Thyroid Cancer after Exposure to Radioiodine in Childhood and Adolescence: 131I-Related Risk and the Role of Selected Host and Environmental Factors
Abstract
1. Introduction
2. Results
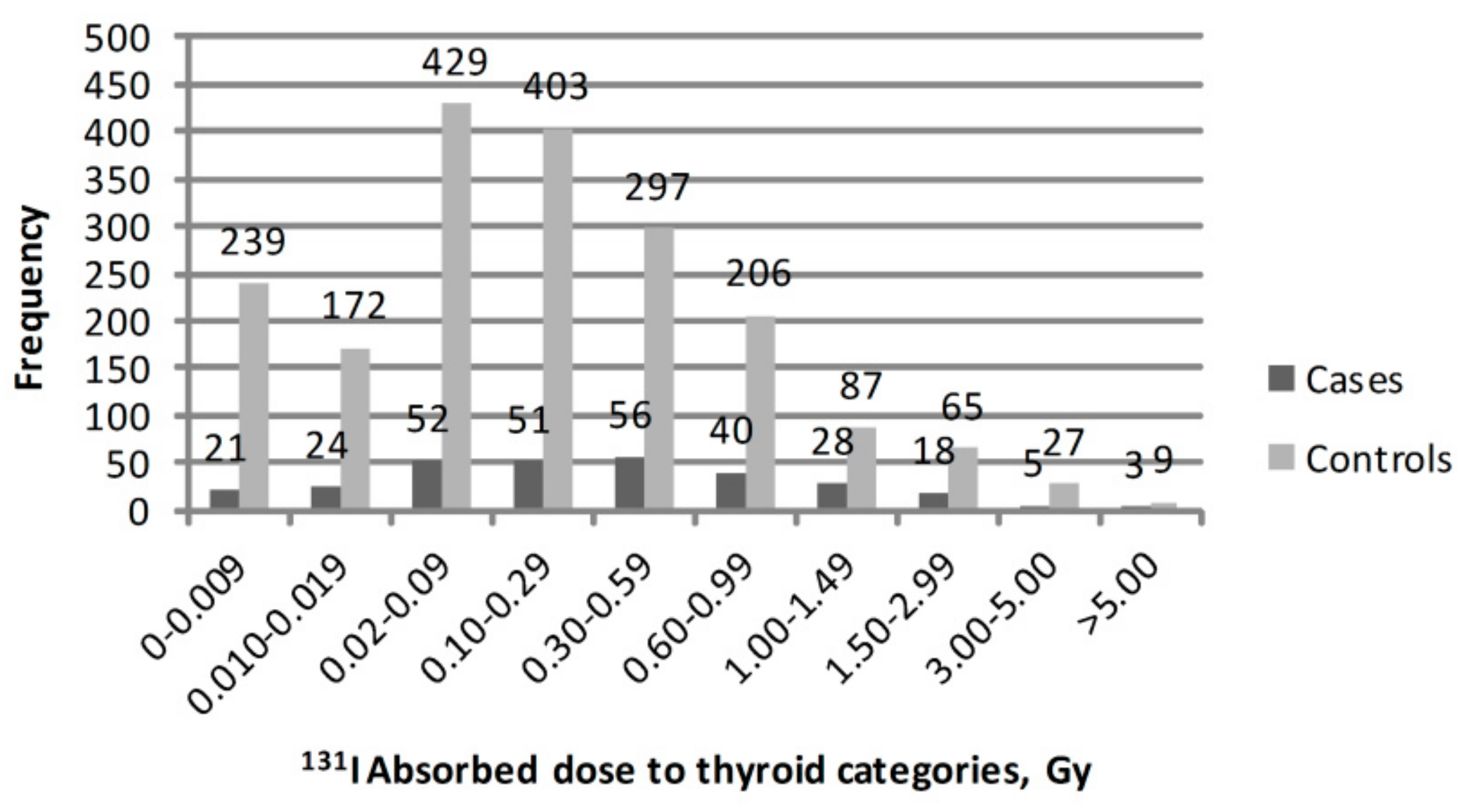
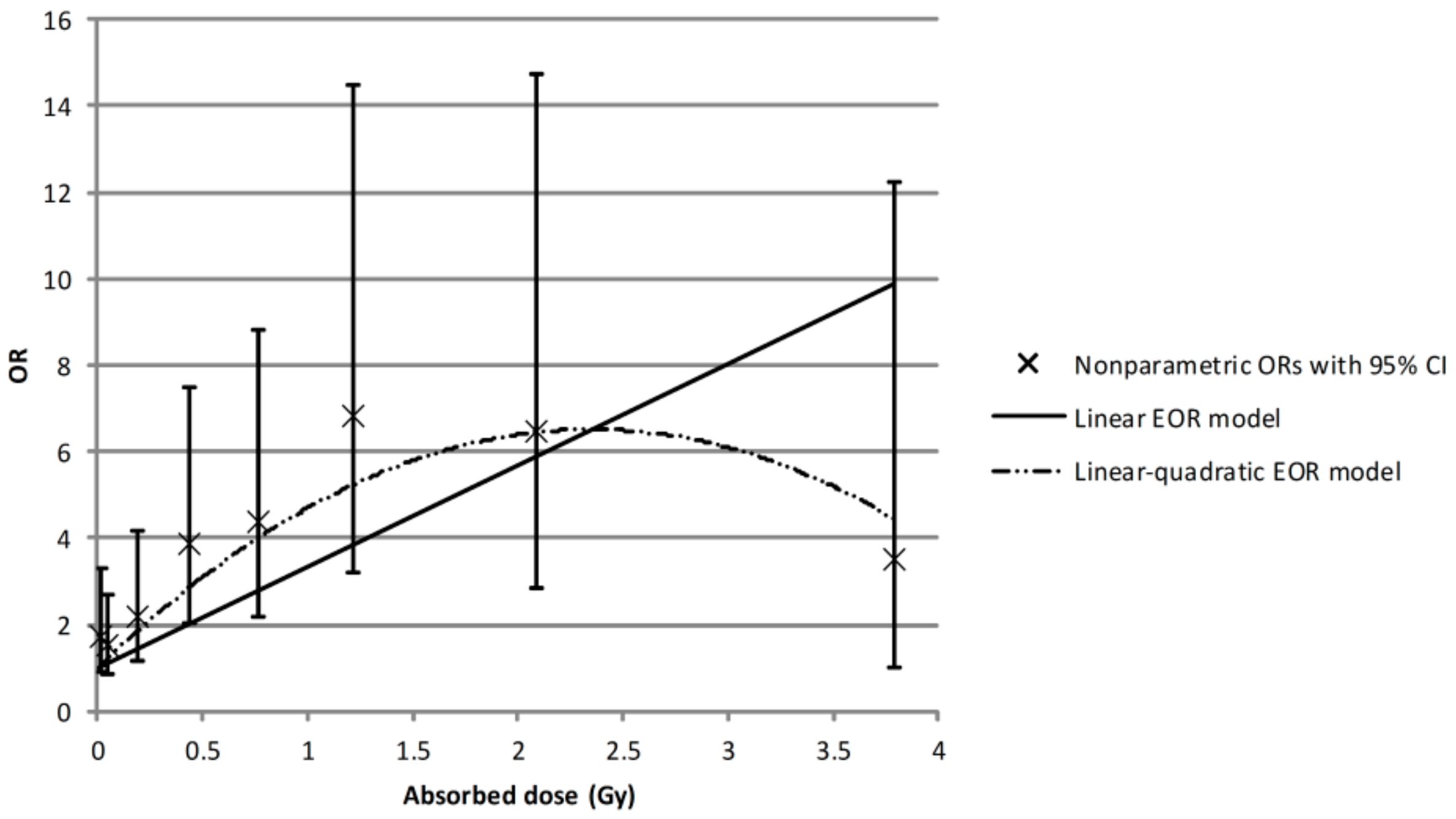
2.1. Association Between 131I Thyroid Dose and Thyroid Cancer Risk
2.2. Association Between Selected Non-Radiation Risk Factors and Thyroid Cancer
2.3. Effect Modification of the Radiation Dose Response
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Thyroid Dose Assessment
4.3. Study Indicators of Iodine Sufficiency
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cardis, E.; Kesminiene, A.; Ivanov, V.; Malakhova, I.; Shibata, Y.; Khrouch, V.; Drozdovitch, V.; Maceika, E.; Zvonova, I.; Vlassov, O.; et al. Risk of Thyroid Cancer after Exposure to 131 I in Childhood. JNCI J. Natl. Cancer Inst. 2005, 97, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Zablotska, L.B.; Ron, E.; Rozhko, A.V.; Hatch, M.; Polyanskaya, O.N.; Brenner, A.V.; Lubin, J.; Romanov, G.N.; McConnell, R.J.; O’Kane, P.; et al. Thyroid cancer risk in Belarus among children and adolescents exposed to radioiodine after the Chornobyl accident. Br. J. Cancer 2011, 104, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Brenner, A.V.; Tronko, M.D.; Hatch, M.; Bogdanova, T.I.; Oliynik, V.A.; Lubin, J.H.; Zablotska, L.B.; Tereschenko, V.P.; McConnell, R.J.; Zamotaeva, G.A.; et al. I-131 dose response for incident thyroid cancers in Ukraine related to the Chornobyl accident. Environ. Health Perspect. 2011, 119, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, K.; Preston, D.; Funamoto, S.; Yonehara, S.; Ito, M.; Tokuoka, S.; Sugiyama, H.; Soda, M.; Ozasa, K.; Mabuchi, K. Long-Term trend of thyroid cancer risk among Japanese Atomic-Bomb survivors: 60 years after exposure. Int. J. Cancer 2013, 132, 1222–1226. [Google Scholar] [CrossRef] [PubMed]
- Veiga, L.H.S.; Holmberg, E.; Anderson, H.; Pottern, L.; Sadetzki, S.; Adams, M.J.; Sakata, R.; Schneider, A.B.; Inskip, P.; Bhatti, P.; et al. Thyroid Cancer after Childhood Exposure to External Radiation: An Updated Pooled Analysis of 12 Studies. Radiat. Res. 2016, 185, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Tronko, M.; Brenner, A.V.; Bogdanova, T.; Shpak, V.; Oliynyk, V.; Cahoon, E.K.; Drozdovitch, V.; Little, M.P.; Tereshchenko, V.; Zamotayeva, G.; et al. Thyroid neoplasia risk is increased nearly 30 years after the Chernobyl accident. Int. J. Cancer 2017, 141, 1585–1588. [Google Scholar] [CrossRef]
- Ostroumova, E.; Rozhko, A.; Hatch, M.; Furukawa, K.; Polyanskaya, O.; McConnell, R.J.; Nadyrov, E.; Petrenko, S.; Romanov, G.; Yauseyenka, V.; et al. Measures of thyroid function among Belarusian children and adolescents exposed to iodine-131 from the accident at the Chernobyl nuclear plant. Environ. Health Perspect. 2013, 121, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Zablotska, L.B.; Nadyrov, E.A.; Polyanskaya, O.N.; McConnell, R.J.; O’Kane, P.; Lubin, J.; Hatch, M.; Little, M.P.; Brenner, A.V.; Veyalkin, I.V.; et al. Risk of thyroid follicular adenoma among children and adolescents in Belarus exposed to iodine-131 after the Chornobyl accident. Am. J. Epidemiol. 2015, 182, 781–790. [Google Scholar] [CrossRef]
- Cahoon, E.K.; Nadyrov, E.A.; Polyanskaya, O.N.; Yauseyenka, V.V.; Veyalkin, I.V.; Yeudachkova, T.I.; Maskvicheva, T.I.; Minenko, V.F.; Liu, W.; Drozdovitch, V.; et al. Risk of Thyroid Nodules in Residents of Belarus Exposed to Chernobyl Fallout as Children and Adolescents. J. Clin. Endocrinol. Metab. 2017, 102, 2207–2217. [Google Scholar] [CrossRef]
- Greene, F.L.; Page, D.L.; Fleming, I.D.; Fritz, A.G.; Balch, C.M.; Haller, D.G.; Morrow, M. (Eds.) AJCC Cancer Staging Manual, 6th ed.; Springer Science + Business Media: New York, NY, USA, 2002. [Google Scholar]
- Stezhko, V.A.; Buglova, E.E.; Danilova, L.I.; Drozd, V.M.; Krysenko, N.A.; Lesnikova, N.R.; Minenko, V.F.; Ostapenko, V.A.; Petrenko, S.V.; Polyanskaya, O.N.; et al. A cohort study of thyroid cancer and other thyroid diseases after the Chornobyl accident: Objectives, design and methods. Radiat. Res. 2004, 161, 481–492. [Google Scholar] [CrossRef]
- Tronko, M.D.; Howe, G.R.; Bogdanova, T.I.; Bouville, A.C.; Epstein, O.V.; Brill, A.B.; Likhtarev, I.A.; Fink, D.J.; Markov, V.V.; Greenebaum, E.; et al. A Cohort Study of Thyroid Cancer and Other Thyroid Diseases After the Chornobyl Accident: Thyroid Cancer in Ukraine Detected During First Screening. JNCI J. Natl. Cancer Inst. 2006, 98, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Land, C.E.; Kwon, D.; Hoffman, F.O.; Moroz, B.; Drozdovitch, V.; Bouville, A.; Beck, H.; Luckyanov, N.; Weinstock, R.M.; Simon, S.L. Accounting for shared and unshared dosimetric uncertainties in the dose response for ultrasound-detected thyroid nodules after exposure to radioactive fallout. Radiat. Res. 2015, 183, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Khokhryakov, V.; Drozhko, E.; Glagolenko, Y.; Rovny, S.; Vasilenko, E.; Suslov, A.; Anspaugh, L.; Napier, B.; Bouville, A.; Khokhryakov, V.; et al. Studies on the Ozyorsk population: Dosimetry. Radiat. Environ. Biophys. 2002, 41, 33–35. [Google Scholar] [CrossRef] [PubMed]
- Mushkacheva, G.; Rabinovich, E.; Privalov, V.; Povolotskaya, S.; Shorokhova, V.; Sokolova, S.; Turdakova, V.; Ryzhova, E.; Hall, P.; Schneider, A.B.; et al. Thyroid Abnormalities Associated with Protracted Childhood Exposure to 131I from Atmospheric Emissions from the Mayak Weapons Facility in Russia. Radiat. Res. 2006, 166, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Kitahara, C.M.; Gamborg, M.; Berrington de González, A.; Sørensen, T.I.A.; Baker, J.L. Childhood height and body mass index were associated with risk of adult thyroid cancer in a large cohort study. Cancer Res. 2014, 74, 235–242. [Google Scholar] [CrossRef]
- Brindel, P.; Doyon, F.; Rachédi, F.; Boissin, J.-L.; Sebbag, J.; Shan, L.; Chungue, V.; Bost-Bezeaud, F.; Petitdidier, P.; Paoaafaite, J.; et al. Anthropometric factors in differentiated thyroid cancer in French Polynesia: A Case–Control study. Cancer Causes Control. 2009, 20, 581–590. [Google Scholar] [CrossRef]
- Popoveniuc, G.; Jonklaas, J. Thyroid nodules. Med. Clin. N. Am. 2012, 96, 329–349. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Hatch, M.; Polyanskaya, O.; McConnell, R.; Gong, Z.; Drozdovitch, V.; Rozhko, A.; Prokopovich, A.; Petrenko, S.; Brenner, A.; Zablotska, L. Urinary Iodine and Goiter Prevalence in Belarus: Experience of the Belarus-American cohort study of thyroid cancer and other thyroid diseases following the Chornobyl nuclear accident. Thyroid 2011, 21, 429–437. [Google Scholar] [CrossRef]
- WHO; UNICEF; ICCIDD. Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers, 3rd ed.; WHO Press: Geneva, Switzerland, 2007; ISBN 978-92-4-159582-7. [Google Scholar]
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). Sources and Effects of Ionizing Radiation-Vol. II Effects; United Nations: New York, NY, USA, 2000. [Google Scholar]
- Drozdovitch, V.; Kesminiene, A.; Moissonnier, M.; Veyalkin, I.; Ostroumova, E. Uncertainties in radiation doses for a Case-Control study of thyroid cancer among residents exposed in childhood to 131I from Chernobyl fallout. Health Phys. 2019, in press. [Google Scholar]
- Drozdovitch, V.; Khrouch, V.; Maceika, E.; Zvonova, I.; Vlasov, O.; Bratilova, A.; Gavrilin, Y.; Goulko, G.; Hoshi, M.; Kesminiene, A.; et al. Reconstruction of radiation doses in a Case-Control study of thyroid cancer following the Chernobyl accident. Health Phys. 2010, 99, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Skryabin, A.M.; Drozdovitch, V.; Belsky, Y.; Leshcheva, S.V.; Mirkhaidarov, A.K.; Voillequé, P.; Luckyanov, N.; Bouville, A. Thyroid mass in children and adolescents living in the most exposed areas to Chernobyl fallout in Belarus. Radiat. Prot. Dosim. 2010, 142, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Shibata, Y. Proceedings of the Fifth Chernobyl Sasakawa Medical Cooperation Symposium, Kyiv, Ukraine, 14–15 October 1996; Chernobyl: A decade: Amsterdam, The Netherlands, 1997. [Google Scholar]
- Drozdovitch, V.; Zhukova, O.; Germenchuk, M.; Khrutchinsky, A.; Kukhta, T.; Luckyanov, N.; Minenko, V.; Podgaiskaya, M.; Savkin, M.; Vakulovsky, S.; et al. Database of meteorological and radiation measurements made in Belarus during the first three months following the Chernobyl accident. J. Environ. Radioact. 2013, 116, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Drozdovitch, V.; Minenko, V.; Golovanov, I.; Khrutchinsky, A.; Kukhta, T.; Kutsen, S.; Luckyanov, N.; Ostroumova, E.; Trofimik, S.; Voillequé, P.; et al. Thyroid Dose Estimates for a Cohort of Belarusian Children Exposed to (131) I from the Chernobyl Accident: Assessment of Uncertainties. Radiat. Res. 2015, 184, 203–218. [Google Scholar] [CrossRef] [PubMed]
- Korobova, E.; Anoshko, Y.; Kesminiene, A.; Kouvyline, A.; Romanov, S.; Tenet, V.; Suonio, E.; Cardis, E. Evaluation of stable iodine status of the areas affected by the Chernobyl accident in an epidemiological study in Belarus and the Russian Federation. Selenium Iodine Anom. Soils Health 2010, 107, 124–135. [Google Scholar] [CrossRef]
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a WHO growth reference for School-Aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef]
- Preston, D.L.; Lubin, J.H.; Pierce, D.A.; McConney, M.E. Epicure User’s Guide; Hirosoft International Corporation: Seattle, WA, USA, 1993. [Google Scholar]
| Characteristics | Cases | Controls | Total |
|---|---|---|---|
| Total | 298 | 1934 | 2232 |
| Sex | |||
| Male | 109 | 700 | 809 |
| Female | 189 | 1234 | 1423 |
| Country/Region | |||
| Belarus | |||
| Gomel | 192 | 1239 | 1431 |
| Mogilev | 36 | 230 | 266 |
| Russian Federation | |||
| Bryansk | 11 | 74 | 85 |
| Kaluga | 10 | 60 | 70 |
| Orel | 27 | 187 | 214 |
| Tula | 22 | 144 | 166 |
| Age at exposure, years | |||
| <2 | 89 | 699 | 788 |
| 2–4 | 84 | 520 | 604 |
| 5–9 | 67 | 364 | 431 |
| 10–14 | 44 | 247 | 291 |
| 15–18 | 14 | 104 | 118 |
| Region | Median Dose for Cases | Median Dose for Controls | Mean Dose for Cases | Mean Dose for Controls |
|---|---|---|---|---|
| Belarus | ||||
| Gomel | 0.51 | 0.31 | 0.77 | 0.58 |
| Mogilev | 0.11 | 0.056 | 0.48 | 0.14 |
| Russian Federation | ||||
| Bryansk | 0.35 | 0.086 | 0.80 | 0.40 |
| Kaluga | 0.011 | 0.019 | 0.031 | 0.031 |
| Orel | 0.014 | 0.018 | 0.028 | 0.031 |
| Tula | 0.019 | 0.021 | 0.037 | 0.036 |
| Total | 0.31 | 0.15 | 0.59 | 0.41 |
| Dose Category, Gy | Mean Dose, Gy | Number of Cases | OR * | 95% CI |
|---|---|---|---|---|
| 0–0.009 | 0.005 | 21 | 1.00 | Reference |
| 0.010–0.019 | 0.014 | 24 | 1.71 | 0.89; 3.29 |
| 0.02–0.09 | 0.05 | 52 | 1.51 | 0.85; 2.70 |
| 0.10–0.29 | 0.19 | 50 | 2.21 | 1.17; 4.19 |
| 0.30–0.59 | 0.43 | 56 | 3.88 | 2.01; 7.50 |
| 0.60–0.99 | 0.77 | 40 | 4.38 | 2.17; 8.83 |
| 1.00–1.49 | 1.22 | 28 | 6.83 | 3.22; 14.48 |
| 1.50–2.99 | 2.10 | 18 | 6.49 | 2.86; 14.75 |
| 3.00–5.00 | 3.77 | 5 | 3.53 | 1.02; 12.22 |
| Variable | N of Cases (%) | N of Controls (%) | OR a (95% CI) |
|---|---|---|---|
| Personal history of thyroid diseases | |||
| Any thyroid disease except thyroid cancer | |||
| Never had | 246 (87.2) | 1680 (92.6) | 1.00 (Referent) |
| Ever had | 36 (12.8) | 134 (7.4) | 1.98 (1.30; 3.00) |
| Benign nodules | |||
| no | 270 (95.7) | 1808 (99.7) | 1.00 (Referent) |
| yes | 12 (4.3) | 6 (0.3) | 14.26 (4.50; 45.18) |
| Goiter | |||
| no | 246 (92.1) | 1680 (93.4) | 1.00 (Referent) |
| yes | 21 (7.9) | 118 (6.6) | 1.29 (0.78; 2.15) |
| Family history of thyroid diseases | |||
| Thyroid cancer | |||
| no | 266 (96.7) | 1745 (99.0) | 1.00 (Referent) |
| yes | 9 (3.3) | 18 (1.0) | 3.37 (1.38; 8.24) |
| Thyroid or other endocrine disease b | |||
| no | 200 (74.4) | 1322 (77.2) | 1.00 (Referent) |
| yes | 69 (25.6) | 391 (22.8) | 1.22 (0.90; 1.66) |
| Nodule | |||
| no | 248 (94.3) | 1639 (96.3) | 1.00 (Referent) |
| yes | 15 (5.7) | 63 (3.7) | 1.51 (0.82; 2.77) |
| Goiter | |||
| no | 228 (87.0) | 1536 (90.3) | 1.00 (Referent) |
| yes | 34 (13.0) | 166 (9.7) | 1.33 (0.88; 2.00) |
| Anthropomorphic factor | |||
| BMI | |||
| Normal | 117 (73.6) | 1385 (84.4) | 1.00 (Referent) |
| Overweight | 42 (26.4) | 257 (15.7) | 1.87 (1.24; 2.82) |
| Stabile iodine status | |||
| Iodine supplementation | |||
| no | 241 (91.3) | 1304 (83.1) | 1.00 (Referent) |
| yes | 23 (8.7) | 265 (16.9) | 0.41 (0.25; 0.66) |
| Stabile iodine intake status at the time of the accident c | |||
| Deficient | 118 (41.8) | 548 (29.8) | 1.63 (1.20; 2.21) |
| Sufficient | 164 (58.2) | 1293 (70.2) | 1.00 (Referent) |
| Variable | N of Thyroid Cancer Cases | OR at 1 Gy (95% CI) a | p-Value for Heterogeneity |
|---|---|---|---|
| Sex | |||
| men | 102 | 3.01 (1.76; 5.14) | 0.78 |
| women | 180 | 3.33 (2.06; 5.36) | |
| Age at the accident, years | |||
| ≤1.5 | 65 | 2.34 (1.41; 3.92) | |
| 1.6–3.0 | 61 | 3.03 (1.74; 5.25) | 0.15 |
| 3.1–8.0 | 87 | 6.22 (3.11; 12.44) | |
| 8.1+ | 69 | 3.17 (0.90; 11.17) | |
| Personal history of thyroid disease except thyroid cancer | |||
| Never had | 246 | 3.44 (2.37; 4.98) | |
| Ever had | 36 | 4.34 (1.98; 9.49) | 0.16 |
| Personal history of benign nodules | |||
| no | 270 | 3.03 (2.12; 4.34) | |
| yes | 12 | 32.61 (5.77; 184.18) | 0.68 |
| Family history of thyroid cancer | |||
| no | 266 | 3.21 (2.23; 4.61) | |
| yes | 9 | 8.31 (2.11; 32.79) | 0.62 |
| BMI | |||
| Normal | 117 | 3.78 (2.19; 6.55) | |
| Overweight | 42 | 6.80 (2.72; 16.96) | 0.92 |
| Iodine supplementation | |||
| no | 241 | 3.62 (2.43; 5.40) | |
| yes | 23 | 0.65 (0.23; 1.81) | 0.05 |
| Stabile iodine intake status at the time of the accident b | |||
| Deficient | 118 | 4.65 (3.01; 7.18) | 0.90 |
| Sufficient | 164 | 2.80 (1.66; 4.70) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zupunski, L.; Ostroumova, E.; Drozdovitch, V.; Veyalkin, I.; Ivanov, V.; Yamashita, S.; Cardis, E.; Kesminiene, A. Thyroid Cancer after Exposure to Radioiodine in Childhood and Adolescence: 131I-Related Risk and the Role of Selected Host and Environmental Factors. Cancers 2019, 11, 1481. https://doi.org/10.3390/cancers11101481
Zupunski L, Ostroumova E, Drozdovitch V, Veyalkin I, Ivanov V, Yamashita S, Cardis E, Kesminiene A. Thyroid Cancer after Exposure to Radioiodine in Childhood and Adolescence: 131I-Related Risk and the Role of Selected Host and Environmental Factors. Cancers. 2019; 11(10):1481. https://doi.org/10.3390/cancers11101481
Chicago/Turabian StyleZupunski, Ljubica, Evgenia Ostroumova, Vladimir Drozdovitch, Ilya Veyalkin, Viktor Ivanov, Shunichi Yamashita, Elisabeth Cardis, and Ausrele Kesminiene. 2019. "Thyroid Cancer after Exposure to Radioiodine in Childhood and Adolescence: 131I-Related Risk and the Role of Selected Host and Environmental Factors" Cancers 11, no. 10: 1481. https://doi.org/10.3390/cancers11101481
APA StyleZupunski, L., Ostroumova, E., Drozdovitch, V., Veyalkin, I., Ivanov, V., Yamashita, S., Cardis, E., & Kesminiene, A. (2019). Thyroid Cancer after Exposure to Radioiodine in Childhood and Adolescence: 131I-Related Risk and the Role of Selected Host and Environmental Factors. Cancers, 11(10), 1481. https://doi.org/10.3390/cancers11101481






