Analysis of A 6-Mirna Signature in Serum from Colorectal Cancer Screening Participants as Non-Invasive Biomarkers for Advanced Adenoma and Colorectal Cancer Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Samples
2.2. Hemolysis Determination
2.3. RNA Extraction
2.4. Analysis of miRNA Expression by Real-Time qRTPCR
2.5. Faecal Haemoglobin (f(Hb)) Determination
2.6. Statistical Analysis
3. Results
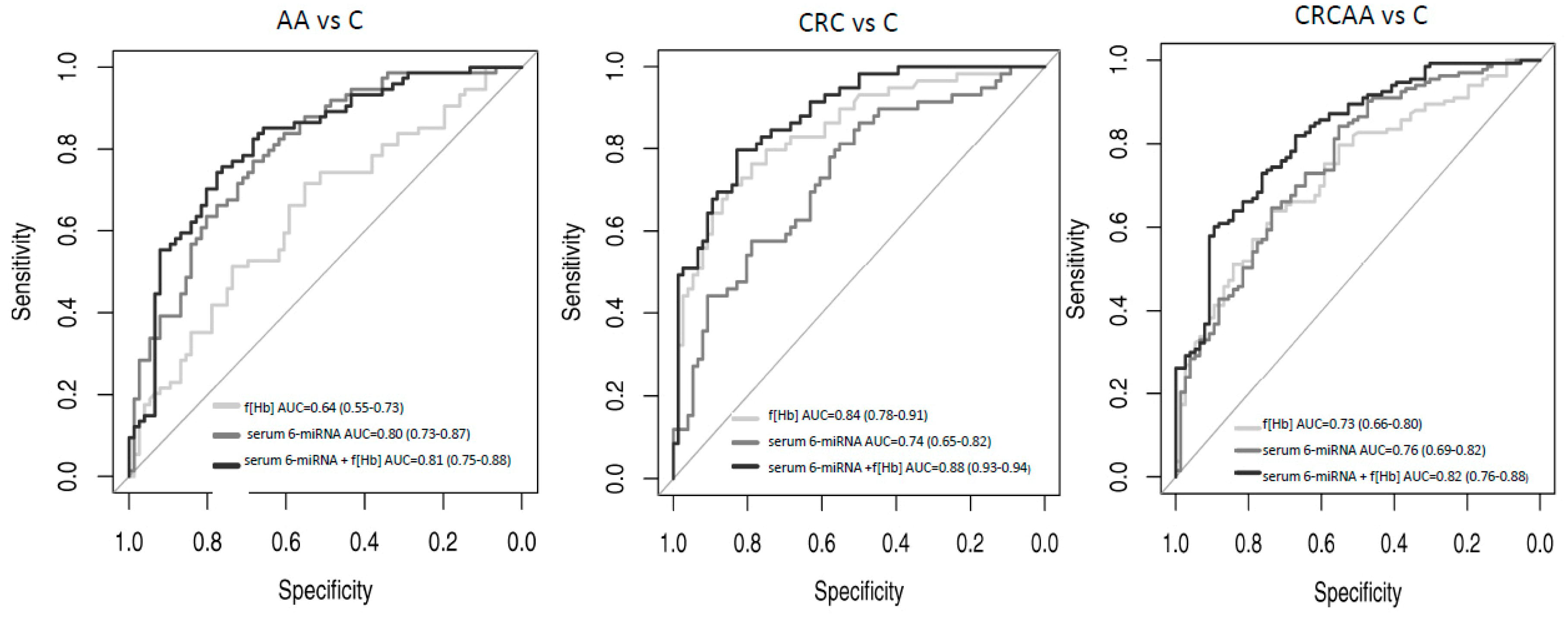
3.1. A Signature Combining 6-miRNAs in Serum and f(Hb) Show High Accuracy for Detecting Patients Harbouring Advanced Adenomas (AA) or Colorectal Cancer (CRC)
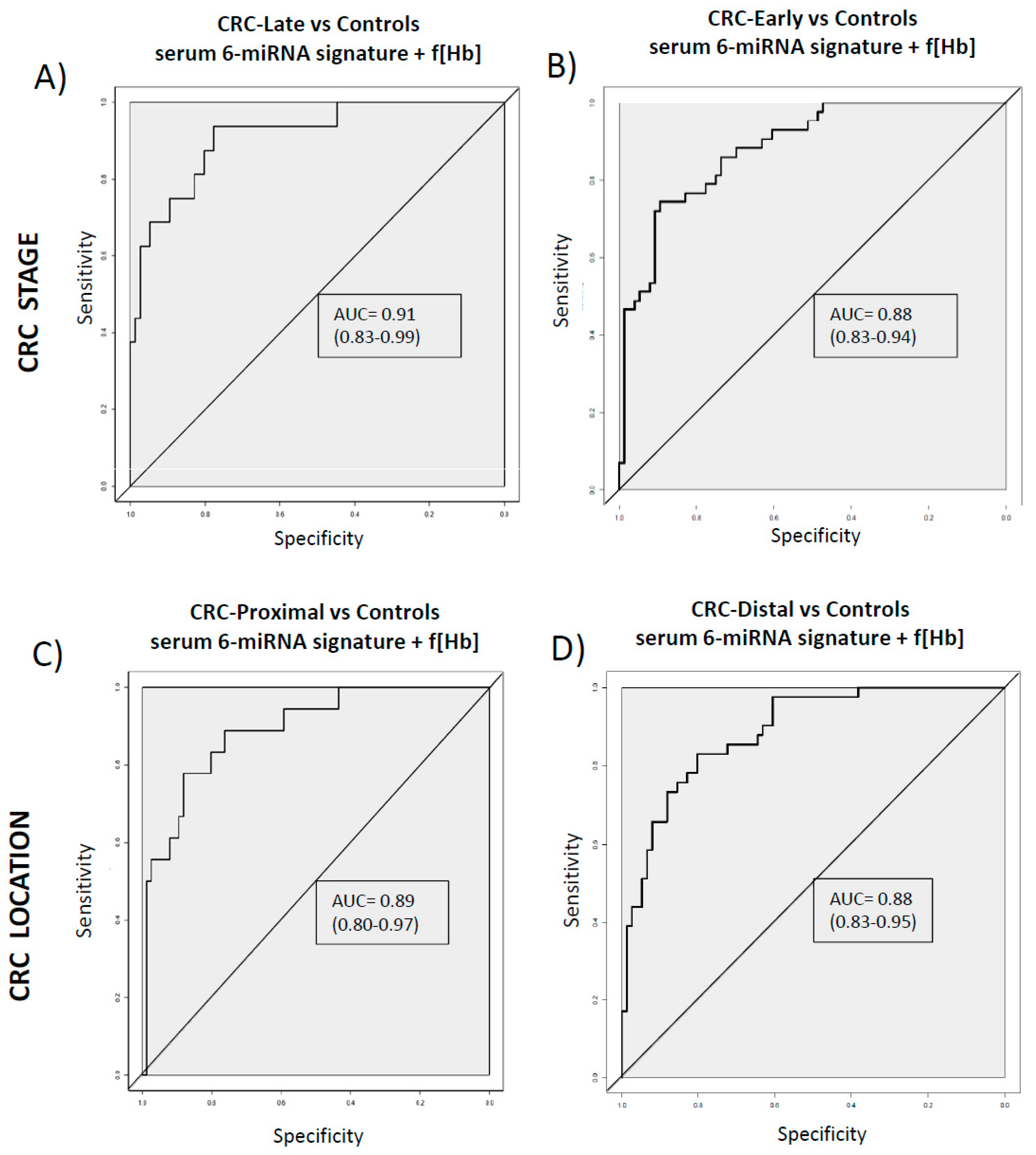
3.2. Diagnostic Performance of the Combined Signature Does not Depend on CRC Stage or Location
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Hofsli, E.; Sjursen, W.; Prestvik, W.S.; Johansen, J.; Rye, M.; Tranø, G.; Wasmuth, H.H.; Hatlevoll, I.; Thommesen, L. Identification of serum microRNA profiles in colon cancer. Br. J. Cancer 2013, 108, 1712–1719. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.J. Clinical uses of tumour markers: A critical review. Crit. Rev. Clin. Lab. Sci. 2001, 38, 225–262. [Google Scholar] [CrossRef] [PubMed]
- Quintero, E.; Castells, A.; Bujanda, L. Colonoscopy versus faecal immunochemical testing in colorectal-cancer screening. Gastroenterol. Endosc. 2012, 54, 1510. [Google Scholar]
- Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W.; García, F.A.R.; Gillman, M.W.; Harper, D.M.; Kemper, A.R.; Krist, A.H.; et al. Screening for colorectal cancer. JAMA 2016, 315, 2564. [Google Scholar]
- Jung, M.; Schaefer, A.; Steiner, I.; Kempkensteffen, C.; Stephan, C.; Erbersdobler, A.; Jung, K. Robust microRNA stability in degraded RNA preparations from human tissue and cell samples. Clin. Chem. 2010, 56, 998–1006. [Google Scholar] [CrossRef]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
- Diosdado, B.; van de Wiel, M.A.; Terhaar, J.S.; Droste, S.; Mongera, S.; Postma, C.; Meijerink, W.J.H.J.; Carvalho, B.; Meijer, G.A.; Terhaar Sive Droste, J.S.; et al. MiR-17-92 cluster is associated with 13q gain and c-myc expression during colorectal adenoma to adenocarcinoma progression. Br. J. Cancer 2009, 101, 707–714. [Google Scholar] [CrossRef]
- Giráldez, M.D.M.D.; Lozano, J.J.J.J.; Ramírez, G.; Hijona, E.; Bujanda, L.; Castells, A.; Gironella, M. Circulating microRNAs as biomarkers of colorectal cancer: Results from a genome-wide profiling and validation study. Clin. Gastroenterol. Hepatol. 2013, 11, 681–688. [Google Scholar] [CrossRef]
- Herreros-Villanueva, M.; Duran-Sanchon, S.; Martín, A.C.A.C.; Pérez-Palacios, R.; Vila-Navarro, E.; Marcuello, M.; Diaz-Centeno, M.; Cubiella, J.; Diez, M.S.M.S.; Bujanda, L.; et al. Plasma microRNA signature validation for early detection of colorectal cancer. Clin. Transl. Gastroenterol. 2019, 10, e00003. [Google Scholar] [CrossRef]
- Zekri, A.R.N.; Youssef, A.S.E.D.; Lotfy, M.M.; Gabr, R.; Ahmed, O.S.; Nassar, A.; Hussein, N.; Omran, D.; Medhat, E.; Eid, S.; et al. Circulating serum miRNAs as diagnostic markers for colorectal cancer. PLoS ONE 2016, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Huang, Z.; Zhu, D.; Zhou, X.; Shan, X.; Qi, L.; Wu, L.; Cheng, W.; Zhu, J.; Zhang, L.; et al. A panel of microRNA signature in serum for colorectal cancer diagnosis. Oncotarget 2017, 8, 17081–17091. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.-J.; Zhou, T.; Liu, Z.-L.; Tian, H.-P.; Xia, S.-S. Plasma miR-200c and miR-18a as potential biomarkers for the detection of colorectal carcinoma. Mol. Clin. Oncol. 2013, 1, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Huang, D.; Ni, S.; Peng, Z.; Sheng, W.; Du, X. Plasma microRNAs are promising novel biomarkers for early detection of colorectal cancer. Int. J. Cancer 2010, 127, 118–126. [Google Scholar] [CrossRef]
- Luo, X.; Stock, C.; Burwinkel, B.; Brenner, H. Identification and evaluation of plasma microRNAs for early detection of colorectal cancer. PLoS ONE 2013, 8, e62880. [Google Scholar] [CrossRef]
- Yamada, A.; Horimatsu, T.; Okugawa, Y.; Nishida, N.; Honjo, H.; Ida, H.; Kou, T.; Kusaka, T.; Sasaki, Y.; Yagi, M.; et al. Serum miR-21, miR-29a, and miR-125b are promising biomarkers for the early detection of colorectal neoplasia. Clin. Cancer Res. 2015, 21, 4234–4242. [Google Scholar] [CrossRef]
- Kanaan, Z.; Roberts, H.; Eichenberger, M.R.; Billeter, A.; Ocheretner, G.; Pan, J.; Rai, S.N.; Jorden, J.; Williford, A.; Galandiuk, S. A plasma MicroRNA panel for detection of colorectal adenomas: A step toward more precise screening for colorectal cancer. Ann. Surg. 2013, 258, 400–406. [Google Scholar] [CrossRef]
- Kirschner, M.B.; Edelman, J.J.B.; Kao, S.C.-H.; Vallely, M.P.; van Zandwijk, N.; Reid, G. The impact of hemolysis on cell-free microRNA biomarkers. Front. Genet. 2013, 4, 94. [Google Scholar] [CrossRef]
- Kirschner, M.B.; Kao, S.C.; Edelman, J.J.; Armstrong, N.J.; Vallely, M.P.; van Zandwijk, N.; Reid, G. Haemolysis during sample preparation alters microRNA content of plasma. PLoS ONE 2011, 6, e24145. [Google Scholar] [CrossRef]
- Fortunato, O.; Boeri, M.; Verri, C.; Conte, D.; Mensah, M.; Suatoni, P.; Pastorino, U.; Sozzi, G. Assessment of circulating micrornas in plasma of lung cancer patients. Molecules 2014, 19, 3038–3054. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, N.P.M.; Bos, A.C.R.K.; Lemmens, V.E.P.P.; Tanis, P.J.; Hugen, N.; Nagtegaal, I.D.; de Wilt, J.H.W.; Verhoeven, R.H.A. An overview of 25 years of incidence, treatment and outcome of colorectal cancer patients. Int. J. Cancer 2018, 143, 2758–2766. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Liles, E.G.; Bent, S.; Levin, T.R.; Corley, D.A. Accuracy of faecal immunochemical tests for colorectal cancer. Ann. Intern. Med. 2014, 160, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Hirai, H.W.; Tsoi, K.K.F.F.; Chan, J.Y.C.C.; Wong, S.H.; Ching, J.Y.L.L.; Wong, M.C.S.S.; Wu, J.C.Y.Y.; Chan, F.K.L.L.; Sung, J.J.Y.Y.; Ng, S.C. Systematic review with meta-analysis: Faecal occult blood tests show lower colorectal cancer detection rates in the proximal colon in colonoscopy-verified diagnostic studies. Aliment. Pharmacol. Ther. 2016, 43, 755–764. [Google Scholar] [CrossRef]
- Petrelli, F.; Tomasello, G.; Borgonovo, K.; Ghidini, M.; Turati, L.; Dallera, P.; Passalacqua, R.; Sgroi, G.; Barni, S. Prognostic survival associated with left-sided vs right-sided colon cancer a systematic review and meta-analysis. JAMA Oncol. 2017, 3, 211–219. [Google Scholar] [CrossRef]
- Missiaglia, E.; Jacobs, B.; D’Ario, G.; Di Narzo, A.F.; Soneson, C.; Budinska, E.; Popovici, V.; Vecchione, L.; Gerster, S.; Yan, P.; et al. Distal and proximal colon cancers differ in terms of molecular, pathological, and clinical features. Ann. Oncol. 2014, 25, 1995–2001. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, R.; Yang, F.; Cheng, R.; Chen, X.; Cui, S.; Gu, Y.; Sun, W.; You, C.; Liu, Z.; et al. miR-19a promotes colorectal cancer proliferation and migration by targeting TIA1. Mol. Cancer 2017, 16, 53. [Google Scholar] [CrossRef]
- Carter, J.V.; Galbraith, N.J.; Yang, D.; Burton, J.F.; Walker, S.P.; Galandiuk, S. Blood-based microRNAs as biomarkers for the diagnosis of colorectal cancer: A systematic review and meta-analysis. Br. J. Cancer 2017, 116, 762–774. [Google Scholar] [CrossRef]
- Huang, L.; Wang, X.; Wen, C.; Yang, X.; Song, M.; Chen, J.; Wang, C.; Zhang, B.; Wang, L.; Iwamoto, A.; et al. Hsa-MIR-19a is associated with lymph metastasis and mediates the TNF-α induced epithelial-to-mesenchymal transition in colorectal cancer. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef]
- Lu, Y.; Yang, H.; Yuan, L.; Liu, G.; Zhang, C.; Hong, M.; Liu, Y.; Zhou, M.; Chen, F.; Li, X. Overexpression of miR-335 confers cell proliferation and tumour growth to colorectal carcinoma cells. Mol. Cell. Biochem. 2016, 412, 235–245. [Google Scholar] [CrossRef]
- Zanutto, S.; Ciniselli, C.M.; Belfiore, A.; Lecchi, M.; Masci, E.; Delconte, G.; Primignani, M.; Tosetti, G.; Dal Fante, M.; Fazzini, L.; et al. Plasma miRNA-based signatures in CRC screening programmes. Int. J. Cancer 2019, 1–10. [Google Scholar] [CrossRef]
- Healy, N.A.A.; Heneghan, H.M.M.; Miller, N.; Osborne, C.K.K.; Schiff, R.; Kerin, M.J.J. Systemic mirnas as potential biomarkers for malignancy. Int. J. Cancer 2012, 131, 2215–2222. [Google Scholar] [CrossRef] [PubMed]
- McDonald, J.S.; Milosevic, D.; Reddi, H.V.; Grebe, S.K.; Algeciras-Schimnich, A. Analysis of circulating microRNA: Preanalytical and analytical challenges. Clin. Chem. 2011, 57, 833–840. [Google Scholar] [CrossRef] [PubMed]
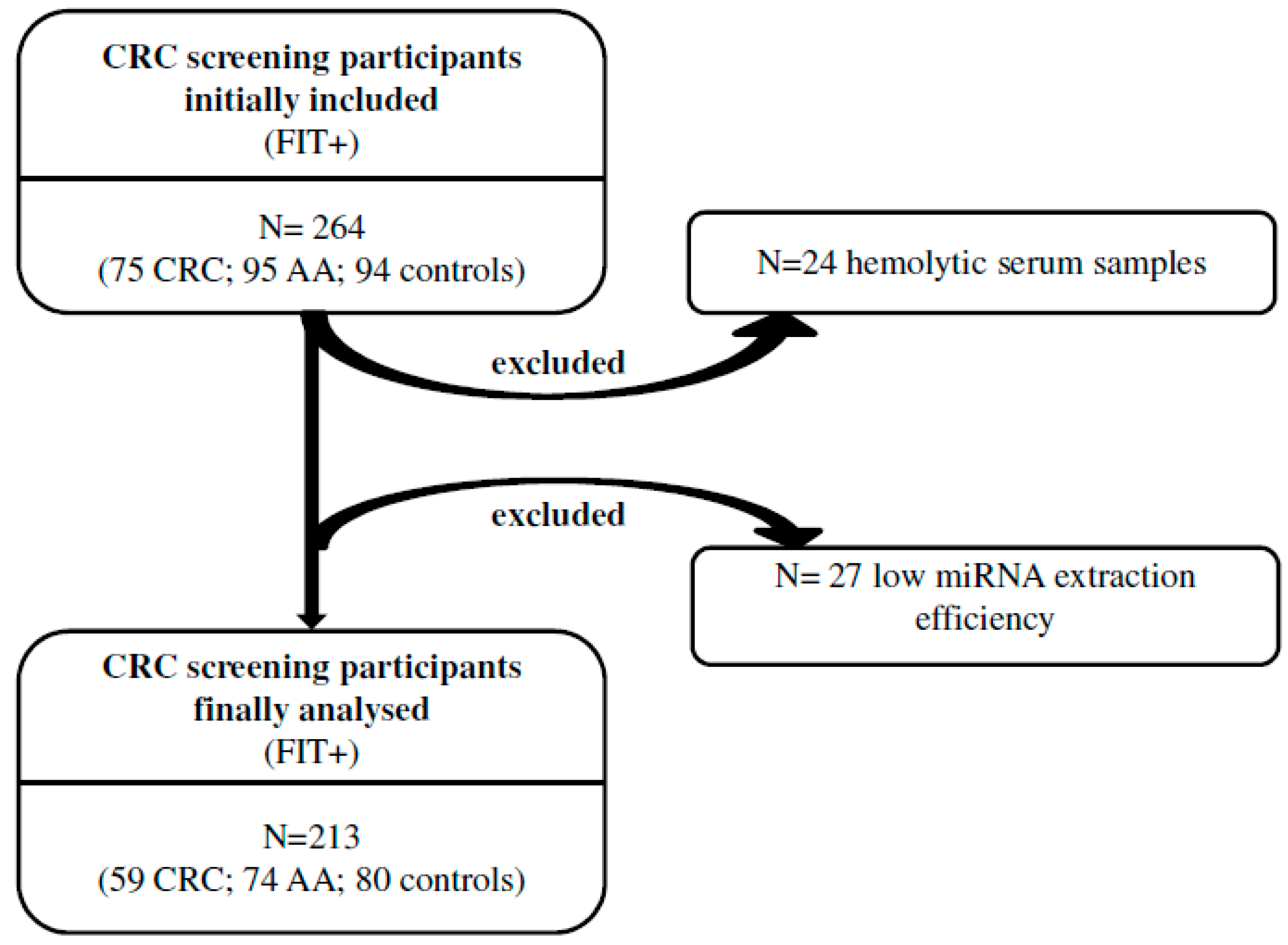
| Clinico-Pathological Characteristics | Total (n = 213) | Control (n = 80) | AA (n = 74) | CRC (n = 59) |
|---|---|---|---|---|
| Mean Age (SD) | 6186 (553) | 6202 (561) | 6154 (559) | 6205 (543) |
| Gender–Number | ||||
| Male | 150 | 55 | 51 | 44 |
| Female | 63 | 25 | 23 | 15 |
| CRC features | ||||
| TNM stage–Number | ||||
| I | 30 | |||
| II | 13 | |||
| Ш | 14 | |||
| Ⅳ | 2 | |||
| Location–Number | ||||
| Proximal | 18 | |||
| Distal | 41 | |||
| AA features | ||||
| High-grade dysplasia–Number | ||||
| Yes | 27 | |||
| No | 47 | |||
| Villous Component–Number | ||||
| Yes | 27 | |||
| No | 47 |
| Groups Compared | Serum 6-miRNA Signature | Faecal Hemoglobin Concentration | Serum 6-miRNA Signature + Faecal Haemoglobin Concentration | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AUC (95% CI) | SN (%) | SP (%) | PPV (%) | NPV (%) | AUC (95% CI) | SN (%) | SP (%) | PPV (%) | NPV (%) | AUC (95% CI) | SN (%) | SP (%) | PPV (%) | NPV (%) | |
| CRC versus Control | 0.74 (0.65–0.82) | 81 | 56 | 58 | 79 | 0.85 (0.78–0.91) | 81 | 73 | 69 | 84 | 0.88 (0.83–0.94) | 81 | 78 | 74 | 84 |
| AA versus Control | 0.80 (0.72–0.87) | 81 | 63 | 68 | 77 | 0.65 (0.56–0.73) | 81 | 35 | 54 | 67 | 0.81 (0.75–0.88) | 81 | 69 | 71 | 79 |
| CRCAA versus Control | 0.76 (0.69–0.82) | 80 | 57 | 76 | 62 | 0.73 (0.67–0.8) | 80 | 55 | 75 | 63 | 0.82 (0.76–0.88) | 80 | 68 | 81 | 66 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcuello, M.; Duran-Sanchon, S.; Moreno, L.; Lozano, J.J.; Bujanda, L.; Castells, A.; Gironella, M. Analysis of A 6-Mirna Signature in Serum from Colorectal Cancer Screening Participants as Non-Invasive Biomarkers for Advanced Adenoma and Colorectal Cancer Detection. Cancers 2019, 11, 1542. https://doi.org/10.3390/cancers11101542
Marcuello M, Duran-Sanchon S, Moreno L, Lozano JJ, Bujanda L, Castells A, Gironella M. Analysis of A 6-Mirna Signature in Serum from Colorectal Cancer Screening Participants as Non-Invasive Biomarkers for Advanced Adenoma and Colorectal Cancer Detection. Cancers. 2019; 11(10):1542. https://doi.org/10.3390/cancers11101542
Chicago/Turabian StyleMarcuello, María, Saray Duran-Sanchon, Lorena Moreno, Juan José Lozano, Luis Bujanda, Antoni Castells, and Meritxell Gironella. 2019. "Analysis of A 6-Mirna Signature in Serum from Colorectal Cancer Screening Participants as Non-Invasive Biomarkers for Advanced Adenoma and Colorectal Cancer Detection" Cancers 11, no. 10: 1542. https://doi.org/10.3390/cancers11101542
APA StyleMarcuello, M., Duran-Sanchon, S., Moreno, L., Lozano, J. J., Bujanda, L., Castells, A., & Gironella, M. (2019). Analysis of A 6-Mirna Signature in Serum from Colorectal Cancer Screening Participants as Non-Invasive Biomarkers for Advanced Adenoma and Colorectal Cancer Detection. Cancers, 11(10), 1542. https://doi.org/10.3390/cancers11101542






