Effectiveness and Healthcare Cost of Adding Trastuzumab to Standard Chemotherapy for First-Line Treatment of Metastatic Gastric Cancer: A Population-Based Cohort Study
Abstract
1. Introduction
2. Results
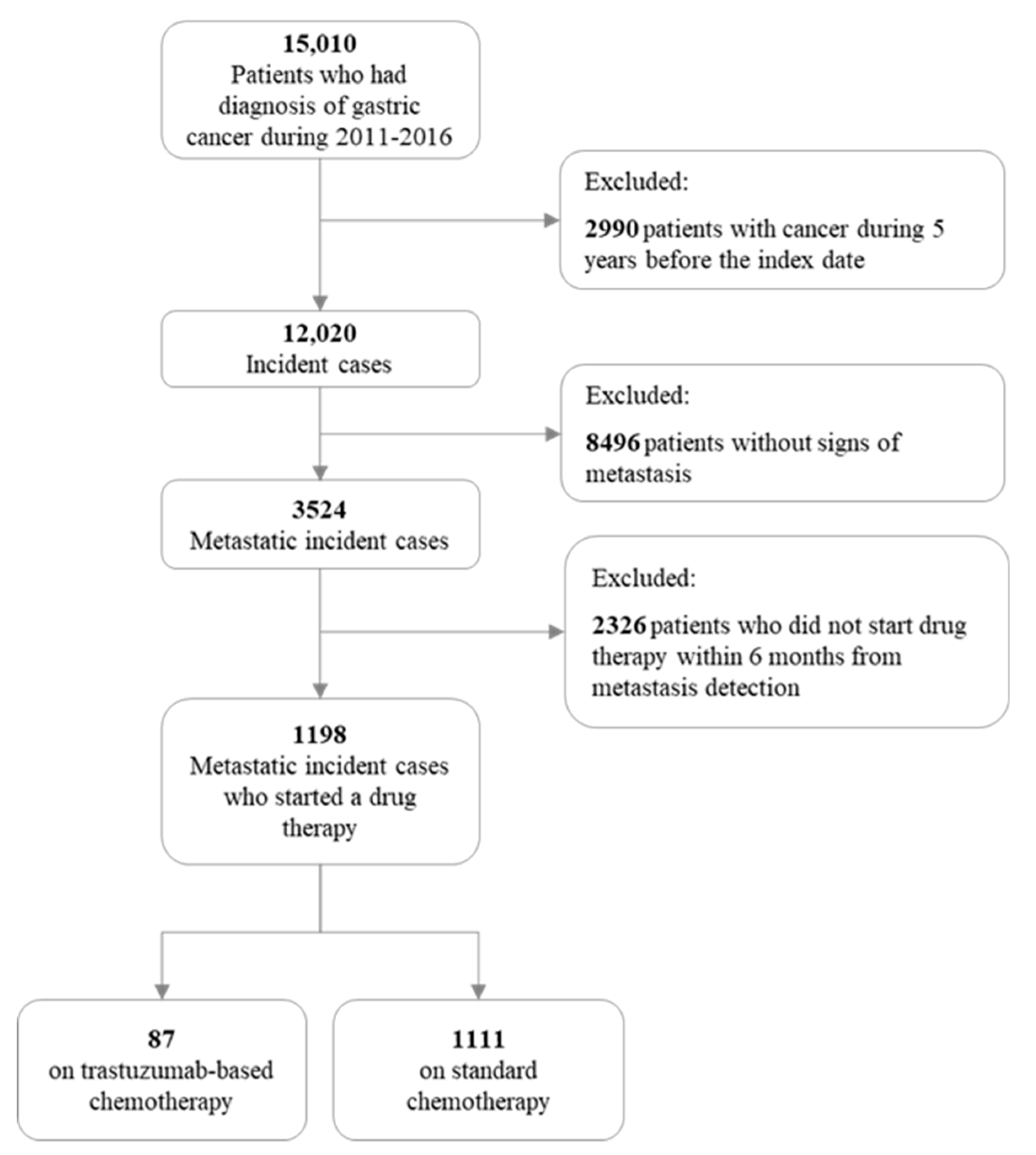
2.1. Patients
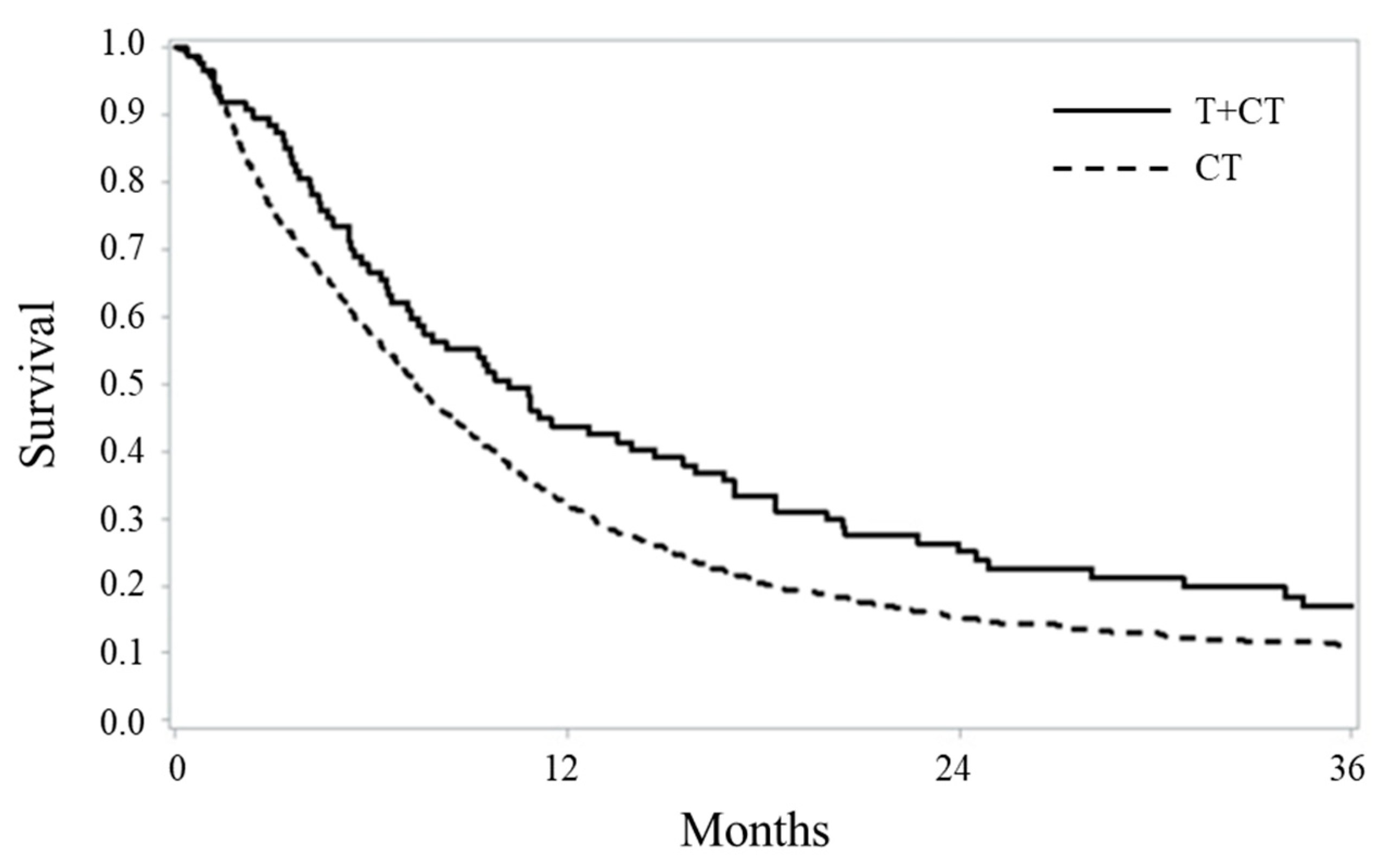
2.2. Survival Comparisons
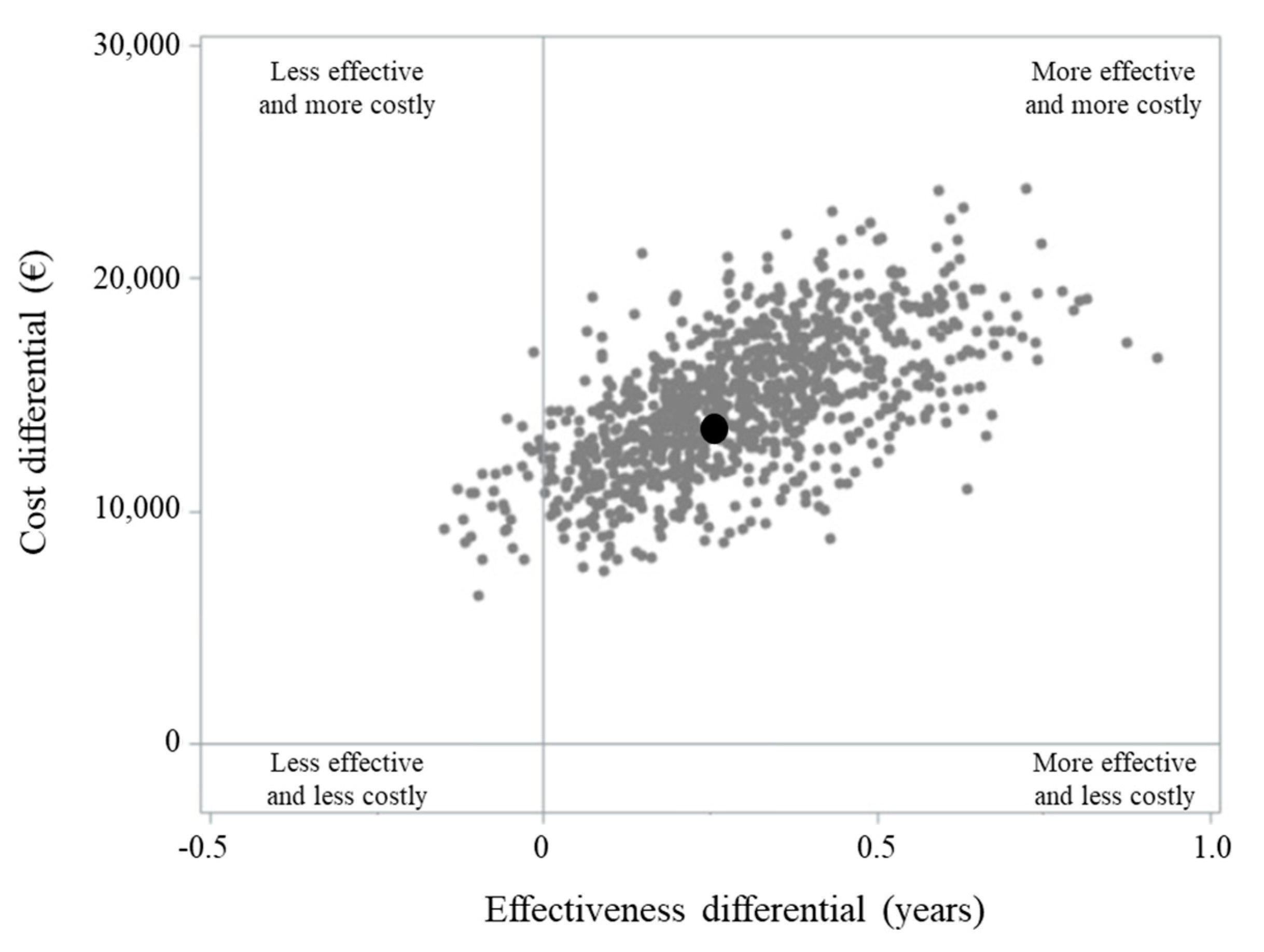
2.3. Healthcare Cost and Cost-Effectiveness Profile
2.4. Sensitivity Analyses
3. Discussion
4. Materials and Methods
4.1. Setting
4.2. Cohort Selection and Follow-Up
4.3. First-Line Therapy
4.4. Baseline Characteristics
4.5. Statistical Analyses
4.6. Sensitivity Analyses
4.7. Considerations on Sample Size
4.8. Ethical Issues
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Herceptin Approval History. Available online: https://www.drugs.com/history/herceptin.html (accessed on 25 April 2020).
- Bang, Y.J.; Van Cutsem, E.; Feyereislova, A.; Chung, H.C.; Shen, L.; Sawaki, A.; Lordick, F.; Ohtsu, A.; Omuro, Y.; Satoh, T.; et al. ToGA Trial Investigators. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet 2010, 376, 687–697. [Google Scholar] [CrossRef]
- Italian Association of Clinical Oncology (Associazione Italiana di Oncologia Medica—AIOM). Guidelines on Stomach Cancer 2019. Available online: https://www.aiom.it/linee-guida-aiom-neoplasie-dello-stomaco-e-della-funzione-gastroesofagea-2019/ (accessed on 25 April 2020).
- Di Tanna, G.L.; Bychenkova, A.; O’Neill, F.; Wirtz, H.S.; Miller, P.; Hartaigh, B.O.; Globe, G. Evaluating Cost-Effectiveness Models for Pharmacologic Interventions in Adults with Heart Failure: A Systematic Literature Review. Pharmacoeconomics 2019, 37, 359–389. [Google Scholar] [CrossRef] [PubMed]
- Eichler, H.G.; Kong, S.X.; Gerth, W.C.; Mavros, P.; Jönsson, B. Use of cost-effectiveness analysis in health-care resource allocation decision-making: How are cost-effectiveness thresholds expected to emerge? Value Health 2004, 7, 518–528. [Google Scholar] [CrossRef]
- Hannan, E.L. Randomized clinical trials and observational studies: Guidelines for assessing respective strengths and limitations. JACC Cardiovasc. Interv. 2008, 1, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Abrahao-Machado, L.F.; Scapulatempo-Neto, C. HER2 testing in gastric cancer: An update. World J. Gastroenterol. 2016, 22, 4619–4625. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Sasako, M.; Mizusawa, J.; Hirota, S.; Ochiai, A.; Kushima, R.; Katai, H.; Tanaka, Y.; Fukushima, N.; Nashimoto, A.; et al. HER2 expression in locally advanced gastric cancer with extensive lymph node (bulky N2 or paraaortic) metastasis (JCOG1005-A trial). Gastric Cancer 2015, 18, 467–475. [Google Scholar] [CrossRef]
- Qiu, M.; Zhou, Y.; Zhang, X.; Wang, Z.; Wang, F.; Shao, J.; Lu, J.; Jin, Y.; Wei, X.; Zhang, D.; et al. Lauren classification combined with HER2 status is a better prognostic factor in Chinese gastric cancer patients. BMC Cancer 2014, 14, 823. [Google Scholar] [CrossRef]
- Xu, C.C.; Yue, L.; Wei, H.J.; Zhao, W.W.; Sui, A.H.; Wang, X.M.; Qiu, W.S. Significance of TFF3 protein and Her-2/neu status in patients with gastric adenocarcinoma. Pathol. Res. Pract. 2013, 209, 479–485. [Google Scholar] [CrossRef]
- Janjigian, Y.Y.; Werner, D.; Pauligk, C.; Steinmetz, K.; Kelsen, D.P.; Jäger, E.; Altmannsberger, H.M.; Robinson, E.; Tafe, L.J.; Tang, L.H.; et al. Prognosis of metastatic gastric and gastroesophageal junction cancer by HER2 status: A European and USA International collaborative analysis. Ann. Oncol. 2012, 23, 2656–2662. [Google Scholar] [CrossRef]
- Chen, C.; Yang, J.M.; Hu, T.T.; Xu, T.J.; Yan, G.; Hu, S.L.; Wei, W.; Xu, W.P. Prognostic role of human epidermal growth factor receptor in gastric cancer: A systematic review and meta-analysis. Arch. Med. Res. 2013, 44, 380–389. [Google Scholar] [CrossRef]
- Liang, J.W.; Zhang, J.J.; Zhang, T.; Zheng, Z.C. Clinicopathological and prognostic significance of HER2 overexpression in gastric cancer: A meta-analysis of the literature. Tumour Biol. 2014, 35, 4849–4858. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.Y.; Huang, J.Y.; Zhao, Q.R.; Jiang, N.; Xu, H.M.; Wang, Z.N.; Li, H.Q.; Zhang, S.B.; Sun, Z. The clinicopathological parameters and prognostic significance of HER2 expression in gastric cancer patients: A meta-analysis of literature. World J. Surg. Oncol. 2017, 15, 68. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zheng, G.; Chen, L.; Xiong, B. Effect of HER-2/neu over-expression on prognosis in gastric cancer: A meta-analysis. Asian Pac. J. Cancer Prev. 2011, 12, 1417–1423. [Google Scholar]
- Gu, J.; Zheng, L.; Wang, Y.; Zhu, M.; Wang, Q.; Li, X. Prognostic significance of HER2 expression based on trastuzumab for gastric cancer (ToGA) criteria in gastric cancer: An updated meta-analysis. Tumour Biol. 2014, 35, 5315–5321. [Google Scholar] [CrossRef] [PubMed]
- Shiroiwa, T.; Fukuda, T.; Shimozuma, K. Cost-effectiveness analysis of trastuzumab to treat HER2-positive advanced gastric cancer based on the randomised ToGA trial. Br. J. Cancer. 2011, 105, 1273–1278. [Google Scholar] [CrossRef]
- Wu, B.; Ye, M.; Chen, H.; Shen, J.F. Costs of trastuzumab in combination with chemotherapy for HER2-positive advanced gastric or gastroesophageal junction cancer: An economic evaluation in the Chinese context. Clin. Ther. 2012, 34, 468–479. [Google Scholar] [CrossRef] [PubMed]
- Franchi, M.; Barni, S.; Tagliabue, G.; Ricci, P.; Mazzucco, W.; Tumino, R.; Caputo, A.; Corrao, G.; GRETA working group. Effectiveness of First-Line Bevacizumab in Metastatic Colorectal Cancer: The Observational Cohort Study GRETA. Oncologist 2019, 24, 358–365. [Google Scholar] [CrossRef]
- Corrao, G.; Rea, F.; Di Martino, M.; De Palma, R.; Scondotto, S.; Fusco, D.; Lallo, A.; Belotti, L.M.B.; Ferrante, M.; Addario, S.P.; et al. Developing and validating a novel multisource comorbidity score from administrative data: A large population-based cohort study from Italy. BMJ Open 2017, 7, e019503. [Google Scholar] [CrossRef]
- Rea, F.; Ieva, F.; Pasotorino, U.; Apolone, G.; Barni, S.; Merlino, L.; Franchi, M.; Corrao, G. Number of lung resections performed and long-term mortality rates of patients after lung cancer surgery: Evidence from an Italian investigation. Eur. J. Cardiothorac. Surg. 2020. [Google Scholar] [CrossRef]
- Bang, H.; Tsiatis, A.A. Median regression with censored cost data. Biometrics 2002, 58, 643–649. [Google Scholar] [CrossRef]
- Zhao, L.; Claggett, B.; Tian, L.; Uno, H.; Pfeffer, M.A.; Solomon, S.D.; Trippa, L.; Wei, L.J. On the restricted mean survival time curve in survival analysis. Biometrics 2016, 72, 215–221. [Google Scholar] [CrossRef]
- Pak, K.; Uno, H.; Kim, D.H.; Tian, L.; Kane, R.C.; Takeuchi, M.; Fu, H.; Claggett, B.; Wei, L.J. Interpretability of cancer clinical trial results using restricted mean survival time as an alternative to the hazard ratio. JAMA Oncol. 2017, 3, 1692–1696. [Google Scholar] [CrossRef]
- Kim, D.H.; Uno, H.; Wei, L.J. Restricted mean survival time as a measure to interpret clinical trial results. JAMA Cardiol. 2017, 2, 1179–1180. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, H. A study on confidence intervals for incremental cost-effectiveness ratios. Biometrical J. 2008, 50, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Canivet, C.; Costa, N.; Ory-Magne, F.; Arcari, C.; Mohara, C.; Pourcel, L.; Dermeaux, H.; Bérard, E.; Bourrel, R.; Molinier, L.; et al. Clinical impact and cost-effectiveness of an education program for PD patients: A randomized controlled trial. PLoS ONE 2016, 11, e0162646. [Google Scholar] [CrossRef] [PubMed]
- European Medicine Agency (EMA). Herceptin—Summary of Product Characteristics. Available online: https://www.ema.europa.eu/en/documents/product-information/herceptin-epar-product-information_en.pdf (accessed on 25 April 2020).
- Schneeweiss, S. Sensitivity analysis and external adjustment for unmeasured confounders in epidemiologic database studies of therapeutics. Pharmacoepidemiol. Drug Saf. 2006, 15, 291–303. [Google Scholar] [CrossRef]
- Haukoos, J.S.; Lewis, R.J. The propensity score. JAMA 2015, 314, 1637–1638. [Google Scholar] [CrossRef]
| Characteristic | Treatment Group | p-Value | |
|---|---|---|---|
| T + CT (N = 87) | CT (N = 1111) | ||
| Age at diagnosis | |||
| <55 | 10 (11.5) | 204 (18.4) | 0.140 * |
| 55–64 | 31 (35.6) | 285 (25.7) | |
| 65–74 | 31 (35.6) | 371 (33.4) | |
| 75 | 15 (17.2) | 251 (22.6) | |
| Median | 67 | 66 | |
| Sex | |||
| Women | 23 (26.4) | 402 (36.2) | 0.067 |
| Men | 64 (73.6) | 709 (63.8) | |
| Year of diagnosis | |||
| 2011 | 15 (17.2) | 243 (21.9) | 0.674 * |
| 2012 | 15 (17.2) | 156 (14.0) | |
| 2013 | 10 (11.5) | 174 (15.7) | |
| 2014 | 21 (24.1) | 176 (15.8) | |
| 2015 | 14 (16.1) | 200 (18.0) | |
| 2016 | 12 (13.8) | 162 (14.6) | |
| Surgery | |||
| No | 62 (71.3) | 862 (77.6) | 0.176 |
| Yes | 25 (28.7) | 249 (22.4) | |
| MCS score | |||
| 0–2 | 54 (62.1) | 669 (60.2) | 0.716 * |
| 3–5 | 27 (31.0) | 349 (31.4) | |
| 6–8 | 4 (4.6) | 73 (6.6) | |
| ≥9 | 2 (2.3) | 20 (1.8) | |
| Variable | N (# Deaths) | Hazard Ratio (HR) (95% CI) |
|---|---|---|
| Exposure | ||
| CT | 1111 (994) | 1 a |
| T + CT | 87 (76) | 0.73 (0.57–0.93) |
| Age | ||
| <55 | 196 (187) | 1 a |
| 55–64 | 297 (284) | 1.08 (0.90–1.31) |
| 65–74 | 362 (363) | 1.06 (0.88–1.27) |
| 75 | 251 (236) | 1.18 (0.96–1.43) |
| Sex | ||
| F | 708 (377) | 1 a |
| M | 398 (693) | 1.23 (1.08–1.40) |
| Year of diagnosis | ||
| 2011 | 258 (235) | 1 a |
| 2012 | 171 (156) | 1.04 (0.85–1.28) |
| 2013 | 184 (174) | 1.05 (0.86–1.29) |
| 2014 | 197 (179) | 1.02 (0.83–1.24) |
| 2015 | 214 (191) | 1.08 (0.89–1.31) |
| 2016 | 174 (135) | 0.84 (0.68–1.04) |
| Surgery | ||
| No | 885 (854) | 1 a |
| Yes | 251 (216) | 0.52 (0.45–0.61) |
| MCS index | ||
| 0–2 | 682 (631) | 1 a |
| 3–5 | 337 (347) | 1.13 (0.98–1.29) |
| 6–8 | 68 (72) | 1.25 (0.97–1.61) |
| 9 | 5 (20) | 1.10 (0.70–1.72) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franchi, M.; Tritto, R.; Torroni, L.; Reno, C.; La Vecchia, C.; Corrao, G. Effectiveness and Healthcare Cost of Adding Trastuzumab to Standard Chemotherapy for First-Line Treatment of Metastatic Gastric Cancer: A Population-Based Cohort Study. Cancers 2020, 12, 1691. https://doi.org/10.3390/cancers12061691
Franchi M, Tritto R, Torroni L, Reno C, La Vecchia C, Corrao G. Effectiveness and Healthcare Cost of Adding Trastuzumab to Standard Chemotherapy for First-Line Treatment of Metastatic Gastric Cancer: A Population-Based Cohort Study. Cancers. 2020; 12(6):1691. https://doi.org/10.3390/cancers12061691
Chicago/Turabian StyleFranchi, Matteo, Roberta Tritto, Lorena Torroni, Chiara Reno, Carlo La Vecchia, and Giovanni Corrao. 2020. "Effectiveness and Healthcare Cost of Adding Trastuzumab to Standard Chemotherapy for First-Line Treatment of Metastatic Gastric Cancer: A Population-Based Cohort Study" Cancers 12, no. 6: 1691. https://doi.org/10.3390/cancers12061691
APA StyleFranchi, M., Tritto, R., Torroni, L., Reno, C., La Vecchia, C., & Corrao, G. (2020). Effectiveness and Healthcare Cost of Adding Trastuzumab to Standard Chemotherapy for First-Line Treatment of Metastatic Gastric Cancer: A Population-Based Cohort Study. Cancers, 12(6), 1691. https://doi.org/10.3390/cancers12061691








