Immunotherapy in Glioblastoma: Current Shortcomings and Future Perspectives
Abstract
1. Introduction
2. Single Immune Therapies are not Effective in Glioblastoma
2.1. Immune Checkpoint Inhibitors
2.1.1. PD-1 Inhibitor Trials
2.1.2. PD-L1 Inhibitor Trials
2.2. Vaccination with Peptides or Dendritic Cells
2.2.1. Peptide Vaccination Trials
2.2.2. DC Vaccination Trials
2.3. Adoptive Transfer of Effector Lymphocytes
2.3.1. Gene-Engineered T cells
2.3.2. Non-Engineered T Cells and Other Lymphocytes
3. Immune Therapy Resistance in Glioblastoma
3.1. Low-Immunogenicity of Glioblastoma
3.2. Immune Privilege of CNS
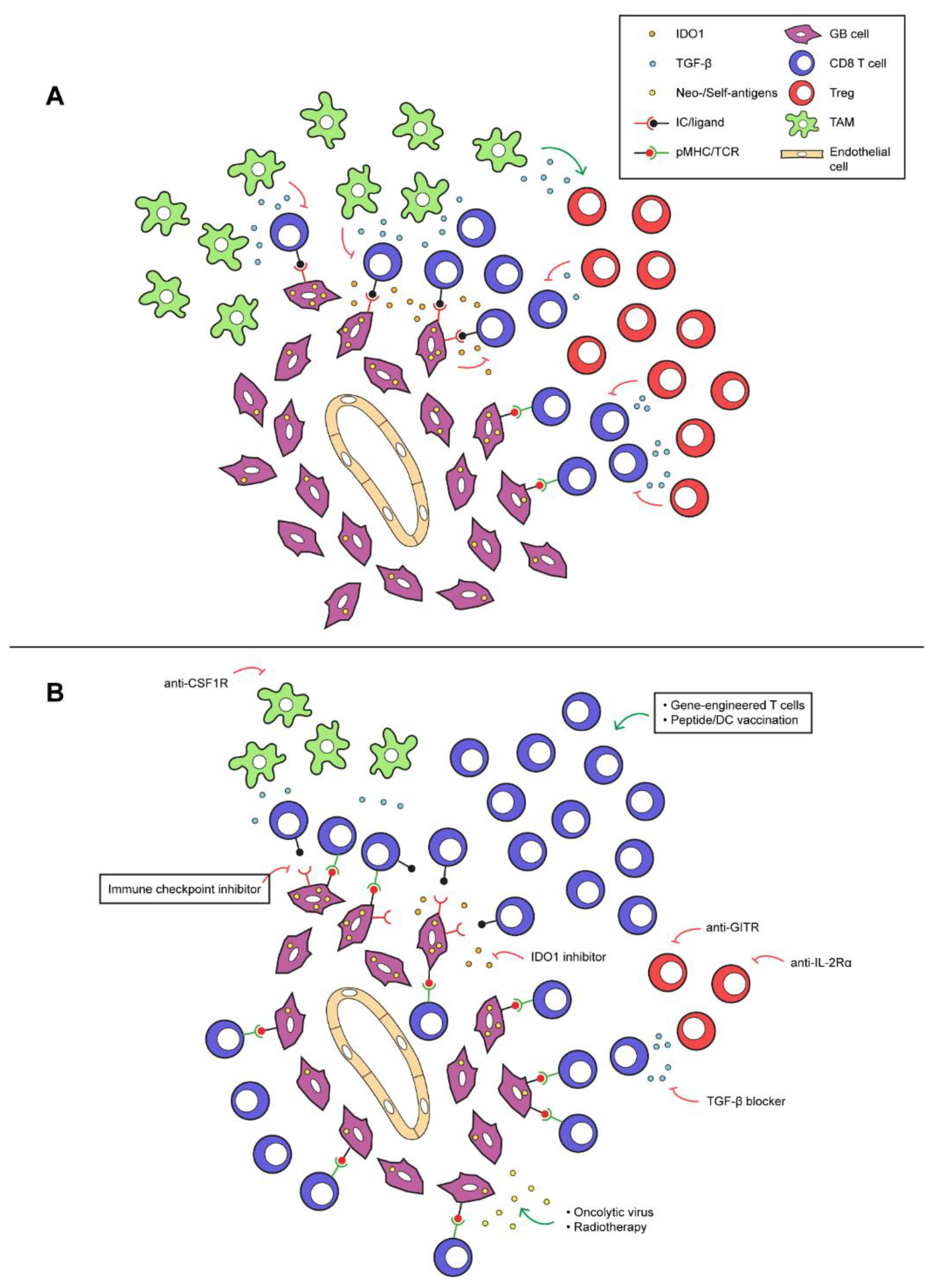
3.3. Immune-Suppressive Micro-Environment
3.3.1. Immune-Suppressive Cells
3.3.2. Immune-Suppressive Mediators
4. Strategies to Sensitize Glioblastoma to Immune Therapies
4.1. Strategies to Enhance the Efficacy of ICI
4.1.1. Downregulating the Immune Suppressive Micro-Environment
4.1.2. Increasing the CD8 T Cell Influx
4.2. Strategies to Enhance the Efficacy of Vaccinations
4.3. Strategies to Enhance the Efficacy of Adoptive Transfer of Effector Lymphocytes
4.4. Combination of ICI, Vaccination, and Adoptive Transfer of Effector Lymphocytes
5. Outstanding Questions for Future Research
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ostrom, Q.T.; Gittleman, H.; Liao, P.; Vecchione-Koval, T.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. Cbtrus statistical report: Primary brain and other central nervous system tumors diagnosed in the united states in 2010–2014. Neuro. Oncol. 2017, 19, v1–v88. [Google Scholar] [CrossRef] [PubMed]
- Darvin, P.; Toor, S.M.; Sasidharan Nair, V.; Elkord, E. Immune checkpoint inhibitors: Recent progress and potential biomarkers. Exp. Mol. Med. 2018, 50, 165. [Google Scholar] [CrossRef] [PubMed]
- Roberts, Z.J.; Better, M.; Bot, A.; Roberts, M.R.; Ribas, A. Axicabtagene ciloleucel, a first-in-class car t cell therapy for aggressive nhl. Leuk. Lymphoma 2018, 59, 1785–1796. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crino, L.; Eberhardt, W.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Escudier, B.; McDermott, D.F.; George, S.; Hammers, H.J.; Srinivas, S.; Tykodi, S.S.; Sosman, J.A.; Procopio, G.; Plimack, E.R.; et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N. Engl. J. Med. 2015, 373, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Schachter, J.; Long, G.V.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.; Lotem, M.; et al. Pembrolizumab versus ipilimumab in advanced melanoma. N. Engl. J. Med. 2015, 372, 2521–2532. [Google Scholar] [CrossRef] [PubMed]
- Reardon, D.A.; Omuro, A.; Brandes, A.A.; Rieger, J.; Wick, A.; Sepulveda, J.; Phuphanich, S.; de Souza, P.; Ahluwalia, M.S.; Lim, M.; et al. Os10.3 randomized phase 3 study evaluating the efficacy and safety of nivolumab vs bevacizumab in patients with recurrent glioblastoma: Checkmate 143. Neuro-Oncology 2017, 19, iii21. [Google Scholar] [CrossRef]
- Sampson, J.H.; Omuro, A.M.P.; Preusser, M.; Lim, M.; Butowski, N.A.; Cloughesy, T.F.; Strauss, L.C.; Latek, R.R.; Paliwal, P.; Weller, M.; et al. A randomized, phase 3, open-label study of nivolumab versus temozolomide (tmz) in combination with radiotherapy (rt) in adult patients (pts) with newly diagnosed, o-6-methylguanine DNA methyltransferase (mgmt)-unmethylated glioblastoma (gbm): Checkmate-498. J. Clin. Oncol. 2016, 34, TPS2079. [Google Scholar] [CrossRef]
- Schalper, K.A.; Rodriguez-Ruiz, M.E.; Diez-Valle, R.; Lopez-Janeiro, A.; Porciuncula, A.; Idoate, M.A.; Inoges, S.; de Andrea, C.; Lopez-Diaz de Cerio, A.; Tejada, S.; et al. Neoadjuvant nivolumab modifies the tumor immune microenvironment in resectable glioblastoma. Nat. Med. 2019, 25, 470–476. [Google Scholar] [CrossRef]
- Cloughesy, T.F.; Mochizuki, A.Y.; Orpilla, J.R.; Hugo, W.; Lee, A.H.; Davidson, T.B.; Wang, A.C.; Ellingson, B.M.; Rytlewski, J.A.; Sanders, C.M.; et al. Neoadjuvant anti-pd-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat. Med. 2019, 25, 477–486. [Google Scholar] [CrossRef]
- Bouffet, E.; Larouche, V.; Campbell, B.B.; Merico, D.; de Borja, R.; Aronson, M.; Durno, C.; Krueger, J.; Cabric, V.; Ramaswamy, V.; et al. Immune checkpoint inhibition for hypermutant glioblastoma multiforme resulting from germline biallelic mismatch repair deficiency. J. Clin. Oncol. 2016, 34, 2206–2211. [Google Scholar] [CrossRef]
- Johanns, T.M.; Miller, C.A.; Dorward, I.G.; Tsien, C.; Chang, E.; Perry, A.; Uppaluri, R.; Ferguson, C.; Schmidt, R.E.; Dahiya, S.; et al. Immunogenomics of hypermutated glioblastoma: A patient with germline pole deficiency treated with checkpoint blockade immunotherapy. Cancer Discov. 2016, 6, 1230–1236. [Google Scholar] [CrossRef]
- Ahmad, H.; Fadul, C.E.; Schiff, D.; Purow, B. Checkpoint inhibitor failure in hypermutated and mismatch repair-mutated recurrent high-grade gliomas. Neurooncol. Pract. 2019, 6, 424–427. [Google Scholar] [CrossRef]
- Hunter, C.; Smith, R.; Cahill, D.P.; Stephens, P.; Stevens, C.; Teague, J.; Greenman, C.; Edkins, S.; Bignell, G.; Davies, H.; et al. A hypermutation phenotype and somatic msh6 mutations in recurrent human malignant gliomas after alkylator chemotherapy. Cancer Res. 2006, 66, 3987–3991. [Google Scholar] [CrossRef]
- Choi, S.; Yu, Y.; Grimmer, M.R.; Wahl, M.; Chang, S.M.; Costello, J.F. Temozolomide-associated hypermutation in gliomas. Neuro. Oncol. 2018, 20, 1300–1309. [Google Scholar] [CrossRef]
- Draaisma, K.; Chatzipli, A.; Taphoorn, M.; Kerkhof, M.; Weyerbrock, A.; Sanson, M.; Hoeben, A.; Lukacova, S.; Lombardi, G.; Leenstra, S.; et al. Molecular evolution of idh wild-type glioblastomas treated with standard of care affects survival and design of precision medicine trials: A report from the eortc 1542 study. J. Clin. Oncol. 2019, JCO1900367. [Google Scholar] [CrossRef]
- Barthel, F.P.; Johnson, K.C.; Varn, F.S.; Moskalik, A.D.; Tanner, G.; Kocakavuk, E.; Anderson, K.J.; Abiola, O.; Aldape, K.; Alfaro, K.D.; et al. Longitudinal molecular trajectories of diffuse glioma in adults. Nature 2019, 576, 112–120. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, B.; Hu, X.; Kim, H.; Squatrito, M.; Scarpace, L.; deCarvalho, A.C.; Lyu, S.; Li, P.; Li, Y.; et al. Tumor evolution of glioma-intrinsic gene expression subtypes associates with immunological changes in the microenvironment. Cancer Cell. 2017, 32, 42–56 e46. [Google Scholar] [CrossRef]
- Neyns, B.; Salama, L.B.; Awada, G.; Cremer, J.D.; Schwarze, J.K.; Seynaeve, L.; Four, S.D.; Fischbuch, L.; Vanbinst, A.-M.; Everaert, H.; et al. Gliavax: A stratified phase ii clinical trial of avelumab and axitinib in patients with recurrent glioblastoma. J. Clin. Oncol. 2019, 37, 2034. [Google Scholar] [CrossRef]
- David, A.R.; Thomas Joseph, K.; Jorg, D.; Jennifer Leigh, C.; Gavin, D.; Michael, L.; Timothy Francis, C.; Hui Kong, G.; Andrew, J.P.; Paul, S.; et al. Phase ii study to evaluate safety and efficacy of medi4736 (durvalumab) + radiotherapy in patients with newly diagnosed unmethylated mgmt glioblastoma (new unmeth gbm). J. Clin. Oncol. 2019, 37, 2032. [Google Scholar]
- French, P.J.; Eoli, M.; Sepulveda, J.M.; de Heer, I.; Kros, J.M.; Walenkamp, A.; Frenel, J.S.; Franceschi, E.; Clement, P.M.; Weller, M.; et al. Defining egfr amplification status for clinical trial inclusion. Neuro. Oncol. 2019, 21, 1263–1272. [Google Scholar] [CrossRef]
- Gao, Y.; Vallentgoed, W.R.; French, P.J. Finding the right way to target egfr in glioblastomas; lessons from lung adenocarcinomas. Cancers 2018, 10, 489. [Google Scholar] [CrossRef]
- Congdon, K.L.; Gedeon, P.C.; Suryadevara, C.M.; Caruso, H.G.; Cooper, L.J.; Heimberger, A.B.; Sampson, J.H. Epidermal growth factor receptor and variant iii targeted immunotherapy. Neuro. Oncol. 2014, 16 (Suppl. 8), viii20–viii25. [Google Scholar] [CrossRef]
- Sampson, J.H.; Heimberger, A.B.; Archer, G.E.; Aldape, K.D.; Friedman, A.H.; Friedman, H.S.; Gilbert, M.R.; Herndon, J.E., 2nd; McLendon, R.E.; Mitchell, D.A.; et al. Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant iii peptide vaccination in patients with newly diagnosed glioblastoma. J. Clin. Oncol. 2010, 28, 4722–4729. [Google Scholar] [CrossRef]
- David, A.R.; James, S.; David Dinh, T.; Karen, L.F.; Louis, B.N.; Gordon, L.; Daniela Annenelie, B.; Rimas Vincas, L.; Annick, D.; Lynn Stuart, A.; et al. React: Overall survival from a randomized phase ii study of rindopepimut (cdx-110) plus bevacizumab in relapsed glioblastoma. J. Clin. Oncol. 2015, 33, 2009. [Google Scholar]
- Sampson, J.H.; Aldape, K.D.; Archer, G.E.; Coan, A.; Desjardins, A.; Friedman, A.H.; Friedman, H.S.; Gilbert, M.R.; Herndon, J.E.; McLendon, R.E.; et al. Greater chemotherapy-induced lymphopenia enhances tumor-specific immune responses that eliminate egfrviii-expressing tumor cells in patients with glioblastoma. Neuro. Oncol. 2011, 13, 324–333. [Google Scholar] [CrossRef]
- Schuster, J.; Lai, R.K.; Recht, L.D.; Reardon, D.A.; Paleologos, N.A.; Groves, M.D.; Mrugala, M.M.; Jensen, R.; Baehring, J.M.; Sloan, A.; et al. A phase ii, multicenter trial of rindopepimut (cdx-110) in newly diagnosed glioblastoma: The act iii study. Neuro. Oncol. 2015, 17, 854–861. [Google Scholar] [CrossRef]
- Weller, M.; Butowski, N.; Tran, D.D.; Recht, L.D.; Lim, M.; Hirte, H.; Ashby, L.; Mechtler, L.; Goldlust, S.A.; Iwamoto, F.; et al. Rindopepimut with temozolomide for patients with newly diagnosed, egfrviii-expressing glioblastoma (act iv): A randomised, double-blind, international phase 3 trial. Lancet Oncol. 2017, 18, 1373–1385. [Google Scholar] [CrossRef]
- Nathanson, D.A.; Gini, B.; Mottahedeh, J.; Visnyei, K.; Koga, T.; Gomez, G.; Eskin, A.; Hwang, K.; Wang, J.; Masui, K.; et al. Targeted therapy resistance mediated by dynamic regulation of extrachromosomal mutant egfr DNA. Science 2014, 343, 72–76. [Google Scholar] [CrossRef]
- van den Bent, M.J.; Gao, Y.; Kerkhof, M.; Kros, J.M.; Gorlia, T.; van Zwieten, K.; Prince, J.; van Duinen, S.; Sillevis Smitt, P.A.; Taphoorn, M.; et al. Changes in the egfr amplification and egfrviii expression between paired primary and recurrent glioblastomas. Neuro. Oncol. 2015, 17, 935–941. [Google Scholar] [CrossRef]
- Liau, L.M.; Ashkan, K.; Tran, D.D.; Campian, J.L.; Trusheim, J.E.; Cobbs, C.S.; Heth, J.A.; Salacz, M.; Taylor, S.; D’Andre, S.D.; et al. First results on survival from a large phase 3 clinical trial of an autologous dendritic cell vaccine in newly diagnosed glioblastoma. J. Transl. Med. 2018, 16, 142. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, D.M.; Nasrallah, M.P.; Desai, A.; Melenhorst, J.J.; Mansfield, K.; Morrissette, J.J.D.; Martinez-Lage, M.; Brem, S.; Maloney, E.; Shen, A.; et al. A single dose of peripherally infused egfrviii-directed car t cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci. Transl. Med. 2017, 9, eaaa0984. [Google Scholar] [CrossRef] [PubMed]
- Goff, S.L.; Morgan, R.A.; Yang, J.C.; Sherry, R.M.; Robbins, P.F.; Restifo, N.P.; Feldman, S.A.; Lu, Y.C.; Lu, L.; Zheng, Z.; et al. Pilot trial of adoptive transfer of chimeric antigen receptor-transduced t cells targeting egfrviii in patients with glioblastoma. J. Immunother. 2019, 42, 126–135. [Google Scholar] [CrossRef]
- Brown, C.E.; Alizadeh, D.; Starr, R.; Weng, L.; Wagner, J.R.; Naranjo, A.; Ostberg, J.R.; Blanchard, M.S.; Kilpatrick, J.; Simpson, J.; et al. Regression of glioblastoma after chimeric antigen receptor t-cell therapy. N. Engl. J. Med. 2016, 375, 2561–2569. [Google Scholar] [CrossRef] [PubMed]
- Quattrocchi, K.B.; Miller, C.H.; Cush, S.; Bernard, S.A.; Dull, S.T.; Smith, M.; Gudeman, S.; Varia, M.A. Pilot study of local autologous tumor infiltrating lymphocytes for the treatment of recurrent malignant gliomas. J. Neurooncol. 1999, 45, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Meng, Q.; Bartek, J., Jr.; Poiret, T.; Persson, O.; Rane, L.; Rangelova, E.; Illies, C.; Peredo, I.H.; Luo, X.; et al. Tumor-infiltrating lymphocytes (tils) from patients with glioma. Oncoimmunology 2017, 6, e1252894. [Google Scholar] [CrossRef] [PubMed]
- Woroniecka, K.; Chongsathidkiet, P.; Rhodin, K.; Kemeny, H.; Dechant, C.; Farber, S.H.; Elsamadicy, A.A.; Cui, X.; Koyama, S.; Jackson, C.; et al. T-cell exhaustion signatures vary with tumor type and are severe in glioblastoma. Clin. Cancer Res. 2018, 24, 4175–4186. [Google Scholar] [CrossRef]
- Mohme, M.; Schliffke, S.; Maire, C.L.; Runger, A.; Glau, L.; Mende, K.C.; Matschke, J.; Gehbauer, C.; Akyuz, N.; Zapf, S.; et al. Immunophenotyping of newly diagnosed and recurrent glioblastoma defines distinct immune exhaustion profiles in peripheral and tumor-infiltrating lymphocytes. Clin. Cancer Res. 2018, 24, 4187–4200. [Google Scholar] [CrossRef]
- Hu, W.; Wang, G.; Huang, D.; Sui, M.; Xu, Y. Cancer immunotherapy based on natural killer cells: Current progress and new opportunities. Front. Immunol. 2019, 10, 1205. [Google Scholar] [CrossRef]
- Holdhoff, M.; Guner, G.; Rodriguez, F.J.; Hicks, J.L.; Zheng, Q.; Forman, M.S.; Ye, X.; Grossman, S.A.; Meeker, A.K.; Heaphy, C.M.; et al. Absence of cytomegalovirus in glioblastoma and other high-grade gliomas by real-time pcr, immunohistochemistry, and in situ hybridization. Clin. Cancer Res. 2017, 23, 3150–3157. [Google Scholar] [CrossRef]
- Reap, E.A.; Suryadevara, C.M.; Batich, K.A.; Sanchez-Perez, L.; Archer, G.E.; Schmittling, R.J.; Norberg, P.K.; Herndon, J.E., 2nd; Healy, P.; Congdon, K.L.; et al. Dendritic cells enhance polyfunctionality of adoptively transferred t cells that target cytomegalovirus in glioblastoma. Cancer Res. 2018, 78, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Cantalupo, P.G.; Katz, J.P.; Pipas, J.M. Viral sequences in human cancer. Virology 2018, 513, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Colli, L.M.; Machiela, M.J.; Myers, T.A.; Jessop, L.; Yu, K.; Chanock, S.J. Burden of nonsynonymous mutations among tcga cancers and candidate immune checkpoint inhibitor responses. Cancer Res. 2016, 76, 3767–3772. [Google Scholar] [CrossRef] [PubMed]
- Weenink, B.; Draaisma, K.; Ooi, H.Z.; Kros, J.M.; Sillevis Smitt, P.A.E.; Debets, R.; French, P.J. Low-grade glioma harbors few cd8 t cells, which is accompanied by decreased expression of chemo-attractants, not immunogenic antigens. Sci. Rep. 2019, 9, 14643. [Google Scholar] [CrossRef]
- Charoentong, P.; Finotello, F.; Angelova, M.; Mayer, C.; Efremova, M.; Rieder, D.; Hackl, H.; Trajanoski, Z. Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 2017, 18, 248–262. [Google Scholar] [CrossRef]
- Hilf, N.; Kuttruff-Coqui, S.; Frenzel, K.; Bukur, V.; Stevanovic, S.; Gouttefangeas, C.; Platten, M.; Tabatabai, G.; Dutoit, V.; van der Burg, S.H.; et al. Actively personalized vaccination trial for newly diagnosed glioblastoma. Nature 2019, 565, 240–245. [Google Scholar] [CrossRef]
- Keskin, D.B.; Anandappa, A.J.; Sun, J.; Tirosh, I.; Mathewson, N.D.; Li, S.; Oliveira, G.; Giobbie-Hurder, A.; Felt, K.; Gjini, E.; et al. Neoantigen vaccine generates intratumoral t cell responses in phase ib glioblastoma trial. Nature 2019, 565, 234–239. [Google Scholar] [CrossRef]
- Dutoit, V.; Migliorini, D.; Dietrich, P.Y.; Walker, P.R. Immunotherapy of malignant tumors in the brain: How different from other sites? Front. Oncol. 2016, 6, 256. [Google Scholar] [CrossRef]
- Shraibman, B.; Barnea, E.; Kadosh, D.M.; Haimovich, Y.; Slobodin, G.; Rosner, I.; Lopez-Larrea, C.; Hilf, N.; Kuttruff, S.; Song, C.; et al. Identification of tumor antigens among the hla peptidomes of glioblastoma tumors and plasma. Mol. Cell. Proteom. 2019, 18, 1255–1268. [Google Scholar] [CrossRef]
- Facoetti, A.; Nano, R.; Zelini, P.; Morbini, P.; Benericetti, E.; Ceroni, M.; Campoli, M.; Ferrone, S. Human leukocyte antigen and antigen processing machinery component defects in astrocytic tumors. Clin. Cancer Res. 2005, 11, 8304–8311. [Google Scholar] [CrossRef]
- Schumacher, T.; Bunse, L.; Pusch, S.; Sahm, F.; Wiestler, B.; Quandt, J.; Menn, O.; Osswald, M.; Oezen, I.; Ott, M.; et al. A vaccine targeting mutant idh1 induces antitumour immunity. Nature 2014, 512, 324–327. [Google Scholar] [CrossRef]
- Ratnam, N.M.; Gilbert, M.R.; Giles, A.J. Immunotherapy in cns cancers: The role of immune cell trafficking. Neuro. Oncol. 2019, 21, 37–46. [Google Scholar] [CrossRef]
- Yin, J.; Valin, K.L.; Dixon, M.L.; Leavenworth, J.W. The role of microglia and macrophages in cns homeostasis, autoimmunity, and cancer. J. Immunol. Res. 2017, 2017, 5150678. [Google Scholar] [CrossRef]
- Orrego, E.; Castaneda, C.A.; Castillo, M.; Bernabe, L.A.; Casavilca, S.; Chakravarti, A.; Meng, W.; Garcia-Corrochano, P.; Villa-Robles, M.R.; Zevallos, R.; et al. Distribution of tumor-infiltrating immune cells in glioblastoma. CNS Oncol. 2018, 7, CNS21. [Google Scholar] [CrossRef]
- Varn, F.S.; Wang, Y.; Mullins, D.W.; Fiering, S.; Cheng, C. Systematic pan-cancer analysis reveals immune cell interactions in the tumor microenvironment. Cancer Res. 2017, 77, 1271–1282. [Google Scholar] [CrossRef]
- Yang, I.; Tihan, T.; Han, S.J.; Wrensch, M.R.; Wiencke, J.; Sughrue, M.E.; Parsa, A.T. Cd8+ t-cell infiltrate in newly diagnosed glioblastoma is associated with long-term survival. J. Clin. Neurosci. 2010, 17, 1381–1385. [Google Scholar] [CrossRef]
- Mostafa, H.; Pala, A.; Hogel, J.; Hlavac, M.; Dietrich, E.; Westhoff, M.A.; Nonnenmacher, L.; Burster, T.; Georgieff, M.; Wirtz, C.R.; et al. Immune phenotypes predict survival in patients with glioblastoma multiforme. J. Hematol. Oncol. 2016, 9, 77. [Google Scholar] [CrossRef]
- Kmiecik, J.; Poli, A.; Brons, N.H.; Waha, A.; Eide, G.E.; Enger, P.O.; Zimmer, J.; Chekenya, M. Elevated cd3+ and cd8+ tumor-infiltrating immune cells correlate with prolonged survival in glioblastoma patients despite integrated immunosuppressive mechanisms in the tumor microenvironment and at the systemic level. J. Neuroimmunol. 2013, 264, 71–83. [Google Scholar] [CrossRef]
- Zhang, J.; Caruso, F.P.; Sa, J.K.; Justesen, S.; Nam, D.H.; Sims, P.; Ceccarelli, M.; Lasorella, A.; Iavarone, A. The combination of neoantigen quality and t lymphocyte infiltrates identifies glioblastomas with the longest survival. Commun. Biol. 2019, 2, 135. [Google Scholar] [CrossRef]
- Hussain, S.F.; Yang, D.; Suki, D.; Aldape, K.; Grimm, E.; Heimberger, A.B. The role of human glioma-infiltrating microglia/macrophages in mediating antitumor immune responses. Neuro. Oncol. 2006, 8, 261–279. [Google Scholar] [CrossRef]
- Platten, M.; Ochs, K.; Lemke, D.; Opitz, C.; Wick, W. Microenvironmental clues for glioma immunotherapy. Curr. Neurol. Neurosci. Rep. 2014, 14, 440. [Google Scholar] [CrossRef]
- Broekman, M.L.; Maas, S.L.N.; Abels, E.R.; Mempel, T.R.; Krichevsky, A.M.; Breakefield, X.O. Multidimensional communication in the microenvirons of glioblastoma. Nat. Rev. Neurol. 2018, 14, 482–495. [Google Scholar] [CrossRef]
- Quail, D.F.; Joyce, J.A. The microenvironmental landscape of brain tumors. Cancer Cell. 2017, 31, 326–341. [Google Scholar] [CrossRef]
- Hambardzumyan, D.; Gutmann, D.H.; Kettenmann, H. The role of microglia and macrophages in glioma maintenance and progression. Nat. Neurosci. 2016, 19, 20–27. [Google Scholar] [CrossRef]
- Platten, M.; Kretz, A.; Naumann, U.; Aulwurm, S.; Egashira, K.; Isenmann, S.; Weller, M. Monocyte chemoattractant protein-1 increases microglial infiltration and aggressiveness of gliomas. Ann. Neurol. 2003, 54, 388–392. [Google Scholar] [CrossRef]
- Kamran, N.; Kadiyala, P.; Saxena, M.; Candolfi, M.; Li, Y.; Moreno-Ayala, M.A.; Raja, N.; Shah, D.; Lowenstein, P.R.; Castro, M.G. Immunosuppressive myeloid cells’ blockade in the glioma microenvironment enhances the efficacy of immune-stimulatory gene therapy. Mol. Ther. 2017, 25, 232–248. [Google Scholar] [CrossRef]
- Jacobs, J.F.; Idema, A.J.; Bol, K.F.; Grotenhuis, J.A.; de Vries, I.J.; Wesseling, P.; Adema, G.J. Prognostic significance and mechanism of treg infiltration in human brain tumors. J. Neuroimmunol. 2010, 225, 195–199. [Google Scholar] [CrossRef]
- El Andaloussi, A.; Lesniak, M.S. An increase in cd4+cd25+foxp3+ regulatory t cells in tumor-infiltrating lymphocytes of human glioblastoma multiforme. Neuro. Oncol. 2006, 8, 234–243. [Google Scholar] [CrossRef]
- Crane, C.A.; Ahn, B.J.; Han, S.J.; Parsa, A.T. Soluble factors secreted by glioblastoma cell lines facilitate recruitment, survival, and expansion of regulatory t cells: Implications for immunotherapy. Neuro. Oncol. 2012, 14, 584–595. [Google Scholar] [CrossRef]
- Roy, L.O.; Poirier, M.B.; Fortin, D. Transforming growth factor-beta and its implication in the malignancy of gliomas. Target. Oncol. 2015, 10, 1–14. [Google Scholar] [CrossRef]
- Frei, K.; Gramatzki, D.; Tritschler, I.; Schroeder, J.J.; Espinoza, L.; Rushing, E.J.; Weller, M. Transforming growth factor-beta pathway activity in glioblastoma. Oncotarget 2015, 6, 5963–5977. [Google Scholar] [CrossRef]
- Lohr, J.; Ratliff, T.; Huppertz, A.; Ge, Y.; Dictus, C.; Ahmadi, R.; Grau, S.; Hiraoka, N.; Eckstein, V.; Ecker, R.C.; et al. Effector t-cell infiltration positively impacts survival of glioblastoma patients and is impaired by tumor-derived tgf-beta. Clin. Cancer Res. 2011, 17, 4296–4308. [Google Scholar] [CrossRef]
- Platten, M.; Weller, M.; Wick, W. Shaping the glioma immune microenvironment through tryptophan metabolism. CNS Oncol. 2012, 1, 99–106. [Google Scholar] [CrossRef]
- Neubert, N.J.; Schmittnaegel, M.; Bordry, N.; Nassiri, S.; Wald, N.; Martignier, C.; Tille, L.; Homicsko, K.; Damsky, W.; Maby-El Hajjami, H.; et al. T cell-induced csf1 promotes melanoma resistance to pd1 blockade. Sci. Transl. Med. 2018, 10, eaan3311. [Google Scholar] [CrossRef]
- Sampson, J.H.; Schmittling, R.J.; Archer, G.E.; Congdon, K.L.; Nair, S.K.; Reap, E.A.; Desjardins, A.; Friedman, A.H.; Friedman, H.S.; Herndon, J.E., 2nd; et al. A pilot study of il-2ralpha blockade during lymphopenia depletes regulatory t-cells and correlates with enhanced immunity in patients with glioblastoma. PLoS ONE 2012, 7, e31046. [Google Scholar] [CrossRef]
- Miska, J.; Rashidi, A.; Chang, A.L.; Muroski, M.E.; Han, Y.; Zhang, L.; Lesniak, M.S. Anti-gitr therapy promotes immunity against malignant glioma in a murine model. Cancer Immunol. Immunother. 2016, 65, 1555–1567. [Google Scholar] [CrossRef]
- Lu, L.; Xu, X.; Zhang, B.; Zhang, R.; Ji, H.; Wang, X. Combined pd-1 blockade and gitr triggering induce a potent antitumor immunity in murine cancer models and synergizes with chemotherapeutic drugs. J. Transl. Med. 2014, 12, 36. [Google Scholar] [CrossRef]
- Brandes, A.A.; Carpentier, A.F.; Kesari, S.; Sepulveda-Sanchez, J.M.; Wheeler, H.R.; Chinot, O.; Cher, L.; Steinbach, J.P.; Capper, D.; Specenier, P.; et al. A phase ii randomized study of galunisertib monotherapy or galunisertib plus lomustine compared with lomustine monotherapy in patients with recurrent glioblastoma. Neuro. Oncol. 2016, 18, 1146–1156. [Google Scholar] [CrossRef]
- Capper, D.; von Deimling, A.; Brandes, A.A.; Carpentier, A.F.; Kesari, S.; Sepulveda-Sanchez, J.M.; Wheeler, H.R.; Chinot, O.; Cher, L.; Steinbach, J.P.; et al. Biomarker and histopathology evaluation of patients with recurrent glioblastoma treated with galunisertib, lomustine, or the combination of galunisertib and lomustine. Int. J. Mol. Sci. 2017, 18, 995. [Google Scholar] [CrossRef]
- Ladomersky, E.; Zhai, L.; Lenzen, A.; Lauing, K.L.; Qian, J.; Scholtens, D.M.; Gritsina, G.; Sun, X.; Liu, Y.; Yu, F.; et al. Ido1 inhibition synergizes with radiation and pd-1 blockade to durably increase survival against advanced glioblastoma. Clin. Cancer Res. 2018, 24, 2559–2573. [Google Scholar] [CrossRef]
- Koks, C.A.; Garg, A.D.; Ehrhardt, M.; Riva, M.; Vandenberk, L.; Boon, L.; De Vleeschouwer, S.; Agostinis, P.; Graf, N.; Van Gool, S.W. Newcastle disease virotherapy induces long-term survival and tumor-specific immune memory in orthotopic glioma through the induction of immunogenic cell death. Int. J. Cancer 2015, 136, E313–E325. [Google Scholar] [CrossRef]
- Martikainen, M.; Essand, M. Virus-based immunotherapy of glioblastoma. Cancers 2019, 11, 186. [Google Scholar] [CrossRef]
- Lang, F.F.; Conrad, C.; Gomez-Manzano, C.; Yung, W.K.A.; Sawaya, R.; Weinberg, J.S.; Prabhu, S.S.; Rao, G.; Fuller, G.N.; Aldape, K.D.; et al. Phase i study of dnx-2401 (delta-24-rgd) oncolytic adenovirus: Replication and immunotherapeutic effects in recurrent malignant glioma. J. Clin. Oncol. 2018, 36, 1419–1427. [Google Scholar] [CrossRef]
- Desjardins, A.; Gromeier, M.; Herndon, J.E., 2nd; Beaubier, N.; Bolognesi, D.P.; Friedman, A.H.; Friedman, H.S.; McSherry, F.; Muscat, A.M.; Nair, S.; et al. Recurrent glioblastoma treated with recombinant poliovirus. N. Engl. J. Med. 2018, 379, 150–161. [Google Scholar] [CrossRef]
- Zeng, J.; See, A.P.; Phallen, J.; Jackson, C.M.; Belcaid, Z.; Ruzevick, J.; Durham, N.; Meyer, C.; Harris, T.J.; Albesiano, E.; et al. Anti-pd-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 343–349. [Google Scholar] [CrossRef]
- Formenti, S.C.; Demaria, S. Combining radiotherapy and cancer immunotherapy: A paradigm shift. J. Natl. Cancer Inst. 2013, 105, 256–265. [Google Scholar] [CrossRef]
- Derer, A.; Spiljar, M.; Baumler, M.; Hecht, M.; Fietkau, R.; Frey, B.; Gaipl, U.S. Chemoradiation increases pd-l1 expression in certain melanoma and glioblastoma cells. Front. Immunol. 2016, 7, 610. [Google Scholar] [CrossRef]
- Qiu, J.; Shi, Z.; Jiang, J. Cyclooxygenase-2 in glioblastoma multiforme. Drug. Discov. Today 2017, 22, 148–156. [Google Scholar] [CrossRef]
- Kleijn, A.; van den Bossche, W.; Haefner, E.S.; Belcaid, Z.; Burghoorn-Maas, C.; Kloezeman, J.J.; Pas, S.D.; Leenstra, S.; Debets, R.; de Vrij, J.; et al. The sequence of delta24-rgd and tmz administration in malignant glioma affects the role of cd8(+)t cell anti-tumor activity. Mol. Ther. Oncolytics 2017, 5, 11–19. [Google Scholar] [CrossRef]
- Pellegatta, S.; Eoli, M.; Cuccarini, V.; Anghileri, E.; Pollo, B.; Pessina, S.; Frigerio, S.; Servida, M.; Cuppini, L.; Antozzi, C.; et al. Survival gain in glioblastoma patients treated with dendritic cell immunotherapy is associated with increased nk but not cd8(+) t cell activation in the presence of adjuvant temozolomide. Oncoimmunology 2018, 7, e1412901. [Google Scholar] [CrossRef]
- Petersen, C.T.; Krenciute, G. Next generation car t cells for the immunotherapy of high-grade glioma. Front. Oncol. 2019, 9, 69. [Google Scholar] [CrossRef]
- Krenciute, G.; Prinzing, B.L.; Yi, Z.; Wu, M.F.; Liu, H.; Dotti, G.; Balyasnikova, I.V.; Gottschalk, S. Transgenic expression of il15 improves antiglioma activity of il13ralpha2-car t cells but results in antigen loss variants. Cancer Immunol. Res. 2017, 5, 571–581. [Google Scholar] [CrossRef]
- Jung, I.Y.; Kim, Y.Y.; Yu, H.S.; Lee, M.; Kim, S.; Lee, J. Crispr/cas9-mediated knockout of dgk improves antitumor activities of human t cells. Cancer Res. 2018, 78, 4692–4703. [Google Scholar] [CrossRef]
- Bielamowicz, K.; Fousek, K.; Byrd, T.T.; Samaha, H.; Mukherjee, M.; Aware, N.; Wu, M.F.; Orange, J.S.; Sumazin, P.; Man, T.K.; et al. Trivalent car t cells overcome interpatient antigenic variability in glioblastoma. Neuro. Oncol. 2018, 20, 506–518. [Google Scholar] [CrossRef]
- Pellegatta, S.; Savoldo, B.; Di Ianni, N.; Corbetta, C.; Chen, Y.; Patane, M.; Sun, C.; Pollo, B.; Ferrone, S.; DiMeco, F.; et al. Constitutive and tnfalpha-inducible expression of chondroitin sulfate proteoglycan 4 in glioblastoma and neurospheres: Implications for car-t cell therapy. Sci. Transl. Med. 2018, 10, eaao2731. [Google Scholar] [CrossRef]
- Tang, X.; Zhao, S.; Zhang, Y.; Wang, Y.; Zhang, Z.; Yang, M.; Zhu, Y.; Zhang, G.; Guo, G.; Tong, A.; et al. B7-h3 as a novel car-t therapeutic target for glioblastoma. Mol. Ther. Oncolytics 2019, 14, 279–287. [Google Scholar] [CrossRef]
- Wang, D.; Starr, R.; Chang, W.; Aguilar, B.; Alizadeh, D.; Wright, S.L.; Yang, X.; Brito, A.; Sarkissian, A.; Ostberg, J.R.; et al. Chlorotoxin redirects chimeric antigen receptor t cells for specific and effective targeting of glioblastoma. bioRxiv 2020. [Google Scholar] [CrossRef]
- Yin, Y.; Boesteanu, A.C.; Binder, Z.A.; Xu, C.; Reid, R.A.; Rodriguez, J.L.; Cook, D.R.; Thokala, R.; Blouch, K.; McGettigan-Croce, B.; et al. Checkpoint blockade reverses anergy in il-13ralpha2 humanized scfv-based car t cells to treat murine and canine gliomas. Mol. Ther. Oncolytics 2018, 11, 20–38. [Google Scholar] [CrossRef]
- Banks, W.A. Characteristics of compounds that cross the blood-brain barrier. BMC Neurol. 2009, 9 (Suppl. 1), S3. [Google Scholar] [CrossRef]
- Tawbi, H.A.; Forsyth, P.A.; Algazi, A.; Hamid, O.; Hodi, F.S.; Moschos, S.J.; Khushalani, N.I.; Lewis, K.; Lao, C.D.; Postow, M.A.; et al. Combined nivolumab and ipilimumab in melanoma metastatic to the brain. N. Engl. J. Med. 2018, 379, 722–730. [Google Scholar] [CrossRef]
- Bailly, C.; Vidal, A.; Bonnemaire, C.; Kraeber-Bodere, F.; Cherel, M.; Pallardy, A.; Rousseau, C.; Garcion, E.; Lacoeuille, F.; Hindre, F.; et al. Potential for nuclear medicine therapy for glioblastoma treatment. Front. Pharmacol. 2019, 10, 772. [Google Scholar] [CrossRef] [PubMed]
| Clinical Trial | Phase | Target | Treatment | Control | Indication | # Patients | Endpoint | Outcome |
|---|---|---|---|---|---|---|---|---|
| CheckMate 143 NCT02017717 PMC5463583 | III | PD-1 | Nivolumab | Bevacizumab | R GB | Each arm: ~185 | OS | No impact |
| CheckMate 498 NCT02617589 | III | PD-1 | RT, nivolumab | SOC | P GB MGMT-unmeth. | Each arm: ~275 | OS | No impact |
| CheckMate 548 NCT02667587 | II | PD-1 | SOC, nivolumab | SOC, placebo | P GB MGMT-meth. | Each arm: ~160 | PFS | No impact |
| Neo-nivo NCT02550249 30742120 | II | PD-1 | Nivolumab (neo-adjuvant), (S)S, nivolumab (adjuvant) | None | P GB R GB | P: 3 R: 27 | (OS) | 7.3 mo |
| NCT02337491 | II | PD-1 | Pembrolizumab, bevacizumab | Pembrolizumab | R GB | Treatment: 50 Control: 30 | (OS) | 8.8 mo vs. 10.3 mo |
| NCT02337686 | II | PD-1 | SS, pembrolizumab | None | R GB | 15 | PFS6 | 53% |
| NCT02852655 30742122 | II | PD-1 | Pembrolizumab (neo-adjuvant), SS, pembrolizumab (adjuvant) | SS, pembrolizumab (adjuvant) | R HGG | Treatment: 16 Control: 19 | (OS) | 13.7 mo vs. 7.5 mo |
| NCT03291314 | II | PD-L1 | Avelumab, axitinib | None | R GB | 32 | PFS6 | 18% |
| SEJ NCT03047473 | II | PD-L1 | SOC, avelumab | None | P GB | 24 | (PFS) | 11.9 mo |
| NCT02336165 | II | PD-L1 | SOC, durvalumab | Historical | P GB MGMT-unmeth. | 40 | OS12 | 60% vs. 50% |
| Clinical Trial | Phase | Target | Treatment | Control | Indication | # Patients | Endpoint | Outcome |
|---|---|---|---|---|---|---|---|---|
| HSPPC-96 NCT00905060 | II | Autologous peptides | SOC, PEP | None | P GB | 46 | OS | 23.8 mo |
| HSPPC-96 NCT00293423 24335700 | II | Autologous peptides | SS, PEP | None | R GB | 41 | OS6 | 90.2% |
| HSPPC-96 NCT01814813 | II | Autologous peptides | PEP Bevacizumab | Bevacizumab | R GB | 30 | OS | No impact |
| ACT-IV NCT01480479 28844499 | III | EGFRvIII | SOC, PEP | SOC, KLH | P GB EGFRvIII+ | 371 | OS | No impact |
| ACT-III 25586468 | II | EGFRvIII | SOC, PEP | Historical | P GB EGFRvIII+ | 65 | PFS5.5 | 66% vs. 45% |
| ACT-II 21149254 | II | EGFRvIII | SOC, PEP | Historical | P GB EGFRvIII+ | 22 | (OS) | 23.6 vs. 15.0 mo |
| ReACT NCT01498328 | II | EGFRvIII | PEP Bevacizumab | KLH Bevacizumab | R GB EFGRvIII+ | 33 | PFS6 | 27% vs. 11% |
| ACTIVATe NCT00643097 20921459 | II | EGFRvIII | SOC, PEP | Historical | P GB EGFRvIII+ | 18 | PFS6 | 94% vs. 59% |
| ITK-1 UMIN000006970 30500939 | III | Multiple TAA | PEP | Placebo | R GB HLA-A24+ | Treatment: 58 Control: 30 | OS | No impact |
| SL-701 NCT02078648 | II | Multiple TAA | PEP Bevacizumab | None | R GB HLA-A2+ | 74 | OS12 | 43% |
| IMA-950 NCT01920191 30753611 | I/II | Multiple TAA | SOC, PEP | None | P GB HLA-A2+ | 16 | (OS) | 19 mo |
| SurVaxM~ NCT02455557 | II | Survivin | SOC, PEP | None | P GB HLA-A2, -A3, -A11 and -A24+ | 55 | OS12 | 70.8% |
| Clinical Trial | Phase | Loading Material for DCs | Treatment | Control | Indication | # Patients | Endpoint | Outcome |
|---|---|---|---|---|---|---|---|---|
| NCT01567202 30159779 | II | GSC antigens | P: SOC, DC R: SS, RT/CT, DC | P: SOC, placebo R: SS, RT/CT, placebo | P GB R GB | Treatment: 22 Control: 21 | PFS | 7.7 mo vs. 6.9 mo |
| NCT02772094 21715171 | I/II | Irradiated tumor cells | SOC/SS, DC | None | P GB | 16 | OS | 17 mo |
| ICT-107 NCT01280552 31320597 | II | Multiple TAA | SOC, DC | SOC, placebo | P GB HLA-A1+ and/or -A2+ | Treatment: 81 Control: 43 | OS | 17.0 vs. 15.0 mo |
| DCVax-L NCT00045968 29843811 | III | Tumor lysate | SOC, DC | SOC, placebo | P GB | 331 | (OS) | 23.1 mo |
| GBM-Vax NCT01213407 30301187 | II | Tumor lysate | SOC, DC | SOC | P GB | 34 | PFS12 | No impact |
| NCT03879512 30054667 | I/II | Tumor lysate | SS, Cyclophosphamide, DC | None | R GB pediatric R GB adult | Pediatric: 6 Adult: 5 | OS6 | 100% |
| DEND/GM NCT01006044 28499389 | II | Tumor lysate | SOC, DC | None | P GB | 31 | PFS | 12.7 mo |
| NCT00576537 18632651 | I/II | Tumor lysate | SOC/SS, DC | None | P GB R GB | P: 11 R: 23 | (OS) | Vaccine responders: 21 mo Nonresponders 14 mo |
| NCT00323115 21499132 | II | Tumor lysate | SOC, DC | None | P GB | 10 | (OS) | 28 mo |
| DENDR2 NCT02820584 | I/II | Tumor lysate | (1) SS, TMZ, DC (2) SS, TTX, DC | None | R GB | (1) 12 (2) 8 | (OS) | (1) 7.4 mo (2) 9.2 mo |
| DENDR1 29632727 | I/II | Tumor lysate | SOC, TMZ, DC | None | P GB | 24 | PFS12 | 41% |
| DC-CAST-GBM NCT00846456 23817721 | I/II | Tumor stem cell mRNA | SOC, DC | None | P GB | 7 | (OS) | 25 mo |
| Clinical Trial | Phase | CAR Generation | Target | Other Treatment | Indication | # Patients | OR (%) | CR (%) |
|---|---|---|---|---|---|---|---|---|
| NCT00730613 26059190 | I | First CD3z | IL13Rα2 | SS | R GB | 3 | 0/3 (0) | 0/3 (0) |
| NCT02208362 28029927 | I | Second 41BB-CD3z | IL13Rα2 | SS | R GB IL13Rα2+ | 1 | 1/1 (100) | 1/1 (100) |
| NCT01109095 28426845 | I | Second CD28-CD3z | HER2 CMV pp65 | SS | R GB HER2+ | 17 | 1/17 (6) | 0/17 (0) |
| NCT02209376 28724573 | I | Second 41BB-CD3z | EGFRvIII | SS | R GB EGFRvIII+ | 10 | 0/10 (0) | 0/10 (0) |
| NCT01454596 30882547 | I | Third CD28-41BB-CD3z | EGFRvIII | Cyclophosphamide, Fludarabine, IL-2 | R GB EGFRvIII+ | 18 | 0/18 (0) | 0/18 (0) |
| Clinical Trial | Phase | Lymphocytes | Other Treatment | Indication | # Patients | OR (%) | CR (%) | OS |
|---|---|---|---|---|---|---|---|---|
| 9390198 | I | Alloreactive CTLs | SS, IL-2 | R A, O grade III-IV | 5 | 0/5 (0) | 0/5 (0) | - |
| 24795429 | I | Autologous CMV-specific T cells | CT | R GB CMV seropositive | 11 | 0/11 (0) | 0/11 (0) | - |
| NCT01588769 29511178 | I | Autologous CTLs and NK cells | None | R GB | 10 | 3/10 (30) | 0/10 (0) | - |
| 9647171 | I | Autologous T lymphocytes | Irradiated tumor cell vaccination | R A grade III-IV | 10 | 3/10 (30) | 0/10 (0) | - |
| NCT00004024 16817692 | II | Autologous T lymphocytes | Irradiated tumor cell vaccination | R A or O grade II-IV | 19 | 8/19 (42) | 1/19 (5) | - |
| 10778730 | I | Autologous TILs | SS, IL-2 | R GB | 6 | 3/6 (50) | 1/6 (17) | - |
| NCT00331526 19816190 | II | Autologous LAK cells | SOC, IL-2 | P GB | 33 | NR | NR | 20.5 mo |
| NCT00003067 8625188 | I | Autologous LAK cells | SS, IL-2 | R GB R A grade III | 19 | 4/19 (21) | 2/19 (11) | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weenink, B.; French, P.J.; Sillevis Smitt, P.A.E.; Debets, R.; Geurts, M. Immunotherapy in Glioblastoma: Current Shortcomings and Future Perspectives. Cancers 2020, 12, 751. https://doi.org/10.3390/cancers12030751
Weenink B, French PJ, Sillevis Smitt PAE, Debets R, Geurts M. Immunotherapy in Glioblastoma: Current Shortcomings and Future Perspectives. Cancers. 2020; 12(3):751. https://doi.org/10.3390/cancers12030751
Chicago/Turabian StyleWeenink, Bas, Pim J. French, Peter A.E. Sillevis Smitt, Reno Debets, and Marjolein Geurts. 2020. "Immunotherapy in Glioblastoma: Current Shortcomings and Future Perspectives" Cancers 12, no. 3: 751. https://doi.org/10.3390/cancers12030751
APA StyleWeenink, B., French, P. J., Sillevis Smitt, P. A. E., Debets, R., & Geurts, M. (2020). Immunotherapy in Glioblastoma: Current Shortcomings and Future Perspectives. Cancers, 12(3), 751. https://doi.org/10.3390/cancers12030751





