marshall, J.F.; Goodman, S.L. Integrins as Therapeutic Targets: Successes and Cancers. Cancers 2017, 9, 110. [Google Scholar] [CrossRef] [PubMed]
- Efthymiou, G.; Saint, A.; Ruff, M.; Rekad, Z.; Ciais, D.; van Obberghen-Schilling, E. Shaping Up the Tumor Microenvironment with Cellular Fibronectin. Front. Oncol. 2020, 10, 641. [Google Scholar] [CrossRef]
- Cox, T.R.; Bird, D.; Baker, A.; Barker, H.E.; Ho, M.W.; Lang, G.; Erler, J.T. LOX-Mediated Collagen Crosslinking is Responsible for Fibrosis-Enhanced Metastasis. Cancer Res. 2013, 73, 1721–1732. [Google Scholar] [CrossRef]
- Lampi, M.C.; Reinhart-King, C.A. Targeting Extracellular Matrix Stiffness to Attenuate Disease: From Molecular Mechanisms to Clinical Trials. Sci. Transl. Med. 2018, 10, eaao0475. [Google Scholar] [CrossRef] [PubMed]
- Riley, D.J.; Kerr, J.S.; Berg, R.A.; Ianni, B.D.; Pietra, G.G.; Edelman, N.H.; Prockop, D.J. Beta-Aminopropionitrile Prevents Bleomycin-Induced Pulmonary Fibrosis in the Hamster. Am. Rev. Respir. Dis. 1982, 125, 67–73. [Google Scholar]
- Yuan, L.; Siegel, M.; Choi, K.; Khosla, C.; Miller, C.R.; Jackson, E.N.; Piwnica-Worms, D.; Rich, K.M. Transglutaminase 2 Inhibitor, KCC009, Disrupts Fibronectin Assembly in the Extracellular Matrix and Sensitizes Orthotopic Glioblastomas to Chemotherapy. Oncogene 2007, 26, 2563–2573. [Google Scholar] [CrossRef]
- Caron, N.S.; Munsie, L.N.; Keillor, J.W.; Truant, R. Using FLIM-FRET to Measure Conformational Changes of Transglutaminase Type 2 in Live Cells. PLoS ONE 2012, 7, e44159. [Google Scholar] [CrossRef]
- Kerr, C.; Szmacinski, H.; Fisher, M.L.; Nance, B.; Lakowicz, J.R.; Akbar, A.; Keillor, J.W.; Lok Wong, T.; Godoy-Ruiz, R.; Toth, E.A.; et al. Transamidase Site-Targeted Agents Alter the Conformation of the Transglutaminase Cancer Stem Cell Survival Protein to Reduce GTP Binding Activity and Cancer Stem Cell Survival. Oncogene 2017, 36, 2981–2990. [Google Scholar] [CrossRef]
- Forni, C.; Braglia, R.; Mulinacci, N.; Urbani, A.; Ronci, M.; Gismondi, A.; Tabolacci, C.; Provenzano, B.; Lentini, A.; Beninati, S. Antineoplastic Activity of Strawberry (Fragaria × Ananassa Duch.) Crude Extracts on B16-F10 Melanoma Cells. Mol. BioSyst. 2014, 1, 1255–1263. [Google Scholar] [CrossRef]
- Kim, S. New Insights into Development of Transglutaminase 2 Inhibitors as Pharmaceutical Lead Compounds. Med. Sci. 2018, 6, 87. [Google Scholar] [CrossRef] [PubMed]
- Besouw, M.; Masereeuw, R.; van den Heuvel, L.; Levtchenko, E. Cysteamine: An Old Drug with New Potential. Drug Discov. Today 2013, 18, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Reversibly Acting Transglutaminase 2 Inhibitors: Drug Candidates for the Treatment of Fibrosis. Available online: https://zedira.com/News/Reversibly-acting-transglutaminase-2-inhibitors-drug-candidates-for-the-treatment-of-fibrosis_127 (accessed on 31 March 2021).
- Harrison, S.A.; Abdelmalek, M.F.; Caldwell, S.; Shiffman, M.L.; Diehl, A.M.; Ghalib, R.; Lawitz, E.J.; Rockey, D.C.; Schall, R.A.; Jia, C.; et al. Simtuzumab is Ineffective for Patients with Bridging Fibrosis rr Compensated Cirrhosis Caused by Nonalcoholic Steatohepatitis. Gastroenterology 2018, 155, 1140–1153. [Google Scholar] [CrossRef] [PubMed]
- Winer, A.; Adams, S.; Mignatti, P. Matrix Metalloproteinase Inhibitors in Cancer Therapy: Turning Past Failures into Future Successes. Mol. Cancer Ther. 2018, 17, 1147–1155. [Google Scholar] [CrossRef] [PubMed]
| Hallmark | Key Mechanisms/Pathways | References |
|---|---|---|
| Sustaining proliferative signals | TGF-β, PI3K/AKT, Β-catenin/Wnt, ERK1/2 | [21,22,23,24] |
| Evading growth suppressors | Regulation of RB/p53 pathways | [25,26,27] |
| Resisting cell death | Caspase-3/Bax, TRAIL | [28,29,30,31,32,33] |
| Enabling replicative immortality | CSCs (CD44), EMT, YAP/TAZ | [34,35,36,37,38,39] |
| Inducing angiogenesis | VEGF, NF-κB/HIF1α, ECM remodelling | [40,41,42,43] |
| Activating invasion | EGF, EMT/TGF-β, type I collagen/β-1 integrins, Rac, ECM alterations | [12,35,36,37,44,45,46,47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tempest, R.; Guarnerio, S.; Maani, R.; Cooper, J.; Peake, N. The Biological and Biomechanical Role of Transglutaminase-2 in the Tumour Microenvironment. Cancers 2021, 13, 2788. https://doi.org/10.3390/cancers13112788
Tempest R, Guarnerio S, Maani R, Cooper J, Peake N. The Biological and Biomechanical Role of Transglutaminase-2 in the Tumour Microenvironment. Cancers. 2021; 13(11):2788. https://doi.org/10.3390/cancers13112788
Chicago/Turabian StyleTempest, Robert, Sonia Guarnerio, Rawan Maani, Jamie Cooper, and Nicholas Peake. 2021. "The Biological and Biomechanical Role of Transglutaminase-2 in the Tumour Microenvironment" Cancers 13, no. 11: 2788. https://doi.org/10.3390/cancers13112788
APA StyleTempest, R., Guarnerio, S., Maani, R., Cooper, J., & Peake, N. (2021). The Biological and Biomechanical Role of Transglutaminase-2 in the Tumour Microenvironment. Cancers, 13(11), 2788. https://doi.org/10.3390/cancers13112788






