New Insights into Molecular Oncogenesis and Therapy of Uveal Melanoma
Abstract
1. Introduction
2. Uveal Melanoma Genetics
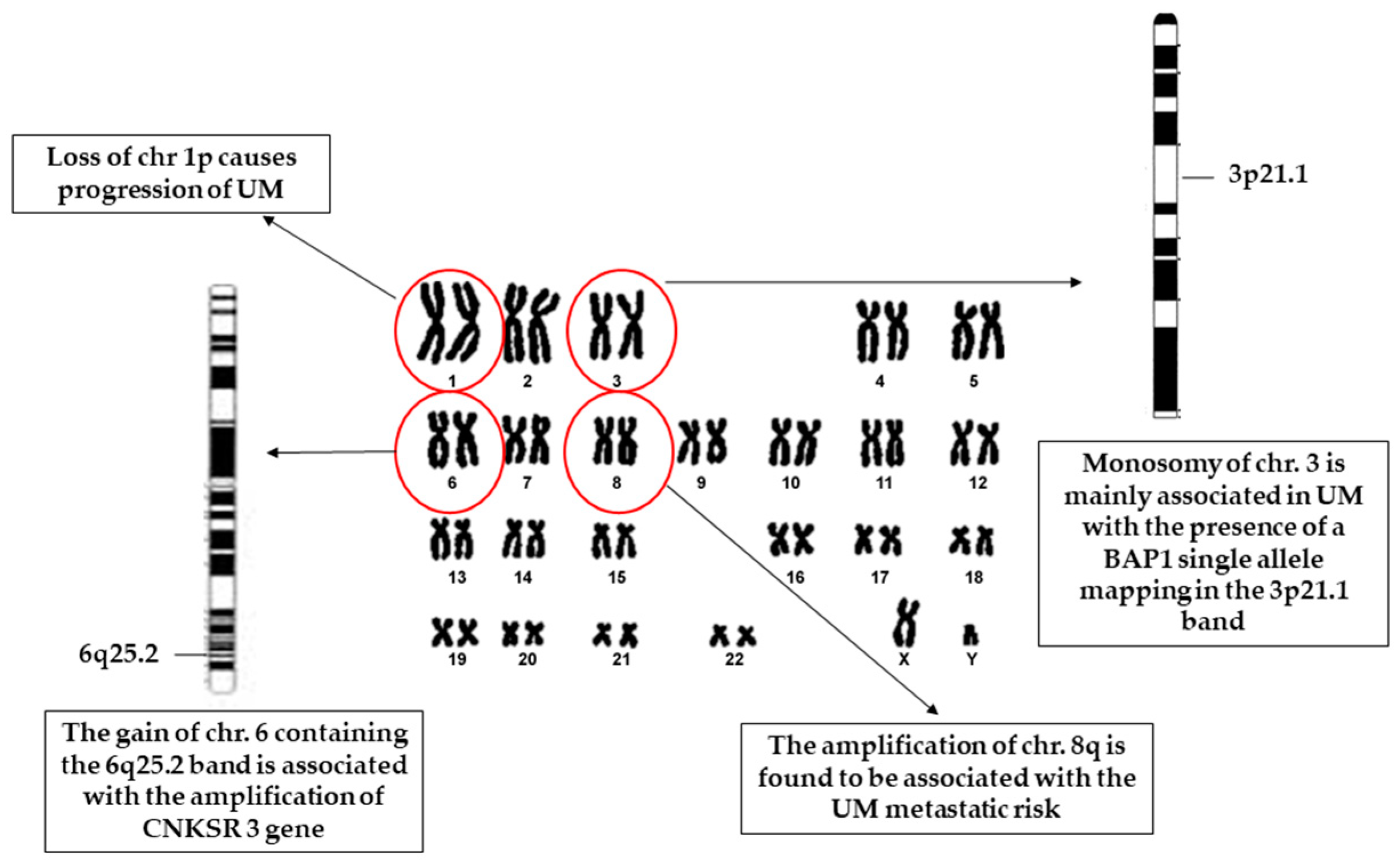
2.1. Cytogenetics
2.2. Gene Mutations
2.2.1. GNAQ/GNA11
2.2.2. CYSLTR2
2.2.3. PLCB4
2.2.4. BAP1
2.2.5. SF3B1
2.2.6. EIF1AX
2.2.7. TERT
3. Genetic Prognostic Tests
4. UM Association with Small DNA Tumour Polyomaviruses BKPyV and SV40
5. Current Therapeutic Options for Primary Tumours
6. Current, Innovative Therapeutic Options for Metastatic Tumours
6.1. Liver-Directed Approach
6.2. Chemotherapy
6.3. Targeted Therapy
6.4. Immunotherapy
6.4.1. Checkpoint Inhibition
6.4.2. Anti-CTLA-4 Therapy
6.5. Anti-PD-1 Therapy
6.6. Combined CTLA-4 and PD-1 Blockade
6.7. Innovative Immune-Based Approaches
6.8. Epigenetic Approaches
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Singh, A.D.; Turell, M.E.; Topham, A.K. Uveal melanoma: Trends in incidence, treatment, and survival. Ophthalmology 2011, 118, 1881–1885. [Google Scholar] [CrossRef]
- Virgili, G.; Gatta, G.; Ciccolallo, L.; Capocaccia, R.; Biggeri, A.; Crocetti, E.; Lutz, J.M.; Paci, E. Survival in patients with uveal melanoma in Europe. Arch. Ophthalmol. 2008, 128, 1413–1418. [Google Scholar]
- Singh, A.D.; Topham, A. Survival rates with uveal melanoma in the United States: 1973–1997. Ophthalmology 2003, 110, 962–965. [Google Scholar] [CrossRef]
- Finger, P.T.; Kurli, M.; Reddy, S.; Tena, L.B.; Pavlick, A.C. Whole body PET/CT for initial staging of choroidal melanoma. Br. J. Ophthalmol. 2005, 89, 1270–1274. [Google Scholar] [CrossRef]
- Kujala, E.; Mäkitie, T.; Kivelä, T. Very Long-Term Prognosis of Patients with Malignant Uveal Melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4651–4659. [Google Scholar] [CrossRef]
- Diener-West, M.; Reynolds, S.M.; Agugliaro, D.J.; Caldwell, R.; Cumming, K.; Earle, J.D.; Green, D.L.; Hawkins, B.S.; Hayman, J.; Jaiyesimi, I.; et al. Screening for Metastasis From Choroidal Melanoma: The Collaborative Ocular Melanoma Study Group Report 23. J. Clin. Oncol. 2004, 22, 2438–2444. [Google Scholar] [CrossRef]
- Shields, J.A.; Shields, C.L. Management of Posterior Uveal Melanoma: Past, Present, and Future. Ophthalmology 2015, 122, 414–428. [Google Scholar] [CrossRef]
- Eskelin, S.; Pyrhönen, S.; Summanen, P.; Hahka-Kemppinen, M.; Kivelä, T. Tumor doubling times in metastatic malignant melanoma of the uvea: Tumor progression before and after treatment. Ophthalmology 2000, 107, 1443–1449. [Google Scholar] [CrossRef]
- Buder, K.; Gesierich, A.; Gelbrich, G.; Goebeler, M. Systemic treatment of metastatic uveal melanoma: Review of literature and future perspectives. Cancer Med. 2013, 2, 674–686. [Google Scholar] [CrossRef]
- Berus, T.; Halon, A.; Markiewicz, A.; Orlowska-Heitzman, J.; Romanowska-Dixon, B.; Donizy, P. Clinical, Histopathological and Cytogenetic Prognosticators in Uveal Melanoma—A Comprehensive Review. Anticancer Res. 2017, 37, 6541–6549. [Google Scholar] [PubMed]
- Sisley, K.; Rennie, I.G.; Cottam, D.W.; Potter, A.M.; Potter, C.W.; Rees, R.C. Cytogenetic findings in six posterior uveal melanomas: Involvement of chromosomes 3, 6, and 8. Genes Chromosomes Cancer 1990, 2, 205–209. [Google Scholar] [CrossRef]
- Prescher, G.; Bornfeld, N.; Becher, R. Nonrandom chromosomal abnormalities in primary uveal melanoma. J. Natl. Cancer Inst. 1990, 82, 1765–1769. [Google Scholar] [CrossRef] [PubMed]
- Prescher, G.; Bornfeld, N.; Hirche, H.; Horsthemke, B.; Jöckel, K.H.; Becher, R. Prognostic implications of monosomy 3 in uveal melanoma. Lancet 1996, 347, 1222–1225. [Google Scholar]
- Horsman, D.E.; Sroka, H.; Rootman, J.; White, V.A. Monosomy 3 and isochromosome 8q in a uveal melanoma. Cancer Genet. Cytogenet. 1990, 45, 249–253. [Google Scholar] [CrossRef]
- Scholes, A.G.M.; Damato, B.E.; Nunn, J.; Hiscott, P.; Grierson, I.; Field, J.K. Monosomy 3 in uveal melanoma: Correlation with clinical and histologic predictors of survival. Invest. Ophthalmol. Vis. Sci. 2003, 44, 1008–1011. [Google Scholar] [CrossRef] [PubMed]
- Damato, B.; Duke, C.; Coupland, S.E.; Hiscott, P.; Smith, P.A.; Campbell, I.; Douglas, A.; Howard, P. Cytogenetics of Uveal Melanoma A 7-Year Clinical Experience. Ophthalmology 2007, 14, 1925–1931. [Google Scholar] [CrossRef]
- Damato, B.; Coupland, S.E. A reappraisal of the significance of largest basal diameter of posterior uveal melanoma. Eye 2009, 23, 2152–2162. [Google Scholar] [CrossRef] [PubMed]
- Shields, C.L.; Furuta, M.; Thangappan, A.; Nagori, S.; Mashayekhi, A.; Lally, D.R.; Kelly, C.C.; Rudich, D.S.; Nagori, A.V.; Wakade, O.A.; et al. Metastasis of Uveal Melanoma Millimeter-by-Millimeter in 8033 Consecutive Eyes. Arch. Ophthalmol. 2009, 127, 989. [Google Scholar] [CrossRef]
- Singh, A.D.; Tubbs, R.; Biscotti, C.; Schoenfield, L.; Trizzoi, P. Chromosomal 3 and 8 status within hepatic metastasis of uveal melanoma. Arch. Pathol. Lab. Med. 2009, 133, 1223–1227. [Google Scholar]
- Sisley, K.; Rennie, I.G.; Andrew Parsons, M.; Jacques, R.; Hammond, D.W.; Bell, S.M.; Potter, A.M.; Rees, R.C. Abnormalities of chromosomes 3 and 8 in posterior uveal melanoma correlate with prognosis. Genes Chromosomes. Cancer 1997, 19, 22–28. [Google Scholar] [CrossRef]
- Damato, B.; Dopierala, J.A.; Coupland, S.E. Genotypic profiling of 452 choroidal melanomas with multiplex ligation-dependent probe amplification. Clin. Cancer Res. 2010, 16, 6083–6092. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, J.G.M.; Koopmans, A.E.; Vaarwater, J.; de Rooi, J.J.; Paridaens, D.; Naus, N.C.; de Klein, A.; Verdijk, R.M.; Kiliç, E. The prognostic value of extraocular extension in relation to monosomy 3 and gain of chromosome 8q in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2014, 55, 1284–1291. [Google Scholar] [CrossRef]
- Harbour, J.W.; Onken, M.D.; Roberson, E.D.O.; Duan, S.; Cao, L.; Worley, L.A.; Council, M.L.; Matatall, K.A.; Helms, C.; Bowcock, A.M. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 2010, 330, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Häusler, T.; Stang, A.; Anastassiou, G.; Jöckel, K.H.; Mrzyk, S.; Horsthemke, B.; Lohmann, D.R.; Zeschnigk, M. Loss of heterozygosity of 1p in uveal melanomas with monosomy 3. Int. J. Cancer 2005, 116, 909–913. [Google Scholar] [CrossRef]
- Damato, B.; Dopierala, J.; Klaasen, A.; van Dijk, M.; Sibbring, J.; Coupland, S.E. Multiplex ligation-dependent probe amplification of uveal melanoma: Correlation with metastatic death. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3048–3055. [Google Scholar] [CrossRef]
- White, V.A.; Chambers, J.D.; Courtright, P.D.; Chang, W.Y.; Horsman, D.E. Correlation of cytogenetic abnormalities with the outcome of patients with uveal melanoma. Cancer 1998, 83, 354–359. [Google Scholar] [CrossRef]
- Landreville, S.; Agapova, O.A.; Hartbour, J.W. Emerging insights into the molecular pathogenesis of uveal melanoma. Futur. Oncol. 2008, 4, 629–636. [Google Scholar] [CrossRef]
- Lake, S.L.; Damato, B.E.; Kalirai, H.; Dodson, A.R.; Taktak, A.F.G.; Lloyd, B.H.; Coupland, S.E. Single nucleotide polymorphism array analysis of uveal melanomas reveals that amplification of CNKSR3 is correlated with improved patient survival. Am. J. Pathol. 2013, 182, 678–687. [Google Scholar] [CrossRef]
- Amaro, A.; Gangemi, R.; Piaggio, F.; Angelini, G.; Barisione, G.; Ferrini, S.; Pfeffer, U. The biology of uveal melanoma. Cancer Metastasis Rev. 2017, 36, 109–140. [Google Scholar] [CrossRef]
- Park, J.J.; Diefenbach, R.J.; Joshua, A.M.; Kefford, R.F.; Carlino, M.S.; Rizos, H. Oncogenic signaling in uveal melanoma. Pigment Cell Melanoma Res. 2018, 31, 661–672. [Google Scholar] [CrossRef]
- Van Raamsdonk, C.D.; Griewank, K.G.; Crosby, M.B.; Garrido, M.C.; Vemula, S.; Wiesner, T.; Obenauf, A.C.; Wackernagel, W.; Green, G.; Bouvier, N.; et al. Mutations in GNA11 in Uveal Melanoma. N. Engl. J. Med. 2010, 363, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Dono, M.; Angelini, G.; Cecconi, M.; Amaro, A.; Esposito, A.I.; Mirisola, V.; Maric, I.; Lanza, F.; Nasciuti, F.; Viaggi, S.; et al. Mutation frequencies of GNAQ, GNA11, BAP1, SF3B1, EIF1AX and TERT in uveal melanoma: Detection of an activating mutation in the TERT gene promoter in a single case of uveal melanoma. Br. J. Cancer 2014, 110, 1058–1065. [Google Scholar] [CrossRef]
- Field, M.G.; Harbour, J.W. Recent developments in prognostic and predictive testing in uveal melanoma. Curr. Opin. Ophthalmol. 2014, 25, 234–239. [Google Scholar] [CrossRef]
- Tarlan, B.; Kıratlı, H. Uveal melanoma: Current trends in diagnosis and management. Turk Oftalmoloiji Derg. 2016, 46, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Shoushtari, A.N.; Carvajal, R.D. GNAQ and GNA11 mutations in uveal melanoma. Melanoma Res. 2014, 24, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Chen, Q.; Gutkind, J.S. Oncotargeting G proteins: The Hippo in the room. Oncotarget 2014, 5, 10997–10999. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.X.; Luo, J.; Mo, J.S.; Liu, G.; Kim, Y.C.; Meng, Z.; Zhao, L.; Peyman, G.; Ouyang, H.; Jiang, W.; et al. Mutant Gq/11 promote uveal melanoma tumorigenesis by activating YAP. Cancer Cell 2014, 25, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.H.; Levin, A.M.; Abramson, D.H. Update on Ophthalmic Oncology 2014: Retinoblastoma and Uveal Melanoma. Asia-Pacific J. Ophthalmol. 2016, 5, 368–382. [Google Scholar] [CrossRef]
- Moore, A.R.; Ceraudo, E.; Sher, J.J.; Guan, Y.; Shoushtari, A.N.; Chang, M.T.; Zhang, J.Q.; Walczak, E.G.; Kazmi, M.A.; Taylor, B.S.; et al. Recurrent activating mutations of G-protein-coupled receptor CYSLTR2 in uveal melanoma. Nat. Genet. 2016, 48, 675–680. [Google Scholar] [CrossRef]
- Uveal Melanomas Harbor Recurrent Activating Mutations in CYSLTR2. Cancer Discov. 2016, 6, 573. [CrossRef]
- Johansson, P.; Aoude, L.G.; Wadt, K.; Glasson, W.J.; Warrier, S.K.; Hewitt, A.W.; Kiilgaard, J.F.; Heegaard, S.; Isaacs, T.; Franchina, M.; et al. Deep sequencing of uveal melanoma identifies a recurrent mutation in PLCB4. Oncotarget 2016, 7, 4624–4631. [Google Scholar] [CrossRef] [PubMed]
- Ventii, K.H.; Devi, N.S.; Friedrich, K.L.; Chernova, T.A.; Tighiouart, M.; Van Meir, E.G.; Wilkinson, K.D. BRCA1-associated protein-1 is a tumor suppressor that requires deubiquitinating activity and nuclear localization. Cancer Res. 2008, 68, 6953–6962. [Google Scholar] [CrossRef]
- Pan, H.; Jia, R.; Zhang, L.; Xu, S.; Wu, Q.; Song, X.; Zhang, H.; Ge, S.; Leon Xu, X.; Fan, X. BAP1 regulates cell cycle progression through E2F1 target genes and mediates transcriptional silencing via H2A monoubiquitination in uveal melanoma cells. Int. J. Biochem. Cell Biol. 2015, 60, 176–184. [Google Scholar] [CrossRef]
- Hanpude, P.; Bhattacharya, S.; Kumar Singh, A.; Kanti Maiti, T. Ubiquitin recognition of BAP1: Understanding its enzymatic function. Biosci. Rep. 2017, 37, BSR20171099. [Google Scholar] [CrossRef]
- Bononi, A.; Giorgi, C.; Patergnani, S.; Larson, D.; Verbruggen, K.; Tanji, M.; Pellegrini, L.; Signorato, V.; Olivetto, F.; Pastorino, S.; et al. BAP1 regulates IP3R3-mediated Ca2+ flux to mitochondria suppressing cell transformation. Nature 2017, 546, 549–553. [Google Scholar] [CrossRef]
- Hebert, L.; Bellanger, D.; Guillas, C.; Campagne, A.; Dingli, F.; Loew, D.; Fievet, A.; Jacquemin, V.; Popova, T.; Jean, D.; et al. Modulating BAP1 expression affects ROS homeostasis, cell motility and mitochondrial function. Oncotarget 2017, 8, 72513–72527. [Google Scholar] [CrossRef]
- Matatall, K.A.; Agapova, O.A.; Onken, M.D.; Worley, L.A.; Bowcock, A.M.; Harbour, J.W. BAP1 deficiency causes loss of melanocytic cell identity in uveal melanoma. BMC Cancer 2013, 13, 371. [Google Scholar] [CrossRef]
- Carbone, M.; Yang, H.; Pass, H.I.; Krausz, T.; Testa, J.R.; Gaudino, G. BAP1 and cancer. Nat. Rev. Cancer 2013, 13, 153–159. [Google Scholar] [CrossRef]
- Murali, R.; Wiesner, T.; Scolyer, R.A. Tumours associated with BAP1 mutations. Pathology 2013, 45, 116–126. [Google Scholar] [CrossRef]
- Onken, M.D.; Li, J.; Cooper, J.A. Uveal melanoma cells utilize a novel route for transendothelial migration. PLoS ONE 2014, 9, e115472. [Google Scholar] [CrossRef] [PubMed]
- Rai, K.; Pilarski, R.; Boru, G.; Rehman, M.; Saqr, A.H.; Massengill, J.B.; Singh, A.; Marino, M.J.; Davidorf, F.H.; Cebulla, C.M.; et al. Germline BAP1 alterations in familial uveal melanoma. Genes. Chromosomes Cancer 2017, 56, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Cebulla, C.M.; Binkley, E.M.; Pilarski, R.; Massengill, J.B.; Rai, K.; Liebner, D.A.; Marino, M.J.; Singh, A.D.; Abdel-Rahman, M.H. Analysis of BAP1 Germline Gene Mutation in Young Uveal Melanoma Patients. Ophthalmic Genet. 2015, 36, 126–131. [Google Scholar] [CrossRef]
- Van De Nes, J.A.P.; Nelles, J.; Kreis, S.; Metz, C.H.D.; Hager, T.; Lohmann, D.R.; Zeschnigk, M. Comparing the prognostic value of BAP1 mutation pattern, chromosome 3 status, and BAP1 immunohistochemistry in uveal melanoma. Am. J. Surg. Pathol. 2016, 40, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Alsafadi, S.; Houy, A.; Battistella, A.; Popova, T.; Wassef, M.; Henry, E.; Tirode, F.; Constantinou, A.; Piperno-Neumann, S.; Roman-Roman, S.; et al. Cancer-associated SF3B1 mutations affect alternative splicing by promoting alternative branchpoint usage. Nat. Commun. 2016, 7, 10615. [Google Scholar] [CrossRef]
- Harbour, J.W.; Roberson, E.D.O.; Anbunathan, H.; Onken, M.D.; Worley, L.A.; Bowcock, A.M. Recurrent mutations at codon 625 of the splicing factor SF3B1 in uveal melanoma. Nat. Genet. 2013, 45, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Maßhöfer, L.; Temming, P.; Rahmann, S.; Metz, C.; Bornfeld, N.; van de Nes, J.; Klein-Hitpass, L.; Hinnebusch, A.G.; Horsthemke, B.; et al. Exome sequencing identifies recurrent somatic mutations in EIF1AX and SF3B1 in uveal melanoma with disomy 3. Nat. Genet. 2013, 45, 933–936. [Google Scholar] [CrossRef] [PubMed]
- Yavuzyigitoglu, S.; Koopmans, A.E.; Verdijk, R.M.; Vaarwater, J.; Eussen, B.; Van Bodegom, A.; Paridaens, D.; Kiliç, E.; De Klein, A. Uveal Melanomas with SF3B1 Mutations: A Distinct Subclass Associated with Late-Onset Metastases. Ophthalmology 2016, 123, 1118–1128. [Google Scholar] [CrossRef]
- Johnson, C.P.; Kim, I.K.; Esmaeli, B.; Amin-Mansour, A.; Treacy, D.J.; Carter, S.L.; Hodis, E.; Wagle, N.; Seepo, S.; Yu, X.; et al. Systematic genomic and translational efficiency studies of uveal melanoma. PLoS ONE 2017, 12, e0178189. [Google Scholar] [CrossRef]
- Ewens, K.G.; Kanetsky, P.A.; Richards-Yutz, J.; Purrazzella, J.; Shields, C.L.; Ganguly, T.; Ganguly, A. Chromosome 3 status combined with BAP1 and EIF1AX mutation profiles are associated with metastasis in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2014, 55, 5160–5167. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, A.E.; Ober, K.; Dubbink, H.J.; Paridaens, D.; Naus, N.C.; Belunek, S.; Krist, B.; Post, E.; Zwarthoff, E.C.; De Klein, A.; et al. Prevalence and implications of TERT promoter mutation in uveal and conjunctival melanoma and in benign and premalignant conjunctival melanocytic lesions. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6024–6030. [Google Scholar] [CrossRef]
- Prescher, G.; Bornfeld, N.; Horsthemke, B.; Becher, R. Chromosomal aberrations defining uveal melanoma of poor prognosis. Lancet 1992, 339, 691–692. [Google Scholar] [CrossRef]
- Seider, M.I.; Mruthyunjaya, P. Molecular prognostics for uveal melanoma. Retina 2018, 38, 211–219. [Google Scholar] [CrossRef]
- Young, T.A.; Rao, N.P.; Glasgow, B.J.; Moral, J.N.; Straatsma, B.R. Fluorescent in situ hybridization for monosomy 3 via 30-Gauge Fine-Needle aspiration biopsy of choroidal melanoma in vivo. Ophthalmology 2007, 114, 142–146. [Google Scholar] [CrossRef]
- McCannel, T.A.; Chang, M.Y.; Burgess, B.L. Multi-year follow-up of fine-needle aspiration biopsy in choroidal melanoma. Ophthalmology 2012, 119, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Aronow, M.; Sun, Y.; Saunthararajah, Y.; Biscotti, C.; Tubbs, R.; Triozzi, P.; Singh, A.D. Monosomy 3 by FISH in Uveal Melanoma: Variability in Techniques and Results. Surv. Ophthalmol. 2012, 57, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Mensink, H.W.; Kiliç, E.; Vaarwater, J.; Douben, H.; Paridaens, D.; de Klein, A. Molecular cytogenetic analysis of archival uveal melanoma with known clinical outcome. Cancer Genet. Cytogenet. 2008, 181, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Lake, S.L.; Kalirai, H.; Dopierala, J.; Damato, B.E.; Coupland, S.E. Comparison of formalin-fixed and snap-frozen samples analyzed by multiplex ligation-dependent probe amplification for prognostic testing in uveal melanoma. Invest. Ophthalmol. Vis. Sci. 2012, 53, 2647–2652. [Google Scholar] [CrossRef]
- Vaarwater, J.; van den Bosch, T.; Mensink, H.W.; van Kempen, C.; Verdijk, R.M.; Naus, N.C.; Paridaens, D.; Brüggenwirth, H.T.; Kiliç, E.; de Klein, A. Multiplex ligation-dependent probe amplification equals fluorescence in-situ hybridization for the identification of patients at risk for metastatic disease in uveal melanoma. Melanoma Res. 2012, 22, 30–37. [Google Scholar] [CrossRef]
- Coupland, S.E.; Lake, S.L.; Damato, B. Molecular pathology of uveal melanoma. In Clinical Ophthalmic Oncology: Uveal Tumors; Springer: Berlin, Germany, 2014; ISBN 9783642542558. [Google Scholar]
- Thomas, S.; Pütter, C.; Weber, S.; Bornfeld, N.; Lohmann, D.R.; Zeschnigk, M. Prognostic significance of chromosome 3 alterations determined by microsatellite analysis in uveal melanoma: A long-term follow-up study. Br. J. Cancer 2012, 106, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Tschentscher, F.; Prescher, G.; Zeschnigk, M.; Horsthemke, B.; Lohmann, D.R. Identification of chromosomes 3, 6, and 8 aberrations in uveal melanoma by microsatellite analysis in comparison to comparative genomic hybridization. Cancer Genet. Cytogenet. 2000, 122, 13–17. [Google Scholar] [CrossRef]
- Shields, C.L.; Ganguly, A.; Materin, M.A.; Teixeira, L.; Mashayekhi, A.; Swanson, L.A.; Marr, B.P.; Shields, J.A. Chromosome 3 analysis of uveal melanoma using fine-needle aspiration biopsy at the time of plaque radiotherapy in 140 consecutive cases. Trans. Am. Ophthalmol. Soc. 2007, 105, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Onken, M.D.; Worley, L.A.; Ehlers, J.P.; Harbour, J.W. Gene expression profiling in uveal melanoma reveals two molecular classes and predicts metastatic death. Cancer Res. 2004, 64, 7205–7209. [Google Scholar] [CrossRef] [PubMed]
- Onken, M.D.; Worley, L.A.; Harbour, J.W. A metastasis modifier locus on human chromosome 8p in uveal melanoma identified by integrative genomic analysis. Clin. Cancer Res. 2008, 14, 3737–3745. [Google Scholar] [CrossRef]
- Field, M.G.; Decatur, C.L.; Kurtenbach, S.; Gezgin, G.; Van Der Velden, P.A.; Jager, M.J.; Kozak, K.N.; Harbour, J.W. PRAME as an independent biomarker for metastasis in uveal melanoma. Clin. Cancer Res. 2016, 22, 1234–1242. [Google Scholar] [CrossRef]
- Field, M.G.; Durante, M.A.; Decatur, C.L.; Tarlan, B.; Oelschlager, K.M.; Stone, J.F.; Kuznetsov, J.; Bowcock, A.M.; Kurtenbach, S.; Harbour, J.W. Epigenetic reprogramming and aberrant expression of PRAME are associated with increased metastatic risk in Class 1 and Class 2 uveal melanomas. Oncotarget 2016, 7, 59209–59219. [Google Scholar] [CrossRef] [PubMed]
- Dogrusöz, M.; Jager, M.J. Genetic prognostication in uveal melanoma. Acta Ophthalmol. 2018, 96, 331–347. [Google Scholar] [CrossRef]
- Bononi, I.; Perri, P.; Begnardi, A.; Martini, A.; Mazzoni, E.; Bosi, S.; Pietrobon, S.; Sebastiani, A.; Tognon, M.; Martini, F. Antibodies reacting with Simian Virus 40 capsid protein mimotopes in serum samples from patients affected by uveal melanoma. J. Hematol. Oncol. 2014, 7, 38. [Google Scholar] [CrossRef]
- Jovanovic, P.; Mihajlovic, M.; Djordjevic-Jocic, J.; Vlajkovic, S.; Cekic, S.; Stefanovic, V. Ocular melanoma: An overview of the current status. Int. J. Clin. Exp. Pathol. 2013, 6, 1230–1244. [Google Scholar] [PubMed]
- Martini, F.; Corallini, A.; Balatti, V.; Sabbioni, S.; Pancaldi, C.; Tognon, M. Simian virus 40 in humans. Infect. Agent. Cancer 2007, 2, 13. [Google Scholar] [CrossRef] [PubMed]
- Tognon, M.; Corallini, A.; Martini, F.; Negrini, M.; Barbanti-Brodano, G. Oncogenic transformation by BK virus and association with human tumors. Oncogene 2003, 22, 5192–5200. [Google Scholar] [CrossRef]
- Barbanti-Brodano, G.; Sabbioni, S.; Martini, F.; Negrini, M.; Corallini, A.; Tognon, M. BK Virus, JC Virus and Simian Virus 40 Infection in Humans, and Association with Human Tumors. In Polyomaviruses and Human Diseases; Springer: New York, NY, USA, 2006; Volume 577, pp. 319–341. [Google Scholar]
- Cardoso, K.M.; Diaz, N.C.; Guimarães, M.A.A.M.; Zalis, M.G.; Delbue, S.; Ferrante, P.; Varella, R.B. Genetic and structural analysis of polyomavirus BK T-antigens reveal a higher density of mutations at inter-domain and hexamerization regions, regardless the status of infection. J. Med. Virol. 2015, 87, 1418–1426. [Google Scholar] [CrossRef]
- Verhalen, B.; Justice, J.L.; Imperiale, M.J.; Jiang, M. Viral DNA Replication-Dependent DNA Damage Response Activation during BK Polyomavirus Infection. J. Virol. 2015, 89, 5032–5039. [Google Scholar] [CrossRef] [PubMed]
- Pietrobon, S.; Bononi, I.; Mazzoni, E.; Lotito, F.; Manfrini, M.; Puozzo, A.; Destro, F.; Guerra, G.; Nocini, P.F.; Martini, F.; et al. Specific IgG Antibodies React to Mimotopes of BK Polyomavirus, a Small DNA Tumor Virus, in Healthy Adult Sera. Front. Immunol. 2017, 8, 236. [Google Scholar] [CrossRef] [PubMed]
- Pietrobon, S.; Bononi, I.; Lotito, F.; Perri, P.; Violanti, S.; Mazzoni, E.; Martini, F.; Tognon, M.G. Specific Detection of Serum Antibodies against BKPyV, A Small DNA Tumour Virus, in Patients Affected by Choroidal Nevi. Front. Microbiol. 2017, 8, 2059. [Google Scholar] [CrossRef] [PubMed]
- Bononi, I.; Mazzoni, E.; Pietrobon, S.; Torreggiani, E.; Rossini, M.; Violanti, S.; Perri, P.; Tognon, M.; Martini, F. High prevalence of serum IgG antibodies reacting to specific mimotopes of BK polyomavirus, a human oncogenic polyomavirus, in patients affected by uveal melanoma. J. Cell. Physiol. 2018, 233, 9052–9059. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, L.E.; McLean, I.W.; Foster, W.D. Does enucleation of the eye containing a malignant melanoma prevent or accelerate the dissemination of tumour cells? Br. J. Ophthalmol. 1978, 62, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Jerry, A.; Shields, C.L.S. Intraocular Tumors: An Atlas and Textbook, 2nd ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2008; ISBN 9781496321343. [Google Scholar]
- Kaliki, S.; Shields, C.L. Uveal melanoma: Relatively rare but deadly cancer. Eye 2017, 31, 241–257. [Google Scholar] [CrossRef]
- Perri, P.; Fiorica, F.; D’Angelo, S.; Lamberti, G.; Parmeggiani, F.; Martini, A.; Carpenteri, F.; Colosimo, C.; Micucci, M.; Perazzini, L.; et al. Ruthenium-106 eye plaque brachytherapy in the conservative treatment of uveal melanoma: A mono-institutional experience. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 1919–1924. [Google Scholar] [PubMed]
- Chattopadhyay, C.; Kim, D.W.; Gombos, D.S.; Oba, J.; Qin, Y.; Williams, M.D.; Esmaeli, B.; Grimm, E.A.; Wargo, J.A.; Woodman, S.E.; et al. Uveal melanoma: From diagnosis to treatment and the science in between. Cancer 2016, 122, 2299–2312. [Google Scholar] [CrossRef]
- Seregard, S.; Damato, B.E. Uveal malignant melanoma: Management options for brachytherapy. In Clinical Ophthalmic Oncology; Damato, B.E., Singh, A.D., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 181–196.2. [Google Scholar]
- American Brachytherapy Society—Ophthalmic Oncology Task Force. The American Brachytherapy Society consensus guidelines for plaque brachytherapy of uveal melanoma and retinoblastoma. Brachytherapy 2014, 13, 1–14. [Google Scholar] [CrossRef]
- Stannard, C.; Sauerwein, W.; Maree, G.; Lecuona, K. Radiotherapy for ocular tumours. Eye 2013, 27, 119–127. [Google Scholar] [CrossRef]
- Takiar, V.; Voong, K.R.; Gombos, D.S.; Mourtada, F.; Rechner, L.A.; Lawyer, A.A.; Morrison, W.H.; Garden, A.S.; Beadle, B.M. A choice of radionuclide: Comparative outcomes and toxicity of ruthenium-106 and iodine-125 in the definitive treatment of uveal melanoma. Pract. Radiat. Oncol. 2015, 5, e169–e176. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Ocular Melanoma Study Group. The COMS Randomized Trial of Iodine 125 Brachytherapy for Choroidal Melanoma. Arch. Ophthalmol. 2006, 124, 1684. [Google Scholar]
- Margo, C.E. The Collaborative Ocular Melanoma Study: An Overview. Cancer Control 2004, 11, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Fine, S.L.; Hawkins, B.S. The Investigators’ Perspective on the Collaborative Ocular Melanoma Study. Arch. Ophthalmol. 2007, 125, 968. [Google Scholar] [CrossRef]
- Damato, B.; Kacperek, A.; Errington, D.; Heimann, H. Ocular Oncology Update Proton beam radiotherapy of uveal melanoma. Saudi J. Ophthalmol. 2013, 27, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, R.D.; Schwartz, G.K.; Tezel, T.; Marr, B.; Francis, J.H.; Nathan, P.D. Metastatic disease from uveal melanoma: Treatment options and future prospects. Br. J. Ophthalmol. 2017, 101, 38–44. [Google Scholar] [CrossRef]
- Mazzeo, V.; Perri, P.; Degli Innocenti, G.; Bragliani, G.B.; Police, G.; Rossi, A. Problèmes d’ évaluation de l’efficacité de la thérapie conservatrice au moyen des plaques de Ru 106/Rh 106. Ophtalmologie 1995, 9, 325–330. [Google Scholar]
- Nichols, E.E.; Richmond, A.; Daniels, A.B. Tumor Characteristics, Genetics, Management, and the Risk of Metastasis in Uveal Melanoma. Semin. Ophthalmol. 2016, 31, 304–309. [Google Scholar] [CrossRef]
- Onken, M.D.; Worley, L.A.; Char, D.H.; Augsburger, J.J.; Correa, Z.M.; Nudleman, E.; Aaberg, T.M.; Altaweel, M.M.; Bardenstein, D.S.; Finger, P.T.; et al. Collaborative Ocular Oncology Group Report Number 1: Prospective Validation of a Multi-Gene Prognostic Assay in Uveal Melanoma. Ophthalmology 2012, 119, 1596–1603. [Google Scholar] [CrossRef]
- Shields, C.L.; Kaliki, S.; Furuta, M.; Fulco, E.; Alarcon, C.; Shields, J.A. American Joint Committee on Cancer Classification of Posterior Uveal Melanoma (Tumor Size Category) Predicts Prognosis in 7731 Patients. Ophthalmology 2013, 120, 2066–2071. [Google Scholar] [CrossRef]
- Collaborative Ocular Melanoma Study Group. Assessment of metastatic disease status at death in 435 patients with large choroidal melanoma in the Collaborative Ocular Melanoma Study (COMS): COMS report no. 15. Arch. Ophthalmol. 2001, 119, 670–676. [Google Scholar] [CrossRef]
- Pereira, P.R.; Odashiro, A.N.; Lim, L.A.; Miyamoto, C.; Blanco, P.L.; Odashiro, M.; Maloney, S.; De Souza, D.F.; Burnier, M.N. Current and emerging treatment options for uveal melanoma. Clin. Ophthalmol. 2013, 7, 1669–1682. [Google Scholar] [CrossRef]
- Aoyama, T.; Mastrangelo, M.J.; Berd, D.; Nathan, F.E.; Shields, C.L.; Shields, J.A.; Rosato, E.L.; Rosato, F.E.; Sato, T. Protracted survival after resection of metastatic uveal melanoma. Cancer 2000, 89, 1561–1568. [Google Scholar] [CrossRef]
- Homsi, J.; Bedikian, A.Y.; Papadopoulos, N.E.; Kim, K.B.; Hwu, W.J.; Mahoney, S.L.; Hwu, P. Phase 2 open-label study of weekly docosahexaenoic acid-paclitaxel in patients with metastatic uveal melanoma. Melanoma Res. 2010, 20, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Kivelä, T.; Suciu, S.; Hansson, J.; Kruit, W.H.J.; Vuoristo, M.S.; Kloke, O.; Gore, M.; Hahka-Kemppinen, M.; Parvinen, L.M.; Kumpulainen, E.; et al. Bleomycin, vincristine, lomustine and dacarbazine (BOLD) in combination with recombinant interferon alpha-2b for metastatic uveal melanoma. Eur. J. Cancer 2003, 39, 1115–1120. [Google Scholar] [CrossRef]
- Schmittel, A.; Scheulen, M.E.; Bechrakis, N.E.; Strumberg, D.; Baumgart, J.; Bornfeld, N.; Foerster, M.H.; Thiel, E.; Keilholz, U. Phase II trial of cisplatin, gemcitabine and treosulfan in patients with metastatic uveal melanoma. Melanoma Res. 2005, 15, 205–207. [Google Scholar] [CrossRef] [PubMed]
- Klingenstein, A.; Haug, A.R.; Zech, C.J.; Schaller, U.C. Radioembolization as Locoregional Therapy of Hepatic Metastases in Uveal Melanoma Patients. Cardiovasc. Intervent. Radiol. 2013, 36, 158–165. [Google Scholar] [CrossRef]
- Huppert, P.E.; Fierlbeck, G.; Pereira, P.; Schanz, S.; Duda, S.H.; Wietholtz, H.; Rozeik, C.; Claussen, C.D. Transarterial chemoembolization of liver metastases in patients with uveal melanoma. Eur. J. Radiol. 2010, 74, e38–e44. [Google Scholar] [CrossRef] [PubMed]
- Mariani, P.; Piperno-Neumann, S.; Servois, V.; Berry, M.G.; Dorval, T.; Plancher, C.; Couturier, J.; Levy-Gabriel, C.; Lumbroso-Le Rouic, L.; Desjardins, L.; et al. Surgical management of liver metastases from uveal melanoma: 16 years’ experience at the Institut Curie. Eur. J. Surg. Oncol. 2009, 35, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Burgmans, M.C.; de Leede, E.M.; Martini, C.H.; Kapiteijn, E.; Vahrmeijer, A.L.; van Erkel, A.R. Percutaneous Isolated Hepatic Perfusion for the Treatment of Unresectable Liver Malignancies. Cardiovasc. Intervent. Radiol. 2016, 39, 801–814. [Google Scholar] [CrossRef] [PubMed]
- Leyvraz, S.; Piperno-Neumann, S.; Suciu, S.; Baurain, J.F.; Zdzienicki, M.; Testori, A.; Marshall, E.; Scheulen, M.; Jouary, T.; Negrier, S.; et al. Hepatic intra-arterial versus intravenous fotemustine in patients with liver metastases from uveal melanoma (EORTC 18021): A multicentric randomized trial. Ann. Oncol. 2014, 25, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.K.; Kesmodel, S.B.; Alexander, H.R., Jr. Isolated hepatic perfusion for patients with liver metastases. Ther. Adv. Med. Oncol. 2014, 6, 180–194. [Google Scholar] [CrossRef]
- Olofsson, R.; Cahlin, C.; All-Ericsson, C.; Hashimi, F.; Mattsson, J.; Rizell, M.; Lindnér, P. Isolated hepatic perfusion for ocular melanoma metastasis: Registry data suggests a survival benefit. Ann. Surg. Oncol. 2014, 21, 466–472. [Google Scholar] [CrossRef]
- Raval, M.; Bande, D.; Pillai, A.K.; Blaszkowsky, L.S.; Ganguli, S.; Beg, M.S.; Kalva, S.P. Yttrium-90 radioembolization of hepatic metastases from colorectal cancer. Front. Oncol. 2014, 4, 120. [Google Scholar] [CrossRef]
- Gonsalves, C.F.; Eschelman, D.J.; Sullivan, K.L.; Anne, P.R.; Doyle, L.; Sato, T. Radioembolization as Salvage Therapy for Hepatic Metastasis of Uveal Melanoma: A Single-Institution Experience. Am. J. Roentgenol. 2011, 196, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, M.E.; Terai, M.; Eschelman, D.J.; Gonsalves, C.F.; Chervoneva, I.; Shields, J.A.; Shields, C.L.; Yamamoto, A.; Sullivan, K.L.; Laudadio, M.; et al. Double-blinded, randomized phase II study using embolization with or without granulocyte-macrophage colony-stimulating factor in uveal melanoma with hepatic metastases. J. Vasc. Interv. Radiol. 2015, 26, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Bedikian, A.Y.; Legha, S.S.; Mavligit, G.; Carrasco, C.H.; Khorana, S.; Plager, C.; Papadopoulos, N.; Benjamin, R.S. Treatment of uveal melanoma metastatic to the liver: A review of the M. D. Anderson Cancer Center experience and prognostic factors. Cancer 1995, 76, 1665–1670. [Google Scholar] [CrossRef]
- Spagnolo, F.; Caltabiano, G.; Queirolo, P. Uveal melanoma. Cancer Treat. Rev. 2012, 38, 549–553. [Google Scholar] [CrossRef]
- Spagnolo, F.; Grosso, M.; Picasso, V.; Tornari, E.; Pesce, M.; Queirolo, P. Treatment of metastatic uveal melanoma with intravenous fotemustine. Melanoma Res. 2013, 23, 196–198. [Google Scholar] [CrossRef]
- Schmittel, A.; Schmidt-Hieber, M.; Martus, P.; Bechrakis, N.E.; Schuster, R.; Siehl, J.M.; Foerster, M.H.; Thiel, E.; Keilholz, U. A randomized phase II trial of gemcitabine plus treosulfan versus treosulfan alone in patients with metastatic uveal melanoma. Ann. Oncol. 2006, 17, 1826–1829. [Google Scholar] [CrossRef] [PubMed]
- Augsburger, J.J.; Corrêa, Z.M.; Shaikh, A.H. Effectiveness of Treatments for Metastatic Uveal Melanoma. Am. J. Ophthalmol. 2009, 148, 119–127. [Google Scholar] [CrossRef]
- Dhillon, A.S.; Hagan, S.; Rath, O.; Kolch, W. MAP kinase signalling pathways in cancer. Oncogene 2007, 26, 3279. [Google Scholar] [CrossRef]
- Luke, J.J.; Ott, P.A.; Shapiro, G.I. The biology and clinical development of MEK inhibitors for cancer. Drugs 2014, 74, 2111–2128. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.; Foote, R.; Coffey, R. Decrease in cranial nerve complications after radiosurgery for acoustic neuromas: A prospective study of dose and volume. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 305–311. [Google Scholar] [CrossRef]
- Zhao, Y.; Adjei, A.A. The clinical development of MEK inhibitors. Nat. Publ. Gr. 2014, 11, 385–400. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, G.; Pratilas, C.A.; Qin, L.-X.; Tadi, M.; Surriga, O.; Carvajal, R.D.; Schwartz, G.K. Identification of Unique MEK-Dependent Genes in GNAQ Mutant Uveal Melanoma Involved in Cell Growth, Tumor Cell Invasion, and MEK Resistance. Clin. Cancer Res. 2012, 18, 3552–3561. [Google Scholar] [CrossRef]
- Carvajal, R.D.; Sosman, J.A.; Quevedo, J.F.; Milhem, M.M.; Joshua, A.M.; Kudchadkar, R.R.; Linette, G.P.; Gajewski, T.F.; Lutzky, J.; Lawson, D.H.; et al. Effect of selumetinib vs. chemotherapy on progression-free survival in uveal melanoma: A randomized clinical trial. JAMA J. Am. Med. Assoc. 2014, 311, 2397–2405. [Google Scholar] [CrossRef]
- Carvajal, R.D.; Schwartz, G.K.; Mann, H.; Smith, I.; Nathan, P.D. Study design and rationale for a randomised, placebo-controlled, double-blind study to assess the efficacy of selumetinib (AZD6244; ARRY-142886) in combination with dacarbazine in patients with metastatic uveal melanoma (SUMIT). BMC Cancer 2015, 15, 467. [Google Scholar] [CrossRef]
- Das Thakur, M.; Salangsang, F.; Landman, A.S.; Sellers, W.R.; Pryer, N.K.; Levesque, M.P.; Dummer, R.; McMahon, M.; Stuart, D.D. Modelling vemurafenib resistance in melanoma reveals a strategy to forestall drug resistance. Nature 2013, 494, 251–255. [Google Scholar] [CrossRef]
- Holt, S.V.; Logié, A.; Odedra, R.; Heier, A.; Heaton, S.P.; Alferez, D.; Davies, B.R.; Wilkinson, R.W.; Smith, P.D. The MEK1/2 inhibitor, selumetinib (AZD6244; ARRY-142886), enhances anti-tumour efficacy when combined with conventional chemotherapeutic agents in human tumour xenograft models. Br. J. Cancer 2012, 106, 858–866. [Google Scholar] [CrossRef]
- MacKeigan, J.P.; Collins, T.S.; Ting, J.P.-Y. MEK Inhibition Enhances Paclitaxel-induced Tumor Apoptosis. J. Biol. Chem. 2000, 275, 38953–38956. [Google Scholar] [CrossRef]
- Shoushtari, A.N.; Kudchadkar, R.R.; Panageas, K.; Murthy, R.K.; Jung, M.; Shah, R.; O’Donnell, B.; Khawaja, T.T.; Shames, Y.; Prempeh-Keteku, N.A.; et al. A randomized phase 2 study of trametinib with or without GSK2141795 in patients with advanced uveal melanoma. J. Clin. Oncol. 2016, 34, 9511. [Google Scholar] [CrossRef]
- SMR Congress 2017 abstracts. Pigment Cell Melanoma Res. 2018, 31, 125–230. [CrossRef]
- Piperno-Neumann, S.; Kapiteijn, E.; Larkin, J.M.G.; Carvajal, R.D.; Luke, J.J.; Seifert, H.; Roozen, I.; Zoubir, M.; Yang, L.; Choudhury, S.; et al. Phase I dose-escalation study of the protein kinase C (PKC) inhibitor AEB071 in patients with metastatic uveal melanoma. J. Clin. Oncol. 2014, 32, 9030. [Google Scholar] [CrossRef]
- Cheng, H.; Chua, V.; Liao, C.; Purwin, T.J.; Terai, M.; Kageyama, K.; Davies, M.A.; Sato, T.; Aplin, A.E. Co-targeting HGF/cMET Signaling with MEK Inhibitors in Metastatic Uveal Melanoma. Mol. Cancer Ther. 2017, 16, 516–528. [Google Scholar] [CrossRef]
- Mouriaux, F.; Servois, V.; Parienti, J.J.; Lesimple, T.; Thyss, A.; Dutriaux, C.; Neidhart-Berard, E.M.; Penel, N.; Delcambre, C.; Peyro Saint Paul, L.; et al. Sorafenib in metastatic uveal melanoma: Efficacy, toxicity and health-related quality of life in a multicentre phase II study. Br. J. Cancer 2016, 115, 20–24. [Google Scholar] [CrossRef]
- Bhatia, S.; Moon, J.; Margolin, K.A.; Weber, J.S.; Lao, C.D.; Othus, M.; Aparicio, A.M.; Ribas, A.; Sondak, V.K. Phase II trial of sorafenib in combination with carboplatin and paclitaxel in patients with metastatic uveal melanoma: SWOG S0512. PLoS ONE 2012, 7, e48787. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved Survival with Ipilimumab in Patients with Metastatic Melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef]
- Robert, C.; Thomas, L.; Bondarenko, I.; O’Day, S.; Weber, J.; Garbe, C.; Lebbe, C.; Baurain, J.-F.; Testori, A.; Grob, J.-J.; et al. Ipilimumab plus Dacarbazine for Previously Untreated Metastatic Melanoma. N. Engl. J. Med. 2011, 364, 2517–2526. [Google Scholar] [CrossRef]
- Snyder, A.; Makarov, V.; Merghoub, T.; Yuan, J.; Zaretsky, J.M.; Desrichard, A.; Walsh, L.A.; Postow, M.A.; Wong, P.; Ho, T.S.; et al. Genetic Basis for Clinical Response to CTLA-4 Blockade in Melanoma. N. Engl. J. Med. 2014, 371, 2189–2199. [Google Scholar] [CrossRef] [PubMed]
- Furney, S.J.; Pedersen, M.; Gentien, D.; Dumont, A.G.; Rapinat, A.; Desjardins, L.; Turajlic, S.; Piperno-Neumann, S.; de la Grange, P.; Roman-Roman, S.; et al. SF3B1 mutations are associated with alternative splicing in uveal melanoma. Cancer Discov. 2013, 3, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, N.A.; Hellmann, M.D.; Snyder, A.; Kvistborg, P.; Makarov, V.; Havel, J.J.; Lee, W.; Yuan, J.; Wong, P.; Ho, T.S.; et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015, 348, 124–128. [Google Scholar] [CrossRef]
- Graziani, G.; Tentori, L.; Navarra, P. Ipilimumab: A novel immunostimulatory monoclonal antibody for the treatment of cancer. Pharmacol. Res. 2012, 65, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Luke, J.J.; Callahan, M.K.; Postow, M.A.; Romano, E.; Ramaiya, N.; Bluth, M.; Giobbie-Hurder, A.; Lawrence, D.P.; Ibrahim, N.; Ott, P.A.; et al. Clinical activity of ipilimumab for metastatic uveal melanoma. Cancer 2013, 119, 3687–3695. [Google Scholar] [CrossRef] [PubMed]
- Deo, M.A. Long-term survival benefit from ipilimumab treatment in metastatic uveal melanoma patients. J. Clin. Oncol. 2014, 32, 3060. [Google Scholar] [CrossRef]
- Maio, M.; Danielli, R.; Chiarion-Sileni, V.; Pigozzo, J.; Parmiani, G.; Ridolfi, R.; De Rosa, F.; Del Vecchio, M.; Di Guardo, L.; Queirolo, P.; et al. Efficacy and safety of ipilimumab in patients with pre-treated, uveal melanoma. Ann. Oncol. 2013, 24, 2911–2915. [Google Scholar] [CrossRef]
- Afzal, M.Z.; Mabaera, R.; Shirai, K. Metastatic uveal melanoma showing durable response to anti-CTLA-4 and anti-PD-1 combination therapy after experiencing progression on anti-PD-1 therapy alone. J. Immunother. Cancer 2018, 6, 13. [Google Scholar] [CrossRef]
- Danielli, R.; Ridolfi, R.; Chiarion-Sileni, V.; Queirolo, P.; Testori, A.; Plummer, R.; Boitano, M.; Calabrò, L.; De Rossi, C.; Di Giacomo, A.M.; et al. Ipilimumab in pretreated patients with metastatic uveal melanoma: Safety and clinical efficacy. Cancer Immunol. Immunother. 2012, 61, 41–48. [Google Scholar] [CrossRef]
- Piulats Rodriguez, J.M.; Ochoa de Olza, M.; Codes, M.; Lopez-Martin, J.A.; Berrocal, A.; García, M.; Gurpide, A.; Homet, B.; Martin-Algarra, S. Phase II study evaluating ipilimumab as a single agent in the first-line treatment of adult patients (Pts) with metastatic uveal melanoma (MUM): The GEM-1 trial. J. Clin. Oncol. 2014, 32, 9033. [Google Scholar] [CrossRef]
- Zimmer, L.; Eigentler, T.K.; Kiecker, F.; Simon, J.; Utikal, J.; Mohr, P.; Berking, C.; Kämpgen, E.; Dippel, E.; Stadler, R.; et al. Open-label, multicenter, single-arm phase II DeCOG-study of ipilimumab in pretreated patients with different subtypes of metastatic melanoma. J. Transl. Med. 2015, 13, 351. [Google Scholar] [CrossRef] [PubMed]
- Joshua, A.M.; Monzon, J.G.; Mihalcioiu, C.; Hogg, D.; Smylie, M.; Cheng, T. A phase 2 study of tremelimumab in patients with advanced uveal melanoma. Melanoma Res. 2015, 25, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.S.; D’Angelo, S.P.; Minor, D.; Hodi, F.S.; Gutzmer, R.; Neyns, B.; Hoeller, C.; Khushalani, N.I.; Miller, W.H.; Lao, C.D.; et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): A randomised, controlled, open-label, phase 3 trial. Lancet. Oncol. 2015, 16, 375–384. [Google Scholar] [CrossRef]
- Robert, C.; Ribas, A.; Wolchok, J.D.; Hodi, F.S.; Hamid, O.; Kefford, R.; Weber, J.S.; Joshua, A.M.; Hwu, W.-J.; Gangadhar, T.C.; et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: A randomised dose-comparison cohort of a phase 1 trial. Lancet 2014, 384, 1109–1117. [Google Scholar] [CrossRef]
- Robert, C.; Long, G.V.; Brady, B.; Dutriaux, C.; Maio, M.; Mortier, L.; Hassel, J.C.; Rutkowski, P.; McNeil, C.; Kalinka-Warzocha, E.; et al. Nivolumab in Previously Untreated Melanoma without BRAF Mutation. N. Engl. J. Med. 2015, 372, 320–330. [Google Scholar] [CrossRef]
- Kottschade, L.A.; McWilliams, R.R.; Markovic, S.N.; Block, M.S.; Villasboas Bisneto, J.; Pham, A.Q.; Esplin, B.L.; Dronca, R.S. The use of pembrolizumab for the treatment of metastatic uveal melanoma. Melanoma Res. 2016, 26, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Karydis, I.; Chan, P.Y.; Wheater, M.; Arriola, E.; Szlosarek, P.W.; Ottensmeier, C.H. Clinical activity and safety of Pembrolizumab in Ipilimumab pre-treated patients with uveal melanoma. Oncoimmunology 2016, 5, e1143997. [Google Scholar] [CrossRef] [PubMed]
- Algazi, A.P.; Tsai, K.K.; Shoushtari, A.N.; Munhoz, R.R.; Eroglu, Z.; Piulats, J.M.; Ott, P.A.; Johnson, D.B.; Hwang, J.; Daud, A.I.; et al. Clinical outcomes in metastatic uveal melanoma treated with PD-1 and PD-L1 antibodies. Cancer 2016, 122, 3344–3353. [Google Scholar] [CrossRef]
- Javed, A.; Arguello, D.; Johnston, C.; Gatalica, Z.; Terai, M.; Weight, R.M.; Orloff, M.; Mastrangelo, M.J.; Sato, T. PD-L1 expression in tumor metastasis is different between uveal melanoma and cutaneous melanoma. Immunotherapy 2017, 9, 1323–1330. [Google Scholar] [CrossRef]
- Johansson, P.A.; Stark, A.; Palmer, J.M.; Bigby, K.; Brooks, K.; Rolfe, O.; Pritchard, A.L.; Whitehead, K.; Warrier, S.; Glasson, W.; et al. Prolonged stable disease in a uveal melanoma patient with germline MBD4 nonsense mutation treated with pembrolizumab and ipilimumab. Immunogenetics 2019. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Mobuchon, L.; Houy, A.; Fiévet, A.; Gardrat, S.; Barnhill, R.L.; Popova, T.; Servois, V.; Rampanou, A.; Mouton, A.; et al. Outlier response to anti-PD1 in uveal melanoma reveals germline MBD4 mutations in hypermutated tumors. Nat. Commun. 2018, 9, 1866. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; Mensink, H.W.; Aarntzen, E.H.J.G.; Schreibelt, G.; Keunen, J.E.E.; Coulie, P.G.; de Klein, A.; Punt, C.J.A.; Paridaens, D.; Figdor, C.G.; et al. Long overall survival after dendritic cell vaccination in metastatic uveal melanoma patients. Am. J. Ophthalmol. 2014, 158, 939–947. [Google Scholar] [CrossRef] [PubMed]
- van Dinten, L.C.; Pul, N.; van Nieuwpoort, A.F.; Out, C.J.; Jager, M.J.; van den Elsen, P.J. Uveal and Cutaneous Melanoma: Shared Expression Characteristics of Melanoma-Associated Antigens. Investig. Opthalmology Vis. Sci. 2005, 46, 24. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, C. Safety, efficacy and biology of the gp100 TCR-based bispecific T cell redirector, IMCgp100 in advanced uveal melanoma in two Phase 1 trials. J.Stem Cell Res. Ther. 2017, 7, 9. [Google Scholar]
- Sato, T.; Nathan, P.D.; Hernandez-Aya, L.; Sacco, J.J.; Orloff, M.M.; Visich, J.; Little, N.; Hulstine, A.-M.; Coughlin, C.M.; Carvajal, R.D. Redirected T cell lysis in patients with metastatic uveal melanoma with gp100-directed TCR IMCgp100: Overall survival findings. J. Clin. Oncol. 2018, 36, 9521. [Google Scholar] [CrossRef]
- Carvajal, R.D.; Sacco, J.; Nathan, P.; Orloff, M.; Little, N.; McAlpine, C.; Krige, D.; Hassan, N.; Hulstine, A.-M.; Coughlin, C. Takami Sato Safety, efficacy and biology of the gp100 TCR-based bispecific T cell redirector IMCgp100 in advanced uveal melanoma. In Proceedings of the Investigative Ophthalmology & Visual Science, Honolulu, HI, USA, 29 April–3 May 2018; Association for Research in Vision and Ophthalmology: Rockville, MD, USA; Volume 59, p. 3622. [Google Scholar]
- Chandran, S.S.; Somerville, R.P.T.; Yang, J.C.; Sherry, R.M.; Klebanoff, C.A.; Goff, S.L.; Wunderlich, J.R.; Danforth, D.N.; Zlott, D.; Paria, B.C.; et al. Treatment of metastatic uveal melanoma with adoptive transfer of tumour-infiltrating lymphocytes: A single-centre, two-stage, single-arm, phase 2 study. Lancet Oncol. 2017, 18, 792–802. [Google Scholar] [CrossRef]
- Rothermel, L.D.; Sabesan, A.C.; Stephens, D.J.; Chandran, S.S.; Paria, B.C.; Srivastava, A.K.; Somerville, R.; Wunderlich, J.R.; Lee, C.-C.R.; Xi, L.; et al. Identification of an Immunogenic Subset of Metastatic Uveal Melanoma. Clin. Cancer Res. 2016, 22, 2237–2249. [Google Scholar] [CrossRef]
- Patel, S.; Lewis, K.D.; Olencki, T. A phase II study of glembatumumab vedotin for metastatic uveal melanoma. In Proceedings of the 2017 World Congress of Melanoma, Brisbane, Australia, 18 October 2017. Presentation SMR09-5. [Google Scholar]
- Rosenberg, S.A.; Yang, J.C.; Sherry, R.M.; Kammula, U.S.; Hughes, M.S.; Phan, G.Q.; Citrin, D.E.; Restifo, N.P.; Robbins, P.F.; Wunderlich, J.R.; et al. Durable Complete Responses in Heavily Pretreated Patients with Metastatic Melanoma Using T-Cell Transfer Immunotherapy. Clin. Cancer Res. 2011, 17, 4550–4557. [Google Scholar] [CrossRef]
- Ott, P.A.; Pavlick, A.C.; Johnson, D.B.; Hart, L.L.; Infante, J.R.; Luke, J.J.; Lutzky, J.; Rothschild, N.E.; Spitler, L.E.; Cowey, C.L.; et al. A phase II study of glembatumumab vedotin (GV), an antibody-drug conjugate (ADC) targeting gpNMB, in advanced melanoma. J. Clin. Oncol. 2017, 35, 109. [Google Scholar] [CrossRef]
- Herlihy, N.; Dogrusöz, M.; van Essen, T.H.; Harbour, J.W.; van der Velden, P.A.; van Eggermond, M.C.J.A.; Haasnoot, G.W.; van den Elsen, P.J.; Jager, M.J. Skewed expression of the genes encoding epigenetic modifiers in high-risk uveal melanoma. Invest. Ophthalmol. Vis. Sci. 2015, 56, 1447–1458. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.H.; Pilarski, R.; Cebulla, C.M.; Massengill, J.B.; Christopher, B.N.; Boru, G.; Hovland, P.; Davidorf, F.H. Germline BAP1 mutation predisposes to uveal melanoma, lung adenocarcinoma, meningioma, and other cancers. J. Med. Genet. 2011, 48, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Landreville, S.; Agapova, O.A.; Matatall, K.A.; Kneass, Z.T.; Onken, M.D.; Lee, R.S.; Bowcock, A.M.; Harbour, J.W. Histone deacetylase inhibitors induce growth arrest and differentiation in uveal melanoma. Clin. Cancer Res. 2012, 18, 408–416. [Google Scholar] [CrossRef]
- Robertson, A.G.; Shih, J.; Yau, C.; Gibb, E.A.; Oba, J.; Mungall, K.L.; Hess, J.M.; Uzunangelov, V.; Walter, V.; Danilova, L.; et al. Integrative Analysis Identifies Four Molecular and Clinical Subsets in Uveal Melanoma. Cancer Cell 2017, 32, 204–220.e15. [Google Scholar] [CrossRef] [PubMed]
- Decatur, C.L.; Ong, E.; Garg, N.; Anbunathan, H.; Bowcock, A.M.; Field, M.G.; Harbour, J.W. Driver Mutations in Uveal Melanoma. JAMA Ophthalmol. 2016, 134, 728. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Bae, S.-C. Histone deacetylase inhibitors: Molecular mechanisms of action and clinical trials as anti-cancer drugs. Am. J. Transl. Res. 2011, 3, 166–179. [Google Scholar] [PubMed]
- Gezgin, G.; Luk, S.J.; Cao, J.; Dogrusöz, M.; van der Steen, D.M.; Hagedoorn, R.S.; Krijgsman, D.; van der Velden, P.A.; Field, M.G.; Luyten, G.P.M.; et al. PRAME as a Potential Target for Immunotherapy in Metastatic Uveal Melanoma. JAMA Ophthalmol. 2017, 135, 541–549. [Google Scholar] [CrossRef]
| Gene Mutation | Gene Function | Dysregulation | Reference |
|---|---|---|---|
| GNAQ/GNA11 | Responsible for extracellular signal transduction and intracellular pathways activation | 91% of UM patients (principal driver of carcinogenesis) GNAQ/GNA11 mutations are mutually exclusive Mutations in GNAQ have a prevalence of 42.2%, whereas in GNA11 is 32.6%. GNAQ/GNA11 mutations activate MEK and the oncoprotein YAP | [31,32,33,34,35,36,37,38] |
| CYSLTR2 | Involved in leukotriene-mediated signalling in inflammation and fibrosis | Leu129Gln substitution founded only in samples lacking mutations in GNAQ, GNA11, and PLCB4 activation of Gαq signalling | [39,40] |
| PLCB4 | Roles in the metabolism of inositol lipids and cancer | PLCB4 p.D630Y mutations are mutually exclusive with mutations in GNA11 and GNAQ | [41] |
| BAP1 | Nuclear ubiquitin carboxyl-terminal hydrolase enzyme with deubiquitinase activity | 22% in familial UM (associated with early UM onset) BAP1 mutations cause loss of protein expression and loss of function and cell phenotype modifications BAP1 depletion increases the amount of transmigration in UM cells | [23,39,40,41,42,43,44,45,46,47,48,49] |
| SF3B1 | Involved in mRNA splicing | Detected in 15%–19% of UM cases Mutation at codon 625 of SF3B1 gene is associated with UM development. Cause splicing dysregulations and alter the transcription process Mutually exclusive to BAP1 mutations Associated with the chromosome 3 disomy Associated with good prognosis and low metastatic potential | [54,55,56,57] |
| EIF1AX | Stimulates the transfer of Met-tRNAi to the ribosomal subunit | Detected in 8%–18.9% of primary UM cases. Alterations in EIF1AX gene are usually missense mutations It was suggested that an abnormal translation process might be responsible for a clonal selective advantage in cells displaying this alteration | [29,32,58,59] |
| TERT | Catalytic subunit of the enzyme telomerase | It is present in about 1% of UM cases, whereas it is more common in other cancers TERT promoter mutations lead to the immortalization of somatic cells. Tumours carrying this mutation also showed GNA11 gene alteration. | [32,60] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Violanti, S.S.; Bononi, I.; Gallenga, C.E.; Martini, F.; Tognon, M.; Perri, P. New Insights into Molecular Oncogenesis and Therapy of Uveal Melanoma. Cancers 2019, 11, 694. https://doi.org/10.3390/cancers11050694
Violanti SS, Bononi I, Gallenga CE, Martini F, Tognon M, Perri P. New Insights into Molecular Oncogenesis and Therapy of Uveal Melanoma. Cancers. 2019; 11(5):694. https://doi.org/10.3390/cancers11050694
Chicago/Turabian StyleViolanti, Sara Silvia, Ilaria Bononi, Carla Enrica Gallenga, Fernanda Martini, Mauro Tognon, and Paolo Perri. 2019. "New Insights into Molecular Oncogenesis and Therapy of Uveal Melanoma" Cancers 11, no. 5: 694. https://doi.org/10.3390/cancers11050694
APA StyleViolanti, S. S., Bononi, I., Gallenga, C. E., Martini, F., Tognon, M., & Perri, P. (2019). New Insights into Molecular Oncogenesis and Therapy of Uveal Melanoma. Cancers, 11(5), 694. https://doi.org/10.3390/cancers11050694






