1. Introduction
Around 10–20% of ovarian cancer (OC) [
1] and 6% breast cancer (BC) [
2] overall are caused by inheritable
BRCA1/BRCA2 mutations. Women carrying
BRCA1/BRCA2 mutations have a 17–44% risk of OC and 69–72% risk of BC until age 80 years [
3]. Most of these cancers can be prevented in unaffected
BRCA1/BRCA2 women carriers. Women can opt for risk-reducing salpingo-oophorectomy (RRSO), to reduce OC risk [
4]. In
BRCA women, RRSO reduces OC risk by 79–96% [
4,
5,
6]. Additionally, they can opt for MRI/mammography screening, chemoprevention with selective estrogen-receptor modulators (SERM) or aromatase inhibitors [
7]; or risk-reducing mastectomy (RRM) [
8,
9] to reduce their BC risk [
10]. RRM reduces BC risk by 90–95% [
8,
9]. Mutation identification also enables women to make timely, informed reproductive/lifestyle choices and consider prenatal/pre-implantation genetic diagnosis.
Despite 25 years of
BRCA testing and effective mechanisms for prevention, current guidelines and access to testing/treatment pathways remain complex and associated with a massive under-utilisation of genetic testing [
11]. Only 20% of eligible US women have accessed/undergone genetic testing [
11]. A UK analysis shows the huge majority (>97%) of
BRCA carriers in the population remain unidentified [
12]. This highlights substantial missed opportunities for early detection and primary prevention. The current approach uses established clinical-criteria/family-history (FH) based
a priori BRCA probability thresholds to identify high-risk individuals eligible for
BRCA testing. These clinical criteria/FH-based criteria are used to calculate mutation probability and have been loosened over the years. Earlier, the threshold for offering
BRCA testing used to be 20% probability. Most countries/health systems now offer
BRCA testing at a
BRCA mutation probability of around 10% [
13]. A number of different strategies ranging from standardised criteria to complex mathematical (Empirical/Mendelian) models have been used to calculate mutation probability and are used in clinical practice. However, this requires individuals and health practitioners to recognise and act on a significant FH.
BRCA carriers, who are unaware of their FH, unappreciative of its risk/significance, not proactive in seeking advice, or lack a strong FH (small families/paternal inheritance/chance) get excluded. Over 50%
BRCA carriers do not fulfil clinical criteria and are missed [
14,
15,
16,
17,
18,
19,
20]. Current detection rates are inadequate to identify all
BRCA carriers and even doubling detection rates will need 165 years to ascertain the ‘clinically detectable’ proportion of
BRCA carriers [
12]. Why should we wait for decades for people to develop cancer before identifying
BRCA carriers and unaffected at-risk family members to offer prevention?
These limitations can be overcome through unrestricted/unselected population based
BRCA testing. Falling
BRCA testing costs, advances in computing/bioinformatics, and next-generation sequencing has made this possible. Jewish population studies show this is feasible, acceptable, has high satisfaction (91–95%), significantly reduces anxiety, doesn’t harm psychological well-being or quality of life, and is extremely cost-effective [
15,
16,
21,
22]. Pilot general population studies are ongoing in the UK/Canada [
23]. However, the potential applicability and scope for primary prevention transcends continents and countries. Health systems, infrastructure, costs, environment, contexts, opportunities, and capacity along with health sector priorities vary considerably across different countries, [
24]. Economic evaluations of health interventions, health perspectives and cost-effectiveness thresholds differ amongst countries. Nevertheless, economic evaluation is important to weigh up costs and health effects of alternative health strategies, to help health policy decision making with respect to cost efficiency and resource allocation. For interventions to be sustainable, they need to be cost-effective and affordable. The World Bank separates countries into four income categories using Gross National Income (GNI) per capita (USA dollars): Low-income (LIC: ≤
$1025), Lower–Middle Income (LMIC:
$1026–
$4035), Upper–Middle Income (UMIC:
$4036–
$12,475), and High Income (HIC: ≥
$12,476). In settings of state funded universal health care coverage, the difference between government and societal perspectives is narrower than countries with a limited social security structure/net, where this gap can be significantly larger and consequences considerable. We for the first time evaluate the cost-effectiveness of population-based
BRCA-testing (compared to clinical-criteria/family-history testing) across multiple countries/health systems: India (LMIC), Brazil (UMIC), China (UMIC), the USA (HIC), the UK (HIC), and the Netherlands (HIC). We present analyses from both health system or payer (here forth called ‘payer’) and societal perspectives.
2. Results
The comparison of lifetime costs and quality-adjusted life year (QALYs) of population testing and clinical-criteria/FH testing for women in different countries along with the country-specific incremental cost-effectiveness ratios (ICERs) and willingness-to-pay (WTP) thresholds are given in
Table 1. Our results show that from a ‘societal perspective’ (using WHO guidelines), population-based
BRCA testing is actually ‘cost-saving’ and contributes to better health in HIC of the UK (ICER =
$−5,639/QALY; life expectancy gained = 3.0 days), USA (ICER =
$−4018/QALY; life expectancy gained = 2.2 days), and The Netherlands (ICER =
$−11,433/QALY; life expectancy gained = 2.8 days). It appears potentially cost-effective in UMICs of China (ICER =
$18,066/QALY; life expectancy gained = 1.8 days) and cost-effective in Brazil (ICER =
$13,579/QALY; life expectancy gained = 3.7 days), but it is not cost-effective in India (ICER =
$23,031/QALY; life expectancy gained = 2.5 days) (LMIC) for the base case.
From a ‘payer perspective’ (using WHO guidelines), population-based BRCA testing is ‘highly’ cost-effective compared with clinical criteria/FH-based testing in HIC, with UK-ICER = $21,191/QALY (life expectancy gained = 3.0 days), USA-ICER = $16,552/QALY (life expectancy gained = 2.2 days), and Netherlands-ICER = $25,215/QALY (life expectancy gained = 2.8 days). In UMIC population-based BRCA testing is cost-effective with ICER = $23,485/QALY in China (life expectancy gained = 1.8 days) and ICER = $20,995/QALY in Brazil (life expectancy gained = 3.7 days). Population-based BRCA testing is not cost-effective in LMIC with ICER = $32,217/QALY in India (life expectancy gained = 2.5 days).
If we consider local, country-specific guidelines for the UK, USA, and the Netherlands, then population-based
BRCA testing is cost-effective from the payer perspective (UK-ICER =
$24,066/QALY; USA-ICER =
$16,552/QALY; Netherlands-ICER =
$17655/QALY), and cost-saving from the societal perspective (UK-ICER = −
$3543/QALY; USA-ICER = −
$4018/QALY; Netherlands ICER = −
$3185/QALY). The corresponding values for life expectancy gained are 2.6 days (UK), 2.2 days (USA) and 4.2 days Netherlands.
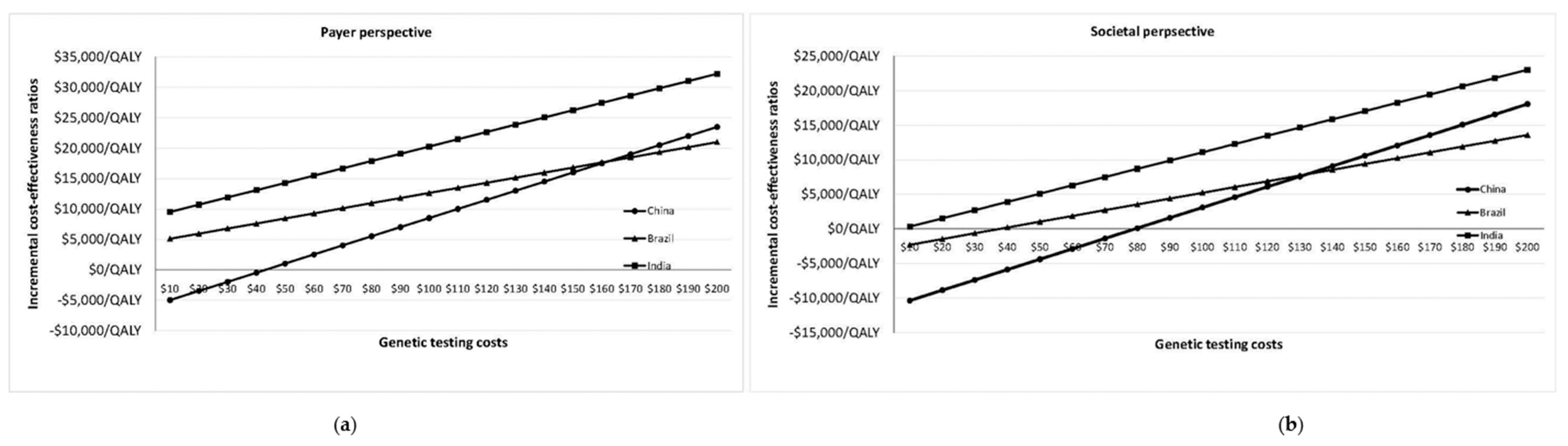
Figure 1a,b plot change in ICER/QALY with varying
BRCA testing costs in Brazil, China and India for payer and societal perspectives. Population testing becomes potentially cost-effective (from a societal perspective) in India if the
BRCA testing cost falls to
$172/test (ICER =
$19,685/QALY) (
Figure 1a;
Appendix D).
BRCA testing costs need to reach
$95/test (ICER =
$19,670/QALY) for cost-effective population testing in India from the payer perspective (
Figure 1b;
Appendix D).
The lifetime population impact (reduction in BC and OC cases and deaths; and excess coronary heart disease (CHD)) of offering population
BRCA testing for the six countries is detailed in
Table 2. A population-based
BRCA testing approach can potentially prevent an additional 2319 to 2666 BC and 327 to 449 OC cases per million women, translating to tens of thousands more BC/OC prevented across the population than the current clinical strategy.
Scenario analyses results are given in
Table 3. Different scenarios analysed include no reduction in BC risk from RRSO, nil compliance with hormone replacement therapy (HRT), reduction in RRM and RRSO rates by half, and reduced genetic testing costs of
$100. Population-based
BRCA testing remains cost-effective from payer and societal perspectives in each HIC and UMIC country at their respective WTP thresholds, even without reduction in BC risk from RRSO, no HRT uptake after RRSO, and 50% lower RRM and RRSO uptake rates (
Table 3). If the
BRCA testing costs fell to
$100/test, it would be highly cost-effective from the payer perspective and cost-saving (negative ICERs) from the societal perspective for HIC; highly cost-effective from payer/societal perspectives for UMIC, and cost-effective from the societal perspective for India (LMIC). The maximum
BRCA testing costs for population testing to remain cost-effective from the payer/societal perspectives respectively are in
Appendix E. At the 3*GDP WTP threshold, these are: UK =
$1254/
$1520; USA =
$1417/
$1577; Netherlands =
$1407/
$1758; China =
$354/
$390; Brazil =
$493/
$582; and India =
$95/
$172. Using UK/USA/Netherlands guideline-based WTP thresholds, these maximum
BRCA testing costs are UK =
$365, USA =
$850–
$1010, and Netherlands =
$800.
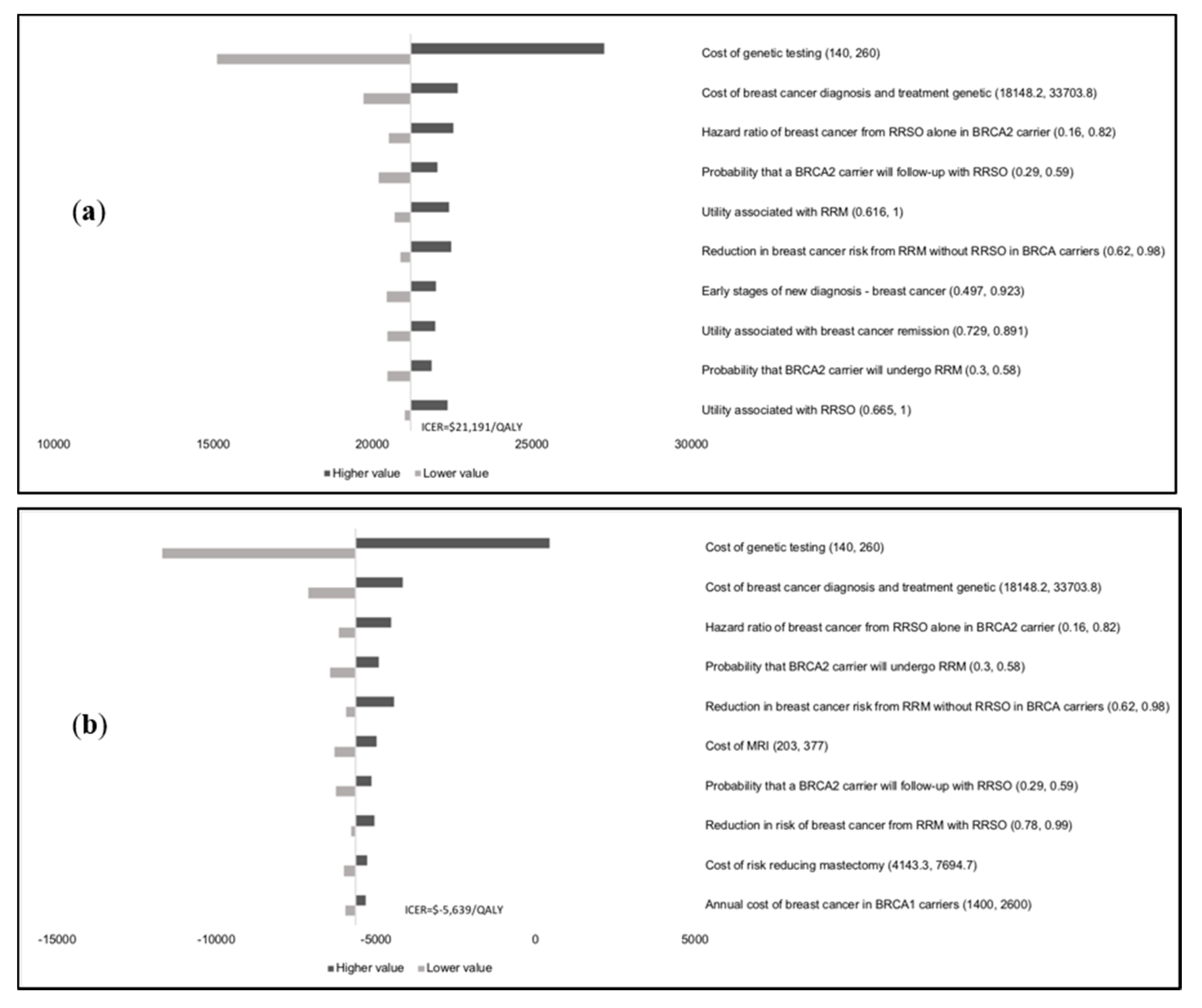
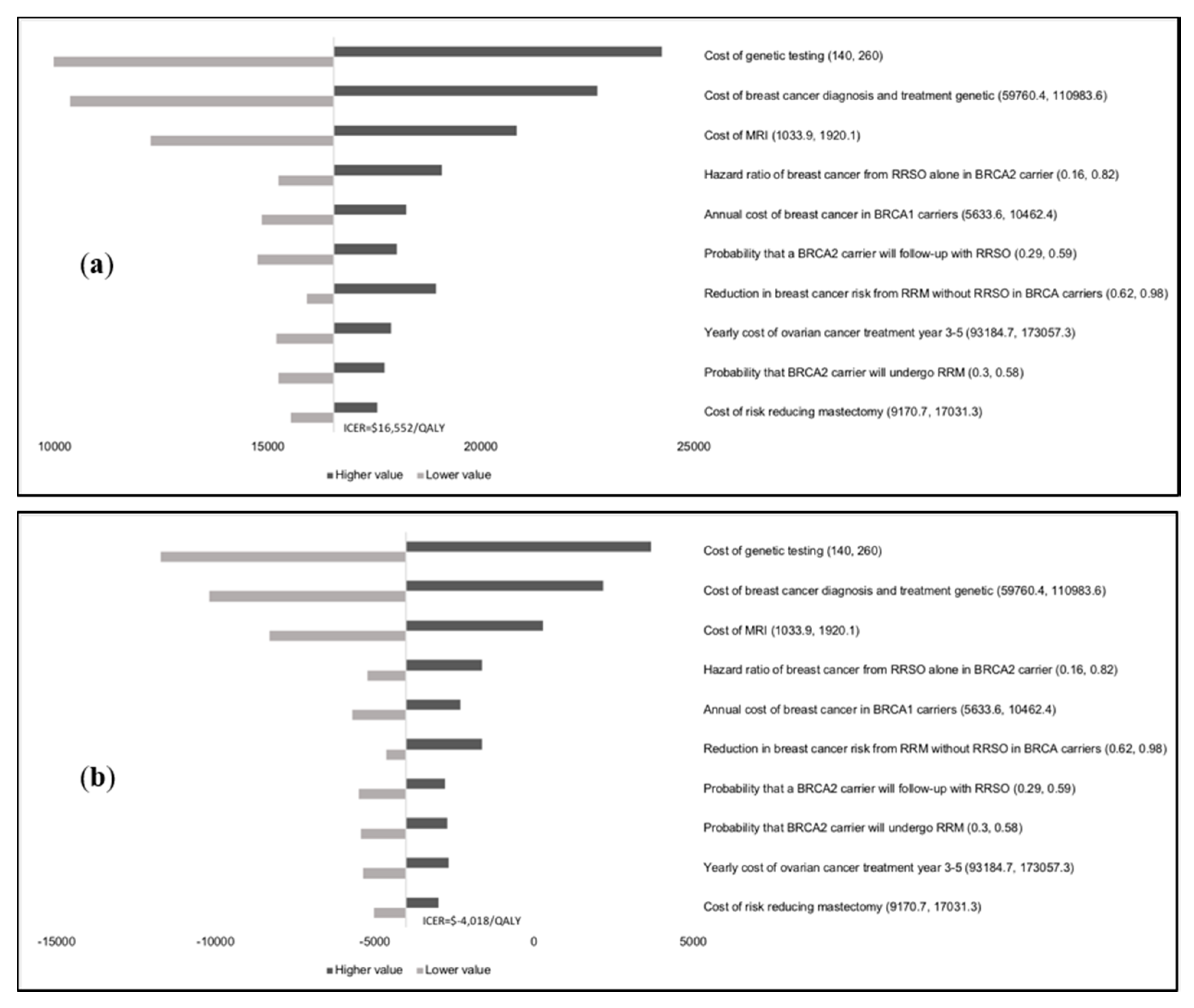
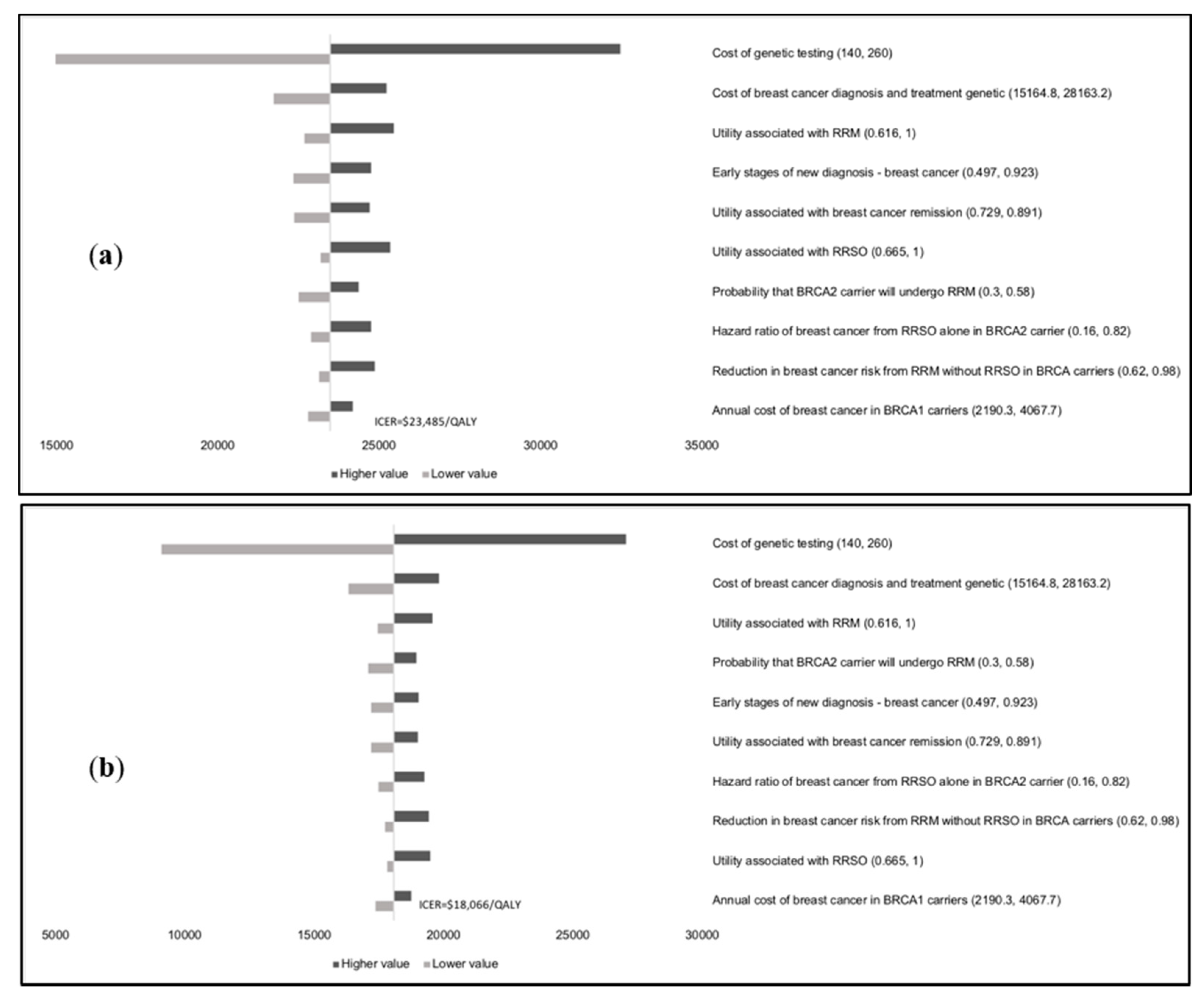
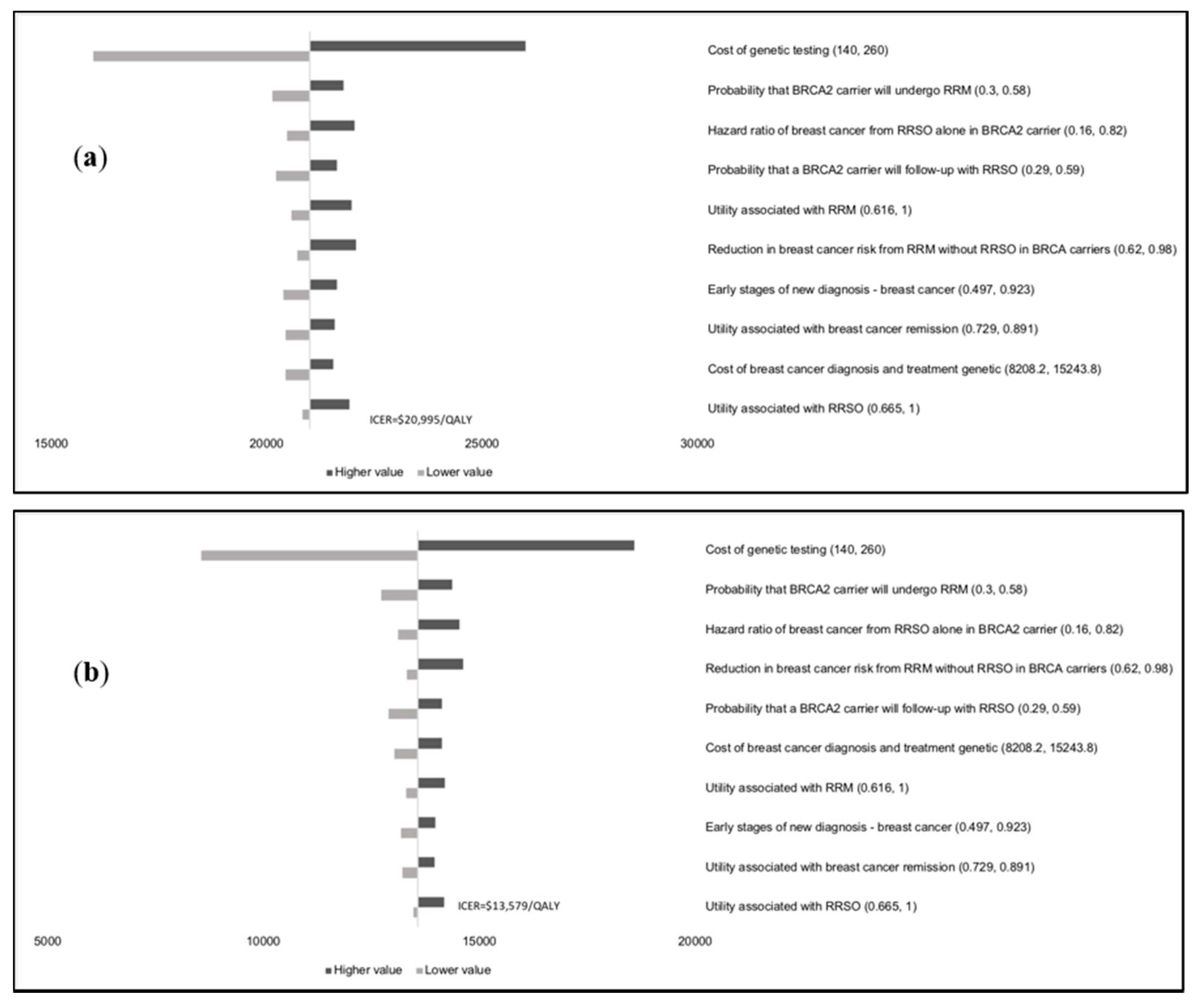
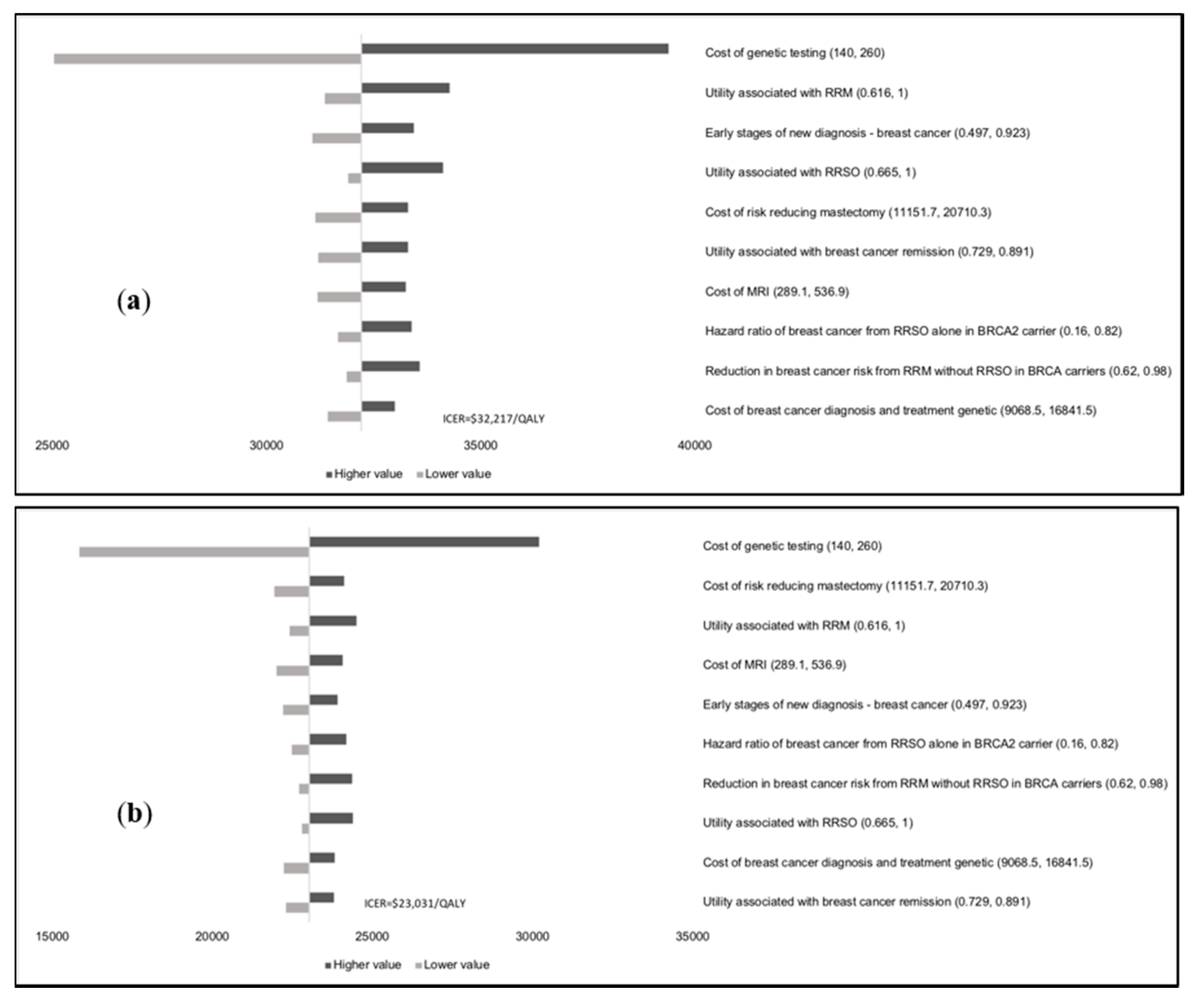
Results of the one-way sensitivity analysis indicate that model outcomes are not impacted much by treatment costs, utility scores, mutation prevalence, and probabilities (
Appendix E). The variable with the maximum effect on ICERs is the cost of
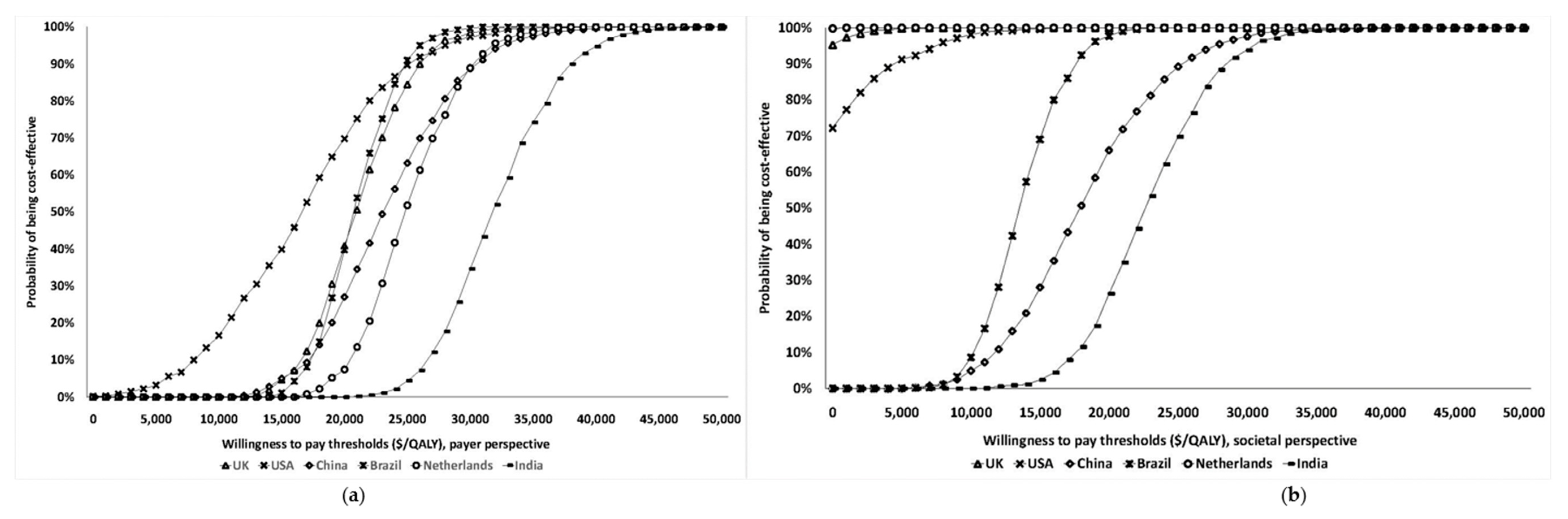
BRCA testing. Probabilistic sensitivity analysis (PSA) results (
Figure 2) show that at the WTP thresholds in each country, a population-testing strategy is cost-effective compared to clinical-criteria/FH-testing strategy from both the payer and societal perspectives for HIC and UMIC but not LMIC countries evaluated. The PSAs were highly cost-effective for the evaluated HIC and UMIC countries. All (100%) simulations are cost-effective at the guideline-specific thresholds for the UK/USA/Netherlands from payer and societal perspectives. For the 3*GDP-based WTP threshold for China/Brazil/India, 100%/100%/22.2% for the societal perspective and 100%/100%/0% simulations for the payer perspective were cost-effective (
Figure 2a,b). However, a population strategy becomes cost-effective in India (LMIC) at
$172/test. At the country-specific WTP thresholds for UK/USA/Netherlands, 84.9%/100%/98.5% of simulations for the payer perspective were cost-effective, and 100% simulations for the societal perspective were cost-effective for all three countries).
3. Discussion
For the first time, we explore the cost-effectiveness of population-based BRCA testing across countries from HIC, UMIC and LMIC health systems. We show that population-based BRCA testing is extremely cost-effective across HIC/UMIC health systems assessed and is potentially cost-saving for HIC health systems (UK/USA/Netherlands) if analysed from a societal perspective. Societal perspective analyses are associated with lower ICER/QALY than the payer perspective, as it incorporates additional costs linked to productivity loss. There is increasing recognition of the importance and need for economic cost-effectiveness evaluations to conform to the societal perspective and is recommended by WHO/international bodies. This is particularly important in middle/lower–income countries that lack a robust/comprehensive state-funded social security system. However, some countries such as the UK only consider a payer perspective when making health policy.
A population-based
BRCA testing approach can potentially prevent an additional 57,708/269,089/15,181/1,050,314/156,299/692,571 BC cases and 9727/43,817/2557/154,756/25,170/97,659 OC cases in the UK/USA/Netherlands/China/Brazil/India respectively (
Table 2) compared to the current clinical strategy. Given the huge under-utilisation of
BRCA testing along with limited access and uptake associated with current treatment pathways [
11,
12], one could postulate that the benefit could be even higher. Our findings are important, as we show that a new population-based approach can have much broader global applicability and a far greater impact on BC/OC burden in the population than current treatment strategies. Cost-effectiveness analyses are necessary to guide policy decisions on healthcare resource allocation. Our findings support a change in paradigm toward population testing to maximise OC/BC prevention and highlights a need for further implementation research in this area.
Our results are sensitive to the cost of testing, particularly in LMIC countries.
BRCA testing costs need to fall further for population testing to be cost-effective in LMIC countries. In India, it would become potentially cost-effective at
$172/test. Although our base case analysis uses costs higher than this, we are aware of Indian providers who offer
BRCA testing for around
$140/test. Genetic testing costs have fallen considerably over the last 5 years and remain on a downward trajectory. While we have used a standard cost for
BRCA testing that is currently available across countries, some providers may charge more than this. Our analysis of maximum cost(s) of
BRCA testing for a population testing strategy remaining cost-effective (
Appendix D) shows that these lie above what is charged by a number of providers today.
The precise definition of an appropriate cost-effectiveness threshold remains an important issue of ongoing debate. While this has been clearly defined in some (particularly HIC) health systems, a WHO-CHOICE 3*GDP threshold is considered too high by some, as it ignores opportunity costs [
30]. Additionally, whilst cost-effectiveness is a key factor for allocating health budgets, it needs to be considered along with context-specific local issues, affordability, budget impact, fairness, and feasibility [
31]. Some advocate against a single fixed threshold and recommend a range of thresholds for different contexts. The Norwegian health system prioritises interventions based on health benefit, resource implications, and health loss to the beneficiary if the intervention was absent (higher priority for higher health loss to the beneficiary) [
32]. We provide a range of cost estimates for
BRCA testing linked to varying potential cost-effectiveness thresholds (ICER/QALY) from payer and societal perspectives to help decision makers in UMIC and LIC. This is important, as the main model parameter impacting the overall result is the cost of
BRCA testing (
Figure 1a,b).
Our analysis has several advantages. We follow the transparency principle to facilitate the interpretation of methodology and results and use current standard of care or best practice as the comparator for measuring costs and effects. As per NICE recommendations, we use QALYs to measure health outcomes, which captures both length of life and quality of life and is generalisable across disease states. Our economic evaluation uses a lifetime horizon that is long enough to capture all costs and effects relevant to the decision problem. Additionally, costs and effects are discounted to reflect their value at the time of decision making, ensuring that the potential time preferences of the relevant population are accounted for. Our base case reflects direct health-care costs and health outcomes, and our analysis includes a societal perspective. We explore heterogeneity through scenario analyses and uncertainty and variability through extensive one-way/PSA analyses, as recommended. Our results remain robust at parameter extremes on one-way analysis (
Appendix E) and with PSA (
Figure 2). Our analysis uses PPP (purchasing power parity), which is a mechanism for accounting for different relative costs of goods when undertaking a comparative analysis of expenditures and incomes in different countries. Besides OC/BC outcomes, we also included excess CHD deaths from premenopausal oophorectomy [
33] and incorporate costs for HRT, excess heart disease, bone health monitoring, and treatment. Our costs also include pre-test counselling for all and post-test genetic counselling for pathogenic mutations and VUS.
Similar to other modelling studies, our study has some limitations. In line with earlier analyses in high-risk and low-risk women, our base case analysis assumes a reduction in BC risk with premenopausal oophorectomy. However, recently, there has been uncertainty around the benefit of BC risk reduction from RRSO. Nonetheless, our scenario analysis shows cost-effectiveness in HIC/UMIC even without BC risk reduction (
Table 3). We use established surgical prevention rates from HIC in the base-case analysis (
Table 4). However, RRM/RRSO rates vary, and lower rates are reported in some populations [
34]. The uptake of breast screening, chemoprevention, and risk-reducing surgery may also be influenced by socioeconomic, demographic, and cultural factors and may vary across populations [
34]. Rates of screening and preventive interventions have also increased with time. Higher rates are reported in the last 10 years compared to earlier decades, as knowledge and awareness of these issues has improved. Rates could be lower in carriers ascertained from population testing, particularly in the absence of cancer burden in the family. More prospective data on the uptake of surgical prevention following population-based testing will be needed. Our scenario analyses confirm cost-effectiveness for both payer and societal perspectives, even at half of standard surgical prevention rates (
Table 3). Although we incorporate a disutility for RRSO and RRM in the analysis, these procedures have potential complication rates of around 3–4% and 21%, respectively [
35,
36]. This needs to be part of the informed consent and decision-making process. While RRSO has been reported to have high satisfaction rates, less cancer worry, and no detriment in generic quality of life; poorer sexual function despite HRT use has been found [
37]. RRM has an adverse association with body image and sexual pleasure but not with sexual activity/habit/discomfort, anxiety/depression, or generic quality of life, and overall satisfaction rates are good. Countries such as India and China lack established national breast cancer screening programmes. The uptake of mammograms is much lower in these countries. The cost-effectiveness of population testing may be higher for these countries than estimated, as the implementation of these interventions in
BRCA carriers are likely to be more beneficial in the absence of routine mammograms in the population. In our analysis, while we included productivity loss, we did not include all indirect costs in the analysis. This may be a limitation. However, including additional indirect costs would improve cost-effectiveness, so our analysis is conservative in that respect. While our analysis covers some important/key countries across different income groups, it does not cover most countries, and therefore, these results are not generalisable globally to all countries across different (HIC/UMIC/LMIC) income groups. While the countries represented in this analysis are from four continents—North America, South America, Europe, and Asia—we do not have representation from Africa or Australia. The populations of countries in our analysis contribute approximately 45% to the global population.
Population-based
BRCA testing implementation studies have been completed in the Jewish population [
15,
21,
22,
38], and pilot ones are being undertaken in the UK and Canadian general populations [
23]. For population testing to be feasible, newer approaches for delivering pre-test information will be needed to facilitate informed decision-making. These will need to be country/region or context-specific. The best modality to deliver pre-test education within the population testing setting remains unresolved. We do not feel there will eventually be a one-size-fits-all model. Although we have costed for pre-test counselling for all in our analyses, whether formal pre-test counselling will be needed for all in the future remains uncertain. Israeli and Canadian Jewish population studies provided only ‘pre-test information’ and post-test genetic counselling for
BRCA carriers, with >90% satisfaction rates [
39,
40]. An Australian Jewish population [
41] and a UK general population study have demonstrated the feasibility of an online web-based decision aid (along with an optional telephone helpline) pre-test education and consent process [
42].
A strategy for the management of variants of unknown significance (VUS) is important and will need developing. People have raised concerns at unnecessary treatment or screening/preventive intervention(s) being undertaken for VUS alone. However, VUS are currently identified through routine clinical testing, too. There is clear acceptance in clinical practice that for a VUS (class-3 variant), no clinical action should be taken based on that variant alone [
43]. A key presumption inherent in a public health screening strategy is that it is not designed to identify ‘all’ individuals with disease, but the large/significant proportion of individuals in a clinically efficient and cost-effective manner. Therefore, some suggest an alternative option of not providing VUS results within a population-testing context [
14]. We incorporate a cost for VUS counselling and management in our analysis.
Chronic disease accounts for 90% US Medicare and 70% UK health care expenditure and is a major challenge facing most health systems, with cancer being its second commonest cause. Between 2006 and 2016, the average annual age-standardised incidence rates for all cancers increased in 130 of 195 countries [
44]. The leading cause for women is BC: 1.7 million cases, 535,000 deaths, 14.9 million disability adjusted life-years (DALYs) [
44]. Globally breast/ovarian cancers in women are predicted to increase by 46.5%/47% and cancer deaths are predicted to increase by 58.3%/58.6% respectively over the next 20 years [
45]. Population testing for
BRCA genes can significantly increase
BRCA carrier detection rates for maximising prevention and reducing cancer burden. It can also serve as an initial model, which subsequently informs the potential applicability of a population testing risk-stratification strategy for other cancer genes and other chronic diseases.
While developing an approach towards implementing population-based
BRCA-testing, it is important to bear in mind the principles of population testing of disease. These were initially proposed by Wilson and Jungner [
46]. Updated criteria have been suggested by the UK National Screening Committee [
47], Khoury [
48], the CDC (ACCE model) [
49], and Burke and Zimmerman (Public Health Foundation) [
50]. Analytic validity, clinical validity, clinical utility, and associated ethical, legal, and social implications remain key principles of the ACCE model, providing a framework for evaluating the applicability of a genetic test [
49]. In our study, we focussed on
BRCA testing, as testing for these genes has well-established clinical utility fulfilling the ACCE principles. Multigene panel testing is widely available in current clinical practice. We are against indiscriminate large-scale commercial panel testing without well-established clinical benefit/utility in the population-testing context. The low incidence of moderate penetrance genes, poor precision, and wide confidence intervals around prevalence and penetrance estimates require more data on the clinical significance of pathogenic variants in multigene panels, and these are reasons against currently implementing large multigene panel testing in the general population [
51,
52]. The USPSTF currently recommends against population testing in the general population [
51]. More data are needed on the ‘E’ (Ethical, legal, and social implications) of a population-based
BRCA testing approach across different populations and health systems. There is an urgent need for multiple implementation studies across countries for evaluating general population
BRCA testing and to develop local/regional and context-specific implementation pathways. These studies will need to provide prospective data on the impact of population testing on psychological well-being, quality of life, long-term health behaviour, socio-ethics, and lifestyle outcomes. A number of challenges and logistic hurdles will need to be overcome, including varying levels of workforce expansion/upskilling and the reorganisation of health services infrastructure. These include increasing public and health-professional awareness, establishing/expanding laboratory testing infrastructure, expanding downstream management pathways, and involving general practitioners, genetics services, gynaecologists, and breast clinicians/services. A framework/structure for data management and legal and regulatory protections will need to be established. These changes will need to be system/country and context-specific.
4. Materials and Methods
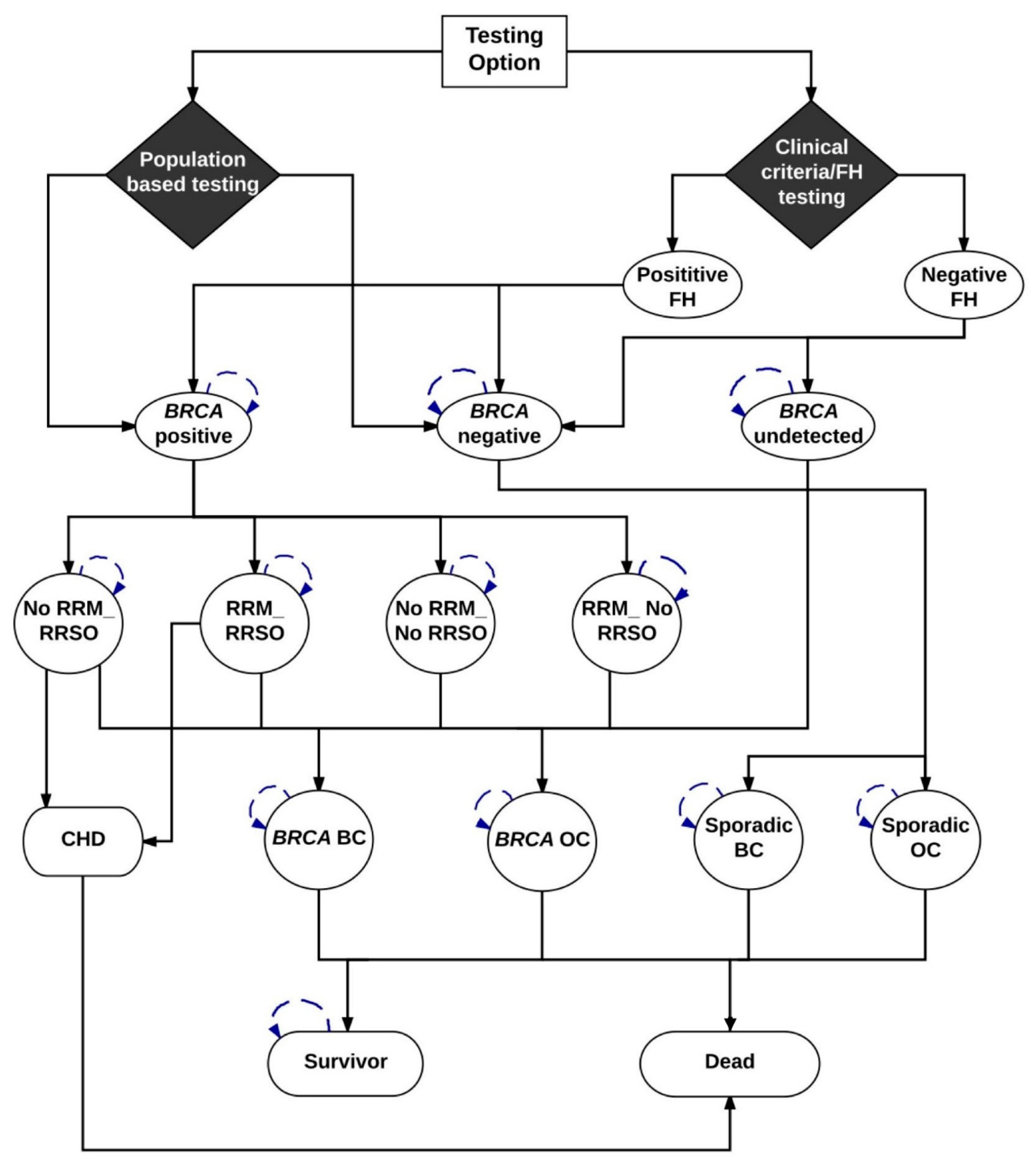
We developed a Markov model (
Figure 3) (TreeAge-Pro-2018 Williamson, MA, USA) to compare the lifetime costs and effects of
BRCA1/BRCA2 testing all general population women ≥30 years compared with clinical-criteria/FH-based testing. We describe separate analyses for populations in the UK, USA, Netherlands, China, Brazil, and India using both payer and societal perspectives. While some countries only consider a payer perspective, a societal perspective is recommended by the WHO and other international bodies [
53]. In the model, all women ≥30 years in the Population testing arm and only those fulfilling clinical/FH criteria in the Clinical-Criteria/FH-based testing arm undergo genetic testing for
BRCA mutations. We include pre-test counselling for all and assume a 70% uptake of genetic testing (from the published literature) [
22]. We include the cost of post-test counselling for mutation carriers as well as the cost of post-test counselling for those with variants of uncertain significance (VUS). We assume a VUS prevalence of 2% [
54]. Model probabilities are described in
Table 4,
Appendix A, and costs are outlined in
Appendix B.
BRCA carriers identified are offered RRSO to reduce OC risk [
4] and MRI/mammography screening, chemoprevention with SERM or RRM [
8] to reduce their BC risk [
10]. OC screening is excluded given the lack of mortality benefit. Women undergoing RRSO receive hormone replacement therapy (HRT) until 51 years. We include the costs of bone health monitoring and dual energy X-ray scans. We incorporate the excess risk and mortality from coronary heart disease (CHD) after premenopausal RRSO for women who do not take HRT (absolute mortality increase = 3.03%) [
33]. Associated costs are modelled over an individual’s lifetime. The Markov cycles’ run depends on life expectancy and these are different across countries (starting from age 30): UK = 53 cycles, US = 52 cycles, Netherlands = 53 cycles, China = 48 cycles, Brazil = 49 cycles, and India = 38 cycles. Cancer incidence is estimated by summing the probabilities of pathways ending in OC or BC.
4.1. Probabilities
The model probabilities for different pathways are given in
Table 4, and a detailed explanation is given in
Appendix A. The age-specific incidence of BC and OC among general population women is obtained from Cancer Research UK [
55,
56], USA Cancer Statistics [
57], and the International Agency for Research on Cancer (GLOBOCAN-2018) [
58]. The BC/OC incidence for
BRCA1/BRCA2 carriers is obtained from the literature [
3].
Figure 3 is a schematic diagram showing the Markov model structure for population and clinical-criteria/family-history (FH)-based
BRCA1/BRCA2 testing. In the Population testing arm, all women ≥30 years old are offered
BRCA1/BRCA2 testing and get classified as
BRCA-positive and
BRCA-negative.
BRCA mutation carriers identified are offered options of risk-reducing mastectomy (RRM) and risk-reducing salpingo-oophorectomy (RRSO). Depending on the probability of
BRCA women undertaking RRM and/or RRSO (+/− chemoprevention), they are placed into different health states and then progress to either
BRCA-associated breast cancer (BC) or
BRCA-associated ovarian cancer (OC). All women undergoing RRSO have an increased risk of fatal coronary heart disease (CHD). In addition, they have a probability of dying from the background all-cause mortality. Hence, patients in the model can go from intervention to death without ever developing breast cancer, ovarian cancer, or coronary artery disease. Patients can move from healthy state to death as they have a probability of dying from the background all-cause mortality.
BRCA-positive women who do not progress or die would stay in the health states and undertake the next cycle.
BRCA1/BRCA2-negative women progress to sporadic non-
BRCA OC or non-
BRCA BC based on the age-dependent probabilities. They also have a probability of dying from the background all-cause mortality. Women do not progress or die would stay in the health states to undertake the next cycle.
In the Clinical criteria/FH arm, only women whose FH fulfil current clinical criteria (based on current guidelines) undergo BRCA1/BRCA2 genetic testing and get classified as BRCA-positive and BRCA-negative. Women with a negative FH are either BRCA negative or have an undetected BRCA mutation. Options of RRM and RRSO and disease progression for identified BRCA mutation carriers and disease progression for BRCA negative women are the same as those in the population testing arm and are described above. All women undergoing RRSO have an increased risk of fatal coronary heart disease (CHD). Undetected BRCA women are not offered RRM or RRSO. Depending on the baseline risk (no risk-reducing options), they progress to BRCA-associated BC or BRCA-associated OC. In addition, they have a probability of dying from the background all-cause mortality. Hence, patients in the model can go from intervention to death without developing breast cancer, ovarian cancer, or coronary artery disease. Patients can move from healthy state to death as they may die from the background all-cause mortality. Women who do not progress or die stay in the health state of BRCA undetected and undertake the next cycle.
Progression through the model is dependent on the probabilities provided in
Table 4.
4.2. Costs
The analysis was conducted from both a payer perspective and societal perspective. All costs are reported at 2016 USA dollars, which was converted by purchasing power parity (PPP) factor [
28]. PPP reflects the value of a country’s currency required to purchase equivalent amounts of goods and services in the domestic market as the USA dollar would buy in the USA. Thus, it is used to translate and compare costs of goods/services between countries using the USA dollar as a common reference point. For comparison, we convert values in all other country currencies (£s, €s, ¥, ₹, R
$) to
$ (USA) using the purchasing power parity (PPP) factor [
28]. In line with the National Institute of Health and Care Excellence (NICE) recommendations, future healthcare costs not associated with BC/OC/heart disease were not considered [
25]. We collected primary data on relevant direct medical costs from the Urban Basic Medical Insurance Database in China [
65]; the Dutch Healthcare Authority (NZA) in Netherlands; Management System of Procedures/Medical drugs/Orthotics/Prosthetics/Special Materials (SIGTAP) [
66], the Health Price Bank (BPS) [
67], and Chamber of Regulation of the Market of Medicines (CMED) [
68] in Brazil; and an accredited cancer centre (Tata Medical Centre) in India (details in
Appendix B). Costing data were obtained from published national health service (NHS) reference costs for the UK [
69,
70] and published literature for the USA (details in
Appendix B). We adopted a standard internationally available
BRCA testing cost (US
$200) for our base case and explored the impact of change in testing costs on the overall results in the sensitivity analyses.
The retirement ages for females are 65 in the UK, 62 in the USA, 50–55 in China, 60 in Brazil, 68 in Netherlands, and 60–65 in India. We used the lower values of the retirement age ranges in China and India to get the conservative estimates of productivity loss. The female labour force participation rates are 56.77% in the UK, 55.99% in the USA, 62.03% in China, 53.32% in Brazil, 58.02% in the Netherlands, and 27.45% in India, which were obtained from the World Bank [
71]. For the hourly wage rates across countries, see
Appendix C. Additionally, we categorised costs due to productivity loss (for details: see
Appendix C) as three subcomponents: (1) temporary disability due to short-term work absences following diagnosis, (2) permanent disability from reduced working hours following return to work or workforce departure; and (3) premature mortality due to death before retirement [
72]. We estimated temporary disability as time absent from work multiplied by age-specific gross earnings. We calculated productivity costs due to permanent disability by applying age-specific gross earnings to the reduction in working hours, or the number of working hours in cases of permanent workforce departure, until retirement age. Regarding productivity loss from premature mortality, we assumed that without cancer, the productive capacity of an individual would continue from the age of diagnosis until the age of retirement. We multiplied the projected years of life lost by the age-specific gross earnings for the remainder of the working life to generate monetary estimates (see
Appendix C). While we included productivity loss, we did not include all indirect costs in the analysis.
4.3. Life Years
Lifetime tables from each country were used to model the lifetime health outcomes, and these were obtained from the World Health Organisation (WHO) [
73]. The median ages for RRM and RRSO in unaffected
BRCA carriers were assumed to be 37 and 40 years [
60]. BC and OC survival were modelled using five-year survival data from the CONCORD global surveillance of cancer survival [
74]. No significant overall long-term survival differences between germ-line and sporadic BC/OC have been found [
75,
76,
77]. After five years, the probability of death was assumed to be the same as that of the general population. Modelling estimated the number of BC cases, OC cases, BC deaths, OC deaths, and excess CHD deaths per million women aged 30 years in the six countries, and it calculated the number of cases prevented and deaths prevented. The actual numbers of cases prevented and deaths prevented were estimated based on the number of female population aged over 30 years in the six countries [
29].
4.4. Quality-Adjusted Life Years (QALY)
QALYs are recommended by NICE as the appropriate summary measure of health effects for economic evaluation. Utility scores multiplied by life years provides QALYs. QALY = (survival in life years) x (utility score). Utility score is an adjustment for quality of life. It is an indication of individual preferences for specific health states where 1 = perfect health and 0 = death. The utility scores for early, advanced, recurrent, and end-stage breast cancer are 0.71, 0.65, 0.45, and 0.16 [
78]. The utility scores used for early, advanced, recurrent, and end-stage OC are 0.81, 0.55, 0.61, and 0.16, respectively [
79]. Additionally, utility scores used for RRM is 0.88 (SD = 0.22) and RRSO is 0.95 (SD = 0.10) [
80].
4.5. Analysis
The Markov model is illustrated in
Figure 3. Model outcomes include OC, BC, and excess deaths from CHD. Future costs and health effects are discounted at WHO-recommended 3% rate for the WHO analyses [
81] and at country-recommended rates for country-specific analyses (see
Table 1). The lifetime costs and QALYs were estimated in both population-testing and clinical-criteria/FH-testing arms. The incremental cost-effectiveness ratio (ICER) was calculated by dividing the difference in cost by the difference in health effects between these two strategies. ICER = (Cost
Population-Testing–Cost
Criteria/FH-testing)/(Effect
Population-Testing–Effect
Criteria/FH-testing). The potential population impact was estimated by calculating the additional reduction in BC and OC incidence/deaths obtained through
BRCA testing women aged >30 years. We present analyses using a range of cost-effectiveness thresholds. For all countries, we present the initial WHO recommendation of three times gross domestic product (GDP) per capita (threshold of being cost-effective) and one-time GDP per capita (threshold for being highly cost-effective) [
82]. For countries (UK [
25], USA [
26], Netherlands [
27]) with specific health economic willingness-to-pay (WTP) threshold guidelines, we also present analysis using those guidelines: UK = £20,000–30,000 [
25]; USA =
$50,000–100,000 [
26]; Netherlands = €20,000–50,000. [
27] Additionally, given the lack of a clear established threshold, we evaluate changes in ICER/QALY with
BRCA testing costs for China, Brazil, and India to identify the
BRCA testing cost threshold for a given economic cost-effectiveness threshold. We use
$ (USA) conversion with PPP for comparison [
28].
We also explored a number of scenario analyses, including: (1) no BC risk reduction from RRSO (p9 = 1); (2) no HRT uptake (p13 = 0); (3) 50% reduction in RRM uptake; (4) 50% reduction in RRSO uptake; (5) lower
BRCA-testing costs of
$100; and (6) the maximum genetic testing costs at which population
BRCA testing remains cost-effective (see
Table 3,
Appendix D). In the one-way sensitivity analysis, each parameter is varied to evaluate their individual impact on results. Probabilities and utility scores were varied according to 95% confidence intervals or ranges where available or by +/−10%. Costs were varied by +/−30%. Probabilistic sensitivity analysis (PSA) was undertaken, and parameters varied simultaneously across their distributions. Costs were specified as having a Gamma distribution, quality of life was specified as having a log-normal distribution, and probability was specified as having a beta distribution, as recommended [
83]. A cost-effectiveness acceptability curve was used to plot the results of 1000 simulations for each country, showing the probability of population-based
BRCA testing being cost-effective at different WTP thresholds. Different curves were generated for payer and societal perspectives.