Baseline and Early Predictors of Good Patient Candidates for Second-Line after Sorafenib Treatment in Unresectable Hepatocellular Carcinoma
Abstract
1. Introduction
2. Methods
2.1. Patients
2.2. Sorafenib Treatment
2.3. Data Collection
2.4. Definition of Criteria of a Good Candidate for Second-Line Treatment
2.5. Statistical Analysis
3. Results
3.1. Baseline Data
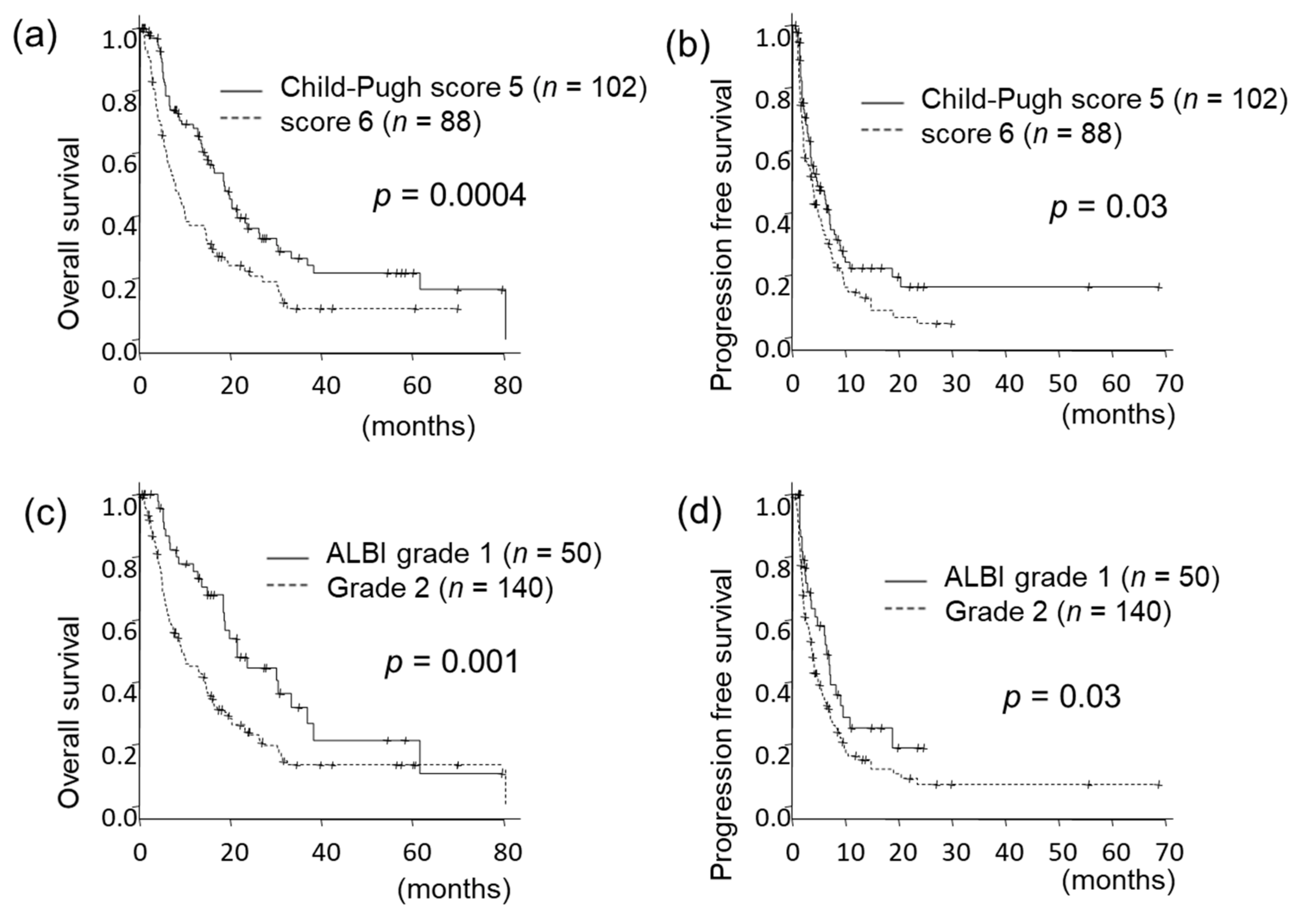
3.2. Prognosis According to Baseline Liver Function
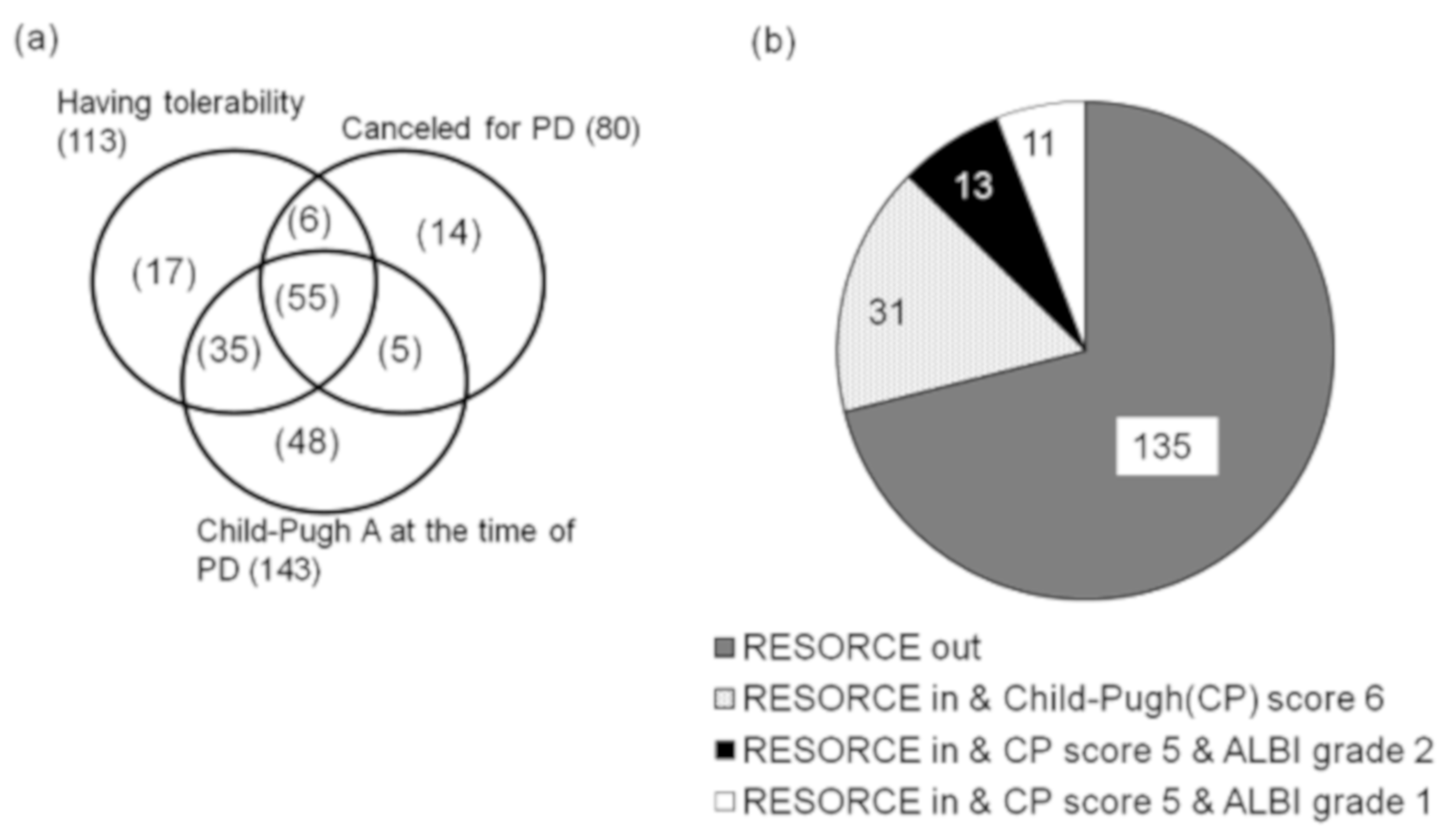
3.3. Frequency of Patients Fulfilling Criteria as a Good Candidate for Second-Line Treatment
3.4. Predictors for Fulfilling the RESORCE Study Inclusion Criteria at the Time of Sorafenib Discontinuation
3.5. Predictors for Fulfilling the RESORCE Study’s Inclusion Criteria plus Child–Pugh Score 5 at the Time of Sorafenib Discontinuation
3.6. Predictors for Fulfilling the RESORCE Study Inclusion Criteria Plus ALBI Grade 1 at the Time of Sorafenib Discontinuation
3.7. Predictive Value of These Criteria
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Lencioni, R.; Marrero, J.; Venook, A.; Ye, S.L.; Kudo, M. Design and rationale for the non-interventional Global Investigation of Therapeutic DEcisions in Hepatocellular Carcinoma and Of its Treatment with Sorafenib (GIDEON) study. Int. J. Clin. Pract. 2010, 64, 1034–1041. [Google Scholar] [CrossRef]
- Takeda, H.; Nishikawa, H.; Osaki, Y.; Tsuchiya, K.; Joko, K.; Ogawa, C.; Taniguchi, H.; Orito, E.; Uchida, Y.; Izumi, N. Proposal of Japan Red Cross score for sorafenib therapy in hepatocellular carcinoma. Hepatol. Res. 2015, 45, E130–E140. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Nishikawa, H.; Osaki, Y.; Tsuchiya, K.; Joko, K.; Ogawa, C.; Taniguchi, H.; Orito, E.; Uchida, Y.; Izumi, N. Clinical features associated with radiological response to sorafenib in unresectable hepatocellular carcinoma: A large multicenter study in Japan. Liver Int. 2015, 35, 1581–1589. [Google Scholar] [CrossRef]
- Bruix, J.; Tak, W.Y.; Gasbarrini, A.; Santoro, A.; Colombo, M.; Lim, H.Y.; Mazzaferro, V.; Wiest, R.; Reig, M.; Wagner, A.; et al. Regorafenib as second-line therapy for intermediate or advanced hepatocellular carcinoma: Multicentre, open-label, phase II safety study. Eur. J. Cancer 2013, 49, 3412–3419. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl. J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef]
- Zhu, A.X.; Park, J.O.; Ryoo, B.Y.; Yen, C.J.; Poon, R.; Pastorelli, D.; Blanc, J.F.; Chung, H.C.; Baron, A.D.; Pfiffer, T.E.; et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): A randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015, 16, 859–870. [Google Scholar] [CrossRef]
- Johnson, P.J.; Berhane, S.; Kagebayashi, C.; Satomura, S.; Teng, M.; Reeves, H.L.; O’Beirne, J.; Fox, R.; Skowronska, A.; Palmer, D.; et al. Assessment of liver function in patients with hepatocellular carcinoma: A new evidence-based approach-the ALBI grade. J. Clin. Oncol. 2015, 33, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, S.; Chiba, T.; Ooka, Y.; Suzuki, E.; Kanogawa, N.; Saito, T.; Motoyama, T.; Tawada, A.; Kanai, F.; Yokosuka, O. Liver function assessment according to the Albumin-Bilirubin (ALBI) grade in sorafenib-treated patients with advanced hepatocellular carcinoma. Investig. New Drugs 2015, 33, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Michitaka, K.; Toyoda, H.; Tada, T.; Ueki, H.; Kaneto, M.; Aibiki, T.; Okudaira, T.; Kawakami, T.; et al. Usefulness of albumin-bilirubin grade for evaluation of prognosis of 2584 Japanese patients with hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2016, 31, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Tada, T.; Kumada, T.; Toyoda, H.; Tsuji, K.; Hiraoka, A.; Michitaka, K.; Deguchi, A.; Ishikawa, T.; Imai, M.; Ochi, H.; et al. Impact of albumin-bilirubin grade on survival in patients with hepatocellular carcinoma who received sorafenib: An analysis using time-dependent receiver operating characteristic. J. Gastroenterol. Hepatol. 2019, 34, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Kariyama, K.; Takaguchi, K.; Atsukawa, M.; Itobayashi, E.; Tsuji, K.; Tajiri, K.; Hirooka, M.; Shimada, N.; et al. Clinical features of lenvatinib for unresectable hepatocellular carcinoma in real-world conditions: Multicenter analysis. Cancer Med. 2019, 8, 137–146. [Google Scholar] [CrossRef]
- Hiraoka, A.; Kumada, T.; Kariyama, K.; Takaguchi, K.; Itobayashi, E.; Shimada, N.; Tajiri, K.; Tsuji, K.; Ishikawa, T.; Ochi, H.; et al. Therapeutic potential of lenvatinib for unresectable hepatocellular carcinoma in clinical practice: Multicenter analysis. Hepatol. Res. 2019, 49, 111–117. [Google Scholar] [CrossRef]
- Heimbach, J.K.; Kulik, L.M.; Finn, R.S.; Sirlin, C.B.; Abecassis, M.M.; Roberts, L.R.; Zhu, A.X.; Murad, M.H.; Marrero, J.A. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018, 67, 358–380. [Google Scholar] [CrossRef]
- Bruix, J.; Sherman, M. Management of hepatocellular carcinoma: An update. Hepatology 2011, 53, 1020–1022. [Google Scholar] [CrossRef]
- World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2000, 284, 3043–3045. [CrossRef]
- World Medical Association. Declaration of Helsinki. Law Med. Health Care 1991, 19, 264–265. [Google Scholar] [CrossRef]
- Kudo, M.; Matsui, O.; Izumi, N.; Iijima, H.; Kadoya, M.; Imai, Y.; Okusaka, T.; Miyayama, S.; Tsuchiya, K.; Ueshima, K.; et al. JSH Consensus-Based Clinical Practice Guidelines for the Management of Hepatocellular Carcinoma: 2014 Update by the Liver Cancer Study Group of Japan. Liver Cancer 2014, 3, 458–468. [Google Scholar] [CrossRef] [PubMed]
- EASL-EORTC clinical practice guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2012, 56, 908–943. [CrossRef] [PubMed]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Kuzuya, T.; Ishigami, M.; Ishizu, Y.; Honda, T.; Hayashi, K.; Ishikawa, T.; Nakano, I.; Hirooka, Y.; Goto, H. Prognostic Factors Associated with Postprogression Survival in Advanced Hepatocellular Carcinoma Patients Treated with Sorafenib Not Eligible for Second-Line Regorafenib Treatment. Oncology 2018, 95, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K. Recent advances in medical management of hepatocellular carcinoma. Hepatol. Res. 2019, 49, 14–32. [Google Scholar] [CrossRef] [PubMed]
- Terashima, T.; Yamashita, T.; Sunagozaka, H.; Arai, K.; Kawaguchi, K.; Kitamura, K.; Sakai, Y.; Mizukoshi, E.; Honda, M.; Kaneko, S. Analysis of the liver functional reserve of patients with advanced hepatocellular carcinoma undergoing sorafenib treatment: Prospects for regorafenib therapy. Hepatol. Res. 2018, 48, 956–966. [Google Scholar] [CrossRef]
- Uchikawa, S.; Kawaoka, T.; Aikata, H.; Kodama, K.; Nishida, Y.; Inagaki, Y.; Hatooka, M.; Morio, K.; Nakahara, T.; Murakami, E.; et al. Clinical outcomes of sorafenib treatment failure for advanced hepatocellular carcinoma and candidates for regorafenib treatment in real-world practice. Hepatol. Res. 2018, 48, 814–820. [Google Scholar] [CrossRef]
- Ogasawara, S.; Chiba, T.; Ooka, Y.; Suzuki, E.; Maeda, T.; Yokoyama, M.; Wakamatsu, T.; Inoue, M.; Saito, T.; Kobayashi, K.; et al. Characteristics of patients with sorafenib-treated advanced hepatocellular carcinoma eligible for second-line treatment. Investig. New Drugs 2018, 36, 332–339. [Google Scholar] [CrossRef]
- Yukimoto, A.; Hirooka, M.; Hiraoka, A.; Michitaka, K.; Ochi, H.; Joko, K.; Imai, Y.; Watanabe, T.; Koizumi, Y.; Yoshida, O.; et al. Using ALBI score at the start of sorafenib treatment to predict regorafenib treatment candidates in patients with hepatocellular carcinoma. Jpn. J. Clin. Oncol. 2019, 49, 42–47. [Google Scholar] [CrossRef]
| Variables | n = 190 |
|---|---|
| Age: years, mean ± SD | 72 ± 9 |
| Male: n (%) | 149 (78%) |
| Maximum size of HCC nodule in the liver: mm, median (range in IQR) | 38 (20–74) |
| Number of HCC nodules: 1/2–3/4 or more, n (%) | 28 (15%)/32 (17%)/130 (68%) |
| Tumor burden (maximum size + number of HCC nodules): mean ± SD | 12 ± 6 |
| BCLC stage: B/C, n (%) | 86 (45%)/104 (55%) |
| Presence of MVI: n (%) | 45 (24%) |
| Presence of metastasis: n (%) | 76 (40%) |
| Albumin: g/dL, mean ± SD | 3.6 ± 0.4 |
| Total bilirubin: g/dL, mean ± SD | 0.9 ± 0.5 |
| Prothrombin time: %, mean ± SD | 98 ± 16 |
| Child–Pugh score: 5/6, n (%) | 102/88 (54%/46%) |
| ALBI predictor, mean ± SD | −2.30 ± 0.42 |
| ALBI grade: 1/2, n (%) | 50/140 (26%/74%) |
| AFP: ng/mL, median (range in IQR) | 181(13–3080) |
| AFP L3 index: % (range in IQR) | 18 (5.8–49) |
| PIVKA-II: mAU/mL, median (range in IQR) | 319 (49–2308) |
| Factors | Univariate | Multivariate Model 1: ALBI | Multivariate Model 2: Alb | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR | p Value | Cut-Off Value | OR | 95% CI | p Value | Cut-Off Value | OR | 95% CI | p Value | |
| Baseline factors | ||||||||||
| Age | 0.67 | 0.37 | ||||||||
| Male | 0.63 | 0.27 | ||||||||
| Tumor burden | 1.0 | 0.91 | ||||||||
| MVI | 0.63 | 0.26 | ||||||||
| Metastasis | 0.91 | 0.74 | ||||||||
| BCLC stage C | 0.67 | 0.19 | ||||||||
| Albumin (g/dL) | 2.5 | 0.01 | - | >3.7 g/dL | 2.7 | 1.4–5.2 | 0.004 | |||
| T.Bil (g/dL) | 0.54 | 0.19 | ||||||||
| Prothrombin time (%) | 0.99 | 0.24 | ||||||||
| ALBI predictor | 3.1 | 0.005 | <−2.33 | 2.5 | 1.3–5.2 | 0.01 | - | |||
| AFP (ng/mL) | 0.71 | 0.34 | ||||||||
| PIVKA-II (mAU/mL) | 1.0 | 0.54 | ||||||||
| Factors at week 4 | ||||||||||
| ΔAlb at week4 (g/dL) | 3.8 | <0.001 | - | <−0.2 g/dL | 2.6 | 1.3–5.2 | 0.005 | |||
| ΔT.Bil at week4 (mg/dL) | 0.56 | 0.35 | ||||||||
| ΔALBI at week4 | 0.29 | 0.015 | <0.255 | 4.9 | 2.1–11 | 0.0001 | - | |||
| Factors | Univariate | Multivariate Model 1: ALBI | Multivariate Model 2: Alb | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR | p Value | Cut-Off Value | OR | 95% CI | p Value | Cut-Off Value | OR | 95% CI | p Value | |
| Baseline factors | ||||||||||
| Age | 1.0 | 0.20 | ||||||||
| Male | 2.1 | 0.26 | ||||||||
| Tumor burden | 0.96 | 0.23 | ||||||||
| MVI | 0.26 | 0.07 | ||||||||
| Metastasis | 0.92 | 0.86 | ||||||||
| BCLC stage C | 0.55 | 0.17 | ||||||||
| Albumin (g/dL) | 2.8 | 0.04 | - | >3.7 g/dL | 2.9 | 1.2–7.2 | 0.02 | |||
| T.Bil (g/dL) | 0.13 | 0.01 | ||||||||
| Prothrombin time (%) | 1.0 | 0.44 | ||||||||
| ALBI predictor | 0.21 | 0.005 | <−2.33 | 5.4 | 1.9–16 | 0.002 | - | |||
| AFP (ng/mL) | 1.0 | 0.63 | ||||||||
| PIVKA-II (mAU/mL) | 1.0 | 0.23 | ||||||||
| Factors at week 4 | ||||||||||
| ΔAlb at week4 (g/dL) | 21 | <0.001 | - | <−0.1 g/dL | 4.1 | 1.6–11 | 0.003 | |||
| ΔT.Bil at week4 (mg/dL) | 0.92 | 0.82 | ||||||||
| ΔALBI at week4 | 0.13 | 0.005 | <0.19 | 5.9 | 1.9–19 | 0.003 | - | |||
| Factors | Univariate | Multivariate Model 1: ALBI | Multivariate Model 2: Alb | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR | p Value | Cut-Off Value | OR | 95% CI | p Value | Cut-Off Value | OR | 95% CI | p Value | |
| Baseline factors | ||||||||||
| Age | 0.99 | 0.18 | ||||||||
| Male | 1.3 | 0.78 | ||||||||
| Tumor burden | 0.87 | 0.01 | <11 | 13 | 1.5–118 | 0.02 | <11 | 12 | 1.5–12 | 0.0002 |
| MVI | 0.81 | 0.77 | ||||||||
| metastasis | 0.36 | 0.11 | ||||||||
| BCLC stage C | 0.43 | 0.90 | ||||||||
| Albumin (g/dL) | 9.8 | 0.002 | - | >3.8 g/dL | 8.7 | 2.0–39 | 0.004 | |||
| T.Bil (g/dL) | 0.42 | 0.34 | ||||||||
| Prothrombin time (%) | 1.0 | 0.53 | ||||||||
| ALBI predictor | 0.07 | 0.002 | <−2.55 | 14 | 2.4–76 | 0.003 | - | |||
| AFP (ng/mL) | 1.0 | 0.79 | ||||||||
| PIVKA-II (mAU/mL) | 1.0 | 0.43 | ||||||||
| Factors at week 4 | ||||||||||
| ΔAlb at week4 (g/dL) | 21 | <0.001 | - | <−0.1 g/dL | 5.4 | 1.2–24 | 0.03 | |||
| ΔT.Bil at week4 (mg/dL) | 0.62 | 0.27 | ||||||||
| ΔALBI at week4 | 0.08 | 0.01 | <0.085 | 20 | 2.2–177 | 0.008 | - | |||
| Factors | RESORCE Study’s Criteria | RESORCE Study’s Criteria + Child–Pugh Score5 | RESORCE Study’s Criteria +ALBI Grade1 |
|---|---|---|---|
| Baseline albumin (g/dL) | 3.7 | 3.7 | 3.8 |
| Baseline ALBI predictor | −2.33 | −2.33 | −2.55 |
| ΔAlb at week 4 (g/dL) | −0.2 | −0.1 | −0.1 |
| ΔALBI at week 4 | 0.255 | 0.19 | 0.085 |
| Tumor burden | NS | NS | 11 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takada, H.; Kurosaki, M.; Tsuchiya, K.; Komiyama, Y.; Itakura, J.; Takahashi, Y.; Nakanishi, H.; Yasui, Y.; Tamaki, N.; Maeyashiki, C.; et al. Baseline and Early Predictors of Good Patient Candidates for Second-Line after Sorafenib Treatment in Unresectable Hepatocellular Carcinoma. Cancers 2019, 11, 1256. https://doi.org/10.3390/cancers11091256
Takada H, Kurosaki M, Tsuchiya K, Komiyama Y, Itakura J, Takahashi Y, Nakanishi H, Yasui Y, Tamaki N, Maeyashiki C, et al. Baseline and Early Predictors of Good Patient Candidates for Second-Line after Sorafenib Treatment in Unresectable Hepatocellular Carcinoma. Cancers. 2019; 11(9):1256. https://doi.org/10.3390/cancers11091256
Chicago/Turabian StyleTakada, Hitomi, Masayuki Kurosaki, Kaoru Tsuchiya, Yasuyuki Komiyama, Jun Itakura, Yuka Takahashi, Hiroyuki Nakanishi, Yutaka Yasui, Nobuharu Tamaki, Chiaki Maeyashiki, and et al. 2019. "Baseline and Early Predictors of Good Patient Candidates for Second-Line after Sorafenib Treatment in Unresectable Hepatocellular Carcinoma" Cancers 11, no. 9: 1256. https://doi.org/10.3390/cancers11091256
APA StyleTakada, H., Kurosaki, M., Tsuchiya, K., Komiyama, Y., Itakura, J., Takahashi, Y., Nakanishi, H., Yasui, Y., Tamaki, N., Maeyashiki, C., Kaneko, S., Takaura, K., Higuchi, M., Okada, M., Wang, W., Osawa, L., Sekiguchi, S., Hayakawa, Y., Yamashita, K., ... Izumi, N. (2019). Baseline and Early Predictors of Good Patient Candidates for Second-Line after Sorafenib Treatment in Unresectable Hepatocellular Carcinoma. Cancers, 11(9), 1256. https://doi.org/10.3390/cancers11091256





