Cardiovascular Health during and after Cancer Therapy
Abstract
Simple Summary
Abstract
1. Introduction
2. Cardiovascular Health Risks with Specific Cancer Therapies
2.1. Radiation
2.2. Anthracyclines
2.3. HER2-Directed Therapies
2.4. Cyclophosphamide
2.5. Fluoropyrimidines
2.6. Platinums
2.7. Tyrosine Kinase Inhibitors and Proteasome Inhibitors
2.8. Immune Checkpoint Inhibitors
2.9. CAR-T Cell Therapy
2.10. Special Considerations before, during, and after Stem Cell or Bone Marrow Transplants
3. Primary and Secondary Prevention after Cancer Therapy
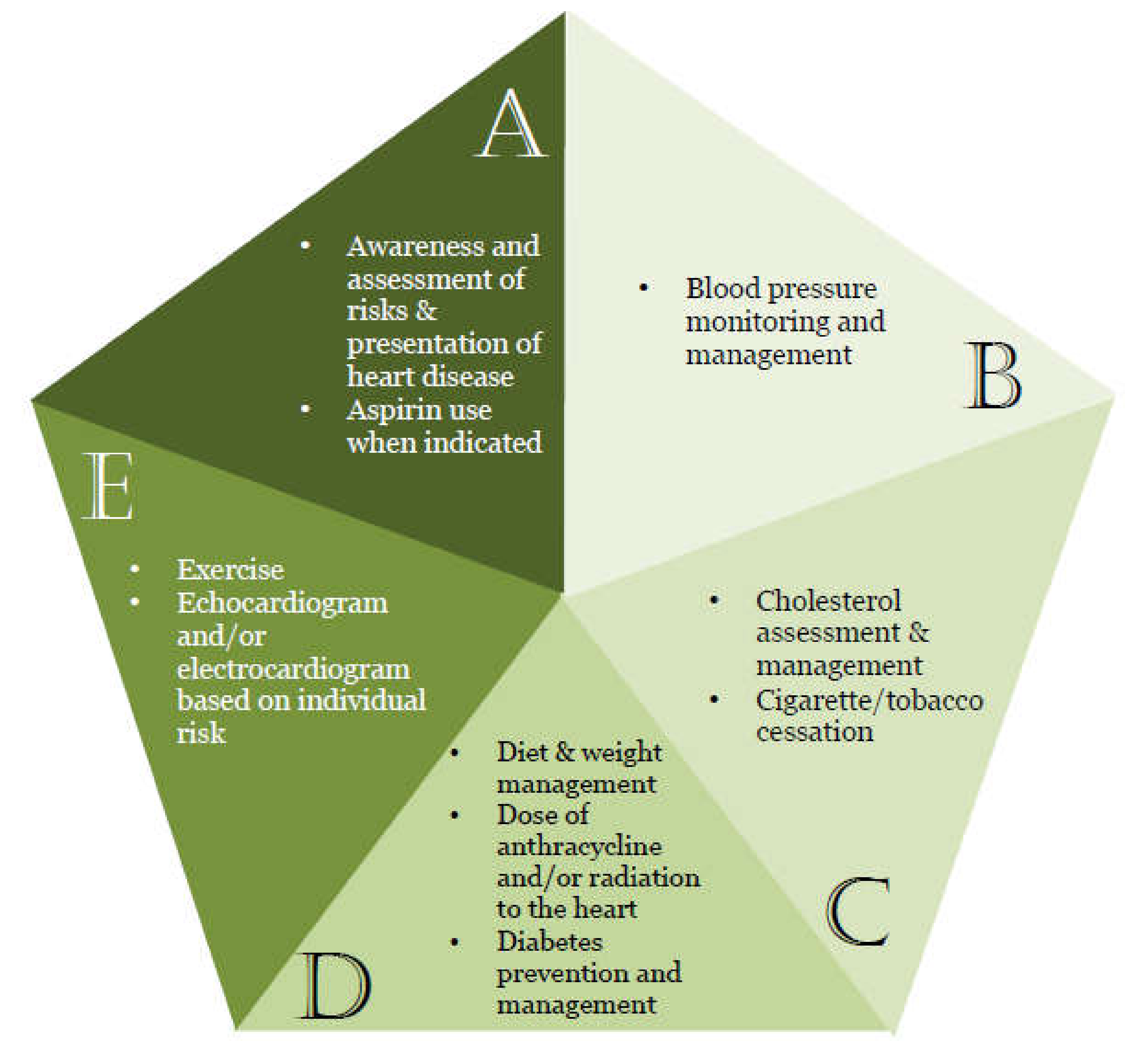
3.1. ABCDE
3.2. Monitoring Cardiovascular Health in Survivors
4. Cardiovascular Considerations at Extremes of Age
5. Next Steps
Author Contributions
Funding
Conflicts of Interest
References
- Lenneman, C.G.; Sawyer, D.B. Cardio-Oncology: An Update on Cardiotoxicity of Cancer-Related Treatment. Circ. Res. 2016, 118, 1008–1020. [Google Scholar] [CrossRef]
- Zamorano, J.L.; Lancellotti, P.; Rodriguez Munoz, D.; Aboyans, V.; Asteggiano, R.; Galderisi, M.; Habib, G.; Lenihan, D.J.; Lip, G.Y.H.; Lyon, A.R.; et al. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: The Task Force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur. Heart J. 2016, 37, 2768–2801. [Google Scholar]
- Roychoudhuri, R.; Robinson, D.; Putcha, V.; Cuzick, J.; Darby, S.; Moller, H. Increased cardiovascular mortality more than fifteen years after radiotherapy for breast cancer: A population-based study. BMC Cancer 2007, 7, 9. [Google Scholar] [CrossRef]
- Van Nimwegen, F.A.; Schaapveld, M.; Janus, C.P.; Krol, A.D.; Petersen, E.J.; Raemaekers, J.M.; Kok, W.E.; Aleman, B.M.; Van Leeuwen, F.E. Cardiovascular disease after Hodgkin lymphoma treatment: 40-year disease risk. JAMA Intern Med. 2015, 175, 1007–1017. [Google Scholar] [CrossRef]
- Stone, C.R.; Mickle, A.T.; Boyne, D.J.; Mohamed, A.; Rabi, D.M.; Brenner, D.R.; Friedenreich, C.M. Treatment for lymphoma and late cardiovascular disease risk: A systematic review and meta-analysis. Health Sci. Rep. 2019, 2, e135. [Google Scholar] [CrossRef] [PubMed]
- Borovac, J.A.; Kwok, C.S.; Iliescu, C.; Lee, H.J.; Kim, P.Y.; Palaskas, N.; Zaman, A.; Butler, R.; Lopez-Mattei, J.; Mamas, M.A. Percutaneous Coronary Intervention and Outcomes in Patients With Lymphoma in the United States (Nationwide Inpatient Sample [NIS] Analysis). Am. J. Cardiol. 2019, 124, 1190–1197. [Google Scholar] [CrossRef] [PubMed]
- Darby, S.; Ewertz, M.; McGale, P.; Bennet, A.M.; Blom-Goldman, U.; Brønnum, D.; Correa, C.; Cutter, D.; Gagliardi, G.; Gigante, B.; et al. Risk of Ischemic Heart Disease in Women after Radiotherapy for Breast Cancer. N. Engl. J. Med. 2013, 368, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Lipshultz, S.E.; Adams, M.J.; Colan, S.D.; Constine, L.S.; Herman, E.H.; Hsu, D.T.; Hudson, M.M.; Kremer, L.C.; Landy, D.C.; Miller, T.L.; et al. Long-term cardiovascular toxicity in children, adolescents, and young adults who receive cancer therapy: Pathophysiology, course, monitoring, management, prevention, and research directions: A scientific statement from the American Heart Association. Circulation 2013, 128, 1927–1995. [Google Scholar] [CrossRef]
- Wu, S.-Z.; Tao, L.-Y.; Wang, J.-N.; Xu, Z.-Q.; Wang, J.; Xue, Y.-J.; Huang, K.-Y.; Lin, J.-F.; Li, L.; Ji, K. Amifostine Pretreatment Attenuates Myocardial Ischemia/Reperfusion Injury by Inhibiting Apoptosis and Oxidative Stress. Oxidative Med. Cell. Longev. 2017, 2017, 1–12. [Google Scholar] [CrossRef]
- Henriksen, P.A. Anthracycline cardiotoxicity: An update on mechanisms, monitoring and prevention. Heart 2018, 104, 971–977. [Google Scholar] [CrossRef]
- Volkova, M. Anthracycline Cardiotoxicity: Prevalence, Pathogenesis and Treatment. Curr. Cardiol. Rev. 2012, 7, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Bristow, M.R.; Thompson, P.D.; Martin, R.P.; Mason, J.W.; Billingham, M.E.; Harrison, D.C. Early anthracycline cardiotoxicity. Am. J. Med. 1978, 65, 823–832. [Google Scholar] [CrossRef]
- Felker, G.M.; Thompson, R.E.; Hare, J.M.; Hruban, R.H.; Clemetson, D.E.; Howard, D.L.; Baughman, K.L.; Kasper, E.K. Underlying Causes and Long-Term Survival in Patients with Initially Unexplained Cardiomyopathy. N. Engl. J. Med. 2000, 342, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Fornaro, A.; Olivotto, I.; Rigacci, L.; Ciaccheri, M.; Tomberli, B.; Poggesi, C.; Coppini, R.; Girolami, F.; Mazzarotto, F.; Chiostri, M.; et al. Comparison of long-term outcome in anthracycline-related versus idiopathic dilated cardiomyopathy: A single centre experience. Eur. J. Hear. Fail. 2017, 20, 898–906. [Google Scholar] [CrossRef]
- Mazur, M.; Wang, F.; Hodge, D.O.; Siontis, B.L.; Beinborn, D.S.; Villarraga, H.R.; Lerman, L.O.; Friedman, P.A.; Herrmann, J. Burden of Cardiac Arrhythmias in Patients With Anthracycline-Related Cardiomyopathy. JACC Clin. Electrophysiol. 2017, 3, 139–150. [Google Scholar] [CrossRef]
- Barbar, T.; Mahmood, S.S.; Liu, J.E. Cardiomyopathy Prevention in Cancer Patients. Cardiol. Clin. 2019, 37, 441–447. [Google Scholar] [CrossRef]
- Lao, J.; Madani, J.; Puértolas, T.; Lvarez, M.; Hernández, A.; Pazo-Cid, R.; Artal, Á.; Torres, A.A. Liposomal Doxorubicin in the Treatment of Breast Cancer Patients: A Review. J. Drug Deliv. 2013, 2013, 1–12. [Google Scholar] [CrossRef]
- Xing, M.; Yan, F.; Yu, S.; Shen, P. Efficacy and Cardiotoxicity of Liposomal Doxorubicin-Based Chemotherapy in Advanced Breast Cancer: A Meta-Analysis of Ten Randomized Controlled Trials. PLoS ONE 2015, 10, e0133569. [Google Scholar] [CrossRef]
- Brown, S.A.; Okwuosa, T.M.; Barac, A.; Volgma, A.S. The Role of Angiotensin-Converting Enzyme Inhibitors and beta-Blockers in Primary Prevention of Cardiac Dysfunction in Breast Cancer Patients. J. Am. Heart Assoc. 2020, 9, e015327. [Google Scholar] [CrossRef]
- Cardinale, D.; Colombo, A.; Sandri, M.T.; LaMantia, G.; Colombo, N.; Civelli, M.; Martinelli, G.; Veglia, F.; Fiorentini, C.; Cipolla, C.M. Prevention of High-Dose Chemotherapy–Induced Cardiotoxicity in High-Risk Patients by Angiotensin-Converting Enzyme Inhibition. Circulation 2006, 114, 2474–2481. [Google Scholar] [CrossRef]
- Gulati, G.; Heck, S.L.; Ree, A.H.; Hoffmann, P.; Schulz-Menger, J.; Fagerland, M.W.; Gravdehaug, B.; von Knobelsdorff-Brenkenhoff, F.; Bratland, Å.; Storås, T.H.; et al. Prevention of cardiac dysfunction during adjuvant breast cancer therapy (PRADA): A 2 × 2 factorial, randomized, placebo-controlled, double-blind clinical trial of candesartan and metoprolol. Eur. Heart J. 2016, 37, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Guglin, M.; Krischer, J.; Tamura, R.; Fink, A.; Bello-Matricaria, L.; McCaskill-Stevens, W.; Munster, P.N. Randomized Trial of Lisinopril Versus Carvedilol to Prevent Trastuzumab Cardiotoxicity in Patients With Breast Cancer. J. Am. Coll. Cardiol. 2019, 73, 2859–2868. [Google Scholar] [CrossRef] [PubMed]
- Cooke, T. What is HER2? Eur. J. Oncol. Nurs. 2000, 4, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Meric-Bernstam, F.; Hurwitz, H.; Raghav, K.P.S.; McWilliams, R.R.; Fakih, M.; VanderWalde, A.; Swanton, C.; Kurzrock, R.; Burris, H.; Sweeney, C.; et al. Pertuzumab plus trastuzumab for HER2-amplified metastatic colorectal cancer (MyPathway): An updated report from a multicentre, open-label, phase 2a, multiple basket study. Lancet Oncol. 2019, 20, 518–530. [Google Scholar] [CrossRef]
- Nemeth, B.T.; Varga, Z.V.; Wu, W.J.; Pacher, P. Trastuzumab cardiotoxicity: From clinical trials to experimental studies. Br. J. Pharmacol. 2016, 174, 3727–3748. [Google Scholar] [CrossRef]
- Ewer, M.S.; Lippman, S.M. Type II Chemotherapy-Related Cardiac Dysfunction: Time to Recognize a New Entity. J. Clin. Oncol. 2005, 23, 2900–2902. [Google Scholar] [CrossRef]
- Dang, C.; Yu, A.F.; Jones, L.W.; Liu, J.; Steingart, R.M.; Argolo, D.F.; Norton, L.; Hudis, C.A. Cardiac Surveillance Guidelines for Trastuzumab-Containing Therapy in Early-Stage Breast Cancer: Getting to the Heart of the Matter. J. Clin. Oncol. 2016, 34, 1030–1033. [Google Scholar] [CrossRef]
- Nowsheen, S.; Aziz, K.; Park, J.Y.; Lerman, A.; Villarraga, H.R.; Ruddy, K.J.; Herrmann, J. Trastuzumab in Female Breast Cancer Patients With Reduced Left Ventricular Ejection Fraction. J. Am. Hear. Assoc. 2018, 7, e008637. [Google Scholar] [CrossRef]
- Lynce, F.; Barac, A.; Geng, X.; Dang, C.; Yu, A.F.; Smith, K.L.; Gallagher, C.; Pohlmann, P.R.; Nunes, R.; Herbolsheimer, P.; et al. Prospective evaluation of the cardiac safety of HER2-targeted therapies in patients with HER2-positive breast cancer and compromised heart function: The SAFE-HEaRt study. Breast Cancer Res. Treat. 2019, 175, 595–603. [Google Scholar] [CrossRef]
- Leong, D.P.; Cosman, T.; Alhussein, M.M.; Tyagi, N.K.; Karampatos, S.; Barron, C.C.; Wright, D.; Tandon, V.; Magloire, P.; Joseph, P.; et al. Safety of Continuing Trastuzumab Despite Mild Cardiotoxicity. JACC CardioOncology 2019, 1, 1–10. [Google Scholar] [CrossRef]
- Pituskin, E.; Mackey, J.R.; Koshman, S.; Jassal, D.; Pitz, M.; Haykowsky, M.J.; Pagano, J.J.; Chow, K.; Thompson, R.B.; Vos, L.; et al. Multidisciplinary Approach to Novel Therapies in Cardio-Oncology Research (MANTICORE 101–Breast): A Randomized Trial for the Prevention of Trastuzumab-Associated Cardiotoxicity. J. Clin. Oncol. 2017, 35, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, E.K.; Kokkinidis, D.G.; Kampaktsis, P.N.; Amir, E.A.; Marwick, T.H.; Gupta, D.; Thavendiranathan, P. Assessment of Prognostic Value of Left Ventricular Global Longitudinal Strain for Early Prediction of Chemotherapy-Induced Cardiotoxicity: A Systematic Review and Meta-analysis. JAMA Cardiol. 2019, 4, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Charbonnel, C.; Convers-Domart, R.; Rigaudeau, S.; Taksin, A.L.; Baron, N.; Lambert, J.; Ghez, S.; Georges, J.-L.; Farhat, H.; Lambert, J.; et al. Assessment of global longitudinal strain at low-dose anthracycline-based chemotherapy, for the prediction of subsequent cardiotoxicity. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Perez, E.A.; Koehler, M.; Byrne, J.; Preston, A.J.; Rappold, E.; Ewer, M.S. Cardiac safety of lapatinib: Pooled analysis of 3689 patients enrolled in clinical trials. Mayo Clin. Proc. 2008, 83, 679–686. [Google Scholar] [CrossRef]
- Pondé, N.; Ameye, L.; Lambertini, M.; Paesmans, M.; Piccart, M.; De Azambuja, E. Trastuzumab emtansine (T-DM1)-associated cardiotoxicity: Pooled analysis in advanced HER2-positive breast cancer. Eur. J. Cancer 2020, 126, 65–73. [Google Scholar] [CrossRef]
- Swain, S.M.; Ewer, M.S.; Cortés, J.; Amadori, D.; Miles, D.; Knott, A.; Clark, E.; Benyunes, M.C.; Ross, G.; Baselga, J. Cardiac Tolerability of Pertuzumab Plus Trastuzumab Plus Docetaxel in Patients With HER2-Positive Metastatic Breast Cancer in CLEOPATRA: A Randomized, Double-Blind, Placebo-Controlled Phase III Study. Oncologist 2013, 18, 257–264. [Google Scholar] [CrossRef]
- AstraZeneca, ENHERTU® (fam-trastuzumab deruxtecan-nxki) for Injection, for Intravenous Use: HIGHLIGHTS OF PRESCRIBING INFORMATION. Initial U.S. Approval: 2019. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/761139s000lbl.pdf (accessed on 15 November 2020).
- Seagen Inc. Highlights of Prescribing Information These Highlights Do Not Include All the Information Needed to use TUKYSA Safely and Effectively. See Full Prescribing Information for TUKYSA. 2020. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/213411s000lbl.pdf (accessed on 15 November 2020).
- GlaxoSmithKline, TYKERB (lapatinib) Tablets; HIGHLIGHTS OF PRESCRIBING INFORMATION. Initial U.S. Approval: 2007. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/022059s3s6lbl.pdf (accessed on 15 November 2020).
- Genetech Inc. KADCYLA® (ado-trastuzumab emtansine) for Injection, for Intravenous Use: HIGHLIGHTS OF PRESCRIBING INFORMATION. Initial U.S. Approval: 2013. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/125427s105lbl.pdf (accessed on 15 November 2020).
- Genetech, I. PERJETATM (pertuzumab) Injection, for Intravenous Use: HIGHLIGHTS OF PRESCRIBING INFORMATION. Initial U.S. Approval: 2012. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/125409lbl.pdf (accessed on 15 November 2020).
- Valachis, A.; Nearchou, A.; Polyzos, N.P.; Lind, P. Cardiac toxicity in breast cancer patients treated with dual HER2 blockade. Int. J. Cancer 2013, 133, 2245–2252. [Google Scholar] [CrossRef]
- Yeh, E.T.; Tong, A.T.; Lenihan, D.J.; Yusuf, S.W.; Swafford, J.; Champion, C.; Durand, J.B.; Gibbs, H.; Zafarmand, A.A.; Ewer, M.S. Cardiovascular complications of cancer therapy: Diagnosis, pathogenesis, and management. Circulation 2004, 109, 3122–3131. [Google Scholar] [CrossRef]
- Atalay, F.; Gulmez, O.; Ugurlu, A.O. Cardiotoxicity following cyclophosphamidetherapy: A case report. J. Med. Case Rep. 2014, 8, 252. [Google Scholar] [CrossRef]
- Anand, A.J. Fluorouracil Cardiotoxicity. Ann. Pharmacother. 1994, 28, 374–378. [Google Scholar] [CrossRef]
- Ang, C.; Kornbluth, M.; Thirlwell, M.; Rajan, R. Capecitabine-induced cardiotoxicity: Case report and review of the literature. Curr. Oncol. 2010, 17, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Kanduri, J.; Asnani, A. Fluoropyrimidine Cardiotoxicity. American College of Cardiology. 2019. Available online: https://www.acc.org/latest-in-cardiology/articles/2019/04/05/07/34/fluoropyrimidine-cardiotoxicity (accessed on 1 December 2020).
- Yuan, C.; Parekh, H.; Allegra, C.; George, T.J.; Starr, J.S. 5-FU induced cardiotoxicity: Case series and review of the literature. Cardio-Oncology 2019, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sara, J.D.; Kaur, J.; Khodadadi, R.; Rehman, M.; Lobo, R.; Chakrabarti, S.; Herrmann, J.; Lerman, A.; Grothey, A. 5-fluorouracil and cardiotoxicity: A review. Ther. Adv. Med. Oncol. 2018, 10, 1758835918780140. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; Hoff, P.M.; Blum, J.L.; Abt, M.; Osterwalder, B. Incidence of cardiotoxicity with the oral fluoropyrimidine capecitabine is typical of that reported with 5-fluorouracil. Ann. Oncol. 2002, 13, 484–485. [Google Scholar] [CrossRef] [PubMed]
- Saif, M.W.; Shah, M.M.; Shah, A.R. Fluoropyrimidine-associated cardiotoxicity: Revisited. Expert Opin. Drug Saf. 2009, 8, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, S.; Sara, J.; Lobo, R.; Eiring, R.A.; Finnes, H.; Mitchell, J.; Hartgers, M.; Okano, A.; Halfdanarson, T.; Grothey, A. Bolus 5-fluorouracil (5-FU) In Combination with Oxaliplatin Is Safe and Well Tolerated in Patients Who Experienced Coronary Vasospasm With Infusional 5-FU or Capecitabine. Clin. Color. Cancer 2019, 18, 52–57. [Google Scholar] [CrossRef]
- Cimino, G.D.; Pan, C.-X.; Henderson, P.T. Personalized medicine for targeted and platinum-based chemotherapy of lung and bladder cancer. Bioanalytical 2013, 5, 369–391. [Google Scholar] [CrossRef]
- Feldman, D.R.; Schaffer, W.L.; Steingart, R.M. Late Cardiovascular Toxicity Following Chemotherapy for Germ Cell Tumors. J. Natl. Compr. Cancer Netw. 2012, 10, 537–544. [Google Scholar] [CrossRef][Green Version]
- Brouwers, E.E.M.; Huitema, A.D.; Beijnen, J.H.; Schellens, J.H.M. Long-term platinum retention after treatment with cisplatin and oxaliplatin. BMC Clin. Pharmacol. 2008, 8, 7–10. [Google Scholar] [CrossRef]
- Haugnes, H.S.; Wethal, T.; Aass, N.; Dahl, O.; Klepp, O.; Langberg, C.W.; Wilsgaard, T.; Bremnes, R.M.; Fosså, S.D. Cardiovascular Risk Factors and Morbidity in Long-Term Survivors of Testicular Cancer: A 20-Year Follow-Up Study. J. Clin. Oncol. 2010, 28, 4649–4657. [Google Scholar] [CrossRef]
- Abu Rmilah, A.A.; Lin, G.; Begna, K.H.; Friedman, P.A.; Herrmann, J. Risk of QTc prolongation among cancer patients treated with tyrosine kinase inhibitors. Int. J. Cancer 2020, 147, 3160–3167. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, J. Vascular toxic effects of cancer therapies. Nat. Rev. Cardiol. 2020, 17, 503–522. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, J. Adverse cardiac effects of cancer therapies: Cardiotoxicity and arrhythmia. Nat. Rev. Cardiol. 2020, 17, 474–502. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M.; Herrmann, J. Cardiotoxicity with vascular endothelial growth factor inhibitor therapy. NPJ Precis. Oncol. 2018, 2, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chari, A.; Stewart, A.K.; Russell, S.D.; Moreau, P.; Herrmann, J.; Banchs, J.; Hajek, R.; Groarke, J.; Lyon, A.R.; Batty, G.N.; et al. Analysis of carfilzomib cardiovascular safety profile across relapsed and/or refractory multiple myeloma clinical trials. Blood Adv. 2018, 2, 1633–1644. [Google Scholar] [CrossRef] [PubMed]
- Soria, J.-C.; Ohe, Y.; Vansteenkiste, J.; Reungwetwattana, T.; Chewaskulyong, B.; Lee, K.H.; Dechaphunkul, A.; Imamura, F.; Nogami, N.; Kurata, T.; et al. Osimertinib in UntreatedEGFR-Mutated Advanced Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 113–125. [Google Scholar] [CrossRef]
- Network N.C.C. NCCN Guidelines. Available online: https://www.nccn.org/professionals/physician_gls/. (accessed on 12 August 2020).
- Kunimasa, K.; Kamada, R.; Oka, T.; Oboshi, M.; Kimura, M.; Inoue, T.; Tamiya, M.; Nishikawa, T.; Yasui, T.; Shioyama, W.; et al. Cardiac Adverse Events in EGFR-Mutated Non-Small Cell Lung Cancer Treated With Osimertinib. JACC CardioOncol. 2020, 2, 1–10. [Google Scholar] [CrossRef]
- Patel, S.B.S.; Kubusek, J.; Mansfield, A.; Duma, N. Osimertinib-induced Cardiomyopathy: Case series and review of the literature. JACC Case Rep. 2019, 2, 651–645. [Google Scholar]
- Archibald, W.J.; Rabe, K.G.; Kabat, B.F.; Herrmann, J.; Ding, W.; Kay, N.E.; Kenderian, S.S.; Muchtar, E.; Leis, J.F.; Wang, Y.; et al. Atrial fibrillation in patients with chronic lymphocytic leukemia (CLL) treated with ibrutinib: Risk prediction, management, and clinical outcomes. Ann. Hematol. 2020. [Google Scholar] [CrossRef]
- Ganatra, S.; Sharma, A.; Shah, S.; Chaudhry, G.M.; Martin, D.T.; Neilan, T.G.; Mahmood, S.S.; Barac, A.; Groarke, J.D.; Hayek, S.S.; et al. Ibrutinib-Associated Atrial Fibrillation. JACC Clin. Electrophysiol. 2018, 4, 1491–1500. [Google Scholar] [CrossRef]
- Agrawal, N.; Khunger, A.; Vachhani, P.; Colvin, T.A.; Hattoum, A.; Spangenthal, E.; Curtis, A.B.; Dy, G.K.; Ernstoff, M.S.; Puzanov, I. Cardiac Toxicity Associated with Immune Checkpoint Inhibitors: Case Series and Review of the Literature. Case Rep. Oncol. 2019, 12, 260–276. [Google Scholar] [CrossRef] [PubMed]
- Bonaca, M.P.; Olenchock, B.A.; Salem, J.E.; Wiviott, S.D.; Ederhy, S.; Cohen, A.; Stewart, G.C.; Choueiri, T.K.; Di Carli, M.; Allenbach, Y.; et al. Myocarditis in the Setting of Cancer Therapeutics: Proposed Case Definitions for Emerging Clinical Syndromes in Cardio-Oncology. Circulation 2019, 140, 80–91. [Google Scholar] [PubMed]
- Champion, S.N.; Stone, J.R. Immune checkpoint inhibitor associated myocarditis occurs in both high-grade and low-grade forms. Mod. Pathol. 2019, 33, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Palaskas, N.; Lopez-Mattei, J.; Durand, J.B.; Iliescu, C.; Deswal, A. Immune Checkpoint Inhibitor Myocarditis: Pathophysiological Characteristics, Diagnosis, and Treatment. J. Am. Hear. Assoc. 2020, 9, e013757. [Google Scholar] [CrossRef]
- Thompson, J.A.; Schneider, B.J.; Brahmer, J.; Andrews, S.; Armand, P.; Bhatia, S.; Budde, L.E.; Costa, L.; Davies, M.; Dunnington, D.; et al. NCCN Guidelines Insights: Management of Immunotherapy-Related Toxicities, Version 1.2020. J. Natl. Compr. Cancer Netw. 2020, 18, 230–241. [Google Scholar] [CrossRef]
- Awadalla, M.; Mahmood, S.S.; Groarke, J.D.; Hassan, M.Z.; Nohria, A.; Rokicki, A.; Murphy, S.P.; Mercaldo, N.D.; Zhang, L.; Zlotoff, D.A.; et al. Global Longitudinal Strain and Cardiac Events in Patients With Immune Checkpoint Inhibitor-Related Myocarditis. J. Am. Coll. Cardiol. 2020, 75, 467–478. [Google Scholar] [CrossRef]
- Mahmood, S.S.; Fradley, M.G.; Cohen, J.V.; Nohria, A.; Reynolds, K.L.; Heinzerling, L.M.; Sullivan, R.J.; Damrongwatanasuk, R.; Chen, C.L.; Gupta, D.; et al. Myocarditis in Patients Treated With Immune Checkpoint Inhibitors. J. Am. Coll. Cardiol. 2018, 71, 1755–1764. [Google Scholar] [CrossRef]
- Alvi, R.M.; Frigault, M.J.; Fradley, M.G.; Jain, M.D.; Mahmood, S.S.; Awadalla, M.; Lee, D.H.; Zlotoff, D.A.; Zhang, L.; Drobni, Z.D.; et al. Cardiovascular Events Among Adults Treated With Chimeric Antigen Receptor T-Cells (CAR-T). J. Am. Coll. Cardiol. 2019, 74, 3099–3108. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Chen, D.H.; Guha, A.; MacKenzie, S.; Walker, J.M.; Roddie, C. CAR T Cell Therapy–Related Cardiovascular Outcomes and Management. JACC CardioOncol. 2020, 2, 97–109. [Google Scholar] [CrossRef]
- Copelan, E.A. Hematopoietic stem-cell transplantation. N. Engl. J. Med. 2006, 354, 1813–1826. [Google Scholar] [CrossRef]
- Shapiro, C.L. Cancer Survivorship. N. Engl. J. Med. 2018, 379, 2438–2450. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Nogueira, L.; Mariotto, A.B.; Rowland, J.H.; Yabroff, K.R.; Alfano, C.M.; Jemal, A.; Kramer, J.L.; Siegel, R.L. Cancer treatment and survivorship statistics, 2019. CA A Cancer J. Clin. 2019, 69, 363–385. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Carter, A.; Sun, C.-L.; Francisco, L.; Baker, K.S.; Gurney, J.G.; Weisdorf, D.G.; Forman, S.J.; Robison, L.L.; Grant, M.; et al. Health Care Utilization by Adult Long-term Survivors of Hematopoietic Cell Transplant: Report from the Bone Marrow Transplant Survivor Study. Cancer Epidemiol. Biomark. Prev. 2007, 16, 834–839. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bhatia, S.; Robison, L.L.; Francisco, L.; Carter, A.; Liu, Y.; Grant, M.; Baker, K.S.; Fung, H.; Gurney, J.G.; McGlave, P.B.; et al. Late mortality in survivors of autologous hematopoietic-cell transplantation: Report from the Bone Marrow Transplant Survivor Study. Blood 2005, 105, 4215–4222. [Google Scholar] [CrossRef] [PubMed]
- Wingard, J.R.; Majhail, N.S.; Brazauskas, R.; Wang, Z.; Sobocinski, K.A.; Jacobsohn, D.; Sorror, M.L.; Horowitz, M.M.; Bolwell, B.; Rizzo, J.D.; et al. Long-Term Survival and Late Deaths After Allogeneic Hematopoietic Cell Transplantation. J. Clin. Oncol. 2011, 29, 2230–2239. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Mueller, B.A.; Baker, K.S.; Cushing-Haugen, K.L.; Flowers, M.E.; Martin, P.J.; Friedman, D.L.; Lee, S.J. Cardiovascular Hospitalizations and Mortality among Recipients of Hematopoietic Stem Cell Transplantation. Ann. Intern. Med. 2011, 155, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Armenian, S.H.; Chemaitilly, W.; Chen, M.; Chow, E.J.; Duncan, C.N.; Jones, L.W.; Pulsipher, M.A.; Remaley, A.T.; Rovo, A.; Salooja, N.; et al. National Institutes of Health Hematopoietic Cell Transplantation Late Effects Initiative: The Cardiovascular Disease and Associated Risk Factors Working Group Report. Biol. Blood Marrow Transplant. 2017, 23, 201–210. [Google Scholar] [CrossRef]
- Armenian, S.H.; Chow, E.J. Cardiovascular disease in survivors of hematopoietic cell transplantation. Cancer 2014, 120, 469–479. [Google Scholar] [CrossRef]
- Armenian, S.H.; Sun, C.-L.; Vase, T.; Ness, K.K.; Blum, E.; Francisco, L.; Venkataraman, K.; Samoa, R.; Wong, F.L.; Forman, S.J.; et al. Cardiovascular risk factors in hematopoietic cell transplantation survivors: Role in development of subsequent cardiovascular disease. Blood 2012, 120, 4505–4512. [Google Scholar] [CrossRef]
- Armenian, S.H.; Sun, C.L.; Francisco, L.; Steinberger, J.; Kurian, S.; Wong, F.L.; Sharp, J.; Sposto, R.; Forman, S.J.; Bhatia, S. Late congestive heart failure after hematopoietic cell transplantation. J. Clin. Oncol. 2008, 26, 5537–5543. [Google Scholar] [CrossRef]
- Armenian, S.H.; Yang, N.; Teh, J.B.; Atencio, L.C.; Gonzales, A.; Wong, F.L.; Leisenring, W.M.; Forman, S.J.; Nakamura, R.; Chow, E.J. Prediction of cardiovascular disease among hematopoietic cell transplantation survivors. Blood Adv. 2018, 2, 1756–1764. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Baker, K.S.; Lee, S.J.; Flowers, M.E.; Cushing-Haugen, K.L.; Inamoto, Y.; Khera, N.; Leisenring, W.M.; Syrjala, K.L.; Martin, P.J. Influence of Conventional Cardiovascular Risk Factors and Lifestyle Characteristics on Cardiovascular Disease After Hematopoietic Cell Transplantation. J. Clin. Oncol. 2014, 32, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Arranto, C.A.; Burkard, T.; Leuppi-Taegtmeyer, A.B.; Gerull, S.; Passweg, J.R.; Pfister, O.; Halter, J. Prevalence of untreated and uncontrolled cardiovascular risk factors in survivors of allogeneic cell transplantation. Bone Marrow Transplant. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, K.; Unitt, C.; Foody, J.M.; Harris, J.R.; Partridge, A.H.; Moslehi, J. ABCDE Steps to Prevent Heart Disease in Breast Cancer Survivors. Circulation 2014, 130, e157–e159. [Google Scholar] [CrossRef] [PubMed]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2019, 74, e177–e232. [Google Scholar]
- American College of Cardiology. ASCVD Risk Estimator Plus. Available online: http://tools.acc.org/ascvd-risk-estimator-plus/#!/calculate/estimate/ (accessed on 10 December 2020).
- Virani, S.S.; Smith, S.C.; Stone, N.J., Jr.; Grundy, S.M. Secondary Prevention for Atherosclerotic Cardiovascular Disease: Comparing Recent US and European Guidelines on Dyslipidemia. Circulation 2020, 141, 1121–1123. [Google Scholar] [CrossRef]
- Gilchrist, S.C.; Barac, A.; Ades, P.A.; Alfano, C.M.; Franklin, B.A.; Jones, L.W.; La Gerche, A.; Ligibel, J.A.; Lopez, G.; Madan, K.; et al. Cardio-Oncology Rehabilitation to Manage Cardiovascular Outcomes in Cancer Patients and Survivors: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e997–e1012. [Google Scholar] [CrossRef] [PubMed]
- D’Amario, D.; Camilli, M.; Migliaro, S.; Canonico, F.; Galli, M.; Arcudi, A.; Montone, R.A.; Borovac, J.A.; Crea, F.; Savarese, G. Sex-Related Differences in Dilated Cardiomyopathy with a Focus on Cardiac Dysfunction in Oncology. Curr. Cardiol. Rep. 2020, 22, 1–11. [Google Scholar] [CrossRef]
- Armenian, S.H.; Lacchetti, C.; Barac, A.; Carver, J.; Constine, L.S.; Denduluri, N.; Dent, S.; Douglas, P.S.; Durand, J.-B.; Ewer, M.; et al. Prevention and Monitoring of Cardiac Dysfunction in Survivors of Adult Cancers: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2017, 35, 893–911. [Google Scholar] [CrossRef]
- Sanft, T.; Denlinger, C.S.; Armenian, S.; Baker, K.S.; Broderick, G.; Demark-Wahnefried, W.; Friedman, D.L.; Goldman, M.; Hudson, M.; Khakpour, N.; et al. NCCN Guidelines Insights: Survivorship, Version 2.2019. J. Natl. Compr. Cancer Netw. 2019, 17, 784–794. [Google Scholar] [CrossRef]
- Ruddy, K.J.; Sangaralingham, L.R.; Van Houten, H.; Nowsheen, S.; Sandhu, N.; Moslehi, J.; Neuman, H.; Jemal, A.; Haddad, T.C.; Blaes, A.H.; et al. Utilization of Cardiac Surveillance Tests in Survivors of Breast Cancer and Lymphoma After Anthracycline-Based Chemotherapy. Circ. Cardiovasc. Qual. Outcomes 2020, 13, e005984. [Google Scholar] [CrossRef] [PubMed]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; Gonzalez-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [PubMed]
- Baker, K.S.; Syrjala, K.L. Long-term complications in adolescent and young adult leukemia survivors. Hematology 2018, 2018, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Hancock, S.L.; Tucker, M.A.; Hoppe, R.T. Factors affecting late mortality from heart disease after treatment of Hodgkin’s disease. JAMA 1993, 270, 1949–1955. [Google Scholar] [CrossRef]
- Mauch, P.M.; Kalish, L.A.; Marcus, K.C.; Shulman, L.N.; Krill, E.; Tarbell, N.J.; Silver, B.; Weinstein, H.; Come, S.; Canellos, G.P.; et al. Long-term survival in Hodgkin’s disease relative impact of mortality, second tumors, infection, and cardiovascular disease. Cancer J. Sci. Am. 1995, 1, 33–42. [Google Scholar]
- Aleman, B.M.; van den Belt-Dusebout, A.W.; De Bruin, M.L.; van’t Veer, M.B.; Baaijens, M.H.; Boer, J.P.D.; Hart, A.A.; Klokman, W.J.; Kuenen, M.A.; Ouwens, G.M.; et al. Late cardiotoxicity after treatment for Hodgkin lymphoma. Blood 2006, 109, 1878–1886. [Google Scholar] [CrossRef]
- Chao, C.; Xu, L.; Bhatia, S.; Cooper, R.; Brar, S.; Wong, F.L.; Armenian, S.H. Cardiovascular Disease Risk Profiles in Survivors of Adolescent and Young Adult (AYA) Cancer: The Kaiser Permanente AYA Cancer Survivors Study. J. Clin. Oncol. 2016, 34, 1626–1633. [Google Scholar] [CrossRef]
- Williams, G.R.; Mackenzie, A.; Magnuson, A.; Olin, R.; Chapman, A.; Mohile, S.; Allore, H.; Somerfield, M.R.; Targia, V.; Extermann, M.; et al. Comorbidity in older adults with cancer. J. Geriatr. Oncol. 2016, 7, 249–257. [Google Scholar] [CrossRef]
- Sogaard, M.; Thomsen, R.W.; Bossen, K.S.; Sørensen, H.H.T.; Nørgaard, M. The impact of comorbidity on cancer survival: A review. Clin. Epidemiol. 2013, 5, 3–29. [Google Scholar] [CrossRef]
- Fourcadier, E.; Trouche-Sabatier, S.; Gras-Aygon, C.; Ecarnot, F.; Daurès, J.-P.; Bessaoud, F. Under-treatment of elderly patients with ovarian cancer: A population based study. BMC Cancer 2015, 15, 937. [Google Scholar] [CrossRef]
- Morrison, V.A.; Hamlin, P.; Soubeyran, P.; Stauder, R.; Wadhwa, P.; Aapro, M.; Lichtman, S.M. Approach to therapy of diffuse large B-cell lymphoma in the elderly: The International Society of Geriatric Oncology (SIOG) expert position commentary. Ann. Oncol. 2015, 26, 1058–1068. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.F.; Ky, B. Roadmap for biomarkers of cancer therapy cardiotoxicity. Heart 2016, 102, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.M.; Rodrigues, J.J.P.C.; De La Torre-Díez, I.; López-Coronado, M.; Saleem, K. Mobile-health: A review of current state in 2015. J. Biomed. Inform. 2015, 56, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Mileski, M.; Kruse, C.S.; Catalani, J.; Haderer, T.; Cianflone, D.; Subasinghe, A. Adopting Telemedicine for the Self-Management of Hypertension: Systematic Review. JMIR Med. Inform. 2017, 5, e41. [Google Scholar] [CrossRef]
- Lee, S.W.H.; Ooi, L.; Lai, Y.K. Telemedicine for the Management of Glycemic Control and Clinical Outcomes of Type 1 Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. Front. Pharmacol. 2017, 8, 330. [Google Scholar] [CrossRef]
- De Simone, V.; Guarise, P.; Guardalben, S.; Padovani, N.; Tondelli, S.; Sandrini, D.; Visentin, E.; Zanotto, G. Telecardiology during the Covid-19 pandemic: Past mistakes and future hopes. Am J Cardiovasc Dis 2020, 10, 34–47. [Google Scholar]
- Chang, H.A.; Armenian, S.H.; Dellinger, T.H. Secondary Neoplasms of the Female Lower Genital Tract after Hematopoietic Cell Transplantation. J. Natl. Compr. Cancer Netw. 2018, 16, 211–218. [Google Scholar] [CrossRef]
- Chung, I.Y.; Jung, M.; Park, Y.R.; Cho, D.; Chung, H.; Min, Y.H.; Park, H.J.; Lee, M.; Lee, S.B.; Chung, S.; et al. Exercise Promotion and Distress Reduction Using a Mobile App-Based Community in Breast Cancer Survivors. Front. Oncol. 2020, 9, 1505. [Google Scholar] [CrossRef]
- Casillas, J.; Schwartz, L.F.; Crespi, C.M.; Ganz, P.A.; Kahn, K.L.; Stuber, M.L.; Bastani, R.; Alquaddomi, F.; Estrin, D.L. The use of mobile technology and peer navigation to promote adolescent and young adult (AYA) cancer survivorship care: Results of a randomized controlled trial. J. Cancer Surviv. 2019, 13, 580–592. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruddy, K.J.; Patel, S.R.; Higgins, A.S.; Armenian, S.H.; Herrmann, J. Cardiovascular Health during and after Cancer Therapy. Cancers 2020, 12, 3737. https://doi.org/10.3390/cancers12123737
Ruddy KJ, Patel SR, Higgins AS, Armenian SH, Herrmann J. Cardiovascular Health during and after Cancer Therapy. Cancers. 2020; 12(12):3737. https://doi.org/10.3390/cancers12123737
Chicago/Turabian StyleRuddy, Kathryn J., Shruti R. Patel, Alexandra S. Higgins, Saro H. Armenian, and Joerg Herrmann. 2020. "Cardiovascular Health during and after Cancer Therapy" Cancers 12, no. 12: 3737. https://doi.org/10.3390/cancers12123737
APA StyleRuddy, K. J., Patel, S. R., Higgins, A. S., Armenian, S. H., & Herrmann, J. (2020). Cardiovascular Health during and after Cancer Therapy. Cancers, 12(12), 3737. https://doi.org/10.3390/cancers12123737




