CYP2D6 Genotype Predicts Plasma Concentrations of Tamoxifen Metabolites in Ethiopian Breast Cancer Patients
Abstract
1. Introduction
2. Results
2.1. Sociodemographic Characterstics
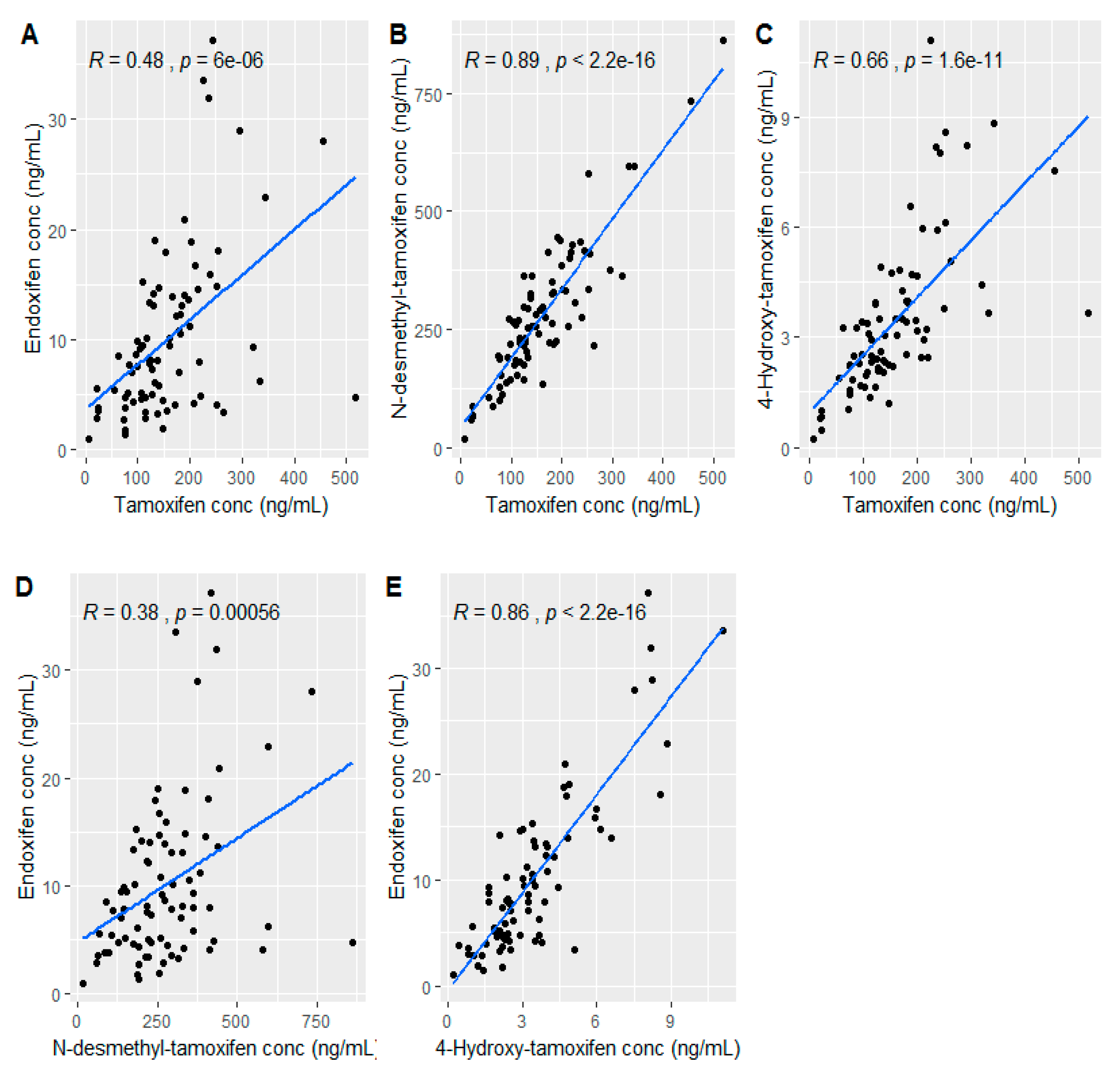
2.2. Plasma Tamoxifen and Its Metabolite Concentration Profile
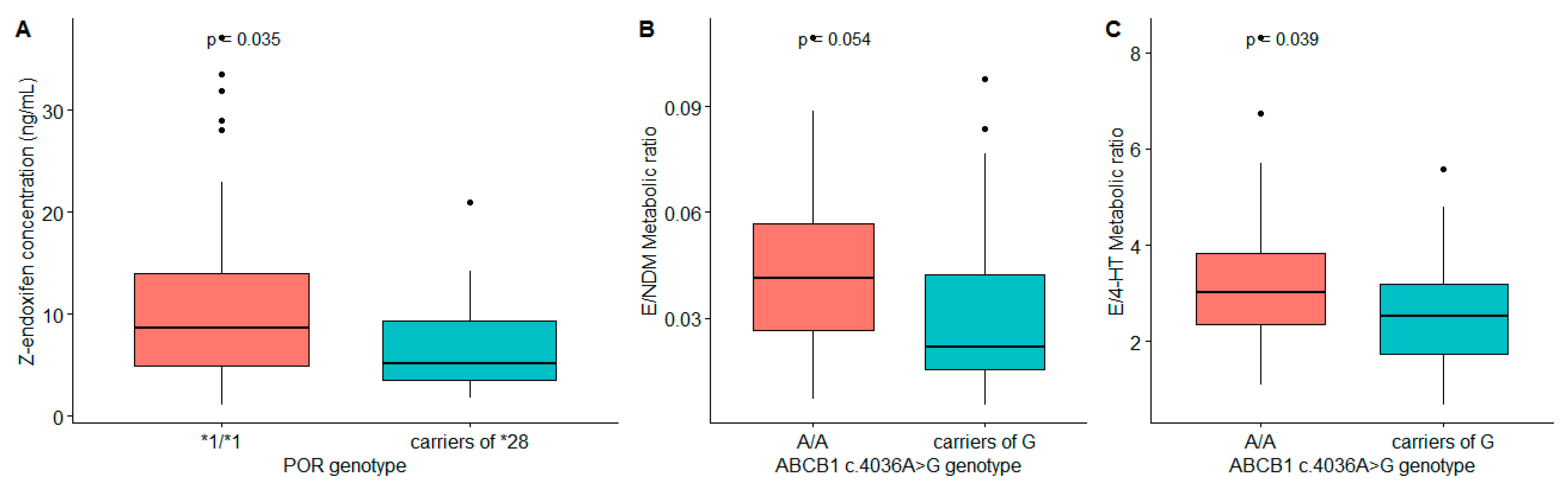
3.3. Association of Pharmacogenetics and Pharmacokinetics of Tamoxifen Its Metabolites
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Genotyping of CYP2D6, CYP2C9, CYP2C19, CYP3A5, POR, ABCB1 and UGT2B15
4.3. Copy Number Variation
4.4. Quantification of Plasma Tamoxifen and Its Metabolites
4.5. Genotype Based Phenotype Assignment
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saladores, P.H.; Precht, J.C.; Schroth, W.; Brauch, H.; Schwab, M. Impact of metabolizing enzymes on drug response of endocrine therapy in breast cancer. Expert Rev. Mol. Diagn. 2013, 13, 349–365. [Google Scholar] [CrossRef] [PubMed]
- EBCTCG. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomised trials. Lancet 2005, 365, 1687–1717. [Google Scholar] [CrossRef]
- Saladores, P.; Mürdter, T.; Eccles, D.; Chowbay, B.; Zgheib, N.K.; Winter, S.; Ganchev, B.; Eccles, B.; Gerty, S.; Tfayli, A.; et al. Tamoxifen metabolism predicts drug concentrations and outcome in premenopausal patients with early breast cancer. Pharm. J. 2015, 15, 84–94. [Google Scholar] [CrossRef] [PubMed]
- De Vries Schultink, A.H.M.; Zwart, W.; Linn, S.C.; Beijnen, J.H.; Huitema, A.D.R. Effects of Pharmacogenetics on the Pharmacokinetics and Pharmacodynamics of Tamoxifen. Clin. Pharm. 2015, 54, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Klein, D.J.; Thorn, C.F.; Desta, Z.; Flockhart, D.A.; Altman, R.B.; Klein, T.E. PharmGKB summary: Tamoxifen pathway, pharmacokinetics. Pharm. Genom. 2013, 23, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Desta, Z.; Stearns, V.; Ward, B.; Ho, H.; Lee, K.-H.; Skaar, T.; Storniolo, A.M.; Li, L.; Araba, A.; et al. CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J. Natl. Cancer Inst. 2005, 97, 30–39. [Google Scholar] [CrossRef]
- Schroth, W.; Winter, S.; Mürdter, T.; Schaeffeler, E.; Eccles, D.; Eccles, B.; Chowbay, B.; Khor, C.C.; Tfayli, A.; Zgheib, N.K.; et al. Improved Prediction of Endoxifen Metabolism by CYP2D6 Genotype in Breast Cancer Patients Treated with Tamoxifen. Front. Pharmacol. 2017, 8, 582. [Google Scholar] [CrossRef] [PubMed]
- Puszkiel, A.; Arellano, C.; Vachoux, C.; Evrard, A.; Le Morvan, V.; Boyer, J.-C.; Robert, J.; Delmas, C.; Dalenc, F.; Debled, M.; et al. Factors Affecting Tamoxifen Metabolism in Patients With Breast Cancer: Preliminary Results of the French PHACS Study. Clin. Pharmacol. Ther. 2019, 106, 585–595. [Google Scholar] [CrossRef]
- Stearns, V.; Johnson, M.D.; Rae, J.M.; Morocho, A.; Novielli, A.; Bhargava, P.; Hayes, D.F.; Desta, Z.; Flockhart, D.A. Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J. Natl. Cancer Inst. 2003, 95, 1758–1764. [Google Scholar] [CrossRef]
- Madlensky, L.; Natarajan, L.; Tchu, S.; Pu, M.; Mortimer, J.; Flatt, S.W.; Nikoloff, D.M.; Hillman, G.; Fontecha, M.R.; Lawrence, H.J.; et al. Tamoxifen metabolite concentrations, CYP2D6 genotype, and breast cancer outcomes. Clin. Pharmacol. Ther. 2011, 89, 718–725. [Google Scholar] [CrossRef]
- Lim, J.S.L.; Chen, X.A.; Singh, O.; Yap, Y.S.; Ng, R.C.H.; Wong, N.S.; Wong, M.; Lee, E.J.D.; Chowbay, B. Impact of CYP2D6, CYP3A5, CYP2C9 and CYP2C19 polymorphisms on tamoxifen pharmacokinetics in Asian breast cancer patients. Br. J. Clin. Pharmacol. 2011, 71, 737–750. [Google Scholar] [CrossRef] [PubMed]
- Mürdter, T.E.; Schroth, W.; Bacchus-Gerybadze, L.; Winter, S.; Heinkele, G.; Simon, W.; Fasching, P.A.; Fehm, T.; German Tamoxifen and AI Clinicians Group; Eichelbaum, M.; et al. Activity levels of tamoxifen metabolites at the estrogen receptor and the impact of genetic polymorphisms of phase I and II enzymes on their concentration levels in plasma. Clin. Pharmacol. Ther. 2011, 89, 708–717. [Google Scholar]
- Livaudais, J.C.; Hershman, D.L.; Habel, L.; Kushi, L.; Gomez, S.L.; Li, C.I.; Neugut, A.I.; Fehrenbacher, L.; Thompson, B.; Coronado, G.D. Racial/ethnic differences in initiation of adjuvant hormonal therapy among women with hormone receptor-positive breast cancer. Breast Cancer Res. Treat 2012, 131, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Farias, A.J.; Du, X.L. Ethnic differences in initiation and timing of adjuvant endocrine therapy among older women with hormone receptor-positive breast cancer enrolled in Medicare Part D. Med. Oncol. 2016, 33, 19. [Google Scholar] [CrossRef] [PubMed]
- Aklillu, E.; Persson, I.; Bertilsson, L.; Johansson, I.; Rodrigues, F.; Ingelman-Sundberg, M. Frequent distribution of ultrarapid metabolizers of debrisoquine in an ethiopian population carrying duplicated and multiduplicated functional CYP2D6 alleles. J. Pharmacol. Exp. Ther. 1996, 278, 441–446. [Google Scholar] [PubMed]
- Iusuf, D.; Teunissen, S.F.; Wagenaar, E.; Rosing, H.; Beijnen, J.H.; Schinkel, A.H. P-glycoprotein (ABCB1) transports the primary active tamoxifen metabolites endoxifen and 4-hydroxytamoxifen and restricts their brain penetration. J. Pharmacol. Exp. Ther. 2011, 337, 710–717. [Google Scholar] [CrossRef]
- Wang, D.; Johnson, A.D.; Papp, A.C.; Kroetz, D.L.; Sadée, W. Multidrug resistance polypeptide 1 (MDR1, ABCB1) variant 3435C>T affects mRNA stability. Pharm. Genom. 2005, 15, 693–704. [Google Scholar] [CrossRef]
- Leschziner, G.D.; Andrew, T.; Pirmohamed, M.; Johnson, M.R. ABCB1 genotype and PGP expression, function and therapeutic drug response: A critical review and recommendations for future research. Pharm. J. 2007, 7, 154–179. [Google Scholar] [CrossRef]
- Tazzite, A.; Kassogue, Y.; Diakité, B.; Jouhadi, H.; Dehbi, H.; Benider, A.; Nadifi, S. Association between ABCB1 C3435T polymorphism and breast cancer risk: A Moroccan case-control study and meta-analysis. BMC Genet. 2016, 17, 126. [Google Scholar] [CrossRef]
- Amjadi, O.; Hedayatizadeh-Omran, A.; Alizadeh-navaei, R. Association between MDR1 (C3435T) gene polymorphism and risk of breast cancer: An Iranian case-control study—WCRJ. WCRJ 2018, 5, e1126. [Google Scholar]
- Mukonzo, J.K.; Okwera, A.; Nakasujja, N.; Luzze, H.; Sebuwufu, D.; Ogwal-Okeng, J.; Waako, P.; Gustafsson, L.L.; Aklillu, E. Influence of efavirenz pharmacokinetics and pharmacogenetics on neuropsychological disorders in Ugandan HIV-positive patients with or without tuberculosis: A prospective cohort study. BMC Infect. Dis. 2013, 13, 261. [Google Scholar] [CrossRef] [PubMed]
- Mukonzo, J.K.; Röshammar, D.; Waako, P.; Andersson, M.; Fukasawa, T.; Milani, L.; Svensson, J.O.; Ogwal-Okeng, J.; Gustafsson, L.L.; Aklillu, E. A novel polymorphism in ABCB1 gene, CYP2B6*6 and sex predict single-dose efavirenz population pharmacokinetics in Ugandans. Br. J. Clin. Pharmacol. 2009, 68, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Swart, M.; Ren, Y.; Smith, P.; Dandara, C. ABCB1 4036A>G and 1236C>T Polymorphisms Affect Plasma Efavirenz Levels in South African HIV/AIDS Patients. Front. Genet. 2012, 3, 236. [Google Scholar] [CrossRef] [PubMed]
- Ngaimisi, E.; Habtewold, A.; Minzi, O.; Makonnen, E.; Mugusi, S.; Amogne, W.; Yimer, G.; Riedel, K.-D.; Janabi, M.; Aderaye, G.; et al. Importance of Ethnicity, CYP2B6 and ABCB1 Genotype for Efavirenz Pharmacokinetics and Treatment Outcomes: A Parallel-Group Prospective Cohort Study in Two Sub-Saharan Africa Populations. PLoS ONE 2013, 8, e67946. [Google Scholar] [CrossRef] [PubMed]
- Elens, L.; Vandercam, B.; Yombi, J.-C.; Lison, D.; Wallemacq, P.; Haufroid, V. Influence of host genetic factors on efavirenz plasma and intracellular pharmacokinetics in HIV-1-infected patients. Pharmacogenomics 2010, 11, 1223–1234. [Google Scholar] [CrossRef]
- Zhou, S.-F. Polymorphism of human cytochrome P450 2D6 and its clinical significance: Part I. Clin. Pharm. 2009, 48, 689–723. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.I.; Lee, S.K.; Kim, J.; Kim, S.W.; Yu, J.; Bae, S.Y.; Lee, J.E.; Nam, S.J.; Lee, S.-Y. Variations in plasma concentrations of tamoxifen metabolites and the effects of genetic polymorphisms on tamoxifen metabolism in Korean patients with breast cancer. Oncotarget 2017, 8, 100296–100311. [Google Scholar] [CrossRef]
- Balkenende, E.M.E.; Dahhan, T.; Linn, S.C.; Jager, N.G.L.; Beijnen, J.H.; Goddijn, M. A prospective case series of women with estrogen receptor-positive breast cancer: Levels of tamoxifen metabolites in controlled ovarian stimulation with high-dose tamoxifen. Hum. Reprod. 2013, 28, 953–959. [Google Scholar] [CrossRef][Green Version]
- Fotoohi, A.K.; Karim, H.; Lafolie, P.; Pohanka, A.; Östervall, J.; Hatschek, T.; Vitols, S. Pronounced Interindividual But Not Intraindividual Variation in Tamoxifen and Metabolite Levels in Plasma During Adjuvant Treatment of Women With Early Breast Cancer. Ther. Drug Monit. 2016, 38, 239–245. [Google Scholar] [CrossRef]
- Wang, D.; Poi, M.J.; Sun, X.; Gaedigk, A.; Leeder, J.S.; Sadee, W. Common CYP2D6 polymorphisms affecting alternative splicing and transcription: Long-range haplotypes with two regulatory variants modulate CYP2D6 activity. Hum. Mol. Genet. 2014, 23, 268–278. [Google Scholar] [CrossRef]
- Zanger, U.M.; Schwab, M. Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther. 2013, 138, 103–141. [Google Scholar] [CrossRef] [PubMed]
- Goetz, M.P.; Sangkuhl, K.; Guchelaar, H.-J.; Schwab, M.; Province, M.; Whirl-Carrillo, M.; Symmans, W.F.; McLeod, H.L.; Ratain, M.J.; Zembutsu, H.; et al. Clinical Pharmacogenetics Implementation Consortium (CPIC) Guideline for CYP2D6 and Tamoxifen Therapy. Clin. Pharmacol. Ther. 2018, 103, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Teft, W.A.; Gong, I.Y.; Dingle, B.; Potvin, K.; Younus, J.; Vandenberg, T.A.; Brackstone, M.; Perera, F.E.; Choi, Y.-H.; Zou, G.; et al. CYP3A4 and seasonal variation in vitamin D status in addition to CYP2D6 contribute to therapeutic endoxifen level during tamoxifen therapy. Breast Cancer Res. Treat. 2013, 139, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Aklillu, E.; Herrlin, K.; Gustafsson, L.L.; Bertilsson, L.; Ingelman-Sundberg, M. Evidence for environmental influence on CYP2D6-catalysed debrisoquine hydroxylation as demonstrated by phenotyping and genotyping of Ethiopians living in Ethiopia or in Sweden. Pharmacogenetics 2002, 12, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Gaedigk, A.; Dinh, J.C.; Jeong, H.; Prasad, B.; Leeder, J.S. Ten Years’ Experience with the CYP2D6 Activity Score: A Perspective on Future Investigations to Improve Clinical Predictions for Precision Therapeutics. J. Pers. Med. 2018, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Koppel, N.; Rekdal, V.M.; Balskus, E.P. Chemical transformation of xenobiotics by the human gut microbiota. Science 2017, 356. [Google Scholar] [CrossRef] [PubMed]
- De Vries, E.M.; Lammers, L.A.; Achterbergh, R.; Klümpen, H.-J.; Mathot, R.A.A.; Boelen, A.; Romijn, J.A. Fasting-Induced Changes in Hepatic P450 Mediated Drug Metabolism Are Largely Independent of the Constitutive Androstane Receptor CAR. PLoS ONE 2016, 11, e0159552. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.N.; Zhong, X.-B. P450 oxidoreductase: Genetic polymorphisms and implications for drug metabolism and toxicity. Expert Opin. Drug Metab. Toxicol. 2008, 4, 439–452. [Google Scholar] [CrossRef]
- Hart, S.N.; Wang, S.; Nakamoto, K.; Wesselman, C.; Li, Y.; Zhong, X. Genetic polymorphisms in cytochrome P450 oxidoreductase influence microsomal P450-catalyzed drug metabolism. Pharmacol. Genom. 2008, 18, 11–24. [Google Scholar] [CrossRef]
- Sandee, D.; Morrissey, K.; Agrawal, V.; Tam, H.K.; Kramer, M.A.; Tracy, T.S.; Giacomini, K.M.; Miller, W.L. Effects of genetic variants of human P450 oxidoreductase on catalysis by CYP2D6 in vitro. Pharmacol. Genom. 2010, 20, 677–686. [Google Scholar] [CrossRef]
- Tulsyan, S.; Mittal, R.D.; Mittal, B. The effect of ABCB1 polymorphisms on the outcome of breast cancer treatment. Pharmgenom. Pers. Med. 2016, 9, 47–58. [Google Scholar]
- Teh, L.K.; Mohamed, N.I.; Salleh, M.Z.; Rohaizak, M.; Shahrun, N.S.; Saladina, J.J.; Shia, J.K.S.; Roslan, H.; Sood, S.; Rajoo, T.S.; et al. The risk of recurrence in breast cancer patients treated with tamoxifen: Polymorphisms of CYP2D6 and ABCB1. AAPS J. 2012, 14, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Sensorn, I.; Sukasem, C.; Sirachainan, E.; Chamnanphon, M.; Pasomsub, E.; Trachu, N.; Supavilai, P.; Pinthong, D.; Wongwaisayawan, S. ABCB1 and ABCC2 and the risk of distant metastasis in Thai breast cancer patients treated with tamoxifen. Onco Targets Ther. 2016, 9, 2121–2129. [Google Scholar] [PubMed]
- Romero-Lorca, A.; Novillo, A.; Gaibar, M.; Bandrés, F.; Fernández-Santander, A. Impacts of the Glucuronidase Genotypes UGT1A4, UGT2B7, UGT2B15 and UGT2B17 on Tamoxifen Metabolism in Breast Cancer Patients. PLoS ONE 2015, 10, e0132269. [Google Scholar] [CrossRef] [PubMed]
- Court, M.; Hao, Q.; Krishnaswamy, S.; Bekaii-Saab, T.; Al-Rohaimi, A.; Von Moltke, L. UDPglucuronosyltransferase (UGT) 2B15 pharmacogenetics: UGT2B15 D85Y genotype and gender are major determinants of oxazepam glucuronidation by human liver. J. Pharmacol. Exp. Ther. 2004, 310, 656–665. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Hesse, L.M.; Hazarika, S.; Masse, G.; Harmatz, J.S.; Greenblatt, D.J.; Court, M.H. Evidence for oxazepam as an in vivo probe of UGT2B15: Oxazepam clearance is reduced by UGT2B15 D85Y polymorphism but unaffected by UGT2B17 deletion. Br. J. Clin. Pharmacol. 2009, 68, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.-Y.; Cho, J.-Y.; Yu, K.-S.; Kim, J.-R.; Jung, H.-R.; Lim, K.-S.; Jang, I.-J.; Shin, S.-G. Effect of the UGT2B15 genotype on the pharmacokinetics, pharmacodynamics, and drug interactions of intravenous lorazepam in healthy volunteers. Clin. Pharmacol. Ther. 2005, 77, 486–494. [Google Scholar] [CrossRef]
- Lee, L.; Cheung, W.Y.; Atkinson, E.; Krzyzanowska, M.K. Impact of comorbidity on chemotherapy use and outcomes in solid tumors: A systematic review. J. Clin. Oncol. 2011, 29, 106–117. [Google Scholar] [CrossRef]
- Borges, S.; Desta, Z.; Li, L.; Skaar, T.C.; Ward, B.A.; Nguyen, A.; Jin, Y.; Storniolo, A.M.; Nikoloff, D.M.; Wu, L.; et al. Quantitative effect of CYP2D6 genotype and inhibitors on tamoxifen metabolism: Implication for optimization of breast cancer treatment. Clin. Pharmacol. Ther. 2006, 80, 61–74. [Google Scholar] [CrossRef]
- Powers, J.L.; Buys, S.S.; Fletcher, D.; Melis, R.; Johnson-Davis, K.L.; Lyon, E.; Malmberg, E.M.; McMillin, G.A. Multigene and Drug Interaction Approach for Tamoxifen Metabolite Patterns Reveals Possible Involvement of CYP2C9, CYP2C19, and ABCB1. J. Clin. Pharm. 2016, 56, 1570–1581. [Google Scholar] [CrossRef]
- Hansten, P.D. The Underrated Risks of Tamoxifen Drug Interactions. Eur. J. Drug Metab. Pharm. 2018, 43, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.H.; Makonnen, E.; Yimer, G.; Seifu, D.; Bekele, A.; Assefa, M.; Aseffa, A.; Howe, R.; Fotoohi, A.; Hassan, M.; et al. CYP2J2∗7 Genotype Predicts Risk of Chemotherapy-Induced Hematologic Toxicity and Reduced Relative Dose Intensity in Ethiopian Breast Cancer Patients. Front. Pharmacol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]

| Parameters | Value | |
|---|---|---|
| Socio-demographics | ||
| Age (years, mean ± SD *) | 39 ± 8.5 | |
| BMI (kg/m2, mean ± SD) | 24.6 (±3.86) | |
| Chemotherapy regimen used | N (%) | |
| FAC ∝ | 37 (45.7) | |
| AC | 11 (11.1) | |
| AC - T | 35 (43.2) | |
| Duration of tamoxifen use (months, median + IQR ♣) | 11 (6–18) | |
| Menopausal status | N (%) | |
| Premenopausal | 56 (69.1) | |
| Postmenopausal | 25 (30.9) | |
| Tumor characteristics | N (%) | |
| Histologic type of tumor | Ductal | 79 (97.5) |
| Lobular | 2 (2.5) | |
| Degree of differentiation | Well differentiated | 21 (25.9) |
| Moderately differentiated | 44(54.3) | |
| Poorly differentiated | 16 (19.8) | |
| Lymph node involvement | Negative | 15 (18.5) |
| Positive | 66 (81.5) | |
| Distant metastatic site | No known distant metastasis | 10 (12.3) |
| Bone, lymph node, or lung only | 63 (77.8) | |
| Liver, CNS, lung + other organs | 8 (9.9) |
| Gene | Variant allele | Allele frequency (%) |
|---|---|---|
| CYP2D6 | *2 | 33.3 |
| *4 | 4.9 | |
| *5 | 4.3 | |
| *10 | 1.9 | |
| *17 | 10.5 | |
| *1×N or *2×N † | 14.8 | |
| CYP2C9 | *2 | 4.3 |
| *3 | 7.4 | |
| CYP2C19 | *2 | 11.7 |
| *3 | 1.2 | |
| CYP3A5 | *3 | 67 |
| POR | *28 | 12.4 |
| ABCB1 c.4036A>G | G | 14.8 |
| ABCB1 c.3435C>T | T | 16.9 |
| UGT2B15 *2 | T | 20.2 |
| UGT2B15 *4 | C | 40.3 |
| CYP2D6 Activity Score | Phenotype Group † | N (%) |
|---|---|---|
| >2 | UM ¥ | 18 (22.2) |
| 2 | NM | 37 (45.7) |
| 1.5 | NM | 12 (14.8) |
| 1 | NM or IM | 10 (12.4) |
| 0.5 | IM | 3 (3.7) |
| 0 | PM | 1 (1.2) |
| Plasma concentrations | values | CV (%) ‡ |
| Tamoxifen (ng/mL, median + IQR #) | 138 (105–200.5) | 56.2 |
| N-Desmethyl-tamoxifen (ng/mL, median + IQR) | 257 (185.5–343) | 53.1 |
| 4-Hydroxy-tamoxifen (ng/mL, median + IQR) | 3.04 (2.15–4.0) | 61 |
| Z-Endoxifen (ng/mL, median + IQR) | 7.94 (4.68–13.75) | 74.7 |
| Metabolic ratio | values | CV (%) ‡ |
| MRεE/NDM (median + IQR) | 0.038 (0.021–0.06) | 59 |
| MRγNDM/Tam (median + IQR) | 1.81 (1.51–2.26) | 28.6 |
| MRλ4-HT/Tam (median + IQR) | 0.022 (0.016–0.029) | 38.8 |
| MR♠E/4-HT (median + IQR) | 2.85 (2.10–3.73) | 43.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, J.H.; Makonnen, E.; Fotoohi, A.; Aseffa, A.; Howe, R.; Aklillu, E. CYP2D6 Genotype Predicts Plasma Concentrations of Tamoxifen Metabolites in Ethiopian Breast Cancer Patients. Cancers 2019, 11, 1353. https://doi.org/10.3390/cancers11091353
Ahmed JH, Makonnen E, Fotoohi A, Aseffa A, Howe R, Aklillu E. CYP2D6 Genotype Predicts Plasma Concentrations of Tamoxifen Metabolites in Ethiopian Breast Cancer Patients. Cancers. 2019; 11(9):1353. https://doi.org/10.3390/cancers11091353
Chicago/Turabian StyleAhmed, Jemal Hussien, Eyasu Makonnen, Alan Fotoohi, Abraham Aseffa, Rawleigh Howe, and Eleni Aklillu. 2019. "CYP2D6 Genotype Predicts Plasma Concentrations of Tamoxifen Metabolites in Ethiopian Breast Cancer Patients" Cancers 11, no. 9: 1353. https://doi.org/10.3390/cancers11091353
APA StyleAhmed, J. H., Makonnen, E., Fotoohi, A., Aseffa, A., Howe, R., & Aklillu, E. (2019). CYP2D6 Genotype Predicts Plasma Concentrations of Tamoxifen Metabolites in Ethiopian Breast Cancer Patients. Cancers, 11(9), 1353. https://doi.org/10.3390/cancers11091353





