Disruption of Glycogen Utilization Markedly Improves the Efficacy of Carboplatin against Preclinical Models of Clear Cell Ovarian Carcinoma
Abstract
1. Introduction
2. Results
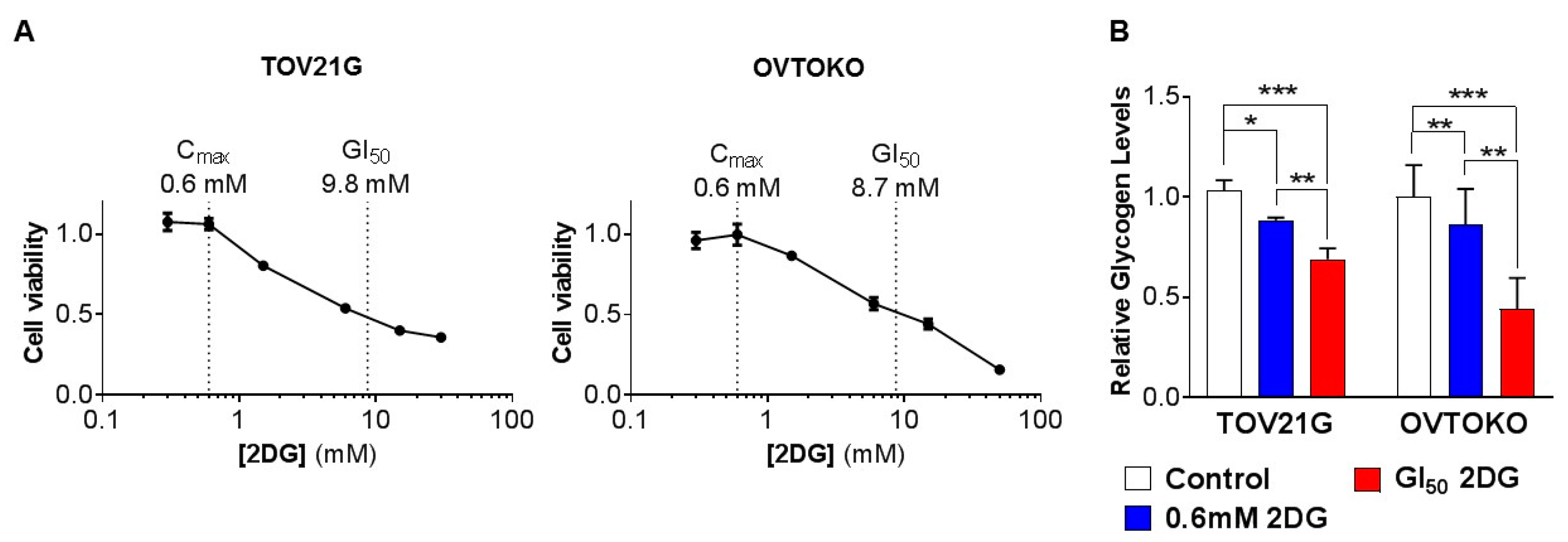
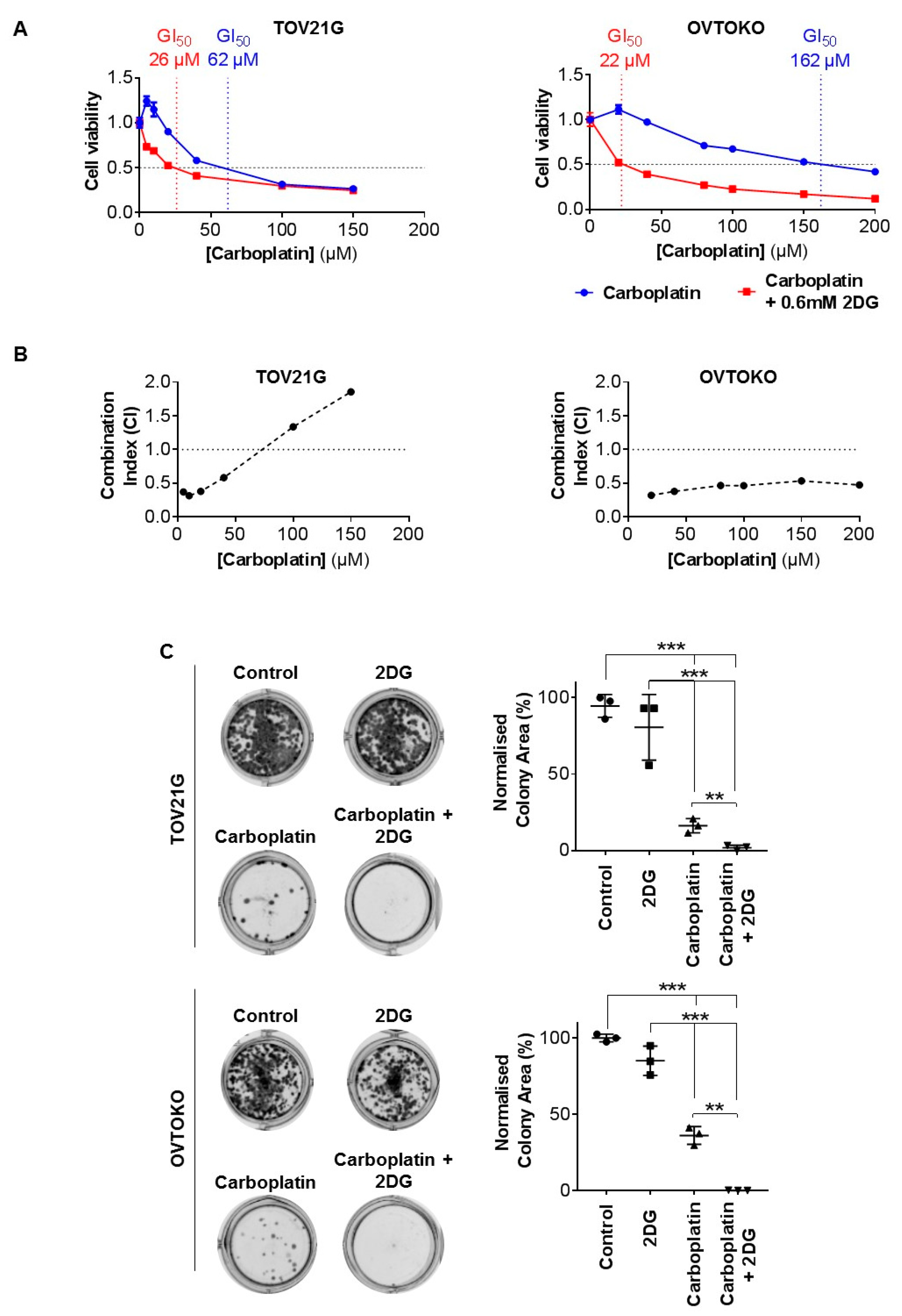
2.1. Effect of 2DG on In Vitro Growth of OCC Cell Lines with Distinct Chemo-Sensitivities
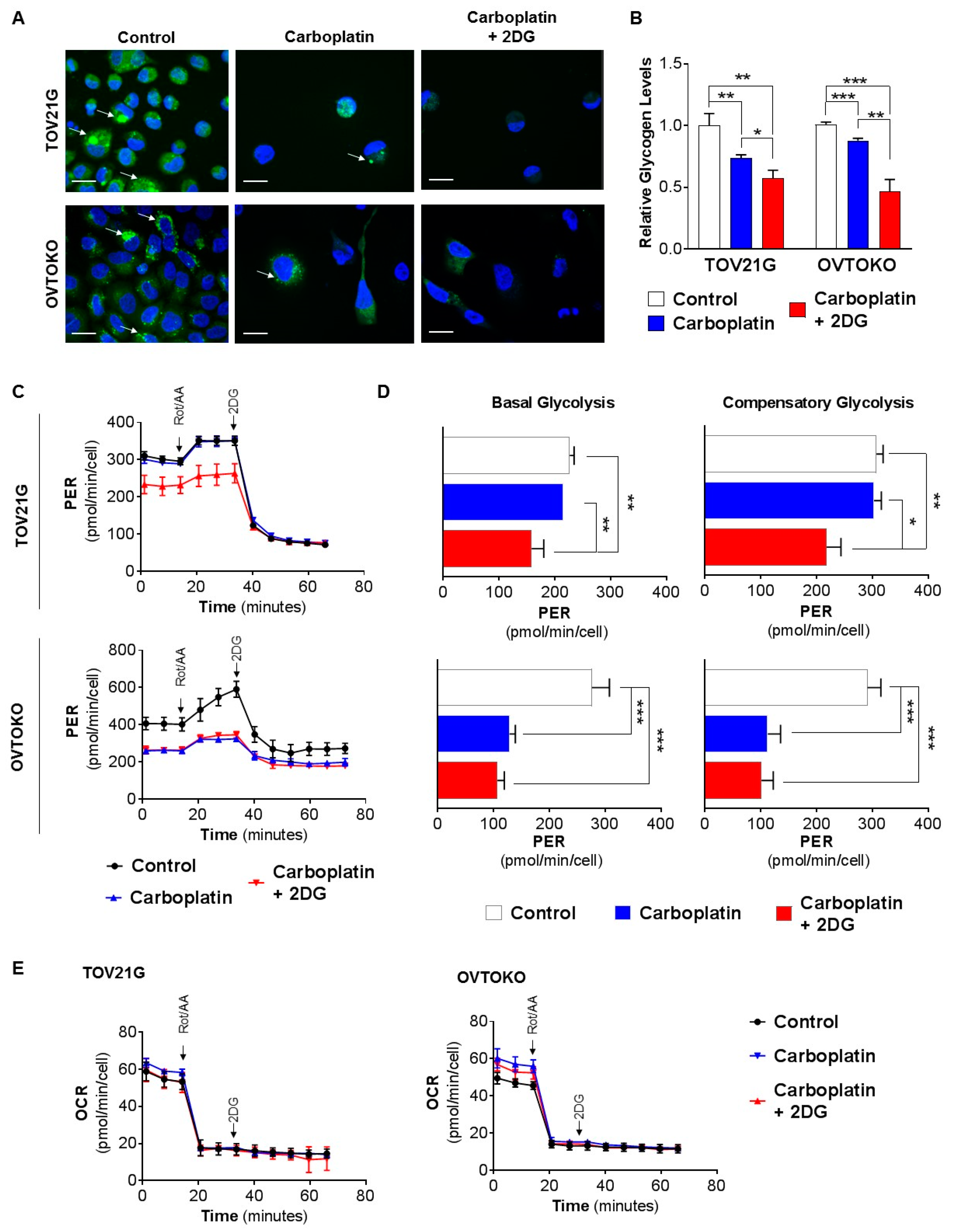
2.2. 2DG in Combination with Carboplatin Reduces Glycogen Levels in OCC Cells In Vitro
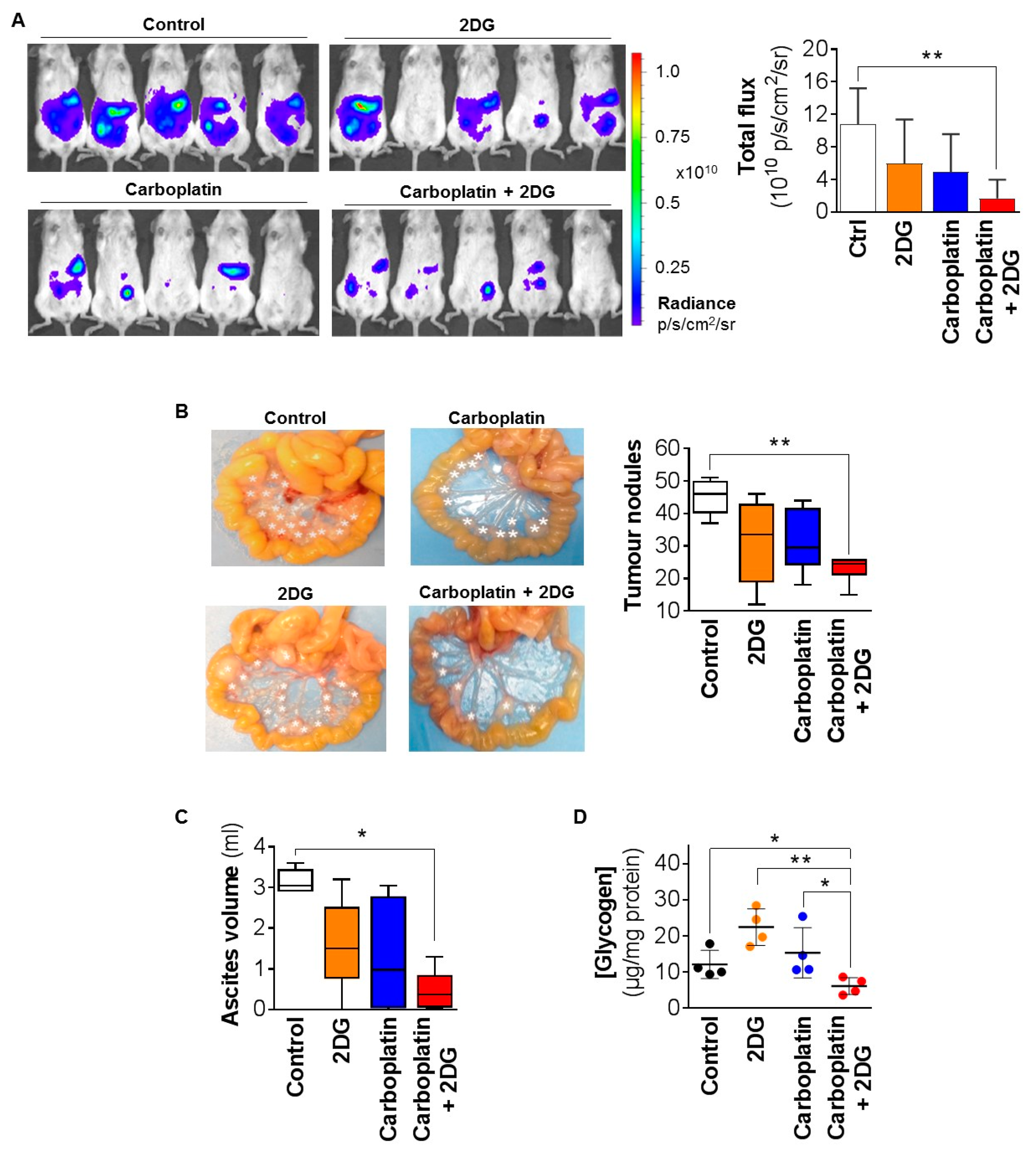
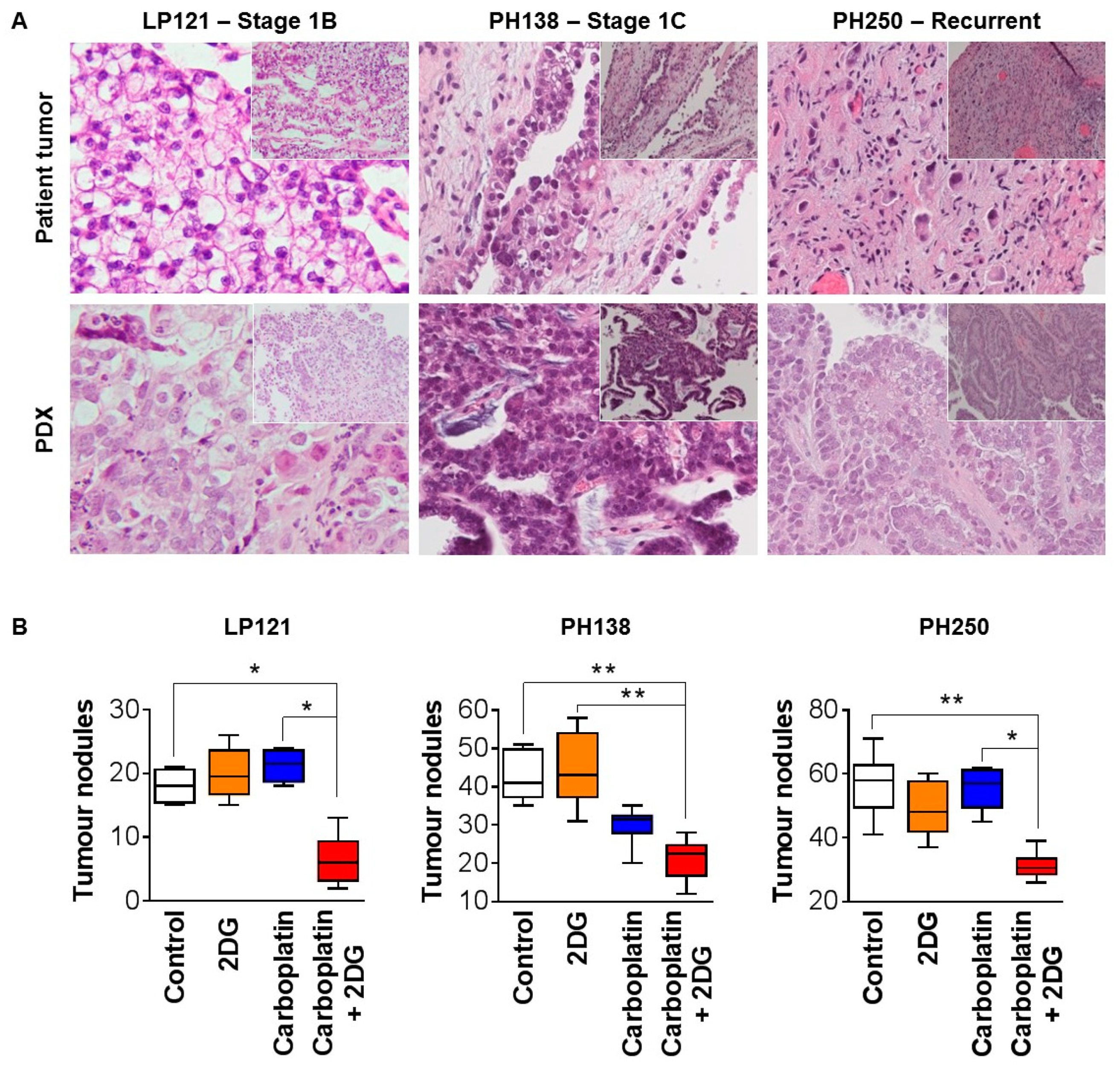
2.3. 2DG Combined with Carboplatin Reduces Tumor Burden of OCC Xenografts in Mice
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Cell Lines and Culture Conditions
4.3. Luciferase Labelling of Cells
4.4. Cell Viability Assay
4.5. Colony Formation Assay
4.6. Fluorescence Microscopy Analysis
4.7. Quantitative Glycogen Content Measurements
4.8. Bioenergetic Analysis of Glycolysis and Mitochondrial Respiration
4.9. Assessment of the Effect of 2DG on the Efficacy of Carboplatin In Vivo
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Anglesio, M.S.; Carey, M.S.; Kobel, M.; Mackay, H.; Huntsman, D.G. Clear cell carcinoma of the ovary: A report from the first ovarian clear cell symposium, June 24th, 2010. Gynecol. Oncol. 2011, 121, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Yamagami, W.; Nagase, S.; Takahashi, F.; Ino, K.; Hachisuga, T.; Aoki, D.; Katabuchi, H. Clinical statistics of gynecologic cancers in japan. J. Gynecol. Oncol. 2017, 28, e32. [Google Scholar] [CrossRef] [PubMed]
- McCluggage, W.G. Morphological subtypes of ovarian carcinoma: A review with emphasis on new developments and pathogenesis. Pathology 2011, 43, 420–432. [Google Scholar] [CrossRef] [PubMed]
- Uekuri, C.; Shigetomi, H.; Ono, S.; Sasaki, Y.; Matsuura, M.; Kobayashi, H. Toward an understanding of the pathophysiology of clear cell carcinoma of the ovary (review). Oncol. Lett. 2013, 6, 1163–1173. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ozols, R.F.; Bundy, B.N.; Greer, B.E.; Fowler, J.M.; Clarke-Pearson, D.; Burger, R.A.; Mannel, R.S.; DeGeest, K.; Hartenbach, E.M.; Baergen, R.; et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: A gynecologic oncology group study. J. Clin. Oncol. 2003, 21, 3194–3200. [Google Scholar] [CrossRef] [PubMed]
- Al-Barrak, J.; Santos, J.L.; Tinker, A.; Hoskins, P.; Gilks, C.B.; Lau, H.; Swenerton, K.D. Exploring palliative treatment outcomes in women with advanced or recurrent ovarian clear cell carcinoma. Gynecol. Oncol. 2011, 122, 107–110. [Google Scholar] [CrossRef]
- He, Y.; Wu, A.C.; Harrington, B.S.; Davies, C.M.; Wallace, S.J.; Adams, M.N.; Palmer, J.S.; Roche, D.K.; Hollier, B.G.; Westbrook, T.F.; et al. Elevated CDCP1 predicts poor patient outcome and mediates ovarian clear cell carcinoma by promoting tumor spheroid formation, cell migration and chemoresistance. Oncogene 2016, 35, 468–478. [Google Scholar] [CrossRef]
- Sugiyama, T.; Kamura, T.; Kigawa, J.; Terakawa, N.; Kikuchi, Y.; Kita, T.; Suzuki, M.; Sato, I.; Taguchi, K. Clinical characteristics of clear cell carcinoma of the ovary. Cancer 2000, 88, 2584–2589. [Google Scholar] [CrossRef]
- Tan, D.S.; Miller, R.E.; Kaye, S.B. New perspectives on molecular targeted therapy in ovarian clear cell carcinoma. Br. J. Cancer 2013, 108, 1553–1559. [Google Scholar] [CrossRef]
- Pectasides, D.; Pectasides, E.; Psyrri, A.; Economopoulos, T. Treatment issues in clear cell carcinoma of the ovary: A different entity? Oncologist 2006, 11, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Gounaris, I.; Brenton, J.D. Molecular pathogenesis of ovarian clear cell carcinoma. Future Oncol. 2015, 11, 1389–1405. [Google Scholar] [CrossRef]
- Zois, C.E.; Harris, A.L. Glycogen metabolism has a key role in the cancer microenvironment and provides new targets for cancer therapy. J. Mol. Med. 2016, 94, 137–154. [Google Scholar] [CrossRef]
- Takehara, K.; Konishi, H.; Kojima, A.; Okame, S.; Shiroyama, Y.; Yokoyama, T.; Nogawa, T.; Sugawara, Y. SUV max of FDG-PET/CT as a prognostic factor in ovarian clear cell adenocarcinoma. J. Clin. Oncol. 2014, 32, e16509. [Google Scholar] [CrossRef]
- Roach, P.J.; Depaoli-Roach, A.A.; Hurley, T.D.; Tagliabracci, V.S. Glycogen and its metabolism: Some new developments and old themes. Biochem. J. 2012, 441, 763–787. [Google Scholar] [CrossRef]
- Dwarakanath, B.S.; Jain, V. Targeting glucose metabolism with 2-deoxy-d-glucose for improving cancer therapy. Future Oncol. 2009, 5, 581–585. [Google Scholar] [CrossRef]
- El Mjiyad, N.; Caro-maldonado, A.; Ramírez-peinado, S.; Muñoz-pinedo, C. Sugar-free approaches to cancer cell killing. Oncogene 2011, 30, 253–264. [Google Scholar] [CrossRef]
- Kurtoglu, M.; Gao, N.; Shang, J.; Maher, J.C.; Lehrman, M.A.; Wangpaichitr, M.; Savaraj, N.; Lane, A.N.; Lampidis, T.J. Under normoxia, 2-deoxy-d-glucose elicits cell death in select tumor types not by inhibition of glycolysis but by interfering with n-linked glycosylation. Mol. Cancer 2007, 6, 3049–3058. [Google Scholar] [CrossRef]
- Bianchi, C.; Meregalli, C.; Bombelli, S.; Di Stefano, V.; Salerno, F.; Torsello, B.; De Marco, S.; Bovo, G.; Cifola, I.; Mangano, E.; et al. The glucose and lipid metabolism reprogramming is grade-dependent in clear cell renal cell carcinoma primary cultures and is targetable to modulate cell viability and proliferation. Oncotarget 2017, 8, 113502–113515. [Google Scholar] [CrossRef]
- Hernlund, E.; Hjerpe, E.; Avall-Lundqvist, E.; Shoshan, M. Ovarian carcinoma cells with low levels of beta-f1-atpase are sensitive to combined platinum and 2-deoxy-d-glucose treatment. Mol. Cancer 2009, 8, 1916–1923. [Google Scholar] [CrossRef]
- Cheng, G.; Zielonka, J.; Dranka, B.P.; McAllister, D.; Mackinnon, A.C., Jr.; Joseph, J.; Kalyanaraman, B. Mitochondria-targeted drugs synergize with 2-deoxyglucose to trigger breast cancer cell death. Cancer Res. 2012, 72, 2634–2644. [Google Scholar] [CrossRef]
- Lucantoni, F.; Dussmann, H.; Prehn, J.H.M. Metabolic targeting of breast cancer cells with the 2-deoxy-d-glucose and the mitochondrial bioenergetics inhibitor mdivi-1. Front. Cell Dev. Biol. 2018, 6, 113. [Google Scholar] [CrossRef]
- Zhang, T.; Zhu, X.; Wu, H.; Jiang, K.; Zhao, G.; Shaukat, A.; Deng, G.; Qiu, C. Targeting the ROS/PI3K/AKT/HIF-1alpha/HK2 axis of breast cancer cells: Combined administration of Polydatin and 2-Deoxy-d-glucose. J. Cell Mol. Med. 2019, 23, 3711–3723. [Google Scholar] [CrossRef]
- Lucantoni, F.; Dussmann, H.; Llorente-Folch, I.; Prehn, J.H.M. BCL2 and BCL(X)L selective inhibitors decrease mitochondrial atp production in breast cancer cells and are synthetically lethal when combined with 2-deoxy-d-glucose. Oncotarget 2018, 9, 26046–26063. [Google Scholar] [CrossRef]
- Lyamzaev, K.G.; Izyumov, D.S.; Avetisyan, A.V.; Yang, F.; Pletjushkina, O.Y.; Chernyak, B.V. Inhibition of mitochondrial bioenergetics: The effects on structure of mitochondria in the cell and on apoptosis. Acta Biochim. Pol. 2004, 51, 553–562. [Google Scholar] [CrossRef]
- Pattni, B.S.; Jhaveri, A.; Dutta, I.; Baleja, J.D.; Degterev, A.; Torchilin, V. Targeting energy metabolism of cancer cells: Combined administration of NCL-240 and 2-DG. Int. J. Pharm. 2017, 532, 149–156. [Google Scholar] [CrossRef]
- Abbadi, S.; Rodarte, J.J.; Abutaleb, A.; Lavell, E.; Smith, C.L.; Ruff, W.; Schiller, J.; Olivi, A.; Levchenko, A.; Guerrero-Cazares, H.; et al. Glucose-6-phosphatase is a key metabolic regulator of glioblastoma invasion. Mol. Cancer Res. 2014, 12, 1547–1559. [Google Scholar] [CrossRef]
- Maher, J.C.; Krishan, A.; Lampidis, T.J. Greater cell cycle inhibition and cytotoxicity induced by 2-deoxy-d-glucose in tumor cells treated under hypoxic vs aerobic conditions. Cancer Chemother. Pharm. 2004, 53, 116–122. [Google Scholar] [CrossRef]
- Stein, M.; Lin, H.; Jeyamohan, C.; Dvorzhinski, D.; Gounder, M.; Bray, K.; Eddy, S.; Goodin, S.; White, E.; Dipaola, R.S. Targeting tumor metabolism with 2-deoxyglucose in patients with castrate-resistant prostate cancer and advanced malignancies. Prostate 2010, 70, 1388–1394. [Google Scholar] [CrossRef]
- Raez, L.E.; Papadopoulos, K.; Ricart, A.D.; Chiorean, E.G.; Dipaola, R.S.; Stein, M.N.; Rocha Lima, C.M.; Schlesselman, J.J.; Tolba, K.; Langmuir, V.K.; et al. A phase i dose-escalation trial of 2-deoxy-d-glucose alone or combined with docetaxel in patients with advanced solid tumors. Cancer Chemother. Pharm. 2013, 71, 523–530. [Google Scholar] [CrossRef]
- Chou, T.-C. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010, 70, 440. [Google Scholar] [CrossRef]
- Louzao, M.C.; Espina, B.; Vieytes, M.R.; Vega, F.V.; Rubiolo, J.A.; Baba, O.; Terashima, T.; Botana, L.M. “Fluorescent glycogen” formation with sensibility for in vivo and in vitro detection. Glycoconj. J. 2008, 25, 503–510. [Google Scholar] [CrossRef]
- Mookerjee, S.A.; Gerencser, A.A.; Nicholls, D.G.; Brand, M.D. Quantifying intracellular rates of glycolytic and oxidative atp production and consumption using extracellular flux measurements. J. Biol. Chem. 2018, 293, 12649–12652. [Google Scholar] [CrossRef]
- Alvero, A.B.; Montagna, M.K.; Sumi, N.J.; Joo, W.D.; Graham, E.; Mor, G. Multiple blocks in the engagement of oxidative phosphorylation in putative ovarian cancer stem cells: Implication for maintenance therapy with glycolysis inhibitors. Oncotarget 2014, 5, 8703–8715. [Google Scholar] [CrossRef]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef]
- Favaro, E.; Bensaad, K.; Chong, M.G.; Tennant, D.A.; Ferguson, D.J.; Snell, C.; Steers, G.; Turley, H.; Li, J.L.; Gunther, U.L.; et al. Glucose utilization via glycogen phosphorylase sustains proliferation and prevents premature senescence in cancer cells. Cell Metab. 2012, 16, 751–764. [Google Scholar] [CrossRef]
- Iida, Y.; Aoki, K.; Asakura, T.; Ueda, K.; Yanaihara, N.; Takakura, S.; Yamada, K.; Okamoto, A.; Tanaka, T.; Ohkawa, K. Hypoxia promotes glycogen synthesis and accumulation in human ovarian clear cell carcinoma. Int. J. Oncol. 2012, 40, 2122–2130. [Google Scholar] [CrossRef]
- Scarman, A.L.; Hooper, J.D.; Boucaut, K.J.; Sit, M.L.; Webb, G.C.; Normyle, J.F.; Antalis, T.M. Organization and chromosomal localization of the murine testisin gene encoding a serine protease temporally expressed during spermatogenesis. Eur. J. Biochem. FEBS 2001, 268, 1250–1258. [Google Scholar] [CrossRef]
- Wu, A.C.; He, Y.; Broomfield, A.; Paatan, N.J.; Harrington, B.S.; Tseng, H.-W.; Beaven, E.A.; Kiernan, D.M.; Swindle, P.; Clubb, A.B.; et al. CD169+ macrophages mediate pathological formation of woven bone in skeletal lesions of prostate cancer. J. Pathol. 2016, 239, 218–230. [Google Scholar] [CrossRef]
- Harrington, B.S.; He, Y.; Davies, C.M.; Wallace, S.J.; Adams, M.N.; Beaven, E.A.; Roche, D.K.; Kennedy, C.; Chetty, N.P.; Crandon, A.J.; et al. Cell line and patient-derived xenograft models reveal elevated CDCP1 as a target in high-grade serous ovarian cancer. Br. J. Cancer 2016, 114, 417–426. [Google Scholar] [CrossRef]
- Crowley, L.C.; Christensen, M.E.; Waterhouse, N.J. Measuring survival of adherent cells with the colony-forming assay. Cold Spring Harb. Protoc. 2016, 8, 721–725. [Google Scholar] [CrossRef]
- Guzman, C.; Bagga, M.; Kaur, A.; Westermarck, J.; Abankwa, D. ColonyArea: An ImageJ plugin to automatically quantify colony formation in clonogenic assays. PLoS ONE 2014, 9, e92444. [Google Scholar] [CrossRef]
- Harrington, B.S.; He, Y.; Khan, T.; Puttick, S.; Conroy, P.J.; Kryza, T.; Cuda, T.; Sokolowski, K.A.; Tse, B.W.C.; Robbins, K.K.; et al. Anti-CDCP1 immuno-conjugates for detection and inhibition of ovarian cancer. Theranostics 2020, 10, 2095–2114. [Google Scholar] [CrossRef]
- Witney, T.H.; Carroll, L.; Alam, I.S.; Chandrashekran, A.; Nguyen, Q.-D.; Sala, R.; Harris, R.; DeBerardinis, R.J.; Agarwal, R.; Aboagye, E.O. A novel radiotracer to image glycogen metabolism in tumors by positron emission tomography. Cancer Res. 2014, 74, 1319–1328. [Google Scholar] [CrossRef]
- Sullivan, M.A.; O’Connor, M.J.; Umana, F.; Roura, E.; Jack, K.; Stapleton, D.I.; Gilbert, R.G. Molecular insights into glycogen α-particle formation. Biomacromolecules 2012, 13, 3805–3813. [Google Scholar] [CrossRef]
- Dier, U.; Shin, D.-H.; Hemachandra, L.P.M.P.; Uusitalo, L.M.; Hempel, N. Bioenergetic analysis of ovarian cancer cell lines: Profiling of histological subtypes and identification of a mitochondria-defective cell line. PLoS ONE 2014, 9, e98479. [Google Scholar] [CrossRef]
- Dar, S.; Chhina, J.; Mert, I.; Chitale, D.; Buekers, T.; Kaur, H.; Giri, S.; Munkarah, A.; Rattan, R. Bioenergetic adaptations in chemoresistant ovarian cancer cells. Sci. Rep. 2017, 7, 8760. [Google Scholar] [CrossRef]
- Weroha, S.J.; Becker, M.A.; Enderica-Gonzalez, S.; Harrington, S.C.; Oberg, A.L.; Maurer, M.J.; Perkins, S.E.; AlHilli, M.; Butler, K.A.; McKinstry, S.; et al. Tumorgrafts as in vivo surrogates for women with ovarian cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 1288–1297. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, T.; He, Y.; Kryza, T.; Harrington, B.S.; Gunter, J.H.; Sullivan, M.A.; Cuda, T.; Rogers, R.; Davies, C.M.; Broomfield, A.; et al. Disruption of Glycogen Utilization Markedly Improves the Efficacy of Carboplatin against Preclinical Models of Clear Cell Ovarian Carcinoma. Cancers 2020, 12, 869. https://doi.org/10.3390/cancers12040869
Khan T, He Y, Kryza T, Harrington BS, Gunter JH, Sullivan MA, Cuda T, Rogers R, Davies CM, Broomfield A, et al. Disruption of Glycogen Utilization Markedly Improves the Efficacy of Carboplatin against Preclinical Models of Clear Cell Ovarian Carcinoma. Cancers. 2020; 12(4):869. https://doi.org/10.3390/cancers12040869
Chicago/Turabian StyleKhan, Tashbib, Yaowu He, Thomas Kryza, Brittney S. Harrington, Jennifer H. Gunter, Mitchell A. Sullivan, Tahleesa Cuda, Rebecca Rogers, Claire M. Davies, Amy Broomfield, and et al. 2020. "Disruption of Glycogen Utilization Markedly Improves the Efficacy of Carboplatin against Preclinical Models of Clear Cell Ovarian Carcinoma" Cancers 12, no. 4: 869. https://doi.org/10.3390/cancers12040869
APA StyleKhan, T., He, Y., Kryza, T., Harrington, B. S., Gunter, J. H., Sullivan, M. A., Cuda, T., Rogers, R., Davies, C. M., Broomfield, A., Gough, M., Wu, A. C., McGann, T., Weroha, S. J., Haluska, P., Forbes, J. M., Armes, J. E., Barry, S. C., Coward, J. I., ... Hooper, J. D. (2020). Disruption of Glycogen Utilization Markedly Improves the Efficacy of Carboplatin against Preclinical Models of Clear Cell Ovarian Carcinoma. Cancers, 12(4), 869. https://doi.org/10.3390/cancers12040869




