Personalized Radiation Therapy in Cancer Pain Management
Abstract
1. Introduction
2. Painful Bone Metastases
2.1. Pathomechanism of Analgesic Effect of Radiation
2.2. External Beam Radiation Therapy for Painful Bone Metastases
2.2.1. Conformal Radiation Therapy
2.2.2. Hemibody Irradiation (HBI)
2.2.3. Stereotactic Surgery/Stereotactic Body Radiation Therapy
2.2.4. Re-Irradiation
3. Brachytherapy for the Treatment of Painful Bone Metastases
Pain Flare Syndrome
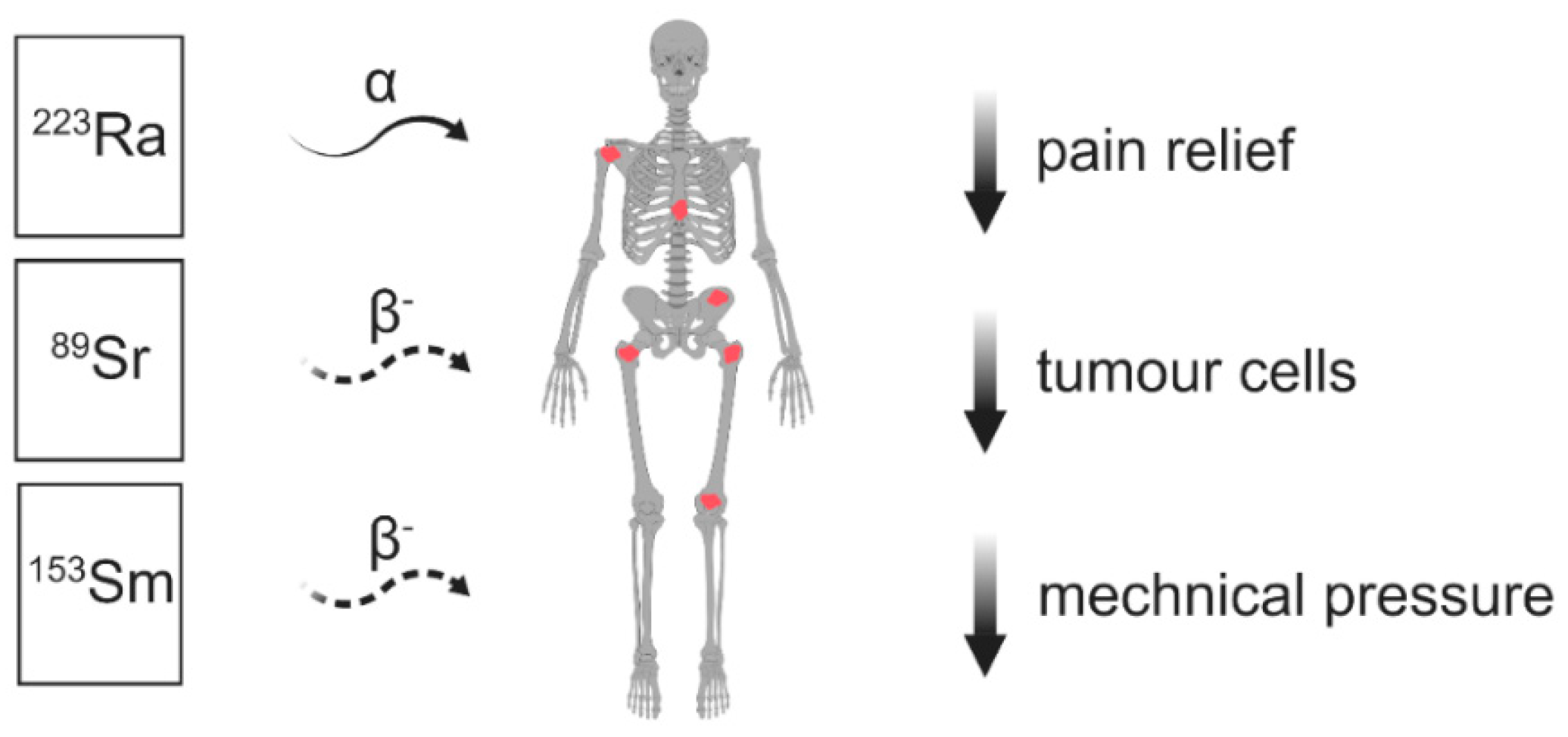
4. Radioactive Isotopes for the Treatment of Painful Bone Metastases
4.1. Several Radionuclides Are Used in Clinical Practice, and Many of Them Are under Investigation
4.1.1. Strontium-89 Chloride
4.1.2. Samarium-153-EDTMP
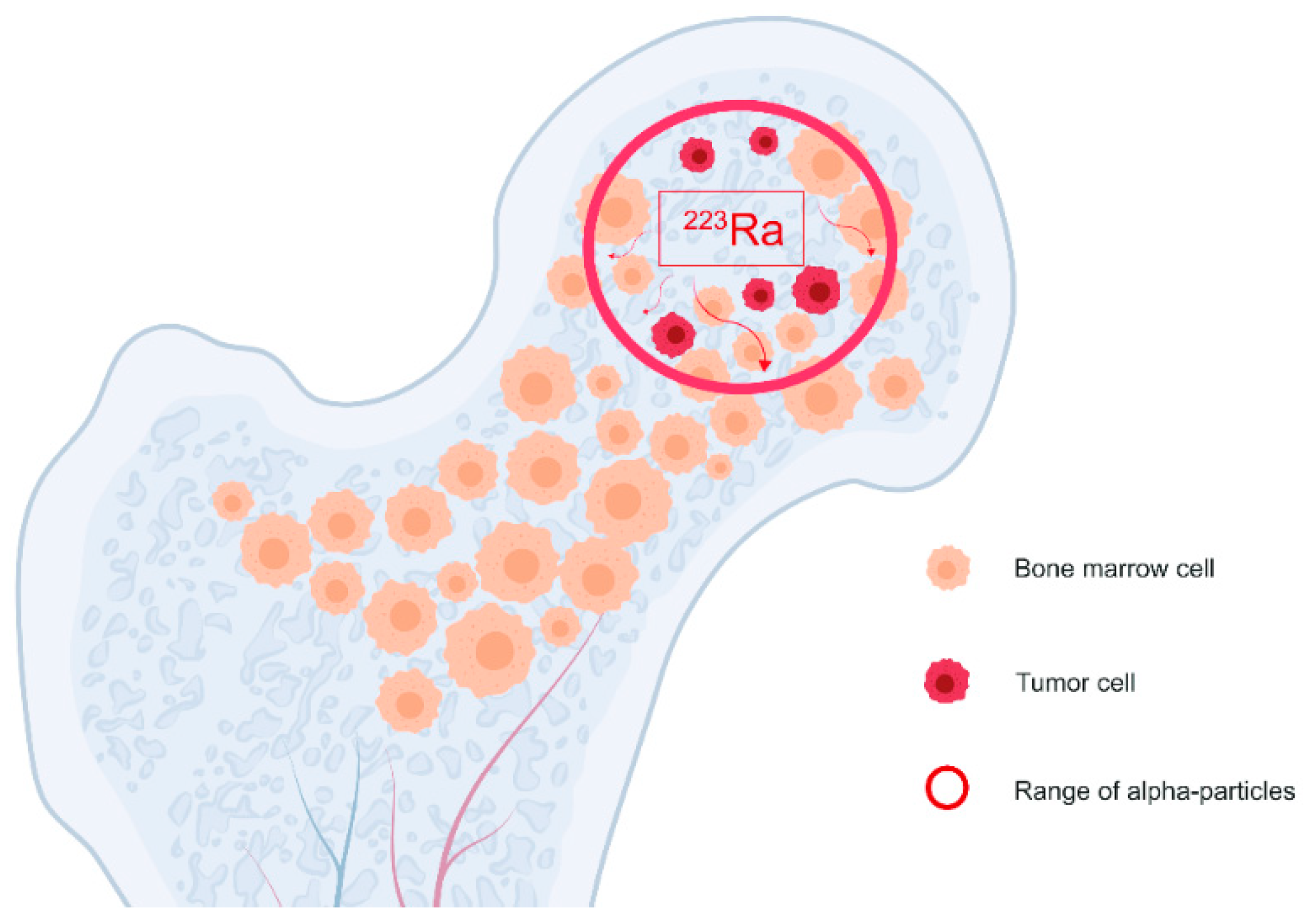
4.1.3. Radium-223-Dichloride
4.1.4. Rhenium-186-HEDP
4.1.5. Rhenium-188-HEDP
4.2. Side Effects of Radionuclide Therapy
5. Radiation Therapy for Painful Primary/Regional/Metastatic Solid Tumours Other Than Bone Tumours
6. Pain Assessment after Radiation Therapy
- Complete response: A pain score of 0 at the treated site and no concomitant increase in analgesic intake, which means stable or reduced analgesics in daily oral morphine equivalent (OMED).
- Partial response: Pain decrease of 2 or more at the treated site on a scale of 0 to 10 without analgesic increase, or analgesic dose decrease of 25% or more from the baseline without an increase in pain intensity.
- Pain progression: Increase in pain score of 2 or more above the baseline at the treated site with stable OMED, or an increase of 25% or more OMED in comparison to the baseline with the pain score stable or 1 point above the baseline.
- Intermediate response: Any response that is not captured by those defined above [159].
7. Future Directions
8. Conclusions
Funding
Conflicts of Interest
References
- Agarawal, J.P.; Swangsilpa, T.; van der Linden, Y.; Rades, D.; Jeremic, B.; Hoskin, P.J. The Role of External Beam Radiotherapy in the Management of Bone Metastases. Clin. Oncol. 2006, 18, 747–760. [Google Scholar] [CrossRef]
- Chow, E.; Zeng, L.; Salvo, N.; Dennis, K.; Tsao, M.; Lutz, S. Update on the Systematic Review of Palliative Radiotherapy Trials for Bone Metastases. Clin. Oncol. 2012, 24, 112–124. [Google Scholar] [CrossRef]
- Konopka-Filippow, M.; Zabrocka, E.; Wõjtowicz, A.; Skalij, P.; Wojtukiewicz, M.Z.; Sierko, E. Pain management during radiotherapy and radiochemotherapy in oropharyngeal cancer patients: Single-institution experience. Int. Dent. J. 2015, 65, 242–248. [Google Scholar] [CrossRef]
- Lutz, S.; Berk, L.; Chang, E.; Chow, E.; Hahn, C.; Hoskin, P.; Howell, D.; Konski, A.; Kachnic, L.; Lo, S.; et al. Palliative radiotherapy for bone metastases: An ASTRO evidence-based guideline. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 965–976. [Google Scholar] [CrossRef]
- Kougioumtzopoulou, A.; Zygogianni, A.; Liakouli, Z.; Kypraiou, E.; Kouloulias, V. The role of radiotherapy in bone metastases: A critical review of current literature. Eur. J. Cancer Care (Engl). 2017, 26, e12724. [Google Scholar] [CrossRef]
- Wei, R.L.; Mattes, M.D.; Yu, J.; Thrasher, A.; Shu, H.-K.; Paganetti, H.; De Los Santos, J.; Koontz, B.; Abraham, C.; Balboni, T. Attitudes of radiation oncologists toward palliative and supportive care in the United States: Report on national membership survey by the American Society for Radiation Oncology (ASTRO). Pract. Radiat. Oncol. 2017, 7, 113–119. [Google Scholar] [CrossRef]
- Arcangeli, G.; Pinnarò, P.; Rambone, R.; Giannarelli, D.; Benassi, M. A phase III randomized study on the sequencing of radiotherapy and chemotherapy in the conservative management of early-stage breast cancer. Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 61–167. [Google Scholar] [CrossRef]
- Sharma, K.; Bahadur, A.; Mohanta, P.; Singh, K.; Rathi, A. Palliative treatment of painful bone metastases: Does fractionation matter? Indian J. Palliat. Care 2008, 14, 7. [Google Scholar] [CrossRef]
- Harstell, W.F.; Santosh, Y. Palliation of bone metastases. In Principles and Practice of Radiotion Oncology, Volume 1.; Halperin, E., Perez, C., Brady, L., Eds.; Lippincott Williams and Wilkin: Philadelphia, PA, USA, 2013; pp. 1778–1791. [Google Scholar]
- Johnstone, C.; Lutz, S.T. External beam radiotherapy and bone metastases. Ann. Palliat. Med. 2014, 3, 114–122. [Google Scholar]
- Lutz, S. The Role of radiation therapy in controlling painful bone metastases. Curr. Pain Headache Rep. 2012, 16, 300–306. [Google Scholar] [CrossRef]
- Goblirsch, M.J.; Zwolak, P.P.; Clohisy, D.R. Biology of Bone Cancer Pain. Clin. Cancer Res. 2006, 12, 6231s–6235s. [Google Scholar] [CrossRef]
- Needham, P.R.; Mithal, N.P.; Hoskin, P.J. Radiotherapy for bone pain. J. R. Soc. Med. 1994, 87, 503–505. [Google Scholar]
- Goblirsch, M.; Mathews, W.; Lynch, C.; Alaei, P.; Gerbi, B.J.; Mantyh, P.W.; Clohisy, D.R. Radiation treatment decreases bone cancer pain, osteolysis and tumor size. Radiat. Res. 2004, 161, 228–234. [Google Scholar] [CrossRef]
- Wu, J.S.Y.; Wong, R.K.S.; Lloyd, N.S.; Johnston, M.; Bezjak, A.; Whelan, T. Radiotherapy fractionation for the palliation of uncomplicated painful bone metastases—An evidence-based practice guideline. BMC Cancer 2004, 4, 71. [Google Scholar] [CrossRef]
- Sprave, T.; Verma, V.; Förster, R.; Schlampp, I.; Hees, K.; Bruckner, T.; Bostel, T.; El Shafie, R.A.; Welzel, T.; Nicolay, N.H.; et al. Bone density and pain response following intensity-modulated radiotherapy versus three-dimensional conformal radiotherapy for vertebral metastases—Secondary results of a randomized trial. Radiat. Oncol. 2018, 13, 212. [Google Scholar] [CrossRef]
- Koswig, S.; Budach, V. Remineralization and pain relief in bone metastases after after different radiotherapy fractions (10 times 3 Gy vs. 1 time 8 Gy). A prospective study. Strahlenther. Onkol. 1999, 175, 500–508. [Google Scholar] [CrossRef]
- Hoskin, P.J.; Stratford, M.R.L.; Folkes, L.K.; Regan, J.; Yarnold, J.R. Effect of local radiotherapy for bone pain on urinary markers of osteoclast activity. Lancet 2000, 355, 1428–1429. [Google Scholar] [CrossRef]
- Chow, E.; DeAngelis, C.; Chen, B.E.; Azad, A.; Meyer, R.M.; Wilson, C.; Kerba, M.; Bezjak, A.; Wilson, P.; Nabid, A.; et al. Effect of re-irradiation for painful bone metastases on urinary markers of osteoclast activity (NCIC CTG SC.20U). Radiother. Oncol. 2015, 115, 141–148. [Google Scholar] [CrossRef]
- Sapkaroski, D.; Osborne, C.; Knight, K.A. A review of stereotactic body radiotherapy—Is volumetric modulated arc therapy the answer? J. Med. Radiat. Sci. 2015, 62, 142–151. [Google Scholar] [CrossRef]
- Falkmer, U.; Järhult, J.; Wersäll, P.; Cavallin-Ståhl, E. A systematic overview of radiation therapy effects in skeletal metastases. Acta Oncol. (Madr). 2003, 42, 620–633. [Google Scholar] [CrossRef]
- Yarnold, J.R. 8 Gy single fraction radiotherapy for the treatment of metastatic skeletal pain: Randomised comparison with a multifraction schedule over 12 months of patient follow-up. Radiother. Oncol. 1999, 52, 111–121. [Google Scholar] [CrossRef]
- Foro Arnalot, P.; Fontanals, A.V.; Galcerán, J.C.; Lynd, F.; Latiesas, X.S.; de Dios, N.R.; Castillejo, A.R.; Bassols, M.L.; Galán, J.L.; Conejo, I.M.; et al. Randomized clinical trial with two palliative radiotherapy regimens in painful bone metastases: 30 Gy in 10 fractions compared with 8 Gy in single fraction. Radiother. Oncol. 2008, 89, 150–155. [Google Scholar] [CrossRef]
- Steenland, E.; Leer, J.; Van Houwelingen, H.; Post, W.J.; Van den Hout, W.B.; Kievit, J.; De Haes, H.; Martijn, H.; Oei, B.; Vonk, E.; et al. The effect of a single fraction compared to multiple fractions on painful bone metastases: A global analysis of the Dutch Bone Metastasis Study. Radiother. Oncol. 1999, 52, 101–109. [Google Scholar] [CrossRef]
- Nielsen, O.S.; Bentzen, S.M.; Sandberg, E.; Gadeberg, C.C.; Timothy, A.R. Randomized trial of single dose versus fractionated palliative radiotherapy of bone metastases. Radiother. Oncol. 1998, 47, 233–240. [Google Scholar] [CrossRef]
- Gaze, M.N.; Kelly, C.G.; Kerr, G.R.; Cull, A.; Cowie, V.J.; Gregor, A.; Howard, G.C.; Rodger, A. Pain relief and quality of life following radiotherapy for bone metastases: a randomised trial of two fractionation schedules. Radiother. Oncol. 1997, 45, 109–116. [Google Scholar] [CrossRef]
- Nongkynrih, A.; Dhull, A.K.; Kaushal, V.; Atri, R.; Dhankhar, R.; Kamboj, K. Comparison of Single Versus Multifraction Radiotherapy in Palliation of Painful Bone Metastases. World J. Oncol. 2018, 9, 91–95. [Google Scholar] [CrossRef]
- Loblaw, D.A.; Wu, J.S.; Kirkbride, P.; Panzarella, T.; Smith, K.; Aslanidis, J.; Warde, P. Pain flare in patients with bone metastases after palliative radiotherapy—A nested randomized control trial. Support. Care Cancer 2007, 15, 451–455. [Google Scholar] [CrossRef]
- Price, P.; Hoskin, P.J.; Easton, D.; Austin, D.; Palmer, S.G.; Yarnold, J.R. Prospective randomised trial of single and multifraction radiotherapy schedules in the treatment of painful bony metastases. Radiother. Oncol. 1986, 6, 247–255. [Google Scholar] [CrossRef]
- Cole, D.J. A randomized trial of a single treatment versus conventional fractionation in the palliative radiotherapy of painful bone metastases. Clin. Oncol. 1989, 1, 59–62. [Google Scholar] [CrossRef]
- Roos, D.E.; Turner, S.L.; O’Brien, P.C.; Smith, J.G.; Spry, N.A.; Burmeister, B.H.; Hoskin, P.J.; Ball, D.L. Randomized trial of 8 Gy in 1 versus 20 Gy in 5 fractions of radiotherapy for neuropathic pain due to bone metastases (Trans-Tasman Radiation Oncology Group, TROG 96.05). Radiother. Oncol. 2005, 75, 54–63. [Google Scholar] [CrossRef]
- Harstell, W.F.; Scott, C.B.; Bruner, D.W.; Scarantino, C.W.; Ivker, R.A.; Roach, M.; Suh, J.H.; Demas, W.F.; Movsas, B.; Petersen, I.A.; et al. Randomized trial of short- versus long-course radiotherapy for palliation of painful bone metastases. J. Natl. Cancer Inst. 2005, 97, 798–804. [Google Scholar]
- Fairchild, A.; Barnes, E.; Ghosh, S.; Ben-Josef, E.; Roos, D.; Hartsell, W.; Holt, T.; Wu, J.; Janjan, N.; Chow, E. International Patterns of Practice in Palliative Radiotherapy for Painful Bone Metastases: Evidence-Based Practice? Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 1501–1510. [Google Scholar] [CrossRef]
- Fischer-Valuck, B.W.; Baumann, B.C.; Apicelli, A.; Rao, Y.J.; Roach, M.; Daly, M.; Dans, M.C.; White, P.; Contreras, J.; Henke, L.; et al. Palliative radiation therapy (RT) for prostate cancer patients with bone metastases at diagnosis: A hospital-based analysis of patterns of care, RT fractionation scheme, and overall survival. Cancer Med. 2018, 7, 4240–4250. [Google Scholar] [CrossRef]
- Kachnic, L.; Berk, L. Palliative Single-Fraction Radiation Therapy: How Much More Evidence Is Needed? JNCI 2005, 97, 786–788. [Google Scholar] [CrossRef]
- Chow, E.; van der Linden, Y.M.; Roos, D.; Hartsell, W.F.; Hoskin, P.; Wu, J.S.Y.; Brundage, M.D.; Nabid, A.; Tissing-Tan, C.J.A.; Oei, B.; et al. Single versus multiple fractions of repeat radiation for painful bone metastases: A randomised, controlled, non-inferiority trial. Lancet Oncol. 2014, 15, 164–171. [Google Scholar] [CrossRef]
- Van den Hout, W.B.; van der Linden, Y.M.; Steenland, E.; Wiggenraad, R.G.J.; Kievit, J.; de Haes, H.; Leer, J.W.H. Single- versus multiple-fraction radiotherapy in patients with painful bone metastases: Cost-utility analysis based on a randomized trial. J. Natl. Cancer Inst. 2003, 95, 222–229. [Google Scholar] [CrossRef]
- Konski, A.; James, J.; Hartsell, W.; Leibenhaut, M.H.; Janjan, N.; Curran, W.; Roach, M.; Watkins-Bruner, D. Economic analysis of Radiation Therapy Oncology Group 97-14: Multiple versus single fraction radiation treatment of patients with bone metastases. Am. J. Clin. Oncol. Cancer Clin. Trials 2009, 32, 423–428. [Google Scholar] [CrossRef]
- Collinson, L.; Kvizhinadze, G.; Nair, N.; McLeod, M.; Blakely, T. Economic evaluation of single-fraction versus multiple-fraction palliative radiotherapy for painful bone metastases in breast, lung and prostate cancer. J. Med. Imaging Radiat. Oncol. 2016, 60, 650–660. [Google Scholar] [CrossRef]
- Bekelman, J.E.; Epstein, A.J.; Emanuel, E.J. Single- vs Multiple-Fraction Radiotherapy for Bone Metastases From Prostate Cancer. JAMA 2013, 310, 1501. [Google Scholar] [CrossRef]
- Schreiber, D.; Safdieh, J.; Becker, D.J.; Schwartz, D. Patterns of care and survival outcomes of palliative radiation for prostate cancer with bone metastases: Comparison of ≤5 fractions to ≥10 fractions. Ann. Palliat. Med. 2017, 6, 55–65. [Google Scholar] [CrossRef]
- Olson, R.A.; Tiwana, M.S.; Barnes, M.; Kiraly, A.; Beecham, K.; Miller, S.; Hoegler, D.; Olivotto, I. Use of single-versus multiple-fraction palliative radiation therapy for bone metastases: Population-based analysis of 16,898 courses in a Canadian Province. Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 1092–1099. [Google Scholar] [CrossRef]
- Olson, R.A.; Tiwana, M.; Barnes, M.; Cai, E.; McGahan, C.; Roden, K.; Yurkowski, E.; Gentles, Q.; French, J.; Halperin, R.; et al. Impact of using audit data to improve the evidence-based use of single-fraction radiation therapy for bone metastases in British Columbia presented at the Canadian Association of Radiation Oncology 2014 Annual Meeting, Aug 25–28, 2014, St. John’s, Newfoun. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 40–47. [Google Scholar] [CrossRef]
- Morgan, K.J.; Anghelescu, D.L. A review of adult and pediatric neuropathic pain assessment tools. Clin. J. Pain 2017, 33, 844–852. [Google Scholar] [CrossRef]
- Roberto, A.; Deandrea, S.; Greco, M.T.; Corli, O.; Negri, E.; Pizzuto, M.; Ruggeri, F. Prevalence of Neuropathic Pain in Cancer Patients: Pooled Estimates from a Systematic Review of Published Literature and Results from a Survey Conducted in 50 Italian Palliative Care Centers. J. Pain Symptom Manag. 2016, 51, 1091–1102e4. [Google Scholar] [CrossRef]
- Vadalouca, A.; Raptis, E.; Moka, E.; Zis, P.; Sykioti, P.; Siafaka, I. Pharmacological Treatment of Neuropathic Cancer Pain: A Comprehensive Review of the Current Literature. Pain Pract. 2012, 12, 219–251. [Google Scholar] [CrossRef]
- Fallon, M.T.; Colvin, L. Neuropathic pain in cancer. Br. J. Anaesth. 2013, 111, 105–111. [Google Scholar] [CrossRef]
- Saito, T.; Tomitaka, E.; Toya, R.; Matsuyama, T.; Ninomura, S.; Watakabe, T.; Oya, N. A neuropathic pain component as a predictor of improvement in pain interference after radiotherapy for painful tumors: A secondary analysis of a prospective observational study. Clin. Transl. Radiat. Oncol. 2018, 12, 34–39. [Google Scholar] [CrossRef]
- Lu, C.-W.; Shao, J.; Wu, Y.-G.; Wang, C.; Wu, J.-H.; Lv, R.-X.; Ding, M.-C.; Shi, Z.-C.; Mao, N.-F. Which Combination Treatment Is Better for Spinal Metastasis. Am. J. Ther. 2019, 26, e38–e44. [Google Scholar] [CrossRef]
- Patchell, R.A.; Tibbs, P.A.; Regine, W.F.; Payne, R.; Saris, S.; Kryscio, R.J.; Mohiuddin, M.; Young, B. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: A randomised trial. Lancet 2005, 366, 643–648. [Google Scholar] [CrossRef]
- Maranzano, E.; Bellavita, R.; Rossi, R.; De Angelis, V.; Frattegiani, A.; Bagnoli, R.; Mignogna, M.; Beneventi, S.; Lupattelli, M.; Ponticelli, P.; et al. Short-course versus split-course radiotherapy in metastatic spinal cord compression: Results of a phase III, randomized, multicenter trial. J. Clin. Oncol. 2005, 23, 3358–3365. [Google Scholar] [CrossRef]
- Arcangeli, G.; Giovinazzo, G.; Saracino, B.; D’Angelo, L.; Giannarelli, D.; Micheli, A. Radiation therapy in the management of symptomatic bone metastases: The effect of total dose and histology on pain relief and response duration. Int. J. Radiat. Oncol. Biol. Phys. 1998, 42, 1119–1126. [Google Scholar] [CrossRef]
- Lutz, S.; Balboni, T.; Jones, J.; Lo, S.; Petit, J.; Rich, S.E.; Wong, R.; Hahn, C. Palliative radiation therapy for bone metastases: Update of an ASTRO Evidence-Based Guideline. Pract. Radiat. Oncol. 2017, 7, 4–12. [Google Scholar] [CrossRef]
- Pal, S.; Dutta, S.; Adhikary, S.; Bhattacharya, B.; Ghosh, B.; Patra, N. Hemi body irradiation: An economical way of palliation of pain in bone metastasis in advanced cancer. South Asian J. Cancer 2014, 3, 28. [Google Scholar] [CrossRef]
- Delinikolas, P.; Patatoukas, G.; Kouloulias, V.; Dilvoi, M.; Plousi, A.; Efstathopoulos, E.; Platoni, K. A novel Hemi-Body Irradiation technique using electron beams (HBIe−). Phys. Medica 2018, 46, 16–24. [Google Scholar] [CrossRef]
- Palma, D.A.; Salama, J.K.; Lo, S.S.; Senan, S.; Treasure, T.; Govindan, R.; Weichselbaum, R. The oligometastatic state-separating truth from wishful thinking. Nat. Rev. Clin. Oncol. 2014, 11, 549–557. [Google Scholar] [CrossRef]
- Osborn, V.W.; Lee, A.; Yamada, Y. Stereotactic Body Radiation Therapy for Spinal Malignancies. Technol. Cancer Res. Treat. 2018, 17. [Google Scholar] [CrossRef]
- Chang, U.K.; Cho, W.I.; Kim, M.S.; Cho, C.K.; Lee, D.H.; Rhee, C.H. Local tumor control after retreatment of spinal metastasis using stereotactic body radiotherapy; comparison with initial treatment group. Acta Oncol. (Madr). 2012, 51, 589–595. [Google Scholar] [CrossRef]
- Ryu, S.; Jin, R.; Jin, J.Y.; Chen, Q.; Rock, J.; Anderson, J.; Movsas, B. Pain Control by Image-Guided Radiosurgery for Solitary Spinal Metastasis. J. Pain Symptom Manag. 2008, 35, 292–298. [Google Scholar] [CrossRef]
- Sprave, T.; Verma, V.; Förster, R.; Schlampp, I.; Bruckner, T.; Bostel, T.; Welte, S.E.; Tonndorf-Martini, E.; Nicolay, N.H.; Debus, J.; et al. Randomized phase II trial evaluating pain response in patients with spinal metastases following stereotactic body radiotherapy versus three-dimensional conformal radiotherapy. Radiother. Oncol. 2018, 128, 274–282. [Google Scholar] [CrossRef]
- Nguyen, Q.N.; Shiu, A.S.; Rhines, L.D.; Wang, H.; Allen, P.K.; Wang, X.S.; Chang, E.L. Management of Spinal Metastases From Renal Cell Carcinoma Using Stereotactic Body Radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 1185–1192. [Google Scholar] [CrossRef]
- Gibbs, I.C.; Kamnerdsupaphon, P.; Ryu, M.R.; Dodd, R.; Kiernan, M.; Chang, S.D.; Adler, J.R. Image-guided robotic radiosurgery for spinal metastases. Radiother. Oncol. 2007, 82, 185–190. [Google Scholar] [CrossRef]
- Pan, H.; Simpson, D.R.; Mell, L.K.; Mundt, A.J.; Lawson, J.D. A survey of stereotactic body radiotherapy use in the United States. Cancer 2011, 117, 4566–4572. [Google Scholar] [CrossRef] [PubMed]
- Sahgal, A.; Ma, L.; Weinberg, V.; Gibbs, I.C.; Chao, S.; Chang, U.K.; Werner-Wasik, M.; Angelov, L.; Chang, E.L.; Sohn, M.J.; et al. Reirradiation human spinal cord tolerance for stereotactic body radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Keum, K.C.; Cha, J.H.; Kim, J.H.; Seong, J.S.; Lee, C.G.; Nam, K.C.; Koom, W.S. Stereotactic Body Radiotherapy with Helical Tomotherapy for Pain Palliation in Spine Metastasis. Technol. Cancer Res. Treat. 2013, 12, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.; Pugh, S.L.; Gerszten, P.C.; Yin, F.F.; Timmerman, R.D.; Hitchcock, Y.J.; Movsas, B.; Kanner, A.A.; Berk, L.B.; Followill, D.S.; et al. RTOG 0631 phase 2/3 study of image guided stereotactic radiosurgery for localized (1-3) spine metastases: Phase 2 results. Pract. Radiat. Oncol. 2014, 4, 76–81. [Google Scholar] [CrossRef] [PubMed]
- McGee, H.M.; Carpenter, T.; Ozbek, U.; Kirkwood, K.; Tzu-Chi, T.; Blacksburg, S.; Germano, I.M.; Green, S.; Buckstein, M. Analysis of Local Control and Pain Control Following Spine Stereotactic Radiosurgery Reveals Inferior Outcomes for Hepatocellular Carcinoma compared to other Radioresistant Histologies. Pract. Radiat. Oncol. 2018. [Google Scholar]
- Ryu, S.; Jin, J.-Y.; Jin, R.; Rock, J.; Ajlouni, M.; Movsas, B.; Rosenblum, M.; Kim, J.H. Partial volume tolerance of the spinal cord and complications of single-dose radiosurgery. Cancer 2007, 109, 628–636. [Google Scholar] [CrossRef]
- Guckenberger, M.; Mantel, F.; Gerszten, P.C.; Flickinger, J.C.; Sahgal, A.; Létourneau, D.; Grills, I.S.; Jawad, M.; Fahim, D.K.; Shin, J.H.; et al. Safety and efficacy of stereotactic body radiotherapy as primary treatment for vertebral metastases: A multi-institutional analysis. Radiat. Oncol. 2014, 9, 226. [Google Scholar] [CrossRef]
- Nieder, C.; Grosu, A.L.; Andratschke, N.H.; Molls, M. Update of human spinal cord reirradiation tolerance based on additional data from 38 patients. Int. J. Radiat. Oncol. Biol. Phys. 2006, 66, 1446–1449. [Google Scholar] [CrossRef] [PubMed]
- Mantel, F.; Flentje, M.; Guckenberger, M. Stereotactic body radiation therapy in the re-irradiation situation—A review. Radiat. Oncol. 2013, 8, 7. [Google Scholar] [CrossRef]
- Myrehaug, S.; Sahgal, A.; Hayashi, M.; Levivier, M.; Ma, L.; Martinez, R.; Paddick, I.; Régis, J.; Ryu, S.; Slotman, B.; et al. Reirradiation spine stereotactic body radiation therapy for spinal metastases: systematic review. J. Neurosurg. Spine 2017, 27, 428–435. [Google Scholar] [CrossRef]
- Choi, C.Y.H.; Adler, J.R.; Gibbs, I.C.; Chang, S.D.; Jackson, P.S.; Minn, A.Y.; Lieberson, R.E.; Soltys, S.G. Stereotactic radiosurgery for treatment of spinal metastases recurring in close proximity to previously irradiated spinal cord. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.K.; Wang, X.-S.; Shiu, A.S.; Allen, P.; Yang, J.; McAleer, M.F.; Azeem, S.; Rhines, L.D.; Chang, E.L. Prospective evaluation of spinal reirradiation by using stereotactic body radiation therapy. Cancer 2011, 117, 3509–3516. [Google Scholar] [CrossRef] [PubMed]
- Damast, S.; Wright, J.; Bilsky, M.; Hsu, M.; Zhang, Z.; Lovelock, M.; Cox, B.; Zatcky, J.; Yamada, Y. Impact of dose on local failure rates after image-guided reirradiation of recurrent paraspinal metastases. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, A.; Floyd, S.; Wong, E.; Jeyapalan, S.; Groff, M.; Kasper, E. Stereotactic body radiotherapy reirradiation for recurrent epidural spinal metastases. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 1500–1505. [Google Scholar] [CrossRef]
- Hashmi, A.; Guckenberger, M.; Kersh, R.; Gerszten, P.C.; Mantel, F.; Grills, I.S.; Flickinger, J.C.; Shin, J.H.; Fahim, D.K.; Winey, B.; et al. Re-irradiation stereotactic body radiotherapy for spinal metastases: a multi-institutional outcome analysis. J. Neurosurg. Spine 2016, 25, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, J.P.; van der Kogel, A.J.; Schultheiss, T.E. Radiation Dose-Volume Effects in the Spinal Cord. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S42–S49. [Google Scholar] [CrossRef] [PubMed]
- Rades, D.; Stalpers, L.J.A.; Veninga, T.; Hoskin, P.J. Spinal reirradiation after short-course RT for metastatic spinal cord compression. Int. J. Radiat. Oncol. Biol. Phys. 2005, 63, 872–875. [Google Scholar] [CrossRef] [PubMed]
- Nieder, C.; Grosu, A.L.; Andratschke, N.H.; Molls, M. Proposal of human spinal cord reirradiation dose based on collection of data from 40 patients. Int. J. Radiat. Oncol. Biol. Phys. 2005, 61, 851–855. [Google Scholar] [CrossRef] [PubMed]
- Laufer, I.; Rubin, D.G.; Lis, E.; Cox, B.W.; Stubblefield, M.D.; Yamada, Y.; Bilsky, M.H. The NOMS Framework: Approach to the Treatment of Spinal Metastatic Tumors. Oncologist 2013, 18, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Zuckerman, S.L.; Lim, J.; Yamada, Y.; Bilsky, M.H.; Laufer, I. Brachytherapy in Spinal Tumors: A Systematic Review. World Neurosurg. 2018, 118, e235–e244. [Google Scholar] [CrossRef]
- Cardoso, E.R.; Ashamalla, H.; Weng, L.; Mokhtar, B.; Ali, S.; Macedon, M.; Guirguis, A. Percutaneous tumor curettage and interstitial delivery of samarium-153 coupled with kyphoplasty for treatment of vertebral metastases. J. Neurosurg. Spine 2009, 10, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Tan, J.; Zhao, R.; Wang, J.; Sun, H.; Wang, X.; Xu, L.; Jiang, H.; Zhang, J. Clinical Investigations on the Spinal Osteoblastic Metastasis Treated by Combination of Percutaneous Vertebroplasty and 125I Seeds Implantation Versus Radiotherapy. Cancer Biother. Radiopharm. 2013, 28, 58–64. [Google Scholar] [CrossRef]
- Cao, Q.; Wang, H.; Meng, N.; Jiang, Y.; Jiang, P.; Gao, Y.; Tian, S.; Liu, C.; Yang, R.; Wang, J.; et al. CT-guidance interstitial 125Iodine seed brachytherapy as a salvage therapy for recurrent spinal primary tumors. Radiat. Oncol. 2014, 9, 301. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Li, J.; Wang, Z.; Liu, B.; Han, D.; Wang, P. A preliminary comparative clinical study of vertebroplasty with multineedle or single-needle interstitial implantation of 125I seeds in the treatment of osteolytic metastatic vertebral tumors. J. Neurosurg. Spine 2014, 20, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Shi, G.; Meng, X. Clinical curative effect of percutaneous vertebroplasty combined with 125I-seed implantation in treating spinal metastatic tumor. Pak. J. Pharm. Sci. 2015, 28, 1039–1042. [Google Scholar] [PubMed]
- Qian, J.; Bao, Z.; Zou, J.; Yang, H. Effect of pedicle fixation combined with 125I seed implantation for metastatic thoracolumbar tumors. J. Pain Res. 2016, 9, 271–278. [Google Scholar] [PubMed]
- Huang, H.; Xu, S.; Du, Z.; Li, F.; Wang, L. Treatment of metastatic thoracolumbar tumors by percutaneous vertebroplasty combined with interstitial implantation of 125I seeds. Zhonghua Zhong Liu Za Zhi 2014, 36, 228–231. [Google Scholar]
- Chow, E.; Ling, A.; Davis, L.; Panzarella, T.; Danjoux, C. Pain flare following external beam radiotherapy and meaningful change in pain scores in the treatment of bone metastases. Radiother. Oncol. 2005, 75, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Hird, A.; Chow, E.; Zhang, L.; Wong, R.; Wu, J.; Sinclair, E.; Danjoux, C.; Tsao, M.; Barnes, E.; Loblaw, A. Determining the Incidence of Pain Flare Following Palliative Radiotherapy for Symptomatic Bone Metastases: Results From Three Canadian Cancer Centers. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 193–197. [Google Scholar] [CrossRef]
- McDonald, R.; Chow, E.; Rowbottom, L.; De Angelis., C.; Soliman, H. Incidence of pain flare in radiation treatment of bone metastases: A literature review. J. Bone Oncol. 2014, 3, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Iturriaga, A.; Cacicedo, J.; Navarro, A.; Morillo, V.; Willisch, P.; Carvajal, C.; Hortelano, E.; Lopez-Guerra, J.L.; Illescas, A.; Casquero, F.; et al. Incidence of pain flare following palliative radiotherapy for symptomatic bone metastases: Multicenter prospective observational study. BMC Palliat. Care 2015, 14, 48. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.-B.; Yan, W.-L.; Dai, J.-C.; Xu, F.; Yuan, Q.; Shi, H.-H. Strontium-89: A desirable therapeutic for bone metastases of prostate cancer. Zhonghua Nan Ke Xue 2008, 14, 819–822. (In Chinese) [Google Scholar] [PubMed]
- Ogawa, K.; Washiyama, K. Bone Target Radiotracers for Palliative Therapy of Bone Metastases. Curr. Med. Chem. 2012, 19, 3290–3300. [Google Scholar] [CrossRef] [PubMed]
- Serafini, A.N. Therapy of metastatic bone pain. J. Nucl. Med. 2001, 42, 895–906. [Google Scholar] [PubMed]
- Finlay, I.G.; Mason, M.D.; Shelley, M. Radioisotopes for the palliation of metastatic bone cancer: A systematic review. Lancet Oncol. 2005, 6, 392–400. [Google Scholar] [CrossRef]
- Pandit-Taskar, N.; Batraki, M.; Divgi, C.R. Radiopharmaceutical therapy for palliation of bone pain from osseous metastases. J. Nucl. Med. 2004, 45, 1358–1365. [Google Scholar] [PubMed]
- Paes, F.M.; Serafini, A.N. Systemic Metabolic Radiopharmaceutical Therapy in the Treatment of Metastatic Bone Pain. Semin. Nucl. Med. 2010, 40, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, E.B.; Taylor, A.T. EANM procedure guidelines for treatment of refractory metastatic bone pain. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, BP7–BP11. [Google Scholar]
- Bodei, L.; Lam, M.; Chiesa, C.; Flux, G.; Brans, B.; Chiti, A.; Giammarile, F. EANM procedure guideline for treatment of refractory metastatic bone pain. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 1934–1940. [Google Scholar] [CrossRef]
- Choi, J.Y. Treatment of Bone Metastasis with Bone-Targeting Radiopharmaceuticals. Nucl. Med. Mol. Imaging 2018, 52, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Fettich, J.; Padhy, A.; Nair, N.; Morales, R.; Tanumihardja, M.; Riccabonna, G.; Nair, G. Comparative clinical efficacy and safety of Phosphorus-32 and Strontium-89 in the palliative treatment of metastatic bone pain: Results of an IAEA Coordinated Research Project. World J. Nucl. Med. 2003, 34, 226–231. [Google Scholar]
- Sciuto, R.; Festa, A.; Pasqualoni, R.; Semprebene, A.; Rea, S.; Bergomi, S.; Maini, C.L. Metastatic bone pain palliation with 89-Sr and 186-Re-HEDP in breast cancer patients. Breast Cancer Res. Treat. 2001, 66, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.L.; Gruenewald, S.; Spry, N.; Gebski, V. Less pain does equal better quality of life following Strontium-89 therapy for metastatic prostate cancer. Br. J. Cancer 2001, 84, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Dafermou, A.; Colamussi, P.; Giganti, M.; Cittanti, C.; Bestagno, M.; Piffanelli, A. A multicentre observational study of radionuclide therapy in patients with painful bone metastases of prostate cancer. Eur. J. Nucl. Med. 2001, 28, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Ashayeri, E.; Omogbehin, A.; Sridhar, R.; Shankar, R.A. Strontium 89 in the treatment of pain due to diffuse osseous metastases: a university hospital experience. J. Natl. Med. Assoc. 2002, 94, 706–711. [Google Scholar]
- Baczyk, M.; Milecki, P.; Baczyk, E.; Sowiński, J. The effectivness of strontium 89 in palliative therapy of painful prostate cancer bone metastases. Ortop. Traumatol. Rehabil. 2003, 5, 364–368. [Google Scholar] [PubMed]
- Liepe, K.; Kotzerke, J. A comparative study of Re-HEDP, Re-HEDP, Sm-EDTMP and Sr in the treatment of painful skeletal metastases. Nucl. Med. Commun. 2007, 28, 623–630. [Google Scholar] [CrossRef]
- Zenda, S.; Nakagami, Y.; Toshima, M.; Arahira, S.; Kawashima, M.; Matsumoto, Y.; Kinoshita, H.; Satake, M.; Akimoto, T. Strontium-89 (Sr-89) chloride in the treatment of various cancer patients with multiple bone metastases. Int. J. Clin. Oncol. 2014, 19, 739–743. [Google Scholar] [CrossRef]
- Laing, A.H.; Ackery, D.M.; Bayly, R.J.; Buchanan, R.B.; Lewington, V.J.; McEwan, A.J.B.; Macleod, P.M.; Zivanovic, M.A. Strontium-89 chloride for pain palliation in prostatic skeletal malignancy. Br. J. Radiol. 1991, 64, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, E.B.; Williams, C. Strontium-89 therapy for the pain of osseous metastases. J. Nucl. Med. 1985, 26, 345–348. [Google Scholar] [PubMed]
- Mertens, W.C.; Stitt, L.; Porter, A.T. Strontium 89 therapy and relief of pain in patients with prostatic carcinoma metastatic to bone: A dose response relationship? Am. J. Clin. Oncol. Cancer Clin. Trials 1993, 16, 238–242. [Google Scholar] [CrossRef]
- Dolezal, J. Systemic radionuclide therapy with Samarium-153-EDTMP for painful bone metastases. Nucl. Med. Rev. Cent. East. Eur. 2000, 3, 161–163. [Google Scholar] [PubMed]
- Wang, R.F.; Zhang, C.L.; Zhu, S.L.; Zhu, M. A comparative study of samarium-153-ethylenediaminetetramethylene phosphonic acid with pamidronate disodium in the treatment of patients with painful metastatic bone cancer. Med. Princ. Pract. 2003, 12, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Sapienza, M.T.; Ono, C.R.; Guimarães, M.I.C.; Watanabe, T.; Costa, P.A.; Buchpiguel, C.A. Retrospective evaluation of bone pain palliation after samarium-153-EDTMP therapy. Rev. Hosp. Clin. Fac. Med. Sao Paulo 2004, 59, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Sá de Camargo Etchebehere, E.C.; Cunha Pereira Neto, C.A.; Lopes de Lima, M.C.; de Oliveira Santos, A.; Ramos, C.D.; Silva, C.M.; Camargo, E.E. Treatment of bone pain secondary to metastases using samarium-153-EDTMP. Sao Paulo Med. J. 2004, 122, 208–212. [Google Scholar] [CrossRef]
- Sartor, O.; Reid, R.H.; Hoskin, P.J.; Quick, D.P.; Ell, P.J.; Coleman, R.E.; Kotler, J.A.; Freeman, L.M.; Olivier, P. Samarium-153-lexidronam complex for treatment of painful bone metastases in hormone-refractory prostate cancer. Urology 2004, 63, 940–945. [Google Scholar] [CrossRef]
- Tripathi, M.; Singhal, T.; Chandrasekhar, N.; Kumar, P.; Bal, C.; Jhulka, P.K.; Bandopadhyaya, G.; Malhotra, A. Samarium-153 ethylenediamine tetramethylene phosphonate therapy for bone pain palliation in skeletal metastases. Indian J. Cancer 2006, 43, 86–92. [Google Scholar] [CrossRef]
- Ripamonti, C.; Fagnoni, E.; Campa, T.; Seregni, E.; Maccauro, M.; Bombardieri, E. Incident pain and analgesic consumption decrease after samarium infusion: A pilot study. Support. Care Cancer 2007, 15, 339–342. [Google Scholar] [CrossRef]
- Dolezal, J.; Vizda, J.; Odrazka, K. Prospective evaluation of samarium-153-EDTMP radionuclide treatment for bone metastases in patients with hormone-refractory prostate cancer. Urol. Int. 2007, 78, 50–57. [Google Scholar] [CrossRef]
- Resche, I.; Chatal, J.F.; Pecking, A.; Ell, P.; Duchesne, G.; Rubens, R.; Fogelman, I.; Houston, S.; Fauser, A.; Fischer, M.; et al. A dose-controlled study of 153Sm-ethylenediaminetetramethylenephosphonate (EDTMP) in the treatment of patients with painful bone metastases. Eur. J. Cancer 1997, 33, 1583–1591. [Google Scholar] [CrossRef]
- Serafini, A.N.; Houston, S.J.; Resche, I.; Quick, D.P.; Grund, F.M.; Ell, P.J.; Bertrand, A.; Ahmann, F.R.; Orihuela, E.; Reid, R.H.; et al. Palliation of pain associated with metastatic bone cancer using samarium-153 lexidronam: A double-blind placebo-controlled clinical trial. J. Clin. Oncol. 1998, 16, 1574–1581. [Google Scholar] [CrossRef]
- Tian, J.H.; Zhang, J.M.; Hou, Q.T.; Oyang, Q.H.; Wang, J.M.; Luan, Z.S.; Chuan, L.; He, Y.J. Multicentre trial on the efficacy and toxicity of single-dose samarium-153-ethylene diamine tetramethylene phosphonate as a palliative treatment for painful skeletal metastases in China. Eur. J. Nucl. Med. 1999, 26, 2–7. [Google Scholar] [CrossRef]
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fosså, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha Emitter Radium-223 and Survival in Metastatic Prostate Cancer. N. Engl. J. Med. 2013, 369, 213–223. [Google Scholar] [CrossRef]
- Parker, C.; Zhan, L.; Cislo, P.; Reuning-Scherer, J.; Vogelzang, N.J.; Nilsson, S.; Sartor, O.; O’Sullivan, J.M.; Coleman, R.E. Effect of radium-223 dichloride (Ra-223) on hospitalisation: An analysis from the phase 3 randomised Alpharadin in Symptomatic Prostate Cancer Patients (ALSYMPCA) trial. Eur. J. Cancer 2017, 71, 1–6. [Google Scholar] [CrossRef]
- Parker, C.; Heinrich, D.; O’Sullivan, J.M.; Fossa, S.; Chodacki, A.; Demkow, T.; Cross, A.; Bolstad, B.; Garcia-Vargas, J.; Sartor, O. Sartor Overall survival benefit of radium-223 chloride (Alpharadin) in the treatment of patients with symptomatic bone metastases in Castration-resistant Prostate Cancer (CRPC): A phase III randomized trial (ALSYMPCA). Eur. J. Cancer 2011, 47, 3. [Google Scholar] [CrossRef]
- Nilsson, S.; Sartor, A.O.; Bruland, O.S.; Fang, F.; Aksnes, A.-K.; Parker, C. Pain analyses from the phase III randomized ALSYMPCA study with radium-223 dichloride (Ra-223) in castration-resistant prostate cancer (CRPC) patients with bone metastases. J. Clin. Oncol. 2013, 31, 5038. [Google Scholar] [CrossRef]
- Quirijnen, J.M.; Han, S.H.; Zonnenberg, B.A.; Klerk, J.M.; het Schip, A.D.; Dijk, A.; Kroode, H.F.; Blijham, G.H.; Rijk, P.P. Efficacy of rhenium-186-etidronate in prostate cancer patients with metastatic bone pain. J. Nucl. Med. 1996, 37, 1511–1515. [Google Scholar]
- Maxon, H.R.; Schroder, L.E.; Thomas, S.R.; Hertzberg, V.S.; Deutsch, E.A.; Scher, H.I.; Samaratunga, R.C.; Libson, K.F.; Williams, C.C.; Moulton, J.S. Re-186(Sn) HEDP for treatment of painful osseous metastases: initial clinical experience in 20 patients with hormone-resistant prostate cancer. Radiology 1990, 176, 155–159. [Google Scholar] [CrossRef]
- Giannakenas, C.; Kalofonos, H.P.; Apostolopoulos, D.J.; Zarakovitis, J.; Kosmas, C.; Vassilakos, P.J. Preliminary results of the use of Re-186-HEDP for palliation of pain in patients with metastatic bone disease. Am. J. Clin. Oncol. 2000, 23, 83–88. [Google Scholar] [CrossRef]
- Kolesnikov-Gauthier, H.; Carpentier, P.; Depreux, P.; Vennin, P.; Caty, A.; Sulman, C. Evaluation of toxicity and efficacy of 186Re-hydroxyethylidene diphosphonate in patients with painful bone metastases of prostate or breast cancer. J. Nucl. Med. 2000, 41, 1689–1694. [Google Scholar]
- Sciuto, R.; Tofani, A.; Festa, A.; Giannarelli, D.; Pasqualoni, R.; Maini, C.L. Short- and long-term effects of 186Re-1,1-hydroxyethylidene diphosphonate in the treatment of painful bone metastases. J. Nucl. Med. 2000, 41, 647–654. [Google Scholar] [PubMed]
- Paes, F.M.; Ernani, V.; Hosein, P.; Serafini, A.N. Radiopharmaceuticals: When and how to use them to treat metastatic bone pain. J. Support. Oncol. 2011, 9, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Liepe, K.; Kotzerke, J. Internal radiotherapy of painful bone metastases. Methods 2011, 55, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Maxon III, H.R.; Schroder, L.E.; Hertzberg, V.S.; Thomas, S.R.; Englaro, E.E.; Samaratunga, R.; Smith, H.; Moulton, J.S.; Williams, C.C.; Ehrhardt, G.J.; et al. Rhenium-186(Sn)HEDP for treatment of painful osseous metastases: Results of a double-blind crossover comparison with placebo. J. Nucl. Med. 1991, 32, 1877–1881. [Google Scholar]
- Maxon, H.R.; Thomas, S.R.; Hertzberg, V.S.; Schroder, L.E.; Englaro, E.E.; Samaratunga, R.; Scher, H.I.; Moulton, J.S.; Deutsch, E.A.; Deutsch, K.F.; et al. Rhenium-186 hydroxyethylidene diphosphonate for the treatment of painful osseous metastases. Semin. Nucl. Med. 1992, 22, 33–40. [Google Scholar] [CrossRef]
- Han, S.H.; Zonneberg, B.A.; de Klerk, J.M.; Quirijnen, J.M.; van het Schip, A.D.; van Dijk, A.; Blijham, G.H.; van Rijk, P.P. 186Re-etidronate in breast cancer patients with metastatic bone pain. J. Nucl. Med. 1999, 40, 639–642. [Google Scholar]
- Han, S.H.; de Klerk, J.M.H.; Tan, S.; van het Schip, A.D.; Derksen, B.H.; van Dijk, A.; Kruitwagen, C.L.J.J.; Blijham, G.H.; van Rijk, P.P.; Zonnenberg, B.A. The PLACORHEN study: A double-blind, placebo-controlled, randomized radionuclide study with (186)Re-etidronate in hormone-resistant prostate cancer patients with painful bone metastases. Placebo Controlled Rhenium Study. J. Nucl. Med. 2002, 43, 1150–1156. [Google Scholar]
- Fuster, D.; Herranz, R.; Alcover, J.; Mateos, J.J.; Martín, F.; Vidal-Sicart, S.; Pons, F. Treatment of metastatic bone pain with repeated doses of strontium-89 in patients with prostate neoplasm. Rev. Esp. Med. Nucl. 2000, 19, 270–274. [Google Scholar] [CrossRef]
- Sartor, O.; Reid, R.H.; Bushnell, D.L.; Quick, D.P.; Ell, P.J. Safety and efficacy of repeat administration of samarium Sm-153 lexidronam to patients with metastatic bone pain. Cancer 2007, 109, 637–643. [Google Scholar] [CrossRef]
- Palmedo, H.; Guhlke, S.; Bender, H.; Sartor, J.; Schoeneich, G.; Risse, J.; Grünwald, F.; Knapp, F.F.; Biersack, H.J. Dose escalation study with rhenium-188 hydroxyethylidene diphosphonate in prostate cancer patients with osseous metastases. Eur. J. Nucl. Med. 2000, 27, 123–130. [Google Scholar] [CrossRef]
- Liepe, K.; Hliscs, R.; Kropp, J.; Gruning, T.; Runge, R.; Koch, R.; Knapp, F.F.J.; Franke, W.G. Rhenium-188-HEDP in the palliative treatment of bone metastases. Cancer Biother. Radiopharm. 2000, 15, 261–265. [Google Scholar] [CrossRef]
- Palmedo, H.; Manka-Waluch, A.; Albers, P.; Schmidt-Wolf, I.G.H.; Reinhardt, M.; Ezziddin, S.; Joe, A.; Roedel, R.; Fimmers, R.; Knapp, F.F.; et al. Repeated bone-targeted therapy for hormone-refractory prostate carcinoma: Randomized phase II trial with the new, high-energy radiopharmaceutical rhenium-188 hydroxyethylidenediphosphonate. J. Clin. Oncol. 2003, 21, 2869–2875. [Google Scholar] [CrossRef]
- Nilsson, S.; Larsen, R.H.; Fosså, S.D.; Balteskard, L.; Borch, K.W.; Westlin, J.E.; Salberg, G.; Bruland, Ø.S. First clinical experience with α-emitting radium-223 in the treatment of skeletal metastases. Clin. Cancer Res. 2005, 11, 4451–4459. [Google Scholar] [CrossRef]
- Bruland, Ø.S.; Nilsson, S.; Fisher, D.R.; Larsen, R.H. High-linear energy transfer irradiation targeted to skeletal metastases by the α-emitter 223Ra: Adjuvant or alternative to conventional modalities? Clin. Cancer Res. 2006, 12, 6250–6258. [Google Scholar] [CrossRef]
- Nilsson, S.; Franzén, L.; Parker, C.; Tyrrell, C.; Blom, R.; Tennvall, J.; Lennernäs, B.; Petersson, U.; Johannessen, D.C.; Sokal, M.; et al. Bone-targeted radium-223 in symptomatic, hormone-refractory prostate cancer: A randomised, multicentre, placebo-controlled phase II study. Lancet Oncol. 2007, 8, 587–594. [Google Scholar] [CrossRef]
- Coleman, R.; Aksnes, A.K.; Naume, B.; Garcia, C.; Jerusalem, G.; Piccart, M.; Vobecky, N.; Thuresson, M.; Flamen, P. A phase IIa, nonrandomized study of radium-223 dichloride in advanced breast cancer patients with bone-dominant disease. Breast Cancer Res. Treat. 2014, 145, 411–418. [Google Scholar] [CrossRef]
- Prelaj, A.; Rebuzzi, S.E.; Buzzacchino, F.; Pozzi, C.; Ferrara, C.; Frantellizzi, V.; Follacchio, G.A.; Civitelli, L.; De Vincentis, G.; Tomao, S.; et al. Radium-223 in patients with metastatic castration-resistant prostate cancer: Efficacy and safety in clinical practice. Oncol. Lett. 2019, 17, 1467–1476. [Google Scholar] [CrossRef]
- Fairchild, A.; Harris, K.; Barnes, E.; Wong, R.; Lutz, S.; Bezjak, A.; Cheung, P.; Chow, E. Palliative thoracic radiotherapy for lung cancer: A systematic review. J. Clin. Oncol. 2008, 26, 4001–4011. [Google Scholar] [CrossRef]
- Tey, J.; Choo, B.A.; Leong, C.N.; Loy, E.Y.; Wong, L.C.; Lim, K.; Lu, J.J.; Koh, W.Y. Clinical outcome of palliative radiotherapy for locally advanced symptomatic gastric cancer in the modern era. Medicine (Baltimore) 2014, 93, e118. [Google Scholar] [CrossRef]
- Rominger, C.J.; Gelber, R.D.; Gunderson, L.L.; Conner, N. Radiation therapy alone or in combination with chemotherapy in the treatment of residual or inoperable carcinoma of the rectum and rectosigmoid or pelvic recurrence following colorectal surgery. Radiation therapy oncology group study (76-16). Am. J. Clin. Oncol. Cancer Clin. Trials 1985, 8, 118–127. [Google Scholar] [CrossRef]
- Bae, S.H.; Park, W.; Choi, D.H.; Nam, H.; Kang, W.K.; Park, Y.S.; Park, J.O.; Chun, H.K.; Lee, W.Y.; Yun, S.H.; et al. Palliative radiotherapy in patients with a symptomatic pelvic mass of metastatic colorectal cancer. Radiat. Oncol. 2011, 6, 52. [Google Scholar] [CrossRef]
- Rosati, L.M.; Herman, J.M. Role of Stereotactic Body Radiotherapy in the Treatment of Elderly and Poor Performance Status Patients With Pancreatic Cancer. J. Oncol. Pract. 2017, 13, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.P.; Koay, E.J. Current and emerging radiotherapy strategies for pancreatic adenocarcinoma: stereotactic, intensity modulated and particle radiotherapy. Ann. Pancreat. Cancer 2018, 1, 22. [Google Scholar] [CrossRef]
- Ryan, J.F.; Rosati, L.M.; Groot, V.P.; Le, D.T.; Zheng, L.; Laheru, D.A.; Shin, E.J.; Jackson, J.; Moore, J.; Narang, A.K.; et al. Stereotactic body radiation therapy for palliative management of pancreatic adenocarcinoma in elderly and medically inoperable patients. Oncotarget 2018, 9, 16427–16436. [Google Scholar] [CrossRef] [PubMed]
- Lam, T.C.; Tseng, Y. Defining the radiation oncologist’s role in palliative care and radiotherapy. Ann. Palliat. Med. 2018, 7, 1002. [Google Scholar] [CrossRef]
- Wang, X.S.; Rhines, L.D.; Shiu, A.S.; Yang, J.N.; Selek, U.; Gning, I.; Liu, P.; Allen, P.K.; Azeem, S.S.; Brown, P.D.; et al. Stereotactic body radiation therapy for management of spinal metastases in patients without spinal cord compression: A phase 1-2 trial. Lancet Oncol. 2012, 13, 395–402. [Google Scholar] [CrossRef]
- Chow, E.; Hoskin, P.; Mitera, G.; Zeng, L.; Lutz, S.; Roos, D.; Hahn, C.; Van Der Linden, Y.; Hartsell, W.; Kumar, E. Update of the international consensus on palliative radiotherapy endpoints for future clinical trials in bone metastases. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- NCT03143322 Standard Treatment +/− SBRT in Solid Tumors Patients With Between 1 and 3 Bone-only Metastases. Available online: https://www.cochranelibrary.com/central/doi/10.1002/central/CN-01580971/full (accessed on 3 March 2019).
- Biological Image Guided Antalgic Stereotactic Body Radiotherapy of Bone Metastases. Available online: https://clinicaltrials.gov/ct2/show/NCT01429493 (accessed on 3 March 2019).
- Sprave, T.; Welte, S.E.; Bruckner, T.; Förster, R.; Bostel, T.; Schlampp, I.; Nicolay, N.H.; Debus, J.; Rief, H. Intensity-modulated radiotherapy with integrated-boost in patients with bone metastasis of the spine: Study protocol for a randomized controlled trial. Trials 2018, 19, 59. [Google Scholar] [CrossRef] [PubMed]
- Randomized Trial Comparing Conventional Radiotherapy With Stereotactic Radiotherapy in Patients With Bone Metastases—VERTICAL Study (VERTICAL). Available online: https://clinicaltrials.gov/ct2/show/NCT02364115 (accessed on 3 March 2019).
- Prospective Evaluation of Interventional Studies on Bone Metastases—The PRESENT Cohort. Available online: https://clinicaltrials.gov/ct2/show/NCT02356497 (accessed on 3 March 2019).
- Leiden University Medical, C. The OPTIMAL Study—A Prospective Cohort of Patients With Bone Metastases of the Long Bones. Available online: https://clinicaltrials.gov/show/NCT02705157 (accessed on 3 March 2019).
- Radiosurgical Hypophysectomy for Bone Metasteses Pain. Available online: https://clinicaltrials.gov/ct2/show/NCT03377517 (accessed on 3 March 2019).
- Habberstad, R.; Frøseth, T.C.S.; Aass, N.; Abramova, T.; Baas, T.; Mørkeset, S.T.; Caraceni, A.; Laird, B.; Boland, J.W.; Rossi, R.; et al. The Palliative Radiotherapy and Inflammation Study (PRAIS)—protocol for a longitudinal observational multicenter study on patients with cancer induced bone pain. BMC Palliat. Care 2018, 17, 110. [Google Scholar] [CrossRef] [PubMed]
- Furfari, A.; Wan, B.A.; Ding, K.; Wong, A.; Zhu, L.; Bezjak, A.; Wong, R.; Wilson, C.F.; DeAngelis, C.; Azad, A.; et al. Genetic biomarkers associated with response to palliative radiotherapy in patients with painful bone metastases. Ann. Palliat. Med. 2017, 6, S233–S239. [Google Scholar] [CrossRef] [PubMed]
- Furfari, A.; Wan, B.A.; Ding, K.; Wong, A.; Zhu, L.; Bezjak, A.; Wong, R.; Wilson, C.F.; DeAngelis, C.; Azad, A.; et al. Genetic biomarkers associated with changes in quality of life and pain following palliative radiotherapy in patients with bone metastases. Ann. Palliat. Med. 2017, 6, S248–S256. [Google Scholar] [CrossRef]
- Furfari, A.; Wan, B.A.; Ding, K.; Wong, A.; Zhu, L.; Bezjak, A.; Wong, R.; Wilson, C.F.; DeAngelis, C.; Azad, A.; et al. Genetic biomarkers associated with pain flare and dexamethasone response following palliative radiotherapy in patients with painful bone metastases. Ann. Palliat. Med. 2017, 6, S240–S247. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.S.; Gordon, D.; Tchekmedyian, S.; Yanagihara, R.; Hirsh, V.; Krzakowski, M.; Pawlicki, M.; de Souza, P.; Zheng, M.; Urbanowitz, G.; et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: A phase III, double-blind, randomized trial—The Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J. Clin. Oncol. 2003, 21, 3150–3157. [Google Scholar] [CrossRef] [PubMed]
- Safety, Tolerability and PK/PD of JMT103 in Patients With Bone Metastases From Tumors. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03550508?cond=Bone+Metastases&draw=2&rank=1 (accessed on 3 March 2019).
- Sousa, S.; Clézardin, P. Bone-Targeted Therapies in Cancer-Induced Bone Disease. Calcif. Tissue Int. 2018, 102, 227–250. [Google Scholar] [CrossRef] [PubMed]
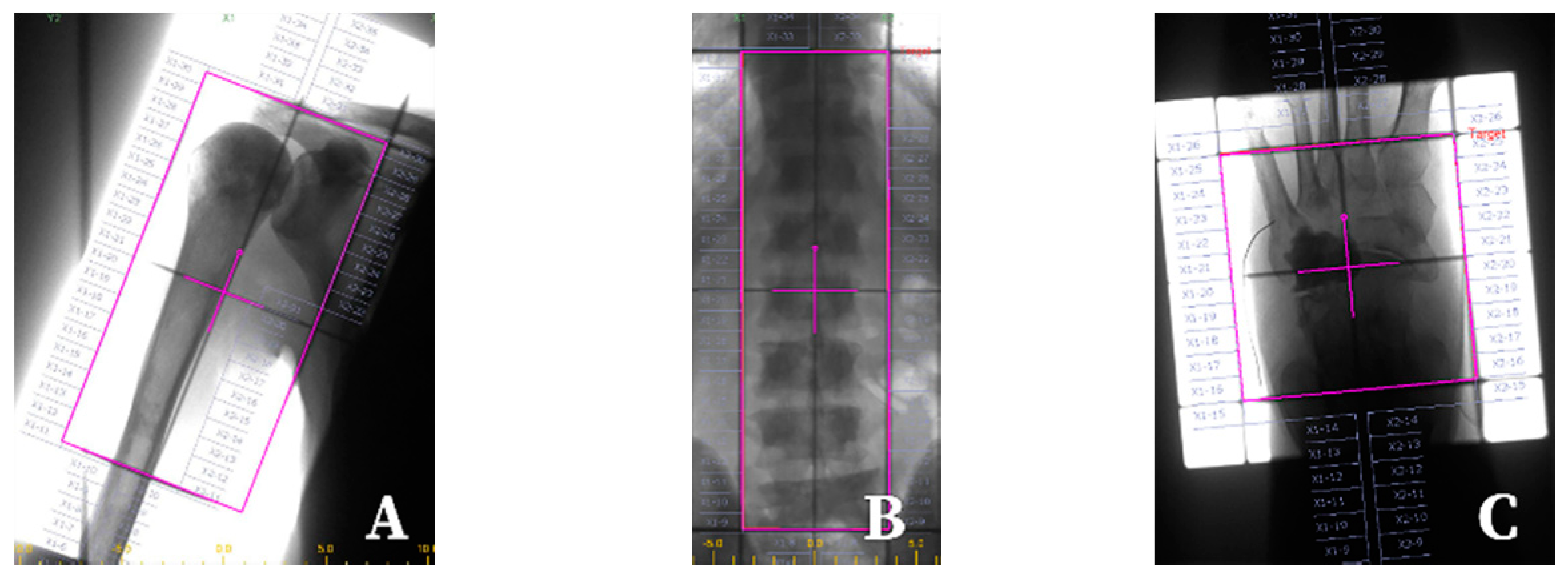
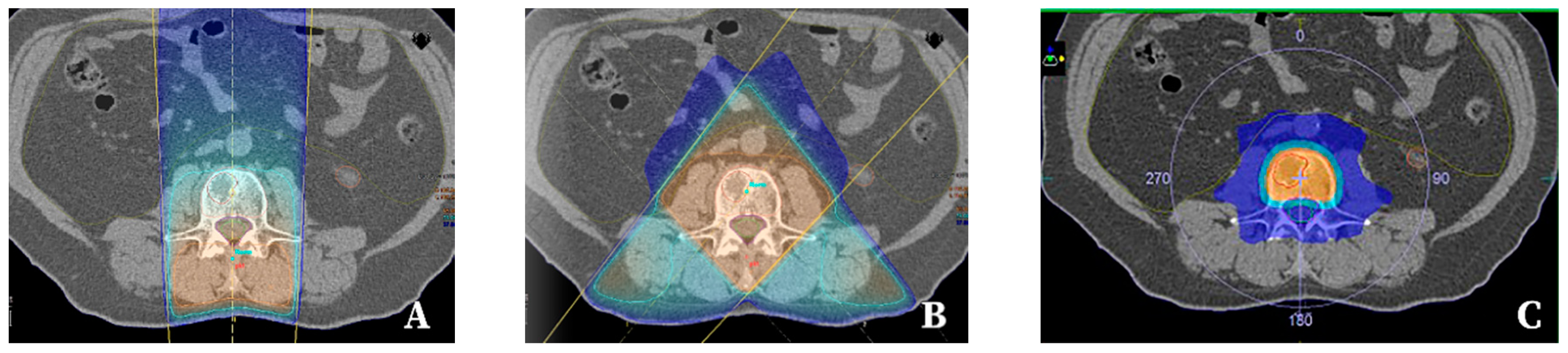
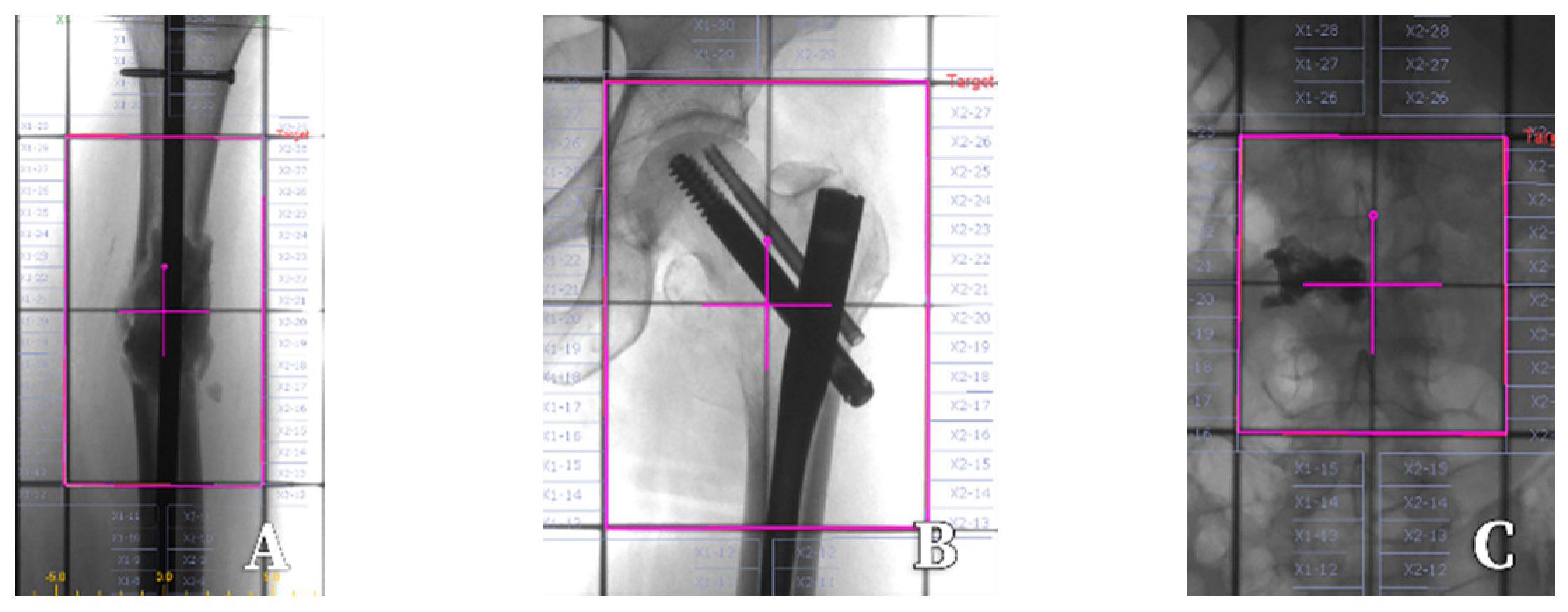
| Patient-Related Factors | Tumour-Related Factors | Logistic Issues |
|---|---|---|
|
|
|
| Trial | Number of Patients | Fractionation | Complete or Partial Pain Response | Complete Pain Response |
|---|---|---|---|---|
| Price et al., 1986 [29] | 288 | 1 × 8 Gy 10 × 3 Gy | 73% 64% | 45% 28% |
| Cole et al., 1989 [30] | 29 | 1 × 8 Gy 6 × 4 Gy | 88% 85% | NR 2 NR 2 |
| Gaze et al., 1997 [26] | 280 | 1 × 10 Gy 5 × 4.5 Gy | 84% 89% | 39% 48% |
| Nielsen et al., 1998 [25] | 241 | 1 × 8 Gy 5 × 4 Gy | 44% 46% | 15% 17% |
| Steenland et al., 1999 [24] | 1171 | 1 × 8 Gy 6 × 4 Gy | 72% 69% | 37% 33% |
| Koswig et al., 1999 [17] | 107 | 1 × 8 Gy 10 × 3 Gy | 79% 82% | 31% 33% |
| BPTWP 1 1999 [22] | 272 | 1 × 8 Gy 5 × 4 Gy | 72% 68% | 52% 51% |
| Roos 2005 [31] | 275 | 1 × 8 Gy 10 × 3 Gy | 61% 53% | 15% 18% |
| Hartsell et al., 2005 [32] | 998 | 1 × 8 Gy 10 × 3 Gy | 65% 66% | 15% 18% |
| Foro Arnalot et al., 2008 [23] | 160 | 1 × 8 Gy 10 × 3 Gy | 75% 86% | 15% 13% |
| Nongkynrih et al., 2018 [27] | 60 | 1 × 8 Gy 5 × 4 Gy 10 × 3 Gy | 80% 75% 85% | 20% 20% 20% |
| Study | Initial RT Dose (Median) | Re-Irradiation Modality | Re-Treatment Dose | Pain Response |
|---|---|---|---|---|
| Choi et al., 2010 [73] | 40 Gy (24.2–50.4) | CyberKnife | Median marginal dose 20 Gy/2fr (range 18/1–25/5) 30 Gy/5fr | 65% improvement in pain |
| Garg et al., 2011 [74] | 30 Gy (30–45) | IG-IMRT | 27 Gy/3fr 20 Gy/5fr | Improvement in pain at 6 months |
| Damast et al., 2011 [75] | 30 Gy (8–66) | IG-IMRT | 20 Gy/5fr 30 Gy/5fr | 77% improvement in pain |
| Mahadevan et al., 2011 [76] | 30 Gy (8–46) | CyberKnife | 25–30 Gy/5fr 24 Gy/3fr | 79% improvement in pain |
| Chang et al., 2012 [58] | 39Gy Gy2 1 | CyberKnife | 20.6 Gy/1fr (18.2–23.7) | 80.8% pain control rate at 1 year |
| Hashmi et al., 2016 [77] | 30 Gy/10fr | IG-IMRT | 16.6 Gy/fr 24 Gy/3fr | 74.3% improvement in pain |
| Study | Brachytherapy Intervention | Pain Control VAS (Mean +/− Standard Deviation) | |
|---|---|---|---|
| Pretreatment | Posttreatment | ||
| Cardoso et al., 2009 [83] | Percutaneous curettage of SM cement augmentation, and bone cement injection with 153Sm | 8.5 +/− 2 | 2.6 +/− 3.1 |
| Yang et al., 2013 [84] | Cement augmentation and percutaneous 125I seed implantation | 8.73 +/− 0.31 | 1.32 +/− 0.37 |
| Cao et al., 2014 [85] | Percutaneous 125I seed implantation | 4.48 +/− 2.03 | 1.18 +/− 1.38 |
| Huang et al., 2014 [89] | Cement augmentation and percutaneous 125I seed implantation | 7.12 +/− 1.48 | 2.26 +/− 1.07 |
| Li et al., 2014 [86] | Cement augmentation and percutaneous 125I seed implantation | 7.7 +/− 1.3 (SN) 8.0 +/− 1.2 (MN) | 2.6 +/− 1.0 2.4 +/− 1.1 |
| Wang et al., 2015 [87] | Cement augmentation and percutaneous 125I seed implantation | 6.37 +/− 1.67 | 1.32 +/− 0.75 |
| Qian et al., 2016 [88] | Pedicle fixation of affected vertebra and implantation of 125I seeds via needles | 7.43 +/− 0.98 | 4.29 +/− 0.98 |
| Radioactive Isotope | Trial | Study Type | Number of Patients | Cancer | Analgesic Effect | Duration of Analgesic Effect |
|---|---|---|---|---|---|---|
| Radium-223-Chloride | Nilsson et al., 2005 [145] | phase I | 25 pts | breast, prostate | 50% pts | 1 WP 52%, 4 WP 60%, 8 WP 56% |
| Bruland et al., 2006 [146] | phase I | 6 pts | prostate | repeated administration was well tolerated | NS | |
| Nilsson et al., 2007 [147] | phase II randomized | 64 pts | prostate | 10 versus 16 (placebo) reported bone pain after injection | NS | |
| Coleman et al., 2014 [148] | phase IIa | 23 pts | breast | BPI pain severity index at week 17 was 0.6 | NS | |
| Parker et al., 2011 ALSYMPCA [127] | phase III randomized | 921 pts | castration-resistant prostate | NS | NS | |
| Prelaj at al., 2019 [149] | retrospective | 32 pts | prostate | 71% | NS | |
| Rhenium-186-HEDP | Maxon et al., 1991 [136] | double-blind | 20 pts | prostate | 80% pts | NS |
| Maxon et al., 1992 [137] | prospective | 43 pts | breast, prostate | 77% pts initial injection 50% second in injection | 7 weeks | |
| Han et al., 1999 [138] | prospective | 30 pts | breast | 58% pts | NS | |
| Han et al., 2002 PLACORHEN [139] | double-blind randomized | 111 pts | prostate | 0–96% (mean 27%) | NS | |
| Rhenium-188-HEDP | Palmedo et al., 2000 [142] | prospective | 22 pts | prostate | 64% | 7.5 weeks |
| Liepe et al., 2000 [143] | prospective | 15 pts | prostate | 76% | NS | |
| Samarium-153-EDTMP | Dolezal et al., 2000 [114] | prospective | 33 pts | prostate, breast, other | 70% | NS |
| Wang et al., 2003 [115] | Comparative randomized | 9 pts | prostate, breast, other | 78% | 3.5 +/− 2.3 months | |
| Sapienza et al., 2004 [116] | retrospective | 73 pts | prostate, breast | 76% | NS | |
| Samarium-153-EDTMP | Etchebehere et al., 2004 [117] | retrospective | 58 pts | prostate, breast, other | 78% | 5.75–6 months |
| Sartor et al., 2004 [118] | phase III randomized | 152 pts | prostate | 64% | NS | |
| Tripathi et al., 2006 [119] | prospective | 86 pts | prostate, breast, other | 73% | 2–8 months | |
| Ripamonti et al., 2007 [120] | prospective | 13 pts | prostate, breast | 61,5% | NS | |
| Liepe et al., 2007 [109] | prospective | 15 pts | prostate, breast | 73% | 10 +/− 1 weeks | |
| Dolezal et al., 2007 [121] | prospective | 32 pts | prostate | 72% | 3 months | |
| Strontium-89 Dichloride | Sciuto. et al., 2001 [104] | randomized | 51 pts | breast | 84% | 2–14 months |
| Turner et al., 2001 [105] | prospective | 93 pts | prostate | 63% | NS | |
| Dafermou et al., 2001 [106] | multicentre observational | 527 pts | prostate | 59.8% | 5.0 +/− 3.5 months | |
| Ashayeri et al., 2002 [107] | prospective | 27 pts | prostate, breast | 81% | up to 1 year | |
| Baczyk et al., 2003 [108] | prospective | 70 pts | prostate | 88% | 3–12 months | |
| Fettich et al., 2003 [103] | prospective | 93 pts | bone mts | 75% | NS | |
| Liepe et al., 2007 [109] | prospective | 15 pts | prostate, breast | 72% | 9 +/− 2 weeks | |
| Ma et al., 2008 [94] | prospective | 116 pts | prostate | 83.6% | 3–12 months | |
| Zenda et al., 2014 [110] | prospective | 54 pts | 26 pts prostate/breast 28 pts other malignancies (lung, head and neck, colorectal, other) | 69.2% 73.1% | 2–6 months |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sierko, E.; Hempel, D.; Zuzda, K.; Wojtukiewicz, M.Z. Personalized Radiation Therapy in Cancer Pain Management. Cancers 2019, 11, 390. https://doi.org/10.3390/cancers11030390
Sierko E, Hempel D, Zuzda K, Wojtukiewicz MZ. Personalized Radiation Therapy in Cancer Pain Management. Cancers. 2019; 11(3):390. https://doi.org/10.3390/cancers11030390
Chicago/Turabian StyleSierko, Ewa, Dominika Hempel, Konrad Zuzda, and Marek Z. Wojtukiewicz. 2019. "Personalized Radiation Therapy in Cancer Pain Management" Cancers 11, no. 3: 390. https://doi.org/10.3390/cancers11030390
APA StyleSierko, E., Hempel, D., Zuzda, K., & Wojtukiewicz, M. Z. (2019). Personalized Radiation Therapy in Cancer Pain Management. Cancers, 11(3), 390. https://doi.org/10.3390/cancers11030390






