Targeting Wnt Signaling for Gastrointestinal Cancer Therapy: Present and Evolving Views
Abstract
Simple Summary
Abstract
1. Introduction
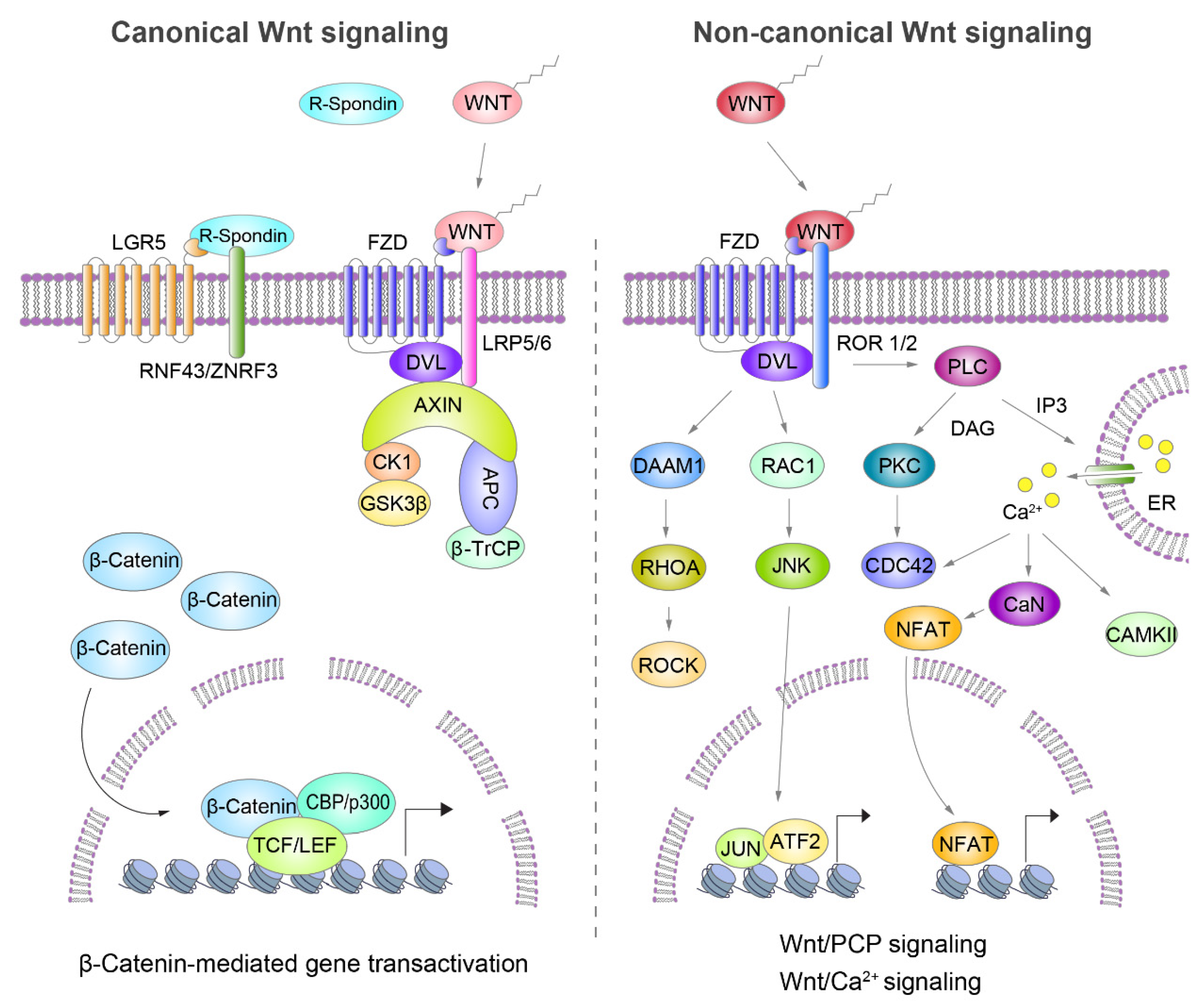
2. Wnt Signaling
3. Wnt Signaling Alteration in GI Cancers
4. Therapeutically Targeting Wnt Signaling in GI Cancer
5. Targeting WNT Ligands
5.1. Inhibiting WNT Ligands
5.2. Targeting Lipid Modification of WNT Ligands
6. Targeting Wnt Receptors and Co-Receptors
6.1. Antibodies against FZDs
6.2. Mimetic Agents Binding to FZDs
6.3. Inhibiting LRP5/6
6.4. Accelerating the Degradation of FZD/LRP Receptors
7. Targeting the Destruction Complex
7.1. Inhibiting the DVL–FZD Interaction
7.2. Stabilizing AXIN
7.3. Stabilizing APC
7.4. Activating CK1 and GSK3
8. Targeting β-Catenin and β-Catenin-Dependent Transcriptional Machinery
8.1. Promoting β-Catenin Degradation
8.2. Inhibiting the β-Catenin–TCF/LEF Complex
8.3. Manipulating TCF/LEF Phosphatases
8.4. Inhibiting Coactivators
9. Caveats in Targeting Wnt Signaling
9.1. Targeting Core Components of Wnt Signaling
9.2. Targeting Upstream vs. Downstream
10. Evolving Views in Targeting Wnt Signaling
10.1. Cancer- and Tissue-Specific Wnt Signaling Targeting
10.2. Efficacy and Combination Therapy
10.3. β-Catenin Paradox
10.4. Generalization of Wnt Targeting Therapy
11. New Candidates for Targeting Wnt Signaling in GI Cancers
12. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nusse, R.; Varmus, H.E. Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell 1982, 31, 99–109. [Google Scholar] [CrossRef]
- Nusslein-Volhard, C.; Wieschaus, E. Mutations affecting segment number and polarity in Drosophila. Nature 1980, 287, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Steinhart, Z.; Angers, S. Wnt signaling in development and tissue homeostasis. Development 2018, 145, dev146589. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H.; Loh, K.M.; Nusse, R. Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science 2014, 346, 1248012. [Google Scholar] [CrossRef]
- Logan, C.Y.; Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef]
- Nusse, R. Wnt signaling in disease and in development. Cell Res. 2005, 15, 28–32. [Google Scholar] [CrossRef]
- Kahn, M. Can we safely target the WNT pathway? Nat. Rev. Drug Discov. 2014, 13, 513–532. [Google Scholar] [CrossRef]
- Anastas, J.N.; Moon, R.T. WNT signalling pathways as therapeutic targets in cancer. Nat. Rev. Cancer 2013, 13, 11–26. [Google Scholar] [CrossRef]
- Jung, Y.S.; Park, J.I. Wnt signaling in cancer: Therapeutic targeting of Wnt signaling beyond beta-catenin and the destruction complex. Exp. Mol. Med. 2020, 52, 183–191. [Google Scholar] [CrossRef]
- Niehrs, C. The complex world of WNT receptor signalling. Nat. Rev. Mol. Cell Biol. 2012, 13, 767–779. [Google Scholar] [CrossRef]
- Li, V.S.; Ng, S.S.; Boersema, P.J.; Low, T.Y.; Karthaus, W.R.; Gerlach, J.P.; Mohammed, S.; Heck, A.J.; Maurice, M.M.; Mahmoudi, T.; et al. Wnt signaling through inhibition of beta-catenin degradation in an intact Axin1 complex. Cell 2012, 149, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- Papkoff, J.; Brown, A.M.; Varmus, H.E. The int-1 proto-oncogene products are glycoproteins that appear to enter the secretory pathway. Mol. Cell Biol. 1987, 7, 3978–3984. [Google Scholar] [CrossRef] [PubMed]
- Grumolato, L.; Liu, G.; Mong, P.; Mudbhary, R.; Biswas, R.; Arroyave, R.; Vijayakumar, S.; Economides, A.N.; Aaronson, S.A. Canonical and noncanonical Wnts use a common mechanism to activate completely unrelated coreceptors. Genes Dev. 2010, 24, 2517–2530. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.H.; Seeling, J.M.; Hill, V.; Yochum, A.; Virshup, D.M. Casein kinase I phosphorylates and destabilizes the beta-catenin degradation complex. Proc. Natl. Acad. Sci. USA 2002, 99, 1182–1187. [Google Scholar] [CrossRef] [PubMed]
- Ha, N.C.; Tonozuka, T.; Stamos, J.L.; Choi, H.J.; Weis, W.I. Mechanism of phosphorylation-dependent binding of APC to beta-catenin and its role in beta-catenin degradation. Mol. Cell 2004, 15, 511–521. [Google Scholar] [CrossRef]
- Liu, C.; Li, Y.; Semenov, M.; Han, C.; Baeg, G.-H.; Tan, Y.; Zhang, Z.; Lin, X.; He, X. Control of beta-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell 2002, 108, 837–847. [Google Scholar] [CrossRef]
- Stamos, J.L.; Weis, W.I. The beta-catenin destruction complex. Cold Spring Harb. Perspect. Biol. 2013, 5, a007898. [Google Scholar] [CrossRef]
- He, X.; Semenov, M.; Tamai, K.; Zeng, X. LDL receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling: Arrows point the way. Development 2004, 131, 1663–1677. [Google Scholar] [CrossRef]
- Kishida, S.; Yamamoto, H.; Hino, S.; Ikeda, S.; Kishida, M.; Kikuchi, A. DIX domains of Dvl and axin are necessary for protein interactions and their ability to regulate beta-catenin stability. Mol. Cell Biol. 1999, 19, 4414–4422. [Google Scholar] [CrossRef]
- Brunner, E.; Peter, O.; Schweizer, L.; Basler, K. Pangolin encodes a Lef-1 homologue that acts downstream of Armadillo to transduce the Wingless signal in Drosophila. Nature 1997, 385, 829–833. [Google Scholar] [CrossRef]
- van de Wetering, M.; Cavallo, R.; Dooijes, D.; van Beest, M.; van Es, J.; Loureiro, J.; Ypma, A.; Hursh, D.; Jones, T.; Bejsovec, A.; et al. Armadillo coactivates transcription driven by the product of the Drosophila segment polarity gene dTCF. Cell 1997, 88, 789–799. [Google Scholar] [CrossRef]
- Takemaru, K.I.; Moon, R.T. The transcriptional coactivator CBP interacts with beta-catenin to activate gene expression. J. Cell Biol. 2000, 149, 249–254. [Google Scholar] [CrossRef]
- Hecht, A.; Vleminckx, K.; Stemmler, M.P.; van Roy, F.; Kemler, R. The p300/CBP acetyltransferases function as transcriptional coactivators of beta-catenin in vertebrates. EMBO J. 2000, 19, 1839–1850. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, A.; Yamamoto, H.; Sato, A.; Matsumoto, S. New insights into the mechanism of Wnt signaling pathway activation. Int. Rev. Cell Mol. Biol. 2011, 291, 21–71. [Google Scholar] [PubMed]
- Simons, M.; Mlodzik, M. Planar cell polarity signaling: From fly development to human disease. Annu. Rev. Genet. 2008, 42, 517–540. [Google Scholar] [CrossRef] [PubMed]
- De, A. Wnt/Ca2+ signaling pathway: A brief overview. Acta Biochim. Biophys. Sin. 2011, 43, 745–756. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012, 487, 330–337. [Google Scholar] [CrossRef]
- Rowan, A.J.; Lamlum, H.; Ilyas, M.; Wheeler, J.; Straub, J.; Papadopoulou, A.; Bicknell, D.; Bodmer, W.F.; Tomlinson, I.P. APC mutations in sporadic colorectal tumors: A mutational “hotspot” and interdependence of the “two hits”. Proc. Natl. Acad. Sci. USA 2000, 97, 3352–3357. [Google Scholar] [CrossRef]
- Russell, J.O.; Monga, S.P. Wnt/beta-catenin signaling in liver development, homeostasis, and pathobiology. Annu. Rev. Pathol. 2018, 13, 351–378. [Google Scholar] [CrossRef]
- Khalaf, A.M.; Fuentes, D.; Morshid, A.I.; Burke, M.R.; Kaseb, A.O.; Hassan, M.; Hazle, J.D.; Elsayes, K.M. Role of Wnt/beta-catenin signaling in hepatocellular carcinoma, pathogenesis, and clinical significance. J. Hepatocell. Carcinoma 2018, 5, 61–73. [Google Scholar] [CrossRef]
- Merle, P.; de la Monte, S.; Kim, M.; Herrmann, M.; Tanaka, S.; Von Dem Bussche, A.; Kew, M.C.; Trepo, C.; Wands, J.R. Functional consequences of frizzled-7 receptor overexpression in human hepatocellular carcinoma. Gastroenterology 2004, 127, 1110–1122. [Google Scholar] [CrossRef]
- Waddell, N.; Pajic, M.; Patch, A.M.; Chang, D.K.; Kassahn, K.S.; Bailey, P.; Johns, A.L.; Miller, D.; Nones, K.; Quek, K.; et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature 2015, 518, 495–501. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.; Tan, D.S.; Subbiah, V.; Weekes, C.D.; Teneggi, V.; Diermayr, V.; Ethirajulu, K.; Yeo, P.; Chen, D.; Blanchard, S. First-in-human phase 1 study of ETC-159 an oral PORCN inhbitor in patients with advanced solid tumours. Am. Soc. Clin. Oncol. 2017, 35. [Google Scholar] [CrossRef]
- Pintova, S.; Dharmupari, S.; Moshier, E.; Zubizarreta, N.; Ang, C.; Holcombe, R.F. Genistein combined with FOLFOX or FOLFOX-Bevacizumab for the treatment of metastatic colorectal cancer: Phase I/II pilot study. Cancer Chemother. Pharmacol. 2019, 84, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Jimeno, A.; Gordon, M.; Chugh, R.; Messersmith, W.; Mendelson, D.; Dupont, J.; Stagg, R.; Kapoun, A.M.; Xu, L.; Uttamsingh, S.; et al. A first-in-human phase I study of the anticancer stem cell agent ipafricept (OMP-54F28), a decoy receptor for Wnt ligands, in patients with advanced solid tumors. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 7490–7497. [Google Scholar] [CrossRef] [PubMed]
- Dotan, E.; Cardin, D.B.; Lenz, H.-J.; Messersmith, W.A.; O’Neil, B.; Cohen, S.J.; Denlinger, C.S.; Shahda, S.; Kapoun, A.M.; Brachmann, R.K.; et al. Phase Ib study of WNT inhibitor ipafricept (IPA) with nab-paclitaxel (Nab-P) and gemcitabine (G) in patients (pts) with previously untreated stage IV pancreatic cancer (mPC). Am. Soc. Clin. Oncol. 2019, 37. [Google Scholar] [CrossRef]
- Vermorken, J.; Cervantes, A.; Morsing, P.; Johansson, K.; Andersson, T.; Roest, N.L.; Gullbo, J.; Salazar, R. P-133 A randomized, multicenter, open-label controlled phase 2 trial of Foxy-5 as neoadjuvant therapy in patients with WNT5A negative colon cancer. Ann. Oncol. 2019, 30 (Suppl. S4). [Google Scholar] [CrossRef]
- Davis, S.L.; Cardin, D.B.; Shahda, S.; Lenz, H.J.; Dotan, E.; O’Neil, B.H.; Kapoun, A.M.; Stagg, R.J.; Berlin, J.; Messersmith, W.A.; et al. A phase 1b dose escalation study of Wnt pathway inhibitor vantictumab in combination with nab-paclitaxel and gemcitabine in patients with previously untreated metastatic pancreatic cancer. Investig. New Drugs 2019, 38, 821–830. [Google Scholar] [CrossRef]
- Ryan, D.; Murphy, J.; Mahalingam, D.; Strickler, J.; Stein, S.; Sirard, C.; Landau, S.; Bendell, J. PD-016 Current results of a phase I study of DKN-01 in combination with paclitaxel (P) in patients (pts) with advanced DKK1+ esophageal cancer (EC) or gastro-esophageal junction tumors (GEJ). Ann. Oncol. 2016, 27 (Suppl. S2), ii108. [Google Scholar] [CrossRef][Green Version]
- Eads, J.R.; Goyal, L.; Stein, S.; El-Khoueiry, A.B.; Manji, G.A.; Abrams, T.A.; Landau, S.B.; Sirard, C.A. Phase I study of DKN-01, an anti-DKK1 antibody, in combination with gemcitabine (G) and cisplatin (C) in patients (pts) with advanced biliary cancer. J. Clin. Oncol. 2016, 34 (Suppl. S15), e15603. [Google Scholar] [CrossRef]
- Bendell, J.; Eckhardt, G.; Hochster, H.; Morris, V.; Strickler, J.; Kapoun, A.; Wang, M.; Xu, L.; McGuire, K.; Dupont, J. Initial results from a phase 1a/b study of OMP-131R10, a first-in-class anti-RSPO3 antibody, in advanced solid tumors and previously treated metastatic colorectal cancer (CRC). Eur. J. Cancer 2016, 69 (Suppl. 1), S29–S30. [Google Scholar] [CrossRef]
- Burock, S.; Daum, S.; Tröger, H.; Kim, T.D.; Krüger, S.; Rieke, D.T.; Ochsenreither, S.; Welter, K.; Herrmann, P.; Sleegers, A. Niclosamide a new chemotherapy agent? Pharmacokinetics of the potential anticancer drug in a patient cohort of the NIKOLO trial. Am. Soc. Clin. Oncol. 2018, 36, e14536. [Google Scholar] [CrossRef]
- Molenaar, R.J.; Coelen, R.J.S.; Khurshed, M.; Roos, E.; Caan, M.W.A.; van Linde, M.E.; Kouwenhoven, M.; Bramer, J.A.M.; Bovee, J.; Mathot, R.A.; et al. Study protocol of a phase IB/II clinical trial of metformin and chloroquine in patients with IDH1-mutated or IDH2-mutated solid tumours. BMJ Open. 2017, 7, e014961. [Google Scholar] [CrossRef] [PubMed]
- Mahalingam, D.; Mita, M.; Sarantopoulos, J.; Wood, L.; Amaravadi, R.K.; Davis, L.E.; Mita, A.C.; Curiel, T.J.; Espitia, C.M.; Nawrocki, S.T.; et al. Combined autophagy and HDAC inhibition: A phase I safety, tolerability, pharmacokinetic, and pharmacodynamic analysis of hydroxychloroquine in combination with the HDAC inhibitor vorinostat in patients with advanced solid tumors. Autophagy 2014, 10, 1403–1414. [Google Scholar] [CrossRef]
- Boone, B.A.; Bahary, N.; Zureikat, A.H.; Moser, A.J.; Normolle, D.P.; Wu, W.C.; Singhi, A.D.; Bao, P.; Bartlett, D.L.; Liotta, L.A.; et al. Safety and biologic response of pre-operative autophagy inhibition in combination with gemcitabine in patients with pancreatic adenocarcinoma. Ann. Surg. Oncol. 2015, 22, 4402–4410. [Google Scholar] [CrossRef]
- Loaiza-Bonilla, A.; O’Hara, M.H.; Redlinger, M.; Damjanov, N.; Teitelbaum, U.R.; Vasilevskaya, I.; Rosen, M.A.; Heitjan, D.F.; Amaravadi, R.K.; O’Dwyer, P.J. Phase II trial of autophagy inhibition using hydroxychloroquine (HCQ) with FOLFOX/bevacizumab in the first-line treatment of advanced colorectal cancer. J. Clin. Oncol. 2015, 33, 3614. [Google Scholar] [CrossRef]
- Wolpin, B.M.; Rubinson, D.A.; Wang, X.; Chan, J.A.; Cleary, J.M.; Enzinger, P.C.; Fuchs, C.S.; McCleary, N.J.; Meyerhardt, J.A.; Ng, K.; et al. Phase II and pharmacodynamic study of autophagy inhibition using hydroxychloroquine in patients with metastatic pancreatic adenocarcinoma. Oncologist 2014, 19, 637–648. [Google Scholar] [CrossRef]
- Hong, T.S.; Wo, J.Y.-L.; Jiang, W.; Yeap, B.Y.; Clark, J.W.; Ryan, D.P.; Blaszkowsky, L.S.; Drapek, L.C.; Mamon, H.J.; Murphy, J.E.; et al. Phase II study of autophagy inhibition with hydroxychloroquine (HCQ) and preoperative (preop) short course chemoradiation (SCRT) followed by early surgery for resectable ductal adenocarcinoma of the head of pancreas (PDAC). J. Clin. Oncol. 2017, 35, 4118. [Google Scholar] [CrossRef]
- Karasic, T.B.; O’Hara, M.H.; Loaiza-Bonilla, A.; Reiss, K.A.; Teitelbaum, U.R.; Borazanci, E.; De Jesus-Acosta, A.; Redlinger, C.; Burrell, J.A.; Laheru, D.A.; et al. Effect of gemcitabine and nab-paclitaxel with or without hydroxychloroquine on patients with advanced pancreatic cancer: A phase 2 randomized clinical trial. JAMA Oncol. 2019, 5, 993–998. [Google Scholar] [CrossRef]
- Arora, S.P.; Tenner, L.L.; Sarantopoulos, J.; Morris, J.L.; Longoria, L.; Liu, Q.; Michalek, J.; Mahalingam, D. Modulation of autophagy: A phase II study of vorinostat (VOR) plus hydroxychloroquine (HCQ) vs regorafenib (RGF) in chemo-refractory metastatic colorectal cancer (mCRC). J. Clin. Oncol. 2019, 37, 3551. [Google Scholar] [CrossRef]
- El-Khoueiry, A.B.; Ning, Y.; Yang, D.; Cole, S.; Kahn, M.; Zoghbi, M.; Berg, J.; Fujimori, M.; Inada, T.; Kouji, H. A phase I first-in-human study of PRI-724 in patients (pts) with advanced solid tumors. Am. Soc. Clin. Oncol. 2013, 31, 2501. [Google Scholar] [CrossRef]
- Ko, A.H.; Chiorean, E.G.; Kwak, E.L.; Lenz, H.-J.; Nadler, P.I.; Wood, D.L.; Fujimori, M.; Inada, T.; Kouji, H.; McWilliams, R.R. Final results of a phase Ib dose-escalation study of PRI-724, a CBP/beta-catenin modulator, plus gemcitabine (GEM) in patients with advanced pancreatic adenocarcinoma (APC) as second-line therapy after FOLFIRINOX or FOLFOX. Am. Soc. Clin. Oncol. 2016, 34, e15721. [Google Scholar] [CrossRef]
- Fischer, M.M.; Cancilla, B.; Yeung, V.P.; Cattaruzza, F.; Chartier, C.; Murriel, C.L.; Cain, J.; Tam, R.; Cheng, C.Y.; Evans, J.W.; et al. WNT antagonists exhibit unique combinatorial antitumor activity with taxanes by potentiating mitotic cell death. Sci. Adv. 2017, 3, e1700090. [Google Scholar] [CrossRef]
- Lopez-Rios, J.; Esteve, P.; Ruiz, J.M.; Bovolenta, P. The Netrin-related domain of Sfrp1 interacts with Wnt ligands and antagonizes their activity in the anterior neural plate. Neural Dev. 2008, 3, 19. [Google Scholar] [CrossRef]
- Bu, Q.; Li, Z.; Zhang, J.; Xu, F.; Liu, J.; Liu, H. The crystal structure of full-length Sizzled from Xenopus laevis yields insights into Wnt-antagonistic function of secreted Frizzled-related proteins. J. Biol. Chem. 2017, 292, 16055–16069. [Google Scholar] [CrossRef]
- Agostino, M.; Pohl, S.O.; Dharmarajan, A. Structure-based prediction of Wnt binding affinities for Frizzled-type cysteine-rich domains. J. Biol. Chem. 2017, 292, 11218–11229. [Google Scholar] [CrossRef]
- Xavier, C.P.; Melikova, M.; Chuman, Y.; Uren, A.; Baljinnyam, B.; Rubin, J.S. Secreted Frizzled-related protein potentiation versus inhibition of Wnt3a/beta-catenin signaling. Cell. Signal. 2014, 26, 94–101. [Google Scholar] [CrossRef]
- Liang, C.J.; Wang, Z.W.; Chang, Y.W.; Lee, K.C.; Lin, W.H.; Lee, J.L. SFRPs are biphasic modulators of Wnt-signaling-elicited cancer stem cell properties beyond extracellular control. Cell Rep. 2019, 28, 1511–1525.e5. [Google Scholar] [CrossRef]
- Malinauskas, T.; Aricescu, A.R.; Lu, W.; Siebold, C.; Jones, E.Y. Modular mechanism of Wnt signaling inhibition by Wnt inhibitory factor 1. Nat. Struct. Mol. Biol. 2011, 18, 886–893. [Google Scholar] [CrossRef]
- Piccolo, S.; Agius, E.; Leyns, L.; Bhattacharyya, S.; Grunz, H.; Bouwmeester, T.; De Robertis, E.M. The head inducer Cerberus is a multifunctional antagonist of Nodal, BMP and Wnt signals. Nature 1999, 397, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Rios-Esteves, J.; Haugen, B.; Resh, M.D. Identification of key residues and regions important for porcupine-mediated Wnt acylation. J. Biol. Chem. 2014, 289, 17009–17019. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Chia, J.; Canning, C.A.; Jones, C.M.; Bard, F.A.; Virshup, D.M. WLS retrograde transport to the endoplasmic reticulum during Wnt secretion. Dev. Cell 2014, 29, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Suciu, R.M.; Cognetta, A.B., 3rd; Potter, Z.E.; Cravatt, B.F. Selective irreversible inhibitors of the Wnt-deacylating enzyme NOTUM developed by activity-based protein profiling. ACS Med. Chem. Lett. 2018, 9, 563–568. [Google Scholar] [CrossRef]
- Moverare-Skrtic, S.; Nilsson, K.H.; Henning, P.; Funck-Brentano, T.; Nethander, M.; Rivadeneira, F.; Nunes, G.C.; Koskela, A.; Tuukkanen, J.; Tuckermann, J.; et al. Osteoblast-derived NOTUM reduces cortical bone mass in mice and the NOTUM locus is associated with bone mineral density in humans. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2019, 33, 11163–11179. [Google Scholar] [CrossRef]
- Zhang, X.; Abreu, J.G.; Yokota, C.; MacDonald, B.T.; Singh, S.; Coburn, K.L.; Cheong, S.M.; Zhang, M.M.; Ye, Q.Z.; Hang, H.C.; et al. Tiki1 is required for head formation via Wnt cleavage-oxidation and inactivation. Cell 2012, 149, 1565–1577. [Google Scholar] [CrossRef]
- Zhang, X.; MacDonald, B.T.; Gao, H.; Shamashkin, M.; Coyle, A.J.; Martinez, R.V.; He, X. Characterization of Tiki, a New Family of Wnt-specific Metalloproteases. J. Biol. Chem. 2016, 291, 2435–2443. [Google Scholar] [CrossRef]
- Steinhart, Z.; Pavlovic, Z.; Chandrashekhar, M.; Hart, T.; Wang, X.; Zhang, X.; Robitaille, M.; Brown, K.R.; Jaksani, S.; Overmeer, R.; et al. Genome-wide CRISPR screens reveal a Wnt-FZD5 signaling circuit as a druggable vulnerability of RNF43-mutant pancreatic tumors. Nat. Med. 2017, 23, 60–68. [Google Scholar] [CrossRef]
- Slusarski, D.C.; Yang-Snyder, J.; Busa, W.B.; Moon, R.T. Modulation of embryonic intracellular Ca2+ signaling by Wnt-5A. Dev. Biol. 1997, 182, 114–120. [Google Scholar] [CrossRef]
- Murphy, L.L.; Hughes, C.C. Endothelial cells stimulate T cell NFAT nuclear translocation in the presence of cyclosporin A: Involvement of the wnt/glycogen synthase kinase-3 beta pathway. J. Immunol. 2002, 169, 3717–3725. [Google Scholar] [CrossRef]
- Sheldahl, L.C.; Park, M.; Malbon, C.C.; Moon, R.T. Protein kinase C is differentially stimulated by Wnt and Frizzled homologs in a G-protein-dependent manner. Curr. Biol. CB 1999, 9, 695–698. [Google Scholar] [CrossRef]
- Kühl, M.; Sheldahl, L.C.; Malbon, C.C.; Moon, R.T. Ca(2+)/calmodulin-dependent protein kinase II is stimulated by Wnt and Frizzled homologs and promotes ventral cell fates in Xenopus. J. Biol. Chem. 2000, 275, 12701–12711. [Google Scholar] [CrossRef] [PubMed]
- Mikels, A.J.; Nusse, R. Purified Wnt5a protein activates or inhibits beta-catenin-TCF signaling depending on receptor context. PLoS Biol. 2006, 4, e115. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Yamamoto, H.; Sakane, H.; Koyama, H.; Kikuchi, A. Wnt5a regulates distinct signalling pathways by binding to Frizzled2. EMBO J. 2010, 29, 41–54. [Google Scholar] [CrossRef]
- Safholm, A.; Tuomela, J.; Rosenkvist, J.; Dejmek, J.; Harkonen, P.; Andersson, T. The Wnt-5a-derived hexapeptide Foxy-5 inhibits breast cancer metastasis in vivo by targeting cell motility. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2008, 14, 6556–6563. [Google Scholar] [CrossRef]
- Safholm, A.; Leandersson, K.; Dejmek, J.; Nielsen, C.K.; Villoutreix, B.O.; Andersson, T. A formylated hexapeptide ligand mimics the ability of Wnt-5a to impair migration of human breast epithelial cells. J. Biol. Chem. 2006, 281, 2740–2749. [Google Scholar] [CrossRef]
- Jönsson, M.; Andersson, T. Repression of Wnt-5a impairs DDR1 phosphorylation and modifies adhesion and migration of mammary cells. J. Cell Sci. 2001, 114 Pt 11, 2043–2053. [Google Scholar]
- Glinka, A.; Wu, W.; Delius, H.; Monaghan, A.P.; Blumenstock, C.; Niehrs, C.J.N. Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature 1998, 391, 357–362. [Google Scholar] [CrossRef]
- Niehrs, C. Function and biological roles of the Dickkopf family of Wnt modulators. Oncogene 2006, 25, 7469–7481. [Google Scholar] [CrossRef]
- Yu, B.; Yang, X.; Xu, Y.; Yao, G.; Shu, H.; Lin, B.; Hood, L.; Wang, H.; Yang, S.; Gu, J.; et al. Elevated expression of DKK1 is associated with cytoplasmic/nuclear beta-catenin accumulation and poor prognosis in hepatocellular carcinomas. J. Hepatol. 2009, 50, 948–957. [Google Scholar] [CrossRef]
- Xu, W.H.; Liu, Z.B.; Yang, C.; Qin, W.; Shao, Z.M. Expression of dickkopf-1 and beta-catenin related to the prognosis of breast cancer patients with triple negative phenotype. PLoS ONE 2012, 7, e37624. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.Y.; Yang, X.R.; Shen, Q.J.; Yang, L.X.; Xu, Y.; Qiu, S.J.; Sun, Y.F.; Zhang, X.; Wang, Z.; Zhu, K.; et al. High expression of Dickkopf-related protein 1 is related to lymphatic metastasis and indicates poor prognosis in intrahepatic cholangiocarcinoma patients after surgery. Cancer 2013, 119, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhou, H.; Zhang, X.; Ma, X.; Liu, Z.; Liu, X. Elevated levels of Dickkopf-1 are associated with beta-catenin accumulation and poor prognosis in patients with chondrosarcoma. PLoS ONE 2014, 9, e105414. [Google Scholar]
- Shi, Y.; Gong, H.L.; Zhou, L.; Tian, J.; Wang, Y. Dickkopf-1 is a novel prognostic biomarker for laryngeal squamous cell carcinoma. Acta Otolaryngol. 2014, 134, 753–759. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, Y.W.; Li, Y.; Zhang, J.W.; Zhang, T.; Fu, B.S.; Zhang, Q.; Jiang, N. Constitutive expression of Wnt/betacatenin target genes promotes proliferation and invasion of liver cancer stem cells. Mol. Med. Rep. 2016, 13, 3466–3474. [Google Scholar] [CrossRef]
- Zhuang, X.; Zhang, H.; Li, X.; Li, X.; Cong, M.; Peng, F.; Yu, J.; Zhang, X.; Yang, Q.; Hu, G. Differential effects on lung and bone metastasis of breast cancer by Wnt signalling inhibitor DKK1. Nat. Cell Biol. 2017, 19, 1274–1285. [Google Scholar] [CrossRef]
- Caneparo, L.; Huang, Y.L.; Staudt, N.; Tada, M.; Ahrendt, R.; Kazanskaya, O.; Niehrs, C.; Houart, C. Dickkopf-1 regulates gastrulation movements by coordinated modulation of Wnt/beta catenin and Wnt/PCP activities, through interaction with the Dally-like homolog Knypek. Genes Dev. 2007, 21, 465–480. [Google Scholar] [CrossRef][Green Version]
- Cha, S.W.; Tadjuidje, E.; Tao, Q.; Wylie, C.; Heasman, J. Wnt5a and Wnt11 interact in a maternal Dkk1-regulated fashion to activate both canonical and non-canonical signaling in Xenopus axis formation. Development 2008, 135, 3719–3729. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Yanagita, M.; Oka, M.; Watabe, T.; Iguchi, H.; Niida, A.; Takahashi, S.; Akiyama, T.; Miyazono, K.; Yanagisawa, M.; Sakurai, T. USAG-1: A bone morphogenetic protein antagonist abundantly expressed in the kidney. Biochem. Biophys. Res. Commun. 2004, 316, 490–500. [Google Scholar] [CrossRef]
- Itasaki, N.; Jones, C.M.; Mercurio, S.; Rowe, A.; Domingos, P.M.; Smith, J.C.; Krumlauf, R. Wise, a context-dependent activator and inhibitor of Wnt signalling. Development 2003, 130, 4295–4305. [Google Scholar] [CrossRef] [PubMed]
- de Lau, W.; Peng, W.C.; Gros, P.; Clevers, H. The R-spondin/Lgr5/Rnf43 module: Regulator of Wnt signal strength. Genes Dev. 2014, 28, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Nusse, R.; Clevers, H. Wnt/beta-catenin signaling, disease, and emerging therapeutic modalities. Cell 2017, 169, 985–999. [Google Scholar] [CrossRef] [PubMed]
- Seshagiri, S.; Stawiski, E.W.; Durinck, S.; Modrusan, Z.; Storm, E.E.; Conboy, C.B.; Chaudhuri, S.; Guan, Y.; Janakiraman, V.; Jaiswal, B.S.; et al. Recurrent R-spondin fusions in colon cancer. Nature 2012, 488, 660–664. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.K.; Spit, M.; Jordens, I.; Low, T.Y.; Stange, D.E.; van de Wetering, M.; van Es, J.H.; Mohammed, S.; Heck, A.J.; Maurice, M.M.; et al. Tumour suppressor RNF43 is a stem-cell E3 ligase that induces endocytosis of Wnt receptors. Nature 2012, 488, 665–669. [Google Scholar] [CrossRef]
- Hao, H.X.; Xie, Y.; Zhang, Y.; Charlat, O.; Oster, E.; Avello, M.; Lei, H.; Mickanin, C.; Liu, D.; Ruffner, H.; et al. ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature 2012, 485, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Inglis, D.J.; Licari, J.; Georgiou, K.R.; Wittwer, N.L.; Hamilton, R.W.; Beaumont, D.M.; Scherer, M.A.; Lavranos, T.C. Abstract 3910: Characterization of BNC101 a human specific monoclonal antibody targeting the GPCR LGR5: First-in-human evidence of target engagement. Cancer Res. 2018, 78 (Suppl. S13), 3910. [Google Scholar]
- Osada, T.; Chen, M.; Yang, X.Y.; Spasojevic, I.; Vandeusen, J.B.; Hsu, D.; Clary, B.M.; Clay, T.M.; Chen, W.; Morse, M.A.; et al. Antihelminth compound niclosamide downregulates Wnt signaling and elicits antitumor responses in tumors with activating APC mutations. Cancer Res. 2011, 71, 4172–4182. [Google Scholar] [CrossRef]
- Chen, M.; Wang, J.; Lu, J.; Bond, M.C.; Ren, X.R.; Lyerly, H.K.; Barak, L.S.; Chen, W. The anti-helminthic niclosamide inhibits Wnt/Frizzled1 signaling. Biochemistry 2009, 48, 10267–10274. [Google Scholar] [CrossRef]
- Lu, W.; Lin, C.; Roberts, M.J.; Waud, W.R.; Piazza, G.A.; Li, Y. Niclosamide suppresses cancer cell growth by inducing Wnt co-receptor LRP6 degradation and inhibiting the Wnt/beta-catenin pathway. PLoS ONE 2011, 6, e29290. [Google Scholar] [CrossRef]
- Wong, H.-C.; Bourdelas, A.; Krauss, A.; Lee, H.-J.; Shao, Y.; Wu, D.; Mlodzik, M.; Shi, D.-L.; Zheng, J. Direct binding of the PDZ domain of Dishevelled to a conserved internal sequence in the C-terminal region of Frizzled. Mol. Cell 2003, 12, 1251–1260. [Google Scholar] [CrossRef]
- Fiedler, M.; Mendoza-Topaz, C.; Rutherford, T.J.; Mieszczanek, J.; Bienz, M. Dishevelled interacts with the DIX domain polymerization interface of Axin to interfere with its function in down-regulating beta-catenin. Proc. Natl. Acad. Sci. USA 2011, 108, 1937–1942. [Google Scholar] [CrossRef] [PubMed]
- Grandy, D.; Shan, J.; Zhang, X.; Rao, S.; Akunuru, S.; Li, H.; Zhang, Y.; Alpatov, I.; Zhang, X.A.; Lang, R.A.; et al. Discovery and characterization of a small molecule inhibitor of the PDZ domain of dishevelled. J. Biol. Chem. 2009, 284, 16256–16263. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; You, L.; Xu, Z.; Uematsu, K.; Shan, J.; He, B.; Mikami, I.; Edmondson, L.R.; Neale, G.; Zheng, J.; et al. An antagonist of dishevelled protein-protein interaction suppresses beta-catenin-dependent tumor cell growth. Cancer Res. 2007, 67, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.; Shi, D.L.; Wang, J.; Zheng, J. Identification of a specific inhibitor of the dishevelled PDZ domain. Biochemistry 2005, 44, 15495–15503. [Google Scholar] [CrossRef]
- Zhang, Y.; Appleton, B.A.; Wiesmann, C.; Lau, T.; Costa, M.; Hannoush, R.N.; Sidhu, S.S. Inhibition of Wnt signaling by Dishevelled PDZ peptides. Nat. Chem. Biol. 2009, 5, 217–219. [Google Scholar] [CrossRef]
- Riffell, J.L.; Lord, C.J.; Ashworth, A. Tankyrase-targeted therapeutics: Expanding opportunities in the PARP family. Nat. Rev. Drug Discov. 2012, 11, 923–936. [Google Scholar] [CrossRef]
- Huang, S.M.; Mishina, Y.M.; Liu, S.; Cheung, A.; Stegmeier, F.; Michaud, G.A.; Charlat, O.; Wiellette, E.; Zhang, Y.; Wiessner, S.; et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 2009, 461, 614–620. [Google Scholar] [CrossRef]
- Huang, J.; Xiao, D.; Li, G.; Ma, J.; Chen, P.; Yuan, W.; Hou, F.; Ge, J.; Zhong, M.; Tang, Y.; et al. EphA2 promotes epithelial-mesenchymal transition through the Wnt/β-catenin pathway in gastric cancer cells. Oncogene 2014, 33, 2737–2747. [Google Scholar] [CrossRef]
- Lau, T.; Chan, E.; Callow, M.; Waaler, J.; Boggs, J.; Blake, R.A.; Magnuson, S.; Sambrone, A.; Schutten, M.; Firestein, R.; et al. A novel tankyrase small-molecule inhibitor suppresses APC mutation-driven colorectal tumor growth. Cancer Res. 2013, 73, 3132–3144. [Google Scholar] [CrossRef]
- Martins-Neves, S.R.; Paiva-Oliveira, D.I.; Fontes-Ribeiro, C.; Bovée, J.; Cleton-Jansen, A.M.; Gomes, C.M.F. IWR-1, a tankyrase inhibitor, attenuates Wnt/β-catenin signaling in cancer stem-like cells and inhibits in vivo the growth of a subcutaneous human osteosarcoma xenograft. Cancer Lett. 2018, 414, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Scarborough, H.A.; Helfrich, B.A.; Casás-Selves, M.; Schuller, A.G.; Grosskurth, S.E.; Kim, J.; Tan, A.C.; Chan, D.C.; Zhang, Z.; Zaberezhnyy, V.; et al. AZ1366: An inhibitor of tankyrase and the canonical Wnt pathway that limits the persistence of non-small cell lung cancer cells following EGFR inhibition. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 1531–1541. [Google Scholar] [CrossRef] [PubMed]
- Waaler, J.; Machon, O.; Tumova, L.; Dinh, H.; Korinek, V.; Wilson, S.R.; Paulsen, J.E.; Pedersen, N.M.; Eide, T.J.; Machonova, O.; et al. A novel tankyrase inhibitor decreases canonical Wnt signaling in colon carcinoma cells and reduces tumor growth in conditional APC mutant mice. Cancer Res. 2012, 72, 2822–2832. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Jiang, B.; Jiang, X.; Charlat, O.; Chen, A.; Mickanin, C.; Bauer, A.; Xu, W.; Yan, X.; Cong, F. The SIAH E3 ubiquitin ligases promote Wnt/beta-catenin signaling through mediating Wnt-induced Axin degradation. Genes Dev. 2017, 31, 904–915. [Google Scholar] [CrossRef]
- Ji, L.; Lu, B.; Zamponi, R.; Charlat, O.; Aversa, R.; Yang, Z.; Sigoillot, F.; Zhu, X.; Hu, T.; Reece-Hoyes, J.S.; et al. USP7 inhibits Wnt/beta-catenin signaling through promoting stabilization of Axin. Nat. Commun. 2019, 10, 4184. [Google Scholar] [CrossRef]
- Jung, Y.S.; Jun, S.; Kim, M.J.; Lee, S.H.; Suh, H.N.; Lien, E.M.; Jung, H.Y.; Lee, S.; Zhang, J.; Yang, J.I.; et al. TMEM9 promotes intestinal tumorigenesis through vacuolar-ATPase-activated Wnt/beta-catenin signalling. Nat. Cell Biol. 2018, 20, 1421–1433. [Google Scholar] [CrossRef]
- Thorne, C.A.; Hanson, A.J.; Schneider, J.; Tahinci, E.; Orton, D.; Cselenyi, C.S.; Jernigan, K.K.; Meyers, K.C.; Hang, B.I.; Waterson, A.G.; et al. Small-molecule inhibition of Wnt signaling through activation of casein kinase 1alpha. Nat. Chem. Biol. 2010, 6, 829–836. [Google Scholar] [CrossRef]
- Hwang, S.Y.; Deng, X.; Byun, S.; Lee, C.; Lee, S.J.; Suh, H.; Zhang, J.; Kang, Q.; Zhang, T.; Westover, K.D.; et al. Direct targeting of beta-catenin by a small molecule stimulates proteasomal degradation and suppresses oncogenic Wnt/beta-catenin signaling. Cell Rep. 2016, 16, 28–36. [Google Scholar] [CrossRef]
- Simonetta, K.R.; Taygerly, J.; Boyle, K.; Basham, S.E.; Padovani, C.; Lou, Y.; Cummins, T.J.; Yung, S.L.; von Soly, S.K.; Kayser, F.; et al. Prospective discovery of small molecule enhancers of an E3 ligase-substrate interaction. Nat. Commun. 2019, 10, 1402. [Google Scholar] [CrossRef]
- Goentoro, L.; Kirschner, M.W. Evidence that fold-change, and not absolute level, of beta-catenin dictates Wnt signaling. Mol. Cell 2009, 36, 872–884. [Google Scholar] [CrossRef]
- Cadigan, K.M.; Waterman, M.L. TCF/LEFs and Wnt signaling in the nucleus. Cold Spring Harb. Perspect. Biol. 2012, 4, a007906. [Google Scholar] [CrossRef] [PubMed]
- Gonsalves, F.C.; Klein, K.; Carson, B.B.; Katz, S.; Ekas, L.A.; Evans, S.; Nagourney, R.; Cardozo, T.; Brown, A.M.; DasGupta, R. An RNAi-based chemical genetic screen identifies three small-molecule inhibitors of the Wnt/wingless signaling pathway. Proc. Natl. Acad. Sci. USA 2011, 108, 5954–5963. [Google Scholar] [CrossRef] [PubMed]
- Sukhdeo, K.; Mani, M.; Zhang, Y.; Dutta, J.; Yasui, H.; Rooney, M.D.; Carrasco, D.E.; Zheng, M.; He, H.; Tai, Y.T.; et al. Targeting the beta-catenin/TCF transcriptional complex in the treatment of multiple myeloma. Proc. Natl. Acad. Sci. USA 2007, 104, 7516–7521. [Google Scholar] [CrossRef] [PubMed]
- Tian, W.; Han, X.; Yan, M.; Xu, Y.; Duggineni, S.; Lin, N.; Luo, G.; Li, Y.M.; Han, X.; Huang, Z.; et al. Structure-based discovery of a novel inhibitor targeting the beta-catenin/Tcf4 interaction. Biochemistry 2012, 51, 724–731. [Google Scholar] [CrossRef]
- Mahmoudi, T.; Li, V.S.; Ng, S.S.; Taouatas, N.; Vries, R.G.; Mohammed, S.; Heck, A.J.; Clevers, H. The kinase TNIK is an essential activator of Wnt target genes. EMBO J. 2009, 28, 3329–3340. [Google Scholar] [CrossRef]
- Masuda, M.; Uno, Y.; Ohbayashi, N.; Ohata, H.; Mimata, A.; Kukimoto-Niino, M.; Moriyama, H.; Kashimoto, S.; Inoue, T.; Goto, N.; et al. TNIK inhibition abrogates colorectal cancer stemness. Nat. Commun. 2016, 7, 12586. [Google Scholar] [CrossRef]
- Shitashige, M.; Satow, R.; Jigami, T.; Aoki, K.; Honda, K.; Shibata, T.; Ono, M.; Hirohashi, S.; Yamada, T. Traf2- and Nck-interacting kinase is essential for Wnt signaling and colorectal cancer growth. Cancer Res. 2010, 70, 5024–5033. [Google Scholar] [CrossRef]
- Lee, Y.; Jung, J.I.; Park, K.Y.; Kim, S.A.; Kim, J. Synergistic inhibition effect of TNIK inhibitor KY-05009 and receptor tyrosine kinase inhibitor dovitinib on IL-6-induced proliferation and Wnt signaling pathway in human multiple myeloma cells. Oncotarget 2017, 8, 41091–41101. [Google Scholar] [CrossRef]
- Ishitani, T.; Ninomiya-Tsuji, J.; Nagai, S.; Nishita, M.; Meneghini, M.; Barker, N.; Waterman, M.; Bowerman, B.; Clevers, H.; Shibuya, H. The TAK1-NLK-MAPK-related pathway antagonizes signalling between beta-catenin and transcription factor TCF. Nature 1999, 399, 798–802. [Google Scholar] [CrossRef]
- Ishitani, T.; Kishida, S.; Hyodo-Miura, J.; Ueno, N.; Yasuda, J.; Waterman, M.; Shibuya, H.; Moon, R.T.; Ninomiya-Tsuji, J.; Matsumoto, K. The TAK1-NLK mitogen-activated protein kinase cascade functions in the Wnt-5a/Ca(2+) pathway to antagonize Wnt/beta-catenin signaling. Mol. Cell Biol. 2003, 23, 131–139. [Google Scholar] [CrossRef]
- Hikasa, H.; Sokol, S.Y. Phosphorylation of TCF proteins by homeodomain-interacting protein kinase 2. J. Biol. Chem. 2011, 286, 12093–120100. [Google Scholar] [CrossRef] [PubMed]
- Hikasa, H.; Ezan, J.; Itoh, K.; Li, X.; Klymkowsky, M.W.; Sokol, S.Y. Regulation of TCF3 by Wnt-dependent phosphorylation during vertebrate axis specification. Dev. Cell 2010, 19, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Emami, K.H.; Nguyen, C.; Ma, H.; Kim, D.H.; Jeong, K.W.; Eguchi, M.; Moon, R.T.; Teo, J.L.; Kim, H.Y.; Moon, S.H.; et al. A small molecule inhibitor of beta-catenin/CREB-binding protein transcription [corrected]. Proc. Natl. Acad. Sci. USA 2004, 101, 12682–12687. [Google Scholar] [CrossRef] [PubMed]
- Miyabayashi, T.; Teo, J.L.; Yamamoto, M.; McMillan, M.; Nguyen, C.; Kahn, M. Wnt/beta-catenin/CBP signaling maintains long-term murine embryonic stem cell pluripotency. Proc. Natl. Acad. Sci. USA 2007, 104, 5668–5673. [Google Scholar] [CrossRef] [PubMed]
- de la Roche, M.; Rutherford, T.J.; Gupta, D.; Veprintsev, D.B.; Saxty, B.; Freund, S.M.; Bienz, M. An intrinsically labile alpha-helix abutting the BCL9-binding site of beta-catenin is required for its inhibition by carnosic acid. Nat. Commun. 2012, 3, 680. [Google Scholar] [CrossRef] [PubMed]
- Tam, B.Y.; Chiu, K.; Chung, H.; Bossard, C.; Nguyen, J.D.; Creger, E.; Eastman, B.W.; Mak, C.C.; Ibanez, M.; Ghias, A.; et al. The CLK inhibitor SM08502 induces anti-tumor activity and reduces Wnt pathway gene expression in gastrointestinal cancer models. Cancer Lett. 2020, 473, 186–197. [Google Scholar] [CrossRef]
- Bossard, C.; Chiu, K.; Chung, H.; Nguyen, J.D.; Creger, E.; Eastman, B.; Mak, C.C.; Do, L.; Cho, S.; KC, S. Effects of SM08502, a novel, oral small-molecule inhibitor of Wnt pathway signaling, on gene expression and antitumor activity in colorectal cancer (CRC) models. Am. Soc. Clin. Oncol. 2019, 37, e15185. [Google Scholar] [CrossRef]
- Bossard, C.; Cruz, N.; Eastman, B.; Mak, C.-C.; Sunil, K.; Tam, B.; Bucci, G.; Stewart, J.; Phalen, T.; Cha, S. Abstract A02: SM08502, a novel, small-molecule CDC-like kinase (CLK) inhibitor, downregulates the Wnt signaling pathway and demonstrates antitumor activity in pancreatic cancer cell lines and in vivo xenograft models. AACR 2019. [Google Scholar] [CrossRef]
- Pinto, D.; Gregorieff, A.; Begthel, H.; Clevers, H. Canonical Wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev. 2003, 17, 1709–1713. [Google Scholar] [CrossRef]
- Fevr, T.; Robine, S.; Louvard, D.; Huelsken, J. Wnt/beta-catenin is essential for intestinal homeostasis and maintenance of intestinal stem cells. Mol. Cell Biol. 2007, 27, 7551–7559. [Google Scholar] [CrossRef]
- Ito, M.; Yang, Z.; Andl, T.; Cui, C.; Kim, N.; Millar, S.E.; Cotsarelis, G. Wnt-dependent de novo hair follicle regeneration in adult mouse skin after wounding. Nature 2007, 447, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Duncan, A.W.; Rattis, F.M.; DiMascio, L.N.; Congdon, K.L.; Pazianos, G.; Zhao, C.; Yoon, K.; Cook, J.M.; Willert, K.; Gaiano, N.; et al. Integration of Notch and Wnt signaling in hematopoietic stem cell maintenance. Nat. Immunol. 2005, 6, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Schepers, A.; Clevers, H. Wnt signaling, stem cells, and cancer of the gastrointestinal tract. Cold Spring Harb. Perspect. Biol. 2012, 4, a007989. [Google Scholar] [CrossRef] [PubMed]
- Barker, N.; Tan, S.; Clevers, H. Lgr proteins in epithelial stem cell biology. Development 2013, 140, 2484–2494. [Google Scholar] [CrossRef] [PubMed]
- Junttila, M.R.; Mao, W.; Wang, X.; Wang, B.E.; Pham, T.; Flygare, J.; Yu, S.F.; Yee, S.; Goldenberg, D.; Fields, C.; et al. Targeting LGR5+ cells with an antibody-drug conjugate for the treatment of colon cancer. Sci. Transl. Med. 2015, 7, 314ra186. [Google Scholar] [CrossRef]
- de Sousa e Melo, F.; Kurtova, A.V.; Harnoss, J.M.; Kljavin, N.; Hoeck, J.D.; Hung, J.; Anderson, J.E.; Storm, E.E.; Modrusan, Z.; Koeppen, H.; et al. A distinct role for Lgr5(+) stem cells in primary and metastatic colon cancer. Nature 2017, 543, 676–680. [Google Scholar] [CrossRef]
- Shimokawa, M.; Ohta, Y.; Nishikori, S.; Matano, M.; Takano, A.; Fujii, M.; Date, S.; Sugimoto, S.; Kanai, T.; Sato, T. Visualization and targeting of LGR5(+) human colon cancer stem cells. Nature 2017, 545, 187–192. [Google Scholar] [CrossRef]
- Gong, X.; Azhdarinia, A.; Ghosh, S.C.; Xiong, W.; An, Z.; Liu, Q.; Carmon, K.S. LGR5-targeted antibody-drug conjugate eradicates gastrointestinal tumors and prevents recurrence. Mol. Cancer Ther. 2016, 15, 1580–1590. [Google Scholar] [CrossRef]
- Salik, B.; Yi, H.; Hassan, N.; Santiappillai, N.; Vick, B.; Connerty, P.; Duly, A.; Trahair, T.; Woo, A.J.; Beck, D.; et al. Targeting RSPO3-LGR4 signaling for leukemia stem cell eradication in acute myeloid leukemia. Cancer Cell 2020, 38, 263–278.e6. [Google Scholar] [CrossRef]
- Buchert, M.; Athineos, D.; Abud, H.E.; Burke, Z.D.; Faux, M.C.; Samuel, M.S.; Jarnicki, A.G.; Winbanks, C.E.; Newton, I.P.; Meniel, V.S.; et al. Genetic dissection of differential signaling threshold requirements for the Wnt/beta-catenin pathway in vivo. PLoS Genet. 2010, 6, e1000816. [Google Scholar] [CrossRef]
- Lenz, H.J.; Kahn, M. Safely targeting cancer stem cells via selective catenin coactivator antagonism. Cancer Sci. 2014, 105, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, D.J.; Barker, N.; Costanzo, N.S.D.; Mason, E.A.; Gurney, A.; Meniel, V.S.; Koushyar, S.; Austin, C.R.; Ernst, M.; Pearson, H.B.; et al. Frizzled-7 is required for Wnt signaling in gastric tumors with and without Apc mutations. Cancer Res. 2019, 79, 970–981. [Google Scholar] [CrossRef] [PubMed]
- Gurney, A.; Axelrod, F.; Bond, C.J.; Cain, J.; Chartier, C.; Donigan, L.; Fischer, M.; Chaudhari, A.; Ji, M.; Kapoun, A.M.; et al. Wnt pathway inhibition via the targeting of Frizzled receptors results in decreased growth and tumorigenicity of human tumors. Proc. Natl. Acad. Sci. USA 2012, 109, 11717–11722. [Google Scholar] [CrossRef] [PubMed]
- Diamond, J.R.; Becerra, C.; Richards, D.; Mita, A.; Osborne, C.; O’Shaughnessy, J.; Zhang, C.; Henner, R.; Kapoun, A.M.; Xu, L.; et al. Phase Ib clinical trial of the anti-frizzled antibody vantictumab (OMP-18R5) plus paclitaxel in patients with locally advanced or metastatic HER2-negative breast cancer. Breast Cancer Res. Treat. 2020, 184, 53–62. [Google Scholar] [CrossRef]
- Brabletz, T.; Jung, A.; Reu, S.; Porzner, M.; Hlubek, F.; Kunz-Schughart, L.A.; Knuechel, R.; Kirchner, T. Variable beta-catenin expression in colorectal cancers indicates tumor progression driven by the tumor environment. Proc. Natl. Acad. Sci. USA 2001, 98, 10356–10361. [Google Scholar] [CrossRef]
- Fodde, R.; Tomlinson, I. Nuclear beta-catenin expression and Wnt signalling: In defence of the dogma. J. Pathol. 2010, 221, 239–241. [Google Scholar] [CrossRef]
- Phelps, R.A.; Chidester, S.; Dehghanizadeh, S.; Phelps, J.; Sandoval, I.T.; Rai, K.; Broadbent, T.; Sarkar, S.; Burt, R.W.; Jones, D.A. A two-step model for colon adenoma initiation and progression caused by APC loss. Cell 2009, 137, 623–634. [Google Scholar] [CrossRef]
- Janssen, K.P.; Alberici, P.; Fsihi, H.; Gaspar, C.; Breukel, C.; Franken, P.; Rosty, C.; Abal, M.; El Marjou, F.; Smits, R.; et al. APC and oncogenic KRAS are synergistic in enhancing Wnt signaling in intestinal tumor formation and progression. Gastroenterology 2006, 131, 1096–1109. [Google Scholar] [CrossRef]
- Horst, D.; Chen, J.; Morikawa, T.; Ogino, S.; Kirchner, T.; Shivdasani, R.A. Differential WNT activity in colorectal cancer confers limited tumorigenic potential and is regulated by MAPK signaling. Cancer Res. 2012, 72, 1547–1556. [Google Scholar] [CrossRef]
- Mzoughi, S.; Zhang, J.; Hequet, D.; Teo, S.X.; Fang, H.; Xing, Q.R.; Bezzi, M.; Seah, M.K.Y.; Ong, S.L.M.; Shin, E.M.; et al. PRDM15 safeguards naive pluripotency by transcriptionally regulating WNT and MAPK-ERK signaling. Nat. Genet. 2017, 49, 1354–1363. [Google Scholar] [CrossRef]
- Jung, Y.S.; Jun, S.; Lee, S.H.; Sharma, A.; Park, J.I. Wnt2 complements Wnt/β-catenin signaling in colorectal cancer. Oncotarget 2015, 6, 37257–37268. [Google Scholar] [CrossRef] [PubMed]
- Tomar, V.S.; Patil, V.; Somasundaram, K. Temozolomide induces activation of Wnt/beta-catenin signaling in glioma cells via PI3K/Akt pathway: Implications in glioma therapy. Cell Biol. Toxicol. 2020, 36, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Prossomariti, A.; Piazzi, G.; Alquati, C.; Ricciardiello, L. Are Wnt/beta-catenin and PI3K/AKT/mTORC1 distinct pathways in colorectal cancer? Cell. Mol. Gastroenterol. Hepatol. 2020, 10, 491–506. [Google Scholar] [CrossRef] [PubMed]
- Shorning, B.Y.; Dass, M.S.; Smalley, M.J.; Pearson, H.B. The PI3K-AKT-mTOR pathway and prostate cancer: At the crossroads of AR, MAPK, and WNT signaling. Int. J. Mol. Sci. 2020, 21, 4507. [Google Scholar] [CrossRef]
- Zhong, Z.; Sepramaniam, S.; Chew, X.H.; Wood, K.; Lee, M.A.; Madan, B.; Virshup, D.M. PORCN inhibition synergizes with PI3K/mTOR inhibition in Wnt-addicted cancers. Oncogene 2019, 38, 6662–6677. [Google Scholar] [CrossRef]
- Jung, Y.S.; Stratton, S.A.; Lee, S.H.; Kim, M.J.; Jun, S.; Zhang, J.; Zheng, B.; Cervantes, C.L.; Cha, J.H.; Barton, M.C.; et al. TMEM9-v-ATPase activates Wnt/beta-catenin signaling via APC lysosomal degradation for liver regeneration and tumorigenesis. Hepatology 2020. [Google Scholar] [CrossRef]
- Voloshanenko, O.; Erdmann, G.; Dubash, T.D.; Augustin, I.; Metzig, M.; Moffa, G.; Hundsrucker, C.; Kerr, G.; Sandmann, T.; Anchang, B.; et al. Wnt secretion is required to maintain high levels of Wnt activity in colon cancer cells. Nat. Commun. 2013, 4, 2610. [Google Scholar] [CrossRef]
- Seino, T.; Kawasaki, S.; Shimokawa, M.; Tamagawa, H.; Toshimitsu, K.; Fujii, M.; Ohta, Y.; Matano, M.; Nanki, K.; Kawasaki, K.; et al. Human pancreatic tumor organoids reveal loss of stem cell niche factor dependence during disease progression. Cell Stem Cell 2018, 22, 454–467.e6. [Google Scholar] [CrossRef]
- Tammela, T.; Sanchez-Rivera, F.J.; Cetinbas, N.M.; Wu, K.; Joshi, N.S.; Helenius, K.; Park, Y.; Azimi, R.; Kerper, N.R.; Wesselhoeft, R.A.; et al. A Wnt-producing niche drives proliferative potential and progression in lung adenocarcinoma. Nature 2017, 545, 355–359. [Google Scholar] [CrossRef]
- Hou, P.; Ma, X.; Zhang, Q.; Wu, C.J.; Liao, W.; Li, J.; Wang, H.; Zhao, J.; Zhou, X.; Guan, C.; et al. USP21 deubiquitinase promotes pancreas cancer cell stemness via Wnt pathway activation. Genes Dev. 2019, 33, 1361–1366. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Wong, C.C.; Zhang, J.; Dong, Y.; Li, X.; Kang, W.; Chan, F.K.L.; Sung, J.J.Y.; Yu, J. RNF6 promotes colorectal cancer by activating the Wnt/beta-catenin pathway via ubiquitination of TLE3. Cancer Res. 2018, 78, 1958–1971. [Google Scholar] [CrossRef] [PubMed]
- Novellasdemunt, L.; Foglizzo, V.; Cuadrado, L.; Antas, P.; Kucharska, A.; Encheva, V.; Snijders, A.P.; Li, V.S.W. USP7 is a tumor-specific WNT activator for APC-mutated colorectal cancer by mediating beta-catenin deubiquitination. Cell Rep. 2017, 21, 612–627. [Google Scholar] [CrossRef] [PubMed]
- Gay, D.M.; Ridgway, R.A.; Muller, M.; Hodder, M.C.; Hedley, A.; Clark, W.; Leach, J.D.; Jackstadt, R.; Nixon, C.; Huels, D.J.; et al. Loss of BCL9/9l suppresses Wnt driven tumourigenesis in models that recapitulate human cancer. Nat. Commun. 2019, 10, 723. [Google Scholar] [CrossRef] [PubMed]
- Deka, J.; Wiedemann, N.; Anderle, P.; Murphy-Seiler, F.; Bultinck, J.; Eyckerman, S.; Stehle, J.C.; Andre, S.; Vilain, N.; Zilian, O.; et al. Bcl9/Bcl9l are critical for Wnt-mediated regulation of stem cell traits in colon epithelium and adenocarcinomas. Cancer Res. 2010, 70, 6619–6628. [Google Scholar] [CrossRef]
- Feng, M.; Jin, J.Q.; Xia, L.; Xiao, T.; Mei, S.; Wang, X.; Huang, X.; Chen, J.; Liu, M.; Chen, C.; et al. Pharmacological inhibition of β-catenin/BCL9 interaction overcomes resistance to immune checkpoint blockades by modulating T(reg) cells. Sci. Adv. 2019, 5, eaau5240. [Google Scholar] [CrossRef]
- Flanagan, D.J.; Barker, N.; Nowell, C.; Clevers, H.; Ernst, M.; Phesse, T.J.; Vincan, E. Loss of the Wnt receptor frizzled 7 in the mouse gastric epithelium is deleterious and triggers rapid repopulation in vivo. Dis. Models Mech. 2017, 10, 971–980. [Google Scholar] [CrossRef]
- Flanagan, D.J.; Phesse, T.J.; Barker, N.; Schwab, R.H.; Amin, N.; Malaterre, J.; Stange, D.E.; Nowell, C.J.; Currie, S.A.; Saw, J.T.; et al. Frizzled7 functions as a Wnt receptor in intestinal epithelial Lgr5(+) stem cells. Stem Cell Rep. 2015, 4, 759–767. [Google Scholar] [CrossRef]
- Jung, H.Y.; Jun, S.; Lee, M.; Kim, H.C.; Wang, X.; Ji, H.; McCrea, P.D.; Park, J.I. PAF and EZH2 induce Wnt/beta-catenin signaling hyperactivation. Mol. Cell 2013, 52, 193–205. [Google Scholar] [CrossRef]
- Kim, M.J.; Xia, B.; Suh, H.N.; Lee, S.H.; Jun, S.; Lien, E.M.; Zhang, J.; Chen, K.; Park, J.I. PAF-Myc-controlled cell stemness is required for intestinal regeneration and tumorigenesis. Dev. Cell 2018, 44, 582–596.e4. [Google Scholar] [CrossRef]
- Schutgens, F.; Clevers, H. Human organoids: Tools for understanding biology and treating diseases. Annu. Rev. Pathol. 2020, 15, 211–234. [Google Scholar] [CrossRef]
- Sato, T.; Vries, R.G.; Snippert, H.J.; van de Wetering, M.; Barker, N.; Stange, D.E.; van Es, J.H.; Abo, A.; Kujala, P.; Peters, P.J.; et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009, 459, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H. Modeling development and disease with organoids. Cell 2016, 165, 1586–1597. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xu, X.; Yang, L.; Zhu, J.; Wan, J.; Shen, L.; Xia, F.; Fu, G.; Deng, Y.; Pan, M.; et al. Patient-derived organoids predict chemoradiation responses of locally advanced rectal cancer. Cell Stem Cell 2020, 26, 17–26.e6. [Google Scholar] [CrossRef] [PubMed]
- Vlachogiannis, G.; Hedayat, S.; Vatsiou, A.; Jamin, Y.; Fernández-Mateos, J.; Khan, K.; Lampis, A.; Eason, K.; Huntingford, I.; Burke, R.; et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science 2018, 359, 920–926. [Google Scholar] [CrossRef]
- Nagle, P.W.; Plukker, J.T.M.; Muijs, C.T.; van Luijk, P.; Coppes, R.P. Patient-derived tumor organoids for prediction of cancer treatment response. Semin. Cancer Biol. 2018, 53, 258–264. [Google Scholar] [CrossRef]

| Agent | Mechanism | Trial | Cancer |
|---|---|---|---|
| LGK974 | PORCN inhibitor | NCT02278133 | BRAF V600-mutated metastatic colorectal cancer |
| Genistein | SFRP2 silencer inhibitor | NCT01985763 | Metastatic colorectal cancer |
| Foxy-5 | WNT5A mimic | Vermorken 2019 | WNT5A-negative colon cancer |
| DKN-01 | Monoclonal antibody against DKK1 | NCT03645980; NCT04166721 | Advanced hepatocellular carcinoma; Advanced gastroesophageal adenocarcinoma |
| Niclosamide | FZD1 inhibitor, LRP6 inhibitor | NCT02519582 | Progressed colorectal cancer |
| PRI-724 | β-catenin/CREBBP inhibitor | NCT02413853 | Metastatic colorectal adenocarcinoma |
| Chloroquine | v-ATPase inhibitor | NCT02496741 | Advanced solid malignancies, including intrahepatic cholangiocarcinoma |
| Hydroxy-chloroquine | v-ATPase inhibitor | NCT01006369, etc. (total 13 trials) | Advanced colorectal carcinoma; Advanced hepatocellular carcinoma; Advanced cholangiocarcinoma; Pancreatic adenocarcinoma |
| Trial | Agent | Mechanism | Design | Cancer | Interventions | Status |
|---|---|---|---|---|---|---|
| NCT02675946 | CGX1321 | Porcupine inhibitor | Phase I; Single group | Advanced GI cancers | CGX1321; CGX1321 + pembrolizumab | Recruiting |
| NCT03507998 | CGX1321 | Porcupine inhibitor | Phase I; Single group | Advanced GI cancers | CGX1321 | Recruiting |
| Ng 2017 (NCT02521844) [34] | ETC-159 | Porcupine inhibitor | Phase I; Single group | Advanced solid malignancies, including colorectal cancer, etc. | ETC-159; ETC-159 + pembrolizumab | Ongoing |
| NCT01351103 | LGK974 | Porcupine inhibitor | Phase I; Single group | Solid malignancies, including esophageal squamous-cell carcinoma, pancreatic adenocarcinoma, BRAF-mutated colorectal cancer, etc. | LGK974; LGK974 + spartalizumab | Recruiting |
| NCT02278133 | LGK974 | Porcupine inhibitor | Phase II; Single group | BRAF V600-mutated metastatic colorectal cancer with RNF43 mutations and/or R-spondin fusions | LGK974 + LGX818 + cetuximab | Completed |
| Pintova 2019 (NCT01985763) [35] | Genistein | SFRP2 silencer inhibitor | Phase II; Single group | Metastatic colorectal cancer | Genistein + FOLFOX; Genistein + FOLFOX + bevacizumab | Completed |
| Jimeno 2017 (NCT01608867) [36] | Ipafricept (OMP-54F28) | WNT decoy receptor | Phase I; Single group | Solid malignancies, including pancreatic cancer, colorectal cancer, etc. | Ipafricept | Completed |
| Dotan 2019 (NCT02050178) [37] | Ipafricept (OMP-54F28) | WNT decoy receptor | Phase I; Single group | Metastatic pancreatic ductal adenocarcinoma | Ipafricept + nab-paclitaxel + gemcitabine | Completed |
| NCT02069145 | Ipafricept (OMP-54F28) | WNT decoy receptor | Phase I; Single group | Advanced hepatocellular carcinoma | Ipafricept + sorafenib | Completed |
| NCT02020291 | Foxy-5 | WNT5A mimic | Phase I; Single group | Metastatic breast, colon, prostate cancer | Foxy-5 | Completed |
| NCT02655952 | Foxy-5 | WNT5A mimic | Phase I; Single group | Metastatic breast, colon, prostate cancer | Foxy-5 | Completed |
| Vermorken 2019 [38] | Foxy-5 | WNT5A mimic | Phase II; Randomized; Parallel | WNT5A-negative colon cancer | Foxy-5 vs placebo | Recruiting |
| Davis 2019 (NCT02005315) [39] | Vantictumab (OMP-18R5) | Monoclonal antibody against FZDs | Phase I; Single group | Metastatic pancreatic ductal adenocarcinoma | Vantictumab + nab-paclitaxel + gemcitabine | Terminated |
| Ryan 2016 (NCT02013154) [40] | DKN-01 | Monoclonal antibody against DKK1 | Phase I; Non-randomized; Parallel | Recurrent or metastatic esophageal cancer, gastro-esophageal junction cancer | DKN-01; DKN-01 vs paclitaxel; DKN-01 vs pembrolizumab | Ongoing |
| Eads 2016 (NCT02375880) [41] | DKN-01 | Monoclonal antibody against DKK1 | Phase I; Single group | Advanced cholangiocarcinoma | DKN-01 + gemcitabine + cisplatin | Ongoing |
| NCT03645980 | DKN-01 | Monoclonal antibody against DKK1 | Phase II; Non-randomized; Sequential | Advanced hepatocellular carcinoma | DKN-01 vs sequential DKN-01 + sorafenib | Recruiting |
| NCT04166721 | DKN-01 | Monoclonal antibody against DKK1 | Phase II; Single group | Advanced gastroesophageal adenocarcinoma | DKN-01 + atezolizumab | Recruiting |
| Bendell 2016 (NCT02482441) [42] | Rosmantuzumab (OMP-131R10) | Monoclonal antibody against RSPO3 | Phase I; Single group | Advanced solid malignancies, including metastatic colorectal cancer, etc. | OMP-131R10 | Completed |
| NIKOLO trial (NCT02519582) [43] | Niclosamide | FZD1 inhibitor, LRP6 inhibitor | Phase II; Single group | Progressed colorectal cancer | Niclosamide | Recruiting |
| NCT02687009 | Niclosamide | FZD1 inhibitor, LRP6 inhibitor | Phase I; Single group | Colorectal adenocarcinoma | Niclosamide | Terminated |
| NCT02726334 | BNC101 | Monoclonal antibody against LGR5 | Phase I; Single group | Metastatic colorectal cancer | BNC101; BNC101+ FOLFIRI | Terminated |
| NCT01777477 | Chloroquine | v-ATPase inhibitor | Phase I; Single group | Advanced pancreatic adenocarcinoma | Chloroquine + gemcitabine | Completed |
| Molenaar 2017 (NCT02496741) [44] | Chloroquine | v-ATPase inhibitor | Phase II; Single group | Advanced solid malignancies, including intrahepatic cholangiocarcinoma | Chloroquine + metformin | Completed |
| NCT01006369 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Non-randomized; Parallel | Metastatic colorectal carcinoma | Hydroxychloroquine + FOLFOX6 + bevacizumab vs Hydroxychloroquine + XELOX + bevacizumab | Completed |
| Mahalingam 2014 (NCT01023737) [45] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase I; Single group | Advanced solid malignancies, including colorectal cancer, etc. | Hydroxychloroquine + vorinostat | Completed |
| Boone 2015 (NCT01128296) [46] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Single group | Unresectable pancreatic ductal adenocarcinoma | Hydroxychloroquine + gemcitabine | Completed |
| Loaiza-Bonilla 2015 (NCT01206530) [47] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Single group | Advanced colorectal adenocarcinoma | Hydroxychloroquine + FOLFOX + bevacizumab | Completed |
| Wolpin 2014 (NCT01273805) [48] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase I; Single group | Metastatic pancreatic cancer | Hydroxychloroquine | Completed |
| Hong 2017 (NCT01494155) [49] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Single group | Early pancreatic ductal carcinoma | Short course radiation therapy preoperatively. Hydroxychloroquine + capecitabine postoperatively | Ongoing |
| Karasic 2019 (NCT01506973) [50] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Randomized; Parallel | Advanced pancreatic adenocarcinoma | Hydroxychloroquine + nab-paclitacel + gemcitabine vs nab-paclitacel + gemcitabine | Ongoing |
| NCT01978184 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Randomized; Parallel | Resectable pancreatic adenocarcinoma | Hydroxychloroquine + nab-paclitacel + gemcitabine vs nab-paclitacel + gemcitabine | Completed |
| NCT02013778 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Single group | Unresectable hepatocellular carcinoma | Hydroxychloroquine + transarterial chemoembolization | Terminated |
| Arora 2019 (NCT02316340) [51] | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Randomized; Crossover | Metastatic colorectal cancer | Hydroxychloroquine + vorinostat vs regorafenib | Completed |
| NCT03037437 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Non-randomized; Parallel | Advanced hepatocellular cancer | Hydroxychloroquine + sorafenib vs sorafenib | Ongoing |
| NCT03215264 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Single group | Metastatic colorectal cancer | Hydroxychloroquine + entinostat + regorafenib | Suspended |
| NCT03344172 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Randomized; Parallel | Resectable pancreatic adenocarcinoma | Hydroxychloroquine + gemcitabine + nab-paclitaxel + avelumab vs hydroxychloroquine + gemcitabine + nab-paclitaxel | Suspended |
| NCT03377179 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Single group | Advanced cholangiocarcinoma | ABC294640; Hydroxychloroquine + ABC294640 | Ongoing |
| NCT03825289 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase I; Single group | Advanced pancreatic cancer | Hydroxychloroquine + trametinib | Ongoing |
| NCT04132505 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase I; Single group | KRAS-mutated metastatic pancreatic adenocarcinoma | Hydroxychloroquine + binimetinib | Ongoing |
| NCT04145297 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase I; Single group | MAPK-mutated GI cancers | Hydroxychloroquine + ulixertinib | Ongoing |
| NCT04214418 | Hydroxy-chloroquine | v-ATPase inhibitor | Phase II; Non-randomized; Sequential | KRAS-mutated advanced solid malignancies, including pancreatic adenocarcinoma, colorectal adenocarcinoma, etc. | Hydroxychloroquine + atezolizumab + cobimetinib | Ongoing |
| El-Khoueiry 2013 (NCT01302405) [52] | PRI-724 | β-catenin/CREBBP inhibitor | Phase I; Single group | Advanced solid malignancies, including colorectal cancer, etc. | PRI-724 | Terminated |
| Ko 2016 (NCT01764477) [53] | PRI-724 | β-catenin/CREBBP inhibitor | Phase I; Single group | Recurrent or advanced pancreatic adenocarcinoma | PRI-724 + gemcitabine | Completed |
| NCT02413853 | PRI-724 | β-catenin/CREBBP inhibitor | Phase II; Randomized; Parallel | Metastatic colorectal adenocarcinoma | mFOLFOX6/Bevacizumab + PRI-724 vs mFOLFOX6/Bevacizumab | Withdrawn |
| NCT03355066 | SM08502 | CLK inhibitor | Phase I; Single group | Advanced solid malignancies, including pancreatic cancer, colorectal cancer, etc. | SM08502 | Recruiting |
| Mechanism | Agents |
|---|---|
| PORCN inhibitor | CGX1321, ETC-159, LGK974, GNF-6231, IWP-2, IWP-3, IWP-4, IWP-12, IWP-L6, IWP-O1, RXC004, WNT-C59 |
| SFRP1 inhibitor | WAY-316606 |
| SFRP2 silencer inhibitor | Genistein |
| WNT5A mimic | Foxy-5 |
| WNT inhibitor | Ant1.4Br/Ant1.4Cl, wogonin |
| WNT decoy receptor | Ipafricept |
| WNT3A-LRP5 complex inhibitor | APCDD1 |
| FZD inhibitor | Vantictumab |
| FZD1&LRP6 inhibitor | Niclosamide |
| FZD4 inhibitor | FzM1 |
| FZD7 inhibitor | Fz7-21 |
| FZD10 inhibitor | OTSA101, OTSA101-DTPA-90Y |
| LGR5 inhibitor | BNC101 |
| LRP6 inhibitor | Gigantol, salinomycin |
| FZD8-LRP6 heterodimer inhibitor | IGFBP-4 |
| DKK1 inhibitor | DKN-01 |
| DVL-PDZ domain inhibitor | Compound 3289-8625, FJ9, NSC668036, peptide Pen-N3 |
| RSPO3 inhibitor | Rosmantuzumab |
| TNKS inhibitor | 2X-121, AZ1366, AZ-6102, G007-LK, G244-LM, IWR-1, JW55, JW67, JW74, K-756, MN-64, MSC2504877, NVP-TNKS656, RK-287107, TC-E5001, WIKI4, XAV939 |
| v-ATPase inhibitor | Apicularen, archazolid, bafilomycin, chloroquine, chondropsine, concanamycin, cruentaren, disulfiramthe, FR167356, FR177995, FR202126, hydroxychloroquine, indolyl, KM91104, lobatamide, NiK12192, oximidine, salicylihamide, SB 242784, tributyltin chloride |
| CK1 activator | Pyrvinium |
| GSK3β fragment mimic | TCS 183 |
| β-catenin inhibitor | 21H7, isoquercitrin, KY1220, KYA1797K, triptonide (NSC 165677, PG 492) |
| β-catenin degrader | MSAB, NRX-252114 |
| β-catenin/TCF inhibitor | BC21, BC2059, CCT031374, CCT036477, CGP049090, CWP232228, ethacrynic acid, FH535, iCRT3, iCRT5, iCRT14, LF3, NLS-StAx-h, PKF115-584, PKF118-310, PKF118-744, PNU-74654, quercetin, ZTM000990 |
| TNIK inhibitor | KY-05009, NCB-0846 |
| β-catenin/EP300 inhibitor | IQ-1, windorphen, YH249/250 |
| β-catenin/CREBBP&EP300 inhibitor | C-82, ICG-001, PRI-724, retinoids, vitamin D3 |
| β-catenin/PYGO inhibitor | Pyrvinium |
| β-catenin/BCL9 inhibitor | Compound 22, carnosic acid, SAH-BCL9 |
| CLK inhibitor | SM08502 |
| Wnt/β-catenin signaling inhibitor | Adavivint (SM04690, lorecivivint), artesunate, cardamonin, cardionogen, CCT031374, diethyl benzylphosphonate, echinacoside, KY02111, pamidronic acid, specnuezhenide |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.J.; Huang, Y.; Park, J.-I. Targeting Wnt Signaling for Gastrointestinal Cancer Therapy: Present and Evolving Views. Cancers 2020, 12, 3638. https://doi.org/10.3390/cancers12123638
Kim MJ, Huang Y, Park J-I. Targeting Wnt Signaling for Gastrointestinal Cancer Therapy: Present and Evolving Views. Cancers. 2020; 12(12):3638. https://doi.org/10.3390/cancers12123638
Chicago/Turabian StyleKim, Moon Jong, Yuanjian Huang, and Jae-Il Park. 2020. "Targeting Wnt Signaling for Gastrointestinal Cancer Therapy: Present and Evolving Views" Cancers 12, no. 12: 3638. https://doi.org/10.3390/cancers12123638
APA StyleKim, M. J., Huang, Y., & Park, J.-I. (2020). Targeting Wnt Signaling for Gastrointestinal Cancer Therapy: Present and Evolving Views. Cancers, 12(12), 3638. https://doi.org/10.3390/cancers12123638





