Acquired Resistance to Antibody-Drug Conjugates
Abstract
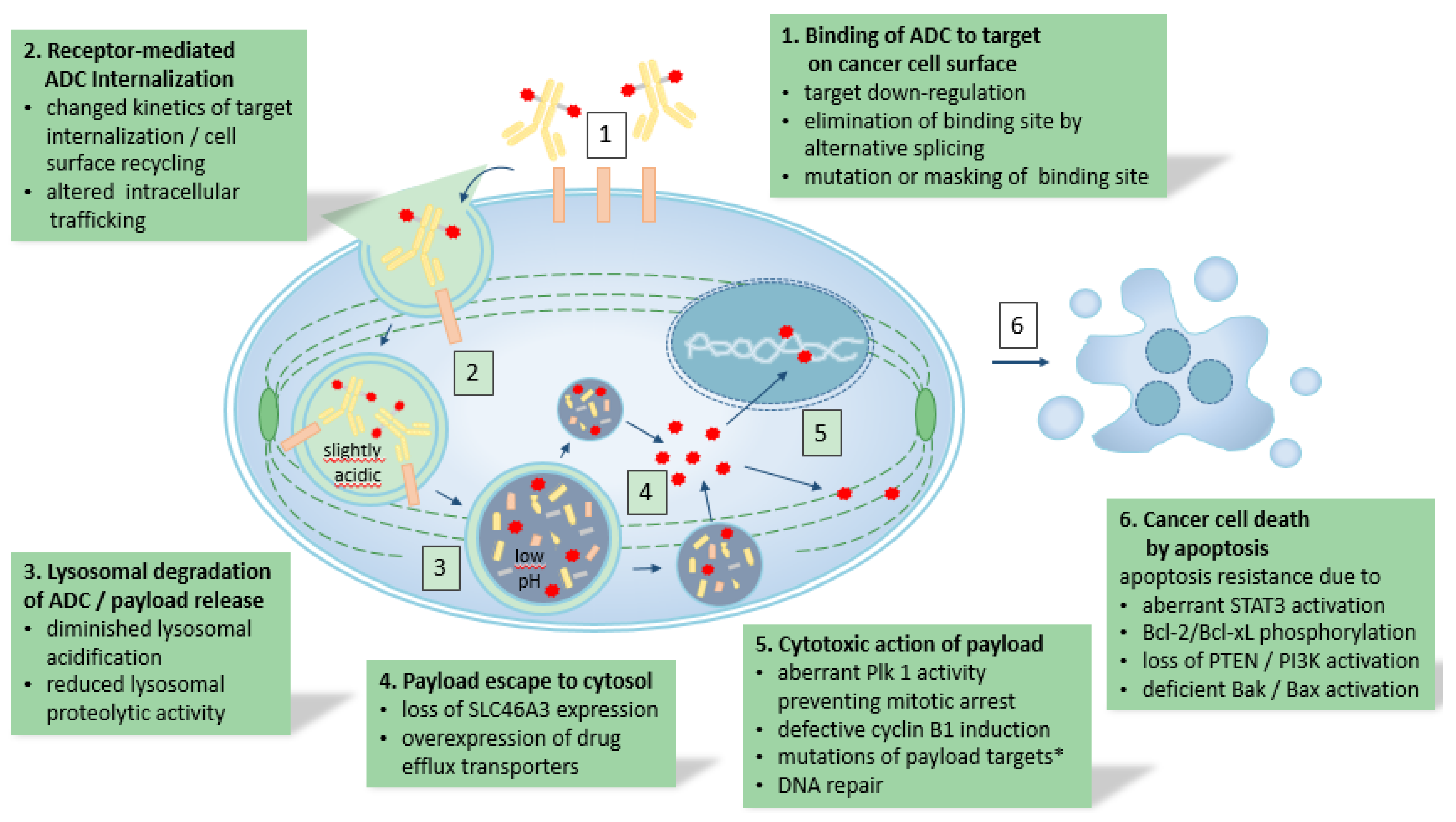
1. Background and Introduction
2. FDA-Approved ADCs
2.1. Gemtuzumab Ozogamicin
2.2. Brentuximab Vedotin
2.3. Trastuzumab Emtansine
2.4. Inotuzumab Ozogamicin
3. Challenges to Overcome ADC Resistance Mechanisms
4. Promising Combination Therapy Approaches
4.1. The Combination with Chemotherapeutics and/or Targeted Agents
4.2. Combination of ADCs with Immune Checkpoint Blockade
5. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Interactive Overview of Antibody Drug Conjugates. Available online: https://adcreview.com/adc-university/adc-drugmap/ (accessed on 30 January 2019).
- Kume, K.; Ikeda, M.; Miura, S.; Ito, K.; Sato, K.A.; Ohmori, Y.; Endo, F.; Katagiri, H.; Ishida, K.; Ito, C.; et al. α-Amanitin Restrains Cancer Relapse from Drug-Tolerant Cell Subpopulations via TAF15. Sci. Rep. 2016, 6, 1–15. [Google Scholar] [CrossRef]
- Zhang, F.; Rothermund, K.; Gangadharan, S.B.; Pommier, Y.; Prochownik, E.V.; Lazo, J.S. Phenotypic Screening Reveals Topoisomerase I as a Breast Cancer Stem Cell Therapeutic Target. Oncotarget 2012, 3, 998. [Google Scholar] [CrossRef] [PubMed]
- Gemtuzumab ozogamicin (Mylotarg®; Pfizer/Wyeth) ADC Review. Available online: https://adcreview.com/gemtuzumab-ozogamicin-mylotarg/ (accessed on 14 March 2019).
- Hamann, P.R.; Hinman, L.M.; Hollander, I.; Beyer, C.F.; Lindh, D.; Holcomb, R.; Hallett, W.; Tsou, H.-R.; Upeslacis, J.; Shochat, D.; et al. Gemtuzumab ozogamicin, a potent and selective anti-CD33 antibody-calicheamicin conjugate for treatment of acute myeloid leukemia. Bioconjug. Chem. 2002, 13, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Tanimoto, M.; Scheinberg, D.A.; Cordon-Cardo, C.; Huie, D.; Clarkson, B.D.; Old, L.J. Restricted expression of an early myeloid and monocytic cell surface antigen defined by monoclonal antibody M195. Leukemia 1989, 3, 339–348. [Google Scholar] [PubMed]
- Ma, H.; Sawas, A. Combining Biology and Chemistry for a New Take on Chemotherapy: Antibody-Drug Conjugates in Hematologic Malignancies. Curr. Hematol. Malig. Rep. 2018, 13, 555–569. [Google Scholar] [CrossRef]
- Hills, R.K.; Castaigne, S.; Appelbaum, F.R.; Delaunay, J.; Petersdorf, S.; Othus, M.; Estey, E.H.; Dombret, H.; Chevret, S.; Ifrah, N.; et al. Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: A meta-analysis of individual patient data from randomised controlled trials. Lancet Oncol. 2014, 15, 986–996. [Google Scholar] [CrossRef]
- Egan, P.C.; Reagan, J.L. The return of gemtuzumab ozogamicin: A humanized anti-CD33 monoclonal antibody–drug conjugate for the treatment of newly diagnosed acute myeloid leukemia. Oncol. Targets Ther. 2018, 11, 8265–8272. [Google Scholar] [CrossRef] [PubMed]
- Fathi, A.T.; Erba, H.P.; Lancet, J.E.; Stein, E.M.; Ravandi, F.; Faderl, S.; Walter, R.B.; Advani, A.S.; DeAngelo, D.J.; Kovacsovics, T.J.; et al. A phase 1 trial of vadastuximab talirine combined with hypomethylating agents in patients with CD33-positive AML. Blood 2018, 132, 1125–1133. [Google Scholar] [CrossRef]
- Khan, N.; Hills, R.K.; Virgo, P.; Couzens, S.; Clark, N.; Gilkes, A.; Richardson, P.; Knapper, S.; Grimwade, D.; Russell, N.H.; et al. Expression of CD33 is a predictive factor for effect of gemtuzumab ozogamicin at different doses in adult acute myeloid leukaemia. Leukemia 2017, 31, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Lamba, J.K.; Chauhan, L.; Shin, M.; Loken, M.R.; Pollard, J.A.; Wang, Y.C.; Ries, R.E.; Aplenc, R.; Hirsch, B.A.; Raimondi, S.C.; et al. CD33 splicing polymorphism determines gemtuzumab ozogamicin response in de novo acute myeloid leukemia: Report from randomized phase III children’s oncology group trial AAML0531. J. Clin. Oncol. 2017, 35, 674–2682. [Google Scholar] [CrossRef] [PubMed]
- Walter, R.B.; Gooley, T.A.; van der Velden, V.H.J.; Loken, M.R.; van Dongen, J.J.M.; Flowers, D.A.; Bernstein, I.D.; Appelbaum, F.R. CD33 expression and P-glycoprotein-mediated drug efflux inversely correlate and predict clinical outcome in patients with acute myeloid leukemia treated with gemtuzumab ozogamicin monotherapy. Blood 2007, 109, 4168–4170. [Google Scholar] [CrossRef]
- Walter, R.B.; Raden, B.W.; Hong, T.C.; Flowers, D.A.; Bernstein, I.D.; Linenberger, M.L. Multidrug resistance protein attenuates gemtuzumab ozogamicin-induced cytotoxicity in acute myeloid leukemia cells. Blood 2003, 102, 1466–1473. [Google Scholar] [CrossRef]
- Matsui, H.; Takeshita, A.; Naito, K.; Shinjo, K.; Shigeno, K.; Maekawa, M.; Yamakawa, Y.; Tanimoto, M.; Kobayashi, M.; Ohnishi, K.; et al. Reduced effect of gemtuzumab ozogamicin (CMA-676) on P-glycoprotein and/or CD34-positive leukemia cells and its restoration by multidrug resistance modifiers. Leukemia 2002, 16, 813–819. [Google Scholar] [CrossRef]
- Matsumoto, T.; Jimi, S.; Hara, S.; Takamatsu, Y.; Suzumiya, J.; Tamura, K. Importance of inducible multidrug resistance 1 expression in HL-60 cells resistant to gemtuzumab ozogamicin. Leuk. Lymphoma 2012, 53, 1399–1405. [Google Scholar] [CrossRef]
- Rosen, D.B.; Harrington, K.H.; Cordeiro, J.A.; Leung, L.Y.; Putta, S.; Lacayo, N.; Laszlo, G.S.; Gudgeon, C.J.; Hogge, D.E.; Hawtin, R.E.; et al. AKT signaling as a novel factor associated with in vitro resistance of human AML to gemtuzumab ozogamicin. PLoS ONE 2013, 8, e53518. [Google Scholar] [CrossRef]
- Haag, P.; Viktorsson, K.; Lindberg, M.L.; Kanter, L.; Lewensohn, R.; Stenke, L. Deficient activation of Bak and Bax confers resistance to gemtuzumab ozogamicin-induced apoptotic cell death in AML. Exp. Hematol. 2009, 37, 755–766. [Google Scholar] [CrossRef]
- Maimaitili, Y.; Inase, A.; Miyata, Y.; Kitao, A.; Mizutani, Y.; Kakiuchi, S.; Shimono, Y.; Saito, Y.; Sonoki, T.; Minami, H.; et al. An mTORC1/2 kinase inhibitor enhances the cytotoxicity of gemtuzumab ozogamicin by activation of lysosomal function. Leuk. Res. 2018, 74, 68–74. [Google Scholar] [CrossRef]
- Forero-Torres, A.; Leonard, J.P.; Younes, A.; Rosenblatt, J.D.; Brice, P.; Bartlett, N.L.; Bosly, A.; Pinter-Brown, L.; Kennedy, D.; Sievers, E.L.; et al. A Phase II study of SGN-30 (anti-CD30 mAb) in Hodgkin lymphoma or systemic anaplastic large cell lymphoma. Br. J. Haematol. 2009, 146, 171–179. [Google Scholar] [CrossRef]
- Donato, E.M.; Fernández-Zarzoso, M.; Hueso, J.A.; de la Rubia, J. Brentuximab vedotin in Hodgkin lymphoma and anaplastic large-cell lymphoma: An evidence-based review. Oncol. Targets Ther. 2018, Volume 11, 4583–4590. [Google Scholar] [CrossRef]
- Chen, R.; Hou, J.; Newman, E.; Kim, Y.; Donohue, C.; Liu, X.; Thomas, S.H.; Forman, S.J.; Kane, S.E. CD30 Downregulation, MMAE Resistance, and MDR1 Upregulation Are All Associated with Resistance to Brentuximab Vedotin. Mol. Cancer Ther. 2015, 14, 1376–1384. [Google Scholar] [CrossRef]
- Beck, A.; Goetsch, L.; Dumontet, C.; Corvaïa, N. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 2017, 16, 315–337. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, N.L.; Chen, R.; Fanale, M.A.; Brice, P.; Gopal, A.; Smith, S.E.; Advani, R.; Matous, J.V.; Ramchandren, R.; Rosenblatt, J.D.; et al. Retreatment with brentuximab vedotin in patients with CD30-positive hematologic malignancies. J. Hematol. Oncol. 2014, 7, 24. [Google Scholar] [CrossRef]
- Dean, L. Trastuzumab (Herceptin) Therapy and ERBB2 (HER2) Genotype; National Center for Biotechnology Information: Bethesda, MD, USA, 2012.
- Vogel, C. Herceptin clinical trials: Past, present and future. Breast Cancer Res. 2001, 3, A66. [Google Scholar] [CrossRef]
- Diéras, V.; Miles, D.; Verma, S.; Pegram, M.; Welslau, M.; Baselga, J.; Krop, I.E.; Blackwell, K.; Hoersch, S.; Xu, J.; et al. Trastuzumab emtansine versus capecitabine plus lapatinib in patients with previously treated HER2-positive advanced breast cancer (EMILIA): A descriptive analysis of final overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2017, 18, 732–742. [Google Scholar] [CrossRef]
- Krop, I.E.; Kim, S.-B.; Martin, A.G.; LoRusso, P.M.; Ferrero, J.-M.; Badovinac-Crnjevic, T.; Hoersch, S.; Smitt, M.; Wildiers, H. Trastuzumab emtansine versus treatment of physician’s choice in patients with previously treated HER2-positive metastatic breast cancer (TH3RESA): Final overall survival results from a randomised open-label phase 3 trial. Lancet. Oncol. 2017, 18, 743–754. [Google Scholar] [CrossRef]
- Krop, I.E.; Kim, S.-B.; González-Martín, A.; LoRusso, P.M.; Ferrero, J.-M.; Smitt, M.; Yu, R.; Leung, A.C.F.; Wildiers, H.; TH3RESA Study Collaborators. Trastuzumab emtansine versus treatment of physician’s choice for pretreated HER2-positive advanced breast cancer (TH3RESA): A randomised, open-label, phase 3 trial. Lancet Oncol. 2014, 15, 689–699. [Google Scholar] [CrossRef]
- Baselga, J.; Lewis Phillips, G.D.; Verma, S.; Ro, J.; Huober, J.; Guardino, A.E.; Samant, M.K.; Olsen, S.; de Haas, S.L.; Pegram, M.D. Relationship between Tumor Biomarkers and Efficacy in EMILIA, a Phase III Study of Trastuzumab Emtansine in HER2-Positive Metastatic Breast Cancer. Clin. Cancer Res. 2016, 22, 3755–3763. [Google Scholar] [CrossRef] [PubMed]
- Perez, E.A.; Barrios, C.; Eiermann, W.; Toi, M.; Im, Y.-H.; Conte, P.; Martin, M.; Pienkowski, T.; Pivot, X.; Burris, H.; et al. Trastuzumab Emtansine With or Without Pertuzumab Versus Trastuzumab Plus Taxane for Human Epidermal Growth Factor Receptor 2-Positive, Advanced Breast Cancer: Primary Results From the Phase III MARIANNE Study. J. Clin. Oncol. 2017, 35, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Buckley, N.E.; Forde, C.; McArt, D.G.; Boyle, D.P.; Mullan, P.B.; James, J.A.; Maxwell, P.; McQuaid, S.; Salto-Tellez, M. Quantification of HER2 heterogeneity in breast cancer-implications for identification of sub-dominant clones for personalised treatment. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ocaña, A.; Amir, E.; Pandiella, A. Dual targeting of HER2-positive breast cancer with trastuzumab emtansine and pertuzumab: Understanding clinical trial results. Oncotarget 2018, 9, 31915–31919. [Google Scholar] [CrossRef]
- Hurvitz, S.A.; Martin, M.; Symmans, W.F.; Jung, K.H.; Huang, C.-S.; Thompson, A.M.; Harbeck, N.; Valero, V.; Stroyakovskiy, D.; Wildiers, H.; et al. Neoadjuvant trastuzumab, pertuzumab, and chemotherapy versus trastuzumab emtansine plus pertuzumab in patients with HER2-positive breast cancer (KRISTINE): A randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2018, 19, 115–126. [Google Scholar] [CrossRef]
- Von Minckwitz, G.; Huang, C.-S.; Mano, M.S.; Loibl, S.; Mamounas, E.P.; Untch, M.; Wolmark, N.; Rastogi, P.; Schneeweiss, A.; Redondo, A.; et al. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. N. Engl. J. Med. 2019, 380, 617–628. [Google Scholar] [CrossRef]
- Rinnerthaler, G.; Gampenrieder, S.P.; Greil, R. HER2 directed antibody-drug-conjugates beyond T-DM1 in breast cancer. Preprints 2019, 20, 1115. [Google Scholar] [CrossRef]
- Al-Saden, N.; Cai, Z.; Reilly, R.M. Tumor uptake and tumor/blood ratios for [89Zr]Zr-DFO-trastuzumab-DM1 on microPET/CT images in NOD/SCID mice with human breast cancer xenografts are directly correlated with HER2 expression and response to trastuzumab-DM1. Nucl. Med. Biol. 2018, 67, 43–51. [Google Scholar] [CrossRef]
- Kim, S.-B.; Wildiers, H.; Krop, I.E.; Smitt, M.; Yu, R.; Lysbet de Haas, S.; Gonzalez-Martin, A. Relationship between tumor biomarkers and efficacy in TH3RESA, a phase III study of trastuzumab emtansine (T-DM1) vs. treatment of physician’s choice in previously treated HER2-positive advanced breast cancer. Int. J. Cancer 2016, 139, 2336–2342. [Google Scholar] [CrossRef]
- Loganzo, F.; Tan, X.; Sung, M.; Jin, G.; Myers, J.S.; Melamud, E.; Wang, F.; Diesl, V.; Follettie, M.T.; Musto, S.; et al. Tumor Cells Chronically Treated with a Trastuzumab-Maytansinoid Antibody-Drug Conjugate Develop Varied Resistance Mechanisms but Respond to Alternate Treatments. Mol. Cancer Ther. 2015, 14, 952–963. [Google Scholar] [CrossRef]
- Sung, M.; Tan, X.; Lu, B.; Golas, J.; Hosselet, C.; Wang, F.; Tylaska, L.; King, L.; Zhou, D.; Dushin, R.; et al. Caveolae-mediated endocytosis as a novel mechanism of resistance to trastuzumab emtansine (T-DM1). Mol. Cancer Ther. 2017, 17, 243–253. [Google Scholar] [CrossRef]
- Wang, H.; Wang, W.; Xu, Y.; Yang, Y.; Chen, X.; Quan, H.; Lou, L. Aberrant intracellular metabolism of T-DM1 confers T-DM1 resistance in human epidermal growth factor receptor 2-positive gastric cancer cells. Cancer Sci. 2017, 108, 1458–1468. [Google Scholar] [CrossRef]
- Ríos-Luci, C.; García-Alonso, S.; Díaz-Rodríguez, E.; Nadal-Serrano, M.; Arribas, J.; Ocaña, A.; Pandiella, A. Resistance to the antibody–drug conjugate T-DM1 is based in a reduction in lysosomal proteolytic activity. Cancer Res. 2017, 77, 4639–4651. [Google Scholar] [CrossRef]
- Hamblett, K.J.; Jacob, A.P.; Gurgel, J.L.; Tometsko, M.E.; Rock, B.M.; Patel, S.K.; Milburn, R.R.; Siu, S.; Ragan, S.P.; Rock, D.A.; et al. SLC46A3 Is Required to Transport Catabolites of Noncleavable Antibody Maytansine Conjugates from the Lysosome to the Cytoplasm. Cancer Res. 2015, 75, 5329–5340. [Google Scholar] [CrossRef]
- Li, G.; Guo, J.; Shen, B.-Q.; Yadav, D.B.; Sliwkowski, M.X.; Crocker, L.M.; Lacap, J.A.; Phillips, G.D.L. Mechanisms of Acquired Resistance to Trastuzumab Emtansine in Breast Cancer Cells. Mol. Cancer Ther. 2018, 17, 1441–1453. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Q.; Gao, M.; Fu, L.; Li, Y.; Quan, H.; Lou, L. STAT3 activation confers trastuzumab-emtansine (T-DM1) resistance in HER2-positive breast cancer. Cancer Sci. 2018, 109, 3305–3315. [Google Scholar] [CrossRef]
- Saatci, Ö.; Borgoni, S.; Akbulut, Ö.; Durmuş, S.; Raza, U.; Eyüpoğlu, E.; Alkan, C.; Akyol, A.; Kütük, Ö.; Wiemann, S.; et al. Targeting PLK1 overcomes T-DM1 resistance via CDK1-dependent phosphorylation and inactivation of Bcl-2/xL in HER2-positive breast cancer. Oncogene 2018, 37, 2251–2269. [Google Scholar] [CrossRef]
- Sabbaghi, M.; Guardia, C.; Servitja, S.; Menendez, S.; Serrano, L.; Salido, M.; Muntasell, A.; Zazo, S.; Eroles, P.; Tusquets, I.; et al. Defective cyclin B1 induction in trastuzumab-emtansine ( T-DM1) acquired resistance in HER2-positive breast cancer. Clin. Cancer Res. 2017, 23, 7006–7019. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Zeng, C.; Hari, M.; Cabral, F. Random Mutagenesis of β-Tubulin Defines a Set of Dispersed Mutations That Confer Paclitaxel Resistance. Pharm. Res. 2012, 29, 2994–3006. [Google Scholar] [CrossRef]
- Nami, B.; Wang, Z. Genetics and Expression Profile of the Tubulin Gene Superfamily in Breast Cancer Subtypes and Its Relation to Taxane Resistance. Cancers 2018, 10, 274. [Google Scholar] [CrossRef]
- Piccaluga, P.P.; Arpinati, M.; Candoni, A.; Laterza, C.; Paolini, S.; Gazzola, A.; Sabattini, E.; Visani, G.; Pileri, S.A. Surface antigens analysis reveals significant expression of candidate targets for immunotherapy in adult acute lymphoid leukemia. Leuk. Lymphoma 2011, 52, 325–327. [Google Scholar] [CrossRef]
- Raponi, S.; De Propris, M.S.; Intoppa, S.; Milani, M.L.; Vitale, A.; Elia, L.; Perbellini, O.; Pizzolo, G.; Foá, R.; Guarini, A. Flow cytometric study of potential target antigens (CD19, CD20, CD22, CD33) for antibody-based immunotherapy in acute lymphoblastic leukemia: Analysis of 552 cases. Leuk. Lymphoma 2011, 52, 1098–1107. [Google Scholar] [CrossRef]
- Shan, D.; Press, O.W. Constitutive endocytosis and degradation of CD22 by human B cells. J. Immunol. 1995, 154, 4466–4475. [Google Scholar]
- Drake, P.M.; Carlson, A.; McFarland, J.M.; Bañas, S.; Barfield, R.M.; Zmolek, W.; Kim, Y.C.; Huang, B.C.B.; Kudirka, R.; Rabuka, D. CAT-02-106, a Site-Specifically Conjugated Anti-CD22 Antibody Bearing an MDR1-Resistant Maytansine Payload Yields Excellent Efficacy and Safety in Preclinical Models. Mol. Cancer Ther. 2018, 17, 161–168. [Google Scholar] [CrossRef]
- Advani, R.H.; Lebovic, D.; Chen, A.; Brunvand, M.; Goy, A.; Chang, J.E.; Hochberg, E.; Yalamanchili, S.; Kahn, R.; Lu, D.; et al. Phase I Study of the Anti-CD22 Antibody–Drug Conjugate Pinatuzumab Vedotin with/without Rituximab in Patients with Relapsed/Refractory B-cell Non-Hodgkin Lymphoma. Clin. Cancer Res. 2017, 23, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Weber, T.; Mavratzas, A.; Kiesgen, S.; Haase, S.; Bötticher, B.; Exner, E.; Mier, W.; Grosse-Hovest, L.; Jäger, D.; Arndt, M.A.E.; et al. A Humanized Anti-CD22-Onconase Antibody-Drug Conjugate Mediates Highly Potent Destruction of Targeted Tumor Cells. J. Immunol. Res. 2015, 2015, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.-F.; Zheng, B.; Go, M.; Lau, J.; Spencer, S.; Raab, H.; Soriano, R.; Jhunjhunwala, S.; Cohen, R.; Caruso, M.; et al. A Novel Anti-CD22 Anthracycline-Based Antibody-Drug Conjugate (ADC) That Overcomes Resistance to Auristatin-Based ADCs. Clin. Cancer Res. 2015, 21, 3298–3306. [Google Scholar] [CrossRef]
- Kantarjian, H.M.; DeAngelo, D.J.; Stelljes, M.; Martinelli, G.; Liedtke, M.; Stock, W.; Gökbuget, N.; O’Brien, S.; Wang, K.; Wang, T.; et al. Inotuzumab Ozogamicin versus Standard Therapy for Acute Lymphoblastic Leukemia. N. Engl. J. Med. 2016, 375, 740–753. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, A.; Shinjo, K.; Yamakage, N.; Ono, T.; Hirano, I.; Matsui, H.; Shigeno, K.; Nakamura, S.; Tobita, T.; Maekawa, M.; et al. CMC-544 (inotuzumab ozogamicin) shows less effect on multidrug resistant cells: Analyses in cell lines and cells from patients with B-cell chronic lymphocytic leukaemia and lymphoma. Br. J. Haematol. 2009, 146, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Loganzo, F.; Sung, M.; Gerber, H.-P. Mechanisms of Resistance to Antibody–Drug Conjugates. Mol. Cancer Ther. 2016, 15, 2825–2834. [Google Scholar] [CrossRef]
- Sendur, M.A.N.; Aksoy, S.; Altundag, K. Cardiotoxicity of novel HER2-targeted therapies. Curr. Med. Res. Opin. 2013, 29, 1015–1024. [Google Scholar] [CrossRef]
- Ward, E.S.; Ober, R.; Kang, J.; Sun, W. Endolysosomal targeting conjugates for improved delivery of cargo molecules to the endolysosomal compartment of target cells. Patent WO2018136455A1 2018.
- Martin, M.; Fumoleau, P.; Dewar, J.A.; Albanell, J.; Limentani, S.A.; Campone, M.; Chang, J.C.; Patre, M.; Strasak, A.; de Haas, S.L.; et al. Trastuzumab emtansine (T-DM1) plus docetaxel with or without pertuzumab in patients with HER2-positive locally advanced or metastatic breast cancer: Results from a phase Ib/IIa study. Ann. Oncol. 2016, 27, 1249–1256. [Google Scholar] [CrossRef]
- Chooniedass, S.; Dillon, R.L.; Premsukh, A.; Hudson, P.J.; Adams, G.P.; MacDonald, G.C.; Cizeau, J. DeBouganin Diabody Fusion Protein Overcomes Drug Resistance to ADCs Comprised of Anti-Microtubule Agents. Molecules 2016, 21, 1741. [Google Scholar] [CrossRef]
- Kolyvas, E.; Rudloff, M.; Poruchynsky, M.; Landsman, R.; Hollevoet, K.; Venzon, D.; Alewine, C. Mesothelin-targeted immunotoxin RG7787 has synergistic anti-tumor activity when combined with taxanes. Oncotarget 2017, 8, 9189–9199. [Google Scholar] [CrossRef] [PubMed]
- Kather, J.N.; Suarez-Carmona, M.; Charoentong, P.; Weis, C.-A.; Hirsch, D.; Bankhead, P.; Horning, M.; Ferber, D.; Kel, I.; Herpel, E.; et al. Topography of cancer-associated immune cells in human solid tumors. eLife 2018, 7, e36967. [Google Scholar] [CrossRef] [PubMed]
- Gerber, H.P.; Sapra, P.; Loganzo, F.; May, C. Combining antibody-drug conjugates and immune-mediated cancer therapy: What to expect? Biochem. Pharmacol. 2015, 102, 1–6. [Google Scholar] [CrossRef]
- Müller, P.; Kreuzaler, M.; Khan, T.; Thommen, D.S.; Martin, K.; Glatz, K.; Savic, S.; Harbeck, N.; Nitz, U.; Gluz, O.; et al. Trastuzumab emtansine (T-DM1) renders HER2+ breast cancer highly susceptible to CTLA-4/PD-1 blockade. Sci. Transl. Med. 2015, 7, 315ra188. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Martin, K.; Theurich, S.; Schreiner, J.; Savic, S.; Terszowski, G.; Lardinois, D.; Heinzelmann-Schwarz, V.A.; Schlaak, M.; Kvasnicka, H.-M.; et al. Microtubule-Depolymerizing Agents Used in Antibody-Drug Conjugates Induce Antitumor Immunity by Stimulation of Dendritic Cells. Cancer Immunol. Res. 2014, 2, 741–755. [Google Scholar] [CrossRef] [PubMed]
| ADC | Resistance Mechanisms Directed Against | ||
|---|---|---|---|
| Targeting Moiety | Linker | Payload | |
| Gemtuzumab ozogamicin | CD33 splice variant lacking antibody epitope | drug efflux transporters, | |
| PI3K pathway activation, | |||
| mTORC1/2 activation, | |||
| deficient Bak/Bax activation | |||
| Brentuximab vedotin | CD30 down-regulation | drug efflux transporter, | |
| MMAE resistance | |||
| Trastuzumab emtansine | HER2 down-regulation, | enhanced trafficking to non-lysosomal compartments, reduced V-ATPase activity * | drug efflux transporter, |
| SLC46A3 down-regulation, | |||
| STAT3 pathway activation, | |||
| altered internalization | PTEN/PI3K activation, | ||
| PLK1 activation, | |||
| failure to induce Cyclin B1 | |||
| Inotuzumab ozogamicin | drug efflux transporters | ||
| Combination | Trial Number | Phase | MoA of Combined Agent(s) | Indication |
|---|---|---|---|---|
| GO and 5-azacitidine | NCT00766116 | I/II | DNA methyl-transferase inhibitor | Relapsed AML |
| GO and the combo of idarubicin, etoposide, cytarabine, pegfilgrastim with ATRA 1 | NCT00893399 | III | Three different DNA damaging agents plus agonists for G-CSF and retinoic acid receptor | AML with NPM1 mutation 2 |
| GO and glasdegib | NCT03390296 | Ib/II | Smoothened inhibitor | RR 3 AML |
| Conditioning therapy with GO plus cyclophosphamide and busulfan chemotherapy followed by ASCT | NCT02221310 | II | Two immune suppressive alkylating agents | High-risk AML or myelodysplastic syndrome (MDS) |
| GO and G-CSF, cladribine, cytarabine, and mitoxantrone | NCT03531918 | I/II | Three different DNA damaging agents plus a G-CSFR agonist | 1st line AML |
| GO and daunorubicin/cytarabine filled liposomes | NCT03672539 | I | Two different DNA damaging agents | RR 3 AML, high-risk MDS |
| Neoadjuvant T-DM1 and lapatinib followed by Abraxane | NCT02073487 | II | A HER1/2 kinase and a microtubule inhibitor | HER2+ breast cancer |
| T-DM1 and poziotinib | NCT03429101 | I | Covalent HER1/2/4 kinase inhibitor | Metastatic HER2+ breast cancer |
| T-DM1 and osimertinib | NCT03784599 | II | HER1 T790M kinase inhibitor | Mutant HER1+, HER2+, stage IV lung cancer |
| T-DM1 and neratinib | NCT02236000 | Ib/II | Irreversible pan-HER inhibitor | Metastatic HER2+ breast cancer |
| T-DM1 and palbociclib | NCT01976169 | Ib | CDK4/6 inhibitor | Recurrent or metastatic HER2+ breast cancer |
| T-DM1 and ribociclib | NCT02657343 | Ib/II | CDK4/6 inhibitor | Metastatic HER2+ breast cancer |
| T-DM1 and palbociclib | NCT03530696 | II | CDK4/6 inhibitor | Metastatic HER2+ breast cancer |
| T-DM1 and taselisib | NCT02390427 | Ib | Phosphoinositide 3-kinase α inhibitor | Advanced HER2+ breast cancer |
| INO and cyclophosphamide, vincristine, prednisone | NCT01925131 | I | An alkylating agent, a microtubule inhibitor, and a glucocorticoid | RR 3 CD22+ acute leukemia |
| INO and low dose chemotherapy (cyclophosphamide/vincristine or methotrexate/cytarabine) | NCT01371630 | I/II | Well tolerated cytostatic agents | Older patients with previously untreated ALL |
| INO and bosutinib | NCT02311998 | I/II | Bcr-Abl kinase inhibitor | CD22 and Philadelphia-chromosome positive ALL and CML |
| INO and intensive chemo-therapy (Hyper-CVAD regimen) | NCT03488225 | II | 11 induction therapy plus two maintenance therapy agents | 1st line B-cell lineage ALL |
| INO and rituximab, cyclophosphamide, vincristine, prednisolone | NCT01679119 | II | Anti-CD20 antibody, well tolerated cytostatic agents | DLBCL patients unfit for anthracycline |
| Combination | Trial Number | Phase | MoA of Combo Agent(s) | Indication |
|---|---|---|---|---|
| T-DM1 and pembrolizumab | NCT03032107 | Ib | PD-1 blocking antibody | Metastatic HER2+ breast cancer |
| T-DM1 and atezolizumab | NCT02924883 | Ib | PD-L1 blocking antibody | Locally advanced or metastatic HER2+ breast cancer |
| Different doses of T-DM1 and atezolizumab | NCT02605915 | Ib | PD-L1 blocking antibody | Locally advanced or metastatic HER2+ breast cancer |
| T-DM1 and utomilumab | NCT03364348 | Ib | Agonistic 4-1BB antibody | HER2+ advanced breast cancer |
| BV and nivolumab | NCT02581631 | I/II | PD-1 blocking antibody | RR 1 CD30+ Hodgkin Lymphoma |
| BV and pembrolizumab | NCT02684292 | III | PD-1 blocking antibody | RR 1 CD30+ Hodgkin Lymphoma |
| BV and nivolumab +/− ipilimumab | NCT01896999 | I/II | PD-1 and CTLA-4 blocking antibodies | RR 1 CD30+ Hodgkin Lymphoma |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collins, D.M.; Bossenmaier, B.; Kollmorgen, G.; Niederfellner, G. Acquired Resistance to Antibody-Drug Conjugates. Cancers 2019, 11, 394. https://doi.org/10.3390/cancers11030394
Collins DM, Bossenmaier B, Kollmorgen G, Niederfellner G. Acquired Resistance to Antibody-Drug Conjugates. Cancers. 2019; 11(3):394. https://doi.org/10.3390/cancers11030394
Chicago/Turabian StyleCollins, Denis M., Birgit Bossenmaier, Gwendlyn Kollmorgen, and Gerhard Niederfellner. 2019. "Acquired Resistance to Antibody-Drug Conjugates" Cancers 11, no. 3: 394. https://doi.org/10.3390/cancers11030394
APA StyleCollins, D. M., Bossenmaier, B., Kollmorgen, G., & Niederfellner, G. (2019). Acquired Resistance to Antibody-Drug Conjugates. Cancers, 11(3), 394. https://doi.org/10.3390/cancers11030394





