(Endo)Cannabinoids and Gynaecological Cancers
Simple Summary
Abstract
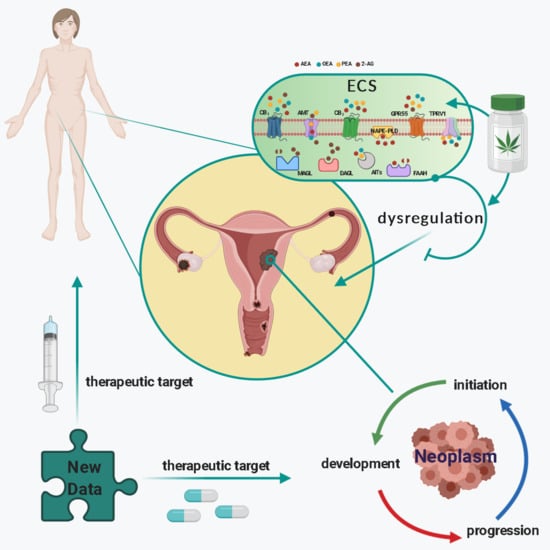
1. Introduction
1.1. Cannabis and Endocannabinoids and the Discovery of the Endocannabinoid System
1.2. The Endocannabinoid System: A Multifaceted Network
1.3. ECS in Female Tissues and Reproductive Events
1.4. The Endocannabinoid System in Relation to Normal Gynaecological Tissues
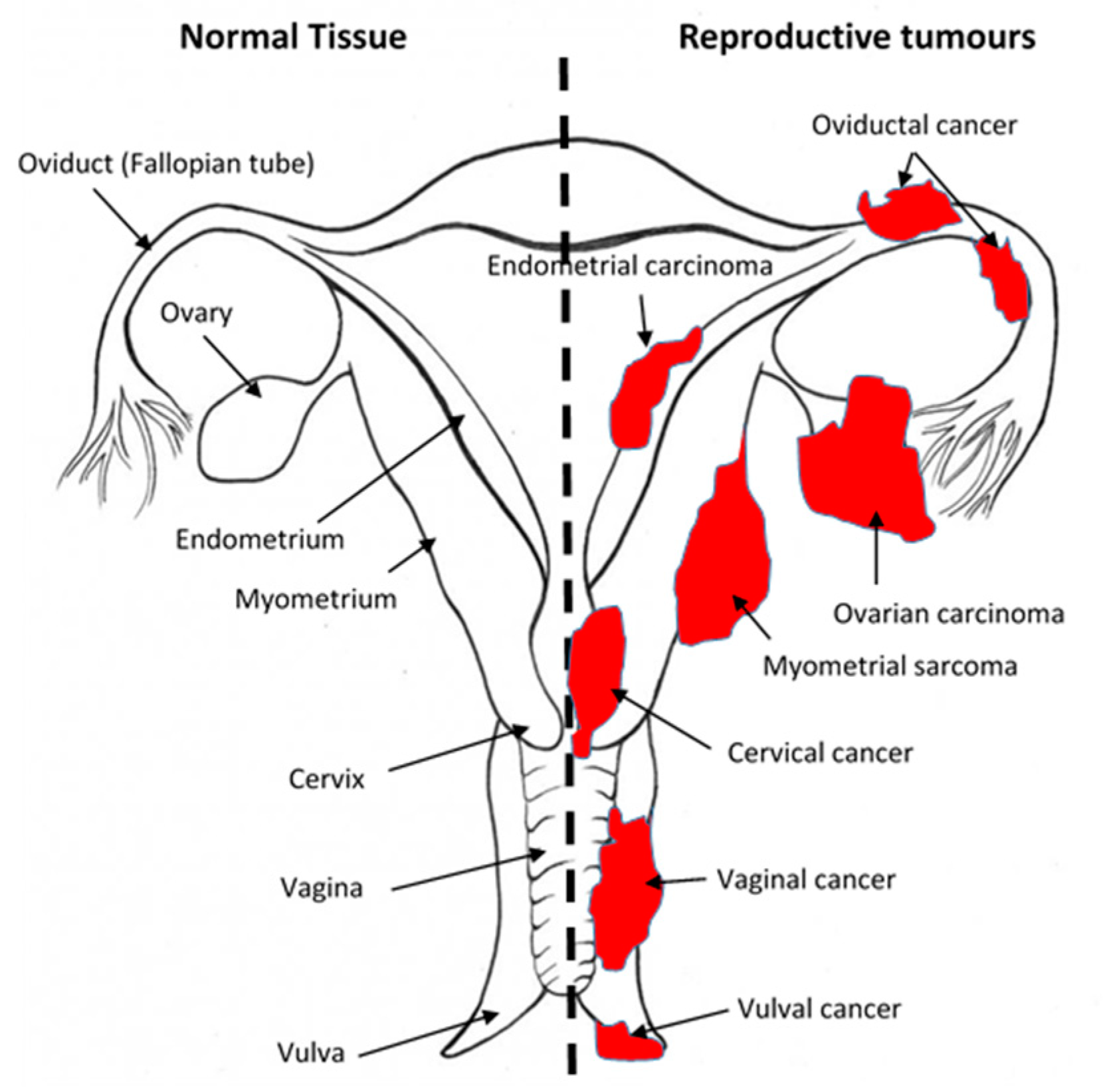
1.5. An Overview of Gynaecological Cancers
1.6. The Effects of Cannabinoids on Gynaecological Cancers
1.6.1. Cannabinoids and Ovarian Cancer
1.6.2. Cannabinoids and Fallopian Tube Cancer
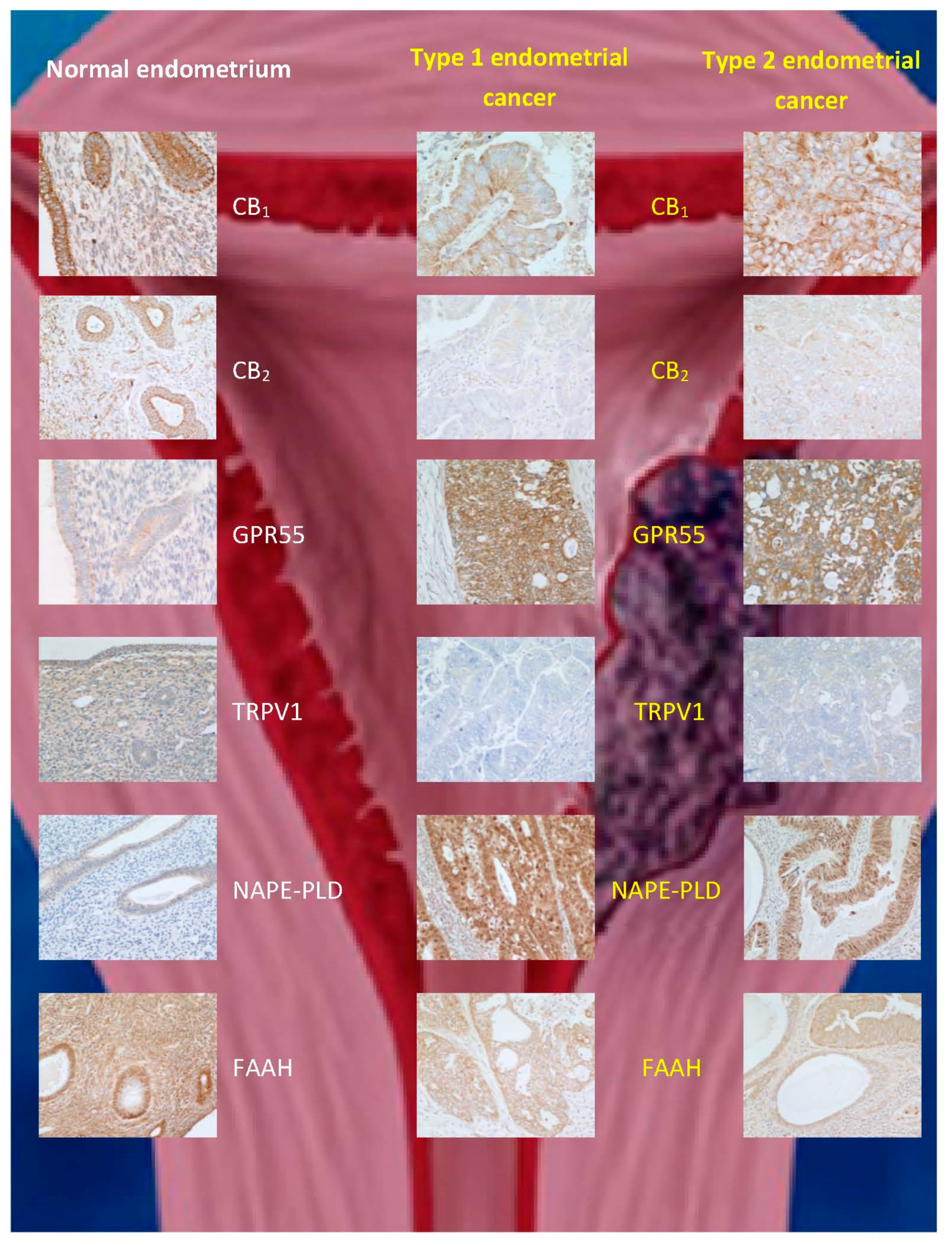
1.6.3. Cannabinoids and Endometrial Cancer
1.6.4. Cannabinoids and Cervical Cancer
1.6.5. Cannabinoids and Vaginal Cancer
1.6.6. Cannabinoids and Vulvar Cancer
1.6.7. Cannabinoids and Choriocarcinoma
2. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Schilling, S.; Melzer, R.; McCabe, P.F. Cannabis sativa. Curr. Biol. 2020, 30, R8–R9. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Hanus, L. A historical overview of chemical research on cannabinoids. Chem. Phys. Lipids 2000, 108, 1–13. [Google Scholar] [CrossRef]
- Brand, E.J.; Zhao, Z. Cannabis in Chinese medicine: Are some traditional indications referenced in ancient literature related to cannabinoids? Front. Pharmacol. 2017, 8, 108. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M.; Bab, I.; Biro, T.; Cabral, G.A.; Dey, S.K.; Di Marzo, V.; Konje, J.C.; Kunos, G.; Mechoulam, R.; Pacher, P.; et al. Endocannabinoid signaling at the periphery: 50 years after THC. Trends Pharmacol. Sci. 2015, 36, 277–296. [Google Scholar] [CrossRef] [PubMed]
- Pellati, F.; Borgonetti, V.; Brighenti, V.; Biagi, M.; Benvenuti, S.; Corsi, L. Cannabis sativa L. and nonpsychoactive cannabinoids: Their chemistry and role against oxidative stress, inflammation, and cancer. Biomed. Res. Int. 2018, 2018, 1691428. [Google Scholar] [CrossRef]
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561–564. [Google Scholar] [CrossRef]
- Munro, S.; Thomas, K.L.; Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993, 365, 61–65. [Google Scholar] [CrossRef]
- Devane, W.A.; Hanus, L.; Breuer, A.; Pertwee, R.G.; Stevenson, L.A.; Griffin, G.; Gibson, D.; Mandelbaum, A.; Etinger, A.; Mechoulam, R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 1992, 258, 1946–1949. [Google Scholar] [CrossRef]
- Mechoulam, R.; Ben-Shabat, S.; Hanus, L.; Ligumsky, M.; Kaminski, N.E.; Schatz, A.R.; Gopher, A.; Almog, S.; Martin, B.R.; Compton, D.R.; et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharmacol. 1995, 50, 83–90. [Google Scholar] [CrossRef]
- Pandey, R.; Mousawy, K.; Nagarkatti, M.; Nagarkatti, P. Endocannabinoids and immune regulation. Pharmacol. Res. 2009, 60, 85–92. [Google Scholar] [CrossRef]
- Bab, I.; Ofek, O.; Tam, J.; Rehnelt, J.; Zimmer, A. Endocannabinoids and the regulation of bone metabolism. J. Neuroendocrinol. 2008, 20 (Suppl. 1), 69–74. [Google Scholar] [CrossRef]
- Cecconi, S.; Rapino, C.; Di Nisio, V.; Rossi, G.; Maccarrone, M. The (endo)cannabinoid signaling in female reproduction: What are the latest advances? Prog. Lipid. Res. 2020, 77, 101019. [Google Scholar] [CrossRef] [PubMed]
- Silver, R.J. The endocannabinoid system of animals. Animals 2019, 9, 686. [Google Scholar] [CrossRef]
- Casajuana Köguel, C.; López-Pelayo, H.; Balcells-Olivero, M.M.; Colom, J.; Gual, A. Psychoactive constituents of cannabis and their clinical implications: A systematic review. Adicciones 2018, 30, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Leuti, A.; Fazio, D.; Fava, M.; Piccoli, A.; Oddi, S.; Maccarrone, M. Bioactive lipids, inflammation and chronic diseases. Adv. Drug. Deliv. Rev. 2020. [Google Scholar] [CrossRef] [PubMed]
- Malfitano, A.M.; Basu, S.; Maresz, K.; Bifulco, M.; Dittel, B.N. What we know and do not know about the cannabinoid receptor 2 (CB2). Semin. Immunol. 2014, 26, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M. Missing pieces to the endocannabinoid puzzle. Trends Mol. Med. 2020, 26, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.A. The enigmatic pharmacology of GPR55. Trends Pharmacol. Sci. 2009, 30, 156–163. [Google Scholar] [CrossRef]
- Syed, S.K.; Bui, H.H.; Beavers, L.S.; Farb, T.B.; Ficorilli, J.; Chesterfield, A.K.; Kuo, M.S.; Bokvist, K.; Barrett, D.G.; Efanov, A.M. Regulation of GPR119 receptor activity with endocannabinoid-like lipids. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E1469–E1478. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid ligands targeting TRP channels. Front. Mol. Neurosci. 2018, 11, 487. [Google Scholar] [CrossRef]
- Bohonyi, N.; Pohoczky, K.; Szalontai, B.; Perkecz, A.; Kovacs, K.; Kajtar, B.; Orban, L.; Varga, T.; Szegedi, S.; Bodis, J.; et al. Local upregulation of transient receptor potential ankyrin 1 and transient receptor potential vanilloid 1 ion channels in rectosigmoid deep infiltrating endometriosis. Mol. Pain 2017, 13, 1744806917705564. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, K.; Held, K.; Van Bree, R.; Meuleman, C.; Peeraer, K.; Tomassetti, C.; Voets, T.; D’Hooghe, T.; Vriens, J. Functional expression of transient receptor potential channels in human endometrial stromal cells during the luteal phase of the menstrual cycle. Hum. Reprod. 2015, 30, 1421–1436. [Google Scholar] [CrossRef] [PubMed]
- Ducza, E.; Csanyi, A.; Szoke, E.; Pohoczky, K.; Hajagos-Toth, J.; Kothencz, A.; Tiszai, Z.; Gaspar, R. Significance of transient receptor potential vanilloid 4 and aquaporin 5 co-expression in the rat uterus at term. Heliyon 2019, 5, e02697. [Google Scholar] [CrossRef]
- Han, J.H.; Choi, H.K.; Kim, S.J. Topical TRPM8 agonist (icilin) relieved vulva pruritus originating from lichen sclerosus et atrophicus. Acta Derm. Venereol. 2012, 92, 561–562. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wu, Y.T.; Zhu, Q.; Zhang, H.Y.; Huang, Z.; Zhang, D.; Qi, H.; Liang, G.L.; He, X.Q.; Wang, X.F.; et al. TRPV4 is involved in levonorgestrel-induced reduction in oviduct ciliary beating. J. Pathol. 2019, 248, 77–87. [Google Scholar] [CrossRef]
- Meseguer, V.; Karashima, Y.; Talavera, K.; D’Hoedt, D.; Donovan-Rodriguez, T.; Viana, F.; Nilius, B.; Voets, T. Transient receptor potential channels in sensory neurons are targets of the antimycotic agent clotrimazole. J. Neurosci. 2008, 28, 576–586. [Google Scholar] [CrossRef]
- Persoons, E.; Hennes, A.; De Clercq, K.; Van Bree, R.; Vriens, G.; Peterse, D.; Vanhie, A.; Meuleman, C.; Voets, T. Functional expression of TRP ion channels in endometrial stromal cells of endometriosis patients. Int. J. Mol. Sci. 2018, 19, 2467. [Google Scholar] [CrossRef]
- Teilmann, S.C.; Byskov, A.G.; Pedersen, P.A.; Wheatley, D.N.; Pazour, G.J.; Christensen, S.T. Localization of transient receptor potential ion channels in primary and motile cilia of the female murine reproductive organs. Mol. Reprod. Dev. 2005, 71, 444–452. [Google Scholar] [CrossRef]
- Tingaker, B.K.; Ekman-Ordeberg, G.; Facer, P.; Irestedt, L.; Anand, P. Influence of pregnancy and labor on the occurrence of nerve fibers expressing the capsaicin receptor TRPV1 in human corpus and cervix uteri. Reprod. Biol. Endocrinol. 2008, 6, 8. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Marczylo, T.H.; Konje, J.C. New insights of uterine leiomyoma pathogenesis: Endocannabinoid system. Med. Sci. Monit. Basic Res. 2019, 25, 76–87. [Google Scholar] [CrossRef]
- Dong, K.; Zhang, M.X.; Liu, Y.; Su, X.L.; Chen, B.; Zhang, X.L. Peroxisome proliferator-activated receptor alpha expression changes in human pregnant myometrium. Reprod. Sci. 2013, 20, 654–660. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Enioutina, E.Y.; Visic, V.D.; Daynes, R.A. Enhancement of common mucosal immunity in aged mice following their supplementation with various antioxidants. Vaccine 2000, 18, 2381–2393. [Google Scholar] [CrossRef]
- Nickkho-Amiry, M.; McVey, R.; Holland, C. Peroxisome proliferator-activated receptors modulate proliferation and angiogenesis in human endometrial carcinoma. Mol. Cancer Res. 2012, 10, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Tao, T.; Wang, Y.; Xu, B.; Mao, X.; Sun, Y.; Liu, W. Role of adiponectin/peroxisome proliferator-activated receptor alpha signaling in human chorionic gonadotropin-induced estradiol synthesis in human luteinized granulosa cells. Mol. Cell Endocrinol. 2019, 493, 110450. [Google Scholar] [CrossRef] [PubMed]
- Tsibris, J.C.; Porter, K.B.; Jazayeri, A.; Tzimas, G.; Nau, H.; Huang, H.; Kuparadze, K.; Porter, G.W.; O’Brien, W.F.; Spellacy, W.N. Human uterine leiomyomata express higher levels of peroxisome proliferator-activated receptor gamma, retinoid X receptor alpha, and all-trans retinoic acid than myometrium. Cancer Res. 1999, 59, 5737–5744. [Google Scholar] [PubMed]
- Yin, Y.; Lin, C.; Veith, G.M.; Chen, H.; Dhandha, M.; Ma, L. Neonatal diethylstilbestrol exposure alters the metabolic profile of uterine epithelial cells. Dis. Model. Mech. 2012, 5, 870–880. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Hou, X.; Gao, S. Stimulation of peroxisome proliferator-activated receptor gamma inhibits estrogen receptor alpha transcriptional activity in endometrial carcinoma cells. Oncol. Rep. 2015, 33, 1227–1234. [Google Scholar] [CrossRef]
- Walker, O.S.; Holloway, A.C.; Raha, S. The role of the endocannabinoid system in female reproductive tissues. J. Ovarian Res. 2019, 12, 3. [Google Scholar] [CrossRef]
- O’Sullivan, S.E. An update on PPAR activation by cannabinoids. Br. J. Pharmacol. 2016, 173, 1899–1910. [Google Scholar] [CrossRef]
- Selvy, P.E.; Lavieri, R.R.; Lindsley, C.W.; Brown, H.A. Phospholipase D: Enzymology, functionality, and chemical modulation. Chem. Rev. 2011, 111, 6064–6119. [Google Scholar] [CrossRef]
- Nicolussi, S.; Gertsch, J. Endocannabinoid transport revisited. Vitam. Horm 2015, 98, 441–485. [Google Scholar] [CrossRef] [PubMed]
- Ligresti, A.; De Petrocellis, L.; Hernan Perez de la Ossa, D.; Aberturas, R.; Cristino, L.; Moriello, A.S.; Finizio, A.; Gil, M.E.; Torres, A.I.; Molpeceres, J.; et al. Exploiting nanotechnologies and TRPV1 channels to investigate the putative anandamide membrane transporter. PLoS ONE 2010, 5, e10239. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M.; Dainese, E.; Oddi, S. Intracellular trafficking of anandamide: New concepts for signaling. Trends Biochem. Sci. 2010, 35, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M. Metabolism of the endocannabinoid anandamide: Open questions after 25 years. Front. Mol. Neurosci. 2017, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- El-Talatini, M.R.; Taylor, A.H.; Elson, J.C.; Brown, L.; Davidson, A.C.; Konje, J.C. Localisation and function of the endocannabinoid system in the human ovary. PLoS ONE 2009, 4, e4579. [Google Scholar] [CrossRef] [PubMed]
- Horne, A.W.; Phillips, J.A., 3rd; Kane, N.; Lourenco, P.C.; McDonald, S.E.; Williams, A.R.; Simon, C.; Dey, S.K.; Critchley, H.O. CB1 expression is attenuated in Fallopian tube and decidua of women with ectopic pregnancy. PLoS ONE 2008, 3, e3969. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.H.; Abbas, M.S.; Habiba, M.A.; Konje, J.C. Histomorphometric evaluation of cannabinoid receptor and anandamide modulating enzyme expression in the human endometrium through the menstrual cycle. Histochem. Cell Biol. 2010, 133, 557–565. [Google Scholar] [CrossRef]
- Taylor, A.H.; Finney, M.; Lam, P.M.; Konje, J.C. Modulation of the endocannabinoid system in viable and non-viable first trimester pregnancies by pregnancy-related hormones. Reprod. Biol. Endocrinol. 2011, 9, 152. [Google Scholar] [CrossRef]
- Gebeh, A.K.; Willets, J.M.; Marczylo, E.L.; Taylor, A.H.; Konje, J.C. Ectopic pregnancy is associated with high anandamide levels and aberrant expression of FAAH and CB1 in fallopian tubes. J. Clin. Endocrinol. Metab. 2012, 97, 2827–2835. [Google Scholar] [CrossRef]
- El-Talatini, M.R.; Taylor, A.H.; Konje, J.C. The relationship between plasma levels of the endocannabinoid, anandamide, sex steroids, and gonadotrophins during the menstrual cycle. Fertil. Steril. 2010, 93, 1989–1996. [Google Scholar] [CrossRef]
- Maia, J.; Fonseca, B.M.; Teixeira, N.; Correia-da-Silva, G. The fundamental role of the endocannabinoid system in endometrium and placenta: Implications in pathophysiological aspects of uterine and pregnancy disorders. Hum. Reprod. Update 2020. [Google Scholar] [CrossRef] [PubMed]
- Habayeb, O.M.; Taylor, A.H.; Bell, S.C.; Taylor, D.J.; Konje, J.C. Expression of the endocannabinoid system in human first trimester placenta and its role in trophoblast proliferation. Endocrinology 2008, 149, 5052–5060. [Google Scholar] [CrossRef]
- Karasu, T.; Marczylo, T.H.; Marczylo, E.L.; Taylor, A.H.; Oloto, E.; Konje, J.C. The effect of mifepristone (RU486) on the endocannabinoid system in human plasma and first-trimester trophoblast of women undergoing termination of pregnancy. J. Clin. Endocrinol. Metab. 2014, 99, 871–880. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fugedi, G.; Molnar, M.; Rigo, J., Jr.; Schonleber, J.; Kovalszky, I.; Molvarec, A. Increased placental expression of cannabinoid receptor 1 in preeclampsia: An observational study. BMC Pregnancy Childbirth 2014, 14, 395. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, B.M.; Cunha, S.C.; Goncalves, D.; Mendes, A.; Braga, J.; Correia-da-Silva, G.; Teixeira, N.A. Decidual NK cell-derived conditioned medium from miscarriages affects endometrial stromal cell decidualisation: Endocannabinoid anandamide and tumour necrosis factor-alpha crosstalk. Hum. Reprod. 2020, 35, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Habayeb, O.M.; Taylor, A.H.; Finney, M.; Evans, M.D.; Konje, J.C. Plasma anandamide concentration and pregnancy outcome in women with threatened miscarriage. JAMA 2008, 299, 1135–1136. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M.; Valensise, H.; Bari, M.; Lazzarin, N.; Romanini, C.; Finazzi-Agro, A. Relation between decreased anandamide hydrolase concentrations in human lymphocytes and miscarriage. Lancet 2000, 355, 1326–1329. [Google Scholar] [CrossRef]
- Helliwell, R.J.; Chamley, L.W.; Blake-Palmer, K.; Mitchell, M.D.; Wu, J.; Kearn, C.S.; Glass, M. Characterization of the endocannabinoid system in early human pregnancy. J. Clin. Endocrinol. Metab. 2004, 89, 5168–5174. [Google Scholar] [CrossRef] [PubMed]
- Schuel, H.; Burkman, L.J.; Lippes, J.; Crickard, K.; Forester, E.; Piomelli, D.; Giuffrida, A. N-Acylethanolamines in human reproductive fluids. Chem. Phys. Lipids 2002, 121, 211–227. [Google Scholar] [CrossRef]
- Kisgeropoulis, E. Inhibition of Ovarian Cancer Cell Proliferation by Oleoylethanolamide and Its Metabolically Stable Analog AM3102. Ohio State, Researchgate.net. 2013. Available online: https://www.researchgate.net/publication/278025965_Inhibition_of_Ovarian_Cancer_Cell_Proliferation_by_Oleoylethanolamide_and_its_Metabolically_Stable_Analog_AM3102 (accessed on 23 December 2020).
- Bradshaw, H.B.; Allard, C. Endogenous cannabinoid production in the rat female reproductive tract is regulated by changes in the hormonal milieu. Pharmaceuticals 2011, 4, 933–949. [Google Scholar] [CrossRef]
- De Domenico, E.; Todaro, F.; Rossi, G.; Dolci, S.; Geremia, R.; Rossi, P.; Grimaldi, P. Overactive type 2 cannabinoid receptor induces meiosis in fetal gonads and impairs ovarian reserve. Cell Death Dis. 2017, 8, e3085. [Google Scholar] [CrossRef] [PubMed]
- Han, G.H.; Chay, D.B.; Nam, S.; Cho, H.; Chung, J.Y.; Kim, J.H. Prognostic significance of transient receptor potential vanilloid type 1 (TRPV1) and phosphatase and tension homolog (PTEN) in epithelial ovarian cancer. Cancer Genom. Proteom. 2020, 17, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, R.; Maffucci, T.; Falasca, M. The putative cannabinoid receptor GPR55 defines a novel autocrine loop in cancer cell proliferation. Oncogene 2011, 30, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Nomura, D.K.; Long, J.Z.; Niessen, S.; Hoover, H.S.; Ng, S.W.; Cravatt, B.F. Monoacylglycerol lipase regulates a fatty acid network that promotes cancer pathogenesis. Cell 2010, 140, 49–61. [Google Scholar] [CrossRef]
- Gervasi, M.G.; Rapanelli, M.; Ribeiro, M.L.; Farina, M.; Billi, S.; Franchi, A.M.; Perez Martinez, S. The endocannabinoid system in bull sperm and bovine oviductal epithelium: Role of anandamide in sperm-oviduct interaction. Reproduction 2009, 137, 403–414. [Google Scholar] [CrossRef]
- Gervasi, M.G.; Marczylo, T.H.; Lam, P.M.; Rana, S.; Franchi, A.M.; Konje, J.C.; Perez-Martinez, S. Anandamide levels fluctuate in the bovine oviduct during the oestrous cycle. PLoS ONE 2013, 8, e72521. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Marczylo, T.H.; Maccarrone, M.; Konje, J.C. Identification of novel predictive biomarkers for endometrial malignancies: N-Acylethanolamines. Front. Oncol. 2019, 9, 430. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.; Willets, J.; Marczylo, T.; Brown, L.; Davies, Q.; Moss, E.; Konje, J. Effect of anandamide on endometrial adenocarcinoma (Ishikawa) cell numbers: Implications for endometrial cancer therapy. Lancet 2015, 385 (Suppl. 1), S20. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Davies, Q.; Moss, E.L.; Konje, J.C. Optimisation of uniplex and duplex reactions is not required for real-time PCR amplification of target genes in endometrial cancer. Insights Obstet. Gynaecol. 2017, 1, 5.1. Available online: https://www.researchgate.net/publication/347910796 (accessed on 24 December 2020).
- Gentilini, D.; Besana, A.; Vigano, P.; Dalino, P.; Vignali, M.; Melandri, M.; Busacca, M.; Di Blasio, A.M. Endocannabinoid system regulates migration of endometrial stromal cells via cannabinoid receptor 1 through the activation of PI3K and ERK1/2 pathways. Fertil. Steril. 2010, 93, 2588–2593. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Konje, J.C. Cannabinoid receptor expression in estrogen-dependent and estrogen-independent endometrial cancer. J. Recept. Signal. Transduct. Res. 2018, 38, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Ayakannu, T.; Taylor, A.H.; Willets, J.M.; Brown, L.; Lambert, D.G.; McDonald, J.; Davies, Q.; Moss, E.L.; Konje, J.C. Validation of endogenous control reference genes for normalizing gene expression studies in endometrial carcinoma. Mol. Hum. Reprod 2015, 21, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Guida, M.; Ligresti, A.; De Filippis, D.; D’Amico, A.; Petrosino, S.; Cipriano, M.; Bifulco, G.; Simonetti, S.; Orlando, P.; Insabato, L.; et al. The levels of the endocannabinoid receptor CB2 and its ligand 2-arachidonoylglycerol are elevated in endometrial carcinoma. Endocrinology 2010, 151, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Risinger, J.I.; Maxwell, G.L.; Chandramouli, G.V.; Jazaeri, A.; Aprelikova, O.; Patterson, T.; Berchuck, A.; Barrett, J.C. Microarray analysis reveals distinct gene expression profiles among different histologic types of endometrial cancer. Cancer Res. 2003, 63, 6–11. [Google Scholar]
- Fonseca, B.M.; Correia-da-Silva, G.; Teixeira, N.A. Cannabinoid-induced cell death in endometrial cancer cells: Involvement of TRPV1 receptors in apoptosis. J. Physiol. Biochem. 2018, 74, 261–272. [Google Scholar] [CrossRef]
- Ayakannu, T. Investigation of Endocannabinoid Signalling and Their Regulations in Endometrial Carcinoma. Available online: https://leicester.figshare.com/articles/Investigation_of_Endocannabinoid_System_Signalling_Pathways_and_Their_Regulations_in_Endometrial_Carcinoma/10216328 (accessed on 31 October 2020).
- Ayakannu, T.; Taylor, A.H.; Bari, M.; Mastrangelo, N.; Maccarrone, M.; Konje, J.C. Expression and function of the endocannabinoid modulating enzymes fatty acid amide hydrolase and N-acylphosphatidylethanolamine-specific phospholipase D in endometrial carcinoma. Front. Oncol. 2019, 9, 1363. [Google Scholar] [CrossRef]
- Maccarrone, M.; DeFelici, M.; Klinger, F.G.; Battista, N.; Fezza, F.; Dainese, E.; Siracusa, G.; Finazzi-Agro, A. Mouse blastocysts release a lipid which activates anandamide hydrolase in intact uterus. Mol. Hum. Reprod. 2004, 10, 215–221. [Google Scholar] [CrossRef]
- Habayeb, O.; Taylor, A.; Bharkhada, R.; Taylor, D.; Bell, S.; Konje, J. Immunohistochemical localisation of cannabinoid receptor CB1 and fatty acid amide hydrolase (FAAH) in maternal and fetal tissues. Proceedings of British Congress of Obstetrics & Gynaecology Abstracts Book; Royal College of Obstetricians and Gynecologists: London, UK, 2004; p. 10. [Google Scholar]
- Wang, H.; Xie, H.; Sun, X.; Kingsley, P.J.; Marnett, L.J.; Cravatt, B.F.; Dey, S.K. Differential regulation of endocannabinoid synthesis and degradation in the uterus during embryo implantation. Prostaglandins Other Lipid Mediat. 2007, 83, 62–74. [Google Scholar] [CrossRef]
- Brighton, P.J.; Marczylo, T.H.; Rana, S.; Konje, J.C.; Willets, J.M. Characterization of the endocannabinoid system, CB(1) receptor signalling and desensitization in human myometrium. Br. J. Pharmacol. 2011, 164, 1479–1494. [Google Scholar] [CrossRef][Green Version]
- Shen, X.; Duan, H.; Wang, S.; Hong, W.; Wang, Y.Y.; Lin, S.L. Expression of cannabinoid receptors in myometrium and its correlation with dysmenorrhea in adenomyosis. Reprod. Sci. 2019, 26, 1618–1625. [Google Scholar] [CrossRef]
- Contassot, E.; Tenan, M.; Schnuriger, V.; Pelte, M.F.; Dietrich, P.Y. Arachidonyl ethanolamide induces apoptosis of uterine cervix cancer cells via aberrantly expressed vanilloid receptor-1. Gynecol. Oncol. 2004, 93, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Eichele, K.; Ramer, R.; Hinz, B. R(+)-methanandamide-induced apoptosis of human cervical carcinoma cells involves a cyclooxygenase-2-dependent pathway. Pharm Res. 2009, 26, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Li, J.; Zhao, T.; Wang, H.; Lai, G. [Over-expression of cannabinoid receptor 2 induces the apoptosis of cervical carcinoma Caski cells]. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2015, 31, 758–762. [Google Scholar] [PubMed]
- Han, G.H.; Chay, D.B.; Nam, S.; Cho, H.; Chung, J.Y.; Kim, J.H. The combination of transient receptor potential vanilloid type 1 (TRPV1) and phosphatase and tension homolog (PTEN) is an effective prognostic biomarker in cervical cancer. Int. J. Gynecol. Pathol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Milando, R.; Friedman, A. Cannabinoids: Potential role in inflammatory and neoplastic skin diseases. Am. J. Clin. Dermatol. 2019, 20, 167–180. [Google Scholar] [CrossRef]
- Marczylo, T.H.; Lam, P.M.; Amoako, A.A.; Konje, J.C. Anandamide levels in human female reproductive tissues: Solid-phase extraction and measurement by ultraperformance liquid chromatography tandem mass spectrometry. Anal. Biochem. 2010, 400, 155–162. [Google Scholar] [CrossRef]
- Brocato, B.; Zoerner, A.A.; Janjetovic, Z.; Skobowiat, C.; Gupta, S.; Moore, B.M., 2nd; Slominski, A.; Zhang, J.; Schenone, M.; Phinehas, R.; et al. Endocannabinoid crosstalk between placenta and maternal fat in a baboon model (Papio spp.) of obesity. Placenta 2013, 34, 983–989. [Google Scholar] [CrossRef]
- Park, B.; Gibbons, H.M.; Mitchell, M.D.; Glass, M. Identification of the CB1 cannabinoid receptor and fatty acid amide hydrolase (FAAH) in the human placenta. Placenta 2003, 24, 990–995. [Google Scholar] [CrossRef]
- Fonseca, B.M.; Teixeira, N.A.; Almada, M.; Taylor, A.H.; Konje, J.C.; Correia-da-Silva, G. Modulation of the novel cannabinoid receptor-GPR55-during rat fetoplacental development. Placenta 2011, 32, 462–469. [Google Scholar] [CrossRef]
- Fonseca, B.M.; Correia-da-Silva, G.; Taylor, A.H.; Lam, P.M.; Marczylo, T.H.; Bell, S.C.; Konje, J.C.; Teixeira, N.A. The endocannabinoid 2-arachidonoylglycerol (2-AG) and metabolizing enzymes during rat fetoplacental development: A role in uterine remodelling. Int. J. Biochem. Cell Biol. 2010, 42, 1884–1892. [Google Scholar] [CrossRef]
- Costa, M.A.; Fonseca, B.M.; Keating, E.; Teixeira, N.A.; Correia-da-Silva, G. 2-arachidonoylglycerol effects in cytotrophoblasts: Metabolic enzymes expression and apoptosis in BeWo cells. Reproduction 2014, 147, 301–311. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Willets, J.M.; Konje, J.C. The evolving role of the endocannabinoid system in gynaecological cancer. Hum. Reprod. Update 2015, 21, 517–535. [Google Scholar] [CrossRef]
- Bagavandoss, P.; Grimshaw, S. Temporal and spatial distribution of the cannabinoid receptors (CB1, CB2) and fatty acid amide hydroxylase in the rat ovary. Anat. Rec. (Hoboken) 2010, 293, 1425–1432. [Google Scholar] [CrossRef]
- Cui, N.; Feng, X.; Zhao, Z.; Zhang, J.; Xu, Y.; Wang, L.; Hao, G. Restored plasma anandamide and endometrial expression of fatty acid amide hydrolase in women with polycystic ovary syndrome by the combination use of Diane-35 and metformin. Clin. Ther. 2017, 39, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Pirone, A.; Lenzi, C.; Briganti, A.; Abbate, F.; Levanti, M.; Abramo, F.; Miragliotta, V. Spatial distribution of cannabinoid receptor 1 and fatty acid amide hydrolase in the cat ovary and oviduct. Acta Histochem. 2017, 119, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Schuel, H. Tuning the oviduct to the anandamide tone. J. Clin. Investig. 2006, 116, 2087–2090. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Guo, Y.; Wang, D.; Kingsley, P.J.; Marnett, L.J.; Das, S.K.; DuBois, R.N.; Dey, S.K. Aberrant cannabinoid signaling impairs oviductal transport of embryos. Nat. Med. 2004, 10, 1074–1080. [Google Scholar] [CrossRef] [PubMed]
- Bambang, K.N.; Karasu, T.; Gebeh, A.; Taylor, A.H.; Marczylo, T.H.; Lam, P.; Willets, J.M.; Konje, J.C. From fertilisation to implantation in mammalian pregnancy-modulation of early human reproduction by the endocannabinoid system. Pharmaceuticals 2010, 3, 2910–2929. [Google Scholar] [CrossRef]
- Abolghasemi, A.; Dirandeh, E.; Ansari Pirsaraei, Z.; Shohreh, B. Dietary conjugated linoleic acid supplementation alters the expression of genes involved in the endocannabinoid system in the bovine endometrium and increases plasma progesterone concentrations. Theriogenology 2016, 86, 1453–1459. [Google Scholar] [CrossRef]
- Dirandeh, E.; Ghaffari, J. Effects of feeding a source of omega-3 fatty acid during the early postpartum period on the endocannabinoid system in the bovine endometrium. Theriogenology 2018, 121, 141–146. [Google Scholar] [CrossRef]
- Scotchie, J.G.; Savaris, R.F.; Martin, C.E.; Young, S.L. Endocannabinoid regulation in human endometrium across the menstrual cycle. Reprod. Sci. 2015, 22, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Duan, H.; Wang, S.; Gan, L.; Xu, Q.; Li, J.J. Decreased expression of cannabinoid receptors in the eutopic and ectopic endometrium of patients with adenomyosis. Biomed. Res. Int. 2019, 2019, 5468954. [Google Scholar] [CrossRef] [PubMed]
- Neradugomma, N.K.; Drafton, K.; Mor, G.G.; Mao, Q. Marijuana-derived cannabinoids inhibit uterine endometrial stromal cell decidualization and compromise trophoblast-endometrium cross-talk. Reprod. Toxicol. 2019, 87, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Neradugomma, N.K.; Drafton, K.; O’Day, D.R.; Liao, M.Z.; Han, L.W.; Glass, I.A.; Mao, Q. Marijuana use differentially affects cannabinoid receptor expression in early gestational human endometrium and placenta. Placenta 2018, 66, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.H.; Amoako, A.A.; Bambang, K.; Karasu, T.; Gebeh, A.; Lam, P.M.; Marzcylo, T.H.; Konje, J.C. Endocannabinoids and pregnancy. Clin. Chim. Acta 2010, 411, 921–930. [Google Scholar] [CrossRef]
- Rapino, C.; Battista, N.; Bari, M.; Maccarrone, M. Endocannabinoids as biomarkers of human reproduction. Hum. Reprod. Update 2014, 20, 501–516. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M. Endocannabinoids: Friends and foes of reproduction. Prog. Lipid Res. 2009, 48, 344–354. [Google Scholar] [CrossRef]
- Maccarrone, M. Endocannabinoids and reproductive biology. Hum. Reprod. 2009, 24, 1771. [Google Scholar] [CrossRef]
- Wang, H.; Dey, S.K.; Maccarrone, M. Jekyll and hyde: Two faces of cannabinoid signaling in male and female fertility. Endocr. Rev. 2006, 27, 427–448. [Google Scholar] [CrossRef]
- El-Talatini, M.R.; Taylor, A.H.; Konje, J.C. Fluctuation in anandamide levels from ovulation to early pregnancy in in-vitro fertilization-embryo transfer women, and its hormonal regulation. Hum. Reprod. 2009, 24, 1989–1998. [Google Scholar] [CrossRef]
- Sankaranarayanan, R.; Ferlay, J. Worldwide burden of gynaecological cancer: The size of the problem. Best Pract. Res. Clin. Obstet. Gynaecol. 2006, 20, 207–225. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G.; Howlett, A.C.; Abood, M.E.; Alexander, S.P.; Di Marzo, V.; Elphick, M.R.; Greasley, P.J.; Hansen, H.S.; Kunos, G.; Mackie, K.; et al. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: Beyond CB(1) and CB(2). Pharmacol. Rev. 2010, 62, 588–631. [Google Scholar] [CrossRef] [PubMed]
- Jove, M.; Gatius, S.; Yeramian, A.; Portero-Otin, M.; Eritja, N.; Santacana, M.; Colas, E.; Ruiz, M.; Pamplona, R.; Matias-Guiu, X. Metabotyping human endometrioid endometrial adenocarcinoma reveals an implication of endocannabinoid metabolism. Oncotarget 2016, 7, 52364–52374. [Google Scholar] [CrossRef]
- Leconte, M.; Nicco, C.; Ngo, C.; Arkwright, S.; Chereau, C.; Guibourdenche, J.; Weill, B.; Chapron, C.; Dousset, B.; Batteux, F. Antiproliferative effects of cannabinoid agonists on deep infiltrating endometriosis. Am. J. Pathol. 2010, 177, 2963–2970. [Google Scholar] [CrossRef]
- Sun, X.; Dey, S.K. Endocannabinoid signaling in female reproduction. ACS Chem. Neurosci. 2012, 3, 349–355. [Google Scholar] [CrossRef]
- Wang, H.; Xie, H.; Dey, S.K. Endocannabinoid signaling directs periimplantation events. AAPS J. 2006, 8, E425–E432. [Google Scholar] [CrossRef]
- Bachkangi, P.; Taylor, A.H.; Bari, M.; Maccarrone, M.; Konje, J.C. Prediction of preterm labour from a single blood test: The role of the endocannabinoid system in predicting preterm birth in high-risk women. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 243, 1–6. [Google Scholar] [CrossRef]
- Bari, M.; Battista, N.; Pirazzi, V.; Maccarrone, M. The manifold actions of endocannabinoids on female and male reproductive events. Front. Biosci. (Landmark Ed.) 2011, 16, 498–516. [Google Scholar] [CrossRef]
- Habayeb, O.M.; Taylor, A.H.; Evans, M.D.; Cooke, M.S.; Taylor, D.J.; Bell, S.C.; Konje, J.C. Plasma levels of the endocannabinoid anandamide in women—A potential role in pregnancy maintenance and labor? J. Clin. Endocrinol. Metab. 2004, 89, 5482–5487. [Google Scholar] [CrossRef]
- McCluggage, W.G.; Oliva, E. Gynaecological pathology: Problematic areas, new concepts and emerging developments. Pathology 2018, 50, 119–121. [Google Scholar] [CrossRef]
- Ledford, L.R.C.; Lockwood, S. Scope and Epidemiology of Gynecologic Cancers: An Overview. Semin. Oncol. Nurs. 2019, 35, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Cancer Research UK. Cancer Research Statistics, Cancer Incidence and Mortality in the UK. Available online: http://www.cancerresearchuk.org/cancer-info/cancerstats/types/uterus/ (accessed on 2 September 2020).
- Canfell, K.; Kim, J.J.; Brisson, M.; Keane, A.; Simms, K.T.; Caruana, M.; Burger, E.A.; Martin, D.; Nguyen, D.T.N.; Bénard, É.; et al. Mortality impact of achieving WHO cervical cancer elimination targets: A comparative modelling analysis in 78 low-income and lower-middle-income countries. Lancet 2020, 395, 591–603. [Google Scholar] [CrossRef]
- Webb, P.M. Environmental (nongenetic) factors in gynecological cancers: Update and future perspectives. Future Oncol. 2015, 11, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Blake, E.A.; Machida, H.; Mandelbaum, R.S.; Roman, L.D.; Wright, J.D. Incidences and risk factors of metachronous vulvar, vaginal, and anal cancers after cervical cancer diagnosis. Gynecol. Oncol. 2018, 150, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Sobocińska, A.A.; Czarnecka, A.M.; Król, M.; Botta, B.; Szczylik, C. The Therapeutic Aspects of the Endocannabinoid System (ECS) for Cancer and their Development: From Nature to Laboratory. Curr. Pharm. Des. 2016, 22, 1756–1766. [Google Scholar] [CrossRef] [PubMed]
- Fraguas-Sánchez, A.I.; Martín-Sabroso, C.; Torres-Suárez, A.I. Insights into the effects of the endocannabinoid system in cancer: A review. Br. J. Pharmacol. 2018, 175, 2566–2580. [Google Scholar] [CrossRef]
- Brown, I.; Cascio, M.G.; Rotondo, D.; Pertwee, R.G.; Heys, S.D.; Wahle, K.W. Cannabinoids and omega-3/6 endocannabinoids as cell death and anticancer modulators. Prog. Lipid Res. 2013, 52, 80–109. [Google Scholar] [CrossRef]
- Suk, K.T.; Mederacke, I.; Gwak, G.Y.; Cho, S.W.; Adeyemi, A.; Friedman, R.; Schwabe, R.F. Opposite roles of cannabinoid receptors 1 and 2 in hepatocarcinogenesis. Gut 2016, 65, 1721–1732. [Google Scholar] [CrossRef]
- Green, M. Cannabis and Your Vagina: Here’s the 411 on Cannabis Suppositories. Available online: https://www.byrdie.com/how-to-use-cannabis-suppositories-4771875 (accessed on 26 June 2020).
- Harper-Gold, C. How to Get Your Vagina High. Available online: https://greenrushdaily.com/women/get-vagina-high/ (accessed on 26 June 2020).
- Lowry, J. Cannabinoids Can Cause Cell Death in Endometrial Cancer. Available online: https://www.cannahealth.org/cannabinoids-can-cause-cell-death-in-endometrial-cancer/ (accessed on 26 June 2020).
- Moore, G.R. Menopause: Cannabinoids & Sexual Health. Available online: https://www.foriawellness.com/blogs/learn/cbd-thc-sexual-health-menopause (accessed on 26 June 2020).
- Smith, D. Cannabis for Endometrial Cancer. Available online: https://cannabis.net/blog/medical/cannabis-for-endometrial-cancer (accessed on 26 June 2020).
- Trassoff-Jilg, V. Cannabinoids and Endometrial Cancer. Available online: https://waayb.com/cannabinoids-and-endometrial-cancer/ (accessed on 26 June 2020).
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef]
- Messalli, E.M.; Grauso, F.; Luise, R.; Angelini, A.; Rossiello, R. Cannabinoid receptor type 1 immunoreactivity and disease severity in human epithelial ovarian tumors. Am. J. Obstet. Gynecol. 2014, 211, P234.E1–P234.E6. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Xiao, Y.J.; Baudhuin, L.M.; Schwartz, B.M. The role and clinical applications of bioactive lysolipids in ovarian cancer. J. Soc. Gynecol. Investig. 2001, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, N.A.; Yang, J.; Trauger, S.A.; Nakayama, H.; Huang, L.; Strunk, D.; Moses, M.A.; Klagsbrun, M.; Bischoff, J.; Graier, W.F. The GPR 55 agonist, L-alpha-lysophosphatidylinositol, mediates ovarian carcinoma cell-induced angiogenesis. Br. J. Pharmacol. 2015, 172, 4107–4118. [Google Scholar] [CrossRef]
- Deng, H.; Li, W. Monoacylglycerol lipase inhibitors: Modulators for lipid metabolism in cancer malignancy, neurological and metabolic disorders. Acta Pharm. Sin. B 2020, 10, 582–602. [Google Scholar] [CrossRef] [PubMed]
- Fraguas-Sanchez, A.I.; Torres-Suarez, A.I.; Cohen, M.; Delie, F.; Bastida-Ruiz, D.; Yart, L.; Martin-Sabroso, C.; Fernandez-Carballido, A. PLGA nanoparticles for the intraperitoneal administration of CBD in the treatment of ovarian cancer: In Vitro and In Ovo assessment. Pharmaceutics 2020, 12, 439. [Google Scholar] [CrossRef]
- Gebeh, A.K.; Willets, J.M.; Bari, M.; Hirst, R.A.; Marczylo, T.H.; Taylor, A.H.; Maccarrone, M.; Konje, J.C. Elevated anandamide and related N-acylethanolamine levels occur in the peripheral blood of women with ectopic pregnancy and are mirrored by changes in peripheral fatty acid amide hydrolase activity. J. Clin Endocrinol. Metab. 2013, 98, 1226–1234. [Google Scholar] [CrossRef][Green Version]
- Bokhman, J.V. Two pathogenetic types of endometrial carcinoma. Gynecol. Oncol. 1983, 15, 10–17. [Google Scholar] [CrossRef]
- Leslie, K.K.; Thiel, K.W.; Goodheart, M.J.; De Geest, K.; Jia, Y.; Yang, S. Endometrial cancer. Obstet. Gynecol. Clin. N. Am. 2012, 39, 255–268. [Google Scholar] [CrossRef]
- Zhang, Y.; Zheng, W.; Shen, K.; Shen, W. 9-tetrahydrocannabinol inhibits epithelial-mesenchymal transition and metastasis by targeting matrix metalloproteinase-9 in endometrial cancer. Oncol. Lett. 2018, 15, 8527–8535. [Google Scholar] [CrossRef]
- Rosado, J. Endometrial Cancer. Available online: https://www.marijuanadoctors.com/conditions/endometrial-cancer/ (accessed on 26 June 2020).
- Freimuth, N.; Ramer, R.; Hinz, B. Antitumorigenic effects of cannabinoids beyond apoptosis. J. Pharmacol. Exp. Ther. 2010, 332, 336–344. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer [Internet]. GLOBOCAN 2012 v1.0 (2013): IARC Publications Website—Cancer Today (powered by GLOBOCAN 2018)–IARC CancerBase No. 15. Available online: https://publications.iarc.fr/577 (accessed on 24 December 2020).
- Ramer, R.; Merkord, J.; Rohde, H.; Hinz, B. Cannabidiol inhibits cancer cell invasion via upregulation of tissue inhibitor of matrix metalloproteinases-1. Biochem. Pharmacol. 2010, 79, 955–966. [Google Scholar] [CrossRef] [PubMed]
- Barrie, A.M.; Gushue, A.C.; Eskander, R.N. Dramatic response to Laetrile and cannabidiol (CBD) oil in a patient with metastatic low grade serous ovarian carcinoma. Gynecol. Oncol. Rep. 2019, 29, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Lukhele, S.T.; Motadi, L.R. Cannabidiol rather than Cannabis sativa extracts inhibit cell growth and induce apoptosis in cervical cancer cells. BMC Complement. Altern. Med. 2016, 16, 335. [Google Scholar] [CrossRef]
- American Cancer Society. Key Statistics for Vaginal Cancer. Available online: https://www.cancer.org/cancer/vaginal-cancer/about/key-statistics.html (accessed on 26 June 2020).
- Rosado, J. Vaginal Cancer. Available online: https://www.marijuanadoctors.com/conditions/vaginal-cancer/ (accessed on 26 June 2020).
- Rogers, L.J.; Cuello, M.A. Cancer of the vulva. Int. J. Gynaecol. Obstet. 2018, 143 (Suppl. 2) (Suppl. 2), 4–13. [Google Scholar] [CrossRef]
- Sangiovanni, E.; Fumagalli, M.; Pacchetti, B.; Piazza, S.; Magnavacca, A.; Khalilpour, S.; Melzi, G.; Martinelli, G.; Dell’Agli, M. Cannabis sativa L. extract and cannabidiol inhibit in vitro mediators of skin inflammation and wound injury. Phytother. Res. 2019, 33, 2083–2093. [Google Scholar] [CrossRef] [PubMed]
- Stabile, C.; Gunn, A.; Sonoda, Y.; Carter, J. Emotional and sexual concerns in women undergoing pelvic surgery and associated treatment for gynecologic cancer. Transl. Androl. Urol. 2015, 4, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, C.; Cantarero, I.; Palomares, B.; Gomez-Canas, M.; Fernandez-Ruiz, J.; Pavicic, C.; Garcia-Martin, A.; Luz Bellido, M.; Ortega-Castro, R.; Perez-Sanchez, C.; et al. VCE-004.3, a cannabidiol aminoquinone derivative, prevents bleomycin-induced skin fibrosis and inflammation through PPARgamma- and CB2 receptor-dependent pathways. Br. J. Pharmacol. 2018, 175, 3813–3831. [Google Scholar] [CrossRef]
- Stinchcomb, A.L.; Valiveti, S.; Hammell, D.C.; Ramsey, D.R. Human skin permeation of Delta8-tetrahydrocannabinol, cannabidiol and cannabinol. J. Pharm. Pharmacol. 2004, 56, 291–297. [Google Scholar] [CrossRef]
- Fonseca, B.M.; Correia-da-Silva, G.; Taylor, A.H.; Lam, P.M.; Marczylo, T.H.; Konje, J.C.; Teixeira, N.A. Characterisation of the endocannabinoid system in rat haemochorial placenta. Reprod. Toxicol. 2012, 34, 347–356. [Google Scholar] [CrossRef]
- Battista, N.; Bari, M.; Rapino, C.; Trasatti, F.; D’Agostino, A.; Maccarrone, M. Regulation of female fertility by the endocannabinoid system. Hum. Fertil. (Camb.) 2007, 10, 207–216. [Google Scholar] [CrossRef]
- Battista, N.; Pasquariello, N.; Di Tommaso, M.; Maccarrone, M. Interplay between endocannabinoids, steroids and cytokines in the control of human reproduction. J. Neuroendocrinol. 2008, 20 (Suppl. 1), 82–89. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M. Endocannabinoids and reproductive endocrinology. Curr. Opin. Investig. Drugs 2009, 10, 305–310. [Google Scholar] [PubMed]
- Lockwood, C.J. Prediction of pregnancy loss. Lancet 2000, 355, 1292–1293. [Google Scholar] [CrossRef]
- Costa, M.A.; Fonseca, B.M.; Marques, F.; Teixeira, N.A.; Correia-da-Silva, G. The psychoactive compound of Cannabis sativa, Delta(9)-tetrahydrocannabinol (THC) inhibits the human trophoblast cell turnover. Toxicology 2015, 334, 94–103. [Google Scholar] [CrossRef]
- Frank, D.A.; Bauchner, H.; Parker, S.; Huber, A.M.; Kyei-Aboagye, K.; Cabral, H.; Zuckerman, B. Neonatal body proportionality and body composition after in utero exposure to cocaine and marijuana. J. Pediatrics 1990, 117, 622–626. [Google Scholar] [CrossRef]
- Sidney, S. Cardiovascular consequences of marijuana use. J. Clin. Pharmacol. 2002, 42, 64S–70S. [Google Scholar] [CrossRef]
- Taylor, A.H.; Abbas, M.S.; Bell, S.C.; Konje, J.C. The inhibitory effect of delta9-tetrahydrocannabinol on trophoblast cell proliferation and transcription is mediated via the CB-2 receptor. Br. J. Obstet. Gynaecol. 2006, 114, 1040. [Google Scholar]
- Taylor, A.H.; Abbas, M.S.; Bell, S.C.; Konje, J.C. The inhibitory effect of delta9-tetrahydrocannabinol on trophoblast cell proliferation and transcription is mainly mediated via the CB-2 receptor. Reprod. Sci. 2007, 14, 720. [Google Scholar]
- Maia, J.; Midao, L.; Cunha, S.C.; Almada, M.; Fonseca, B.M.; Braga, J.; Goncalves, D.; Teixeira, N.; Correia-da-Silva, G. Effects of cannabis tetrahydrocannabinol on endocannabinoid homeostasis in human placenta. Arch. Toxicol. 2019, 93, 649–658. [Google Scholar] [CrossRef]
- Sarrafpour, S.; Urits, I.; Powell, J.; Nguyen, D.; Callan, J.; Orhurhu, V.; Simopoulos, T.; Viswanath, O.; Kaye, A.D.; Kaye, R.J.; et al. Considerations and implications of cannabidiol use during pregnancy. Curr. Pain Headache Rep. 2020, 24, 38. [Google Scholar] [CrossRef]
- Taylor, B.N.; Mueller, M.; Sauls, R.S. Cannaboinoid antiemetic therapy. In StatPearls; Statpearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Almada, M.; Amaral, C.; Oliveira, A.; Fernandes, P.A.; Ramos, M.J.; Fonseca, B.M.; Correia-da-Silva, G.; Teixeira, N. Cannabidiol (CBD) but not tetrahydrocannabinol (THC) dysregulate in vitro decidualization of human endometrial stromal cells by disruption of estrogen signaling. Reprod. Toxicol. 2020, 93, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Feinshtein, V.; Erez, O.; Ben-Zvi, Z.; Erez, N.; Eshkoli, T.; Sheizaf, B.; Sheiner, E.; Huleihel, M.; Holcberg, G. Cannabidiol changes P-gp and BCRP expression in trophoblast cell lines. PeerJ 2013, 1, e153. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Baillie, G.L.; Phillips, A.M.; Razdan, R.K.; Ross, R.A.; Pertwee, R.G. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br. J. Pharmacol. 2007, 150, 613–623. [Google Scholar] [CrossRef]
- Correa, F.; Wolfson, M.L.; Valchi, P.; Aisemberg, J.; Franchi, A.M. Endocannabinoid system and pregnancy. Reproduction 2016, 152, R191–R200. [Google Scholar] [CrossRef] [PubMed]
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for medical use: A systematic review and meta-analysis. JAMA 2015, 313, 2456–2473. [Google Scholar] [CrossRef]
- Blake, E.A.; Ross, M.; Ihenacho, U.; Figueroa, L.; Silverstein, E.; Flink, D.; Mendez-Ishizaki, Y.; Yessaian, A.; Brunette, L.L.; Matsuo, K.; et al. Non-prescription cannabis use for symptom management amongst women with gynecologic malignancies. Gynecol. Oncol. Rep. 2019, 30, 100497. [Google Scholar] [CrossRef]
- Totorikaguena, L.; Olabarrieta, E.; Lolicato, F.; Romero-Aguirregomezcorta, J.; Smitz, J.; Agirregoitia, N.; Agirregoitia, E. The endocannabinoid system modulates the ovarian physiology and its activation can improve in vitro oocyte maturation. J. Cell Physiol. 2020. [Google Scholar] [CrossRef]
- Maccarrone, M.; Falciglia, K.; Di Rienzo, M.; Finazzi-Agro, A. Endocannabinoids, hormone-cytokine networks and human fertility. Prostaglandins Leukot. Essent. Fatty Acids 2002, 66, 309–317. [Google Scholar] [CrossRef]
- Guzman, M. Cannabis for the management of cancer symptoms: THC version 2.0? Cannabis Cannabinoid Res. 2018, 3, 117–119. [Google Scholar] [CrossRef]
- Maccarrone, M. Phytocannabinoids and endocannabinoids: Different in nature. Rend. Lincei 2020, 31, 931–938. [Google Scholar] [CrossRef]
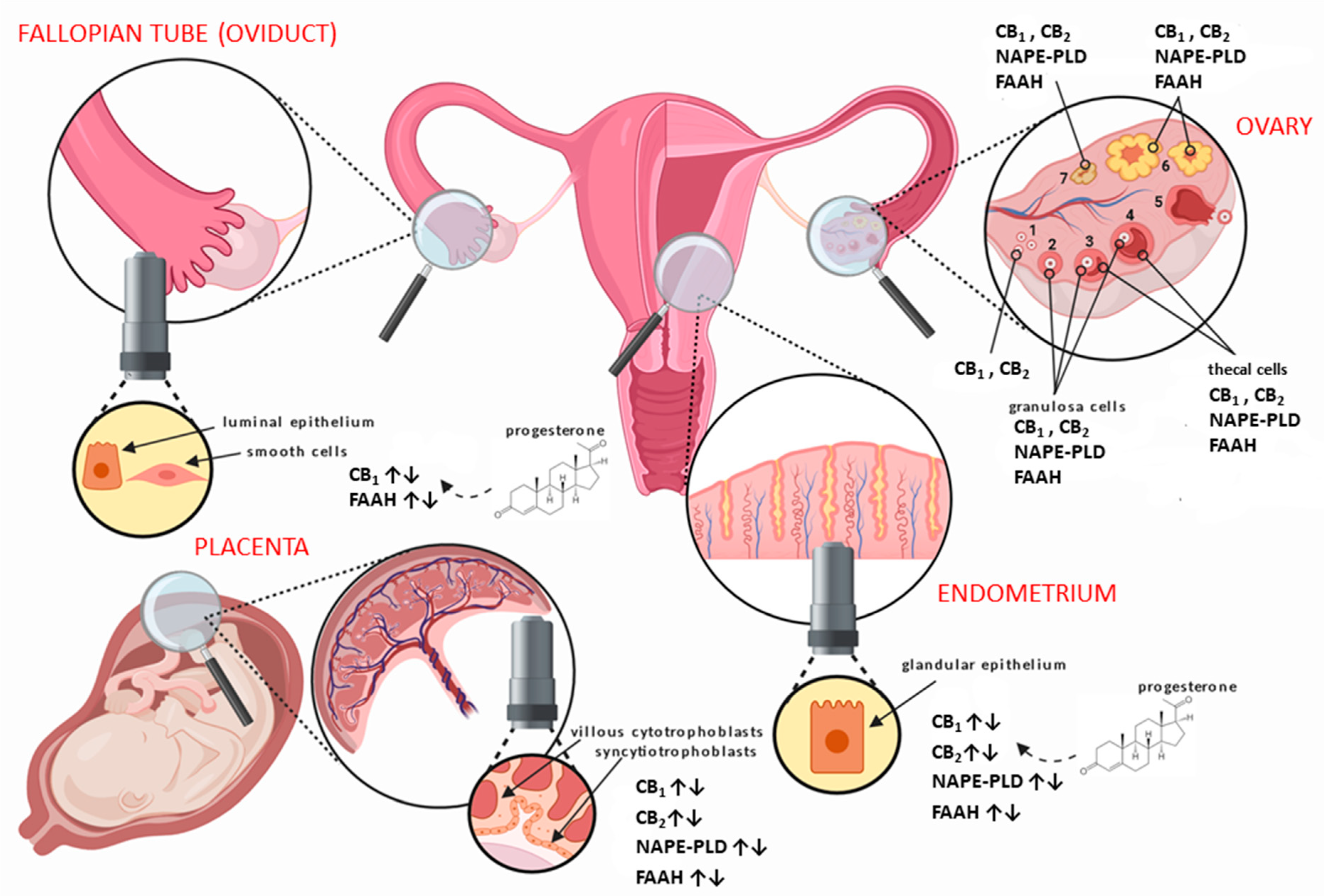
| Tissue | ECS Component | Normal Tissue | Change in Cancer | Pertinent References |
|---|---|---|---|---|
| Ovary | AEA | yes | ↑ | [45,59] |
| OEA | yes | ↑ | [59,60] | |
| PEA | yes | ↑ | [59] | |
| 2-AG | yes | ↑ | [61] | |
| CB1 | yes | ? | [45] | |
| CB2 | yes | ? | [45,62] | |
| TRPV1 | yes | ↑ | [63] | |
| GPR55 | no | ↑ | [64] | |
| NAPE-PLD | yes | ? | [45] | |
| FAAH | yes | ? | [45] | |
| DAGL | ? | ? | none | |
| MAGL | ? | *↑ | [65] | |
| Fallopian Tube (Oviduct) | AEA | yes | ? | [59] |
| OEA | yes | ? | [59] | |
| PEA | yes | ? | [59] | |
| 2-AG | ? | ? | none | |
| CB1 | yes | ? | [46,49,66] | |
| CB2 | yes | ? | [49,66] | |
| TRPV1 | ? | ? | none | |
| GPR55 | ? | ? | none | |
| NAPE-PLD | yes | ? | [49] | |
| FAAH | yes | ? | [49,67] | |
| DAGL | ? | ? | none | |
| MAGL | ? | ? | none | |
| Uterus (endometrium) | AEA | yes | ↑, ± | [50,55,68,69] |
| OEA | yes | ↑ | [68] | |
| PEA | yes | ↑ | [68] | |
| 2-AG | yes | ↑ | [12] | |
| CB1 | yes | ↓, ±, ↑ | [47,70,71,72,73,74,75] | |
| CB2 | yes | ↑, ↓ | [47,72,74] | |
| TRPV1 | yes | ↓ | [76,77] | |
| GPR55 | yes | ↑ | [77] | |
| NAPE-PLD | yes | ↑ | [47,78] | |
| FAAH | yes | ↓ | [47,78,79,80] | |
| DAGL | yes | ? | [74,81] | |
| MAGL | yes | ? | [74,81] | |
| Uterus (myometrium) ** | AEA | Yes | ? | [30] |
| OEA | Yes | ? | [30] | |
| PEA | Yes | ? | [30] | |
| 2-AG | ? | ? | none | |
| CB1 | Yes | ? | [30,82,83] | |
| CB2 | Yes | ? | [30,83] | |
| TRPV1 | Yes | ? | [30] | |
| GPR55 | yes | ? | [30] | |
| NAPE-PLD | yes | ? | [30,82] | |
| FAAH | yes | ? | [30,82] | |
| DAGL | yes | ? | [81] | |
| MAGL | yes | ? | [81] | |
| Cervix | AEA | yes | ? | [84,85] |
| OEA | ? | ? | none | |
| PEA | ? | ? | none | |
| 2-AG | ? | ? | none | |
| CB1 | yes | ? | [80,84] | |
| CB2 | yes | ? | [80,84,86] | |
| TRPV1 | yes | ? | [84,85,87] | |
| GPR55 | ? | ? | none | |
| NAPE-PLD | ? | ? | none | |
| FAAH | yes | ? | [80] | |
| DAGL | ? | ? | none | |
| MAGL | ? | ? | none | |
| Vagina | AEA | ? | ? | none |
| OEA | ? | ? | none | |
| PEA | ? | ? | none | |
| 2-AG | ? | ? | none | |
| CB1 | yes | ? | [80] | |
| CB2 | ? | ? | none | |
| TRPV1 | ? | ? | none | |
| GPR55 | ? | ? | none | |
| NAPE-PLD | ? | ? | none | |
| FAAH | yes | ? | [80] | |
| DAGL | ? | ? | none | |
| MAGL | ? | ? | none | |
| Vulva | AEA | ? | ? | none |
| OEA | ? | ? | none | |
| PEA | ? | ? | none | |
| 2-AG | ? | ? | none | |
| CB1 | yes | ? | [80,88] | |
| CB2 | yes | ? | [88] | |
| TRPV1 | yes | ? | [88] | |
| GPR55 | yes | ? | [88] | |
| NAPE-PLD | ? | ? | none | |
| FAAH | yes | ? | [80] | |
| DAGL | ? | ? | none | |
| MAGL | ? | ? | none | |
| Placenta (Trophoblast) *** | AEA | yes | ? | [53,89] |
| OEA | yes | ? | [53] | |
| PEA | yes | ? | [53] | |
| 2-AG | yes | ? | [90] | |
| CB1 | yes | ? | [48,53,58,80,91] | |
| CB2 | yes | ? | [48,53,58] | |
| TRPV1 | yes | ? | [48] | |
| GPR55 | yes | ? | [92] | |
| NAPE-PLD | yes | ? | [48,53] | |
| FAAH | yes | ? | [48,53,58,80,91] | |
| DAGL | yes | ? | [93,94] | |
| MAGL | yes | ? | [93,94] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taylor, A.H.; Tortolani, D.; Ayakannu, T.; Konje, J.C.; Maccarrone, M. (Endo)Cannabinoids and Gynaecological Cancers. Cancers 2021, 13, 37. https://doi.org/10.3390/cancers13010037
Taylor AH, Tortolani D, Ayakannu T, Konje JC, Maccarrone M. (Endo)Cannabinoids and Gynaecological Cancers. Cancers. 2021; 13(1):37. https://doi.org/10.3390/cancers13010037
Chicago/Turabian StyleTaylor, Anthony H., Daniel Tortolani, Thangesweran Ayakannu, Justin C. Konje, and Mauro Maccarrone. 2021. "(Endo)Cannabinoids and Gynaecological Cancers" Cancers 13, no. 1: 37. https://doi.org/10.3390/cancers13010037
APA StyleTaylor, A. H., Tortolani, D., Ayakannu, T., Konje, J. C., & Maccarrone, M. (2021). (Endo)Cannabinoids and Gynaecological Cancers. Cancers, 13(1), 37. https://doi.org/10.3390/cancers13010037