Vinorelbine Augments Radiotherapy in Hepatocellular Carcinoma
Abstract
1. Introduction
2. Results
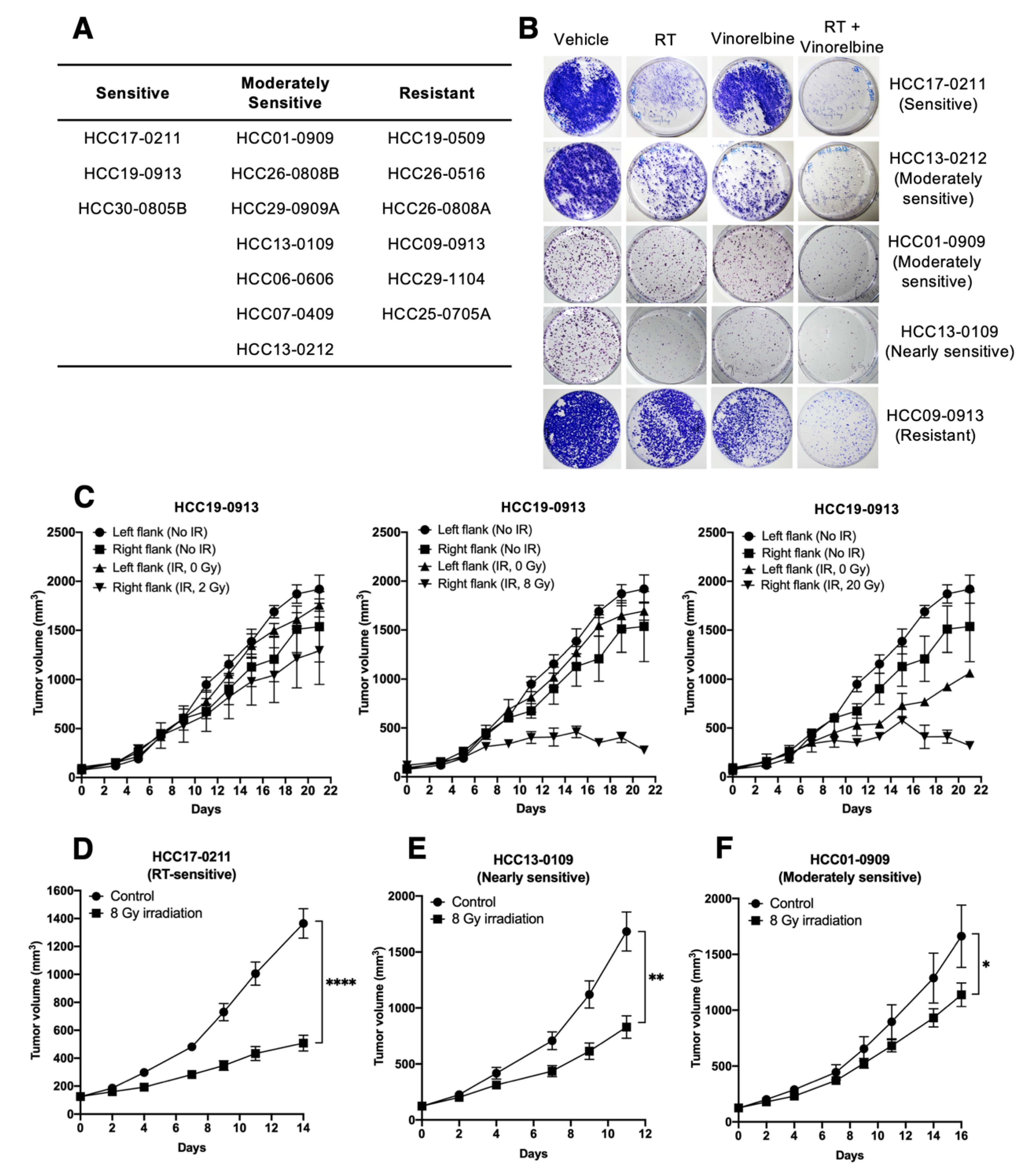
2.1. Screening of Organoid Cultures In Vitro and PDX Models in Mice for Sensitivity to Irradiation
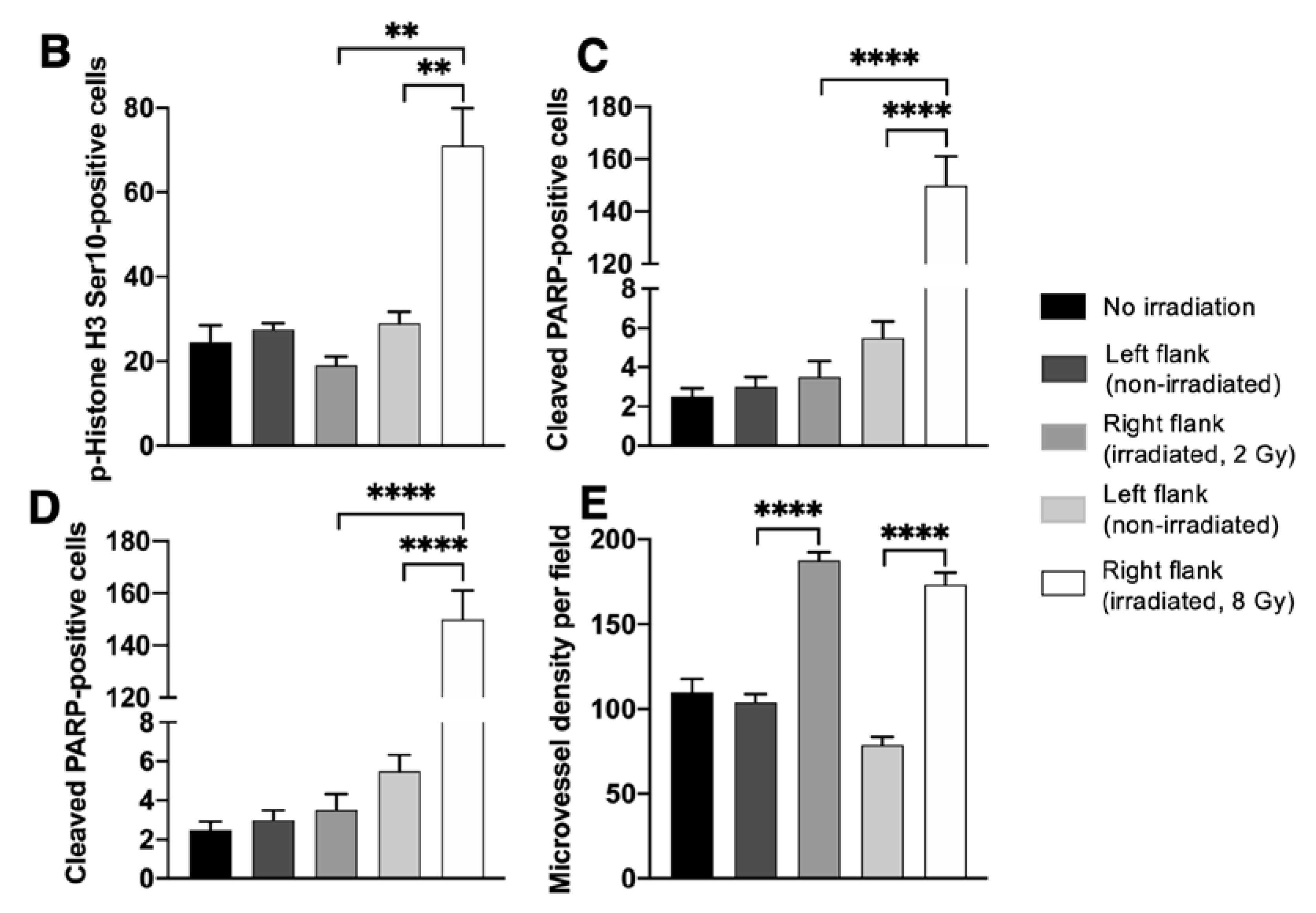
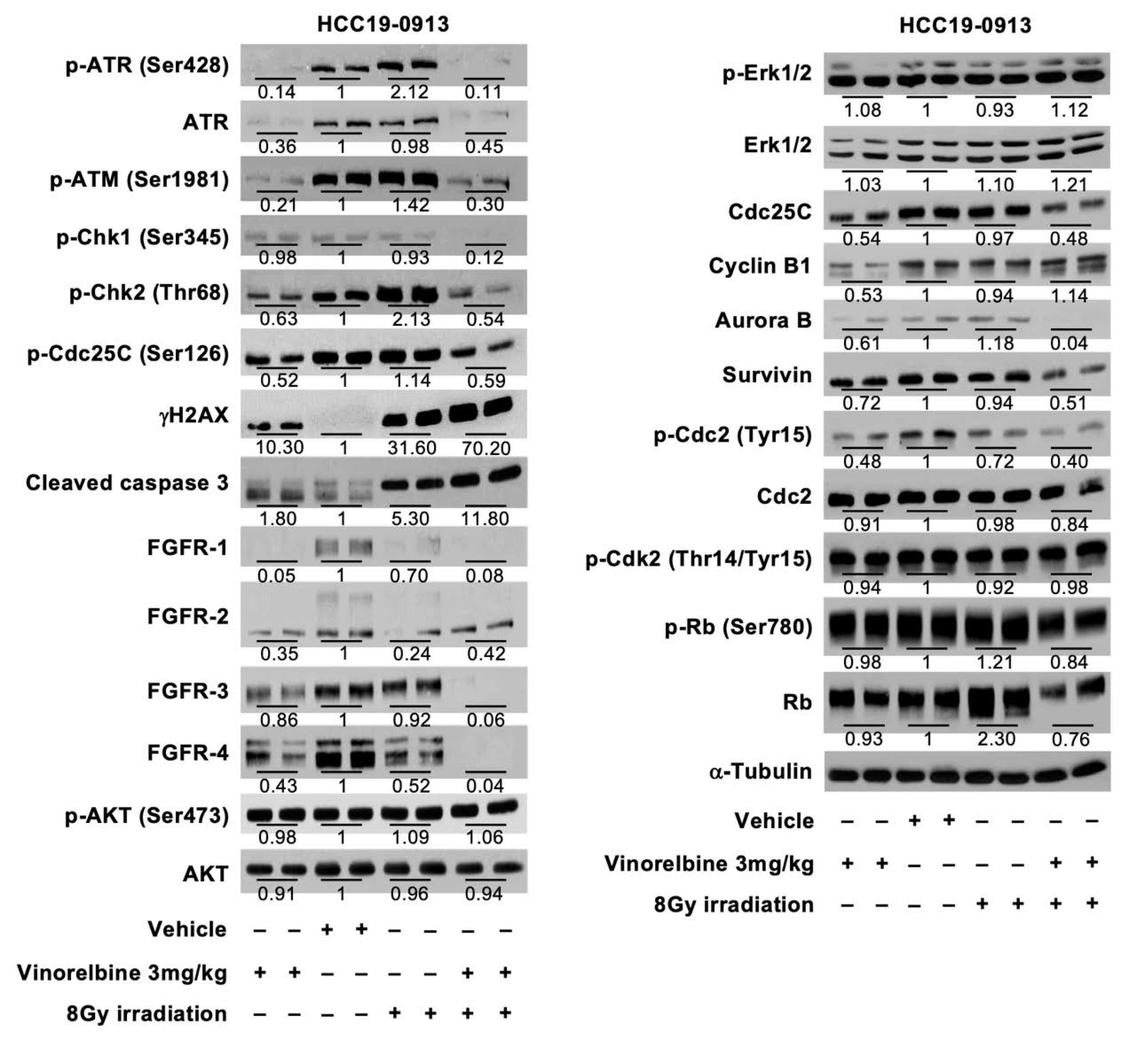
2.2. Effects of Localized Irradiation on Tumor Growth, Angiogenesis, DNA Damage, Apoptosis, and Cell Proliferation
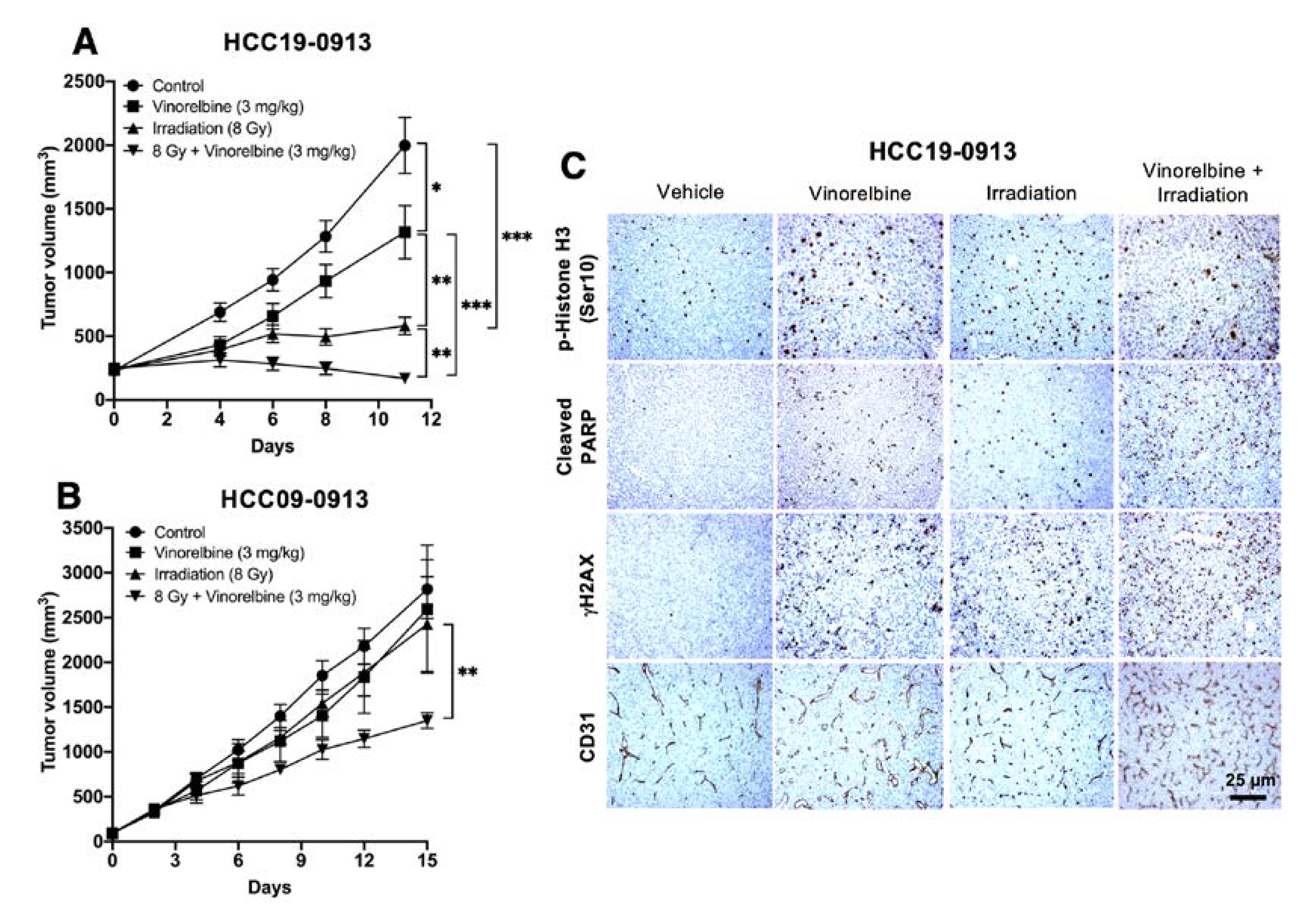
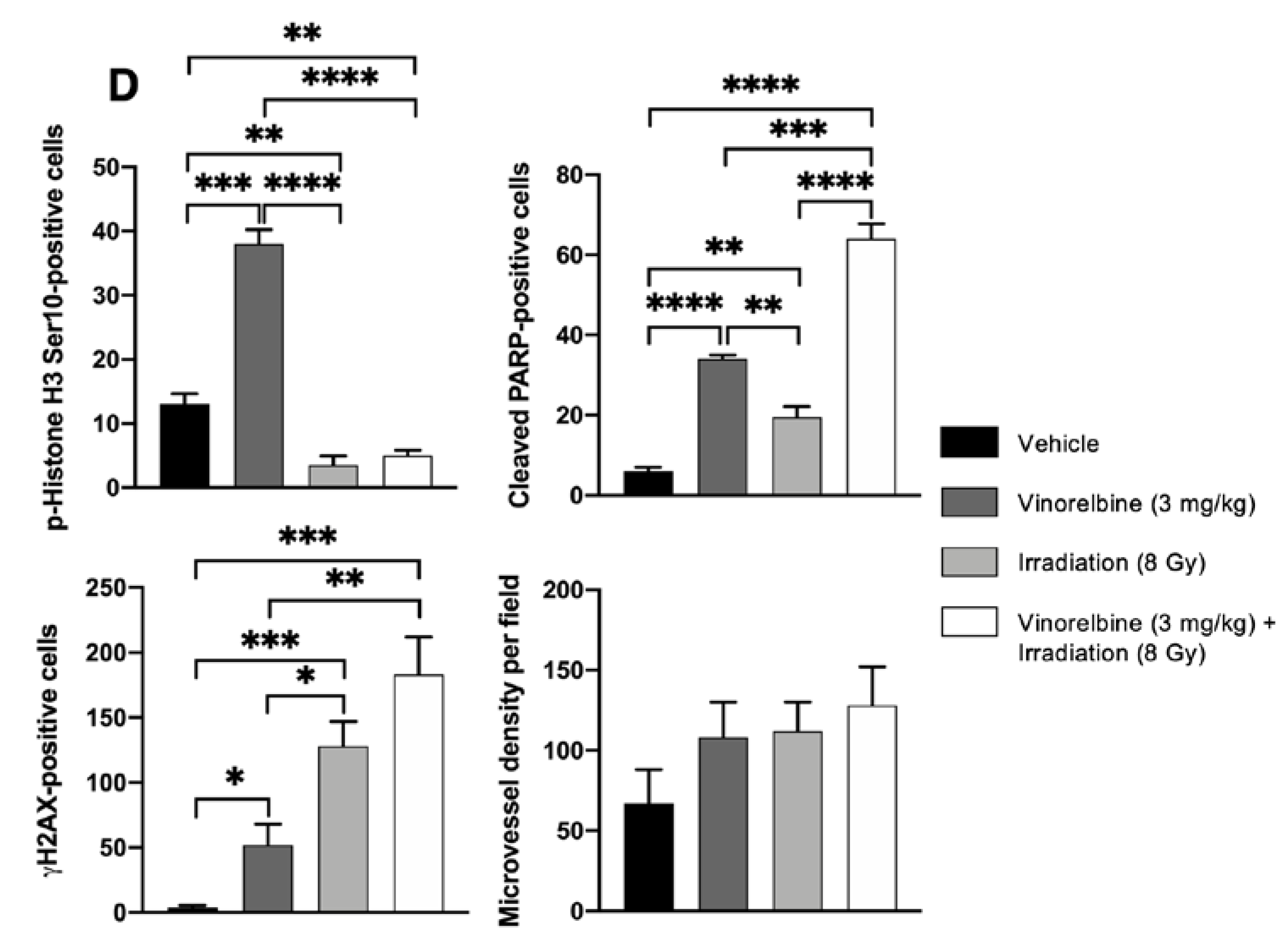
2.3. Vinorelbine Potentiates Antitumor Activity of RT in HCC Models
3. Discussion
4. Materials and Methods
4.1. In Vitro Irradiation
4.2. Establishment of the HCC PDX Model and In Vivo Irradiation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
Appendix A.1. Reagents
Appendix A.2. Primary Cell Culture and Clonogenic Cell Survival Assay
Appendix A.3. Tumor Measurement
Appendix A.4. Flow Cytometry Analysis
Appendix A.5. Western Blot Analysis
Appendix A.6. Immunohistochemistry
Appendix A.7. Statistical Analysis
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, J.; Kosuge, T.; Takayama, T.; Shimada, K.; Yamasaki, S.; Ozaki, H.; Yamaguchi, N.; Makuuchi, M. Recurrence of hepatocellular carcinoma after surgery. Br. J. Surg. 1996, 83, 1219–1222. [Google Scholar] [CrossRef] [PubMed]
- Lai, E.C.; Fan, S.T.; Lo, C.M.; Chu, K.M.; Liu, C.L.; Wong, J. Hepatic resection for hepatocellular carcinoma. An audit of 343 patients. Ann. Surg. 1995, 221, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, K.; Kawahara, N.; Yamamoto, K.; Kajiyama, K.; Maeda, T.; Itasaka, H.; Shirabe, K.; Nishizaki, T.; Yanaga, K.; Sugimachi, K. Results of 280 liver resections for hepatocellular carcinoma. Arch. Surg. 1996, 131, 71–76. [Google Scholar] [CrossRef]
- Stotz, M.; Gerger, A.; Haybaeck, J.; Kiesslich, T.; Bullock, M.D.; Pichler, M. Molecular Targeted Therapies in Hepatocellular Carcinoma: Past, Present and Future. Anticancer Res. 2015, 35, 5737–5744. [Google Scholar]
- Paravati, A.J.; Healy, E.; Murphy, J.D.; Song, W.; Hattangadi-Gluth, J. Stereotactic body radiation therapy for primary hepatic malignancies and metastases to liver: A technical and literature review. Transl. Cancer Res. 2013, 2, 507–520. [Google Scholar]
- Llovet, J.M.; Real, M.I.; Montana, X.; Planas, R.; Coll, S.; Aponte, J.; Ayuso, C.; Sala, M.; Muchart, J.; Sola, R.; et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: A randomised controlled trial. Lancet 2002, 359, 1734–1739. [Google Scholar] [CrossRef]
- Lo, C.-M.; Ngan, H.; Tso, W.-K.; Liu, C.-L.; Lam, C.-M.; Poon, R.T.-P.; Fan, S.-T.; Wong, J. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 2002, 35, 1164–1171. [Google Scholar] [CrossRef]
- Salem, R.; Lewandowski, R.J.; Mulcahy, M.F.; Riaz, A.; Ryu, R.K.; Ibrahim, S.; Atassi, B.; Baker, T.; Gates, V.; Miller, F.H.; et al. Radioembolization for hepatocellular carcinoma using Yttrium-90 microspheres: A comprehensive report of long-term outcomes. Gastroenterology 2010, 138, 52–64. [Google Scholar] [CrossRef]
- Brown, K.T.; Do, R.K.; Gonen, M.; Covey, A.M.; Getrajdman, G.I.; Sofocleous, C.T.; Jarnagin, W.R.; D’Angelica, M.I.; Allen, P.J.; Erinjeri, J.P.; et al. Randomized Trial of Hepatic Artery Embolization for Hepatocellular Carcinoma Using Doxorubicin-Eluting Microspheres Compared With Embolization With Microspheres Alone. J. Clin. Oncol. 2016, 34, 2046–2053. [Google Scholar] [CrossRef]
- Cheng, A.-L.; Kang, Y.-K.; Chen, Z.; Tsao, C.-J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.-S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet. Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.-H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.-L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.-Y.; Cicin, I.; Merle, P.; Park, J.-W.; Blanc, J.-F.; et al. Cabozantinib (C) versus placebo (P) in patients (pts) with advanced hepatocellular carcinoma (HCC) who have received prior sorafenib: Results from the randomized phase III CELESTIAL trial. J. Clin. Oncol. 2018, 36, 207. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.-H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.-W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Sangro, B.; Gomez-Martin, C.; de la Mata, M.; Inarrairaegui, M.; Garralda, E.; Barrera, P.; Riezu-Boj, J.I.; Larrea, E.; Alfaro, C.; Sarobe, P.; et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J. Hepatol. 2013, 59, 81–88. [Google Scholar] [CrossRef]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.-Y.; Choo, S.-P.; Trojan, J.; Welling, T.H.R.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Ringborg, U.; Bergqvist, D.; Brorsson, B.; Cavallin-Stahl, E.; Ceberg, J.; Einhorn, N.; Frodin, J.-E.; Jarhult, J.; Lamnevik, G.; Lindholm, C.; et al. The Swedish Council on Technology Assessment in Health Care (SBU) systematic overview of radiotherapy for cancer including a prospective survey of radiotherapy practice in Sweden 2001--summary and conclusions. Acta Oncol. 2003, 42, 357–365. [Google Scholar] [CrossRef]
- Delaney, G.; Jacob, S.; Featherstone, C.; Barton, M. The role of radiotherapy in cancer treatment: Estimating optimal utilization from a review of evidence-based clinical guidelines. Cancer 2005, 104, 1129–1137. [Google Scholar] [CrossRef]
- Pan, C.C.; Kavanagh, B.D.; Dawson, L.A.; Li, X.A.; Das, S.K.; Miften, M.; Ten Haken, R.K. Radiation-associated liver injury. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S94–S100. [Google Scholar] [CrossRef]
- Edelstein, M.P.; Wolfe, L.A., III; Duch, D.S. Potentiation of radiation therapy by vinorelbine (Navelbine) in non-small cell lung cancer. Semin. Oncol. 1996, 23, 41–47. [Google Scholar] [PubMed]
- Fukuoka, K.; Arioka, H.; Iwamoto, Y.; Fukumoto, H.; Kurokawa, H.; Ishida, T.; Tomonari, A.; Suzuki, T.; Usuda, J.; Kanzawa, F.; et al. Mechanism of the radiosensitization induced by vinorelbine in human non-small cell lung cancer cells. Lung Cancer 2001, 34, 451–460. [Google Scholar] [CrossRef]
- Krzakowski, M.; Lucas, C.; Gridelli, C. Fractionated scheme of oral vinorelbine as single-agent therapy or in combination with cisplatin concomitantly with thoracic radiotherapy in stage III non-small-cell lung cancer: Dose-escalation phase I trial. Clin. Lung Cancer 2014, 15, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Strom, H.H.; Bremnes, R.M.; Sundstrom, S.H.; Helbekkmo, N.; Aasebo, U. Poor prognosis patients with inoperable locally advanced NSCLC and large tumors benefit from palliative chemoradiotherapy: A subset analysis from a randomized clinical phase III trial. J. Thorac. Oncol. 2014, 9, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Huynh, H.; Soo, K.C.; Chow, P.K.H.; Panasci, L.; Tran, E. Xenografts of human hepatocellular carcinoma: A useful model for testing drugs. Clin. Cancer Res. 2006, 12, 4306–4314. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Cid, M.; Larripa, I.; Slavutsky, I. Vinorelbine: Cell cycle kinetics and differential sensitivity of human lymphocyte subpopulations. Toxicol. Lett. 1997, 93, 171–176. [Google Scholar] [CrossRef]
- Stillwagon, G.B.; Order, S.E.; Guse, C.; Klein, J.L.; Leichner, P.K.; Leibel, S.A.; Fishman, E.K. 194 hepatocellular cancers treated by radiation and chemotherapy combinations: Toxicity and response: A Radiation Therapy Oncology Group Study. Int. J. Radiat. Oncol. Biol. Phys. 1989, 17, 1223–1229. [Google Scholar] [CrossRef]
- Han, K.-H.; Seong, J.; Kim, J.K.; Ahn, S.H.; Lee, D.Y.; Chon, C.Y. Pilot clinical trial of localized concurrent chemoradiation therapy for locally advanced hepatocellular carcinoma with portal vein thrombosis. Cancer 2008, 113, 995–1003. [Google Scholar] [CrossRef]
- McIntosh, A.; Hagspiel, K.D.; Al-Osaimi, A.M.; Northup, P.; Caldwell, S.; Berg, C.; Angle, J.F.; Argo, C.; Weiss, G.; Rich, T.A. Accelerated treatment using intensity-modulated radiation therapy plus concurrent capecitabine for unresectable hepatocellular carcinoma. Cancer 2009, 115, 5117–5125. [Google Scholar] [CrossRef]
- Ch’ang, H.-J.; Hsu, C.; Chen, C.-H.; Chang, Y.-H.; Chang, J.S.; Chen, L.-T. Phase II study of concomitant thalidomide during radiotherapy for hepatocellular carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 817–825. [Google Scholar] [CrossRef]
- Chon, Y.E.; Seong, J.; Kim, B.K.; Cha, J.; Kim, S.U.; Park, J.Y.; Ahn, S.H.; Han, K.-H.; Chon, C.Y.; Shin, S.K.; et al. Gastroduodenal complications after concurrent chemoradiation therapy in patients with hepatocellular carcinoma: Endoscopic findings and risk factors. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Ahn, S.H.; Seong, J.S.; Park, J.Y.; Kim, D.Y.; Kim, J.K.; Lee, D.Y.; Lee, K.H.; Han, K.-H. Early alpha-fetoprotein response as a predictor for clinical outcome after localized concurrent chemoradiotherapy for advanced hepatocellular carcinoma. Liver Int. 2011, 31, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Kang, W.J.; Kim, J.K.; Seong, J.; Park, J.Y.; Kim, D.Y.; Ahn, S.H.; Lee, D.Y.; Lee, K.H.; Lee, J.D.; et al. 18F-fluorodeoxyglucose uptake on positron emission tomography as a prognostic predictor in locally advanced hepatocellular carcinoma. Cancer 2011, 117, 4779–4787. [Google Scholar] [CrossRef] [PubMed]
- Du, R.; Lu, K.V.; Petritsch, C.; Liu, P.; Ganss, R.; Passegue, E.; Song, H.; Vandenberg, S.; Johnson, R.S.; Werb, Z.; et al. HIF1alpha induces the recruitment of bone marrow-derived vascular modulatory cells to regulate tumor angiogenesis and invasion. Cancer Cell 2008, 13, 206–220. [Google Scholar] [CrossRef]
- Murdoch, C.; Muthana, M.; Coffelt, S.B.; Lewis, C.E. The role of myeloid cells in the promotion of tumour angiogenesis. Nat. Rev. Cancer 2008, 8, 618–631. [Google Scholar] [CrossRef]
- Pollard, J.W. Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer 2004, 4, 71–78. [Google Scholar] [CrossRef]
- Harimoto, N.; Taguchi, K.; Shirabe, K.; Adachi, E.; Sakaguchi, Y.; Toh, Y.; Okamura, T.; Kayashima, H.; Taketomi, A.; Maehara, Y. The significance of fibroblast growth factor receptor 2 expression in differentiation of hepatocellular carcinoma. Oncology 2010, 78, 361–368. [Google Scholar] [CrossRef]
- Paur, J.; Nika, L.; Maier, C.; Moscu-Gregor, A.; Kostka, J.; Huber, D.; Mohr, T.; Heffeter, P.; Schrottmaier, W.C.; Kappel, S.; et al. Fibroblast growth factor receptor 3 isoforms: Novel therapeutic targets for hepatocellular carcinoma? Hepatology 2015, 62, 1767–1778. [Google Scholar] [CrossRef]
- Huynh, H.; Lee, L.Y.; Goh, K.Y.; Ong, R.; Hao, H.-X.; Huang, A.; Wang, Y.; Graus Porta, D.; Chow, P.; Chung, A. Infigratinib Mediates Vascular Normalization, Impairs Metastasis, and Improves Chemotherapy in Hepatocellular Carcinoma. Hepatology 2019, 69, 943–958. [Google Scholar] [CrossRef]
- Hoffman, R.M. In vitro sensitivity assays in cancer: A review, analysis, and prognosis. J. Clin. Lab. Anal. 1991, 5, 133–143. [Google Scholar] [CrossRef]
- Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; ISBN 9780309154000.
- Tallarida, R.J. Drug Synergism: Its Detection and Applications. J. Pharmacol. Exp. Ther. 2001, 298, 865–872. [Google Scholar] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeoh, K.W.; Prawira, A.; Saad, M.Z.B.; Lee, K.M.; Lee, E.M.H.; Low, G.K.; Mohd Nasir, M.H.B.; Phua, J.H.; Chow, W.W.L.; Lim, I.J.H.; et al. Vinorelbine Augments Radiotherapy in Hepatocellular Carcinoma. Cancers 2020, 12, 872. https://doi.org/10.3390/cancers12040872
Yeoh KW, Prawira A, Saad MZB, Lee KM, Lee EMH, Low GK, Mohd Nasir MHB, Phua JH, Chow WWL, Lim IJH, et al. Vinorelbine Augments Radiotherapy in Hepatocellular Carcinoma. Cancers. 2020; 12(4):872. https://doi.org/10.3390/cancers12040872
Chicago/Turabian StyleYeoh, Kheng Wei, Aldo Prawira, Muhammad Zafrie Bin Saad, Kok Ming Lee, Eric Ming Hon Lee, Gee Keng Low, Mohamed Hakim Bin Mohd Nasir, Jun Hao Phua, Wendy Wan Li Chow, Iris Jiu Hia Lim, and et al. 2020. "Vinorelbine Augments Radiotherapy in Hepatocellular Carcinoma" Cancers 12, no. 4: 872. https://doi.org/10.3390/cancers12040872
APA StyleYeoh, K. W., Prawira, A., Saad, M. Z. B., Lee, K. M., Lee, E. M. H., Low, G. K., Mohd Nasir, M. H. B., Phua, J. H., Chow, W. W. L., Lim, I. J. H., Omar, Y. B., Ho, R. Z. W., Le, T. B. U., Vu, T. C., Soo, K. C., & Huynh, H. (2020). Vinorelbine Augments Radiotherapy in Hepatocellular Carcinoma. Cancers, 12(4), 872. https://doi.org/10.3390/cancers12040872




