Circulating Tumor Cell Detection Technologies and Clinical Utility: Challenges and Opportunities
Abstract
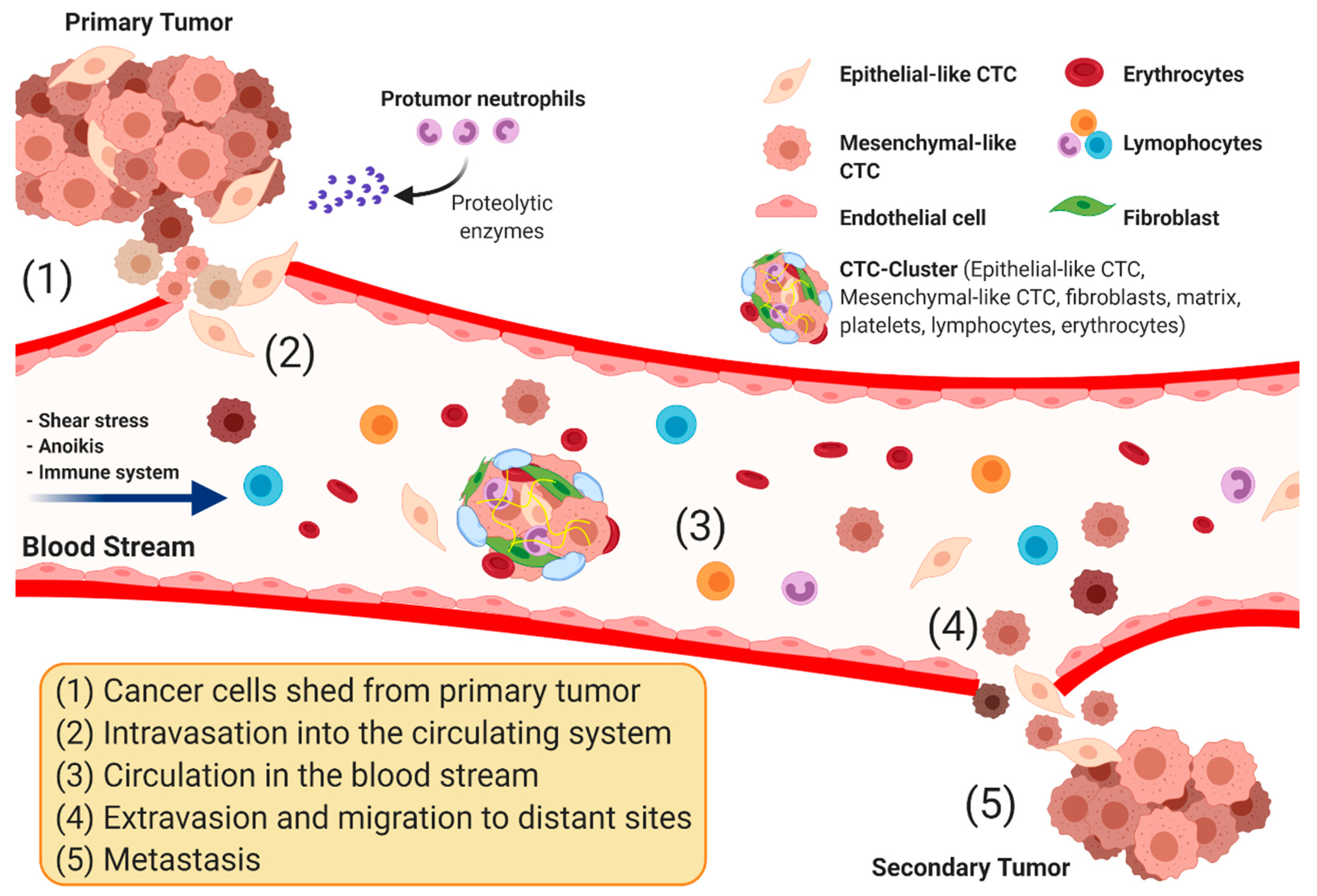
1. Introduction
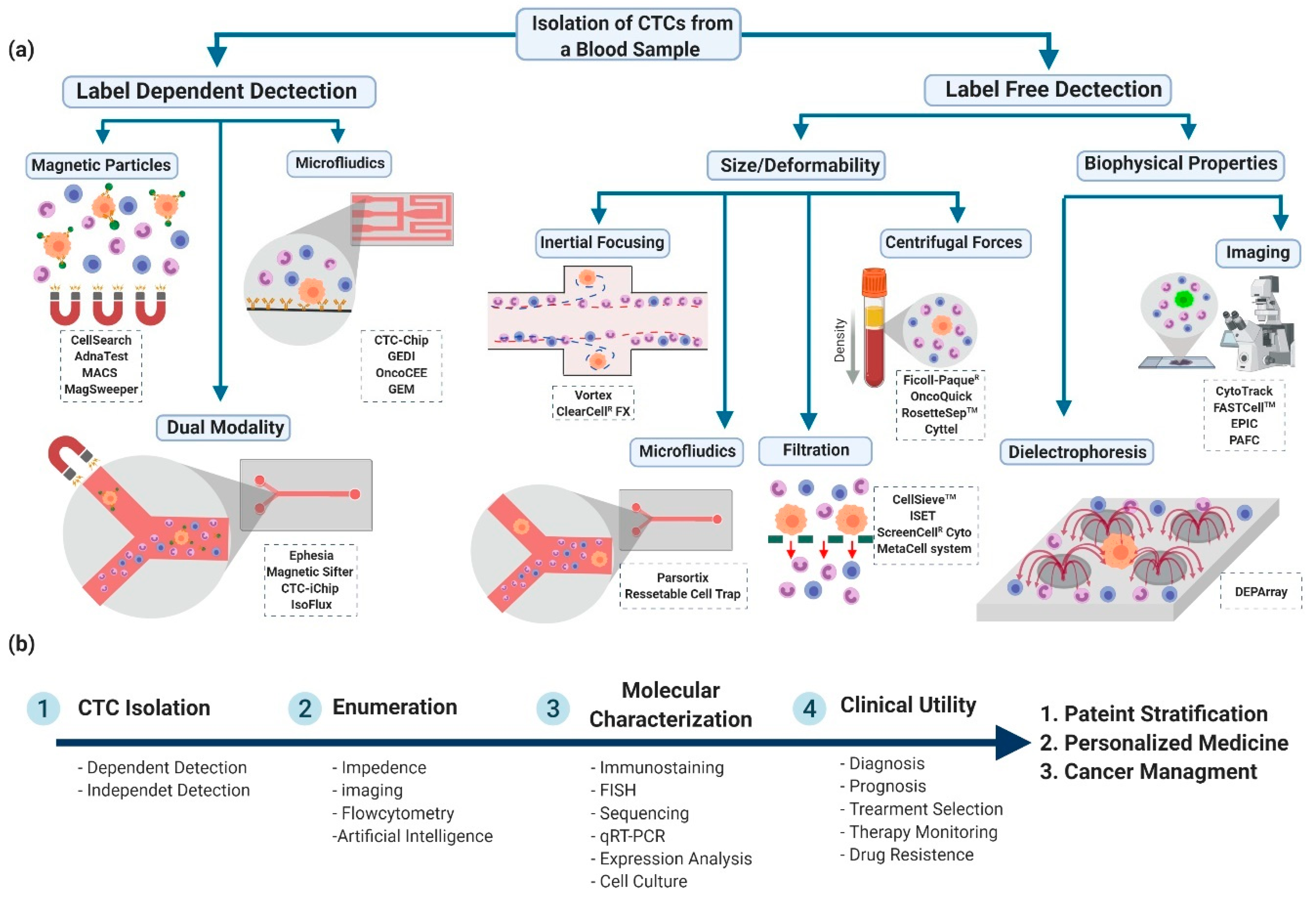
2. Commercialized CTC Detection Technologies
2.1. Label-Dependent Detection Technologies (Cell Surface Markers)
2.1.1. Immuno-Nano and -Magnetic Particles Platforms
2.1.2. Microfluidic Platforms
2.1.3. Dual Modality Platforms
2.2. Label-Free Detection Technologies (Cell Surface Markers)
2.2.1. Size-Based Separation
Filter-Based Detection
Microfluidic Chips
Centrifugal Forces
Inertial Focusing
2.2.2. Direct Imaging and Biophysical Properties-based
Direct Imaging
Dielectrophoresis
3. CTC Clinical Utility: Reporting Capabilities
3.1. Prognosis: Pretreatment Staging Assesmnet
3.2. Prognosis: Response to Therapy
3.3. Beyond Diagnosis and Prognosis: Molecular Characterization and Treatment Decisions
4. Discussion
5. Conclusions and Future Prospects
Funding
Conflicts of Interest
References
- American Cancer Society. Cancer Facts & Figures 2019; American Cancer Society: Atlanta, GA, USA, 2019. [Google Scholar]
- Pantel, K.; Brakenhoff, R.H. Dissecting the metastatic cascade. Nat. Rev. Cancer 2004, 4, 448. [Google Scholar] [CrossRef] [PubMed]
- Pantel, K.; Speicher, M.R. The biology of circulating tumor cells. Oncogene 2016, 35, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- Woo, D.; Yu, M. Circulating tumor cells as “liquid biopsies” to understand cancer metastasis. Transl. Res. 2018, 201, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Butler, T.P.; Gullino, P.M. Quantitation of cell shedding into efferent blood of mammary adenocarcinoma. Cancer Res. 1975, 35, 512–516. [Google Scholar]
- Krebs, M.G.; Hou, J.-M.; Ward, T.H.; Blackhall, F.H.; Dive, C. Circulating tumour cells: Their utility in cancer management and predicting outcomes. Ther. Adv. Med Oncol. 2010, 2, 351–365. [Google Scholar] [CrossRef]
- Bidard, F.-C.; Peeters, D.J.; Fehm, T.; Nolé, F.; Gisbert-Criado, R.; Mavroudis, D.; Grisanti, S.; Generali, D.; Garcia-Saenz, J.A.; Stebbing, J.; et al. Clinical validity of circulating tumour cells in patients with metastatic breast cancer: A pooled analysis of individual patient data. Lancet Oncol. 2014, 15, 406–414. [Google Scholar] [CrossRef]
- De Bono, J.S.; Scher, H.I.; Montgomery, R.B.; Parker, C.; Miller, M.C.; Tissing, H.; Doyle, G.V.; Terstappen, L.W.; Pienta, K.J.; Raghavan, D. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin. Cancer Res. 2008, 14, 6302–6309. [Google Scholar] [CrossRef]
- Krebs, M.G.; Sloane, R.; Priest, L.; Lancashire, L.; Hou, J.-M.; Greystoke, A.; Ward, T.H.; Ferraldeschi, R.; Hughes, A.; Clack, G.; et al. Evaluation and prognostic significance of circulating tumor cells in patients with non-small-cell lung cancer. J Clin. Oncol. 2011, 29, 1556–1563. [Google Scholar] [CrossRef]
- Cohen, S.J.; Punt, C.; Iannotti, N.; Saidman, B.H.; Sabbath, K.D.; Gabrail, N.Y.; Picus, J.; Morse, M.; Mitchell, E.; Miller, M.C.; et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. Clin. Oncol. 2008, 26, 3213–3221. [Google Scholar] [CrossRef]
- Gorges, T.M.; Tinhofer, I.; Drosch, M.; Röse, L.; Zollner, T.M.; Krahn, T.; von Ahsen, O. Circulating tumour cells escape from EpCAM-based detection due to epithelial-to-mesenchymal transition. BMC Cancer 2012, 12, 178. [Google Scholar] [CrossRef]
- Patriarca, C.; Macchi, R.M.; Marschner, A.K.; Mellstedt, H. Epithelial cell adhesion molecule expression (CD326) in cancer: A short review. Cancer Treat. Rev. 2012, 38, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Swennenhuis, J.F.; van Dalum, G.; Zeune, L.L.; Terstappen, L.W.M.M. Improving the CellSearch® system. Expert Rev. Mol. Diagn. 2016, 16, 1291–1305. [Google Scholar] [CrossRef]
- Murlidhar, V.; Rivera-Báez, L.; Nagrath, S. Affinity versus label-free isolation of circulating tumor cells: Who wins? Small 2016, 12, 4450–4463. [Google Scholar] [CrossRef]
- Shen, Z.; Wu, A.; Chen, X. Current detection technologies for circulating tumor cells. Chem. Soc. Rev. 2017, 46, 2038–2056. [Google Scholar] [CrossRef]
- Wang, L.; Balasubramanian, P.; Chen, A.P.; Kummar, S.; Evrard, Y.A.; Kinders, R.J. Promise and limits of the CellSearch platform for evaluating pharmacodynamics in circulating tumor cells. Semin Oncol. 2016, 43, 464–475. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.; Zu, Y. Detecting circulating tumor cells: Current challenges and new trends. Theranostics 2013, 3, 377. [Google Scholar] [CrossRef] [PubMed]
- Danila, D.C.; Samoila, A.; Patel, C.; Schreiber, N.; Herkal, A.; Anand, A.; Bastos, D.; Heller, G.; Fleisher, M.; Scher, H.I. Clinical validity of detecting circulating tumor cells by AdnaTest assay compared to direct detection of tumor mRNA in stabilized whole blood, as a biomarker predicting overall survival for metastatic castration-resistant prostate cancer patients. Cancer J. (Sudbury Mass.) 2016, 22, 315. [Google Scholar] [CrossRef]
- Müller, V.; Riethdorf, S.; Rack, B.; Janni, W.; Fasching, P.A.; Solomayer, E.; Aktas, B.; Kasimir-Bauer, S.; Pantel, K.; Fehm, T.; et al. Prognostic impact of circulating tumor cells assessed with the CellSearch System™ and AdnaTest Breast™ in metastatic breast cancer patients: The DETECT study. Breast Cancer Res. 2012, 14, R118. [Google Scholar] [CrossRef]
- Chinen, L.T.D.; De Carvalho, F.M.; Rocha, B.M.M.; Aguiar, C.M.; Abdallah, E.A.; Campanha, D.; Mingues, N.B.; de Oliveira, T.B.; Maciel, M.S.; Cervantes, G.M.; et al. Cytokeratin-based CTC counting unrelated to clinical follow up. J. Thorac. Dis. 2013, 5, 593. [Google Scholar] [PubMed]
- Giordano, A.; Gao, H.; Anfossi, S.; Cohen, E.; Mego, M.; Lee, B.-N.; Tin, S.; De Laurentiis, M.; Parker, C.A.; Alvarez, R.H.; et al. Epithelial–mesenchymal transition and stem cell markers in patients with HER2-positive metastatic breast cancer. Mol. Cancer Ther. 2012, 11, 2526–2534. [Google Scholar] [CrossRef] [PubMed]
- Ramsköld, D.; Luo, S.; Wang, Y.-C.; Li, R.; Deng, Q.; Faridani, O.R.; Daniels, G.A.; Khrebtukova, I.; Loring, J.F.; Laurent, L.C.; et al. Full-length mRNA-Seq from single-cell levels of RNA and individual circulating tumor cells. Nat. Biotechnol. 2012, 30, 777. [Google Scholar] [CrossRef] [PubMed]
- Scherag, F.D.; Niestroj-Pahl, R.; Krusekopf, S.; Lücke, K.; Brandstetter, T.; Rühe, J. Highly selective capture surfaces on medical wires for fishing tumor cells in whole blood. Anal. Chem. 2017, 89, 1846–1854. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Skelley, A.M.; Merdek, K.D.; Sprott, K.M.; Jiang, C.; Pierceall, W.E.; Lin, J.; Stocum, M.; Carney, W.P.; Smirnov, D.A. Microfluidics and circulating tumor cells. J. Mol. Diagn. 2013, 15, 149–157. [Google Scholar] [CrossRef]
- Kamande, J.W.; Hupert, M.L.; Witek, M.A.; Wang, H.; Torphy, R.; Dharmasiri, U.; Njoroge, S.K.; Jackson, J.M.; Aufforth, R.; Snavely, A.; et al. Modular microsystem for the isolation, enumeration, and phenotyping of circulating tumor cells in patients with pancreatic cancer. Anal. Chem. 2013, 85, 9092–9100. [Google Scholar] [CrossRef]
- Gleghorn, J.P.; Pratt, E.D.; Denning, D.; Liu, H.; Bander, N.H.; Tagawa, S.T.; Nanus, D.M.; Giannakakou, P.A.; Kirby, B.J. Capture of circulating tumor cells from whole blood of prostate cancer patients using geometrically enhanced differential immunocapture (GEDI) and a prostate-specific antibody. Lab Chip 2010, 10, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wong, K.H.; Khankhel, A.H.; Zeinali, M.; Reategui, E.; Phillips, M.J.; Luo, X.; Aceto, N.; Fachin, F.; Hoang, A.N.; et al. Microfluidic isolation of platelet-covered circulating tumor cells. Lab Chip 2017, 17, 3498–3503. [Google Scholar] [CrossRef] [PubMed]
- Stott, S.L.; Hsu, C.-H.; Tsukrov, D.I.; Yu, M.; Miyamoto, D.T.; Waltman, B.A.; Rothenberg, S.M.; Shah, A.M.; Smas, M.E.; Korir, G.K.; et al. Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc. Natl. Acad. Sci. USA 2010, 107, 18392–18397. [Google Scholar] [CrossRef] [PubMed]
- Sheng, W.; Ogunwobi, O.O.; Chen, T.; Zhang, J.; George, T.J.; Liu, C.; Fan, Z.H. Capture, release and culture of circulating tumor cells from pancreatic cancer patients using an enhanced mixing chip. Lab Chip 2014, 14, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J.A.; Pham, T.; Wong, K.L.; Scoggin, J.; Sales, E.V.; Clarin, T.; Pircher, T.J.; Mikolajczyk, S.D.; Cotter, P.D.; Bischoff, F.Z. FISH-based determination of HER2 status in circulating tumor cells isolated with the microfluidic CEE™ platform. Cancer Genet. 2011, 204, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Winer-Jones, J.P.; Vahidi, B.; Arquilevich, N.; Fang, C.; Ferguson, S.; Harkins, D.; Hill, C.; Klem, E.; Pagano, P.C.; Peasley, C.; et al. Circulating tumor cells: Clinically relevant molecular access based on a novel CTC flow cell. PLoS ONE 2014, 9, e86717. [Google Scholar] [CrossRef]
- Yoon, H.J.; Kim, T.H.; Zhang, Z.; Azizi, E.; Pham, T.M.; Paoletti, C.; Lin, J.; Ramnath, N.; Wicha, M.S.; Hayes, D.F.; et al. Sensitive capture of circulating tumour cells by functionalized graphene oxide nanosheets. Nat. Nanotechnol. 2013, 8, 735. [Google Scholar] [CrossRef] [PubMed]
- Svobodova, Z.; Kucerova, J.; Autebert, J.; Horak, D.; Bruckova, L.; Viovy, J.L.; Bilkova, Z. Application of an improved magnetic immunosorbent in an Ephesia chip designed for circulating tumor cell capture. Electrophoresis 2014, 35, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Autebert, J.; Coudert, B.; Champ, J.; Saias, L.; Guneri, E.T.; Lebofsky, R.; Bidard, F.-C.; Pierga, J.-Y.; Farace, F.; Descroix, S.; et al. High purity microfluidic sorting and analysis of circulating tumor cells: Towards routine mutation detection. Lab Chip 2015, 15, 2090–2101. [Google Scholar] [CrossRef] [PubMed]
- Harb, W.; Fan, A.; Tran, T.; Danila, D.C.; Keys, D.; Schwartz, M.; Ionescu-Zanetti, C. Mutational analysis of circulating tumor cells using a novel microfluidic collection device and qPCR assay. Transl. Oncol. 2013, 6, 528. [Google Scholar] [CrossRef]
- Wu, Y.; Deighan, C.J.; Miller, B.L.; Balasubramanian, P.; Lustberg, M.B.; Zborowski, M.; Chalmers, J.J. Isolation and analysis of rare cells in the blood of cancer patients using a negative depletion methodology. Methods 2013, 64, 169–182. [Google Scholar] [CrossRef]
- Ozkumur, E.; Shah, A.M.; Ciciliano, J.C.; Emmink, B.L.; Miyamoto, D.T.; Brachtel, E.; Yu, M.; Chen, P.-I.; Morgan, B.; Trautwein, J.; et al. Inertial focusing for tumor antigen–dependent and –independent sorting of rare circulating tumor cells. Sci. Transl. Med. 2013, 5, 179ra147. [Google Scholar] [CrossRef]
- Karabacak, N.M.; Spuhler, P.S.; Fachin, F.; Lim, E.J.; Pai, V.; Ozkumur, E.; Martel, J.M.; Kojic, N.; Smith, K.; Chen, P.-I.; et al. Microfluidic, marker-free isolation of circulating tumor cells from blood samples. Nat. Protoc. 2014, 9, 694. [Google Scholar] [CrossRef]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef]
- Riethdorf, S.; Fritsche, H.; Müller, V.; Rau, T.; Schindlbeck, C.; Rack, B.; Janni, W.; Coith, C.; Beck, K.; Jänicke, F.; et al. Detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer: A validation study of the CellSearch system. Clin. Cancer Res. 2007, 13, 920–928. [Google Scholar] [CrossRef]
- Bailey, P.C.; Martin, S.S. Insights on CTC biology and clinical impact emerging from advances in capture technology. Cells 2019, 8, 553. [Google Scholar] [CrossRef]
- Gabriel, M.T.; Calleja, L.R.; Chalopin, A.; Ory, B.; Heymann, D. Circulating tumor cells: A review of non–EpCAM-based approaches for cell enrichment and isolation. Clin. Chem. 2016, 62, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Hauch, S.; Zimmermann, S.; Lankiewicz, S.; Zieglschmid, V.; Boecher, O.; Albert, W.H. The clinical significance of circulating tumour cells in breast cancer and colorectal cancer patients. Anticancer Res. 2007, 27, 1337–1341. [Google Scholar] [PubMed]
- Zou, D.; Cui, D. Advances in isolation and detection of circulating tumor cells based on microfluidics. Cancer Biol. Med. 2018, 15, 335. [Google Scholar] [PubMed]
- Yu, M.; Bardia, A.; Wittner, B.S.; Stott, S.L.; Smas, M.E.; Ting, D.T.; Isakoff, S.J.; Ciciliano, J.C.; Wells, M.N.; Shah, A.M.; et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 2013, 339, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Bobek, V.; Gurlich, R.; Eliasova, P.; Kolostova, K. Circulating tumor cells in pancreatic cancer patients: Enrichment and cultivation. World J. Gastroenterol. WJG 2014, 20, 17163. [Google Scholar] [CrossRef]
- Krebs, M.G.; Hou, J.-M.; Sloane, R.; Lancashire, L.; Priest, L.; Nonaka, D.; Ward, T.H.; Backen, A.; Clack, G.; Hughes, A.; et al. Analysis of circulating tumor cells in patients with non-small cell lung cancer using epithelial marker-dependent and-independent approaches. J. Thorac. Oncol. 2012, 7, 306–315. [Google Scholar] [CrossRef]
- Morris, K.L.; Tugwood, J.D.; Khoja, L.; Lancashire, M.; Sloane, R.; Burt, D.; Shenjere, P.; Zhou, C.; Hodgson, C.; Ohtomo, T.; et al. Circulating biomarkers in hepatocellular carcinoma. Cancer Chemother. Pharmacol. 2014, 74, 323–332. [Google Scholar] [CrossRef]
- Xu, T.; Lu, B.; Tai, Y.-C.; Goldkorn, A. A cancer detection platform which measures telomerase activity from live circulating tumor cells captured on a microfilter. Cancer Res. 2010, 70, 6420–6426. [Google Scholar] [CrossRef]
- Schuur, E.R. Rapid and simple isolation of circulating tumor cells for clinical and research applications using ScreenCell®. ScreenCell® Devices: A Flexible ctc Platform. 2012. Available online: https://pdfs.semanticscholar.org/c8ce/414bed0455d52ecacb3977708dd7260495d7.pdf (accessed on 18 April 2020).
- Adams, D.L.; Stefansson, S.; Haudenschild, C.; Martin, S.S.; Charpentier, M.; Chumsri, S.; Cristofanilli, M.; Tang, C.M.; Alpaugh, R.K. Cytometric characterization of circulating tumor cells captured by microfiltration and their correlation to the cellsearch® CTC test. Cytom. Part A 2015, 87, 137–144. [Google Scholar] [CrossRef]
- Obermayr, E.; Maritschnegg, E.; Speiser, P.; Singer, C.; Schuster, E.; Holzer, B.; Danzinger, S.; Pecha, N.; Newland, A.; O’Brian, M.; et al. Circulating rare cells enable highly efficient cancer detection. In Proceedings of the 106th Annual Meeting of the American Association for Cancer Research, Philadelphia, PA, USA, 18–22 April 2015. [Google Scholar]
- Lapin, M.; Tjensvoll, K.; Oltedal, S.; Buhl, T.; Gilje, B.; Smaaland, R.; Nordgård, O. MINDEC-an enhanced negative depletion strategy for circulating tumour cell enrichment. Sci. Rep. 2016, 6, 28929. [Google Scholar] [CrossRef]
- Königsberg, R.; Obermayr, E.; Bises, G.; Pfeiler, G.; Gneist, M.; Wrba, F.; de Santis, M.; Zeillinger, R.; Hudec, M.; Dittrich, C. Detection of EpCAM positive and negative circulating tumor cells in metastatic breast cancer patients. Acta Oncol. 2011, 50, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, R.; Gertler, R.; Friederichs, J.; Fuehrer, K.; Dahm, M.; Phelps, R.; Thorban, S.; Nekarda, H.; Siewert, J. Comparison of two density gradient centrifugation systems for the enrichment of disseminated tumor cells in blood. Cytom. J. Int. Soc. Anal. Cytol. 2002, 49, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Tong, B.; Xu, Y.; Zhao, J.; Chen, M.; Xing, J.; Zhong, W.; Wang, M. Prognostic significance of circulating tumor cells in non-small cell lung cancer patients undergoing chemotherapy. Oncotarget 2017, 8, 86615. [Google Scholar] [CrossRef]
- Ramirez, A.B.; Crist, S.B.; Yeats, T.; Werbin, J.L.; Stilwell, J.L.; Kaldjian, E.P. Simultaneous visual assessment of RNA and protein expression in circulating tumor cells using the AccuCyte-Cytefinder system. In Proceedings of the AACR Annual Meeting 2017, Washington, DC, USA, 1–5 April 2017. [Google Scholar]
- Ramirez, J.-M.; Fehm, T.; Orsini, M.; Cayrefourcq, L.; Maudelonde, T.; Pantel, K.; Alix-Panabieres, C. Prognostic relevance of viable circulating tumor cells detected by EPISPOT in metastatic breast cancer patients. Clin. Chem. 2014, 60, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, T.W.; Ngo, V.T.; Dong, H.; Premasekharan, G.; Weinberg, V.; Doty, S.; Zhao, Q.; Gilbert, E.G.; Ryan, C.J.; Chen, W.T.; et al. Detection and characterization of invasive circulating tumor cells derived from men with metastatic castration-resistant prostate cancer. Int. J. Cancer 2014, 134, 2284–2293. [Google Scholar] [CrossRef]
- Hillig, T.; Horn, P.; Nygaard, A.-B.; Haugaard, A.S.; Nejlund, S.; Brandslund, I.; Sölétormos, G. In vitro detection of circulating tumor cells compared by the CytoTrack and CellSearch methods. Tumor Biol. 2015, 36, 4597–4601. [Google Scholar] [CrossRef] [PubMed]
- Somlo, G.; Lau, S.K.; Frankel, P.; Hsieh, H.B.; Liu, X.; Yang, L.; Krivacic, R.; Bruce, R.H. Multiple biomarker expression on circulating tumor cells in comparison to tumor tissues from primary and metastatic sites in patients with locally advanced/inflammatory, and stage IV breast cancer, using a novel detection technology. Breast Cancer Res. Treat. 2011, 128, 155–163. [Google Scholar] [CrossRef]
- Scher, H.I.; Lu, D.; Schreiber, N.A.; Louw, J.; Graf, R.P.; Vargas, H.A.; Johnson, A.; Jendrisak, A.; Bambury, R.; Danila, D.; et al. Association of AR-V7 on circulating tumor cells as a treatment-specific biomarker with outcomes and survival in castration-resistant prostate cancer. JAMA Oncol. 2016, 2, 1441–1449. [Google Scholar] [CrossRef]
- Ogle, L.F.; Orr, J.G.; Willoughby, C.E.; Hutton, C.; McPherson, S.; Plummer, R.; Boddy, A.V.; Curtin, N.J.; Jamieson, D.; Reeves, H.L. Imagestream detection and characterisation of circulating tumour cells—A liquid biopsy for hepatocellular carcinoma? J. Hepatol. 2016, 65, 305–313. [Google Scholar] [CrossRef]
- De Luca, F.; Rotunno, G.; Salvianti, F.; Galardi, F.; Pestrin, M.; Gabellini, S.; Simi, L.; Mancini, I.; Vannucchi, A.M.; Pazzagli, M.; et al. Mutational analysis of single circulating tumor cells by next generation sequencing in metastatic breast cancer. Oncotarget 2016, 7, 26107. [Google Scholar] [CrossRef]
- Gupta, V.; Jafferji, I.; Garza, M.; Melnikova, V.O.; Hasegawa, D.K.; Pethig, R.; Davis, D.W. ApoStream™, a new dielectrophoretic device for antibody independent isolation and recovery of viable cancer cells from blood. Biomicrofluidics 2012, 6, 024133. [Google Scholar] [CrossRef] [PubMed]
- Sollier, E.; Go, D.E.; Che, J.; Gossett, D.R.; O’Byrne, S.; Weaver, W.M.; Kummer, N.; Rettig, M.; Goldman, J.; Nickols, N. Size-selective collection of circulating tumor cells using Vortex technology. Lab Chip 2014, 14, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Guan, G.; Bhagat, A.A. ClearCell® FX, a label-free microfluidics technology for enrichment of viable circulating tumor cells. Cytom. Part A 2018, 93, 1251–1254. [Google Scholar] [CrossRef] [PubMed]
- Vona, G.; Sabile, A.; Louha, M.; Sitruk, V.; Romana, S.; Schütze, K.; Capron, F.; Franco, D.; Pazzagli, M.; Vekemans, M.; et al. Isolation by size of epithelial tumor cells: A new method for the immunomorphological and molecular characterization of circulating tumor cells. Am. J. Pathol. 2000, 156, 57–63. [Google Scholar] [CrossRef]
- Pinzani, P.; Salvadori, B.; Simi, L.; Bianchi, S.; Distante, V.; Cataliotti, L.; Pazzagli, M.; Orlando, C. Isolation by size of epithelial tumor cells in peripheral blood of patients with breast cancer: Correlation with real-time reverse transcriptase–polymerase chain reaction results and feasibility of molecular analysis by laser microdissection. Hum. Pathol. 2006, 37, 711–718. [Google Scholar] [CrossRef]
- De Giorgi, V.; Pinzani, P.; Salvianti, F.; Panelos, J.; Paglierani, M.; Janowska, A.; Grazzini, M.; Wechsler, J.; Orlando, C.; Santucci, M.; et al. Application of a filtration-and isolation-by-size technique for the detection of circulating tumor cells in cutaneous melanoma. J. Investig. Dermatol. 2010, 130, 2440–2447. [Google Scholar] [CrossRef]
- Coumans, F.A.; van Dalum, G.; Beck, M.; Terstappen, L.W. Filtration parameters influencing circulating tumor cell enrichment from whole blood. PLoS ONE 2013, 8, e61774. [Google Scholar] [CrossRef]
- Miller, M.C.; Robinson, P.S.; Wagner, C.; O’Shannessy, D.J. The Parsortix™ Cell Separation System—A versatile liquid biopsy platform. Cytom. Part A 2018, 93, 1234–1239. [Google Scholar] [CrossRef]
- Jacob, K.; Sollier, C.; Jabado, N. Circulating tumor cells: Detection, molecular profiling and future prospects. Expert Rev. Proteom. 2007, 4, 741–756. [Google Scholar] [CrossRef]
- Low, W.S.; Abas, W.; Bakar, W.A. Benchtop technologies for circulating tumor cells separation based on biophysical properties. BioMed Res. Int. 2015, 2015, 239362. [Google Scholar] [CrossRef]
- Zhang, Z.; Xiao, Y.; Zhao, J.; Chen, M.; Xu, Y.; Zhong, W.; Xing, J.; Wang, M. Relationship between circulating tumour cell count and prognosis following chemotherapy in patients with advanced non-small-cell lung cancer. Respirology 2016, 21, 519–525. [Google Scholar] [CrossRef]
- Bhagat, A.A.S.; Kuntaegowdanahalli, S.S.; Papautsky, I. Inertial microfluidics for continuous particle filtration and extraction. Microfluid. Nanofluid. 2009, 7, 217–226. [Google Scholar] [CrossRef]
- Segre, G.; Silberberg, A. Radial particle displacements in Poiseuille flow of suspensions. Nature 1961, 189, 209. [Google Scholar] [CrossRef]
- Harouaka, R.A.; Nisic, M.; Zheng, S.-Y. Circulating tumor cell enrichment based on physical properties. J. Lab. Autom. 2013, 18, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Riess, J.W.; Frankel, P.; Schwartz, E.; Bennis, R.; Hsieh, H.B.; Liu, X.; Ly, J.C.; Zhou, L.; Nieva, J.J.; et al. ERCC1 expression in circulating tumor cells (CTCs) using a novel detection platform correlates with progression-free survival (PFS) in patients with metastatic non-small-cell lung cancer (NSCLC) receiving platinum chemotherapy. Lung Cancer 2012, 77, 421–426. [Google Scholar] [CrossRef] [PubMed]
- López-Riquelme, N.; Minguela, A.; Villar-Permuy, F.; Ciprian, D.; Castillejo, A.; Álvarez-López, M.R.; Soto, J.L. Imaging cytometry for counting circulating tumor cells: Comparative analysis of the CellSearch vs. ImageStream systems. Apmis 2013, 121, 1139–1143. [Google Scholar] [CrossRef]
- Pethig, R. Dielectrophoresis: Status of the theory, technology, and applications. Biomicrofluidics 2010, 4, 022811. [Google Scholar] [CrossRef] [PubMed]
- Alshareef, M.; Metrakos, N.; Juarez Perez, E.; Azer, F.; Yang, F.; Yang, X.; Wang, G. Separation of tumor cells with dielectrophoresis-based microfluidic chip. Biomicrofluidics 2013, 7, 011803. [Google Scholar] [CrossRef]
- Ignatiadis, M.; Lee, M.; Jeffrey, S.S. Circulating tumor cells and circulating tumor DNA: Challenges and opportunities on the path to clinical utility. Clin. Cancer Res. 2015, 21, 4786–4800. [Google Scholar] [CrossRef]
- Pearl, M.L.; Zhao, Q.; Yang, J.; Dong, H.; Tulley, S.; Zhang, Q.; Golightly, M.; Zucker, S.; Chen, W.-T. Prognostic analysis of invasive circulating tumor cells (iCTCs) in epithelial ovarian cancer. Gynecol. Oncol. 2014, 134, 581–590. [Google Scholar] [CrossRef]
- Khan, M.S.; Kirkwood, A.A.; Tsigani, T.; Lowe, H.; Goldstein, R.; Hartley, J.A.; Caplin, M.E.; Meyer, T. Early changes in circulating tumor cells are associated with response and survival following treatment of metastatic neuroendocrine neoplasms. Clin. Cancer Res. 2016, 22, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Kirkwood, A.; Tsigani, T.; Garcia-Hernandez, J.; Hartley, J.A.; Caplin, M.E.; Meyer, T. Circulating tumor cells as prognostic markers in neuroendocrine tumors. J. Clin. Oncol. 2013, 31, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Janni, W.J.; Rack, B.; Terstappen, L.W.; Pierga, J.-Y.; Taran, F.-A.; Fehm, T.; Hall, C.; De Groot, M.R.; Bidard, F.-C.; Friedl, T.W.; et al. Pooled analysis of the prognostic relevance of circulating tumor cells in primary breast cancer. Clin. Cancer Res. 2016, 22, 2583–2593. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.-M.; Krebs, M.G.; Lancashire, L.; Sloane, R.; Backen, A.; Swain, R.K.; Priest, L.; Greystoke, A.; Zhou, C.; Morris, K.; et al. Clinical significance and molecular characteristics of circulating tumor cells and circulating tumor microemboli in patients with small-cell lung cancer. J. Clin. Oncol. 2012, 30, 525–532. [Google Scholar] [CrossRef]
- Wang, J.; Wang, K.; Xu, J.; Huang, J.; Zhang, T. Prognostic significance of circulating tumor cells in non-small-cell lung cancer patients: A meta-analysis. PLoS ONE 2013, 8, e78070. [Google Scholar] [CrossRef]
- Fiorelli, A.; Accardo, M.; Carelli, E.; Angioletti, D.; Santini, M.; Di Domenico, M. Circulating tumor cells in diagnosing lung cancer: Clinical and morphologic analysis. Ann. Thorac. Surg. 2015, 99, 1899–1905. [Google Scholar] [CrossRef]
- Franken, B.; De Groot, M.R.; Mastboom, W.J.; Vermes, I.; van der Palen, J.; Tibbe, A.G.; Terstappen, L.W. Circulating tumor cells, disease recurrence and survival in newly diagnosed breast cancer. Breast Cancer Res. 2012, 14, R133. [Google Scholar] [CrossRef]
- Bork, U.; Rahbari, N.; Schölch, S.; Reissfelder, C.; Kahlert, C.; Büchler, M.; Weitz, J.; Koch, M. Circulating tumour cells and outcome in non-metastatic colorectal cancer: A prospective study. Br. J. Cancer 2015, 112, 1306–1313. [Google Scholar] [CrossRef]
- Reeh, M.; Effenberger, K.E.; Koenig, A.M.; Riethdorf, S.; Eichstädt, D.; Vettorazzi, E.; Uzunoglu, F.G.; Vashist, Y.K.; Izbicki, J.R.; Pantel, K.; et al. Circulating tumor cells as a biomarker for preoperative prognostic staging in patients with esophageal cancer. Ann. Surg. 2015, 261, 1124–1130. [Google Scholar] [CrossRef]
- Garrel, R.; Mazel, M.; Perriard, F.; Vinches, M.; Cayrefourcq, L.; Guigay, J.; Digue, L.; Aubry, K.; Alfonsi, M.; Delord, J.-P.; et al. Circulating tumor cells as a prognostic factor in recurrent or metastatic head and neck squamous cell carcinoma: The CIRCUTEC prospective study. Clin. Chem. 2019, 65, 1267–1275. [Google Scholar] [CrossRef]
- Chou, W.-C.; Wu, M.-H.; Chang, P.-H.; Hsu, H.-C.; Chang, G.-J.; Huang, W.-K.; Wu, C.-E.; Hsieh, J.C.-H. A prognostic model based on circulating tumour cells is useful for identifying the poorest survival outcome in patients with metastatic colorectal cancer. Int. J. Biol. Sci. 2018, 14, 137. [Google Scholar] [CrossRef] [PubMed]
- Carter, L.; Rothwell, D.G.; Mesquita, B.; Smowton, C.; Leong, H.S.; Fernandez-Gutierrez, F.; Li, Y.; Burt, D.J.; Antonello, J.; Morrow, C.J.; et al. Molecular analysis of circulating tumor cells identifies distinct copy-number profiles in patients with chemosensitive and chemorefractory small-cell lung cancer. Nat. Med. 2017, 23, 114. [Google Scholar] [CrossRef] [PubMed]
- Freidin, M.B.; Freydina, D.V.; Leung, M.; Montero Fernandez, A.; Nicholson, A.G.; Lim, E. Circulating tumor DNA outperforms circulating tumor cells for KRAS mutation detection in thoracic malignancies. Clin. Chem. 2015, 61, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Punnoose, E.A.; Atwal, S.; Liu, W.; Raja, R.; Fine, B.M.; Hughes, B.G.; Hicks, R.J.; Hampton, G.M.; Amler, L.C.; Pirzkall, A.; et al. Evaluation of circulating tumor cells and circulating tumor DNA in non–small cell lung cancer: Association with clinical endpoints in a phase II clinical trial of pertuzumab and erlotinib. Clin. Cancer Res. 2012, 18, 2391–2401. [Google Scholar] [CrossRef]
- Hwang, S.W.; Lee, D.H. Is endoscopic ultrasonography still the modality of choice in preoperative staging of gastric cancer? World J. Gastroenterol. 2014, 20, 13775. [Google Scholar] [CrossRef]
- Judson, P.L.; Geller, M.A.; Bliss, R.L.; Boente, M.P.; Downs, L.S., Jr.; Argenta, P.A.; Carson, L.F. Preoperative detection of peripherally circulating cancer cells and its prognostic significance in ovarian cancer. Gynecol. Oncol. 2003, 91, 389–394. [Google Scholar] [CrossRef]
- De Bono, J.S.; Attard, G.; Adjei, A.; Pollak, M.N.; Fong, P.C.; Haluska, P.; Roberts, L.; Melvin, C.; Repollet, M.; Chianese, D.; et al. Potential applications for circulating tumor cells expressing the insulin-like growth factor-I receptor. Clin. Cancer Res. 2007, 13, 3611–3616. [Google Scholar] [CrossRef]
- Sarioglu, A.F.; Aceto, N.; Kojic, N.; Donaldson, M.C.; Zeinali, M.; Hamza, B.; Engstrom, A.; Zhu, H.; Sundaresan, T.K.; Miyamoto, D.T.; et al. A microfluidic device for label-free, physical capture of circulating tumor cell clusters. Nat. Methods 2015, 12, 685–691. [Google Scholar] [CrossRef]
- Smerage, J.B.; Barlow, W.E.; Hortobagyi, G.N.; Winer, E.P.; Leyland-Jones, B.; Srkalovic, G.; Tejwani, S.; Schott, A.F.; O’Rourke, M.A.; Lew, D.L.; et al. Circulating tumor cells and response to chemotherapy in metastatic breast cancer: SWOG S0500. J. Clin. Oncol. 2014, 32, 3483. [Google Scholar] [CrossRef]
- Keup, C.; Storbeck, M.; Hauch, S.; Hahn, P.; Sprenger-Haussels, M.; Tewes, M.; Mach, P.; Hoffmann, O.; Kimmig, R.; Kasimir-Bauer, S. Cell-free DNA variant sequencing using CTC-depleted blood for comprehensive liquid biopsy testing in metastatic breast cancer. Cancers 2019, 11, 238. [Google Scholar] [CrossRef]
- Marrugo-Ramírez, J.; Mir, M.; Samitier, J. Blood-based cancer biomarkers in liquid biopsy: A promising non-invasive alternative to tissue biopsy. Int. J. Mol. Sci. 2018, 19, 2877. [Google Scholar] [CrossRef] [PubMed]
- Abbosh, C.; Birkbak, N.J.; Wilson, G.A.; Jamal-Hanjani, M.; Constantin, T.; Salari, R.; Le Quesne, J.; Moore, D.A.; Veeriah, S.; Rosenthal, R.; et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 2017, 545, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, M.; Dawson, S.-J.; Tsui, D.W.; Gale, D.; Forshew, T.; Piskorz, A.M.; Parkinson, C.; Chin, S.-F.; Kingsbury, Z.; Wong, A.S.; et al. Non-invasive analysis of acquired resistance to cancer therapy by sequencing of plasma DNA. Nature 2013, 497, 108–112. [Google Scholar] [CrossRef] [PubMed]
| Device | Technology | Tumor Type | Clinical Value | Remarks | Ref. |
|---|---|---|---|---|---|
| Magnetic Nanoparticles | |||||
| CELLSEARCH® (Janssen Diagnostics) | EpCAM-coated ferrofluid nanoparticles for the selection of EpCAM+ cells. The captured cells are then confirmed by IF staining of CK 8, 18,19 and the lack of CD45 | -Metastatic breast cancer -Colorectal -Prostate | -Prognosis -Treatment | -FDA approved -use of antibodies (markers dependence) -Low purity of captured CTC -Sensitivity: 27%, 32%, 70% -Specificity: 89%, 99.7%, 93% -Most clinically validated capture technique | [16,17] |
| AdnaTest (Adnagen) | Magnetic beads coated with a cocktail of antibodies (EpCAM, MUC-1, etc.). Captured CTC are then analyzed by multiplex RT-PCR gene panels | -Breast -Prostate -Ovarian -Colon | -Prognosis -Treatment regimen | -Analyzes blood and bone marrow samples -Downstream RNA analysis post enrichment by RT-PCR -High sensitivity -High contamination with WBCs -Detection limit: >2 CTCs/7.5 mL a -Sensitivity: 73% a | [18,19] |
| MACS system (Miltenyi Biotec) | Immunomagnetic CTC enrichment by antibodies against cell surface markers or by an intracellular anti-pan CK antibody | -Non-small-cell lung cancer (NSCLC) -Breast (HER2+) | -Prognosis | Identifies EpCAM negative CTCs but not CK negative ones Can work with leukocytes depletion (negative enrichment by anti CD45) | [20,21] |
| MagSweeper (Illumina) | Immunomagnetic isolation of CTC by antibodies against EpCAM and cell surface markers from unfractionated blood samples | -Breast -Prostate -Colorectal | -Genetic profiling -Drug discovery | -High purity of captured CTC (almost 100%) -High throughput processing (9mL/hr) -Detects 1–3 CTCs/mL -Sensitivity: 100% b | [17,22] |
| In vivo | |||||
| GILUPI CellCollector™ | EpCAM-coated wire placed intravenously in patients for CTC collection | -Breast | -Detection | -In vivo based-detection of CTC -Processes large volumes of blood -Invasive-Time consuming | [23] |
| Microfluidics | |||||
| Modular Sinusoidal Microsystems (BioFluidica) | Combination of three modules for CTC selection, counting, and enumerating. The chip consists of 320 sinusoidal microchannels coated with antibodies for the capture of CTC followed by phenotypic identification. | -Pancreatic | -Diagnosis | -Electrical sensor for counting and determining the viability -Cell enumeration is based on impedance sensor -High purity (>86%) with an excellent yield of recovered -Processes 7.5 mL/h | [24,25] |
| GEDI | Geometrically enhanced differential immunocapture where antibodies against HER2 and PSMA are immobilized with high capture specificity from unprocessed blood | -Breast -Prostate | -Treatment regimen -Correlation between CTC and primary tumor | -High capture specificity -Detects up to 27 CTCs/mL -high purity (around 26%) -Sensitivity: 94% a | [26] |
| Herringbone (HB) Chip | High throughput microfluidic mixing device that allows efficient capture of CTC on antibody-coated surfaces | -Prostate | -Histological analysis | -Minimal studies have been assessed and all are preclinical -Processes 4.8 mL/h -Can detect up to 12 CTCs/mL -Purity of captured CTC is around 14% | [27,28] |
| GEM chip | Geometrically Enhanced Mixing chip structure that allows enhanced capture of CTC on antibody-coated surfaces | -Pancreatic | -Monitoring treatment response -Diagnosis | -Uses of antibodies or cocktail of antibodies -High selection efficiency -Processes 3.6 mL/h | [29] |
| OncoCEE (cell enrichment and extraction) (Biocept) | Microfluidic chip with internal surfaces functionalized with a cocktail of antibodies against biotumor-associated markers and mesenchymal markers. | -Breast | -Prognosis -Diagnosis -Treatment regiment | -High probability of CTC capture -Analysis of CK+ and CK− CTC is feasible -Sensitivity: 95% b -Specificity: 92% b | [30] |
| LiquidBiopsy® (Cynvenio) | Microfluidic chip with functionalized surfaces (coated with antibodies) for positive selection of CTC with direct automated DNA analysis | -Breast -Lung | -Genetic profiling of CTC | -Processes 5mL/h -20% error accuracy and 25% error precision -High purity of detected cells -Sheath flow decreases non-specific binding | [31] |
| Graphene oxide (GO) Chip | Nanosheets of graphene oxides functionalized with capture antibodies against cell surface markers of CTC with high sensitivity | -Breast -Pancreatic -Lung | -Prognosis | -High capture yield -Processes 1–3 mL/h -Minimal studies have been assessed and all are preclinical -Sensitivity: 73 ± 32.4 at 3–5 cells per mL of blood c | [32] |
| Dual modality | |||||
| Ephesia (CTC-chip) | Micromagnetic particles functionalized with EpCAM antibodies are self-assembled in a microfluidic platform (columns) | -Breast -NSCLC -Prostate -Colorectal | -Prognosis -Diagnosis | -High capture specificity -Processes more than 3 mL/h -Viability of captured cells maintained at 98% -Sensitivity: 99.1% c -Specificity: 100% c | [33,34] |
| IsoFlux (Fluxion) | A microfluidic platform of controlled flow and immunomagnetic capture bead system | -Breast -Prostate | -Diagnosis | -50% rate of capture -Capacity to detect genetic alterations | [35] |
| Quadrupole magnetic separator | Negative CTC enrichment by combining viscous flow stress and magnetic force for the recovery of unlabeled CTC | -Breast | -Prognosis -Diagnosis -Treatment selection | -Detects heterogeneity among CTC by IF -Multiparameter analysis is required -Minimal studies have been assessed and all are preclinical | [36] |
| CTC-iChip | Deterministic lateral displacement, inertial focusing, and magnetophoresis for rapid isolation of leukocytes using anti CD45 and anti CD66B antibodies (negative enrichment) or EpCAM activated beads for CTC enrichment (positive enrichment) | -EpCAM positive cancer -EpCAM negative cancer | -Diagnosis | -Developed at Janssen Diagnostics (in progress) -Positive and negative enrichment -Combines size-based separation of WBCs -Processes 8 mL/h -Low purity of captured CTC (around 8%) -Detection limit: <30 CTCs/7.5 mL d | [37,38] |
| Method | Advantages | Disadvantages |
|---|---|---|
| Magnetic Nanoparticles |
|
|
| Microfluidic chip |
|
|
| Dual Modality |
|
|
| Device | Technology | Tumor Type | Clinical Value | Remarks | Ref. |
|---|---|---|---|---|---|
| Filtration | |||||
| ISET® (Rarecells Diagnostics) | Filter based isolation and enrichment (PCL based filters) | -Lung (NSCLC) -Breast -Melanoma -Hepatocellular carcinoma | -Prognosis -Treatment regiment | -High efficiency capture compared to CellSearch -Label-free (no need to use antibodies) -Detection limit: 1 CTC/mL -Sensitivity: 76.37% a -Specificity: 82.39% a | [47,48] |
| MetaCell® system (MetaCell Ltd.) | size-based enrichment and separation | -Esophageal -Lung -Pancreatic | -Diagnosis -Prognosis | -Allows post-capture analysis and cell culture | [46] |
| Parylene filter (Circulogix) | Filter based isolation and enrichment | -Breast | -Diagnosis -Prognosis | -Post-capture downstream analysis for enumeration and immunophenotypic characterization -Fixation prior to capture eliminates post capture functional assays (cell culture and protein extraction and analysis) -Studies have been assessed in preclinical setups -Detection limit: 25 CTCs/7.5 mL -Capture efficiency: ~90% | [49] |
| ScreenCell® Cyto | Filter based size-exclusion separation and enrichment of CTC | -Melanoma | -Diagnostics -Treatment regimen (personalized medicine) | -Post-capture analysis and cell culture -Allows microscopic examinations of collected CTC -Minimal studies have been assessed and all are preclinical | [50] |
| CellSieve (Creatv MicroTech) | micofilter based isolation and enrichment | -Breast -Prostate | -Prognosis -Diagnosis | -High efficiency isolation compared to CellSearch technology -Post capture histo- and immune-phenotypic characterization of CTC | [51] |
| Microfluidics | |||||
| Parsortix™ technology (Angle plc) | Microfluidic separation of CTC based on their size and deformability. Viable cells are released by reversing the flow. | -Ovarian | -Diagnosis | -Antigen-independent capture with subsequent molecular analysis -Minimal studies have been assessed and all are preclinical -Sensitivity: 92% b (in primary and relapse ovarian cancer) -Specificity: 100% b | [52] |
| Density gradient separation | |||||
| RosetteSep™ CTC Enrichment Cocktail/EasyStep CD45 Depletion (STEMCELL Technologies) | Immunodensity negative selection for CTC using tetrameric antibody complexes that recognizes CD45, CD66b and glycophorin on WBC and RBC | -Pancreatic -Breast | -Prognosis | Unwanted cells are targeted for removal with Tetrameric Antibody Complexes that pellets with RBCs | [53] |
| OncoQuick(Greiner BioOne, Frickenhausen, Germany) | Separation of erythrocytes and some leukocytes from CTC by porous membrane filtration followed by density-grade centrifugation for CTC enrichment | -Gastrointestinal cancer -Advanced breast cancer | -prognosis | -Dual technology for separation of CTC based on size and buoyant density -High tumor cell rate recovery compared to other density-gradient techniques -Post-CTC capture processing is possible | [54,55] |
| Cyttel | Negative immune-magnetic selection of WBC (CD45 antibody) followed by gradient centrifugation and slide smearing of isolated CTC | -Lung (NSCLC) | -Prognosis -Treatment regimen | -High detection rate (bimodal identification of CTC: negative selection followed by in situ hybridization) | [56] |
| AccuCyte–CyteFinder (RareCyte) | Automated rapid imaging of single rare cells, CTC in this case, preceded by density-based cell separation | -adenocarcinoma | -prognosis | -Dual technology platform for single-cell analysis -high sensitivity detection of CD positive CTC -ability to analyze RNA post capture and enrichment -minimal studies have been assessed and all are preclinical | [57] |
| Functional Assays | |||||
| EPISPOT | Negative enrichment using CD45 depletion and short-term culture | -Breast | -Prognosis | -Allows CTC detection based on protein secretion -High sensitivity and specificity -Independent of tumor antigen phenotype capture -Allows quantification of CTC | [58] |
| Vita-Assay (Vitatex) | Functional cell separation using density gradient centrifugation followed by preferential adhesion of CTC to collagen adhesion matrix (CAM-enrichment). | -Prostate | -N.A. | -Allows CTC detection based on invasion properties -Low purity (0.5–35%) | [59] |
| Imaging | |||||
| CytoTrack | The blood sample is speed on a glass disc that is rotated at high speed. Fluorescently labelled cells against EpCAM are scanned with laser beam | -Breast | -Diagnosis | -Analyzes 100M cells/min -Low recovery rates of CTC | [60] |
| FASTcell (SRI) | Fiber optic array scanning technology (FAST) | -Breast | -Prognosis -Guided therapy (personalized treatments) | -High sensitivity of CTC detection based on biomarkers expression -Allows simultaneous detection of multiple tumor specific biomarkers -Analyzes 25M cells/min | [61] |
| Epic (Epic Sciences) | RBC lysis and IF for CK, CD45, and DAPI and other markers followed by high-definition imaging | -Prostate | -prognosis -treatment regimen | -Unbiased screen of all blood nucleated cells for detection of individual CTCs and clusters | [62] |
| ImageStream® (Amnis) | Immunogenetic sorting of blood followed by flow cytometery and fluorescent microscopy for CTC enumeration. | -Hepatocellular carcinoma | -Diagnosis | -Low precision when CTC count is low -Analyzes 5000 cells/s -Sensitivity: 68.75% c -Specificity: 72.97% c with likelihood ratio of 2.544 | [63] |
| Dielectrophoresis | |||||
| DEPArray™ (Silicon Biosystems) | Moving dielectrophoretic cages for cell capture coupled with sanger sequencing | -Breast | -Tumor and treatment monitoring -Prognosis | Isolation of single CTCs for dowstream gene analysis | [64] |
| ApoStream® (ApoCell) | Detection of CTC based on dielectrophoric Field-flow Fractionation (DEP-FFF) in a microfluidic chamber | Breast | -N.A. | -Detection independent of EpCAM expression; useful for viability analysis and culture -processes more than 10 mL/h -Studies have been assessed in preclinical setups | [65] |
| Inertial focusing | |||||
| Vortex | CTC extraction using microscale vortices and inertial focusing | -Breast -Lung | -Prognosis -Diagnosis -Treatment regimen | -Fast processing time of samples (20 min per 7.5 mL of blood) -High CTC integrity and purity post-detection (>50% up to 94%) | [66] |
| ClearCell® FX (Clearbridge BioMedics) | Separation of CTC based on size using Dean Flow Fractionation (DFF) (inertial focusing) | -Lung | -Molecular diagnostics | -Captured CTC can be analyzed post-capture and enrichment in culture -Low recovery rates -Label free (no need for antibody use) -Single step isolation and retrieval process from any type of body fluids -Processes 1–1.5 mL/min -Sensitivity: 80.4% c -Specificity: 85.7% c | [67] |
| Method | Advantages | Disadvantages |
|---|---|---|
| Microfiltration |
|
|
| Density gradient centrifugation |
|
|
| Inertial Focusing |
|
|
| Direct Imaging |
|
|
| Dielectrophoresis |
|
|
| Technology | Cancer (Subtype) | Clinical Utility | Number of Patients | Clinical Scenario | Outcome Measure | Remarks | Ref. |
|---|---|---|---|---|---|---|---|
| Cell Adhesion Matrix (CAM)-initiated CTC enrichment and flowcytometry | Advanced Epithelial Ovarian Cancer (stages I–IV) | Diagnosis Prognosis | 129 | -Invasive subpopulation of CTC (bind to collagen matrix type I & verified by flow cytometry). -iCTC threshold >5 CTCs/1 mL | OS and PFS | -88.6% had iCTC > 5/1 mL -More iCTCs in higher stage disease (38.5% in stage I vs. 95.2% in stage IV patients) -iCTC above threshold correlated with inferior OS and PFS -iCTC correlated better with OS and PFS compared to CA125 | [84] |
| CellSearch | Metastatic breast cancer | Prognosis | Pooled analysis of 1944 patients from 20 studies | -New line of treatment -CTC threshold ≥5 CTCs/7.5 mL | OS and PFS | -46.9% patients had ≥5 CTCs/7.5 mL -CTCs ≥ 5/7.5 mL had worse OS (HR:2.78) & PFS (HR:1.92) -An increase in CTC count post treatment correlated with decreased OS and PFS | [7] |
| CellSearch | Metastatic neuro-endocrine neoplasms | Prognosis | 138 | -New line of treatment -CTC threshold in 3 groups (1, 1–8, >8 /7.5mL) | OS and PFS | 10–15 weeks post treatment: strong association between CTC count and OS and PFS | [85] |
| CellSearch | Metastatic neuro-endocrine tumors | Prognosis | 176 | -CTC threshold ≥1 CTCs/7.5 mL | OS and PSF | -49% of patients had ≥1 CTC/7.5 mL -CTCs ≥ 1/7.5 mL was associated with inferior OS (HR: 8) and PFS (HR: 6.6) -holds in multivariate analysis | [86] |
| CellSearch | Non-Metastatic breast cancer (stages I–III) | Diagnosis Prognosis | Pooled analysis from 3173 patients | -CTC threshold ≥1 CTCs/7.5 mL | DFS, breast cancer-specific survival, OS | -CTCs in 20.2% of patients -presence of CTC associated with unfavorable clinical features -CTCs ≥ 1/7.5 mL associated with inferior DFS (HR: 1.82), distant-DFS (HR: 1.89), breast cancer specific survival (HR: 2.04), OS (HR: 1.97) | [87] |
| CellSearch | Non-Small-Cell Lung Cancer | Prognosis | 97 | -Before and after one cycle of chemotherapy -CTC threshold ≥50 CTCs/7.5 mL | OS and PFS | -85% tested positive for CTC -Before one cycle of chemotherapy: * CTCs ≥ 50/7.5 mL was associated with significantly worse OS & PFS -After one cycle of chemotherapy: * CTCs < 50/7.5 mL was associated with similar OS but better PFS | [88] |
| CellSearch and RT-PCR among others | Non-Small-Cell Lung Cancer | -Diagnosis -Prognosis | Pooled analysis from 1576 patients from 20 studies | -CTC threshold ≥1 CTCs/7.5 mL | OS and PFS | -CTC was correlated positively with lymph node metastasis (OR = 2.06) and tumor stage (OR = 1.95). -CTC were associated with shorter OS (RR = 2.19) and PFS (RR = 2.14) indicating poor prognosis | [89] |
| ScreenCell Cyto filtration device (size-based isolation) | Lung cancer (majority were stage I and II) | -Diagnosis | 77 | -Differentiate benign from malignant lesions using CTC count as well as clinicopathologic and histologic features | Diagnosis | -CTCs divided into 3 groups: Malignant features (MG), Undefined Malignant features (UMF) and benign features (BF). -CTC-MF count correlated with tumor size and stage with high sensitivity and specificity. -CTC-UMF were detected in 8% of malignant patients and 5% of benign patients -CTC-BF were detected in 88% of benign patients and 1% in malignant patients | [90] |
| CellSearch | Newly diagnosed breast cancer | -Diagnosis -Prognosis | 404 | -CTC assessment before undergoing surgical treatment -CTC threshold ≥1 CTCs/30 mL | Breast cancer-related death (BRD) and Relapse-free survival (RFS) at 4-years | -CTCs were detected more frequently in high stage tumors: * 15% of benign tumor patients * 19% in DCIS patients * 16% in stage I patients * 18% in stage II patients * 31% in stage III patients -11.6% of CTC negative patients developed recurrence compared to 21.1% of CTC positive patients -RFS was 88.4% in CTC negative patients compared to 78.9% in CTC positive patients -BRD was 4.3% in CTC negative patients compared to 14.5% in CTC positive patients | [91] |
| CellSearch | Curable colorectal cancer (stages I–IV) | -Diagnosis -Prognosis | 287 | -Preoperative assessment of CTC -CTC threshold ≥1 CTCs/7.5 mL | OS and PFS | -CTC was detected more frequently in metastatic patients -CTC were not associated with clinicopathological variables in non-metastatic patients -in preoperative CTC detection: CTCs≥1/7.5 mL was associated significantly with worse OS (HR = 5.5) and PFS (HR = 12.7) | [92] |
| CellSearch | Resectable esophageal cancer | -Prognosis | 100 | -Preoperative assessment of CTC -CTC threshold ≥1 CTCs/7.5 mL | OS and RFS | -CTCs in 18% of patients -CTC positive patients had inferior OS (HR: 3.128) and RFS (HR: 5.063), holds in multivariate analysis | [93] |
| Epic (Epic Sciences) | Metastatic castration-resistant prostate cancer | -Prognosis | 161 | -New line of treatment at the first follow-up | OS, rPFS (r:radio) | -CTC negative patients have better OS and rPFS compared to CTC positive patients -all AR-V7-CTC positive patients were resistant to ARS inhibitors (63% of CTC-positive cases) -AR-V7-CTC positive patients treated with Taxane had favorable OS compared to those treated with ARS inhibitors (HR = 0.24) | [62] |
| EPISOT, CellSearch and flowcytometry preceded by enrichment using RosetteSep™ (STEMCELL technologies) | Head and neck squamous cell carcinoma | -Prognosis | 65 | -New line of chemotherapy and cetuximab -CTC was assessed at Day 0 (D0, before treatment), D7, & D21 -CTC threshold ≥1 CTCs/10–15 mL | PFS | -Pretreatment, CTC was detected with EPISOT, CellSearch and Flowcytometry in 69%, 21% and 11% of patients respectively. -at D0, the median PFS was 5.3 months for all the 65 patients with 0.08 probability of survival at 12 months. -PFS was significantly higher in patients with no CTC, or reduction between D0 and D7, compared to stable or increased CTC -CTC count on D21 was not significantly associated with PFS | [94] |
| RosetteSep™ (STEMCELL technologies) enrichment followed by flowcytometry | Metastatic colorectal cancer | -Prognosis | 55 | -CTC assessment after the first cycle of treatment -CTC threshold >30 CTCs/mL | OS and FPS | -CTCs were detected in all patients -CTCs>30 /mL associated with inferior OS and PFS, holds in multivariate analysis | [95] |
| CellSearch and DEPArray | Chemosensitive and chemo-refractory small-cell lung cancer | -Diagnosis -Prognosis | 13 | -CTCs from pretreated patients examined for copy-number aberrations (CNAs) using NGS (Illumina) | OS and PFS | -88 single CTCs were testes from 31 patients. -The classifier correctly assigned 83.3% of cases as either chemorefractory or chemo-sensitive. -Significant difference in PFS, but not OS, between chemorefractory and chemosensitive patients | [96] |
| ScreenCell® | Lung Cancer | -Diagnosis -Mutation Screening | 93 | -KRAS mutation assessment in CTC and cell tumor DNA (ctDNA) using COLD-PCR/HRM assay | KRAS mutation | -KRAS mutation was identified in 21.3% of patients -Mutation analysis in matched CTC DNA revealed 20 mutations in 23.2% of the patients -Mutation analysis in matched ctDNA samples revealed 26 mutations in 30.5% of the patients -Greater sensitivity and specificity for KRAS mutation detection in ctDNA than in CTCs | [97] |
| CellSearch | -Advanced Non-Small-Cell Lung Cancer | -Prognosis | 41 | -CTC threshold >1 CTC/7.5 mL -EGFR mutation analysis in a single-armed phase II clinical trial of erlotinib and pertuzumab using TaqMan Gene Expression Assay | PFS and FDG-PET tomographic scan | -CTCs were detected in 78% of patients -Greater sensitivity for EGFR mutation detection in ctDNA than in CTCs -Lower CTC count was associated with longer PFS | [98] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Habli, Z.; AlChamaa, W.; Saab, R.; Kadara, H.; Khraiche, M.L. Circulating Tumor Cell Detection Technologies and Clinical Utility: Challenges and Opportunities. Cancers 2020, 12, 1930. https://doi.org/10.3390/cancers12071930
Habli Z, AlChamaa W, Saab R, Kadara H, Khraiche ML. Circulating Tumor Cell Detection Technologies and Clinical Utility: Challenges and Opportunities. Cancers. 2020; 12(7):1930. https://doi.org/10.3390/cancers12071930
Chicago/Turabian StyleHabli, Zeina, Walid AlChamaa, Raya Saab, Humam Kadara, and Massoud L. Khraiche. 2020. "Circulating Tumor Cell Detection Technologies and Clinical Utility: Challenges and Opportunities" Cancers 12, no. 7: 1930. https://doi.org/10.3390/cancers12071930
APA StyleHabli, Z., AlChamaa, W., Saab, R., Kadara, H., & Khraiche, M. L. (2020). Circulating Tumor Cell Detection Technologies and Clinical Utility: Challenges and Opportunities. Cancers, 12(7), 1930. https://doi.org/10.3390/cancers12071930





