Predictors of Response and Survival in Immune Checkpoint Inhibitor-Treated Unresectable Hepatocellular Carcinoma
Abstract
1. Introduction
2. Results
2.1. Demographic Characteristics of the Study Cohort
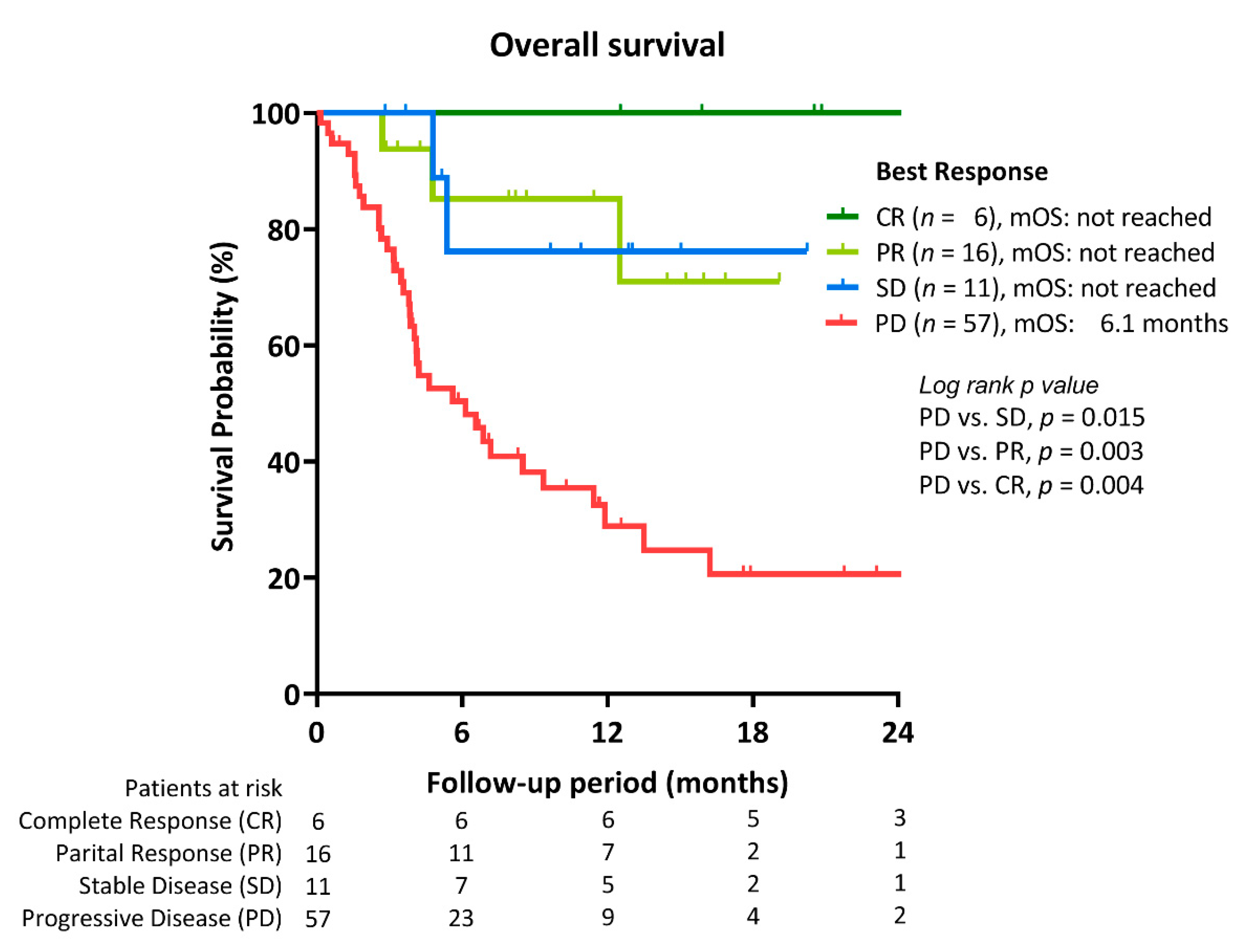
2.2. Treatment Response to ICI Therapy
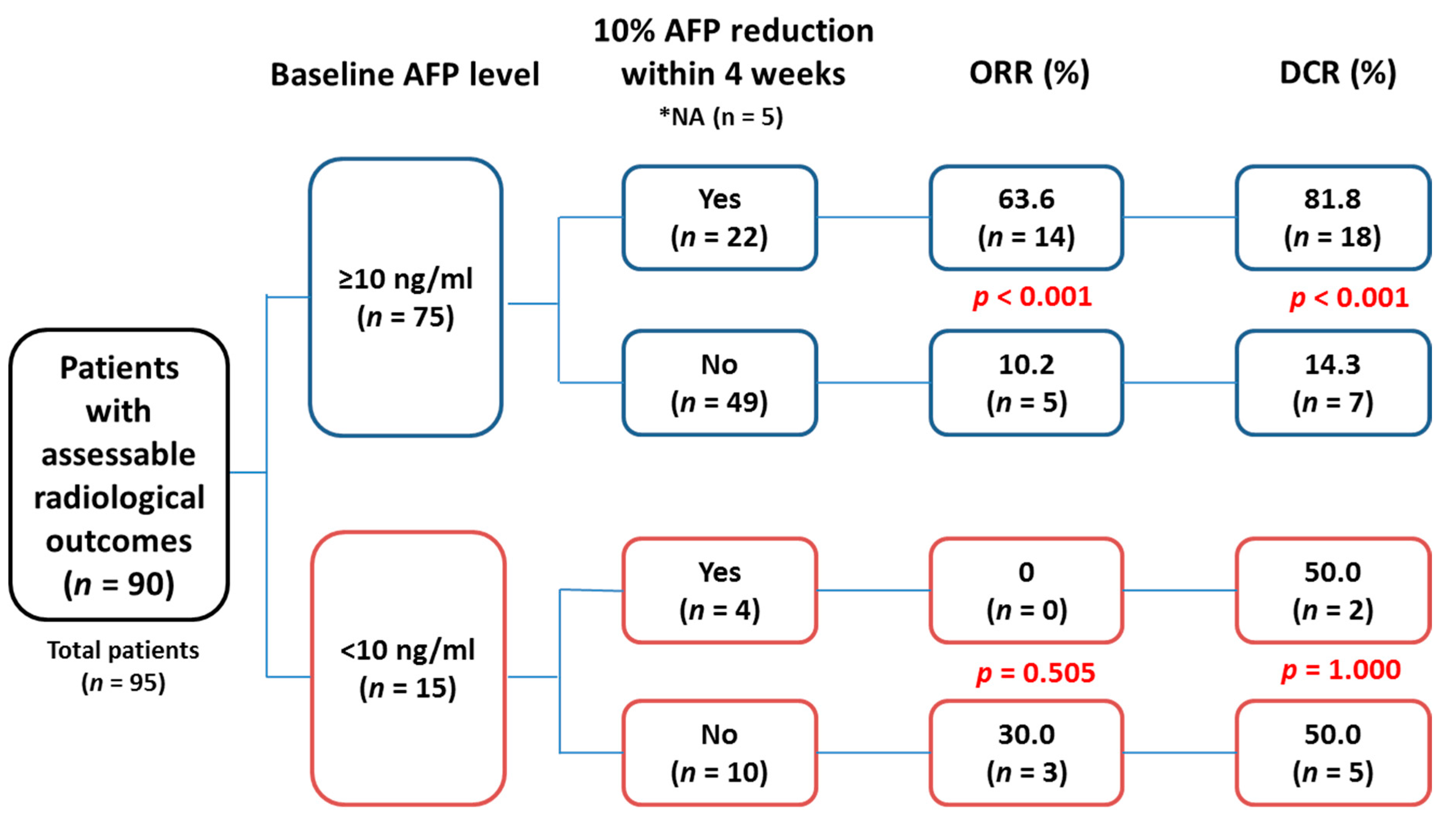
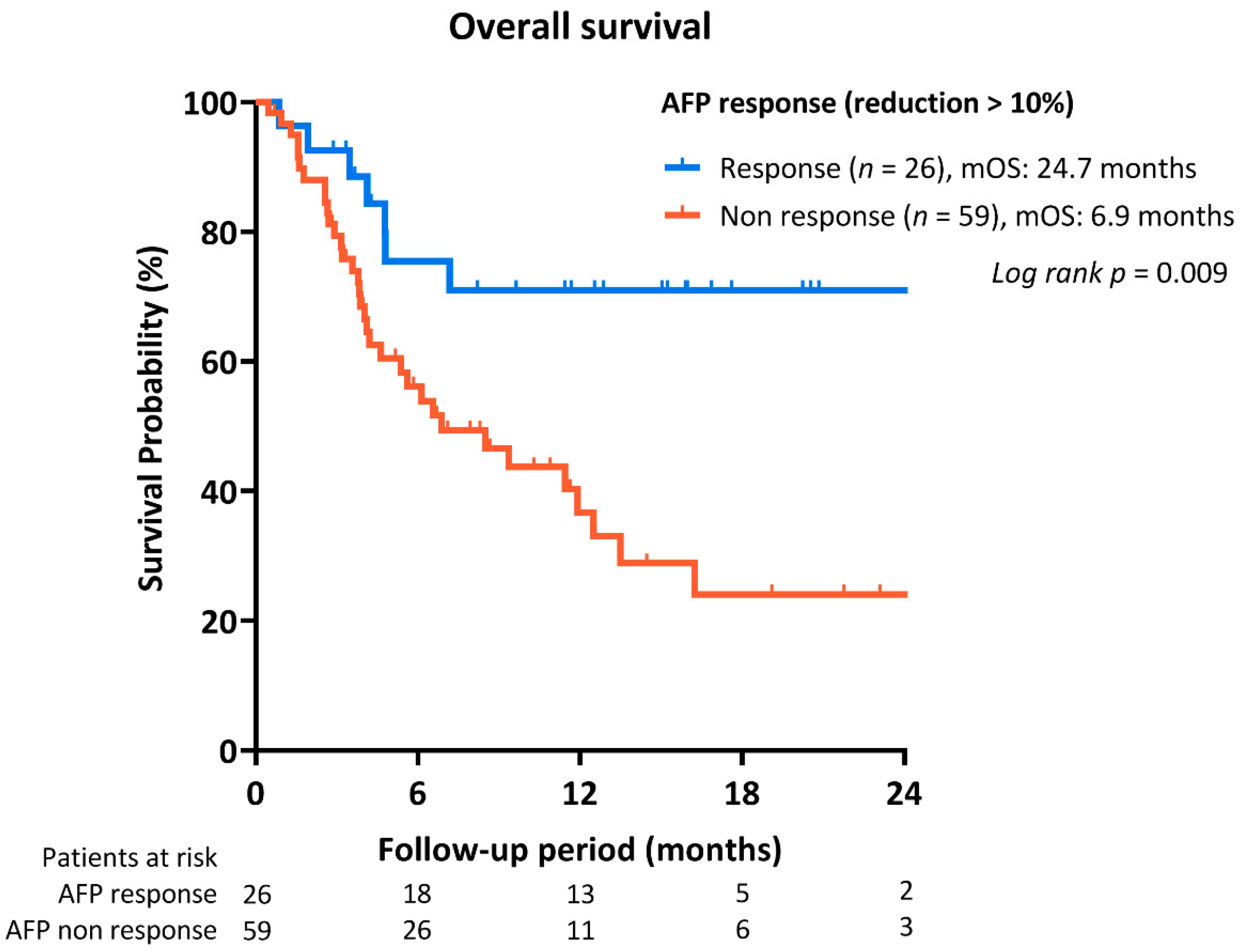
2.3. Association between Tumor Response and Early AFP Response
2.4. Response in HCC Patients with Available PD-L1 Level and Evaluable Images
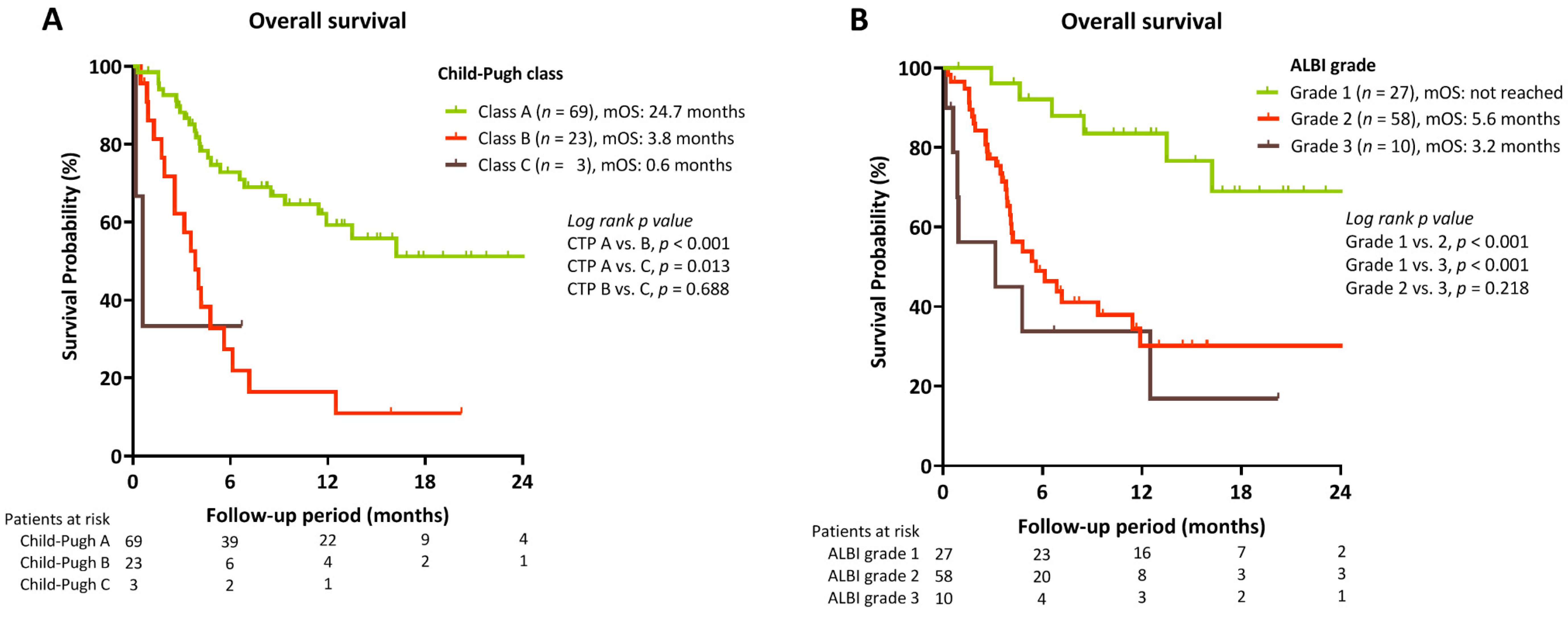
2.5. Uni- and Multivariate Analysis for Factors Associated with OS for All HCC Patients
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Treatment and Outcome Assessment
4.3. PD-L1 Expression Analysis
4.4. Biochemical Tests
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.L.; Schirmacher, P.; Vilgrain, V. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.Y.; Choo, S.P.; Trojan, J.; Welling, T.H.R.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Zhu, A.X.; Finn, R.S.; Edeline, J.; Cattan, S.; Ogasawara, S.; Palmer, D.; Verslype, C.; Zagonel, V.; Fartoux, L.; Vogel, A.; et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial. Lancet Oncol. 2018, 19, 940–952. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Results of KEYNOTE-240: Phase 3 study of pembrolizumab (Pembro) vs best supportive care (BSC) for second line therapy in advanced hepatocellular carcinoma (HCC). J. Clin. Oncol. 2019, 37, 4004. [Google Scholar] [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef] [PubMed]
- Finkelmeier, F.; Waidmann, O.; Trojan, J. Nivolumab for the treatment of hepatocellular carcinoma. Expert Rev. Anticancer Ther. 2018, 18, 1169–1175. [Google Scholar] [CrossRef] [PubMed]
- Garon, E.B.; Rizvi, N.A.; Hui, R.; Leighl, N.; Balmanoukian, A.S.; Eder, J.P.; Patnaik, A.; Aggarwal, C.; Gubens, M.; Horn, L.; et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N. Engl. J. Med. 2015, 372, 2018–2028. [Google Scholar] [CrossRef]
- Fuchs, C.S.; Doi, T.; Jang, R.W.; Muro, K.; Satoh, T.; Machado, M.; Sun, W.; Jalal, S.I.; Shah, M.A.; Metges, J.P.; et al. Safety and Efficacy of Pembrolizumab Monotherapy in Patients With Previously Treated Advanced Gastric and Gastroesophageal Junction Cancer: Phase 2 Clinical KEYNOTE-059 Trial. JAMA Oncol. 2018, 4, e180013. [Google Scholar] [CrossRef] [PubMed]
- Khunger, M.; Hernandez, A.V.; Pasupuleti, V.; Rakshit, S.; Pennell, N.A.; Stevenson, J.; Mukhopadhyay, S.; Schalper, K.; Velcheti, V. Programmed Cell Death 1 ( PD-1 ) Ligand ( PD-L 1 ) Expression in Solid Tumors As a Predictive Biomarker of Benefit From PD-1/PD-L 1 Axis Inhibitors: A Systematic Review and Meta-Analysis. JCO Precis. Oncol. 2017, 1. [Google Scholar] [CrossRef]
- Finkelmeier, F.; Czauderna, C.; Perkhofer, L.; Ettrich, T.J.; Trojan, J.; Weinmann, A.; Marquardt, J.U.; Vermehren, J.; Waidmann, O. Feasibility and safety of nivolumab in advanced hepatocellular carcinoma: Real-life experience from three German centers. J. Cancer Res. Clin. Oncol. 2019, 145, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Memon, K.; Kulik, L.; Lewandowski, R.J.; Wang, E.; Ryu, R.K.; Riaz, A.; Nikolaidis, P.; Miller, F.H.; Yaghmai, V.; Baker, T.; et al. Alpha-fetoprotein response correlates with EASL response and survival in solitary hepatocellular carcinoma treated with transarterial therapies: A subgroup analysis. J. Hepatol. 2012, 56, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Yoon, S.M.; Han, S.; Shim, J.H.; Kim, K.M.; Lim, Y.S.; Lee, H.C.; Kim, S.Y.; Park, J.H.; Kim, J.H. Alpha-fetoprotein normalization as a prognostic surrogate in small hepatocellular carcinoma after stereotactic body radiotherapy: A propensity score matching analysis. BMC Cancer 2015, 15, 987. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.; Dodge, J.L.; Roberts, J.P.; Hirose, R.; Yao, F.Y. Alpha-Fetoprotein Decrease from > 1, 000 to <500 ng/mL in Patients with Hepatocellular Carcinoma Leads to Improved Posttransplant Outcomes. Hepatology 2019, 69, 1193–1205. [Google Scholar]
- Shao, Y.Y.; Lin, Z.Z.; Hsu, C.; Shen, Y.C.; Hsu, C.H.; Cheng, A.L. Early alpha-fetoprotein response predicts treatment efficacy of antiangiogenic systemic therapy in patients with advanced hepatocellular carcinoma. Cancer 2010, 116, 4590–4596. [Google Scholar] [CrossRef]
- Meyer, T.; Melero, I.; Yau, T.; Hsu, C.; Kudo, M.; Choo, S.P.; Trojan, J.; Welling, T.; Kang, Y.-K.; Yeo, W.; et al. Hepatic safety and biomarker assessments in sorafenib-experienced patients with advanced hepatocellular carcinoma treated with nivolumab in the CheckMate-040 study. J. Hepatol. 2018, 68 (Suppl. 1), S16. [Google Scholar] [CrossRef]
- Scheiner, B.; Kirstein, M.M.; Hucke, F.; Finkelmeier, F.; Schulze, K.; von Felden, J.; Koch, S.; Schwabl, P.; Hinrichs, J.B.; Waneck, F.; et al. Programmed cell death protein-1 (PD-1)-targeted immunotherapy in advanced hepatocellular carcinoma: Efficacy and safety data from an international multicentre real-world cohort. Aliment. Pharmacol. Ther. 2019, 49, 1323–1333. [Google Scholar] [CrossRef]
- Shao, Y.Y.; Liu, T.H.; Hsu, C.; Lu, L.C.; Shen, Y.C.; Lin, Z.Z.; Cheng, A.L.; Hsu, C.H. Early alpha-foetoprotein response associated with treatment efficacy of immune checkpoint inhibitors for advanced hepatocellular carcinoma. Liver Int. 2019. [Google Scholar] [CrossRef]
- Lee, P.C.; Chen, Y.T.; Chao, Y.; Huo, T.I.; Li, C.P.; Su, C.W.; Lee, M.H.; Hou, M.C.; Lee, F.Y.; Lin, H.C.; et al. Validation of the albumin-bilirubin grade-based integrated model as a predictor for sorafenib-failed hepatocellular carcinoma. Liver Int. 2018, 38, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Yau, T.; Hsu, C.; Kim, T.Y.; Choo, S.P.; Kang, Y.K.; Hou, M.M.; Numata, K.; Yeo, W.; Chopra, A.; Ikeda, M.; et al. Nivolumab in advanced hepatocellular carcinoma: Sorafenib-experienced Asian cohort analysis. J. Hepatol. 2019, 71, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Reig, M.; Rimola, J.; Torres, F.; Darnell, A.; Rodriguez-Lope, C.; Forner, A.; Llarch, N.; Rios, J.; Ayuso, C.; Bruix, J. Postprogression survival of patients with advanced hepatocellular carcinoma: Rationale for second-line trial design. Hepatology 2013, 58, 2023–2031. [Google Scholar] [CrossRef] [PubMed]
- Iavarone, M.; Cabibbo, G.; Biolato, M.; Della Corte, C.; Maida, M.; Barbara, M.; Basso, M.; Vavassori, S.; Craxi, A.; Grieco, A.; et al. Predictors of survival in patients with advanced hepatocellular carcinoma who permanently discontinued sorafenib. Hepatology 2015, 62, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Pinato, D.J.; Yen, C.; Bettinger, D.; Ramaswami, R.; Arizumi, T.; Ward, C.; Pirisi, M.; Burlone, M.E.; Thimme, R.; Kudo, M.; et al. The albumin-bilirubin grade improves hepatic reserve estimation post-sorafenib failure: Implications for drug development. Aliment. Pharmacol. Ther. 2017, 45, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Matilla, A.; Santoro, A.; Melero, I.; Gracian, A.C.; Acosta-Rivera, M.; Choo, S.P.; El-Khoueiry, A.B.; Kuromatsu, R.; El-Rayes, B.F.; et al. Checkmate-040: Nivolumab (NIVO) in patients (pts) with advanced hepatocellular carcinoma (aHCC) and Child-Pugh B (CPB) status. J. Clin. Oncol. 2019, 37, 327. [Google Scholar] [CrossRef]
- Kambhampati, S.; Bauer, K.E.; Bracci, P.M.; Keenan, B.P.; Behr, S.C.; Gordan, J.D.; Kelley, R.K. Nivolumab in patients with advanced hepatocellular carcinoma and Child-Pugh class B cirrhosis: Safety and clinical outcomes in a retrospective case series. Cancer 2019, 125, 3234–3241. [Google Scholar] [CrossRef]
- Cabibbo, G.; Petta, S.; Barbara, M.; Attardo, S.; Bucci, L.; Farinati, F.; Giannini, E.G.; Negrini, G.; Ciccarese, F.; Rapaccini, G.L.; et al. Hepatic decompensation is the major driver of death in HCV-infected cirrhotic patients with successfully treated early hepatocellular carcinoma. J. Hepatol. 2017, 67, 65–71. [Google Scholar] [CrossRef]
- Kudo, M. Targeted and immune therapies for hepatocellular carcinoma: Predictions for 2019 and beyond. World J. Gastroenterol. 2019, 25, 789–807. [Google Scholar] [CrossRef]
- Ikeda, M.; Sung, M.W.; Kudo, M.; Kobayashi, M.; Baron, A.D.; Finn, R.S.; Kaneko, S.; Zhu, A.X.; Kubota, T.; Kraljevic, S.; et al. A phase 1b trial of lenvatinib (LEN) plus pembrolizumab (PEM) in patients (pts) with unresectable hepatocellular carcinoma (uHCC). J. Clin. Oncol. 2018, 36, 4076. [Google Scholar] [CrossRef]
- Stein, S.; Pishvaian, M.J.; Lee, M.S.; Lee, K.-H.; Hernandez, S.; Kwan, A.; Liu, B.; Grossman, W.; Iizuka, K.; Ryoo, B.-Y. Safety and clinical activity of 1L atezolizumab + bevacizumab in a phase Ib study in hepatocellular carcinoma (HCC). J. Clin. Oncol. 2018, 36, 4074. [Google Scholar] [CrossRef]
- Yau, T.; Kang, Y.-K.; Kim, T.-Y.; El-Khoueiry, A.B.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.-M.; Matilla, A.; et al. Nivolumab (NIVO) + ipilimumab (IPI) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): Results from CheckMate 040. J. Clin. Oncol. 2019, 37, 4012. [Google Scholar] [CrossRef]
- Calderaro, J.; Rousseau, B.; Amaddeo, G.; Mercey, M.; Charpy, C.; Costentin, C.; Luciani, A.; Zafrani, E.S.; Laurent, A.; Azoulay, D.; et al. Programmed death ligand 1 expression in hepatocellular carcinoma: Relationship With clinical and pathological features. Hepatology 2016, 64, 2038–2046. [Google Scholar] [CrossRef] [PubMed]
- Heimbach, J.K.; Kulik, L.M.; Finn, R.S.; Sirlin, C.B.; Abecassis, M.M.; Roberts, L.R.; Zhu, A.X.; Murad, M.H.; Marrero, J.A. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018, 67, 358–380. [Google Scholar] [CrossRef]
- Johnson, P.J.; Berhane, S.; Kagebayashi, C.; Satomura, S.; Teng, M.; Reeves, H.L.; O’Beirne, J.; Fox, R.; Skowronska, A.; Palmer, D.; et al. Assessment of liver function in patients with hepatocellular carcinoma: A new evidence-based approach-the ALBI grade. J. Clin. Oncol. 2015, 33, 550–558. [Google Scholar] [CrossRef]
- Liu, P.H.; Hsu, C.Y.; Hsia, C.Y.; Lee, Y.H.; Chiou, Y.Y.; Huang, Y.H.; Lee, F.Y.; Lin, H.C.; Hou, M.C.; Huo, T.I. ALBI and PALBI grade predict survival for HCC across treatment modalities and BCLC stages in the MELD Era. J. Gastroenterol. Hepatol. 2017, 32, 879–886. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
| Characteristics | n = 95 |
|---|---|
| Age, y | 65.5 (57.2–72.9) |
| Sex (male), n (%) | 73 (76.8) |
| HBsAg-positive, n (%) | 62 (65.3) |
| Anti-HCV-positive, n (%) | 21 (22.1) |
| Max. tumor size, cm | 5.2 (2.3–8.8) |
| Tumor >50% liver volume, n (%) | 30 (31.6) |
| Multiple tumors, n (%) | 89 (93.7) |
| Extrahepatic metastasis, n (%) | 48 (50.5) |
| Portal vein invasion, n (%) | 51 (53.7) |
| AFP, ng/mL | 609.7 (37.5–4832.3) |
| <10 ng/mL, n (%) | 15 (15.8) |
| 10–400 ng/mL, n (%) | 27 (28.4) |
| ≥400 ng/mL, n (%) | 53 (55.8) |
| BCLC stage B/C, n (%) | 20/75 (21.1/78.9) |
| Prothrombin time, INR | 1.10 (1.05–1.23) |
| Platelet count, K/cumm | 145 (102–218) |
| ALT, U/L | 39 (25–61) |
| AST, U/L | 57 (35–97) |
| Total bilirubin, mg/dL | 1.03 (0.55–1.52) |
| Albumin, g/dL | 3.6 (3.2–4.0) |
| Neutrophil-lymphocyte ratio | 4.16 (2.89–6.85) |
| Presence of ascites, n (%) | 37 (38.9) |
| Child–Pugh score | 6 (5–7) |
| Child–Pugh class A/B/C, n (%) | 69/23/3 (72.6/24.2/3.2) |
| ALBI grade 1/2/3, n (%) | 27/58/10 (28.4/61.1/10.5) |
| First line systemic therapy, n (%) | 39 (41.1) |
| Prior therapy to ICI, n (%) | |
| Surgical resection | 35 (36.8) |
| RFA/PEIT/MWA | 31/9/1 (32.6/9.5/1.1) |
| TACE/RT/TARE (Y-90) | 55/23/5 (57.9/24.2/5.3) |
| Sorafenib | 56 (58.9) |
| Nivolumab/Pembrolizumab, n (%) | 92/3 (96.8/3.2) |
| Combined ICI with TKI, n (%) | 13 (13.7) |
| Immune-related AEs | |
| Skin reactions/Pneumonitis/Hepatitis | 6/4/3 (6.3/4.2/3.2) |
| Post PD treatment, n (%) | |
| TACE/RT/TARE (Y-90) | 9/8/2 (9.5/8.4/2.1) |
| Regorafenib/Lenvatinib/Carbozantinib | 8/16/2 (8.4/16.8/2.1) |
| Ramucirumab | 4 (4.2) |
| Sorafenib/Traditional CT | 7/6 (7.4/6.3) |
| Death | 47 (49.5) |
| Evaluable Response | All Patients (n = 95) | Child–Pugh A (n = 69) | Child–Pugh B (n = 23) | Child–Pugh C (n = 3) | Combination Treatment (n = 13) | Monotherapy (n = 82) |
|---|---|---|---|---|---|---|
| Best Response, n (%) | ||||||
| Complete response | 6 (6.7) | 5 (7.5) | 1 (5.0) | 0 | 1 (7.7) | 5 (6.5) |
| Partial response | 16 (17.8) | 13 (19.4) | 3 (15.0) | 0 | 5 (38.5) | 11 (14.3) |
| Stable disease | 11 (12.2) | 10 (14.9) | 1 (5.0) | 0 | 1 (7.7) | 10 (13.0) |
| Progressive disease | 57 (63.3) | 39 (58.2) | 15 (75.0) | 3 (100.0) | 6 (46.2) | 51 (66.2) |
| Non-assessable | 5 | 2 | 3 | 0 | 0 | 5 |
| Objective response rate | 22 (24.4) | 18 (26.9) | 4 (20.0) | 0 | 6 (46.2) | 16 (20.8) |
| Disease control rate | 33 (36.7) | 28 (41.8) | 5 (25.0) | 0 | 7 (53.8) | 26 (33.8) |
| For Responders | ||||||
| Time to response (days) | 63 (48–75) | 64 (52–76) | 52 (21–72) | – | 57 (43–73) | 63 (55–77) |
| Duration of response (months) | Not yet reached (16 ongoing) | Not yet reached (13 ongoing) | Not yet reached (three ongoing) | – | Not yet reached (five ongoing) | Not yet reached (11 ongoing) |
| Characteristics | Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|---|
| OR | 95% CI | p Value | OR | 95% CI | p Value | ||
| Age, y | >60 vs. ≤60 | 0.447 | 0.167–1.192 | 0.108 | |||
| Sex | Male vs. Female | 0.691 | 0.228–2.092 | 0.514 | |||
| HBsAg-positive | Yes vs. No | 1.651 | 0.573–4.756 | 0.353 | |||
| Anti-HCV-positive | Yes vs. No | 0.722 | 0.213–2.446 | 0.601 | |||
| Tumor size, cm | >7 vs. ≤7 | 0.754 | 0.271–2.094 | 0.588 | |||
| Tumor number | Multiple vs. single | 0.625 | 0.106–3.670 | 0.625 | |||
| Tumor shape | Infiltrative vs. nodular | 2.250 | 0.813–6.227 | 0.118 | |||
| Tumor/Liver volume | >50% vs. ≤50% | 0.900 | 0.308–2.633 | 0.847 | |||
| Portal vein invasion | Yes vs. No | 1.131 | 0.431–2.969 | 0.802 | |||
| Main portal vein invasion | Yes vs. No PVI | 1.046 | 0.278–3.932 | 0.947 | |||
| Portal branches invasion | Yes vs. No PVI | 1.295 | 0.441–3.803 | 0.638 | |||
| Extrahepatic metastasis | Yes vs. No | 0.580 | 0.219–1.537 | 0.273 | |||
| BCLC stage | Stage C vs. B | 1.385 | 0.409–4.689 | 0.601 | |||
| AFP, ng/mL | >400 vs. ≤400 | 0.789 | 0.301–2.068 | 0.630 | |||
| AFP, ng/mL | <10 vs. ≤10 | 0.737 | 0.188–2.894 | 0.662 | |||
| NLR | >2.5 vs. ≤2.5 | 1.529 | 0.390–5.992 | 0.542 | |||
| Prothrombin time, INR | >1.2 vs. ≤1.2 | 1.211 | 0.422–3.470 | 0.722 | |||
| Platelet count | >100K vs. ≤100K | 0.821 | 0.275–2.447 | 0.723 | |||
| ALT, U/L | > 40 vs. ≤40 | 0.294 | 0.097–0.888 | 0.030 | 0.384 | 0.109–1.349 | 0.135 |
| AST, U/L | > 40 vs. ≤40 | 0.465 | 0.172–1.255 | 0.131 | |||
| Ascites | Yes vs. No | 0.536 | 0.186–1.539 | 0.246 | |||
| Child–Pugh class | Class B, C vs. A | 0.537 | 0.172–1.914 | 0.366 | |||
| ALBI grade | Grade 2,3 vs. 1 | 0.520 | 0.190–1.422 | 0.203 | |||
| Prior Sorafenib treatment | Yes vs. No | 1.011 | 0.380–2.687 | 0.982 | |||
| Combined treatment * | Yes vs. No | 3.813 | 1.083–13.419 | 0.037 | 2.522 | 0.572–11.111 | 0.222 |
| AFP reduction at fourth week † | Yes vs. No | 7.437 | 2.545–21.735 | <0.001 | 7.259 | 2.359–22.337 | 0.001 |
| IO related AEs | Yes vs. No | 0.916 | 0.228–3.678 | 0.901 | |||
| Characteristics | Univariate | Multivariate (Model 1) # | Multivariate (Model 2) # | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | HR | 95% CI | p | ||
| Age, y | >60 vs. ≤60 | 1.252 | 0.676–2.318 | 0.476 | NA | NA | ||||
| Sex | Male vs. Female | 0.632 | 0.337–1.186 | 0.153 | NA | NA | ||||
| HBsAg-positive | Yes vs. No | 1.020 | 0.555–1.874 | 0.950 | NA | NA | ||||
| Anti-HCV-positive | Yes vs. No | 1.393 | 0.729–2.661 | 0.315 | NA | NA | ||||
| Tumor size, cm | >7 vs. ≤7 | 2.450 | 1.362–4.409 | 0.003 | NS | NS | ||||
| Tumor number | Multiple vs. single | 3.709 | 0.510–26.946 | 0.195 | NA | NA | ||||
| Tumor/Liver volume | >50% vs. ≤50% | 2.425 | 1.323–4.444 | 0.004 | NS | NS | ||||
| Portal vein invasion | Yes vs. No | 1.829 | 1.008–3.321 | 0.047 | NS | NS | ||||
| Extrahepatic metastasis | Yes vs. No | 1.444 | 0.804–2.591 | 0.219 | NA | NA | ||||
| BCLC stage | Stage C vs. B | 1.854 | 0.828–4.154 | 0.134 | NA | NA | ||||
| AFP, ng/mL | >400 vs. ≤400 | 2.039 | 1.102–3.773 | 0.023 | NS | NS | ||||
| AFP, ng/mL | <10 vs. ≤10 | 0.255 | 0.079–0.826 | 0.023 | NS | NS | ||||
| NLR | >2.5 vs. ≤2.5 | 1.010 | 0.467–2.185 | 0.981 | NA | NA | ||||
| Prothrombin time, INR | >1.2 vs. ≤1.2 | 1.585 | 0.842–2.983 | 0.154 | NS | NS | ||||
| Platelet count | >100K vs. ≤100K | 0.928 | 0.479–1.799 | 0.825 | NA | NA | ||||
| ALT, U/L | >40 vs. ≤40 | 2.463 | 1.370–4.428 | 0.003 | NS | NS | ||||
| AST, U/L | >40 vs. ≤40 | 4.762 | 2.015–11.255 | <0.001 | NS | NS | ||||
| Ascites | Yes vs. No | 2.782 | 1.551–4.989 | 0.001 | NA | NS | ||||
| Child–Pugh class | Class A vs. B | 0.260 | 0.143–0.472 | <0.001 | 0.289 | 0.134–0.624 | 0.002 | NA | ||
| ALBI grade | Grade1 vs. 2/3 | 0.189 | 0.079–0.453 | <0.001 | NA | 0.220 | 0.084–0.576 | 0.002 | ||
| Prior Sorafenib treatment | Yes vs. No | 0.952 | 0.528–1.717 | 0.870 | NA | NA | ||||
| Combined treatment * | Yes vs. No | 0.408 | 0.125–1.331 | 0.137 | NS | NS | ||||
| AFP reduction at fourth week † | Yes vs. No | 0.372 | 0.172–0.809 | 0.013 | 0.234 | 0.096–0.569 | 0.001 | 0.243 | 0.104–0.565 | 0.001 |
| Immunotherapy related AEs | Yes vs. No | 0.746 | 0.294–1.893 | 0.537 | NA | NA | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, P.-C.; Chao, Y.; Chen, M.-H.; Lan, K.-H.; Lee, C.-J.; Lee, I.-C.; Chen, S.-C.; Hou, M.-C.; Huang, Y.-H. Predictors of Response and Survival in Immune Checkpoint Inhibitor-Treated Unresectable Hepatocellular Carcinoma. Cancers 2020, 12, 182. https://doi.org/10.3390/cancers12010182
Lee P-C, Chao Y, Chen M-H, Lan K-H, Lee C-J, Lee I-C, Chen S-C, Hou M-C, Huang Y-H. Predictors of Response and Survival in Immune Checkpoint Inhibitor-Treated Unresectable Hepatocellular Carcinoma. Cancers. 2020; 12(1):182. https://doi.org/10.3390/cancers12010182
Chicago/Turabian StyleLee, Pei-Chang, Yee Chao, Ming-Huang Chen, Keng-Hsin Lan, Chieh-Ju Lee, I-Cheng Lee, San-Chi Chen, Ming-Chih Hou, and Yi-Hsiang Huang. 2020. "Predictors of Response and Survival in Immune Checkpoint Inhibitor-Treated Unresectable Hepatocellular Carcinoma" Cancers 12, no. 1: 182. https://doi.org/10.3390/cancers12010182
APA StyleLee, P.-C., Chao, Y., Chen, M.-H., Lan, K.-H., Lee, C.-J., Lee, I.-C., Chen, S.-C., Hou, M.-C., & Huang, Y.-H. (2020). Predictors of Response and Survival in Immune Checkpoint Inhibitor-Treated Unresectable Hepatocellular Carcinoma. Cancers, 12(1), 182. https://doi.org/10.3390/cancers12010182





