Down-Regulation of Cannabinoid Type 1 (CB1) Receptor and its Downstream Signaling Pathways in Metastatic Colorectal Cancer
Abstract
1. Introduction
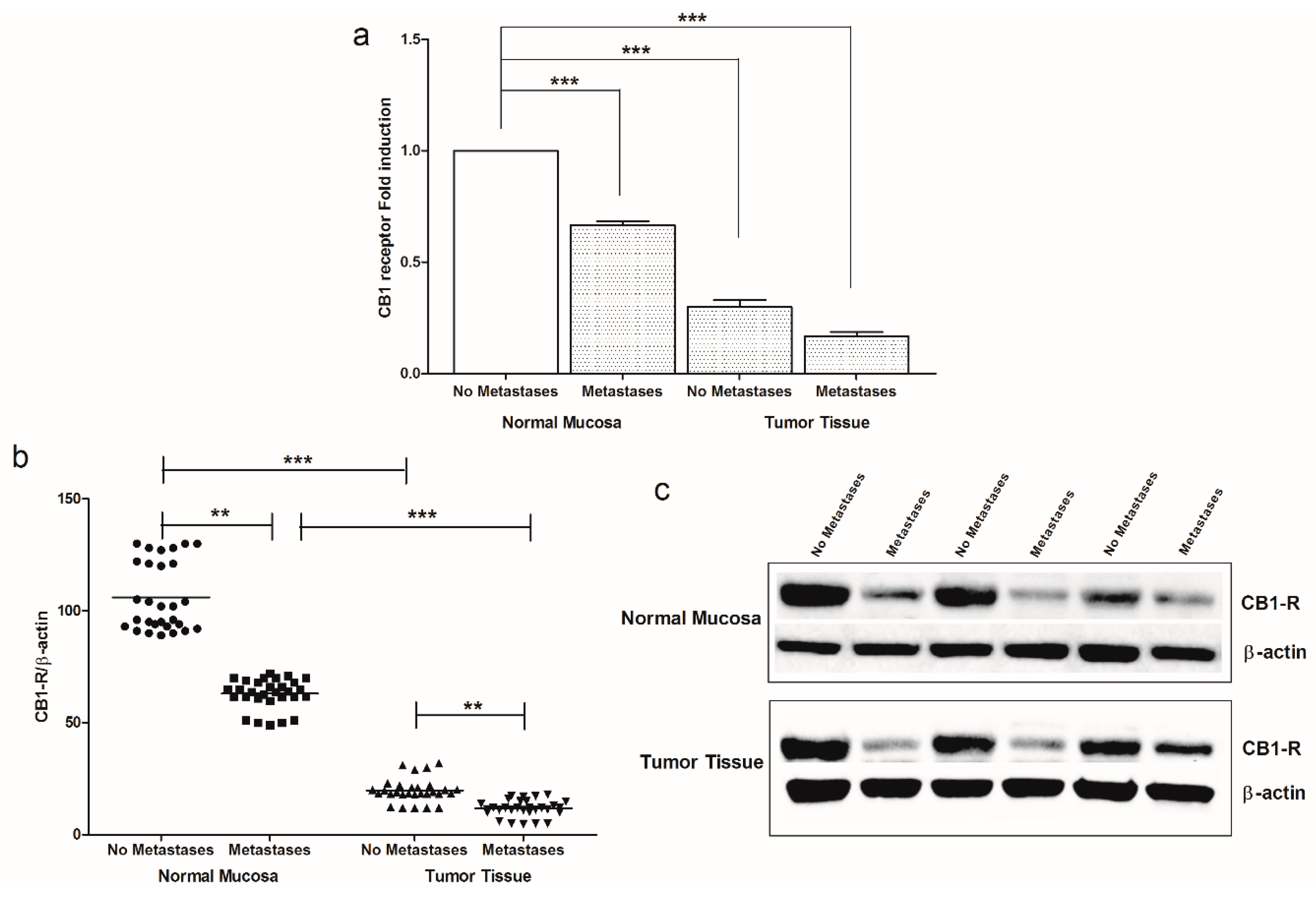
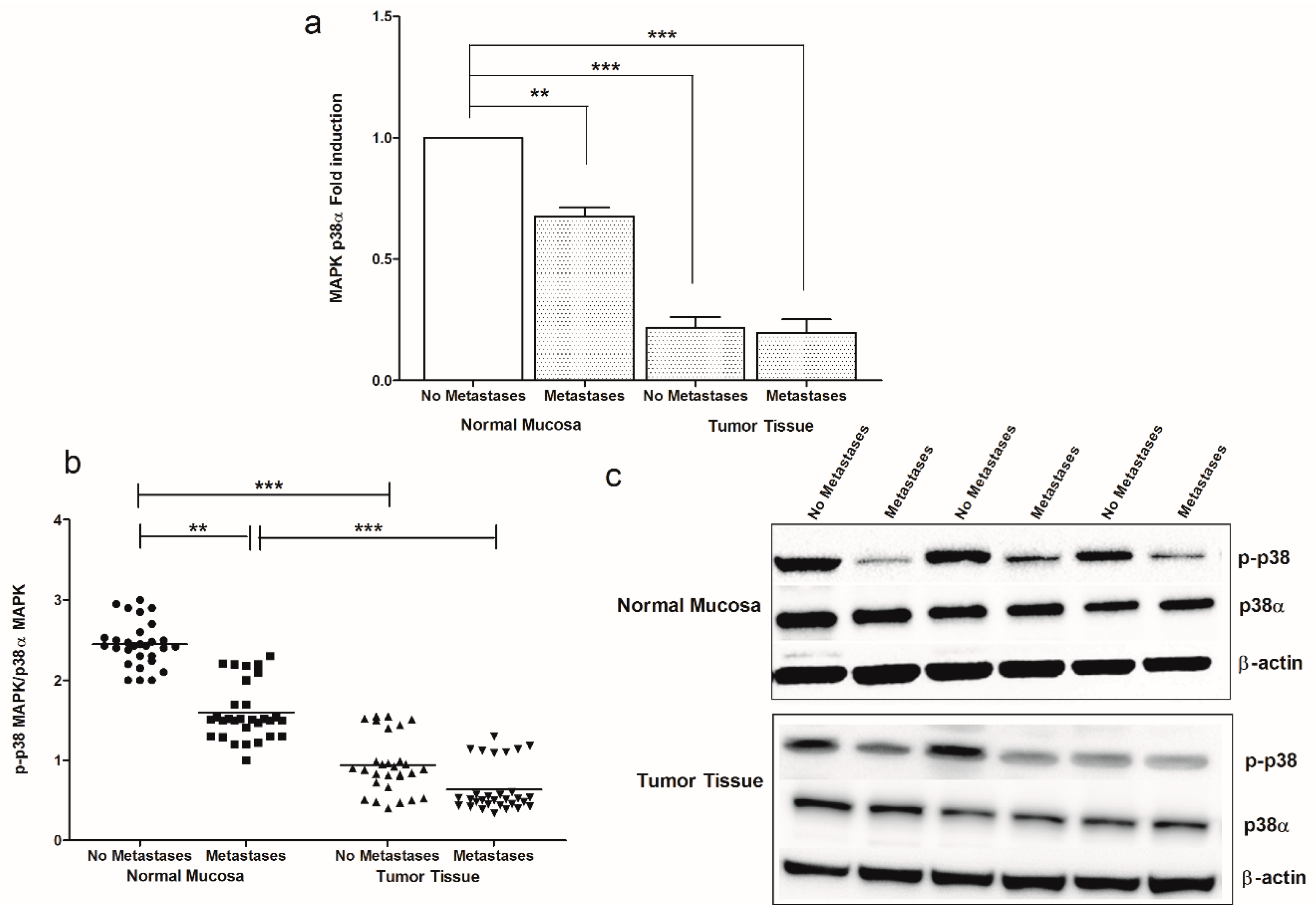
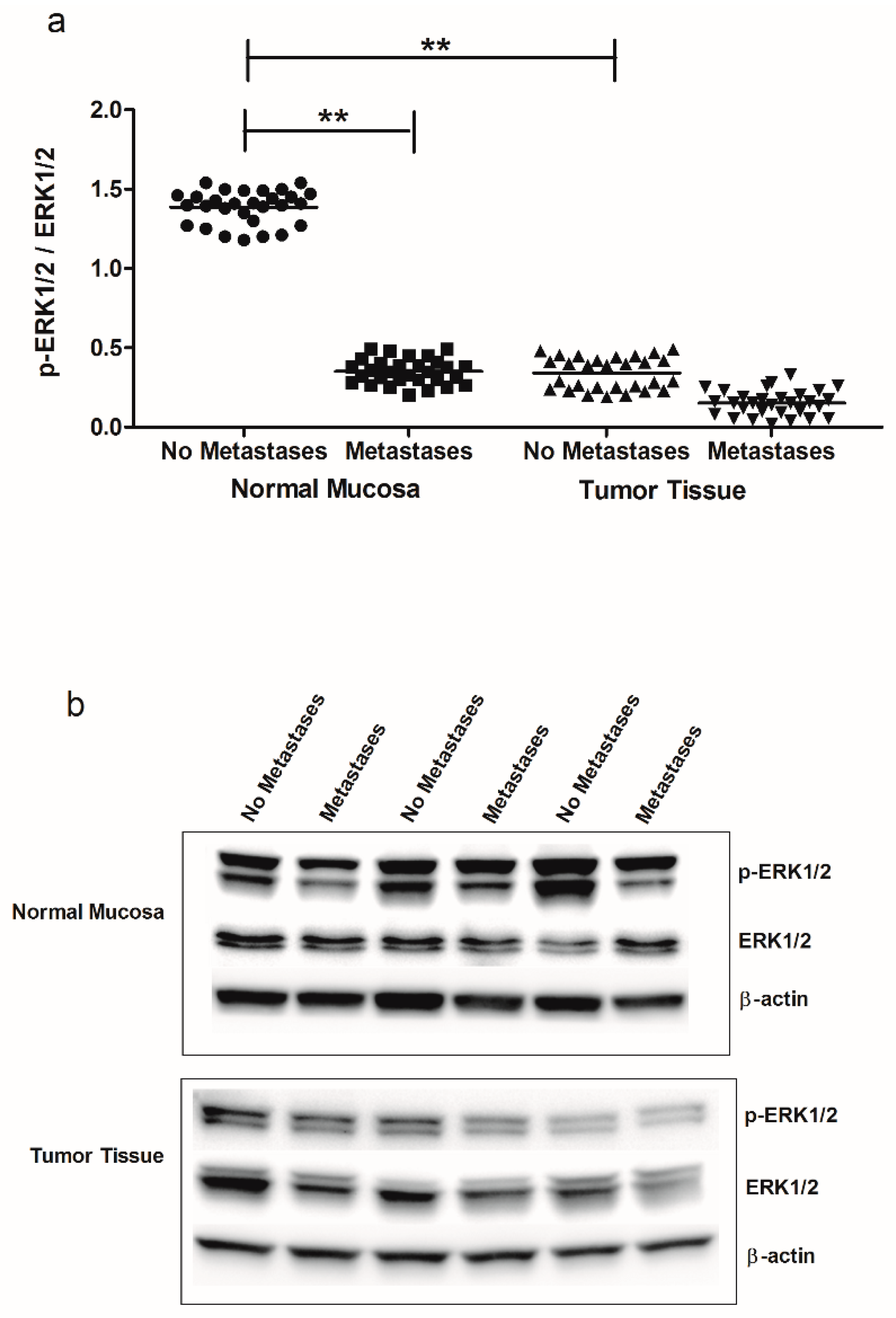
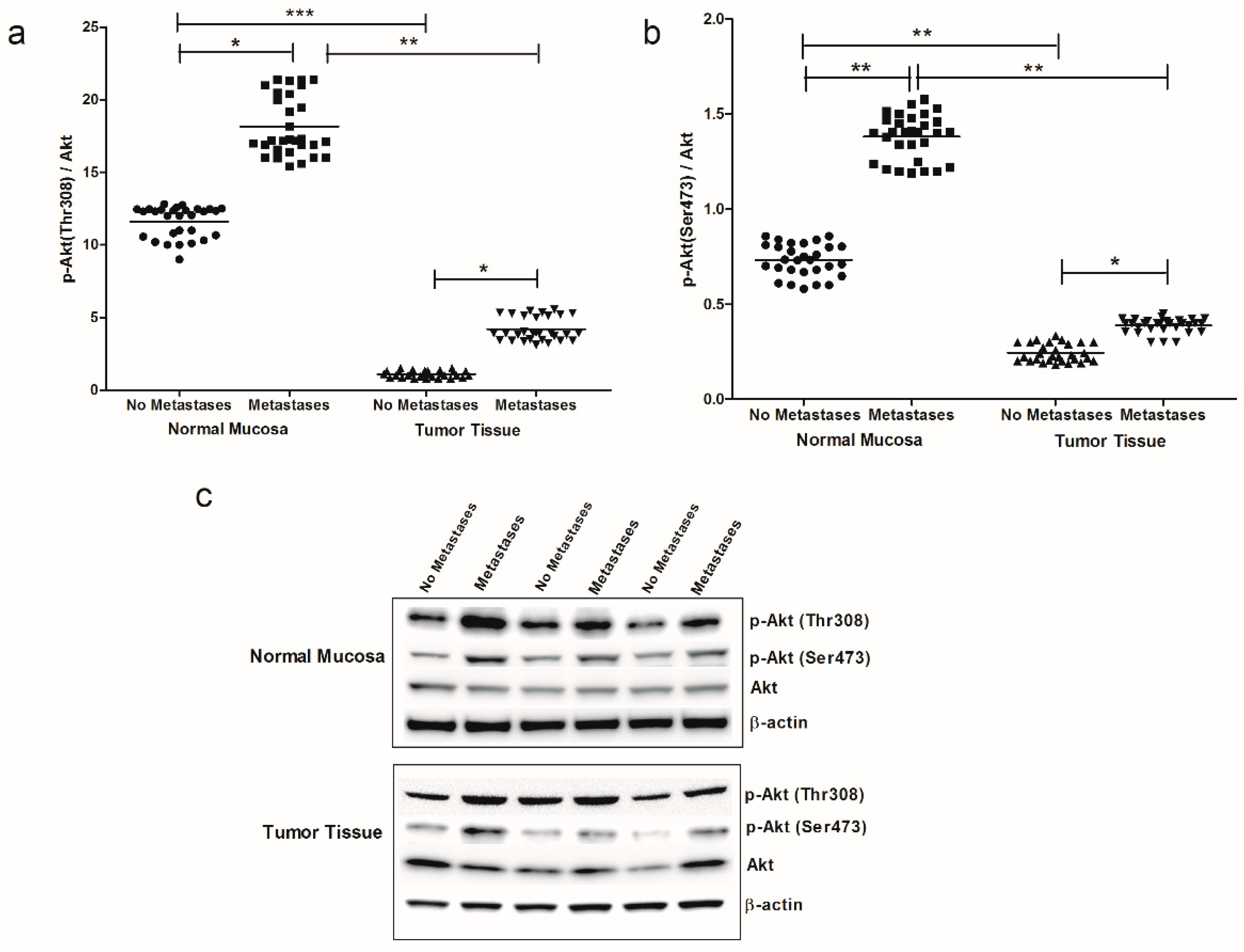
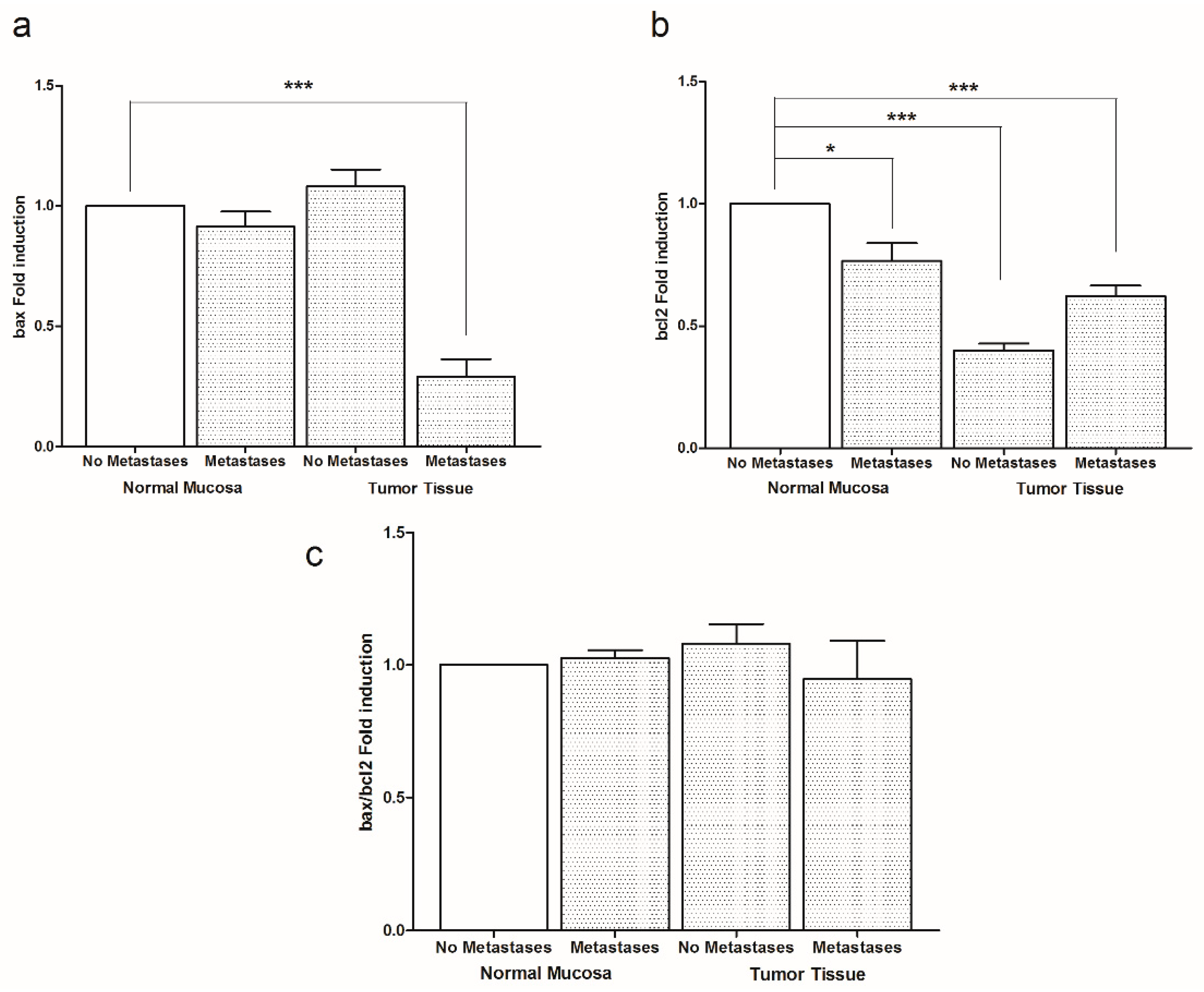
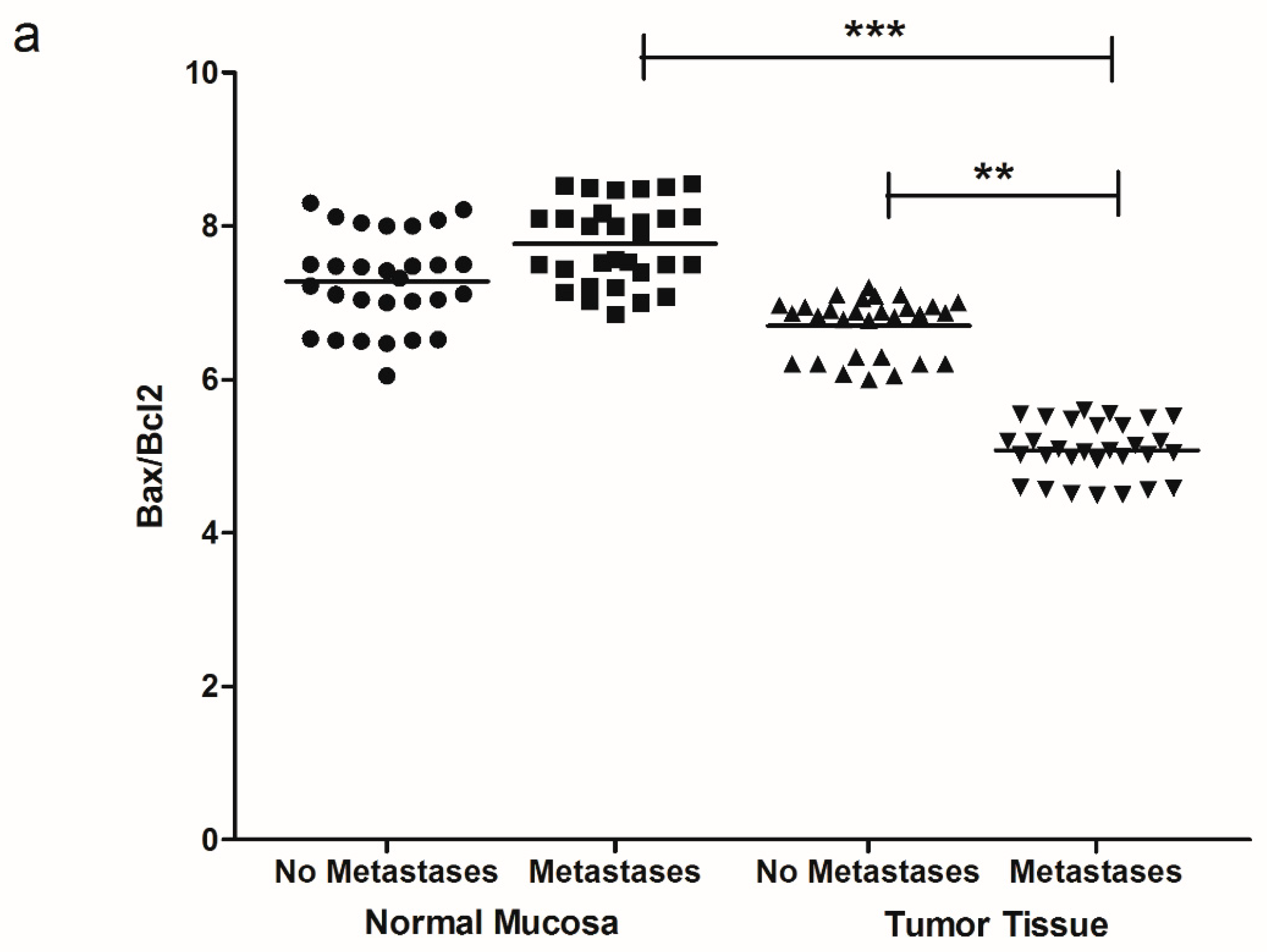
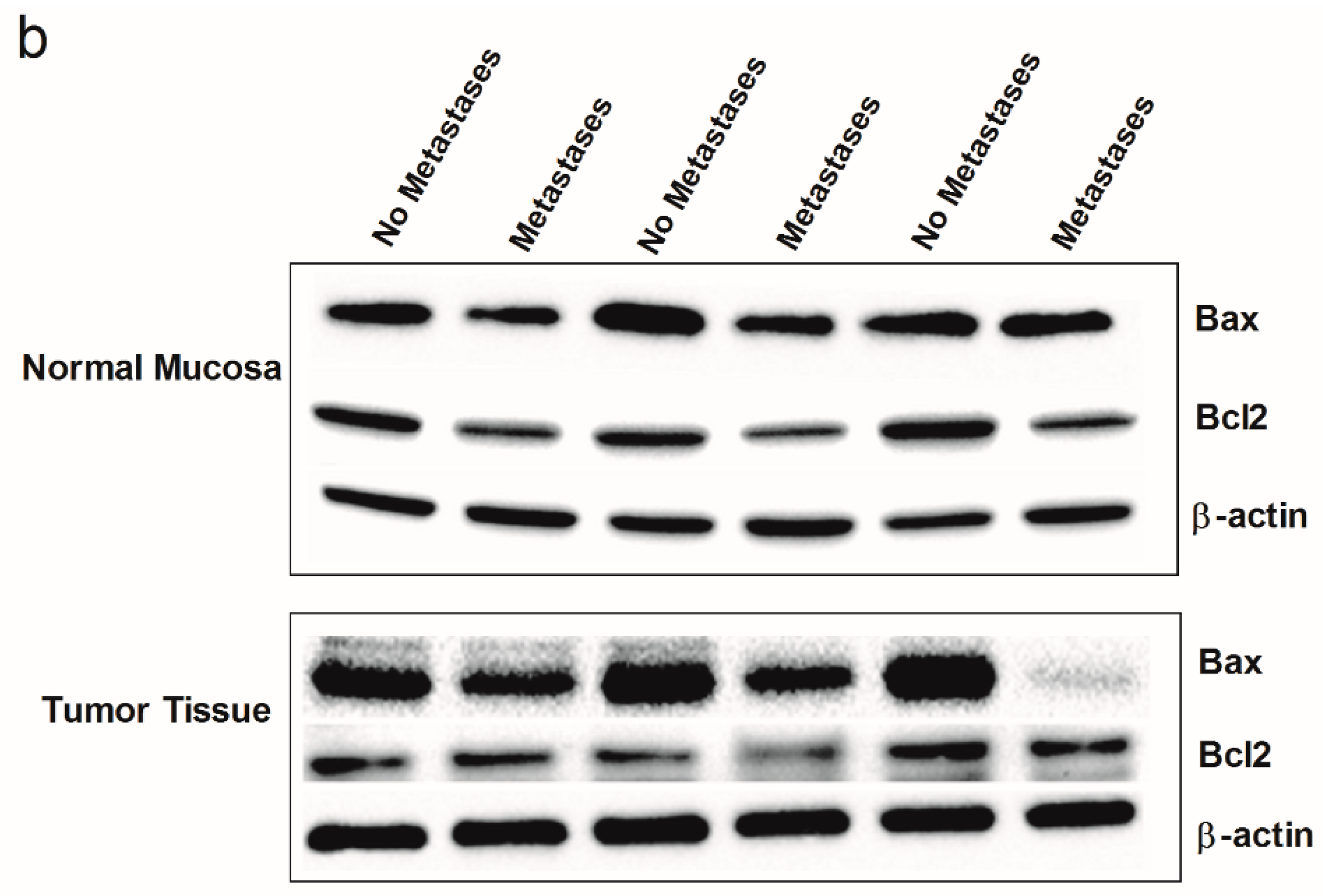
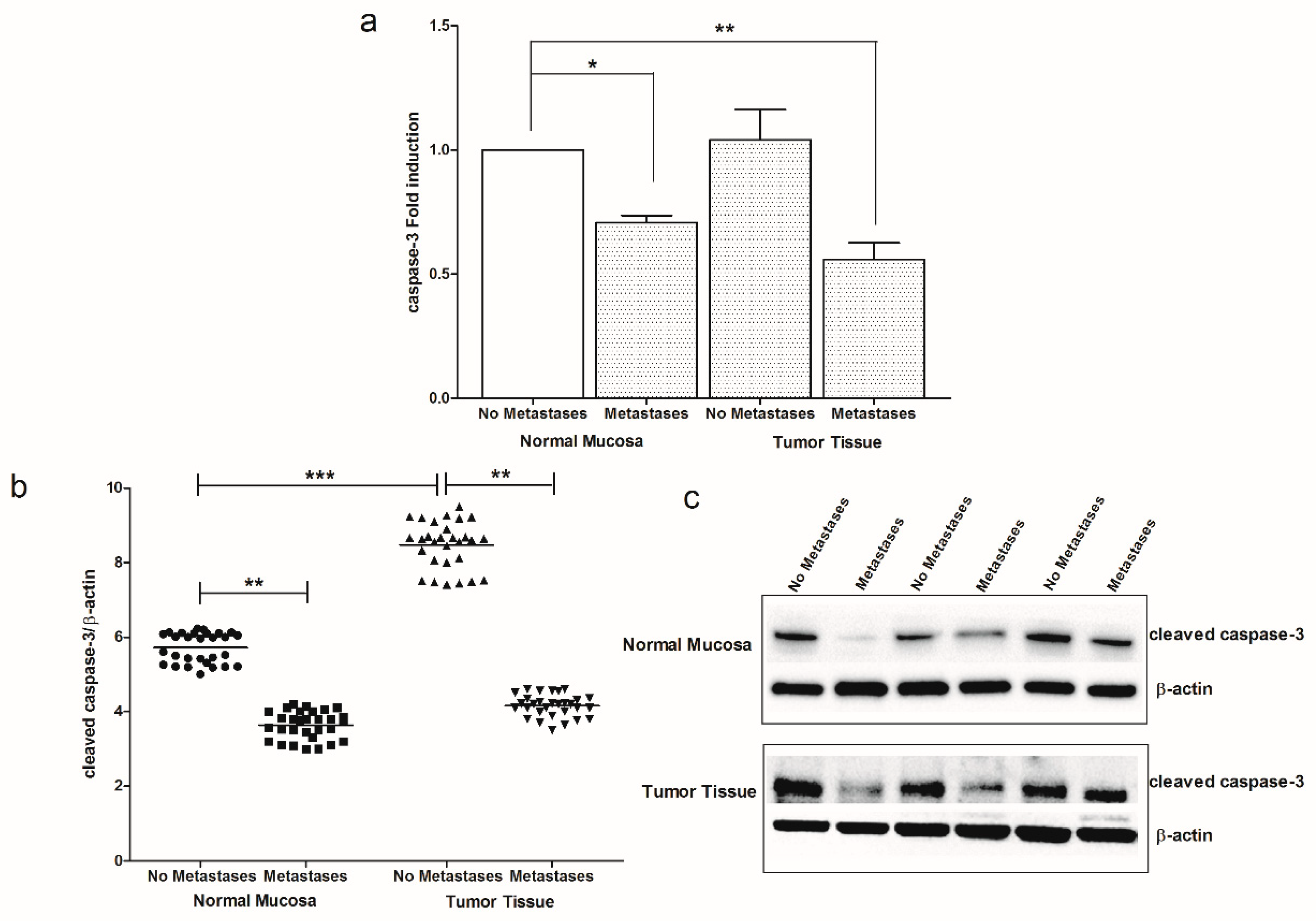
2. Results
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Gene Expression Analysis
4.3. Western Blotting
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Pertwee, R.G. Emerging strategies for exploiting cannabinoid receptor agonists as medicines. Br. J. Pharmacol. 2009, 156, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, V.; Bifulco, M.; De Petrocellis, L. The endocannabinoid system and its therapeutic exploitation. Nat. Rev. Drug. Discov. 2004, 3, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Velasco, G.; Sànchez, C.; Guzmàn, M. Anticancer mechanisms of cannabinoids. Curr. Oncol. 2016, 23, S23–S32. [Google Scholar] [PubMed]
- Guzman, N. Cannabinoids: Potential anticancer agents. Nat. Rev. Cancer 2003, 3, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Malfitano, A.M.; Ciaglia, E.; Gangemi, G.; Gazzerro, P.; Laezza, C.; Bifulco, M. Update on the endocannabinoid system as an anticancer target. Expert Opin. Ther. Targets 2011, 15, 297–308. [Google Scholar] [CrossRef]
- Pisanti, S.; Picardi, P.; D’Alessandro, A.; Laezza, C.; Bifulco, M. The endocannabinoid signaling system in cancer. Trends Pharmacol. Sci. 2013, 34, 273–282. [Google Scholar] [CrossRef]
- Michalski, C.W.; Oti, F.E.; Erkan, M.; Sauliunaite, D.; Bergmann, F.; Pacher, P.; Batkai, S.; Müller, M.W.; Giese, N.A.; Friess, H.; et al. Cannabinoids in pancreatic cancer: Correlation with survival and pain. Int. J. Cancer 2008, 122, 742–750. [Google Scholar] [CrossRef]
- Xu, X.; Liu, Y.; Huang, S.; Liu, G.; Xie, C.; Zhou, J.; Fan, W.; Li, Q.; Wang, Q.; Zhong, D.; et al. Overexpression of cannabinoid receptors CB1 and CB2 correlates with improved prognosis of patients with improved prognosis of patients with hepatocellular carcinoma. Cancer Genet. Cytogenet. 2006, 171, 31–38. [Google Scholar] [CrossRef]
- Wang, D.; Wang, H.; Ning, W.; Backlund, M.G.; Dey, S.K.; DuBois, R.N. Loss of cannabinoid receptor 1 accelerates intestinal tumor growth. Cancer Res. 2008, 68, 6468–6476. [Google Scholar] [CrossRef]
- Massa, F.; Marsicano, G.; Hermann, H.; Cannich, A.; Monory, K.; Cravatt, B.F.; Ferri, G.L.; Sibaev, A.; Storr, M.; Lutz, B. The endogenous cannabinoid system protects against colonic inflammation. J. Clin. Invest. 2004, 113, 1202–1209. [Google Scholar] [CrossRef]
- Notarnicola, M.; Tutino, V.; De Nunzio, V.; Dituri, F.; Caruso, M.G.; Giannelli, G. Dietary omega-3 polyunsaturated fatty acids inhibit tumor growth in transgenic ApcMin/+ mice, correlating with CB1 receptor up-regulation. IJMS 2017, 18, 485. [Google Scholar] [CrossRef]
- Refolo, M.G.; D’alessandro, R.; Malerba, N.; Laezza, C.; Bifulco, M.; Messa, C.; Caruso, M.G.; Notarnicola, M.; Tutino, V. Anti proliferative and pro apoptotic effects of flavonoid quercetin are mediated by CB1 receptor in human colon cancer cell lines. J. Cell. Physiol. 2015, 230, 2973–2980. [Google Scholar] [CrossRef]
- Linsalata, M.; Notarnicola, M.; Tutino, V.; Bifulco, M.; Santoro, A.; Laezza, C.; Messa, C.; Orlando, A.; Caruso, M.G. Effects of Anandamide on Polyamine Levels and cell growth in human colon cancer cells. Anticancer. Res. 2010, 30, 2583–2590. [Google Scholar]
- Olea-Herrero, N.; Vara, D.; Malagarie-Cazenave, S.; Díaz-Laviada, I. Inhibition of human tumor prostate PC-3 cell growth by cannabinoids R+ methanandamide and JWH-015: Involvement of CB2. Br. J. Cancer 2009, 101, 940–950. [Google Scholar] [CrossRef]
- Grimaldi, C.; Pisanti, S.; Laezza, C.; Malfitano, A.M.; Santoro, A.; Vitale, M.; Caruso, M.G.; Notarnicola, M.; Iacuzzo, I.; Portella, G.; et al. Anandamide inhibits adhesion and migration of breast cancer cells. Exp. Cell. Res. 2016, 312, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Bogdanovic, V.; Mrdjanovic, J.; Borisev, I. A review of therapeutic antitumor potential of cannabinoids. JACM 2017, 11, 831–836. [Google Scholar] [CrossRef]
- Gazzerro, P.; Malfitano, A.M.; Santoro, A.; Pisanti, S.; Caruso, M.G.; Notarnicola, M.; Messa, C.; Laezza, C.; Misso, G.; Caraglia, M.; et al. Synergistic inhibition of human colon cancer cell growth by the cannabinoid CB1 receptor antagonist rimonabant and oxaliplatin. Oncol. Rep. 2010, 23, 171–175. [Google Scholar] [PubMed]
- Blanchard, T.G.; Czinn, S.J.; Banerjee, V.; Sharda, N.; Bafford, A.C.; Mubariz, F.; Morozov, D.; Passaniti, A.; Ahmed, H.; Banerjee, A. Identification of cross talk between FoxM1 and RASSF1A as a Therapeutic target of colon cancer. Cancers 2019, 11, 199. [Google Scholar] [CrossRef]
- Burge, M.; Price, T.; Karapetis, C.S. First-line therapy for metastatic colorectal cancer: Current perspectives and future directions. Asia-Pac. J. Clin. Oncol. 2019, 15, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Joseph, J.; Niggemann, B.; Zaenker, K.S.; Entschladen, F. Anandamide is an endogenous inhibitor for the migration of tumor cells and T lymphocytes. Cancer Immunol. Immunother. 2004, 53, 723–728. [Google Scholar] [CrossRef]
- Pisanti, S.; Borselli, C.; Oliviero, O.; Laezza, C.; Gazzerro, P.; Bifulco, M. Antiangiogenic activity of the endocannabinoid anandamide: Correlation to its tumor-suppressor efficacy. J. Cell. Physiol. 2007, 211, 495–503. [Google Scholar] [CrossRef]
- Ciaglia, E.; Torelli, G.; Pisanti, S.; Picardi, P.; D’Alessandro, A.; Laezza, C.; Malfitano, A.M.; Fiore, D.; Pagano Zottola, A.C.; Proto, M.C.; et al. Cannabinoid receptor CB1 regulates STAT3 activity and its expression dictates the responsiveness to SR141716 treatment in human glioma patients’ cells. Oncotarget 2015, 6, 15464–15481. [Google Scholar] [CrossRef] [PubMed]
- Dunn, S.L.; Wilkinson, J.M.; Crawford, A.; Bunning, R.A.D.; Le Maitre, C.L. Expression of Cannabinoid Receptors in Human Osteoarthritic Cartilage: Implications for Future Therapies. Cannabis Cannabinoid Res. 2016, 1, 3–15. [Google Scholar] [CrossRef]
- Pal, D.; Tyagi, A.; Chandrasekaran, B.; Alattasi, H.; Ankem, M.K.; Sharma, A.K.; Damodaran, C. Suppression of Notch1 and AKT mediated epithelial to mesenchymal transition by Verrucarin J in metastatic colon cancer. Cell. Death Dis. 2018, 9, 798. [Google Scholar] [CrossRef] [PubMed]
- Alpini, G.; De Morrow, S. Changes in the endocannabinoid system may give insight into new and effective treatments for cancer. Vitam. Horm. 2009, 81, 469–485. [Google Scholar]
- Dalton, G.D.; Bass, C.E.; Van Horn, C.G.; Howlett, A.C. Signal transduction via cannabinoid receptors. CNS Neur. Disord. Drug. Targets 2009, 8, 422–431. [Google Scholar] [CrossRef]
- Maccarone, M.; Finazzi-Agrò, A. The endocannabinoid system, anandamide and the regulation of mammalian cell apoptosis. Cell. Death Differ. 2003, 10, 946–955. [Google Scholar] [CrossRef]
- Chung, S.C.; Hammarsten, P.; Josefsson, A.; Stattin, P.; Granfors, T.; Egevad, L.; Mancini, G.; Lutz, B.; Bergh, A.; Fowler, C.J. A high cannabinoid CB1 receptor immunoreactivity is associated with disease severity and outcome in prostate cancer. Eur. J. Cancer 2009, 45, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Salazar, M.; Carracedo, A.; Salanueva, I.J.; Hernández-Tiedra, S.; Lorente, M.; Egia, A.; Vázquez, P.; Blázquez, C.; Torres, S.; García, S.; et al. Cannabinoid action induces autophagy-mediated cell death through stimulation of ER stress in human glioma cells. J. Clin. Invest. 2009, 119, 1359–1372. [Google Scholar] [CrossRef]
- Shrivastava, A.; Kuzontkoski, P.M.; Groopman, J.E.; Prasad, A. Cannabidiol induces programmed cell death in breast cancer cells by coordinating the cross-talk between apoptosis and autophagy. Mol. Cancer Ther. 2011, 10, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Vara, D.; Salazar, M.; Olea-Herrero, N.; Guzmán, M.; Velasco, G.; Díaz-Laviada, I. Anti-tumoral action of cannabinoids on hepatocellular carcinoma: Role of AMPK-dependent activation of autophagy. Cell. Death Differ. 2011, 18, 1099–1111. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.K.; Kang, W.K.; Park, J.M.; Ahn, H.J.; Kim, S.W.; TaekOh, S.; Choi, K.Y. Expression of the cannabinoid type I receptor and prognosis following surgery in colorectal cancer. Oncol. Lett. 2013, 5, 870–876. [Google Scholar] [CrossRef] [PubMed]
| CRC Patients | ||
|---|---|---|
| Variables | No Metastases (n = 29) | Metastases (n = 30) |
| Age | 69.7 ± 15.2 | 68.3 ± 11 |
| Sex | ||
| Male | 16 | 20 |
| Female | 13 | 10 |
| Tumor Side a | ||
| Right | 11 | 10 |
| Left | 18 | 20 |
| Tumor Stage b | ||
| Stage I | 5 | 2 |
| Stage II | 20 | 0 |
| Stage III | 3 | 18 |
| Stage IV | 1 | 10 |
| Histological Grading | ||
| Well-differentiated (G1) | 3 | 0 |
| Moderately-differentiated (G2) | 16 | 16 |
| Poorly-differentiated (G3) | 10 | 14 |
| Metastases Site | ||
| Liver | 0 | 12 |
| Visceral lymphnodes | 0 | 16 |
| Bone | 0 | 1 |
| Lung metastases | 0 | 1 |
| Gene | Primer |
|---|---|
| CB1 receptor | |
| Forward | 5′-GGAGAACATCCAGTGTGGGG-3′ |
| Reverse | 5′-CATTGGGGCTGTCTTTACGG-3′ |
| caspase-3 | |
| Forward | 5′-TGAGGCGGTTGTAGAAGAGTTT-3′ |
| Reverse | 5′-TTAACGAAAACCAGAGCGCC-3′ |
| MAPK p38α | |
| Forward | 5′-ACTCAGATGCCGAAGATGAAC-3′ |
| Reverse | 5′-GTGCTCAGGACTCCATCTCT-3′ |
| bax | |
| Forward | 5′-CAGGATGCGTCCACCAAGAA-3′ |
| Reverse | 5′-GCTCCCGGAGGAAGTCCAAT-3′ |
| bcl2 | |
| Forward | 5′-GTGGAGGAGCTCTTCAGGGA-3′ |
| Reverse | 5′-AGGCACCCAGGGTGATG-CAA-3′ |
| β-actin | |
| Forward | 5′-AAAGACCTGTACGCCAACACAGTGCTGTCTGG-3′ |
| Reverse | 5′-CGTCATACTCCTGCTTGCTGATCCACATCTGC-3′ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tutino, V.; Caruso, M.G.; De Nunzio, V.; Lorusso, D.; Veronese, N.; Gigante, I.; Notarnicola, M.; Giannelli, G. Down-Regulation of Cannabinoid Type 1 (CB1) Receptor and its Downstream Signaling Pathways in Metastatic Colorectal Cancer. Cancers 2019, 11, 708. https://doi.org/10.3390/cancers11050708
Tutino V, Caruso MG, De Nunzio V, Lorusso D, Veronese N, Gigante I, Notarnicola M, Giannelli G. Down-Regulation of Cannabinoid Type 1 (CB1) Receptor and its Downstream Signaling Pathways in Metastatic Colorectal Cancer. Cancers. 2019; 11(5):708. https://doi.org/10.3390/cancers11050708
Chicago/Turabian StyleTutino, Valeria, Maria Gabriella Caruso, Valentina De Nunzio, Dionigi Lorusso, Nicola Veronese, Isabella Gigante, Maria Notarnicola, and Gianluigi Giannelli. 2019. "Down-Regulation of Cannabinoid Type 1 (CB1) Receptor and its Downstream Signaling Pathways in Metastatic Colorectal Cancer" Cancers 11, no. 5: 708. https://doi.org/10.3390/cancers11050708
APA StyleTutino, V., Caruso, M. G., De Nunzio, V., Lorusso, D., Veronese, N., Gigante, I., Notarnicola, M., & Giannelli, G. (2019). Down-Regulation of Cannabinoid Type 1 (CB1) Receptor and its Downstream Signaling Pathways in Metastatic Colorectal Cancer. Cancers, 11(5), 708. https://doi.org/10.3390/cancers11050708







