ELAVL1 Role in Cell Fusion and Tunneling Membrane Nanotube Formations with Implication to Treat Glioma Heterogeneity
Simple Summary
Abstract
1. Introduction
2. Historical Background
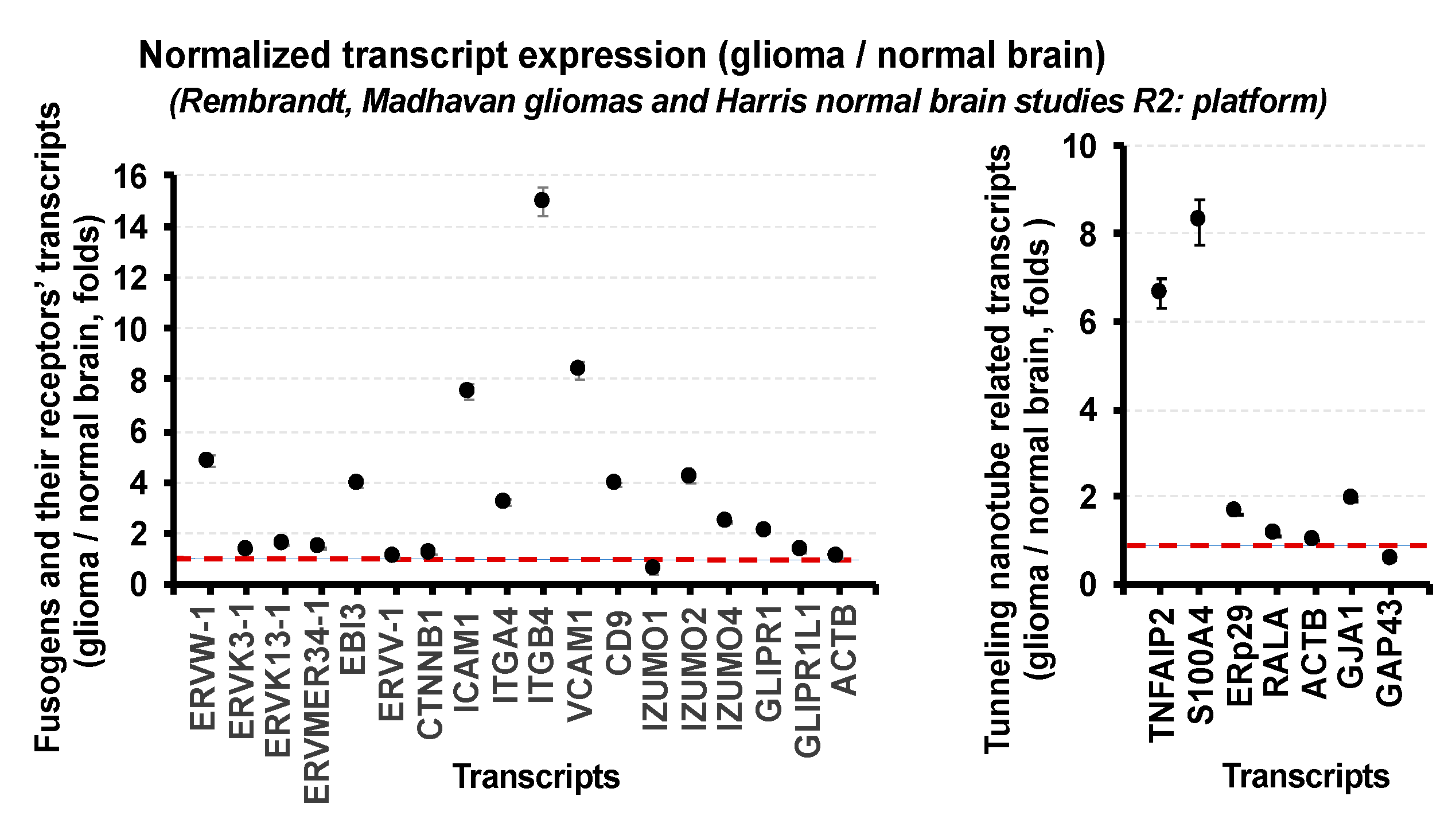
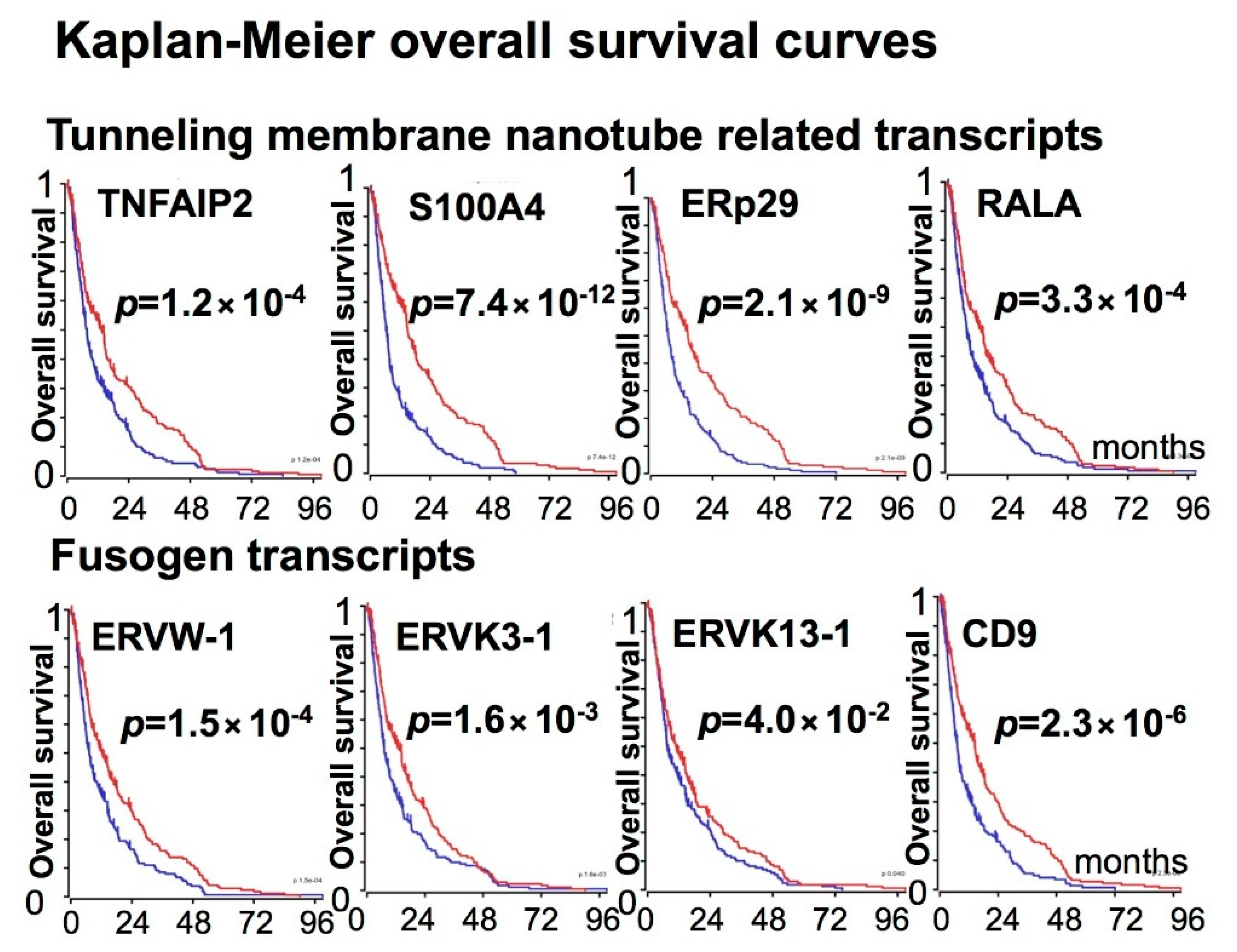
3. Expression of Fusogens, Fusogen Receptors, and Tunneling Membrane Nanotube-Related Transcripts in Gliomas
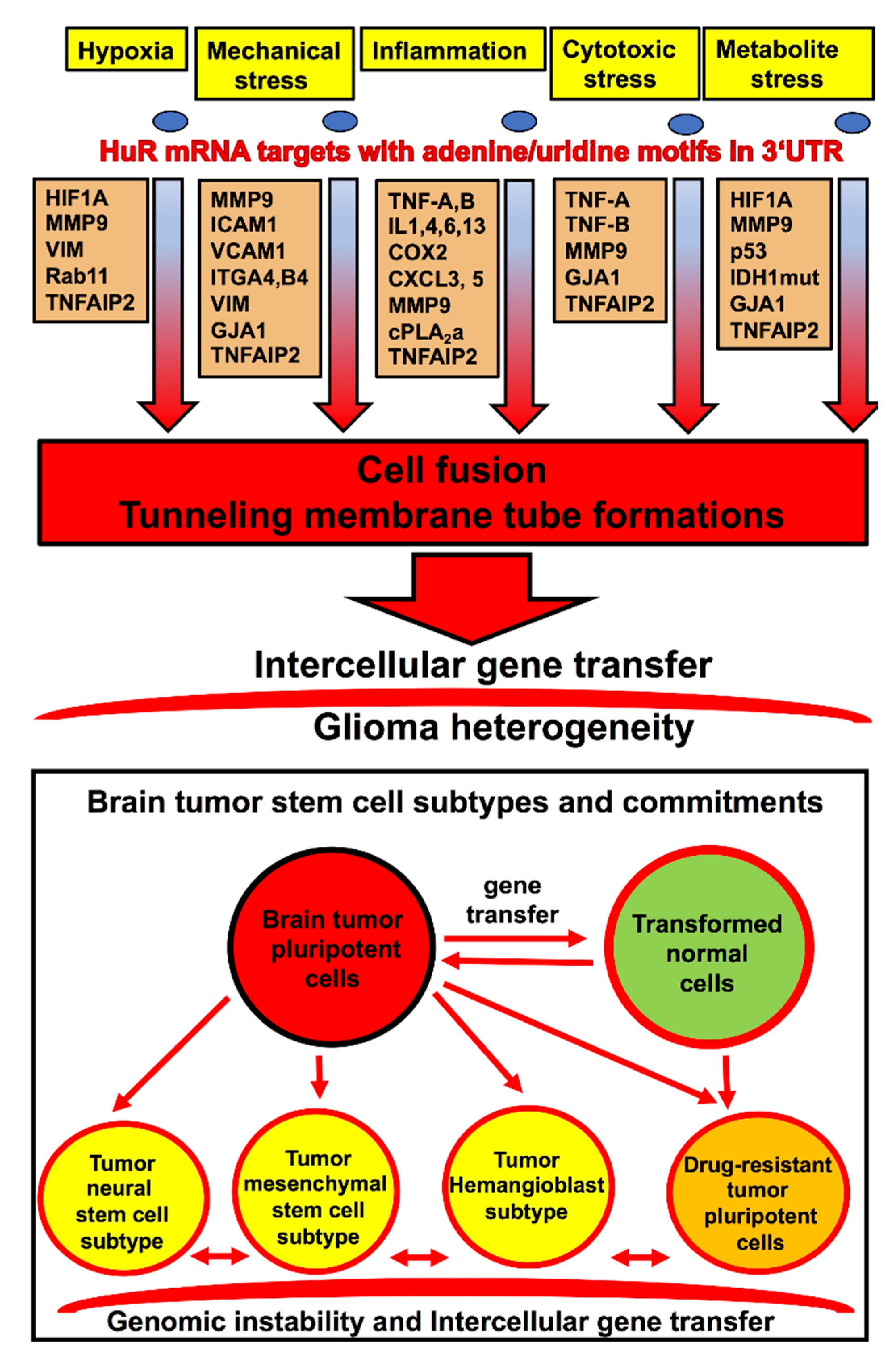
4. HuR-Dependent Cell-Signaling Pathways of Cell Fusion and Tunneling Nanotube Formations Leading to Glioma Heterogeneity
5. Potential Pharmacological Modulators of Cell Fusion
6. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | Arachidonic Acid |
| CNS | Central Nervous System |
| CARs | T cells expressing Chimeric Antigen Receptors |
| cPLA2α | cytosolic Phospholipase A2-alpha |
| COX2 | Cytochrome Oxidase Subunit 2 |
| FDA | Food and Drug Administration |
| GEO | Gene Expression Omnibus repository database |
| HCQ | Hydroxychloroquine |
| MCD | Methyl-β-cyclodextrin |
| Novantrone | mitoxantrone hydrochloride |
| NSAIDs | Non-Steroid Anti-Inflammatory Drugs |
| Optune treatment | an application of the electric field, which induces apoptosis in rapidly dividing tumor cells |
| PDGx | Patient-Derived Glioma Xenografts |
| PGE2 | Prostaglandin E2 |
| PG | Prostaglandins |
| ROSc | Reactive Oxygen Species |
| R2: | Genomic Analysis and Visualization platform R: |
| TMZ | Temozolomide |
| TNFAIP2 | TNF Alpha Induced Protein 2 |
| TNT | Tunneling Membrane Nanotubes |
| MT | Membrane Microtubes |
| WASP | Wiskott-Aldrich Syndrome Protein |
References
- Parker, N.R.; Ekhong, P.; Parkinson, J.F.; Howell, V.M.; Wheeler, H.R. Molecular Heterogeneity in Glioblastoma: Potential Clinical Implications. Front. Oncol. 2015, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; Von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Walid, M.S. Prognostic Factors for Long-Term Survival after Glioblastoma. Perm. J. 2008, 12, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, A.B.; Yabroff, K.R.; Shao, Y.; Feuer, E.J.; Brown, M.L. Projections of the Cost of Cancer Care in the United States: 2010–2020. J. Natl. Cancer Inst. 2011, 103, 117–128. [Google Scholar] [CrossRef]
- Rusthoven, C.G.; Koshy, M.; Sher, D.J.; Perry, J.R.; Laperriere, N.; Mason, W.P. Radiation plus Temozolomide in Patients with Glioblastoma. N. Engl. J. Med. 2017, 376, 2195–2197. [Google Scholar] [CrossRef] [PubMed]
- Tamimi, A.F.; Juweid, M. Epidemiology and Outcome of Glioblastoma. Glioblastoma 2017, 8, 143–153. [Google Scholar] [CrossRef]
- Bagley, S.J.; Desai, A.S.; Linette, G.P.; June, C.H.; O’Rourke, D.M. CAR T-cell therapy for glioblastoma: Recent clinical advances and future challenges. Neuro-Oncology 2018, 20, 1429–1438. [Google Scholar] [CrossRef] [PubMed]
- Branter, J.; Basu, S.; Smith, S. Tumour treating fields in a combinational therapeutic approach. Oncotarget 2018, 9, 36631–36644. [Google Scholar] [CrossRef]
- Mukherjee, P.; Augur, Z.M.; Li, M.; Hill, C.; Greenwood, B.; Domin, M.A.; Kondakci, G.; Narain, N.R.; Kiebish, M.A.; Bronson, R.T.; et al. Therapeutic benefit of combining calorie-restricted ketogenic diet and glutamine targeting in late-stage experimental glioblastoma. Commun. Biol. 2019, 2, 1–14. [Google Scholar] [CrossRef]
- Hernández, J.M.; Podbilewicz, B. The hallmarks of cell-cell fusion. Development 2017, 144, 4481–4495. [Google Scholar] [CrossRef]
- Sapir, A.; Avinoam, O.; Podbilewicz, B.; Chernomordik, L.V. Viral and Developmental Cell Fusion Mechanisms: Conservation and Divergence. Dev. Cell 2008, 14, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Segev, N.; Avinoam, O.; Podbilewicz, B. Fusogens. Curr. Biol. 2018, 28, R378–R380. [Google Scholar] [CrossRef] [PubMed]
- Rustom, A.; Saffrich, R.; Markovic, I.; Walther, P.; Gerdes, H.-H. Nanotubular Highways for Intercellular Organelle Transport. Science 2004, 303, 1007–1010. [Google Scholar] [CrossRef] [PubMed]
- Jash, E.; Prasad, P.; Kumar, N.; Sharma, T.; Goldman, A.; Sehrawat, S. Perspective on nanochannels as cellular mediators in different disease conditions. Cell Commun. Signal. 2018, 16, 76. [Google Scholar] [CrossRef]
- Sowinski, S.; Jolly, C.; Berninghausen, O.; Purbhoo, M.A.; Chauveau, A.; Köhler, K.; Oddos, S.; Eissmann, P.; Brodsky, F.M.; Hopkins, C.; et al. Membrane nanotubes physically connect T cells over long distances presenting a novel route for HIV-1 transmission. Nat. Cell Biol. 2008, 10, 211–219. [Google Scholar] [CrossRef]
- Abounit, S.; Bousset, L.; Loria, F.; Zhu, S.; De Chaumont, F.; Pieri, L.; Olivo-Marin, J.; Melki, R.; Zurzolo, C. Tunneling nanotubes spread fibrillar α-synuclein by intercellular trafficking of lysosomes. EMBO J. 2016, 35, 2120–2138. [Google Scholar] [CrossRef]
- Wang, X.; Yu, X.; Xie, C.; Tan, Z.; Tian, Q.; Zhu, D.; Liu, M.; Guan, Y. Rescue of Brain Function Using Tunneling Nanotubes Between Neural Stem Cells and Brain Microvascular Endothelial Cells. Mol. Neurobiol. 2015, 53, 2480–2488. [Google Scholar] [CrossRef]
- Ahmad, T.; Mukherjee, S.; Pattnaik, B.; Kumar, M.; Singh, S.; Rehman, R.; Tiwari, B.K.; Jha, K.A.; Barhanpurkar, A.P.; Wani, M.R.; et al. Miro1 regulates intercellular mitochondrial transport & enhances mesenchymal stem cell rescue efficacy. EMBO J. 2014, 33, 994–1010. [Google Scholar] [CrossRef]
- Torralba, D.; Baixauli, F.; Sánchez-Madrid, F. Mitochondria Know No Boundaries: Mechanisms and Functions of Intercellular Mitochondrial Transfer. Front. Cell Dev. Biol. 2016, 4, 107. [Google Scholar] [CrossRef]
- Roehlecke, C.; Schmidt, M.H. Tunneling Nanotubes and Tumor Microtubes in Cancer. Cancers 2020, 12, 857. [Google Scholar] [CrossRef]
- English, K.; Shepherd, A.J.; Nzor, N.-E.; Trinh, R.; Kavelaars, A.; Heijnen, C.J. Astrocytes rescue neuronal health after cisplatin treatment through mitochondrial transfer. Acta Neuropathol. Commun. 2020, 8, 36. [Google Scholar] [CrossRef]
- Weil, S.; Osswald, M.; Solecki, G.; Grosch, J.; Jung, E.; Lemke, D.; Ratliff, M.; Hänggi, D.; Wick, W.; Winkler, F. Tumor microtubes convey resistance to surgical lesions and chemotherapy in gliomas. Neuro-Oncology 2017, 19, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
- Pasquier, J.; Guerrouahen, B.S.; Al Thawadi, H.; Ghiabi, P.; Maleki, M.; Abu-Kaoud, N.; Jacob, A.; Mirshahi, M.; Galas, L.; Rafii, S.; et al. Preferential transfer of mitochondria from endothelial to cancer cells through tunneling nanotubes modulates chemoresistance. J. Transl. Med. 2013, 11, 94. [Google Scholar] [CrossRef]
- Shabo, I.; Svanvik, J.; Lindström, A.; Lechertier, T.; Trabulo, S.; Hulit, J.; Sparey, T.; Pawelek, J. Roles of cell fusion, hybridization and polyploid cell formation in cancer metastasis. World J. Clin. Oncol. 2020, 11, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Kaur, E.; Rajendra, J.; Jadhav, S.; Shridhar, E.; Goda, J.S.; Moiyadi, A.; Dutt, S. Radiation-induced homotypic cell fusions of innately resistant glioblastoma cells mediate their sustained survival and recurrence. Carcinogenesis 2015, 36, 685–695. [Google Scholar] [CrossRef]
- Mercapide, J.; Rappa, G.; Lorico, A. The intrinsic fusogenicity of glioma cells as a factor of transformation and progression in the tumor microenvironment. Int. J. Cancer 2011, 131, 334–343. [Google Scholar] [CrossRef]
- Lu, X.; Kang, Y. Cell fusion as a hidden force in tumor progression. Cancer Res. 2009, 69, 8536–8539. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Zhao, D.; Dai, X.; Chen, J.; Rong, X.; Wang, H.; Wang, A.; Li, M.; Dong, J.; Huang, Q.; et al. Fusion of cancer stem cells and mesenchymal stem cells contributes to glioma neovascularization. Oncol. Rep. 2015, 34, 2022–2030. [Google Scholar] [CrossRef] [PubMed]
- El Hallani, S.; Colin, C.; El Houfi, Y.; Idbaih, A.; Boisselier, B.; Marie, Y.; Ravassard, P.; Labussiere, M.; Mokhtari, K.; Thomas, J.-L.; et al. Tumor and Endothelial Cell Hybrids Participate in Glioblastoma Vasculature. BioMed Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Y. Tunneling nanotubes between rat primary astrocytes and C6 glioma cells alter proliferation potential of glioma cells. Neurosci. Bull. 2015, 31, 371–378. [Google Scholar] [CrossRef]
- Bukoreshtliev, N.V.; Wang, X.; Hodneland, E.; Gurke, S.; Barroso, J.F.; Gerdes, H.-H. Selective block of tunneling nanotube (TNT) formation inhibits intercellular organelle transfer between PC12 cells. FEBS Lett. 2009, 583, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- Osswald, M.; Jung, E.; Sahm, F.; Solecki, G.; Venkataramani, V.; Blaes, J.; Weil, S.; Horstmann, H.; Wiestler, B.; Syed, M.; et al. Brain tumour cells interconnect to a functional and resistant network. Nat. Cell Biol. 2015, 528, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Osswald, M.; Solecki, G.; Wick, W.; Winkler, F. A malignant cellular network in gliomas: Potential clinical implications. Neuro-Oncology 2016, 18, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Errede, M.; Mangieri, D.; Longo, G.; Girolamo, F.; De Trizio, I.; Vimercati, A.; Serio, G.; Frei, K.; Perris, R.; Virgintino, D. Tunneling nanotubes evoke pericyte/endothelial communication during normal and tumoral angiogenesis. Fluids Barriers CNS 2018, 15, 28. [Google Scholar] [CrossRef] [PubMed]
- Liang, D. A Salutary Role of Reactive Oxygen Species in Intercellular Tunnel-Mediated Communication. Front. Cell Dev. Biol. 2018, 6, 2. [Google Scholar] [CrossRef]
- Hase, K.; Kimura, S.; Takatsu, H.; Ohmae, M.; Kawano, S.; Kitamura, H.; Ito, M.; Watarai, H.; Hazelett, C.C.; Yeaman, C.; et al. M-Sec promotes membrane nanotube formation by interacting with Ral and the exocyst complex. Nat. Cell Biol. 2009, 11, 1427–1432. [Google Scholar] [CrossRef]
- Ohno, H.; Hase, K.; Kimura, S. M-Sec. Commun. Integr. Biol. 2010, 3, 231–233. [Google Scholar] [CrossRef]
- Pergu, R.; Dagar, S.; Kumar, H.; Kumar, R.; Bhattacharya, J.; Mylavarapu, S.V.S. The chaperone ERp29 is required for tunneling nanotube formation by stabilizing MSec. J. Biol. Chem. 2019, 294, 7177–7193. [Google Scholar] [CrossRef]
- Sun, X.; Wang, Y.-P.; Zhang, J.; Tu, J.; Wang, X.-J.; Su, X.-D.; Wang, L.; Zhang, Y. Tunneling-nanotube direction determination in neurons and astrocytes. Cell Death Dis. 2012, 3, e438. [Google Scholar] [CrossRef]
- Gerdes, H.-H.; Rustom, A.; Wang, X. Tunneling nanotubes, an emerging intercellular communication route in development. Mech. Dev. 2013, 130, 381–387. [Google Scholar] [CrossRef]
- Ribeiro-Rodrigues, T.M.; Martins-Marques, T.; Morel, S.; Kwak, B.R.; Girão, H. Role of connexin 43 in different forms of intercellular communication–gap junctions, extracellular vesicles and tunnelling nanotubes. J. Cell Sci. 2017, 130, 3619–3630. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.; Osswald, M.; Blaes, J.; Wiestler, B.; Sahm, F.; Schmenger, T.; Solecki, G.; Deumelandt, K.; Kurz, F.T.; Xie, R.; et al. Tweety-Homolog 1 Drives Brain Colonization of Gliomas. J. Neurosci. 2017, 37, 6837–6850. [Google Scholar] [CrossRef] [PubMed]
- Wesseling, P.; Bent, M.V.D.; Perry, A. Oligodendroglioma: Pathology, molecular mechanisms and markers. Acta Neuropathol. 2015, 129, 809–827. [Google Scholar] [CrossRef]
- Desir, S.; Dickson, E.L.; Vogel, R.I.; Thayanithy, V.; Wong, P.; Teoh, D.; Geller, M.A.; Steer, C.J.; Subramanian, S.; Lou, E. Tunneling nanotube formation is stimulated by hypoxia in ovarian cancer cells. Oncotarget 2016, 7, 43150–43161. [Google Scholar] [CrossRef] [PubMed]
- Kemerdere, R.; Akgun, M.Y.; Toklu, S.; Alizada, O.; Tanriverdi, T. Preoperative systemic inflammatory markers in low- and high-grade gliomas: A retrospective analysis of 171 patients. Heliyon 2019, 5, e01681. [Google Scholar] [CrossRef] [PubMed]
- Galvão, R.P.; Zong, H. Inflammation and Gliomagenesis: Bi-Directional Communication at Early and Late Stages of Tumor Progression. Curr. Pathobiol. Rep. 2013, 1, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Katrib, A.; Jeong, H.-H.; Fransen, N.L.; Henzel, K.S.; Miller, J.A. An Inflammatory Landscape for Preoperative Neurologic Deficits in Glioblastoma. Front. Genet. 2019, 10, 488. [Google Scholar] [CrossRef]
- Nabors, L.B.; Gillespie, G.Y.; Harkins, L.; King, P.H. HuR, a RNA stability factor, is expressed in malignant brain tumors and binds to adenine- and uridine-rich elements within the 3′ untranslated regions of cytokine and angiogenic factor mRNAs. Cancer Res. 2001, 61, 2154–2161. [Google Scholar] [PubMed]
- Filippova, N.; Yang, X.; Wang, Y.; Gillespie, G.Y.; Langford, C.; King, P.; Wheeler, C.; Nabors, L.B. The RNA-binding protein HuR promotes glioma growth and treatment resistance. Mol. Cancer Res. 2011, 9, 648–659. Available online: https://mcr.aacrjournals.org/content/9/5/648 (accessed on 6 July 2020). [CrossRef]
- Bolognani, F.; Gallani, A.-I.; Sokol, L.; Baskin, D.S.; Meisner-Kober, N. mRNA stability alterations mediated by HuR are necessary to sustain the fast growth of glioma cells. J. Neurooncol. 2011, 106, 531–542. [Google Scholar] [CrossRef]
- Galbán, S.; Kuwano, Y.; Pullmann, R.; Martindale, J.L.; Kim, H.H.; Lal, A.; Abdelmohsen, K.; Yang, X.; Dang, Y.; Liu, J.O.; et al. RNA-Binding Proteins HuR and PTB Promote the Translation of Hypoxia-Inducible Factor 1α. Mol. Cell. Biol. 2007, 28, 93–107. [Google Scholar] [CrossRef]
- Wang, J.; Leavenworth, J.W.; Hjelmeland, A.B.; Smith, R.; Patel, N.; Borg, B.; Si, Y.; King, P. Deletion of the RNA regulator HuR in tumor-associated microglia and macrophages stimulates anti-tumor immunity and attenuates glioma growth. Glia 2019, 67, 2424–2439. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, K.; Gorospe, M. Posttranscriptional regulation of cancer traits by HuR. Wiley Interdiscip. Rev. RNA 2010, 1, 214–229. [Google Scholar] [CrossRef] [PubMed]
- Filippova, N.; Yang, X.; Ananthan, S.; Sorochinsky, A.; Hackney, J.R.; Gentry, Z.; Bae, S.; King, P.; Nabors, L.B. Hu antigen R (HuR) multimerization contributes to glioma disease progression. J. Biol. Chem. 2017, 292, 16999–17010. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Lal, S.; Vaziri-Gohar, A.; O’Hayer, K.; Gunda, V.; Singh, P.K.; Brody, J.R.; Winter, J.M. RNA-Binding Protein HuR Regulates Both Mutant and Wild-Type IDH1 in IDH1-Mutated Cancer. Mol. Cancer Res. 2018, 17, 508–520. [Google Scholar] [CrossRef]
- Uren, P.J.; Burns, S.C.; Ruan, J.; Singh, K.K.; Smith, A.D.; Penalva, L.O.F. Genomic Analyses of the RNA-binding Protein Hu Antigen R (HuR) Identify a Complex Network of Target Genes and Novel Characteristics of Its Binding Sites. J. Biol. Chem. 2011, 286, 37063–37066. [Google Scholar] [CrossRef]
- Astanina, K.; Koch, M.; Jüngst, C.; Zumbusch, A.; Kiemer, A.K. Lipid droplets as a novel cargo of tunnelling nanotubes in endothelial cells. Sci. Rep. 2015, 5, 11453. [Google Scholar] [CrossRef]
- Nakamura, H.; Murayama, T. The Role of Sphingolipids in Arachidonic Acid Metabolism. J. Pharmacol. Sci. 2014, 124, 307–312. [Google Scholar] [CrossRef]
- Lin, W.-N.; Lin, C.-C.; Cheng, H.-Y.; Yang, C.-M. Regulation of cyclooxygenase-2 and cytosolic phospholipase A2gene expression by lipopolysaccharide through the RNA-binding protein HuR: Involvement of NADPH oxidase, reactive oxygen species and mitogen-activated protein kinases. Br. J. Pharmacol. 2011, 163, 1691–1706. [Google Scholar] [CrossRef]
- Liao, W.-L.; Wang, W.-C.; Chang, W.-C.; Tseng, J.T. The RNA-binding Protein HuR Stabilizes Cytosolic Phospholipase A2α mRNA under Interleukin-1β Treatment in Non-small Cell Lung Cancer A549 Cells. J. Biol. Chem. 2011, 286, 35499–35508. [Google Scholar] [CrossRef] [PubMed]
- Linkous, A.G.; Yazlovitskaya, E.M.; Hallahan, D.E. Cytosolic Phospholipase A2 and Lysophospholipids in Tumor Angiogenesis. J. Natl. Cancer Inst. 2010, 102, 1398–1412. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, H. Expression of Cytosolic Phospholipase A2 Alpha in Glioblastoma Is Associated with Resistance to Chemotherapy. Am. J. Med. Sci. 2018, 356, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Dixon, D.A.; Tolley, N.D.; King, P.H.; Nabors, L.B.; McIntyre, T.M.; Zimmerman, G.A.; Prescott, S.M. Altered expression of the mRNA stability factor HuR promotes cyclooxygenase-2 expression in colon cancer cells. J. Clin. Investig. 2001, 108, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, P.; Lombardi, F.; Augello, F.R.; Giusti, I.; Dolo, V.; Leocata, P.; Cifone, M.G.; Cinque, B. Biological effects of selective COX-2 inhibitor NS398 on human glioblastoma cell lines. Cancer Cell Int. 2020, 20, 1–17. [Google Scholar] [CrossRef]
- Sengupta, S.; Jang, B.-C.; Wu, M.-T.; Paik, J.-H.; Furneaux, H.; Hla, T. The RNA-binding Protein HuR Regulates the Expression of Cyclooxygenase-2. J. Biol. Chem. 2003, 278, 25227–25233. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, X.; Sun, D.; Jia, X.; Xu, L.; Qiao, Y.; Jin, Y. COX-2 expression in ovarian cancer: An updated meta-analysis. Oncotarget 2017, 8, 88152–88162. [Google Scholar] [CrossRef]
- Wang, J.; Hjelmeland, A.B.; Nabors, L.B.; King, P. Anti-cancer effects of the HuR inhibitor, MS-444, in malignant glioma cells. Cancer Biol. Ther. 2019, 20, 979–988. [Google Scholar] [CrossRef]
- Chernomordik, L.V.; Kozlov, M.M. Mechanics of membrane fusion. Nat. Struct. Mol. Biol. 2008, 15, 675–683. [Google Scholar] [CrossRef]
- Jin, R.; Junutula, J.R.; Matern, H.T.; Ervin, K.E.; Scheller, R.H.; Brunger, A.T. Exo84 and Sec5 are competitive regulatory Sec6/8 effectors to the RalA GTPase. EMBO J. 2005, 24, 2064–2074. [Google Scholar] [CrossRef]
- Dormoy-Raclet, V.; Ménard, I.; Clair, E.; Kurban, G.; Mazroui, R.; Di Marco, S.; Von Roretz, C.; Pause, A.; Gallouzi, I.E. The RNA-Binding Protein HuR Promotes Cell Migration and Cell Invasion by Stabilizing the β-actin mRNA in a U-Rich-Element-Dependent Manner. Mol. Cell. Biol. 2007, 27, 5365–5380. [Google Scholar] [CrossRef]
- Zybura-Broda, K.; Wolder-Gontarek, M.; Ambrozek-Latecka, M.; Choros, A.; Bogusz, A.; Wilemska-Dziaduszycka, J.; Rylski, M. HuR (Elavl1) and HuB (Elavl2) Stabilize Matrix Metalloproteinase-9 mRNA During Seizure-Induced Mmp-9 Expression in Neurons. Front. Neurosci. 2018, 12, 224. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, K.; Srikantan, S.; Yang, X.; Lal, A.; Kim, H.H.; Kuwano, Y.; Galban, S.; Becker, K.G.; Kamara, D.; De Cabo, R.; et al. Ubiquitin-mediated proteolysis of HuR by heat shock. EMBO J. 2009, 28, 1271–1282. [Google Scholar] [CrossRef] [PubMed]
- D’Aloia, A.; Berruti, G.; Costa, B.; Schiller, C.; Ambrosini, R.; Pastori, V.; Martegani, E.; Ceriani, M. RalGPS2 is involved in tunneling nanotubes formation in 5637 bladder cancer cells. Exp. Cell Res. 2018, 362, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Fifadara, N.H.; Beer, F.; Ono, S.; Ono, S.J. Interaction between activated chemokine receptor 1 and FcεRI at membrane rafts promotes communication and F-actin-rich cytoneme extensions between mast cells. Int. Immunol. 2010, 22, 113–128. [Google Scholar] [CrossRef]
- Palanisamy, K.; Tsai, T.-H.; Yu, T.-M.; Sun, K.-T.; Yu, S.-H.; Lin, F.-Y.; Wang, I.-K.; Li, C.-Y. RNA-binding protein, human antigen R regulates hypoxia-induced autophagy by targeting ATG7/ATG16L1 expressions and autophagosome formation. J. Cell. Physiol. 2018, 234, 7448–7458. [Google Scholar] [CrossRef]
- Ji, E.; Kim, C.; Kang, H.; Ahn, S.; Jung, M.; Hong, Y.; Tak, H.; Lee, S.; Kim, W.; Lee, E.K. RNA Binding Protein HuR Promotes Autophagosome Formation by Regulating Expression of Autophagy-Related Proteins 5, 12, and 16 in Human Hepatocellular Carcinoma Cells. Mol. Cell. Biol. 2019, 39, e00508-18. [Google Scholar] [CrossRef]
- Sun, C.; Dai, X.; Zhao, D.; Wang, H.; Rong, X.; Huang, Q.; Lan, Q. Mesenchymal stem cells promote glioma neovascularization in vivo by fusing with cancer stem cells. BMC Cancer 2019, 19, 1240. [Google Scholar] [CrossRef]
- Goldenberg, D.M.; Zagzag, D.; Heselmeyer-Haddad, K.M.; Garcia, L.Y.B.; Ried, T.; Loo, M.; Chang, C.-H.; Gold, D.V. Horizontal transmission and retention of malignancy, as well as functional human genes, after spontaneous fusion of human glioblastoma and hamster host cells in vivo. Int. J. Cancer 2011, 131, 49–58. [Google Scholar] [CrossRef]
- Cao, M.-F.; Chen, L.; Dang, W.-Q.; Zhang, X.-C.; Zhang, X.; Shi, Y.; Yao, X.-H.; Li, Q.; Zhu, J.; Lin, Y.; et al. Hybrids by tumor-associated macrophages × glioblastoma cells entail nuclear reprogramming and glioblastoma invasion. Cancer Lett. 2019, 442, 445–452. [Google Scholar] [CrossRef]
- Manjunath, Y.; Porciani, D.; Mitchem, J.; Suvilesh, K.N.; Avella, D.M.; Kimchi, E.T.; Staveley-O’Carroll, K.F.; Burke, D.H.; Li, G.; Kaifi, J.T. Tumor-Cell–Macrophage Fusion Cells as Liquid Biomarkers and Tumor Enhancers in Cancer. Int. J. Mol. Sci. 2020, 21, 1872. [Google Scholar] [CrossRef]
- Bracq, L.; Xie, M.; Lambelé, M.; Vu, L.-T.; Matz, J.; Schmitt, A.; Delon, J.; Zhou, P.; Randriamampita, C.; Bouchet, J.; et al. T Cell-Macrophage Fusion Triggers Multinucleated Giant Cell Formation for HIV-1 Spreading. J. Virol. 2017, 91, e01237-17. [Google Scholar] [CrossRef]
- Chen, K.A.; Laywell, E.D.; Marshall, G.; Walton, N.; Zheng, T.; Steindler, D.A. Fusion of neural stem cells in culture. Exp. Neurol. 2006, 198, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, E.; Presta, I.; Maisano, D.; Donato, A.; Pirrone, C.K.; Cardillo, G.; Corrado, S.D.; Mignogna, C.; Mancuso, T.; Donato, G.; et al. Role of Macrophages in Brain Tumor Growth and Progression. Int. J. Mol. Sci. 2018, 19, 1005. [Google Scholar] [CrossRef] [PubMed]
- Civita, P.; Leite, D.M.; Pilkington, G.J. Pre-Clinical Drug Testing in 2D and 3D Human In Vitro Models of Glioblastoma Incorporating Non-Neoplastic Astrocytes: Tunneling Nano Tubules and Mitochondrial Transfer Modulates Cell Behavior and Therapeutic Respons. Int. J. Mol. Sci. 2019, 20, 6017. [Google Scholar] [CrossRef] [PubMed]
- Zaccard, C.R.; Rinaldo, C.R.; Mailliard, R. Linked in: Immunologic membrane nanotube networks. J. Leukoc. Biol. 2016, 100, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Chen, P.; Gupta, P.; Ott, M.; Zamler, D.; Kassab, C.; Bhat, K.P.; Curran, M.A.; De Groot, J.F.; Heimberger, A.B. Immune biology of glioma associated macrophages and microglia: Functional and therapeutic implications. Neuro-Oncology 2019. [Google Scholar] [CrossRef]
- Hanna, S.J.; McCoy-Simandle, K.; Leung, E.; Genna, A.; Condeelis, J.; Cox, D. Tunneling nanotubes, a novel mode of tumor cell–macrophage communication in tumor cell invasion. J. Cell Sci. 2019, 132, jcs223321. [Google Scholar] [CrossRef]
- Morrison, A.R.; Yarovinsky, T.O.; Young, B.D.; Moraes, F.; Ross, T.D.; Ceneri, N.; Zhang, J.; Zhuang, Z.W.; Sinusas, A.J.; Pardi, R.; et al. Chemokine-coupled β2 integrin–induced macrophage Rac2–Myosin IIA interaction regulates VEGF-A mRNA stability and arteriogenesis. J. Exp. Med. 2014, 211, 1957–1968. [Google Scholar] [CrossRef]
- Zhang, J.; Modi, Y.; Yarovinsky, T.; Yu, J.; Collinge, M.; Kyriakides, T.; Zhu, Y.; Sessa, W.; Pardi, R.; Bender, J.R. Macrophage β2 Integrin–Mediated, HuR-Dependent Stabilization of Angiogenic Factor–Encoding mRNAs in Inflammatory Angiogenesis. Am. J. Pathol. 2012, 180, 1751–1760. [Google Scholar] [CrossRef]
- Filippova, N.; Yang, X.; Nabors, L.B. Growth Factor Dependent Regulation of Centrosome Function and Genomic Instability by HuR. Biomolecules 2015, 5, 263–281. [Google Scholar] [CrossRef]
- Dybdal-Hargreaves, N.; Risinger, A.L.; Mooberry, S.L. Eribulin mesylate: Mechanism of action of a unique microtubule-targeting agent. Clin. Cancer Res. 2015, 21, 2445–2452. [Google Scholar] [CrossRef]
- Mundhara, N.; Majumder, A.; Panda, D. Methyl-β-cyclodextrin, an actin depolymerizer augments the antiproliferative potential of microtubule-targeting agents. Sci. Rep. 2019, 9, 7638. [Google Scholar] [CrossRef] [PubMed]
- Zalba, S.; Hagen, T.L.T. Cell membrane modulation as adjuvant in cancer therapy. Cancer Treat. Rev. 2017, 52, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Takabatake, Y.; Takahashi, A.; Isaka, Y. Chloroquine in Cancer Therapy: A Double-Edged Sword of Autophagy. Cancer Res. 2013, 73, 3–7. [Google Scholar] [CrossRef]
- Jones, T.M.; Carew, J.S.; Nawrocki, S.T. Therapeutic Targeting of Autophagy for Renal Cell Carcinoma Therapy. Cancers 2020, 12, 1185. [Google Scholar] [CrossRef] [PubMed]
- Kast, R.E.; Skuli, N.; Karpel-Massler, G.; Frosina, G.; Ryken, T.; Halatsch, M.-E. Blocking epithelial-to-mesenchymal transition in glioblastoma with a sextet of repurposed drugs: The EIS regimen. Oncotarget 2017, 8, 60727–60749. [Google Scholar] [CrossRef]
- Hanna, S.J.; McCoy-Simandle, K.; Miskolci, V.; Guo, P.; Cammer, M.; Hodgson, L.; Cox, D. The Role of Rho-GTPases and actin polymerization during Macrophage Tunneling Nanotube Biogenesis. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Kim, B.-W.; Lee, J.-S.; Ko, Y.-G. Mycoplasma exploits mammalian tunneling nanotubes for cell-to-cell dissemination. BMB Rep. 2019, 52, 490–495. [Google Scholar] [CrossRef]
- Delage, E.; Cervantes, D.C.; Pénard, E.; Schmitt, C.; Syan, S.; Disanza, A.; Scita, G.; Zurzolo, C. Differential identity of Filopodia and Tunneling Nanotubes revealed by the opposite functions of actin regulatory complexes. Sci. Rep. 2016, 6, 39632. [Google Scholar] [CrossRef]
- Meisner, N.-C.; Hintersteiner, M.; Mueller, K.; Bauer, R.; Seifert, J.-M.; Naegeli, H.-U.; Ottl, J.; Oberer, L.; Guenat, C.; Moss, S.; et al. Identification and mechanistic characterization of low-molecular-weight inhibitors for HuR. Nat. Chem. Biol. 2007, 3, 508–515. [Google Scholar] [CrossRef]
- Muralidharan, R.; Mehta, M.; Ahmed, R.; Roy, S.; Xu, L.; Aubé, J.; Chen, A.; Zhao, Y.D.; Herman, T.; Ramesh, R.; et al. HuR-targeted small molecule inhibitor exhibits cytotoxicity towards human lung cancer cells. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chae, M.-J.; Sung, H.Y.; Kim, E.-H.; Lee, M.; Kwak, H.; Chae, C.H.; Kim, S.; Park, W.-Y. Chemical inhibitors destabilize HuR binding to the AU-rich element of TNF-α mRNA. Exp. Mol. Med. 2009, 41, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Wu, X.; Fields, J.K.; Johnson, D.K.; Lan, L.; Pratt, M.; Somoza, A.D.; Wang, C.C.C.; Karanicolas, J.; Oakley, B.R.; et al. The fungal natural product azaphilone-9 binds to HuR and inhibits HuR-RNA interaction in vitro. PLoS ONE 2017, 12, e0175471. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, V.G.; Lal, P.; Mantelli, B.; Tiedje, C.; Zucal, C.; Thongon, N.; Gaestel, M.; Latorre, E.; Marinelli, L.; Seneci, P.; et al. Dihydrotanshinone-I interferes with the RNA-binding activity of HuR affecting its post-transcriptional function. Sci. Rep. 2015, 5, 16478. [Google Scholar] [CrossRef] [PubMed]
- Della Volpe, S.; Nasti, R.; Queirolo, M.; Unver, M.Y.; Jumde, V.K.; Dömling, A.; Vasile, F.; Potenza, D.; Ambrosio, F.A.; Costa, G.; et al. Novel Compounds Targeting the RNA-Binding Protein HuR. Structure-Based Design, Synthesis, and Interaction Studies. ACS Med. Chem. Lett. 2019, 10, 615–620. [Google Scholar] [CrossRef]
- Kakuguchi, W.; Nomura, T.; Kitamura, T.; Otsuguro, S.; Matsushita, K.; Sakaitani, M.; Maenaka, K.; Tei, K. Suramin, screened from an approved drug library, inhibits HuR functions and attenuates malignant phenotype of oral cancer cells. Cancer Med. 2018, 7, 6269–6280. [Google Scholar] [CrossRef]
- Wang, Z.; Bhattacharya, A.; Ivanov, D.N. Identification of Small-Molecule Inhibitors of the HuR/RNA Interaction Using a Fluorescence Polarization Screening Assay Followed by NMR Validation. PLoS ONE 2015, 10, e0138780. [Google Scholar] [CrossRef]
- Hostetter, C.; Licata, L.A.; Costantino, C.L.; Witkiewicz, A.; Yeo, C.J.; Brody, J.R.; Keen, J.C. Cytoplasmic accumulation of the RNA binding protein HuR is central to tamoxifen resistance in estrogen receptor positive breast cancer cells. Cancer Biol. Ther. 2008, 7, 1496–1506. [Google Scholar] [CrossRef]
- Guo, J.; Lv, J.; Chang, S.; Chen, Z.; Lu, W.; Xu, C.; Liu, M.; Pang, X. Inhibiting cytoplasmic accumulation of HuR synergizes genotoxic agents in urothelial carcinoma of the bladder. Oncotarget 2016, 7, 45249–45262. [Google Scholar] [CrossRef]
- Marriott, J.J.; Miyasaki, J.M.; Gronseth, G.; O’Connor, P.W. Evidence Report: The efficacy and safety of mitoxantrone (Novantrone) in the treatment of multiple sclerosis. Neurology 2010, 74, 1463–1470. [Google Scholar] [CrossRef]
- Wu, X.; Lan, L.; Wilson, D.M.; Marquez, R.T.; Tsao, W.-C.; Gao, P.; Roy, A.; Turner, B.A.; McDonald, P.; Tunge, J.A.; et al. Identification and Validation of Novel Small Molecule Disruptors of HuR-mRNA Interaction. ACS Chem. Biol. 2015, 10, 1476–1484. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, J.; Khera, L.; Gaur, N.; Paul, C.; Kaul, R. Role of Modulator of Inflammation Cyclooxygenase-2 in Gammaherpesvirus Mediated Tumorigenesis. Front. Microbiol. 2017, 8, 538. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Qu, L.; Yan, S. Cyclooxygenase-2 promotes tumor growth and suppresses tumor immunity. Cancer Cell Int. 2015, 15, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Singgih, M.F.; Huldani; Achmad, H.; Sukmana, B.I.; Carmelita, A.B.; Putra, A.P.; Ramadhany, S.; Putri, A.P. A Review of Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Medications in Dentistry: Uses and Side Effects. Syst. Rev. Pharm. 2020, 11, 293–298. [Google Scholar] [CrossRef]
- Schroer, J.; Shenk, T. Inhibition of cyclooxygenase activity blocks cell-to-cell spread of human cytomegalovirus. Proc. Natl. Acad. Sci. USA 2008, 105, 19468–19473. [Google Scholar] [CrossRef]
- Deb, J.; Lakshman, T.R.; Ghosh, I.; Jana, S.S.; Paine, T.K. Mechanistic studies of in vitro anti-proliferative and anti-inflammatory activities of the Zn(ii)–NSAID complexes of 1,10-phenanthroline-5,6-dione in MDA-MB-231 cells. Dalton Trans. 2020, 49, 11375–11384. [Google Scholar] [CrossRef]
- Katsanou, V.; Milatos, S.; Yiakouvaki, A.; Sgantzis, N.; Kotsoni, A.; Alexiou, M.; Harokopos, V.; Aidinis, V.; Hemberger, M.; Kontoyiannis, D.L. The RNA-Binding Protein Elavl1/HuR Is Essential for Placental Branching Morphogenesis and Embryonic Development. Mol. Cell. Biol. 2009, 29, 2762–2776. [Google Scholar] [CrossRef]
- Figueroa, A.; Cuadrado, A.; Fan, J.; Atasoy, U.; Muscat, G.E.; Muñoz-Canoves, P.; Gorospe, M.; Emuñoz, A. Role of HuR in Skeletal Myogenesis through Coordinate Regulation of Muscle Differentiation Genes. Mol. Cell. Biol. 2003, 23, 4991–5004. [Google Scholar] [CrossRef]
- Dormoy-Raclet, V.; Cammas, A.; Celona, B.; Lian, X.J.; Van Der Giessen, K.; Zivojnovic, M.; Brunelli, S.; Riuzzi, F.; Sorci, G.; Wilhelm, B.T.; et al. HuR and miR-1192 regulate myogenesis by modulating the translation of HMGB1 mRNA. Nat. Commun. 2013, 4, 2388. [Google Scholar] [CrossRef]
- Lenard, A.; Daetwyler, S.; Betz, C.; Ellertsdóttir, E.; Belting, H.-G.; Huisken, J.; Affolter, M. Endothelial Cell Self-fusion during Vascular Pruning. PLoS Biol. 2015, 13, e1002126. [Google Scholar] [CrossRef]
- Naito, H.; Iba, T.; Takakura, N. Mechanisms of new blood-vessel formation and proliferative heterogeneity of endothelial cells. Int. Immunol. 2020, 32, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Kelaini, S.; Caines, R.; Margariti, A. RBPs Play Important Roles in Vascular Endothelial Dysfunction Under Diabetic Conditions. Front. Physiol. 2018, 9, 1310. [Google Scholar] [CrossRef]
- Bibli, S.-I.; Hu, J.; Sigala, F.; Wittig, I.; Heidler, J.; Zukunft, S.; Tsilimigras, D.I.; Randriamboavonjy, V.; Wittig, J.; Kojonazarov, B.; et al. Cystathionine γ Lyase Sulfhydrates the RNA Binding Protein Human Antigen R to Preserve Endothelial Cell Function and Delay Atherogenesis. Circulation 2019, 139, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Chi, M.N.; Auriol, J.; Jégou, B.; Kontoyiannis, D.L.; Turner, J.M.; Rooij, D.G.D.; Morello, D. The RNA-binding protein ELAVL1/HuR is essential for mouse spermatogenesis, acting both at meiotic and postmeiotic stages. Mol. Biol. Cell 2011, 22, 2875–2885. [Google Scholar] [CrossRef]
- Tsai, I.-T.; Kuo, C.-C.; Liou, J.-P.; Chang, J.-Y. Novel microtubule inhibitor MPT0B098 inhibits hypoxia-induced epithelial-to-mesenchymal transition in head and neck squamous cell carcinoma. J. Biomed. Sci. 2018, 25, 1–12. [Google Scholar] [CrossRef]
- Sun, J.; Gu, X.; Wu, N.; Zhang, P.; Liu, Y.; Jiang, S. Human antigen R enhances the epithelial-mesenchymal transition via regulation of ZEB-1 in the human airway epithelium. Respir. Res. 2018, 19, 109. [Google Scholar] [CrossRef]
- Ale-Agha, N.; Galban, S.; Sobieroy, C.; Abdelmohsen, K.; Gorospe, M.; Sies, H.; Klotz, L.-O. HuR regulates gap junctional intercellular communication by controlling β-catenin levels and adherens junction integrity. Hepatology 2009, 50, 1567–1576. [Google Scholar] [CrossRef]
- Salat, C.; Muñoz, M.J.; Sesé, M.; Cajal, S.R.Y.; Aasen, T. Post-transcriptional regulation of connexins. Biochem. Soc. Trans. 2015, 43, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Rimkute, L.; Jotautis, V.; Marandykina, A.; Sveikatienė, R.; Antanavičiūtė, I.; Skeberdis, V.A. The role of neural connexins in HeLa cell mobility and intercellular communication through tunneling tubes. BMC Cell Biol. 2016, 17, 1. [Google Scholar] [CrossRef]
- Zhou, Z.; Licklider, L.J.; Gygi, S.P.; Reed, R. Comprehensive proteomic analysis of the human spliceosome. Nat. Cell Biol. 2002, 419, 182–185. [Google Scholar] [CrossRef] [PubMed]

| Endogenous Retroviral Fusogens | |||||
| PDGx1 | PDGx2 | PDGx3 | PDGx4 | PDGx5 | |
| ERVK13-1 | +++ | +++ | +++ | +++ | +++ |
| ERV3-1 | ++ | +++ | +++ | +++ | ++ |
| ERVMER34-1 | + | + | + | + | + |
| ERVW-1 | + | + | + | + | none |
| EBI3 | none | none | + | none | ++ |
| ERVV-1 | none | none | + | none | none |
| ERVV-2 | + | none | none | + | none |
| Fusogen Receptors | |||||
| PDGx1 | PDGx2 | PDGx3 | PDGx4 | PDGx5 | |
| CTNNB1 | +++ | +++ | +++ | +++ | +++ |
| ICAM1 | none | + | + | + | +++ |
| ITGA4 | none | ++ | + | ++ | ++ |
| ITGB4 | + | + | + | ++ | ++ |
| VCAM1 | none | none | ++ | none | + |
| Germ Cell Fusogens | |||||
| PDGx1 | PDGx2 | PDGx3 | PDGx4 | PDGx5 | |
| CD9 | +++ | +++ | +++ | +++ | ++ |
| IZUMO1 | + | + | + | + | none |
| IZUMOR1 | + | + | + | + | none |
| IZUMO3 | none | + | none | none | none |
| IZUMO4 | ++ | ++ | ++ | ++ | + |
| SPA17 | ++ | ++ | ++ | ++ | + |
| SPAG8 | + | + | + | + | + |
| Myoblast Fusion Factors | |||||
| PDGx1 | PDGx2 | PDGx3 | PDGx4 | PDGx5 | |
| MYMK | none | none | none | none | + |
| MYMX | + | none | + | none | + |
| TNT Type i and Type ii Transcripts | |||||
|---|---|---|---|---|---|
| PDGx1 | PDGx2 | PDGx3 | PDGx4 | PDGx5 | |
| TNFAIP2 | +++ | ++ | ++ | +++ | +++ |
| RALA | +++ | +++ | +++ | +++ | ++ |
| EXOC6 | ++ | ++ | +++ | +++ | ++ |
| S100A4 | +++ | + | +++ | ++ | +++ |
| GJA1 | +++ | +++ | +++ | +++ | none |
| GAP43 | +++ | +++ | +++ | +++ | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filippova, N.; Nabors, L.B. ELAVL1 Role in Cell Fusion and Tunneling Membrane Nanotube Formations with Implication to Treat Glioma Heterogeneity. Cancers 2020, 12, 3069. https://doi.org/10.3390/cancers12103069
Filippova N, Nabors LB. ELAVL1 Role in Cell Fusion and Tunneling Membrane Nanotube Formations with Implication to Treat Glioma Heterogeneity. Cancers. 2020; 12(10):3069. https://doi.org/10.3390/cancers12103069
Chicago/Turabian StyleFilippova, Natalia, and Louis B. Nabors. 2020. "ELAVL1 Role in Cell Fusion and Tunneling Membrane Nanotube Formations with Implication to Treat Glioma Heterogeneity" Cancers 12, no. 10: 3069. https://doi.org/10.3390/cancers12103069
APA StyleFilippova, N., & Nabors, L. B. (2020). ELAVL1 Role in Cell Fusion and Tunneling Membrane Nanotube Formations with Implication to Treat Glioma Heterogeneity. Cancers, 12(10), 3069. https://doi.org/10.3390/cancers12103069






