Neuroprotection of Radiosensitive Juvenile Mice by Ultra-High Dose Rate FLASH Irradiation
Abstract
1. Introduction
2. Results
2.1. FLASH-RT Preserves Memory Updating in OUL over Time
2.2. FLASH-RT Minimizes Radiation-Induced Novel Object Memory Impairments as Well as Anxiety-Like Behaviors
2.3. FLASH-RT Spares the Neurogenic Niche in Juvenile Mice
2.4. FLASH-RT Preserves Neurogenesis in Juvenile Mice
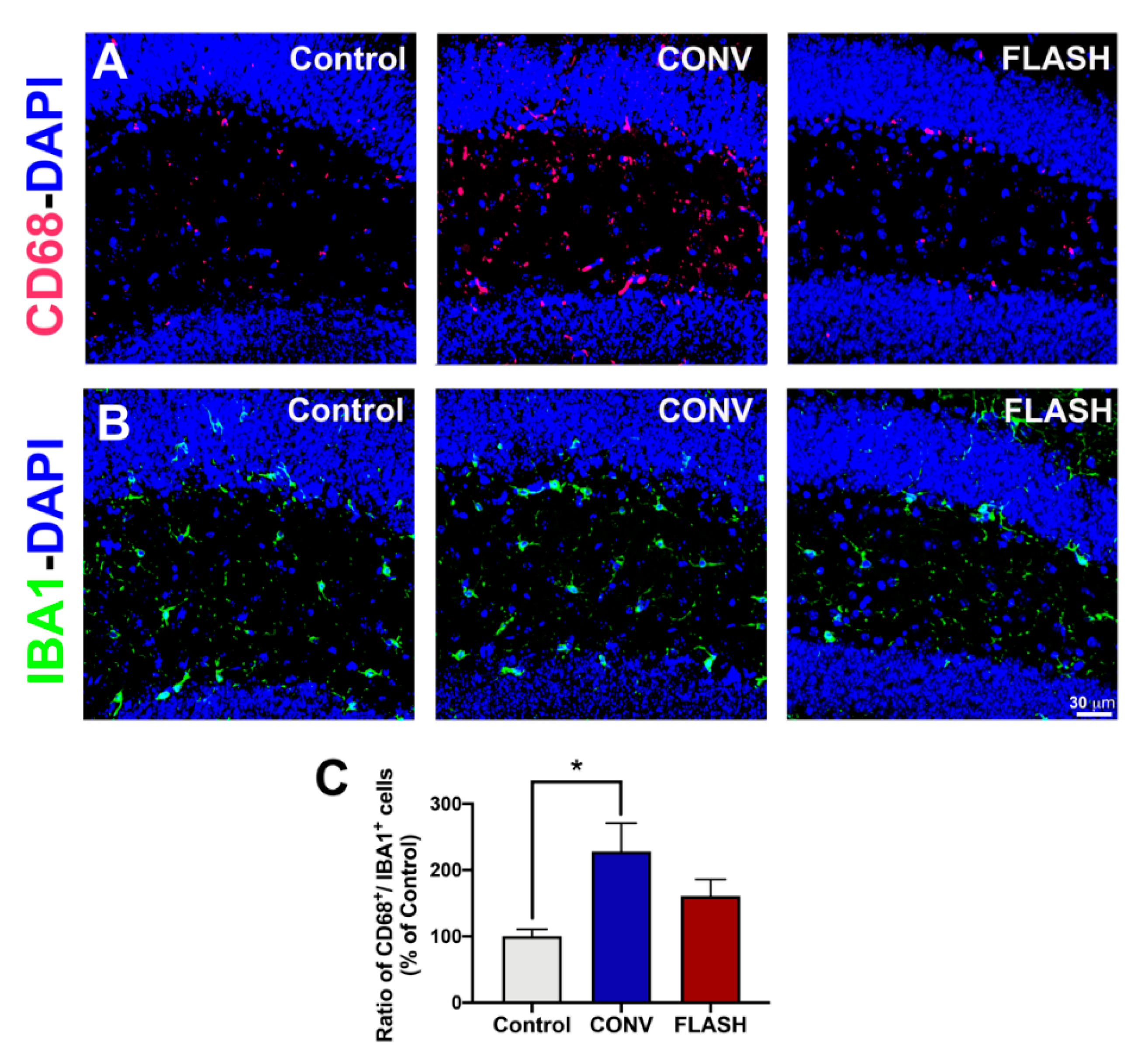
2.5. Attenuation of Activated Microglia by FLASH-RT
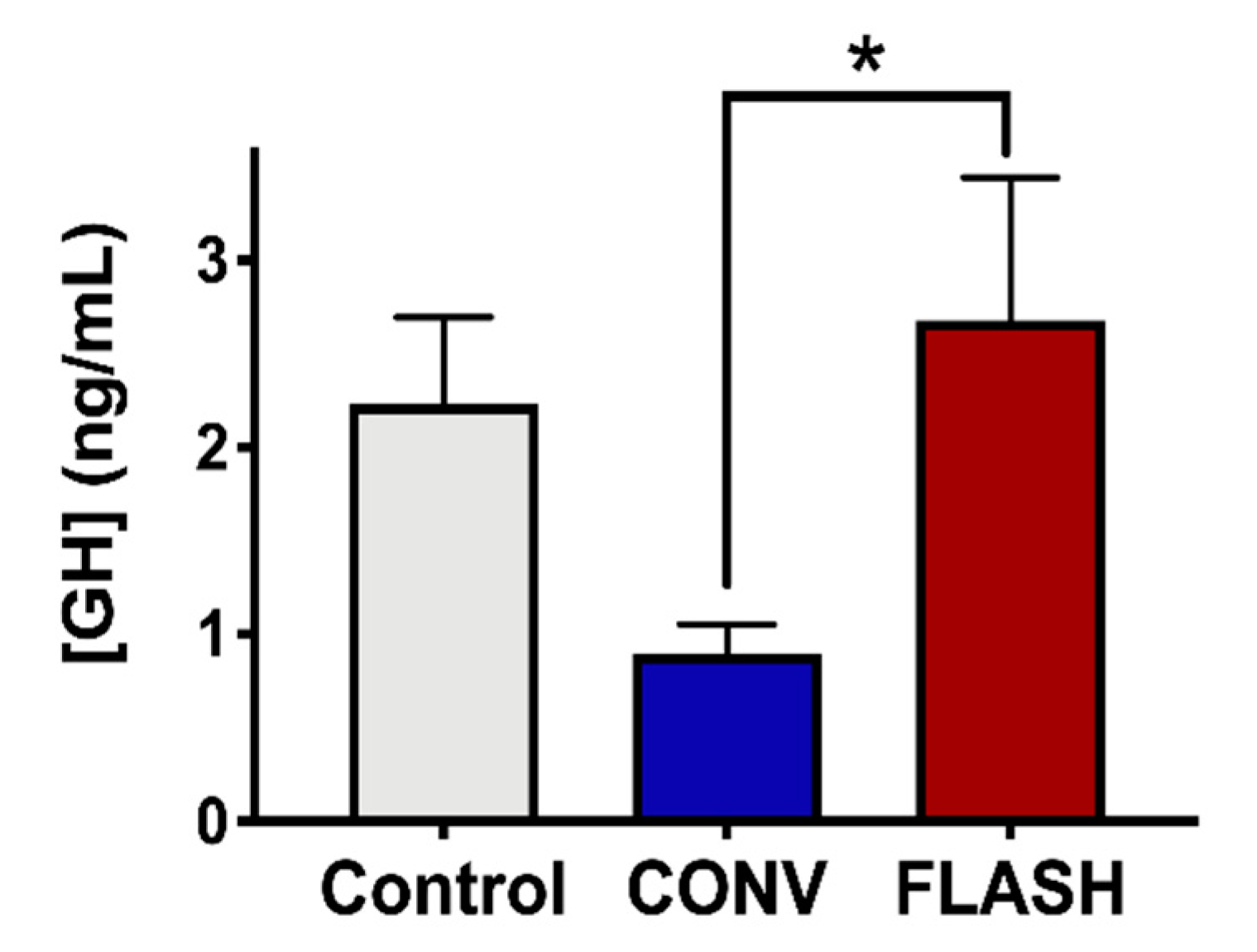
2.6. Preservation of the Endocrine System by FLASH-RT
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Whole Brain Irradiations
4.3. Cognitive Testing
4.4. Apparatus
4.5. Objects in Updated Locations (OUL) Task
4.6. Novel Object Recognition (NOR)
4.7. Light-Dark Box (LDB) Test
4.8. Social Interaction Test (SIT)
4.9. Fear Extinction Testing
4.10. BrdU Treatment and Immunohistochemistry Analyses
4.11. Plasma Growth Hormone Quantification
4.12. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smee, R.I.; Williams, J.R.; De-Loyde, K.J.; Meagher, N.S.; Cohn, R. Medulloblastoma: Progress over time. J. Med. Imaging Radiat. Oncol. 2012, 56, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Udaka, Y.T.; Packer, R.J. Pediatric Brain Tumors. Neurol. Clin. 2018, 36, 533–556. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.M.; Raabe, E.; Eberhart, C.; Cohen, K.J. Management of pediatric and adult patients with medulloblastoma. Curr. Treat. Options Oncol. 2014, 15, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Mulhern, R.K.; Palmer, S.L.; Reddick, W.E.; Glass, J.O.; Kun, L.E.; Taylor, J.; Langston, J.; Gajjar, A. Risks of young age for selected neurocognitive deficits in medulloblastoma are associated with white matter loss. J. Clin. Oncol. 2001, 19, 472–479. [Google Scholar] [CrossRef]
- Saury, J.M.; Emanuelson, I. Cognitive consequences of the treatment of medulloblastoma among children. Pediatr. Neurol. 2011, 44, 21–30. [Google Scholar] [CrossRef]
- Roongpiboonsopit, D.; Kuijf, H.J.; Charidimou, A.; Xiong, L.; Vashkevich, A.; Martinez-Ramirez, S.; Shih, H.A.; Gill, C.M.; Viswanathan, A.; Dietrich, J. Evolution of cerebral microbleeds after cranial irradiation in medulloblastoma patients. Neurology 2017, 88, 789–796. [Google Scholar] [CrossRef]
- Tanyildizi, Y.; Keweloh, S.; Neu, M.A.; Russo, A.; Wingerter, A.; Weyer-Elberich, V.; Stockinger, M.; Schmidberger, H.; Brockmann, M.A.; Faber, J. Radiation-induced vascular changes in the intracranial irradiation field in medulloblastoma survivors: An MRI study. Radiother. Oncol. 2019, 136, 50–55. [Google Scholar] [CrossRef]
- Edelstein, K.; Spiegler, B.J.; Fung, S.; Panzarella, T.; Mabbott, D.J.; Jewitt, N.; D’Agostino, N.M.; Mason, W.P.; Bouffet, E.; Tabori, U.; et al. Early aging in adult survivors of childhood medulloblastoma: Long-term neurocognitive, functional, and physical outcomes. Neuro-Oncology 2011, 13, 536–545. [Google Scholar] [CrossRef]
- Grill, J.; Renaux, V.K.; Bulteau, C.; Viguier, D.; Levy-Piebois, C.; Sainte-Rose, C.; Dellatolas, G.; Raquin, M.A.; Jambaque, I.; Kalifa, C. Long-term intellectual outcome in children with posterior fossa tumors according to radiation doses and volumes. Int. J. Radiat. Oncol. Biol. Phys. 1999, 45, 137–145. [Google Scholar] [CrossRef]
- Packer, R.J.; Zhou, T.; Holmes, E.; Vezina, G.; Gajjar, A. Survival and secondary tumors in children with medulloblastoma receiving radiotherapy and adjuvant chemotherapy: Results of Children’s Oncology Group trial A9961. Neuro-Oncology 2013, 15, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Favaudon, V.; Caplier, L.; Monceau, V.; Pouzoulet, F.; Sayarath, M.; Fouillade, C.; Poupon, M.F.; Brito, I.; Hupe, P.; Bourhis, J.; et al. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci. Transl. Med. 2014, 6, 245ra293. [Google Scholar] [CrossRef] [PubMed]
- Montay-Gruel, P.; Petersson, K.; Jaccard, M.; Boivin, G.; Germond, J.F.; Petit, B.; Doenlen, R.; Favaudon, V.; Bochud, F.; Bailat, C.; et al. Irradiation in a flash: Unique sparing of memory in mice after whole brain irradiation with dose rates above 100 Gy/s. Radiother. Oncol. 2017, 124, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Bourhis, J.; Montay-Gruel, P.; Goncalves Jorge, P.; Bailat, C.; Petit, B.; Ollivier, J.; Jeanneret-Sozzi, W.; Ozsahin, M.; Bochud, F.; Moeckli, R.; et al. Clinical translation of FLASH radiotherapy: Why and how? Radiother. Oncol. 2019. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Acharya, M.M.; Petersson, K.; Alikhani, L.; Yakkala, C.; Allen, B.D.; Ollivier, J.; Petit, B.; Jorge, P.G.; Syage, A.R.; et al. Long-term neurocognitive benefits of FLASH radiotherapy driven by reduced reactive oxygen species. Proc. Natl. Acad. Sci. USA 2019, 116, 10943–10951. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Bouchet, A.; Jaccard, M.; Patin, D.; Serduc, R.; Aim, W.; Petersson, K.; Petit, B.; Bailat, C.; Bourhis, J.; et al. X-rays can trigger the FLASH effect: Ultra-high dose-rate synchrotron light source prevents normal brain injury after whole brain irradiation in mice. Radiother. Oncol. 2018, 129, 582–588. [Google Scholar] [CrossRef]
- Petersson, K.; Adrian, G.; Butterworth, K.; McMahon, S.J. A quantitative analysis of the role of oxygen tension in FLASH radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2020. [Google Scholar] [CrossRef]
- Simmons, D.A.; Lartey, F.M.; Schuler, E.; Rafat, M.; King, G.; Kim, A.; Ko, R.; Semaan, S.; Gonzalez, S.; Jenkins, M.; et al. Reduced cognitive deficits after FLASH irradiation of whole mouse brain are associated with less hippocampal dendritic spine loss and neuroinflammation. Radiother. Oncol. 2019, 139, 4–10. [Google Scholar] [CrossRef]
- Vozenin, M.C.; De Fornel, P.; Petersson, K.; Favaudon, V.; Jaccard, M.; Germond, J.F.; Petit, B.; Burki, M.; Ferrand, G.; Patin, D.; et al. The Advantage of FLASH Radiotherapy Confirmed in Mini-pig and Cat-cancer Patients. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019, 25, 35–42. [Google Scholar] [CrossRef]
- Makale, M.T.; McDonald, C.R.; Hattangadi-Gluth, J.A.; Kesari, S. Mechanisms of radiotherapy-associated cognitive disability in patients with brain tumours. Nat. Rev. Neurol. 2017, 13, 52–64. [Google Scholar] [CrossRef]
- Tofilon, P.J.; Fike, J.R. The radioresponse of the central nervous system: A dynamic process. Radiat. Res. 2000, 153, 357–370. [Google Scholar] [CrossRef]
- Vozenin, M.C.; Hendry, J.H.; Limoli, C.L. Biological Benefits of Ultra-high Dose Rate FLASH Radiotherapy: Sleeping Beauty Awoken. Clin. Oncol. R. Coll. Radiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Acharya, M.M.; Green, K.N.; Allen, B.D.; Najafi, A.R.; Syage, A.; Minasyan, H.; Le, M.T.; Kawashita, T.; Giedzinski, E.; Parihar, V.K.; et al. Elimination of microglia improves cognitive function following cranial irradiation. Sci. Rep. 2016, 6, 31545. [Google Scholar] [CrossRef]
- Craver, B.M.; Acharya, M.M.; Allen, B.D.; Benke, S.N.; Hultgren, N.W.; Baulch, J.E.; Limoli, C.L. 3D surface analysis of hippocampal microvasculature in the irradiated brain. Environ. Mol. Mutagen. 2016, 57, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Parihar, V.K.; Limoli, C.L. Cranial irradiation compromises neuronal architecture in the hippocampus. Proc. Natl. Acad. Sci. USA 2013, 110, 12822–12827. [Google Scholar] [CrossRef]
- Smith, S.M.; Giedzinski, E.; Angulo, M.C.; Lui, T.; Lu, C.; Park, A.L.; Tang, S.; Martirosian, V.; Ru, N.; Chmielewski, N.N.; et al. Functional Equivalence of Stem Cell and Stem Cell-Derived Extracellular Vesicle Transplantation to Repair the Irradiated Brain. Stem Cells Transl. Med. 2019. [Google Scholar] [CrossRef]
- Kwapis, J.L.; Alaghband, Y.; Lopez, A.J.; Long, J.M.; Li, X.; Shu, G.; Bodinayake, K.K.; Matheos, D.P.; Rapp, P.R.; Wood, M.A. HDAC3-Mediated Repression of the Nr4a Family Contributes to Age-Related Impairments in Long-Term Memory. J. Neurosci. 2019, 39, 4999–5009. [Google Scholar] [CrossRef]
- Butler, J.M.; Rapp, S.R.; Shaw, E.G. Managing the cognitive effects of brain tumor radiation therapy. Curr. Treat. Options Oncol. 2006, 7, 517–523. [Google Scholar] [CrossRef]
- Caceres, L.G.; Cid, M.P.; Uran, S.L.; Zorrilla Zubilete, M.A.; Salvatierra, N.A.; Guelman, L.R. Pharmacological alterations that could underlie radiation-induced changes in associative memory and anxiety. Pharm. Biochem. Behav. 2013, 111, 37–43. [Google Scholar] [CrossRef]
- Tang, Y.; Luo, D.; Rong, X.; Shi, X.; Peng, Y. Psychological disorders, cognitive dysfunction and quality of life in nasopharyngeal carcinoma patients with radiation-induced brain injury. PLoS ONE 2012, 7, e36529. [Google Scholar] [CrossRef]
- Yen, P.N.; Lin, I.F.; Chang, W.P.; Wang, J.D.; Chang, T.C.; Kuo, K.L.; Hwang, J.S.; Liu, I.C.; Chen, Y.T.; Yang, C.C. Risk factors of depression after prolonged low-dose rate environmental radiation exposure. Int. J. Radiat. Biol. 2014, 90, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Bourin, M.; Hascoet, M. The mouse light/dark box test. Eur. J. Pharmacol. 2003, 463, 55–65. [Google Scholar] [CrossRef]
- Finlay, J.M.; Dunham, G.A.; Isherwood, A.M.; Newton, C.J.; Nguyen, T.V.; Reppar, P.C.; Snitkovski, I.; Paschall, S.A.; Greene, R.W. Effects of prefrontal cortex and hippocampal NMDA NR1-subunit deletion on complex cognitive and social behaviors. Brain Res. 2015, 1600, 70–83. [Google Scholar] [CrossRef] [PubMed]
- Ko, J. Neuroanatomical Substrates of Rodent Social Behavior: The Medial Prefrontal Cortex and Its Projection Patterns. Front. Neural Circuits 2017, 11, 41. [Google Scholar] [CrossRef]
- Gunaydin, L.A.; Grosenick, L.; Finkelstein, J.C.; Kauvar, I.V.; Fenno, L.E.; Adhikari, A.; Lammel, S.; Mirzabekov, J.J.; Airan, R.D.; Zalocusky, K.A.; et al. Natural neural projection dynamics underlying social behavior. Cell 2014, 157, 1535–1551. [Google Scholar] [CrossRef]
- Sato, A.; Mizuguchi, M.; Ikeda, K. Social interaction test: A sensitive method for examining autism-related behavioral deficits. Protoc. Exch. 2013, 1–9. [Google Scholar] [CrossRef]
- Winslow, J.T. Mouse social recognition and preference. Curr. Protoc. Neurosci. 2003, 22, 8–16. [Google Scholar] [CrossRef]
- Fike, J.R.; Rosi, S.; Limoli, C.L. Neural precursor cells and central nervous system radiation sensitivity. Semin. Radiat. Oncol. 2009, 19, 122–132. [Google Scholar] [CrossRef]
- Duffner, P.K. Long-term effects of radiation therapy on cognitive and endocrine function in children with leukemia and brain tumors. Neurologist 2004, 10, 293–310. [Google Scholar] [CrossRef]
- Harrington, K.J. Ultrahigh Dose-rate Radiotherapy: Next Steps for FLASH-RT. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019, 25, 3–5. [Google Scholar] [CrossRef]
- King, A.A.; Seidel, K.; Di, C.; Leisenring, W.M.; Perkins, S.M.; Krull, K.R.; Sklar, C.A.; Green, D.M.; Armstrong, G.T.; Zeltzer, L.K.; et al. Long-term neurologic health and psychosocial function of adult survivors of childhood medulloblastoma/PNET: A report from the Childhood Cancer Survivor Study. Neuro-Oncology 2017, 19, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Gajjar, A.; Chintagumpala, M.; Ashley, D.; Kellie, S.; Kun, L.E.; Merchant, T.E.; Woo, S.; Wheeler, G.; Ahern, V.; Krasin, M.J.; et al. Risk-adapted craniospinal radiotherapy followed by high-dose chemotherapy and stem-cell rescue in children with newly diagnosed medulloblastoma (St Jude Medulloblastoma-96): Long-term results from a prospective, multicentre trial. Lancet Oncol. 2006, 7, 813–820. [Google Scholar] [CrossRef]
- Komotar, R.J.; Otten, M.L.; Garrett, M.C.; Anderson, R.C. Treatment of early childhood medulloblastoma by postoperative chemotherapy alone-a critical review. Clin. Med. Oncol. 2009, 3, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Parkes, J.; Hendricks, M.; Ssenyonga, P.; Mugamba, J.; Molyneux, E.; Schouten-van Meeteren, A.; Qaddoumi, I.; Fieggen, G.; Luna-Fineman, S.; Howard, S.; et al. SIOP PODC adapted treatment recommendations for standard-risk medulloblastoma in low and middle income settings. Pediatr. Blood Cancer 2015, 62, 553–564. [Google Scholar] [CrossRef] [PubMed]
- Kahalley, L.S.; Ris, M.D.; Grosshans, D.R.; Okcu, M.F.; Paulino, A.C.; Chintagumpala, M.; Moore, B.D.; Guffey, D.; Minard, C.G.; Stancel, H.H.; et al. Comparing Intelligence Quotient Change After Treatment With Proton Versus Photon Radiation Therapy for Pediatric Brain Tumors. J. Clin. Oncol. 2016, 34, 1043–1049. [Google Scholar] [CrossRef]
- Palmer, S.L.; Gajjar, A.; Reddick, W.E.; Glass, J.O.; Kun, L.E.; Wu, S.; Xiong, X.; Mulhern, R.K. Predicting intellectual outcome among children treated with 35–40 Gy craniospinal irradiation for medulloblastoma. Neuropsychology 2003, 17, 548–555. [Google Scholar] [CrossRef]
- Glaser, A.W.; Abdul Rashid, N.F.; U, C.L.; Walker, D.A. School behaviour and health status after central nervous system tumours in childhood. Br. J. Cancer 1997, 76, 643–650. [Google Scholar] [CrossRef]
- Vinchon, M.; Leblond, P.; Caron, S.; Delestret, I.; Baroncini, M.; Coche, B. Radiation-induced tumors in children irradiated for brain tumor: A longitudinal study. Child’s Nerv. Syst. ChNs Off. J. Int. Soc. Pediatric Neurosurg. 2011, 27, 445–453. [Google Scholar] [CrossRef]
- Dvorak-Carbone, H.; Schuman, E.M. Long-term depression of temporoammonic-CA1 hippocampal synaptic transmission. J. Neurophysiol. 1999, 81, 1036–1044. [Google Scholar] [CrossRef]
- Remondes, M.; Schuman, E.M. Role for a cortical input to hippocampal area CA1 in the consolidation of a long-term memory. Nature 2004, 431, 699–703. [Google Scholar] [CrossRef]
- Frankland, P.W.; Bontempi, B. The organization of recent and remote memories. Nat. Rev. Neurosci. 2005, 6, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Alaghband, Y.; Kwapis, J.L.; Lopez, A.J.; White, A.O.; Aimiuwu, O.V.; Al-Kachak, A.; Bodinayake, K.K.; Oparaugo, N.C.; Dang, R.; Astarabadi, M.; et al. Distinct roles for the deacetylase domain of HDAC3 in the hippocampus and medial prefrontal cortex in the formation and extinction of memory. Neurobiol. Learn. Mem. 2017, 145, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Balderas, I.; Rodriguez-Ortiz, C.J.; Salgado-Tonda, P.; Chavez-Hurtado, J.; McGaugh, J.L.; Bermudez-Rattoni, F. The consolidation of object and context recognition memory involve different regions of the temporal lobe. Learn Mem. 2008, 15, 618–624. [Google Scholar] [CrossRef]
- Roozendaal, B.; Hernandez, A.; Cabrera, S.M.; Hagewoud, R.; Malvaez, M.; Stefanko, D.P.; Haettig, J.; Wood, M.A. Membrane-associated glucocorticoid activity is necessary for modulation of long-term memory via chromatin modification. J. Neurosci. 2010, 30, 5037–5046. [Google Scholar] [CrossRef] [PubMed]
- Spalding, K.L.; Bergmann, O.; Alkass, K.; Bernard, S.; Salehpour, M.; Huttner, H.B.; Bostrom, E.; Westerlund, I.; Vial, C.; Buchholz, B.A.; et al. Dynamics of hippocampal neurogenesis in adult humans. Cell 2013, 153, 1219–1227. [Google Scholar] [CrossRef]
- Sorrells, S.F.; Paredes, M.F.; Cebrian-Silla, A.; Sandoval, K.; Qi, D.; Kelley, K.W.; James, D.; Mayer, S.; Chang, J.; Auguste, K.I.; et al. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature 2018, 555, 377–381. [Google Scholar] [CrossRef]
- Duffner, P.K.; Cohen, M.E.; Voorhess, M.L.; MacGillivray, M.H.; Brecher, M.L.; Panahon, A.; Gilani, B.B. Long-term effects of cranial irradiation on endocrine function in children with brain tumors. A prospective study. Cancer 1985, 56, 2189–2193. [Google Scholar] [CrossRef]
- Shalet, S.M. Disorders of the endocrine system due to radiation and cytotoxic chemotherapy. Clin. Endocrinol. 1983, 19, 637–659. [Google Scholar] [CrossRef]
- Shalet, S.M.; Beardwell, C.G.; Pearson, D.; Jones, P.H. The effect of varying doses of cerebral irradiation on growth hormone production in childhood. Clin. Endocrinol. 1976, 5, 287–290. [Google Scholar] [CrossRef]
- Dickinson, W.P.; Berry, D.H.; Dickinson, L.; Irvin, M.; Schedewie, H.; Fiser, R.H.; Elders, M.J. Differential effects of cranial radiation on growth hormone response to arginine and insulin infusion. J. Pediatr. 1978, 92, 754–757. [Google Scholar] [CrossRef]
- Wara, W.M.; Richards, G.E.; Grumbach, M.M.; Kaplan, S.L.; Sheline, G.E.; Conte, F.A. Hypopituitarism after irradiation in children. Int. J. Radiat. Oncol. Biol. Phys. 1977, 2, 549–552. [Google Scholar] [CrossRef]
- Jaccard, M.; Duran, M.T.; Petersson, K.; Germond, J.F.; Liger, P.; Vozenin, M.C.; Bourhis, J.; Bochud, F.; Bailat, C. High dose-per-pulse electron beam dosimetry: Commissioning of the Oriatron eRT6 prototype linear accelerator for preclinical use. Med. Phys. 2018, 45, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Jaccard, M.; Petersson, K.; Buchillier, T.; Germond, J.F.; Duran, M.T.; Vozenin, M.C.; Bourhis, J.; Bochud, F.O.; Bailat, C. High dose-per-pulse electron beam dosimetry: Usability and dose-rate independence of EBT3 Gafchromic films. Med. Phys. 2017, 44, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Petersson, K.; Jaccard, M.; Germond, J.F.; Buchillier, T.; Bochud, F.; Bourhis, J.; Vozenin, M.C.; Bailat, C. High dose-per-pulse electron beam dosimetry—A model to correct for the ion recombination in the Advanced Markus ionization chamber. Med. Phys. 2017, 44, 1157–1167. [Google Scholar] [CrossRef]
- Jorge, P.G.; Jaccard, M.; Petersson, K.; Gondre, M.; Duran, M.T.; Desorgher, L.; Germond, J.F.; Liger, P.; Vozenin, M.C.; Bourhis, J.; et al. Dosimetric and preparation procedures for irradiating biological models with pulsed electron beam at ultra-high dose-rate. Radiother. Oncol. 2019, 139, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Barker, G.R.; Bird, F.; Alexander, V.; Warburton, E.C. Recognition memory for objects, place, and temporal order: A disconnection analysis of the role of the medial prefrontal cortex and perirhinal cortex. J. Neurosci. 2007, 27, 2948–2957. [Google Scholar] [CrossRef] [PubMed]
- Barker, G.R.; Warburton, E.C. When is the hippocampus involved in recognition memory? J. Neurosci. 2011, 31, 10721–10731. [Google Scholar] [CrossRef]
- Petit-Demouliere, B.; Chenu, F.; Bourin, M. Forced swimming test in mice: A review of antidepressant activity. Psychopharmacology 2005, 177, 245–255. [Google Scholar] [CrossRef]
- Parihar, V.K.; Maroso, M.; Syage, A.; Allen, B.D.; Angulo, M.C.; Soltesz, I.; Limoli, C.L. Persistent nature of alterations in cognition and neuronal circuit excitability after exposure to simulated cosmic radiation in mice. Exp. Neurol. 2018, 305, 44–55. [Google Scholar] [CrossRef]
- Cain, C.K.; Blouin, A.M.; Barad, M. Temporally massed CS presentations generate more fear extinction than spaced presentations. J. Exp. Psychol. Anim. Behav. Process. 2003, 29, 323–333. [Google Scholar] [CrossRef]
- Baulch, J.E.; Acharya, M.M.; Allen, B.D.; Ru, N.; Chmielewski, N.N.; Martirosian, V.; Giedzinski, E.; Syage, A.; Park, A.L.; Benke, S.N.; et al. Cranial grafting of stem cell-derived microvesicles improves cognition and reduces neuropathology in the irradiated brain. Proc. Natl. Acad. Sci. USA 2016, 113, 4836–4841. [Google Scholar] [CrossRef] [PubMed]
- Christie, L.A.; Acharya, M.M.; Parihar, V.K.; Nguyen, A.; Martirosian, V.; Limoli, C.L. Impaired cognitive function and hippocampal neurogenesis following cancer chemotherapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 1954–1965. [Google Scholar] [CrossRef] [PubMed]
- Bourhis, J.; Sozzi, W.J.; Jorge, P.G.; Gaide, O.; Bailat, C.; Duclos, F.; Patin, D.; Ozsahin, M.; Bochud, F.; Germond, J.F.; et al. Treatment of a first patient with FLASH-radiotherapy. Radiother. Oncol. 2019, 139, 18–22. [Google Scholar] [CrossRef] [PubMed]
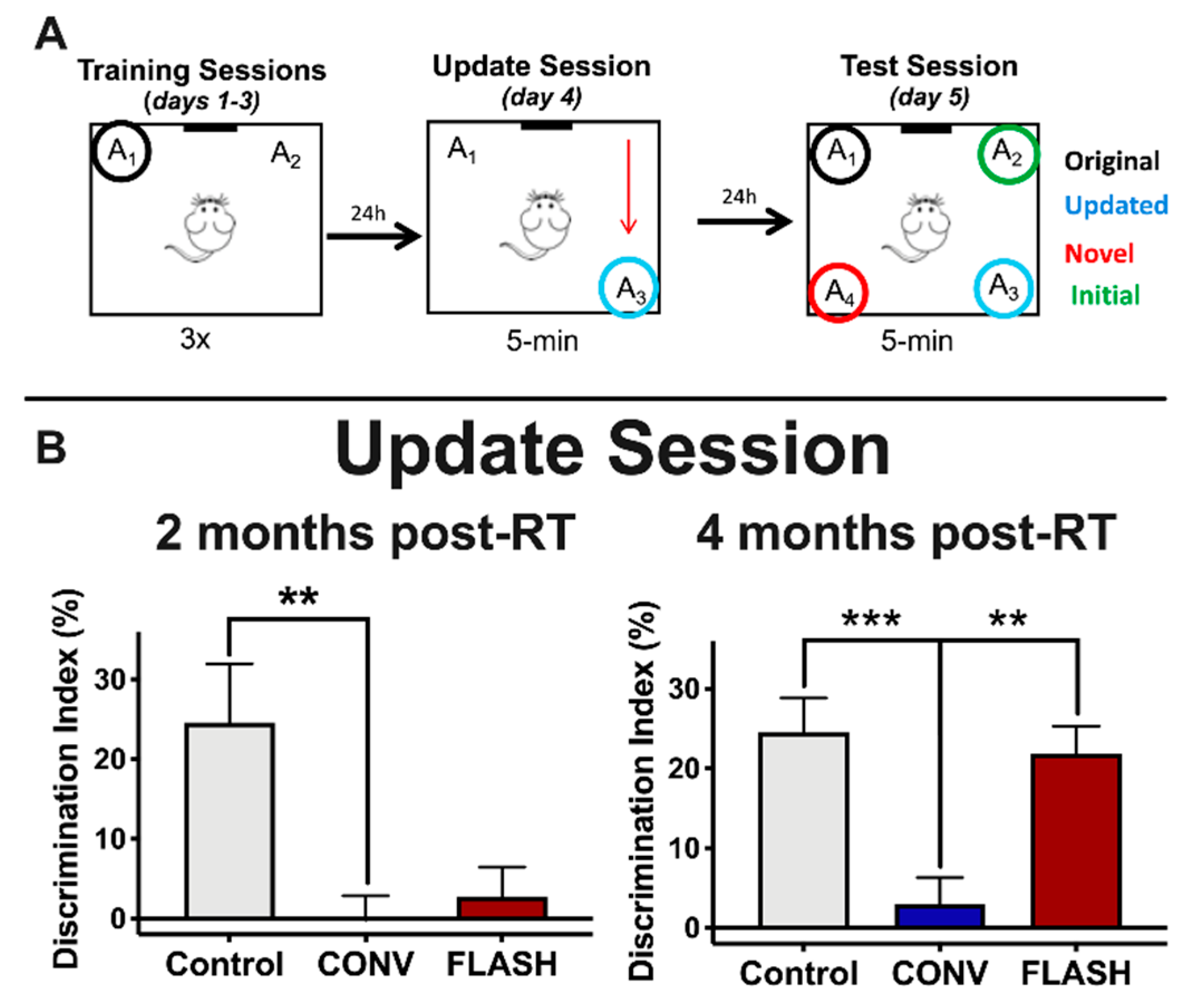
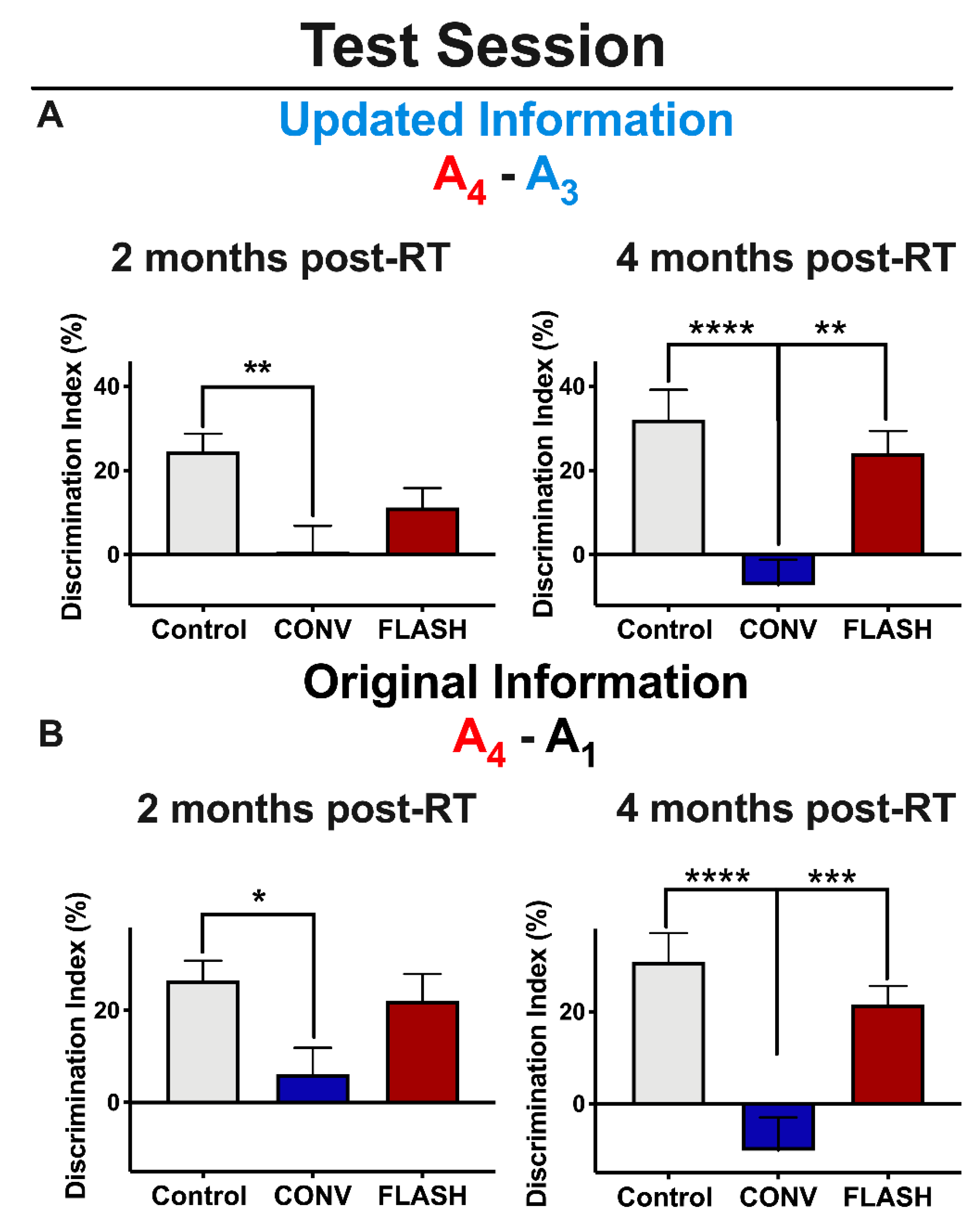
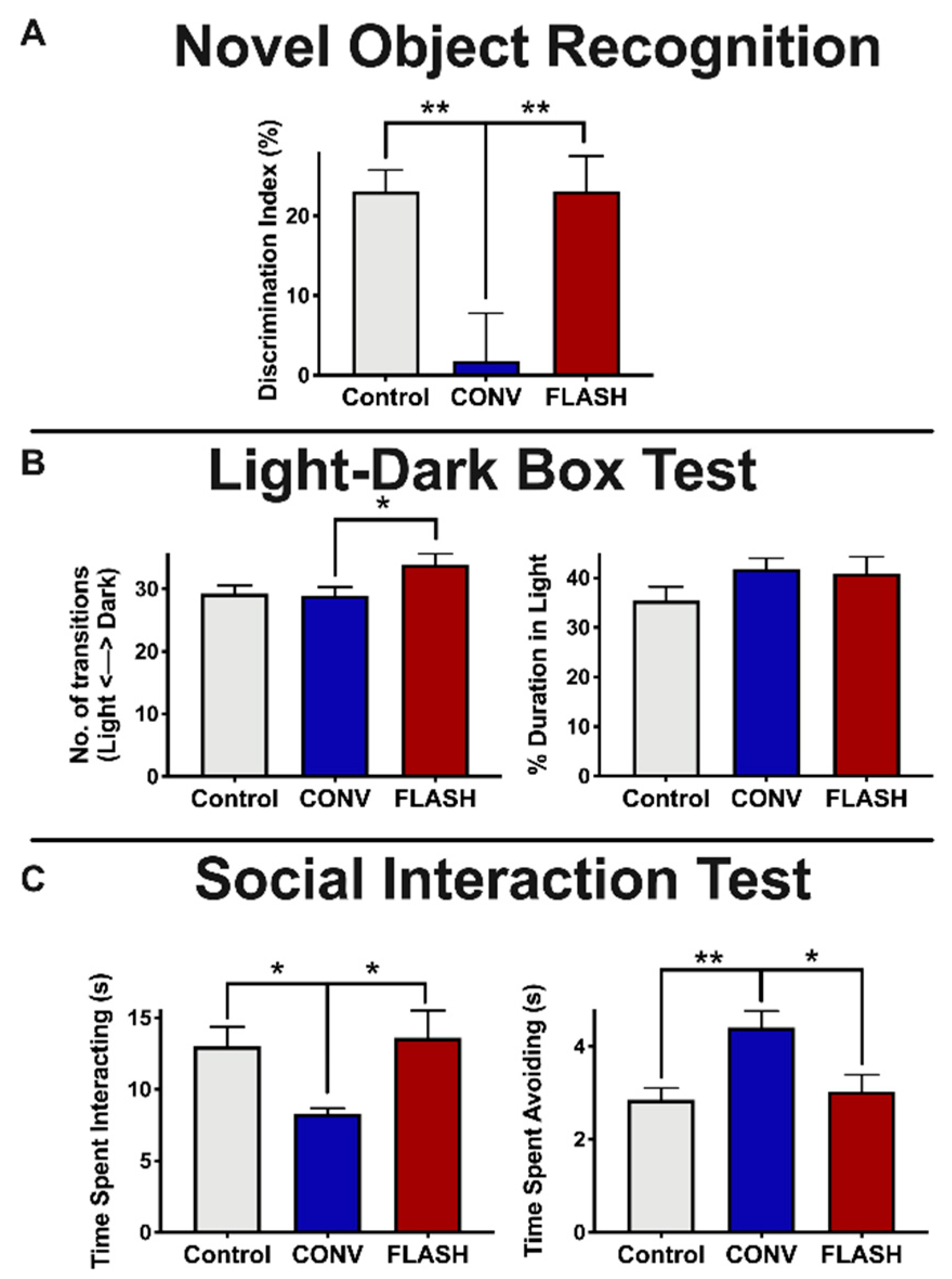


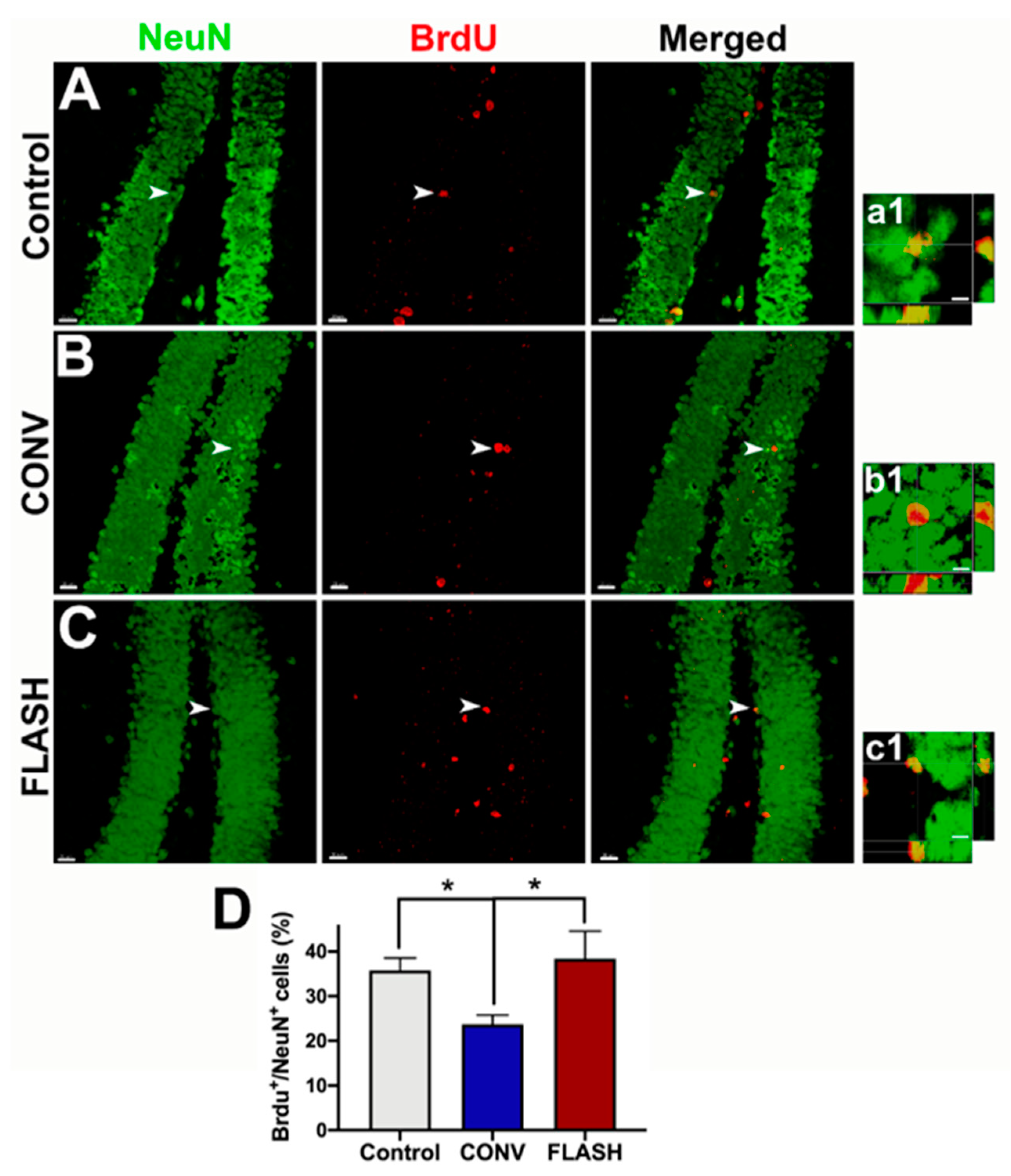
| Delivery Mode | Prescribed Dose (Gy) | Graphite Applicator Type and Size (mm) | Beam Parameters | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Source-to-Surface Distance (mm) | Pulse Repetition Frequency (Hz) | Pulse Width (µs) | Number of Pulses | Treatment Time (s) | Mean Dose Rate (Gy/s) | InstantanEous Dose Rate (Gy/s) | |||
| CONV | 8 | Semicircular ∅17 | 798 | 10 | 1.0 | 1033 | 103.2 | 0.077 | 7.7 × 103 |
| FLASH | 8 | Semicircular ∅17 | 383 | 100 | 1.8 | 1 | 1.8 × 10−6 | 4.4 × 106 | 4.4 × 106 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alaghband, Y.; Cheeks, S.N.; Allen, B.D.; Montay-Gruel, P.; Doan, N.-L.; Petit, B.; Jorge, P.G.; Giedzinski, E.; Acharya, M.M.; Vozenin, M.-C.; et al. Neuroprotection of Radiosensitive Juvenile Mice by Ultra-High Dose Rate FLASH Irradiation. Cancers 2020, 12, 1671. https://doi.org/10.3390/cancers12061671
Alaghband Y, Cheeks SN, Allen BD, Montay-Gruel P, Doan N-L, Petit B, Jorge PG, Giedzinski E, Acharya MM, Vozenin M-C, et al. Neuroprotection of Radiosensitive Juvenile Mice by Ultra-High Dose Rate FLASH Irradiation. Cancers. 2020; 12(6):1671. https://doi.org/10.3390/cancers12061671
Chicago/Turabian StyleAlaghband, Yasaman, Samantha N. Cheeks, Barrett D. Allen, Pierre Montay-Gruel, Ngoc-Lien Doan, Benoit Petit, Patrik Goncalves Jorge, Erich Giedzinski, Munjal M. Acharya, Marie-Catherine Vozenin, and et al. 2020. "Neuroprotection of Radiosensitive Juvenile Mice by Ultra-High Dose Rate FLASH Irradiation" Cancers 12, no. 6: 1671. https://doi.org/10.3390/cancers12061671
APA StyleAlaghband, Y., Cheeks, S. N., Allen, B. D., Montay-Gruel, P., Doan, N.-L., Petit, B., Jorge, P. G., Giedzinski, E., Acharya, M. M., Vozenin, M.-C., & Limoli, C. L. (2020). Neuroprotection of Radiosensitive Juvenile Mice by Ultra-High Dose Rate FLASH Irradiation. Cancers, 12(6), 1671. https://doi.org/10.3390/cancers12061671






