Combination of Irreversible Electroporation and STING Agonist for Effective Cancer Immunotherapy
Simple Summary
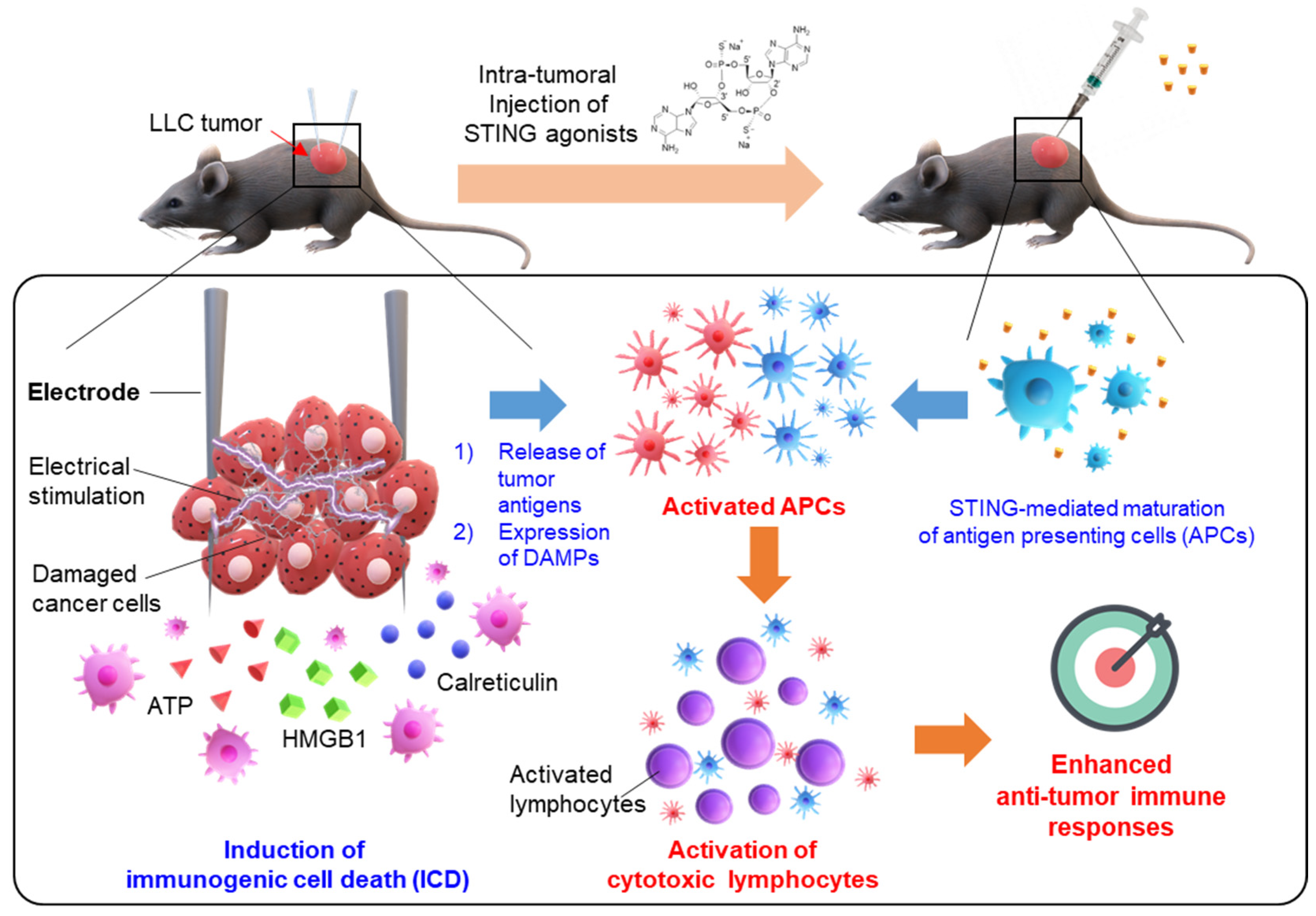
Abstract
1. Introduction
2. Materials and Methods
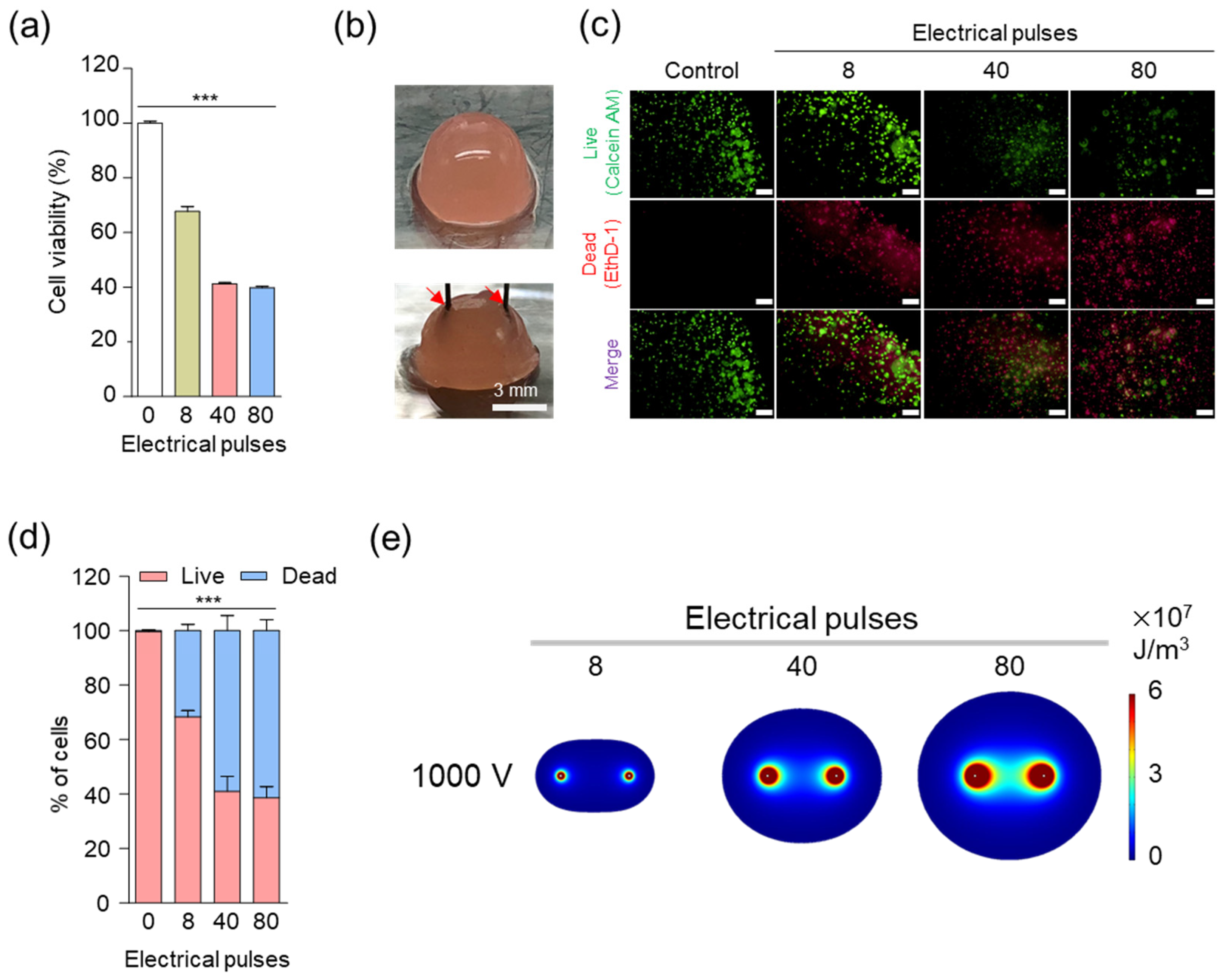
2.1. Finite Element Method (FEM) Simulation
2.2. In Vitro Cell Culture
2.3. In Vitro 3D Tumor Model
2.4. In Vitro IRE Treatment
2.5. In Vitro Cell Viability Analysis
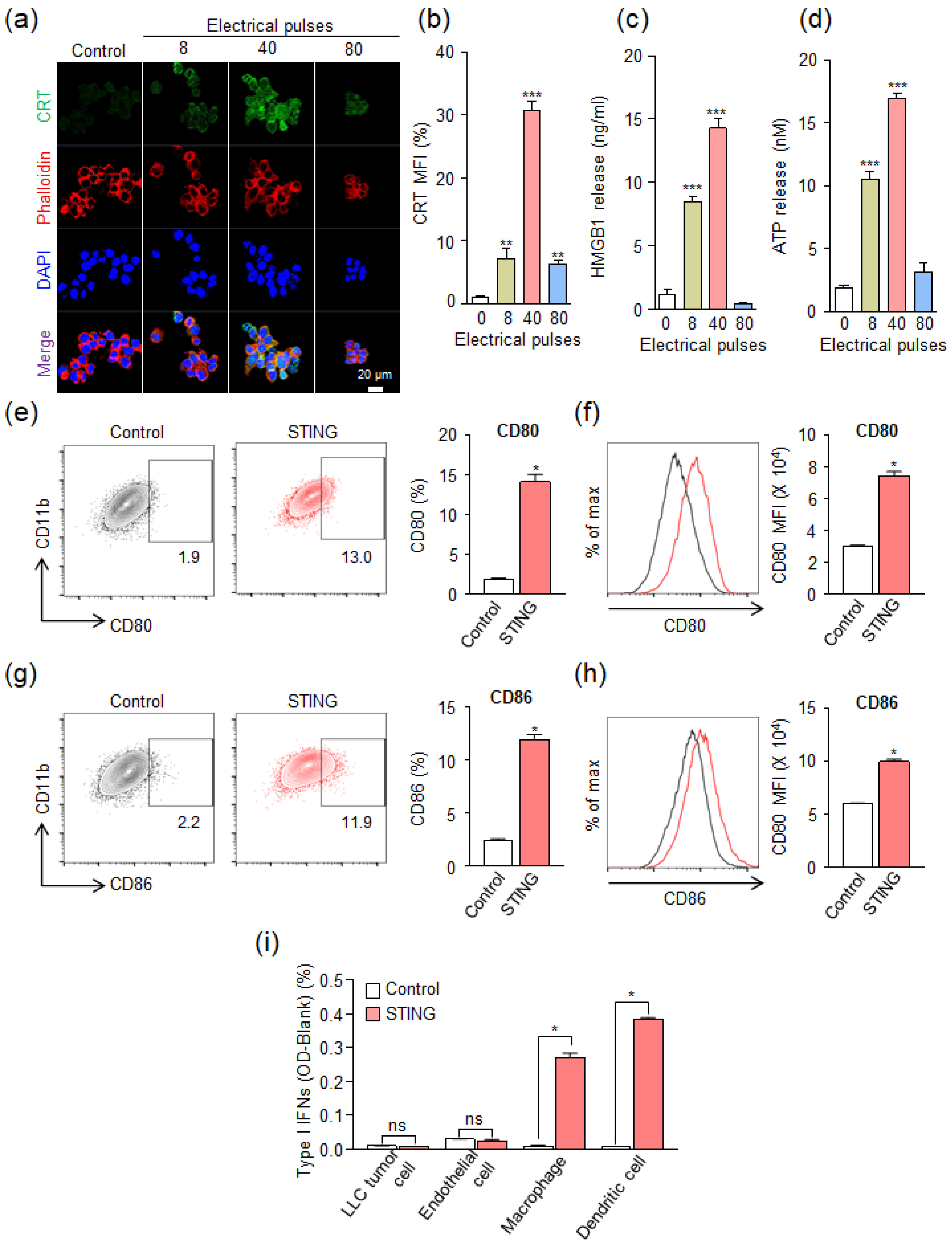
2.6. In Vitro Analysis of the Expression of DMAPs
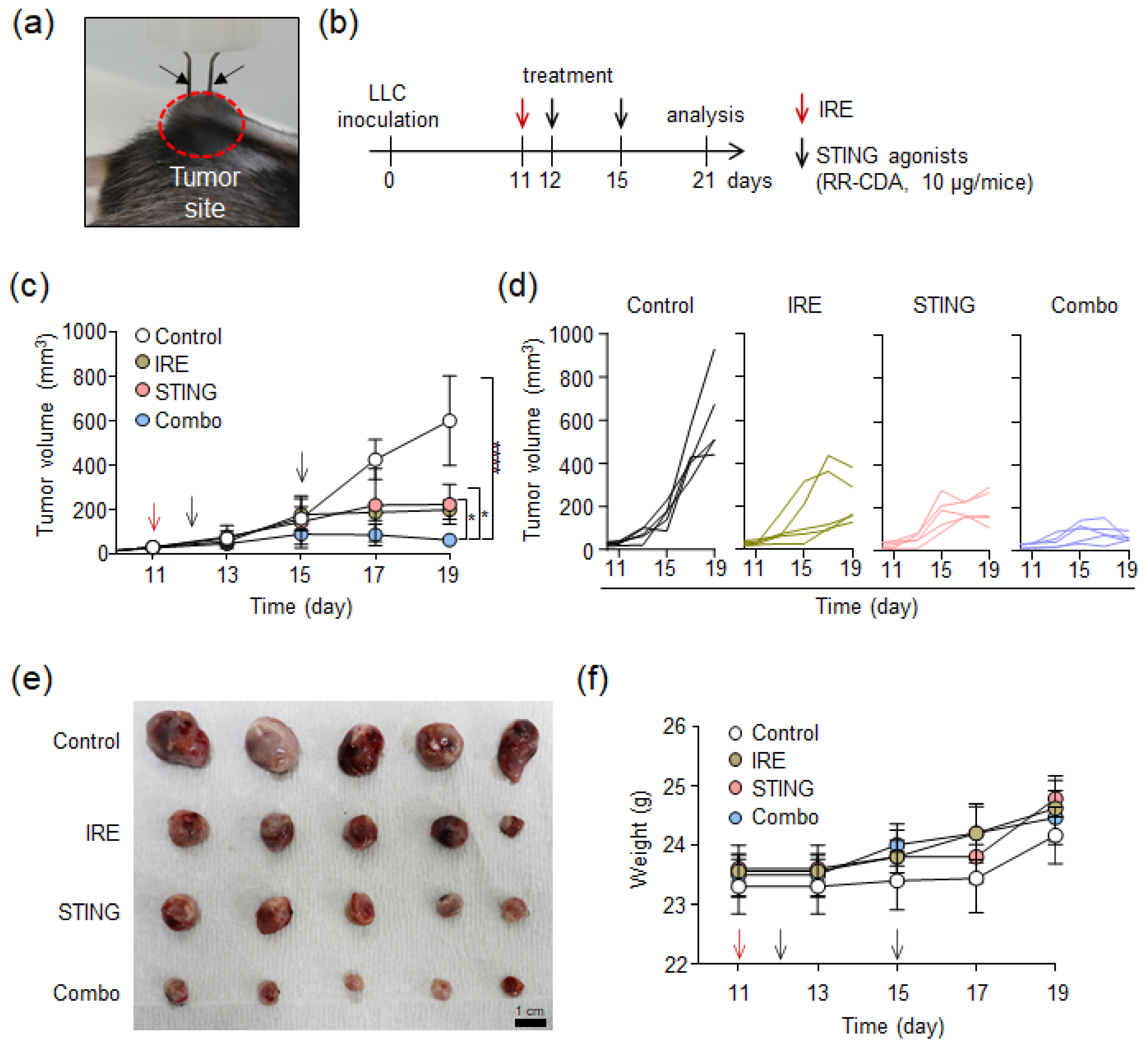
2.7. In Vivo Tumor Treatment with a Combination of IRE and STING Agonist
2.8. Tumor-Infiltrating Immune Cell Analysis Using Flow Cytometry
2.9. Statistical Analysis
3. Results and Discussion
3.1. In Vitro Cellular Experiments to Establish Electrical Conditions for IRE
3.2. In Vitro Immunogenic Cell Death Induced by IRE
3.3. In Vivo Tumor Suppression by Combinational Treatment with IRE and STING Agonist in an LLC Tumor Model
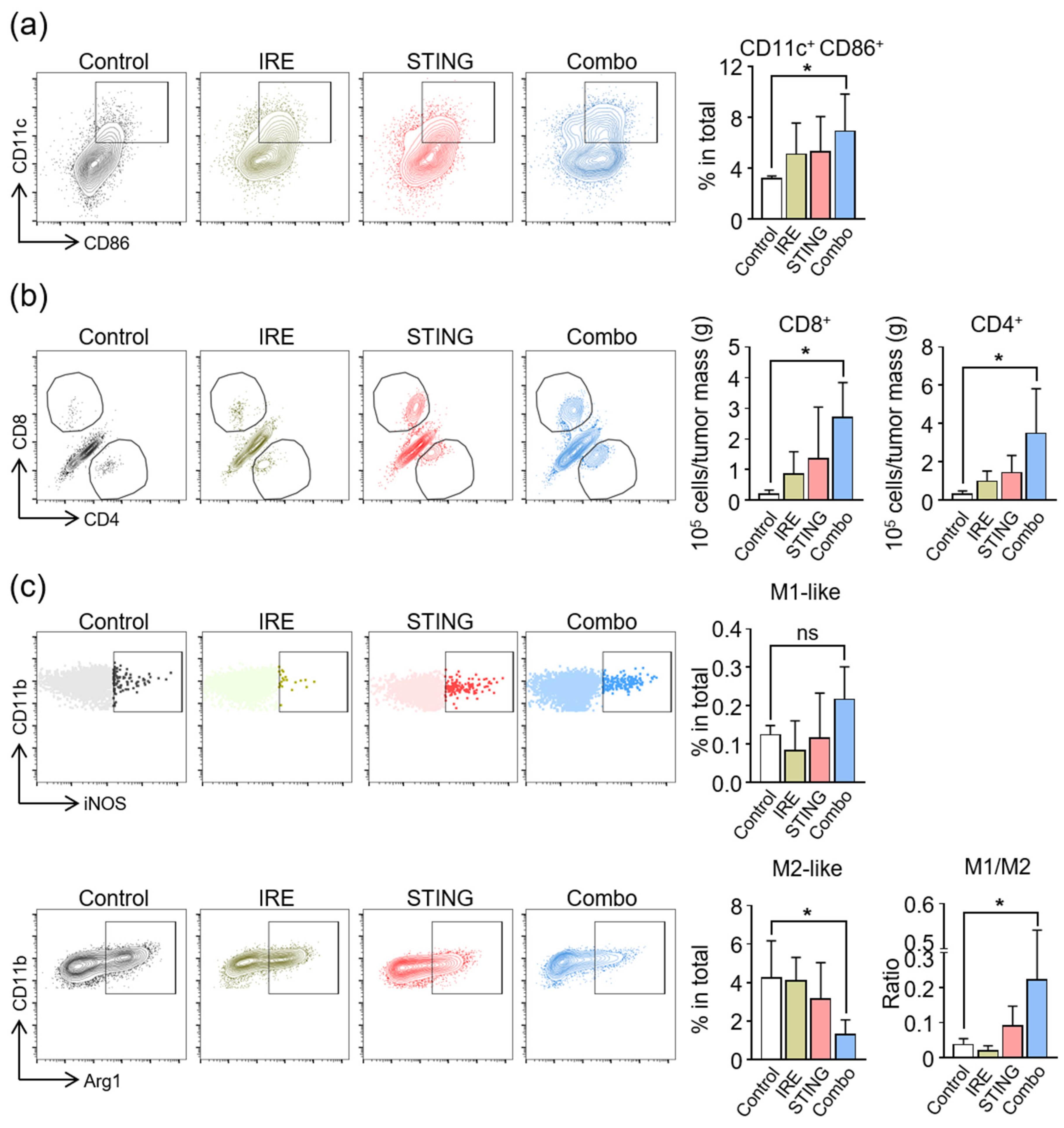
3.4. Induction of Anti-Cancer Immunity in TME by a Combination of IRE and STING Agonists
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ribas, A.; Wolchok, J.D. Cancer immunotherapy using checkpoint blockade. Science 2018, 359, 1350–1355. [Google Scholar] [CrossRef]
- Chen, D.S.; Mellman, I. Oncology meets immunology: The cancer-immunity cycle. Immunity 2013, 39, 1–10. [Google Scholar] [CrossRef]
- Gajewski, T.F.; Schreiber, H.; Fu, Y.-X. Innate and adaptive immune cells in the tumor microenvironment. Nat. Immunol. 2013, 14, 1014–1022. [Google Scholar] [CrossRef]
- Sharma, P.; Hu-Lieskovan, S.; Wargo, J.A.; Ribas, A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 2017, 168, 707–723. [Google Scholar] [CrossRef]
- Gajewski, T.F. The next hurdle in cancer immunotherapy: Overcoming the non-T-cell-inflamed tumor microenvironment. Semin. Oncol. 2015, 42, 663–671. [Google Scholar] [CrossRef]
- Park, W.; Song, K.H.; Lim, J.; Park, C.G.; Doh, J.; Han, D.K. Biomaterial-based strategies to prime dendritic cell-mediated anti-cancer immune responses. Int. Mater. Rev. 2020. [Google Scholar] [CrossRef]
- Park, W.; Gordon, A.C.; Cho, S.; Huang, X.; Harris, K.R.; Larson, A.C.; Kim, D.-H. Immunomodulatory magnetic microspheres for augmenting tumor-specific infiltration of natural killer (NK) cells. ACS Appl. Mater. Interfaces 2017, 9, 13819–13824. [Google Scholar] [CrossRef]
- Kim, K.-S.; Han, J.-H.; Park, J.-H.; Kim, H.-K.; Choi, S.H.; Kim, G.R.; Song, H.; An, H.J.; Han, D.K.; Park, W.; et al. Multifunctional nanoparticles for genetic engineering and bioimaging of natural killer (NK) cell therapeutics. Biomaterials 2019, 221, 119418. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.; Son, S.; Park, K.S.; Zou, W.; Shea, L.D.; Moon, J.J. Cancer nanomedicine for combination cancer immunotherapy. Nat. Rev. Mater. 2019, 4, 398–414. [Google Scholar] [CrossRef]
- Park, W.; Heo, Y.-J.; Han, D.K. New opportunities for nanoparticles in cancer immunotherapy. Biomater. Res. 2018, 22, 211–220. [Google Scholar] [CrossRef]
- Yim, H.; Park, W.; Kim, D.; Fahmy, T.M.; Na, K. A self-assembled polymeric micellar immunomodulator for cancer treatment based on cationic amphiphilic polymers. Biomaterials 2014, 35, 9912–9919. [Google Scholar] [CrossRef] [PubMed]
- Davalos, R.V.; Mir, L.; Rubinsky, B. Tissue ablation with irreversible electroporation. Ann. Biomed. Eng. 2005, 33, 223–231. [Google Scholar] [CrossRef]
- Rubinsky, B. Irreversible electroporation in medicine. Technol. Cancer Res. Treat. 2007, 6, 255–260. [Google Scholar] [CrossRef]
- Bassim Al-Sakere, F.A.; Bernat, C.; Connault, E.; Opolon, P.; Davalos, R.V.; Rubinsky, B.; Mir, L.M. Tumor ablation with irreversible electroporation. PLoS ONE 2007, 2, e1135. [Google Scholar]
- Miller, L.; Leor, J.; Rubinsky, B. Cancer cells ablation with irreversible electroporation. Technol. Ccancer Res. Treat. 2005, 4, 699–705. [Google Scholar] [CrossRef]
- Onik, G.; Mikus, P.; Rubinsky, B. Irreversible electroporation: Implications for prostate ablation. Technol. Cancer Res. Treat. 2007, 6, 295–300. [Google Scholar] [CrossRef]
- Thomson, K.R.; Cheung, W.; Ellis, S.J.; Federman, D.; Kavnoudias, H.; Loader-Oliver, D.; Roberts, S.; Evans, P.; Ball, C.; Haydon, A. Investigation of the safety of irreversible electroporation in humans. J. Vasc. Interv. Radiol. 2011, 22, 611–621. [Google Scholar] [CrossRef]
- Zhao, J.; Wen, X.; Tian, L.; Li, T.; Xu, C.; Wen, X.; Melancon, M.P.; Gupta, S.; Shen, B.; Peng, W.; et al. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat. Commun. 2019, 10, 899. [Google Scholar] [CrossRef]
- Vivas, I.; Iribarren, K.; Lozano, T.; Cano, D.; Lasarte-Cia, A.; Chocarro, S.; Gorraiz, M.; Sarobe, P.; Hervás-Stubbs, S.; Bilbao, J.I. Therapeutic Effect of Irreversible Electroporation in Combination with Poly-ICLC Adjuvant in Preclinical Models of Hepatocellular Carcinoma. J. Vasc. Interv. Radiol. 2019, 30, 1098–1105. [Google Scholar] [CrossRef]
- Scheffer, H.J.; Stam, A.G.; Geboers, B.; Vroomen, L.G.; Ruarus, A.; de Bruijn, B.; van den Tol, M.P.; Kazemier, G.; Meijerink, M.R.; de Gruijl, T.D. Irreversible electroporation of locally advanced pancreatic cancer transiently alleviates immune suppression and creates a window for antitumor T cell activation. Oncoimmunology 2019, 8, 1652532. [Google Scholar] [CrossRef]
- Martin, R.C.; Kwon, D.; Chalikonda, S.; Sellers, M.; Kotz, E.; Scoggins, C.; McMasters, K.M.; Watkins, K. Treatment of 200 locally advanced (stage III) pancreatic adenocarcinoma patients with irreversible electroporation: Safety and efficacy. Ann. Surg. 2015, 262, 486–494. [Google Scholar] [CrossRef]
- Cannon, R.; Ellis, S.; Hayes, D.; Narayanan, G.; Martin, R.C. Safety and early efficacy of irreversible electroporation for hepatic tumors in proximity to vital structures. J. Surg. Oncol. 2013, 107, 544–549. [Google Scholar] [CrossRef]
- Scheffer, H.J.; Nielsen, K.; de Jong, M.C.; van Tilborg, A.A.; Vieveen, J.M.; Bouwman, A.R.; Meijer, S.; van Kuijk, C.; van den Tol, P.M.; Meijerink, M.R. Irreversible electroporation for nonthermal tumor ablation in the clinical setting: A systematic review of safety and efficacy. J. Vasc. Interv. Radiol. 2014, 25, 997–1011. [Google Scholar] [CrossRef]
- Lin, M.; Alnaggar, M.; Liang, S.; Wang, X.; Liang, Y.; Zhang, M.; Chen, J.; Niu, L.; Xu, K. An important discovery on combination of irreversible electroporation and allogeneic natural killer cell immunotherapy for unresectable pancreatic cancer. Oncotarget 2017, 8, 101795–101807. [Google Scholar] [CrossRef]
- Yang, Y.; Qin, Z.; Du, D.; Wu, Y.; Qiu, S.; Mu, F.; Xu, K.; Chen, J. Safety and short-term efficacy of irreversible electroporation and allogenic natural killer cell immunotherapy combination in the treatment of patients with unresectable primary liver cancer. Cardiovasc. Interv. Radiol. 2019, 42, 48–59. [Google Scholar] [CrossRef]
- Woo, S.-R.; Fuertes, M.B.; Corrales, L.; Spranger, S.; Furdyna, M.J.; Leung, M.Y.; Duggan, R.; Wang, Y.; Barber, G.N.; Fitzgerald, K.A. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity 2014, 41, 830–842. [Google Scholar] [CrossRef]
- Corrales, L.; Gajewski, T.F. Molecular pathways: Targeting the stimulator of interferon genes (STING) in the immunotherapy of cancer. Clin. Cancer Res. 2015, 21, 4774–4779. [Google Scholar] [CrossRef]
- Iurescia, S.; Fioretti, D.; Rinaldi, M. Targeting cytosolic nucleic acid-sensing pathways for cancer immunotherapies. Front. Immunol. 2018, 9, 711. [Google Scholar] [CrossRef]
- Berger, G.; Marloye, M.; Lawler, S.E. Pharmacological modulation of the STING pathway for cancer immunotherapy. Trends Mol. Med. 2019, 25, 412–427. [Google Scholar] [CrossRef]
- Chon, H.J.; Kim, H.; Noh, J.H.; Yang, H.; Lee, W.S.; Kong, S.J.; Lee, S.J.; Lee, Y.S.; Kim, W.R.; Kim, J.H. STING signaling is a potential immunotherapeutic target in colorectal cancer. J. Cancer 2019, 10, 4932–4938. [Google Scholar] [CrossRef]
- Zhu, Y.; An, X.; Zhang, X.; Qiao, Y.; Zheng, T.; Li, X. STING: A master regulator in the cancer-immunity cycle. Mol. Cancer 2019, 18, 152. [Google Scholar] [CrossRef]
- Demaria, O.; De Gassart, A.; Coso, S.; Gestermann, N.; Di Domizio, J.; Flatz, L.; Gaide, O.; Michielin, O.; Hwu, P.; Petrova, T.V. STING activation of tumor endothelial cells initiates spontaneous and therapeutic antitumor immunity. Proc. Natl. Acad. Sci. USA 2015, 112, 15408–15413. [Google Scholar] [CrossRef]
- Yang, H.; Lee, W.S.; Kong, S.J.; Kim, C.G.; Kim, J.H.; Chang, S.K.; Kim, S.; Kim, G.; Chon, H.J.; Kim, C. STING activation reprograms tumor vasculatures and synergizes with VEGFR2 blockade. J. Clin. Investig. 2019, 129, 4350–4364. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; McAllister, D.; Vonderhaar, E.P.; Palen, K.; Riese, M.J.; Gershan, J.; Johnson, B.D.; Dwinell, M.B. STING agonist inflames the pancreatic cancer immune microenvironment and reduces tumor burden in mouse models. J. Immunother. Cancer 2019, 7, 115. [Google Scholar] [CrossRef]
- Rolong, A.; Prokop, K.J.; Davalos, R.V. Impact of the Use of Nanoparticles on Electric Field Distribution during Irreversible Electroporation Treatments: Can the Lesion be Enhanced Beyond IRE Margin? In Proceedings of the 6th European Conference of the International Federation for Medical and Biological Engineering, Dubrovnik, Croatia, 7–11 September 2014; Springer: Berlin/Heidelberg, Germany, 2015; pp. 793–796. [Google Scholar]
- Sung, C.K.; Kim, H.B.; Jung, J.H.; Baik, K.Y.; Moon, K.W.; Kim, H.-S.; Yi, J.-H.; Chung, J.H. Histological and mathematical analysis of the irreversibly electroporated liver tissue. Technol. Cancer Res. Treat. 2017, 16, 488–496. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, Y.; Ragin, A.B.; Lewandowski, R.J.; Yang, G.-Y.; Nijm, G.M.; Sahakian, A.V.; Omary, R.A.; Larson, A.C.J.R. MR imaging to assess immediate response to irreversible electroporation for targeted ablation of liver tissues: Preclinical feasibility studies in a rodent model. Radiology 2010, 256, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Sano, M.B.; Arena, C.B.; Bittleman, K.R.; DeWitt, M.R.; Cho, H.J.; Szot, C.S.; Saur, D.; Cissell, J.M.; Robertson, J.; Lee, Y.W. Bursts of bipolar microsecond pulses inhibit tumor growth. Sci. Rep. 2015, 5, 14999. [Google Scholar] [CrossRef] [PubMed]
- Ivey, J.; Wasson, E.; Alinezhadbalalami, N.; Kanitkar, A.; Debinski, W.; Sheng, Z.; Davalos, R.; Verbridge, S. Characterization of ablation thresholds for 3D-cultured patient-derived glioma stem cells in response to high-frequency irreversible electroporation. Research 2019, 8081315. [Google Scholar] [CrossRef]
- Sundararajan, R.; Poompavai, S.; Madhivanan, S.; Prabakaran, S.; Camarillo, I.G. Electro therapies for sarcomas. J. Cancer Prev. Curr. Res. 2019, 10, 119–124. [Google Scholar] [CrossRef]
- Jung, B.; Hong, S.; Kim, S.C.; Hwang, C. In Vivo Observation of Endothelial Cell-Assisted Vascularization in Pancreatic Cancer Xenograft Engineering. Tissue Eng. Regen. Med. 2018, 15, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, S.-H.; Lee, B.-K.; Park, S.-H.; Cho, Y.-S.; Park, Y. Fabrication of Microchannels and Evaluation of Guided Vascularization in Biomimetic Hydrogels. Tissue Eng. Regen. Med. 2018, 15, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; Park, K.D. In Situ Cross-Linkable Hydrogels as a Dynamic Matrix for Tissue Regenerative Medicine. Tissue Eng. Regen. Med. 2018, 15, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, G.; Chen, Y.; Wang, H.; Hua, Y.; Cai, Z. Immunogenic cell death in cancer therapy: Present and emerging inducers. J. Cell. Mol. Med. 2019, 23, 4854–4865. [Google Scholar] [CrossRef]
- Nuccitelli, R.; McDaniel, A.; Anand, S.; Cha, J.; Mallon, Z.; Berridge, J.C.; Uecker, D. Nano-Pulse Stimulation is a physical modality that can trigger immunogenic tumor cell death. J. Immunother. Cancer 2017, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Xiong, Z.; Liu, Y.; Yao, C.; Li, C. Low voltage irreversible electroporation induced apoptosis in HeLa cells. J. Cancer Res. Ther. 2012, 8, 80–85. [Google Scholar] [PubMed]
- Diederich, M. Natural compound inducers of immunogenic cell death. Arch. Pharm. Res. 2019, 42, 629–645. [Google Scholar] [CrossRef]
- Le, Q.-V.; Choi, J.; Oh, Y.-K. Nano delivery systems and cancer immunotherapy. J. Pharm. Investig. 2018, 48, 527–539. [Google Scholar] [CrossRef]
- Kuai, R.; Yuan, W.; Son, S.; Nam, J.; Xu, Y.; Fan, Y.; Schwendeman, A.; Moon, J.J. Elimination of established tumors with nanodisc-based combination chemoimmunotherapy. Sci. Adv. 2018, 4, eaao1736. [Google Scholar] [CrossRef]
- Corrales, L.; Glickman, L.H.; McWhirter, S.M.; Kanne, D.B.; Sivick, K.E.; Katibah, G.E.; Woo, S.-R.; Lemmens, E.; Banda, T.; Leong, J.J. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 2015, 11, 1018–1030. [Google Scholar] [CrossRef]
- Ager, C.R.; Reilley, M.J.; Nicholas, C.; Bartkowiak, T.; Jaiswal, A.R.; Curran, M.A. Intratumoral STING activation with T-cell checkpoint modulation generates systemic antitumor immunity. Cancer Immunol. Res. 2017, 5, 676–684. [Google Scholar] [CrossRef]
- Ohkuri, T.; Kosaka, A.; Nagato, T.; Kobayashi, H. Effects of STING stimulation on macrophages: STING agonists polarize into “classically” or “alternatively” activated macrophages? Hum. Vaccines Immunother. 2018, 14, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, S.; Liu, Y.-H.; Fang, Z.-S.; Lin, C.-L.; Lin, J.-C.; Yao, B.-Y.; Hu, C.-M.J. Synthetic Immunogenic Cell Death Mediated by Intracellular Delivery of STING Agonist Nanoshells Enhances Anticancer Chemo-immunotherapy. Nano Lett. 2020, 20, 2246–2256. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Crowe, W.N.; Wang, L.; Lu, Y.; Petty, W.J.; Habib, A.A.; Zhao, D. An inhalable nanoparticulate STING agonist synergizes with radiotherapy to confer long-term control of lung metastases. Nat. Commun. 2019, 10, 5108. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Go, E.-J.; Yang, H.; Chon, H.J.; Yang, D.; Ryu, W.; Kim, D.-H.; Han, D.K.; Kim, C.; Park, W. Combination of Irreversible Electroporation and STING Agonist for Effective Cancer Immunotherapy. Cancers 2020, 12, 3123. https://doi.org/10.3390/cancers12113123
Go E-J, Yang H, Chon HJ, Yang D, Ryu W, Kim D-H, Han DK, Kim C, Park W. Combination of Irreversible Electroporation and STING Agonist for Effective Cancer Immunotherapy. Cancers. 2020; 12(11):3123. https://doi.org/10.3390/cancers12113123
Chicago/Turabian StyleGo, Eun-Jin, Hannah Yang, Hong Jae Chon, DaSom Yang, WonHyoung Ryu, Dong-Hyun Kim, Dong Keun Han, Chan Kim, and Wooram Park. 2020. "Combination of Irreversible Electroporation and STING Agonist for Effective Cancer Immunotherapy" Cancers 12, no. 11: 3123. https://doi.org/10.3390/cancers12113123
APA StyleGo, E.-J., Yang, H., Chon, H. J., Yang, D., Ryu, W., Kim, D.-H., Han, D. K., Kim, C., & Park, W. (2020). Combination of Irreversible Electroporation and STING Agonist for Effective Cancer Immunotherapy. Cancers, 12(11), 3123. https://doi.org/10.3390/cancers12113123







