Efficacy, Safety, and Tolerability of Approved Combination BRAF and MEK Inhibitor Regimens for BRAF-Mutant Melanoma
Abstract
:1. Introduction
2. Results
2.1. Baseline Characteristics
2.2. Efficacy
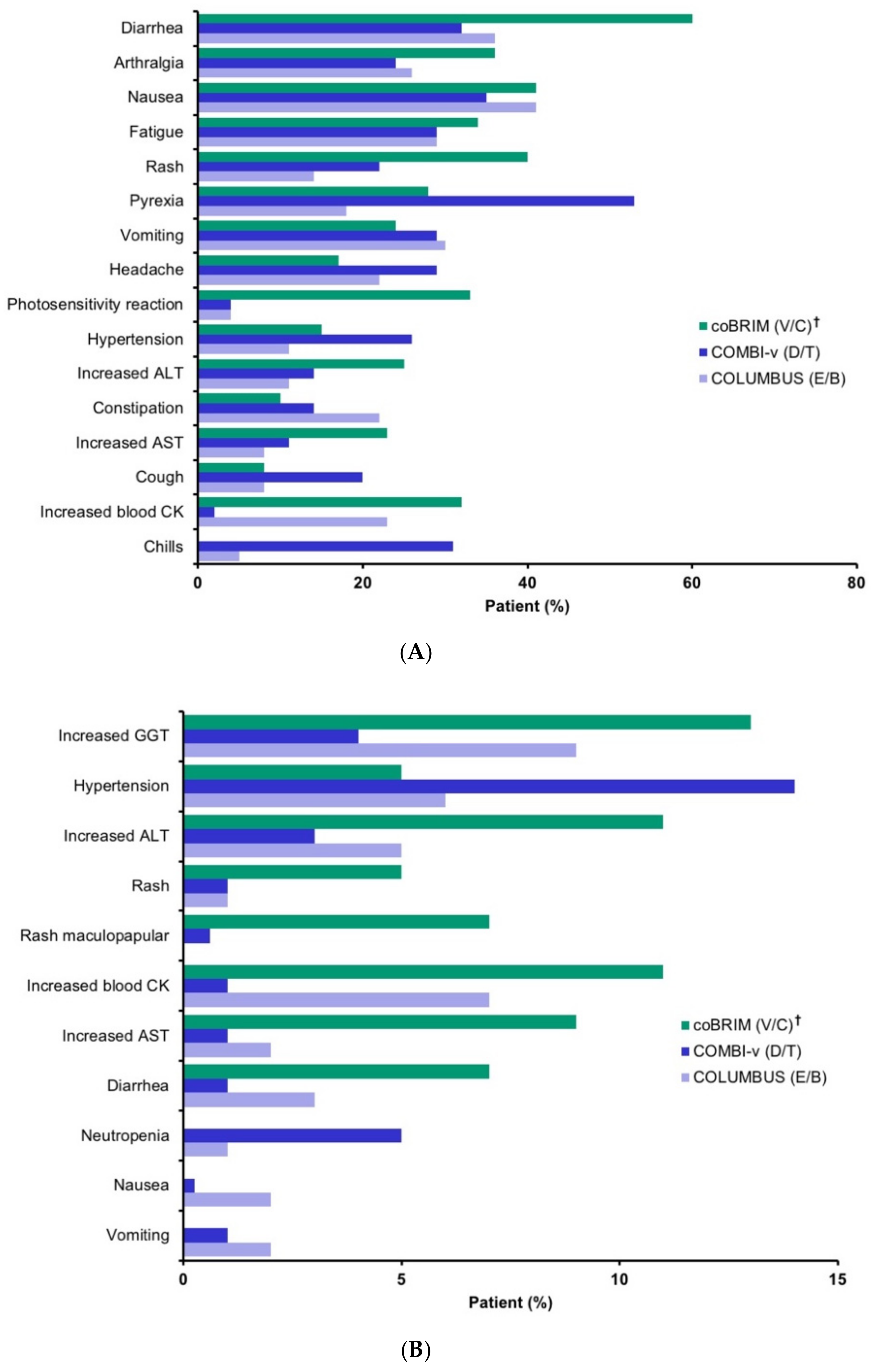
2.3. Safety
- Pyrexia includes pyrexia, increased body temperature, hyperpyrexia, hyperthermia;
- Peripheral edema includes peripheral edema, local swelling, localized edema, edema, peripheral swelling;
- Vomiting includes vomiting, retching;
- Arthralgia includes arthralgia, arthropathy, joint stiffness;
- Rash includes rash, exfoliative rash, erythematous rash, follicular rash, generalized rash, macular rash, maculo-papular rash, papular rash, pruritic rash, vesicular rash;
- Visual impairment includes visual impairment, blurred vision, reduced visual acuity;
- Serous retinopathy includes retinal detachment, chorioretinitis, chorioretinopathy, cystoid macular edema, macular retinal pigment epithelium detachment, retinal pigment epithelium detachment, macular detachment, macular edema, metamorphopsia, retinal disorder, retinal exudates, retinal edema, retinal pigment epitheliopathy, retinopathy, subretinal fluid;
- Hemorrhage includes rectal hemorrhage, hematochezia, hematuria, cerebral hemorrhage, epistaxis, hemorrhoidal hemorrhage, menorrhagia, metrorrhagia, retinal hemorrhage, conjunctival hemorrhage, gastric ulcer hemorrhage, gastrointestinal hemorrhage, hematospermia, hemorrhagic cyst, intracranial tumor hemorrhage, polymenorrhea, subdural hematoma, uterine hemorrhage, hemorrhagic diarrhea, hemoptysis, mucosal hemorrhage, occult blood, post procedural hemorrhage, postmenopausal hemorrhage, pulmonary alveolar hemorrhage, tumor hemorrhage, vaginal hemorrhage, wound hemorrhage.
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Balch, C.M.; Soong, S.J.; Gershenwald, J.E.; Thompson, J.F.; Reintgen, D.S.; Cascinelli, N.; Urist, M.; McMasters, K.M.; Ross, M.I.; Kirkwood, J.M.; et al. Prognostic factors analysis of 17,600 melanoma patients: Validation of the American Joint Committee on Cancer melanoma staging system. J. Clin. Oncol. 2001, 19, 3622–3634. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Cancer Facts and Figures 2018; American Cancer Society: Atlanta, GA, USA, 2018; Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2018/cancer-facts-and-figures-2018.pdf (accessed on 30 November 2018).
- GBD (Disease and Injury Incidence and Prevalence Collaborators). Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Karimkhani, C.; Green, A.C.; Nijsten, T.; Weinstock, M.A.; Dellavalle, R.P.; Naghavi, M.; Fitzmaurice, C. The global burden of melanoma: Results from the Global Burden of Disease Study 2015. Br. J. Dermatol. 2017, 177, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Colombino, M.; Capone, M.; Lissia, A.; Cossu, A.; Rubino, C.; De Giorgi, V.; Massi, D.; Fonsatti, E.; Staibano, S.; Nappi, O.; et al. BRAF/NRAS mutation frequencies among primary tumors and metastases in patients with melanoma. J. Clin. Oncol. 2012, 30, 2522–2529. [Google Scholar] [CrossRef] [PubMed]
- Hauschild, A.; Grob, J.J.; Demidov, L.V.; Jouary, T.; Gutzmer, R.; Millward, M.; Rutkowski, P.; Blank, C.U.; Miller, W.H., Jr.; Kaempgen, E.; et al. Dabrafenib in BRAF-mutated metastatic melanoma: A multicentre, open-label, phase 3 randomised controlled trial. Lancet 2012, 380, 358–365. [Google Scholar] [CrossRef]
- Chapman, P.B.; Hauschild, A.; Robert, C.; Haanen, J.B.; Ascierto, P.; Larkin, J.; Dummer, R.; Garbe, C.; Testori, A.; Maio, M.; et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 2011, 364, 2507–2516. [Google Scholar] [CrossRef]
- Poulikakos, P.I.; Zhang, C.; Bollag, G.; Shokat, K.M.; Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature 2010, 464, 427–430. [Google Scholar] [CrossRef] [Green Version]
- Robert, C.; Karaszewska, B.; Schachter, J.; Rutkowski, P.; Mackiewicz, A.; Stroiakovski, D.; Lichinitser, M.; Dummer, R.; Grange, F.; Mortier, L.; et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N. Engl. J. Med. 2015, 372, 30–39. [Google Scholar] [CrossRef]
- Larkin, J.; Ascierto, P.A.; Dreno, B.; Atkinson, V.; Liszkay, G.; Maio, M.; Mandala, M.; Demidov, L.; Stroyakovskiy, D.; Thomas, L.; et al. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N. Engl. J. Med. 2014, 371, 1867–1876. [Google Scholar] [CrossRef]
- Dummer, R.; Hauschild, A.; Lindenblatt, N.; Pentheroudakis, G.; Keilholz, U. Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26 (Suppl. 5), v126–v132. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology, Neuroendocrine Tumors, 3rd ed.; National Comprehensive Cancer Network: Upper Dublin Township, PA, USA, 2017. [Google Scholar]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology, Cutaneous Melanoma 2019, 1st ed.; National Comprehensive Cancer Network: Upper Dublin Township, PA, USA, 2018. [Google Scholar]
- Ascierto, P.A.; McArthur, G.A.; Dreno, B.; Atkinson, V.; Liszkay, G.; Di Giacomo, A.M.; Mandala, M.; Demidov, L.; Stroyakovskiy, D.; Thomas, L.; et al. Cobimetinib combined with vemurafenib in advanced BRAF(V600)-mutant melanoma (coBRIM): Updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016, 17, 1248–1260. [Google Scholar] [CrossRef]
- Long, G.V.; Stroyakovsky, D.L.; Gogas, H.; Levchenko, E.; de Braud, F.; Larkin, J.M.G.; Garbe, C.; Jouary, T.; Hauschild, A.; Grob, J.J.; et al. COMBI-d: A randomized, double-blinded, Phase III study comparing the combination of dabrafenib and trametinib to dabrafenib and trametinib placebo as first-line therapy in patients (pts) with unresectable or metastatic BRAFV600E/K mutation-positive cutaneous melanoma. J. Clin. Oncol. 2014, 32, 9011. [Google Scholar] [CrossRef]
- Long, G.V.; Stroyakovskiy, D.; Gogas, H.; Levchenko, E.; de Braud, F.; Larkin, J.; Garbe, C.; Jouary, T.; Hauschild, A.; Grob, J.J.; et al. Dabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: A multicentre, double-blind, phase 3 randomised controlled trial. Lancet 2015, 386, 444–451. [Google Scholar] [CrossRef]
- Robert, C.; Karaszewska, B.; Schachter, J.; Rutkowski, P.; Mackiewicz, A.; Stroyakovskiy, D.; Dummer, R.; Grange, F.; Mortier, L.; Chiarion-Sileni, V.; et al. Three-year estimate of overall survival in COMBI-v, a randomized phase 3 study evaluating first-line dabrafenib (D) + trametinib (T) in patients (pts) with unresectable or metastatic BRAF V600E/K–mutant cutaneous melanoma. Ann. Oncol. 2016, 27, LBA40. [Google Scholar] [CrossRef]
- Long, G.V.; Stroyakovskiy, D.; Gogas, H.; Levchenko, E.; de Braud, F.; Larkin, J.; Garbe, C.; Jouary, T.; Hauschild, A.; Grob, J.J.; et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N. Engl. J. Med. 2014, 371, 1877–1888. [Google Scholar] [CrossRef] [PubMed]
- Array BioPharma Inc. Braftovi [Package Insert]; Array BioPharma Inc.: Boulder, CO, USA, 2018. [Google Scholar]
- Array BioPharma Inc. Mektovi [Package Insert]; Array BioPharma Inc.: Boulder, CO, USA, 2018. [Google Scholar]
- European Medicines Agency. European Medicines Agency. European Public Assessment Report (EPAR): Braftovi. Available online: https://www.ema.europa.eu/documents/overview/braftovi-epar-medicine-overview_en.pdf (accessed on 8 December 2018).
- European Medicines Agency. European Medicines Agency. European Public Assessment Report (EPAR): Mektovi. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/mektovi (accessed on 8 December 2018).
- Dummer, R.; Ascierto, P.A.; Gogas, H.J.; Arance, A.; Mandala, M.; Liszkay, G.; Garbe, C.; Schadendorf, D.; Krajsova, I.; Gutzmer, R.; et al. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): A multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2018, 19, 603–615. [Google Scholar] [CrossRef]
- Delord, J.P.; Robert, C.; Nyakas, M.; McArthur, G.A.; Kudchakar, R.; Mahipal, A.; Yamada, Y.; Sullivan, R.; Arance, A.; Kefford, R.F.; et al. Phase I dose-escalation and -expansion study of the BRAF inhibitor encorafenib (LGX818) in metastatic BRAF- mutant melanoma. Clin. Cancer Res. 2017, 23, 5339–5348. [Google Scholar] [CrossRef]
- Adelmann, C.H.; Ching, G.; Du, L.; Saporito, R.C.; Bansal, V.; Pence, L.J.; Liang, R.; Lee, W.; Tsai, K.Y. Comparative profiles of BRAF inhibitors: The paradox index as a predictor of clinical toxicity. Oncotarget 2016, 7, 30453–30460. [Google Scholar] [CrossRef]
- Daud, A.; Gill, J.; Kamra, S.; Chen, L.; Ahuja, A. Indirect treatment comparison of dabrafenib plus trametinib versus vemurafenib plus cobimetinib in previously untreated metastatic melanoma patients. J. Hematol. Oncol. 2017, 10, 3. [Google Scholar] [CrossRef]
- Novartis Pharmaceuticals Corporation. Tafinlar [Package Insert]; Novartis Pharmaceuticals Corporation: East Hanover, NJ, USA, 2018. [Google Scholar]
- Robert, C.; Karaszewska, B.; Schachter, J.; Rutkowski, P.; Mackiewicz, A.; Stroyakovskiy, D.; Lichinitser, M.; Dummer, R.; Grange, F.; Mortier, L.; et al. 3301 Two year estimate of overall survival in COMBI-v, a randomized, open-label, phase III study comparing the combination of dabrafenib (D) and trametinib (T) with vemurafenib (Vem) as first-line therapy in patients (pts) with unresectable or metastatic BRAF V600E/K mutation-positive cutaneous melanoma. Eur. J. Cancer 2015, 51, S663. [Google Scholar] [CrossRef]
- Genentech USA Inc. Cotellic [Package Insert]; Genentech USA Inc.: South San Francisco, CA, USA, 2018. [Google Scholar]
- Genentech. Zelboraf [Package Insert]; Genentech: South San Francisco, CA, USA, 2017. [Google Scholar]
- European Medicines Agency Public Assessment Report. Cotellic EMA/685908/2015. Available online: https://www.ema.europa.eu/documents/assessment-report/cotellic-epar-public-assessment-report_en.pdf (accessed on 20 December 2018).
- Dummer, R.; Ascierto, P.A.; Gogas, H.J.; Arance, A.; Mandala, M.; Liszkay, G.; Garbe, C.; Schadendorf, D.; Krajsova, I.; Gutzmer, R.; et al. Overall survival in patients with BRAF-mutant melanoma receiving encorafenib plus binimetinib versus vemurafenib or encorafenib (COLUMBUS): A multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 1315–1327. [Google Scholar] [CrossRef]
- European Medicines Agency Public Assessment Report. Mekinist (Trametinib) and Tafinlar (Dabrafenib) EMEA/H/C/WS/0736. Available online: https://www.ema.europa.eu/documents/variation-report/mekinist-epar-assessment-report-variation_en.pdf (accessed on 11 January 2019).
- Giusti, R. NDA 206192: Cobimetinib in Combination with Vemurafenib Medical Review; Food and Drug Administration Center for Drug Evaluation and Research: Washington, DC, USA, 2015. [Google Scholar]
- ICH (The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use). Harmonised Tripartite Guideline Clinical Safety Data Management: Definitions and Standards for Expedited Reporting E2A. Available online: https://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Efficacy/E2A/Step4/E2A_Guideline.pdf (accessed on 20 December 2018).
- Food and Drug Administration. Guidance for Industry-Adverse Reactions Section of Labeling for Human Prescription Drug and Biological Products—Content and Format. Available online: https://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm075057.pdf (accessed on 4 March 2019).
- Dreno, B.; Ribas, A.; Larkin, J.; Ascierto, P.A.; Hauschild, A.; Thomas, L.; Grob, J.J.; Koralek, D.O.; Rooney, I.; Hsu, J.J.; et al. Incidence, course, and management of toxicities associated with cobimetinib in combination with vemurafenib in the coBRIM study. Ann. Oncol. 2017, 28, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Novartis Pharmaceuticals. Mekinist [Package Insert]; Novartis Pharmaceuticals: East Hanover, NJ, USA, 2018. [Google Scholar]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- ICH (The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use). Understanding MedDRA-the Medical Dictionary for Regulatory Activities (MedDRA). Available online: https://www.meddra.org/sites/default/files/main_page_slideshow/meddra2013.pdf (accessed on 11 January 2019).

| Characteristics | COMBI-v | coBRIM | COLUMBUS | |||
|---|---|---|---|---|---|---|
| D/T | V | V/C | V | E/B | V | |
| Intent-to-treat population | 352 | 352 | 247 | 248 | 192 | 191 |
| Age (year) Median (range) | 55 (18–91) | 54 (18–88) | 56 (23–88) | 55 (25–85) | 57 (20–89) | 56 (21–82) |
| Male sex, n (%) | 208 (59) | 180 (51) | 146 (59) | 140 (56) | 115 (60) | 111 (58) |
| ECOG performance score n/total n (%) | ||||||
| 0 | 248/350 (71) | 248/352 (70) | 184/243 (76) | 164/244 (67) | 136 (71) | 140 (73) |
| 1 | 102/350 (29) | 104/352 (30) | 58/243 (24) | 80/244 (33) | 56 (29) | 51 (27) |
| 2 | 0/350 | 0/352 | 1/243 (<1) | 0/244 | 0 | 0 |
| Metastatic status n/total n (%) | ||||||
| M0 | 14/351 (4) | 26/351 (7) | 21 (9) | 13 (5) | 9 (5) | 11 (6) |
| M1a | 55/351 (16) | 50/351 (14) | 40 (16) | 40 (16) | 26 (14) | 24 (13) |
| M1b | 61/351 (17) | 67/351 (19) | 40 (16) | 42 (17) | 34 (18) | 31 (16) |
| M1c | 221/351 (63) | 208/351 (59) | 146 (59) | 153 (62) | 123 (64) | 125 (65) |
| Number of organs involved n/total n (%) | ||||||
| <3 | 177/351 (50) | 201/352 (57) | NR | NR | 105/192 (54) | 104/191 (54) |
| ≥3 | 174/351 (50) | 151/352 (43) | 87/192 (45) | 87/191 (46) | ||
| Elevated LDH n /total n (%) | 118/351 (34) | 114/352 (32) | 112/242 (46) | 104/242 (43) | 55/192 (29) | 52/191 (27) |
| BRAF mutation n/total n (%) | ||||||
| V600E | 312/346 (90) | 317/351 (90) | 170/194 (88) | 174/206 (84) | 170/192 (89) | 168/191 (88) |
| V600K | 34/346 (10) | 34 /351 (10) | 24/194 (12) | 32/206 (16) | 22/192 (11) | 23/191 (12) |
| Efficacy Outcome | COMBI-v [27,28] | coBRIM [30,31] | COLUMBUS [22,23,32] | |||
|---|---|---|---|---|---|---|
| D/T n = 352 | V n = 352 | V/C n = 247 | V n = 248 | E/B n = 192 | V n = 191 | |
| PFS *,†, median (95% CI), mo | 11.4 (9.9−14.9) | 7.3 (5.8−7.8) | 12.3 (9.5–13.4) | 7.2 (5.6–7.5) | 14.8 (10.4−18.4) | 7.3 (5.7−8.5) |
| HR (95% CI) | 0.56 (0.46−0.69) | 0.58 (0.46−0.72) | 0.49 (0.37−0.64) | |||
| ORR * (95% CI), % | 64 (59−69) | 51 (46−56) | 70 (64−75) | 50 (44−56) | 75 (68−81) | 49 (42−57) |
| Median DOR * (95% CI), mo | 13.8 (11.0−NR) | 7.5 (7.3−9.3) | 13.0 (11.1−16.6) | 9.2 (7.5−12.8) | 16.2 ** (11.1−20.4) | 8.4 ** (5.8− 11.0) |
| Median OS (95% CI), mo | 25.6 (18.3−NR) | 17.2 (16.4−NR) | 22.3 (20.3−NE) | 17.4 (15.0−19.8) | 33.6 (24.4−39.2) | 16.9 (14.0−24.5) |
| HR (95% CI) | 0.69 (0.53−0.89) | 0.69 (0.54−0.88) | 0.61 (0.47−0.79) | |||
| AE type, n (%) | COMBI-v [9,27,28] | coBRIM [31,34] | COLUMBUS [22,23,32] | |||
|---|---|---|---|---|---|---|
| D/T n = 350 | V n = 349 | V/C n = 247 | V n = 246 | E/B n = 192 | V n = 186 | |
| Any AE | 343 (98) | 345 (99) | 244 (98.8) | 240 (97.6) | 189 (98) | 185 (99) |
| Any serious AE | 131 (37) | 122 (35) | 85 (34.4) | 64 (26) | 66 (34) | 69 (37) |
| AE leading to death | 3 (1) | 3 (1) | 5 (2) | 3 (1.2) | 6 (3) | 2 (1) |
| Any grade ≥3 AE | 183 (52) | 221 (63) | 176 (71.3) | 146 (59.3) | 111 (58) | 118 (63) |
| Any dose interruptions/modifications | 192 (55) | 197 (56) | 110 (44.5) | 87 (35.4) | 102 (53) | 115 (62) |
| Discontinuation due to AE | 44 (13) | 41 (12) | 37 (15) | 20 (8.1) | 29 (15) | 32 (17) |
| ADR, % | D/T * n = 209 | V/C n = 247 | E/B † n = 192 | |||
|---|---|---|---|---|---|---|
| All Grades | Grade 3/4 | All Grades | Grade 3/4 | All Grades | Grade 3/4 | |
| General | ||||||
| Pyrexia | 57 ‡ | 7 | 28 | 2 | 18 | 4 |
| Peripheral edema | 25 | 1.4 | 12.6 [31] | NR | 13 | 1 |
| Chills | 31 | 0 | 10 | 0 | 0 | 0 |
| Gastrointestinal disorders | ||||||
| Nausea | 34 | 0.5 | 41 | 1 | 41 | 2 |
| Vomiting | 25 | 1.0 | 24 | 1 | 30 | 2 |
| Diarrhea | 30 | 1.4 | 60 | 6 | 36 | 3 |
| Arthralgia | 26 | 0.9 | 36 [31] | 2.4 [31] | 26 | 1 |
| Skin | ||||||
| Rash | 42 | 0 | 73 [37] | 17 [37] | 22 | 1 |
| Acneiform dermatitis | 10 [33] | NR | 16 [31] | 2 [31] | 4.4 [23] | 0 [23] |
| PPE syndrome | 5 [33] | NR | 6 [34] | 0 [34] | 6.2 [23] | 0 [23] |
| cuSCC § | 3 | NR | 6 | NR | 2.6 | 0 |
| Basal cell carcinoma | 3.3 | NR | 4.5 | NR | 1.6 | 0 |
| LV dysfunction ¶ | 6 # | NR | 9 [34] | 2 [34] | 7 | 1.6 ** |
| Creatine kinase increased †† | Not monitored ‡‡ | 79 | 14 | 58 | 5 | |
| Photosensitivity ¶¶ | 2 [31] | NR | 46 | 4 | 4 [23] | 0.4 [23] |
| Liver function tests †† | ||||||
| ALT increased | 44 | 3.8 | 68 | 11 | 29 | 6 |
| AST increased | 60 | 4.3 | 73 | 8 | 27 | 3 |
| ALP increased | 50 | 1.0 | 71 | 7 | 21 | 1 |
| Hemorrhage | 19 | 1.9 | 13 | 1 | 19 | 3.2 |
| Ocular toxicity | ||||||
| Serous retinopathy | Not monitored ## | 26 ††† | NR | 20 ††† | 3 | |
| Visual impairment | NR | NR | 15 ‡‡‡ | <1 | 20 ‡‡‡ | 0 |
| Uveitis | 2 [33] | NR | 2 [34] | NR | 4 | 0 |
| Venous thromboembolism | 2.8 §§§ | NR | NR | NR | 6 | 0 |
| ECG QT prolonged | 0.8 [33] | 0 [33] | NR | 1.6 [31] | 0.5 ### | 0 |
| Hypertension | 25 | 6 | 15 | 4 | 11 | 6 |
| Study Design Characteristic | COMBI-v | coBRIM | COLUMBUS |
|---|---|---|---|
| Population | Unresectable locally advanced or metastatic melanoma with BRAF V600E/K mutation | Unresectable locally advanced or metastatic melanoma with BRAF V600 mutation | Unresectable locally advanced or metastatic melanoma with BRAF V600E and/or V600K mutation |
| Enrollment | 704 patients (June 2012–Oct 2013) | 495 patients (Jan 2013–Jan 2014) | 577 patients (Dec 2013–April 2015) |
| Randomization | 1:1 | 1:1 | 1:1:1 |
| Treatments | dabrafenib 150 mg BID + trametinib 2 mg QD | vemurafenib 960 mg BID + cobimetinib 60 mg QD | encorafenib 450 mg QD + binimetinib 45 mg BID |
| vemurafenib 960 mg BID | vemurafenib 960 mg BID | vemurafenib 960 mg BID encorafenib 300 mg QD * | |
| Investigator/Patient blinding | no | yes | no |
| Prior systemic therapy permitted | none | none | first-line immunotherapy |
| Primary endpoint | OS | PFS (local) | PFS (central) |
| Secondary endpoints | PFS (local) ORR DOR | PFS (central) OS ORR DOR | PFS (local) OS ORR DOR TTR |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamid, O.; Cowey, C.L.; Offner, M.; Faries, M.; Carvajal, R.D. Efficacy, Safety, and Tolerability of Approved Combination BRAF and MEK Inhibitor Regimens for BRAF-Mutant Melanoma. Cancers 2019, 11, 1642. https://doi.org/10.3390/cancers11111642
Hamid O, Cowey CL, Offner M, Faries M, Carvajal RD. Efficacy, Safety, and Tolerability of Approved Combination BRAF and MEK Inhibitor Regimens for BRAF-Mutant Melanoma. Cancers. 2019; 11(11):1642. https://doi.org/10.3390/cancers11111642
Chicago/Turabian StyleHamid, Omid, C. Lance Cowey, Michelle Offner, Mark Faries, and Richard D. Carvajal. 2019. "Efficacy, Safety, and Tolerability of Approved Combination BRAF and MEK Inhibitor Regimens for BRAF-Mutant Melanoma" Cancers 11, no. 11: 1642. https://doi.org/10.3390/cancers11111642
APA StyleHamid, O., Cowey, C. L., Offner, M., Faries, M., & Carvajal, R. D. (2019). Efficacy, Safety, and Tolerability of Approved Combination BRAF and MEK Inhibitor Regimens for BRAF-Mutant Melanoma. Cancers, 11(11), 1642. https://doi.org/10.3390/cancers11111642






