Circulating Plasma Gelsolin: A Predictor of Favorable Clinical Outcomes in Head and Neck Cancer and Sensitive Biomarker for Early Disease Diagnosis Combined with Soluble Fas Ligand
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
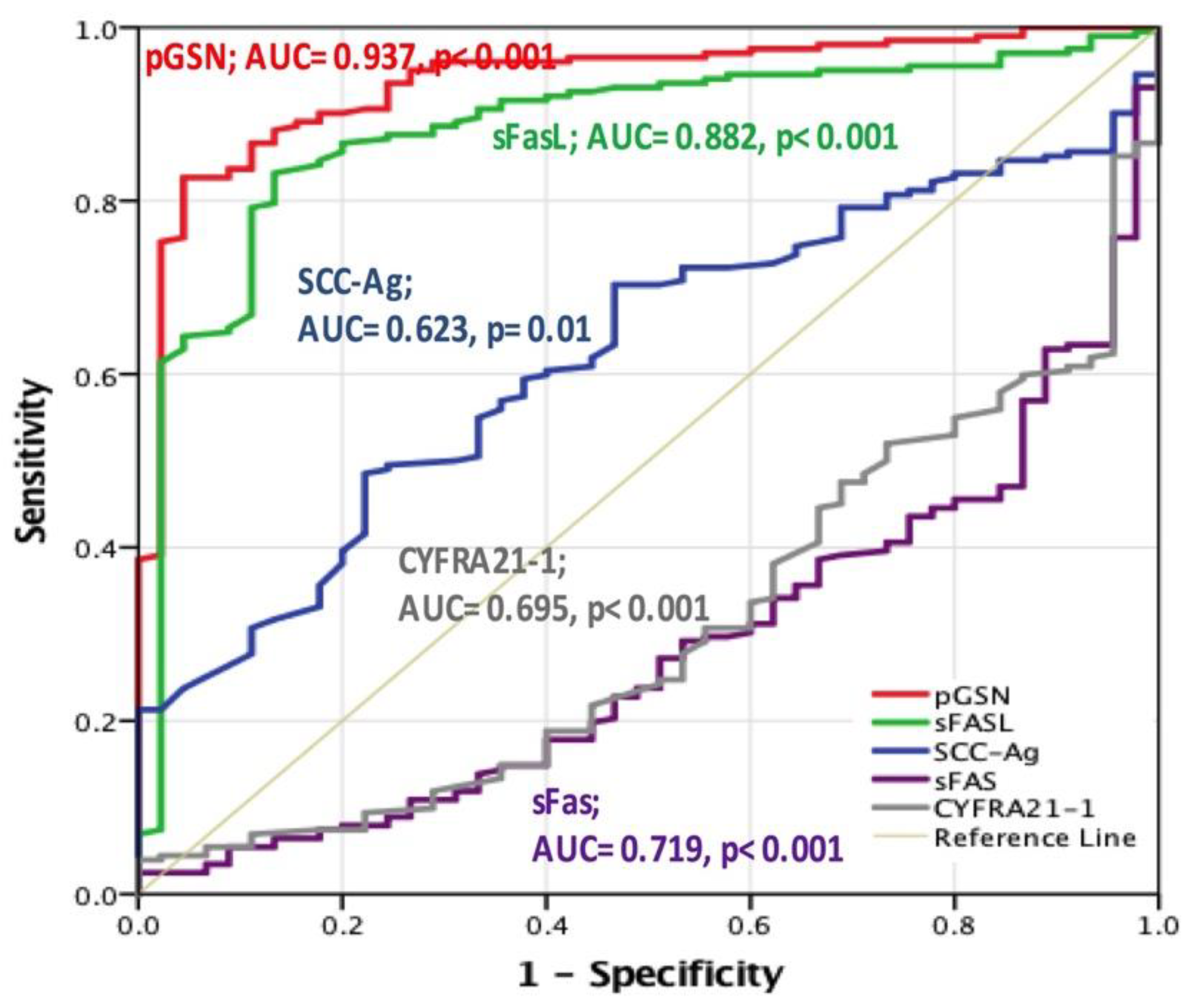
2.2. Diagnostic Value of Circulating pGSN in Patients with HNC
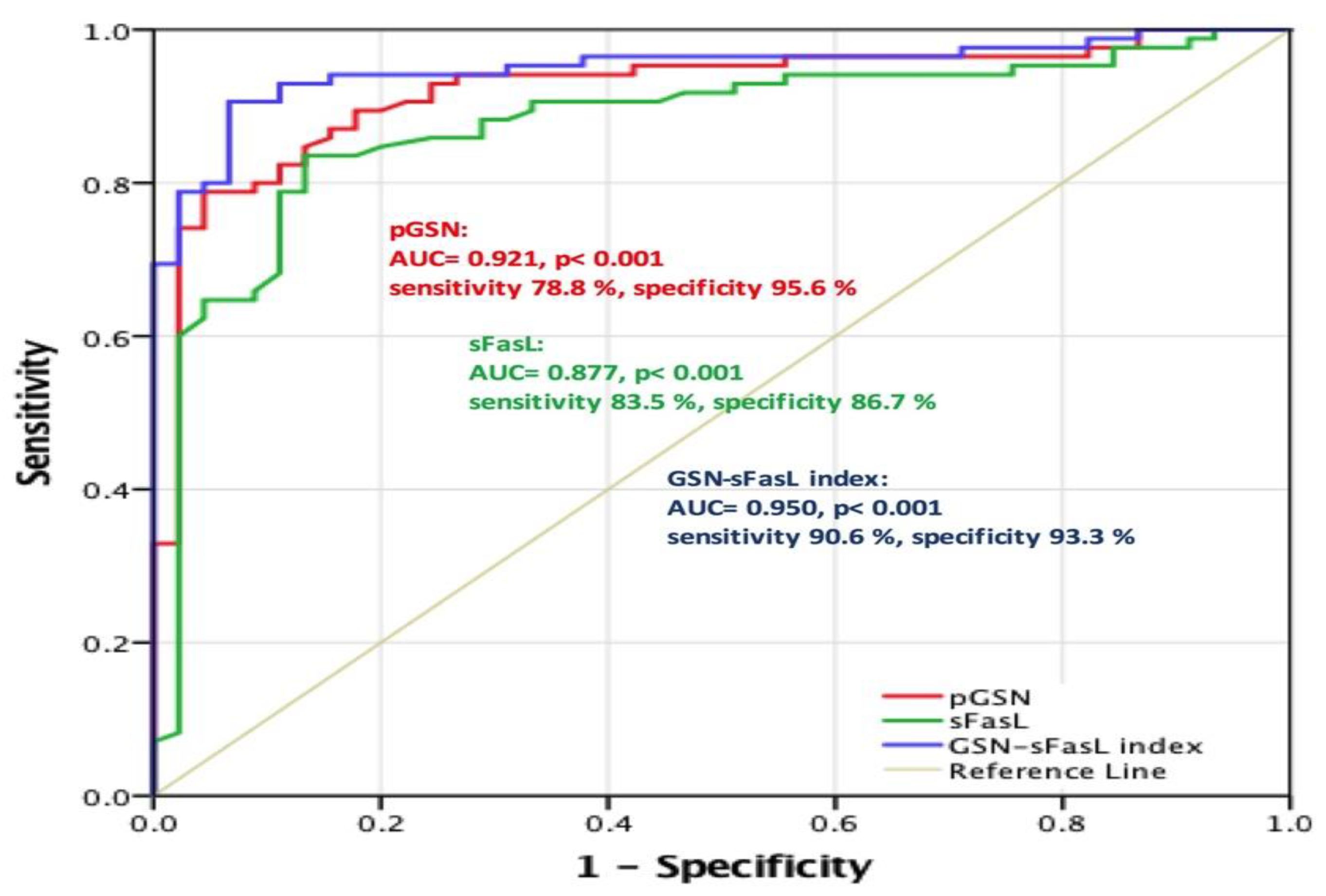
2.3. pGSN as a Valuable Diagnostic Tool for Early HNC Stage
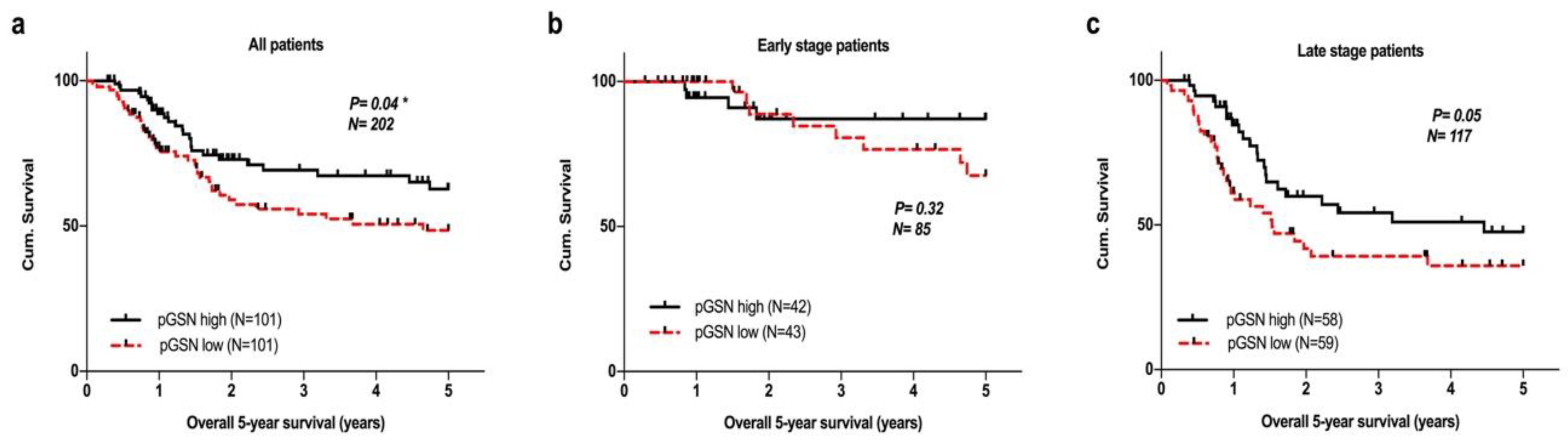
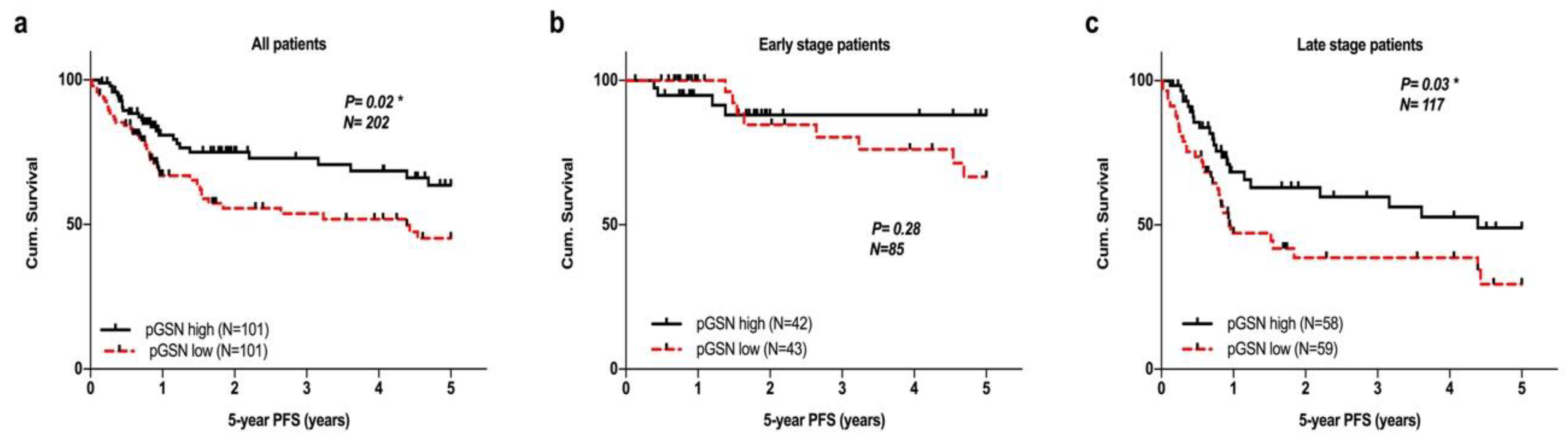
2.4. Survival Analysis of Circulating pGSN in Validation Datasets
2.5. Prognostic Impact of Circulating pGSN and Its Relationship with Other Clinicopathological Parameters
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Clinicopathological Characteristics and Collection of Plasma Samples
4.3. Circulating pGSN and SCC-Ag Detection from Patient Plasma Using Sandwich ELISA
4.4. Circulating Biomarker Detection
4.5. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.J.; Lo, W.C.; Yang, Y.W.; You, S.L.; Chen, C.J.; Lai, M.S. Incidence and survival of adult cancer patients in Taiwan, 2002–2012. J. Formos. Med. Assoc. 2016, 115, 1076–1088. [Google Scholar] [CrossRef] [PubMed]
- Paez, J.G. EGFR Mutations in Lung Cancer: Correlation with Clinical Response to Gefitinib Therapy. Science 2004, 304, 1497–1500. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.C.; Wu, Y.L.; Chen, G.Y.; Feng, J.F.; Liu, X.Q.; Wang, C.L.; Zhang, S.C.; Wang, J.; Zhou, S.W.; Ren, S.X.; et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): A multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011, 12, 735–742. [Google Scholar] [CrossRef]
- Rosas, S.L.; Koch, W.; da Costa Carvalho, M.G.; Wu, L.; Califano, J.; Westra, W.; Jen, J.; Sidransky, D. Promoter hypermethylation patterns of p16, O6-methylguanine-DNA-methyltransferase, and death-associated protein kinase in tumors and saliva of head and neck cancer patients. Cancer Res. 2001, 61, 939–942. [Google Scholar] [PubMed]
- Ranuncolo, S.M.; Puricelli, L.I.; Joffe, E.B.D.; Roffo, A.H.; Matos, E.; Loria, D.; Vilensky, M. Circulating 92-kilodalton matrix metalloproteinase (MMP-9) activity is enhanced in the euglobulin plasma fraction of head and neck squamous cell carcinoma—Reply. Cancer 2003, 97, 203–204. [Google Scholar] [CrossRef]
- Ferris, R.L.; Martinez, I.; Sirianni, N.; Wang, J.; Lopez-Albaitero, A.; Gollin, S.M.; Johnson, J.T.; Khan, S. Human papillomavirus-16 associated squamous cell carcinoma of the head and neck (SCCHN): A natural disease model provides insights into viral carcinogenesis. Eur. J. Cancer 2005, 41, 807–815. [Google Scholar] [CrossRef]
- Margolis, M.L.; Hyzy, J.B.; Schenken, L.L.; Schepart, B.S. Serum tumor markers in non-small cell lung cancer. A comparative analysis. Cancer 1994, 73, 605–609. [Google Scholar] [CrossRef]
- Kato, H.; Tamai, K.; Morioka, H.; Nagai, M.; Nagaya, T.; Torigoe, T. Tumor-antigen TA-4 in the detection of recurrence in cervical squamous cell carcinoma. Cancer 1984, 54, 1544–1546. [Google Scholar] [CrossRef]
- Chen, I.H.; Liao, C.T.; Wang, H.M.; Huang, J.J.; Kang, C.J.; Huang, S.F. Using SCC Antigen and CRP Levels as Prognostic Biomarkers in Recurrent Oral Cavity Squamous Cell Carcinoma. PLoS ONE 2014, 9, e103265. [Google Scholar] [CrossRef]
- Yuan, C.; Yang, K.; Tang, H.; Chen, D. Diagnostic values of serum tumor markers Cyfra21-1, SCCAg, ferritin, CEA, CA19-9, and AFP in oral/oropharyngeal squamous cell carcinoma. Oncol. Targets Ther. 2016, 9, 3381–3386. [Google Scholar]
- Reed, J.C. Dysregulation of apoptosis in cancer. J. Clin. Oncol. 1999, 17, 2941–2953. [Google Scholar] [CrossRef]
- Ashkenazi, A.; Dixit, V.M. Death receptors: Signaling and modulation. Science 1998, 281, 1305–1308. [Google Scholar] [CrossRef] [PubMed]
- Gastman, B.R.; Atarshi, Y.; Reichert, T.E.; Saito, T.; Balkir, L.; Rabinowich, H.; Whiteside, T.L. Fas ligand is expressed on human squamous cell carcinomas of the head and neck, and it promotes apoptosis of T lymphocytes. Cancer Res. 1999, 59, 5356–5364. [Google Scholar] [PubMed]
- Wang, P.W.; Abedini, M.R.; Yang, L.X.; Ding, A.A.; Figeys, D.; Chang, J.Y.; Tsang, B.K.; Shieh, D.B. Gelsolin regulates cisplatin sensitivity in human head-and-neck cancer. Int. J. Cancer 2014, 135, 2760–2769. [Google Scholar] [CrossRef]
- Abedini, M.R.; Wang, P.W.; Huang, Y.F.; Cao, M.J.; Chou, C.Y.; Shieh, D.B.; Tsang, B.K. Cell fate regulation by gelsolin in human gynecologic cancers. Proc. Natl. Acad. Sci. USA 2014, 111, 17684. [Google Scholar] [CrossRef]
- Kwiatkowski, D.J.; Mehl, R.; Izumo, S.; Nadal-Ginard, B.; Yin, H.L. Muscle Is the Major Source of Plasma Gelsolin. J. Biol. Chem. 1988, 263, 8239–8243. [Google Scholar]
- Ankenbauer, T.; Kleinschmidt, J.A.; Vandekerckhove, J.; Franke, W.W. Proteins Regulating Actin Assembly in Oogenesis and Early Embryogenesis of Xenopus-Laevis-Gelsolin Is the Major Cytoplasmic Actin-Binding Protein. J. Cell Biol. 1988, 107, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Suhler, E.; Lin, W.; Yin, H.L.; Lee, W.M. Decreased plasma gelsolin concentrations in acute liver failure, myocardial infarction, septic shock, and myonecrosis. Crit. Care Med. 1997, 25, 594–598. [Google Scholar] [CrossRef]
- Dahl, B.; Schiødt, F.V.; Ott, P.; Gvozdenovic, R.; Yin, H.L.; Lee, W.M. Plasma gelsolin is reduced in trauma patients. Shock 1999, 12, 102–104. [Google Scholar] [CrossRef]
- Lind, S.E.; Smith, D.B.; Janmey, P.A.; Stossel, T.P. Depression of gelsolin levels and detection of gelsolin-actin complexes in plasma of patients with acute lung injury. Am. Rev. Respir. Dis. 1988, 138, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Gay, F.; Estornes, Y.; Saurin, J.C.; Joly-Pharaboz, M.O.; Friederich, E.; Scoazec, J.Y.; Abello, J. In colon carcinogenesis, the cytoskeletal protein gelsolin is down-regulated during the transition from adenoma to carcinoma. Hum. Pathol. 2008, 39, 1420–1430. [Google Scholar] [CrossRef] [PubMed]
- Noske, A.; Denkert, C.; Schober, H.; Sers, C. Loss of Gelsolin expression in human ovarian carcinomas. Eur. J. Cancer 2005, 41, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Chiou, S.H.; Yang, C.L.; Chow, K.C.; Lin, T.Y.; Chang, H.W.; You, W.C.; Huang, H.W.; Chen, C.M.; Chen, N.C.; et al. Secreted gelsolin desensitizes and induces apoptosis of infiltrated lymphocytes in prostate cancer. Oncotarget 2017, 8, 77152–77167. [Google Scholar] [CrossRef]
- Peter, M.E.; Hadji, A.; Murmann, A.E.; Brockway, S.; Putzbach, W.; Pattanayak, A.; Ceppi, P. The role of CD95 and CD95 ligand in cancer. Cell Death Differ. 2015, 22, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Carlomagno, N.; Incollingo, P.; Tammaro, V.; Peluso, G.; Rupealta, N.; Chiacchio, G.; Sandoval Sotelo, M.L.; Minieri, G.; Pisani, A.; Riccio, E.; et al. Diagnostic, Predictive, Prognostic, and Therapeutic Molecular Biomarkers in Third Millennium: A Breakthrough in Gastric Cancer. BioMed Res. Int. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
- Imai, R.; Takenaka, Y.; Yasui, T.; Nakahara, S.; Yamamoto, Y.; Hanamoto, A.; Takemoto, N.; Fukusumi, T.; Cho, H.; Yamamoto, M.; et al. Prognostic significance of serum squamous cell carcinoma antigen in patients with head and neck cancer. Acta Otolaryngol. 2015, 135, 295–301. [Google Scholar] [CrossRef]
- Barak, V.; Meirovitz, A.; Leibovici, V.; Rachmut, J.; Peretz, T.; Eliashar, R.; Gross, M. The Diagnostic and Prognostic Value of Tumor Markers (CEA, SCC, CYFRA 21-1, TPS) in Head and Neck Cancer Patients. Anticancer Res. 2015, 35, 5519–5524. [Google Scholar]
- Marino, N.; Marshall, J.C.; Collins, J.W.; Zhou, M.; Qian, Y.; Veenstra, T.; Steeg, P.S. Nm23-h1 binds to gelsolin and inactivates its actin-severing capacity to promote tumor cell motility and metastasis. Cancer Res. 2013, 73, 5949–5962. [Google Scholar] [CrossRef]
- Matarrese, P.; Abbruzzese, C.; Mileo, A.M.; Vona, R.; Ascione, B.; Visca, P.; Rollo, F.; Benevolo, M.; Malorni, W.; Paggi, M.G. Interaction between the human papillomavirus 16 E7 oncoprotein and gelsolin ignites cancer cell motility and invasiveness. Oncotarget 2016, 7, 50972. [Google Scholar] [CrossRef][Green Version]
- Kothakota, S.; Azuma, T.; Reinhard, C.; Klippel, A.; Tang, J.; Chu, K.; McGarry, T.J.; Kirschner, M.W.; Koths, K.; Kwiatkowski, D.J.; et al. Caspase-3-generated fragment of gelsolin: Effector of morphological change in apoptosis. Science 1997, 278, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Shieh, D.-B.; Godleski, J.; Herndon, J.E.; Azuma, T.; Mercer, H.; Sugarbaker, D.J.; Kwiatkowski, D.J. Cell motility as a prognostic factor in Stage I nonsmall cell lung carcinoma. Cancer 1999, 85, 47–57. [Google Scholar] [CrossRef]
- Shieh, D.B.; Chen, I.W.; Wei, T.Y.; Shao, C.Y.; Chang, H.J.; Chung, C.H.; Wong, T.Y.; Jin, Y.T. Tissue expression of gelsolin in oral carcinogenesis progression and its clinicopathological implications. Oral Oncol. 2006, 42, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Winston, J.S.; Asch, H.L.; Zhang, P.J.; Edge, S.B.; Hyland, A.; Asch, B.B. Downregulation of gelsolin correlates with the progression to breast carcinoma. Breast Cancer Res. Treat. 2001, 65, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Vasconcellos, C.A.; Lind, S.E. Coordinated inhibition of actin-induced platelet aggregation by plasma gelsolin and vitamin D-binding protein. Blood 1993, 82, 3648–3657. [Google Scholar] [CrossRef] [PubMed]
- Epstein, F.H.; Lee, W.M.; Galbraith, R.M. The Extracellular Actin-Scavenger System and Actin Toxicity. N. Engl. J. Med. 1992, 326, 1335–1341. [Google Scholar] [CrossRef]
- Horváth-Szalai, Z.; Kustán, P.; Mühl, D.; Ludány, A.; Bugyi, B.; Kőszegi, T. Antagonistic sepsis markers: Serum gelsolin and actin/gelsolin ratio. Clin. Biochem. 2017, 50, 127–133. [Google Scholar] [CrossRef]
- Asare-Werehene, M.; Nakka, K.; Reunov, A.; Chiu, C.T.; Lee, W.T.; Abedini, M.R.; Wang, P.W.; Shieh, D.B.; Dilworth, F.J.; Carmona, E.; et al. The exosome-mediated autocrine and paracrine actions of plasma gelsolin in ovarian cancer chemoresistance. Oncogene 2020, 39, 1600–1616. [Google Scholar] [CrossRef]
- Asare-Werehene, M.; Communal, L.; Carmona, E.; Le, T.; Provencher, D.; Mes-Masson, A.M.; Tsang, B.K. Pre-operative Circulating Plasma Gelsolin Predicts Residual Disease and Detects Early Stage Ovarian Cancer. Sci. Rep. 2019, 9, 13924. [Google Scholar] [CrossRef]
- Chen, Z.; Li, K.; Yin, X.; Li, H.; Li, Y.; Zhang, Q.; Wang, H.; Qiu, Y. Lower Expression of Gelsolin in Colon Cancer and Its Diagnostic Value in Colon Cancer Patients. J. Cancer 2019, 10, 1288–1296. [Google Scholar] [CrossRef]
- Hoffmann, T.K.; Dworacki, G.; Tsukihiro, T.; Meidenbauer, N.; Gooding, W.; Johnson, J.T.; Whiteside, T.L. Spontaneous apoptosis of circulating T lymphocytes in patients with head and neck cancer and its clinical importance. Clin. Cancer Res. 2002, 8, 2553–2562. [Google Scholar] [PubMed]
- Yu, J.S.; Chen, Y.T.; Chiang, W.F.; Hsiao, Y.C.; Chu, L.J.; See, L.C.; Wu, C.S.; Tu, H.T.; Chen, H.W.; Chen, C.C.; et al. Saliva protein biomarkers to detect oral squamous cell carcinoma in a high-risk population in Taiwan. Proc. Natl. Acad. Sci. USA 2016, 113, 11549–11554. [Google Scholar] [CrossRef] [PubMed]
- Igney, F.H.; Krammer, P.H. Tumor counterattack: Fact or fiction? Cancer Immunol. Immunother. 2005, 54, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Boroumand-Noughabi, S.; Sima, H.R.; Ghaffarzadehgan, K.; Jafarzadeh, M.; Raziee, H.R.; Hosseinnezhad, H.; Moaven, O.; Rajabi-Mashhadi, M.T.; Azarian, A.A.; Mashhadinejad, M.; et al. Soluble Fas might serve as a diagnostic tool for gastric adenocarcinoma. BMC Cancer 2010, 10, 275. [Google Scholar] [CrossRef]
- Bermejo-Pareja, F.; Antequera, D.; Vargas, T.; Molina, J.A.; Carro, E. Saliva levels of Abeta1-42 as potential biomarker of Alzheimer’s disease: A pilot study. BMC Neurol. 2010, 10, 108. [Google Scholar] [CrossRef]
- Bonomi, M.; Patsias, A.; Posner, M.; Sikora, A. The role of inflammation in head and neck cancer. Adv. Exp. Med. Biol. 2014, 816, 107–127. [Google Scholar]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Pils, D.; Tong, D.; Hager, G.; Obermayr, E.; Aust, S.; Heinze, G.; Kohl, M.; Schuster, E.; Wolf, A.; Sehouli, J.; et al. A combined blood based gene expression and plasma protein abundance signature for diagnosis of epithelial ovarian cancer—A study of the OVCAD consortium. BMC Cancer 2013, 13, 178. [Google Scholar] [CrossRef]
- Grambsch, P.M.; Therneau, T.M. Proportional Hazards Tests and Diagnostics Based on Weighted Residuals. Biometrika 1994, 81, 515. [Google Scholar] [CrossRef]

| Variables | AUC | Significance | Sensitivity (%) | Specificity (%) |
|---|---|---|---|---|
| All patients | ||||
| pGSN | 0.937 | < 0.001 | 82.7 | 95.6 |
| Early-stage patients | ||||
| pGSN | 0.921 | < 0.001 | 78.8 | 95.6 |
| sFasL | 0.877 | < 0.001 | 83.5 | 86.7 |
| pGSN–sFasL index | 0.950 | < 0.001 | 90.6 | 93.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, C.-T.; Wang, P.-W.; Asare-Werehene, M.; Tsang, B.K.; Shieh, D.-B. Circulating Plasma Gelsolin: A Predictor of Favorable Clinical Outcomes in Head and Neck Cancer and Sensitive Biomarker for Early Disease Diagnosis Combined with Soluble Fas Ligand. Cancers 2020, 12, 1569. https://doi.org/10.3390/cancers12061569
Chiu C-T, Wang P-W, Asare-Werehene M, Tsang BK, Shieh D-B. Circulating Plasma Gelsolin: A Predictor of Favorable Clinical Outcomes in Head and Neck Cancer and Sensitive Biomarker for Early Disease Diagnosis Combined with Soluble Fas Ligand. Cancers. 2020; 12(6):1569. https://doi.org/10.3390/cancers12061569
Chicago/Turabian StyleChiu, Chen-Tzu, Pei-Wen Wang, Meshach Asare-Werehene, Benjamin K. Tsang, and Dar-Bin Shieh. 2020. "Circulating Plasma Gelsolin: A Predictor of Favorable Clinical Outcomes in Head and Neck Cancer and Sensitive Biomarker for Early Disease Diagnosis Combined with Soluble Fas Ligand" Cancers 12, no. 6: 1569. https://doi.org/10.3390/cancers12061569
APA StyleChiu, C.-T., Wang, P.-W., Asare-Werehene, M., Tsang, B. K., & Shieh, D.-B. (2020). Circulating Plasma Gelsolin: A Predictor of Favorable Clinical Outcomes in Head and Neck Cancer and Sensitive Biomarker for Early Disease Diagnosis Combined with Soluble Fas Ligand. Cancers, 12(6), 1569. https://doi.org/10.3390/cancers12061569





