Kinases and Cancer
Data Availability
Acknowledgments
Conflicts of Interest
References
- Hunter, T.; Cooper, J.A. Protein-tyrosine kinases. Annu. Rev. Biochem. 1985, 54, 897–930. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, P.; Michel, P.A.; Zahn-Zabal, M.; Britan, A.; Cusin, I.; Domagalski, M.; Duek, P.D.; Gateau, A.; Gleizes, A.; Hinard, V.; et al. The nextprot knowledgebase on human proteins: 2017 update. Nucleic Acids Res. 2017, 45, D177–D182. [Google Scholar] [CrossRef] [PubMed]
- Nukaga, S.; Yasuda, H.; Tsuchihara, K.; Hamamoto, J.; Masuzawa, K.; Kawada, I.; Naoki, K.; Matsumoto, S.; Mimaki, S.; Ikemura, S.; et al. Amplification of egfr wild-type alleles in non-small cell lung cancer cells confers acquired resistance to mutation-selective egfr tyrosine kinase inhibitors. Cancer Res. 2017, 77, 2078–2089. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Zeng, Z.; Shia, J.; Paty, P.B. Egfr gene amplification and kras mutation predict response to combination targeted therapy in metastatic colorectal cancer. Pathol. Oncol. Res. 2017, 23, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Chang, N.; Lee, H.W.; Lim, J.E.; Jeong, D.E.; Song, H.J.; Kim, S.; Nam, D.H.; Sung, H.H.; Jeong, B.C.; Seo, S.I.; et al. Establishment and antitumor effects of dasatinib and pki-587 in bd-138t, a patient-derived muscle invasive bladder cancer preclinical platform with concomitant egfr amplification and pten deletion. Oncotarget 2016, 7, 51626–51639. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Zhu, L.; Ji, J.; Ding, F.; Wang, C.; Cai, Q.; Yu, Y.; Zhu, Z.; Zhang, J. Egfr high expression, but not kras status, predicts sensitivity of pancreatic cancer cells to nimotuzumab treatment in vivo. Curr. Cancer Drug Targets 2017, 17, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.Y.; Choi, Y.L.; Han, J.J.; Kim, K.M.; Oh, Y.L. Expression and amplification of her2, egfr and cyclin d1 in breast cancer: Immunohistochemistry and chromogenic in situ hybridization. Pathol. Int. 2008, 58, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Morey, A.L.; Brown, B.; Farshid, G.; Fox, S.B.; Francis, G.D.; McCue, G.; von Neumann-Cosel, V.; Bilous, M. Determining her2 (erbb2) amplification status in women with breast cancer: Final results from the australian in situ hybridisation program. Pathology 2016, 48, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Pasch, S.; Schamberger, T.; Maneck, M.; Mohlendick, B.; Schumacher, S.; Brockhoff, G.; Knoefel, W.T.; Izbicki, J.; Polzer, B.; et al. Diagnostic pathology of early systemic cancer: Erbb2 gene amplification in single disseminated cancer cells determines patient survival in operable esophageal cancer. Int. J. Cancer 2017, 142, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; Yang, B.F.; Yun, T.; Zhu, C.Y.; Li, C.Y.; Jiang, B.; Wang, G.P.; Wang, S.N.; Li, Y.Y.; Zhu, M.L. Methods and significance of the combined detection of her2 gene amplification and chemosensitivity in gastric cancer. Cancer Biomark. 2017, 21, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Han, C.P.; Hsu, J.D.; Yao, C.C.; Lee, M.Y.; Ruan, A.; Tyan, Y.S.; Yang, S.F.; Chiang, H. Her2 gene amplification in primary mucinous ovarian cancer: A potential therapeutic target. Histopathology 2010, 57, 763–764. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.W.; Wang, W.X.; Wu, M.J.; Zhu, Y.C.; Zhuang, W.; Lin, G.; Du, K.Q.; Huang, Y.J.; Chen, Y.P.; Chen, G.; et al. Comparison of the c-met gene amplification between primary tumor and metastatic lymph nodes in non-small cell lung cancer. Thorac. Cancer 2017, 8, 417–422. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Wang, F.; Shao, Q.; Wang, F.H.; Wang, Z.Q.; Wang, Z.Q.; Chen, C.; Li, C.; Luo, H.Y.; Zhang, D.S.; et al. Met amplification is not rare and predicts unfavorable clinical outcomes in patients with recurrent/metastatic gastric cancer after chemotherapy. Cancer 2014, 120, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Di Renzo, M.F.; Olivero, M.; Giacomini, A.; Porte, H.; Chastre, E.; Mirossay, L.; Nordlinger, B.; Bretti, S.; Bottardi, S.; Giordano, S.; et al. Overexpression and amplification of the met/hgf receptor gene during the progression of colorectal cancer. Clin. Cancer Res. 1995, 1, 147–154. [Google Scholar] [PubMed]
- Miwa, W.; Yasuda, J.; Murakami, Y.; Yashima, K.; Sugano, K.; Sekine, T.; Kono, A.; Egawa, S.; Yamaguchi, K.; Hayashizaki, Y.; et al. Isolation of DNA sequences amplified at chromosome 19q13.1-q13.2 including the akt2 locus in human pancreatic cancer. Biochem. Biophys. Res. Commun. 1996, 225, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.Q.; Godwin, A.K.; Bellacosa, A.; Taguchi, T.; Franke, T.F.; Hamilton, T.C.; Tsichlis, P.N.; Testa, J.R. Akt2, a putative oncogene encoding a member of a subfamily of protein-serine/threonine kinases, is amplified in human ovarian carcinomas. Proc. Natl. Acad. Sci. USA 1992, 89, 9267–9271. [Google Scholar] [CrossRef] [PubMed]
- Alterio, D.; Marvaso, G.; Maffini, F.; Gandini, S.; Chiocca, S.; Ferrari, A.; Preda, L.; Rocca, M.C.; Lepanto, D.; Fodor, C.; et al. Role of egfr as prognostic factor in head and neck cancer patients treated with surgery and postoperative radiotherapy: Proposal of a new approach behind the egfr overexpression. Med. Oncol. 2017, 34, 107. [Google Scholar] [CrossRef] [PubMed]
- Curigliano, G.; Viale, G.; Bagnardi, V.; Fumagalli, L.; Locatelli, M.; Rotmensz, N.; Ghisini, R.; Colleoni, M.; Munzone, E.; Veronesi, P.; et al. Clinical relevance of her2 overexpression/amplification in patients with small tumor size and node-negative breast cancer. J. Clin. Oncol. 2009, 27, 5693–5699. [Google Scholar] [CrossRef] [PubMed]
- Hou, F.; Yuan, W.; Huang, J.; Qian, L.; Chen, Z.; Ge, J.; Wu, S.; Chen, J.; Wang, J.; Chen, Z. Overexpression of epha2 correlates with epithelial-mesenchymal transition-related proteins in gastric cancer and their prognostic importance for postoperative patients. Med. Oncol. 2012, 29, 2691–2700. [Google Scholar] [CrossRef] [PubMed]
- Archewa, P.; Pata, S.; Chotjumlong, P.; Supanchart, C.; Krisanaprakornkit, S.; Iamaroon, A. Akt2 and p-akt overexpression in oral cancer cells is due to a reduced rate of protein degradation. J. Investig. Clin. Dent. 2017, 8, e12194. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J. The potential role of the egfr/erbb2 heterodimer in breast cancer. Expert Opin. Ther. Pat. 2007, 17, 6. [Google Scholar] [CrossRef]
- Kanematsu, T.; Yano, S.; Uehara, H.; Bando, Y.; Sone, S. Phosphorylation, but not overexpression, of epidermal growth factor receptor is associated with poor prognosis of non-small cell lung cancer patients. Oncol. Res. 2003, 13, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Urban, P.; Kung, W.; Vuaroqueaux, V.; Labuhn, M.; Wight, E.; Eppenberger, U.; Eppenberger-Castori, S. Phosphorylation of tyrosine 1248-erbb2 measured by chemiluminescence-linked immunoassay is an independent predictor of poor prognosis in primary breast cancer patients. Eur. J. Cancer 2006, 42, 636–645. [Google Scholar] [CrossRef] [PubMed]
- DiGiovanna, M.P.; Stern, D.F.; Edgerton, S.M.; Whalen, S.G.; Moore, D., 2nd; Thor, A.D. Relationship of epidermal growth factor receptor expression to erbb-2 signaling activity and prognosis in breast cancer patients. J. Clin. Oncol. 2005, 23, 1152–1160. [Google Scholar] [CrossRef] [PubMed]
- Milde-Langosch, K.; Bamberger, A.M.; Rieck, G.; Grund, D.; Hemminger, G.; Muller, V.; Loning, T. Expression and prognostic relevance of activated extracellular-regulated kinases (erk1/2) in breast cancer. Br. J. Cancer 2005, 92, 2206–2215. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, S.; Kudo, Y.; Ogawa, I.; Tatsuka, M.; Kawai, H.; Pagano, M.; Takata, T. Constitutive phosphorylation of aurora-a on ser51 induces its stabilization and consequent overexpression in cancer. PLoS ONE 2007, 2, e944. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.J.; Wan, X.B.; Fu, X.H.; Wu, P.H.; Chen, D.K.; Wang, P.N.; Jiang, L.; Wang, D.H.; Chen, Z.T.; Huang, Y.; et al. Phosphorylated p38, a negative prognostic biomarker, complements tnm staging prognostication in colorectal cancer. Tumour Biol. 2014, 35, 10487–10495. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Urban, P.; Vuaroqueaux, V.; Labuhn, M.; Kung, W.; Wight, E.; Mayhew, M.; Eppenberger, U.; Eppenberger-Castori, S. Increased level of phosphorylated akt measured by chemiluminescence-linked immunosorbent assay is a predictor of poor prognosis in primary breast cancer overexpressing erbb-2. Breast Cancer Res. 2005, 7, R394–R401. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J. The potential role of akt phosphorylation in human cancers. Int. J. Biol. Markers 2008, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Kung, W.; Eppenberger, U.; Eppenberger-Castori, S. Increased level of phosphorylated shca measured by chemiluminescence-linked immunoassay is a predictor of good prognosis in primary breast cancer expressing low levels of estrogen receptor. Cancers 2010, 2, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Chen, J.S.; Zhou, X.; Sun, P.R.; Lee, D.F.; Liao, Y.; Zhou, B.P.; Hung, M.C. Phosphorylation/cytoplasmic localization of p21cip1/waf1 is associated with her2/neu overexpression and provides a novel combination predictor for poor prognosis in breast cancer patients. Clin. Cancer Res. 2004, 10, 3815–3824. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R.B. P27kip1 phosphorylation by pkb/akt leads to poor breast cancer prognosis. Breast Cancer Res. 2003, 5, 162–163. [Google Scholar] [CrossRef] [PubMed]
- Willder, J.M.; Heng, S.J.; McCall, P.; Adams, C.E.; Tannahill, C.; Fyffe, G.; Seywright, M.; Horgan, P.G.; Leung, H.Y.; Underwood, M.A.; et al. Androgen receptor phosphorylation at serine 515 by cdk1 predicts biochemical relapse in prostate cancer patients. Br. J. Cancer 2013, 108, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Derenzini, M.; Montanaro, L.; Vici, M.; Barbieri, S.; Ceccarelli, C.; Santini, D.; Taffurelli, M.; Martinelli, G.N.; Trere, D. Relationship between the rb1 mrna level and the expression of phosphorylated rb protein in human breast cancers: Their relevance in cell proliferation activity and patient clinical outcome. Histol. Histopathol. 2007, 22, 505–513. [Google Scholar] [PubMed]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W.; et al. Mutations of the braf gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Ritterhouse, L.L.; Barletta, J.A. Braf v600e mutation-specific antibody: A review. Semin. Diagn. Pathol. 2015, 32, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Tamosaitis, L.; Kvederaviciute, K.; Tarvydas, R.; Staniute, G.; Kalyan, K.; Meskinyte-Kausiliene, E.; Stankevicius, V.; Valius, M. Kras, nras and braf mutations in colorectal cancer and melanoma. Med. Oncol. 2017, 34, 26. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R.; Ugolini, C.; Viola, D.; Lupi, C.; Biagini, A.; Giannini, R.; Romei, C.; Miccoli, P.; Pinchera, A.; Basolo, F. Braf(v600e) mutation and outcome of patients with papillary thyroid carcinoma: A 15-year median follow-up study. J. Clin. Endocrinol. Metab. 2008, 93, 3943–3949. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, S.I. Targeted therapies in non-small cell lung cancer-beyond egfr and alk. Cancers 2015, 7, 930–949. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zou, L.; Zhao, W.; Wang, Y.; Liu, B.; Yao, H.; Yu, H. Clinicopathological significance of c-kit mutation in gastrointestinal stromal tumors: A systematic review and meta-analysis. Sci. Rep. 2015, 5, 13718. [Google Scholar] [CrossRef] [PubMed]
- Inal, C.; Yilmaz, E.; Piperdi, B.; Perez-Soler, R.; Cheng, H. Emerging treatment for advanced lung cancer with egfr mutation. Expert Opin. Emerg. Drugs 2015, 20, 597–612. [Google Scholar] [CrossRef] [PubMed]
- Kiyoi, H.; Naoe, T. Biology, clinical relevance, and molecularly targeted therapy in acute leukemia with flt3 mutation. Int. J. Hematol. 2006, 83, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Melo, J.V. Bcr-abl gene variants. Baillieres Clin. Haematol. 1997, 10, 203–222. [Google Scholar] [CrossRef]
- Talpaz, M.; Shah, N.P.; Kantarjian, H.; Donato, N.; Nicoll, J.; Paquette, R.; Cortes, J.; O'Brien, S.; Nicaise, C.; Bleickardt, E.; et al. Dasatinib in imatinib-resistant philadelphia chromosome-positive leukemias. N. Engl. J. Med. 2006, 354, 2531–2541. [Google Scholar] [CrossRef] [PubMed]
- Sabir, S.R.; Yeoh, S.; Jackson, G.; Bayliss, R. Eml4-alk variants: Biological and molecular properties, and the implications for patients. Cancers 2017, 9, 118. [Google Scholar] [CrossRef] [PubMed]
- Gotlib, J.; Cools, J. Five years since the discovery of fip1l1-pdgfra: What we have learned about the fusion and other molecularly defined eosinophilias. Leukemia 2008, 22, 1999–2010. [Google Scholar] [CrossRef] [PubMed]
- Griseri, P.; Garrone, O.; Lo Sardo, A.; Monteverde, M.; Rusmini, M.; Tonissi, F.; Merlano, M.; Bruzzi, P.; Lo Nigro, C.; Ceccherini, I. Genetic and epigenetic factors affect ret gene expression in breast cancer cell lines and influence survival in patients. Oncotarget 2016, 7, 26465–26479. [Google Scholar] [CrossRef] [PubMed]
- Haag, T.; Herkt, C.E.; Walesch, S.K.; Richter, A.M.; Dammann, R.H. The apoptosis associated tyrosine kinase gene is frequently hypermethylated in human cancer and is regulated by epigenetic mechanisms. Genes Cancer 2014, 5, 365–374. [Google Scholar] [PubMed]
- Fu, D.Y.; Wang, Z.M.; Wang, B.L.; Chen, L.; Yang, W.T.; Shen, Z.Z.; Huang, W.; Shao, Z.M. Frequent epigenetic inactivation of the receptor tyrosine kinase epha5 by promoter methylation in human breast cancer. Hum. Pathol. 2010, 41, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Kim, M.J.; Lee, J.Y.; Lee, S.M.; Choi, J.E.; Lee, S.Y.; Park, J.Y. Epigenetic inactivation of checkpoint kinase 2 gene in non-small cell lung cancer and its relationship with clinicopathological features. Lung Cancer 2009, 65, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Jang, H.R.; Kim, J.H.; Noh, S.M.; Song, K.S.; Cho, J.S.; Jeong, H.Y.; Norman, J.C.; Caswell, P.T.; Kang, G.H.; et al. Epigenetic inactivation of protein kinase d1 in gastric cancer and its role in gastric cancer cell migration and invasion. Carcinogenesis 2008, 29, 629–637. [Google Scholar] [CrossRef] [PubMed]
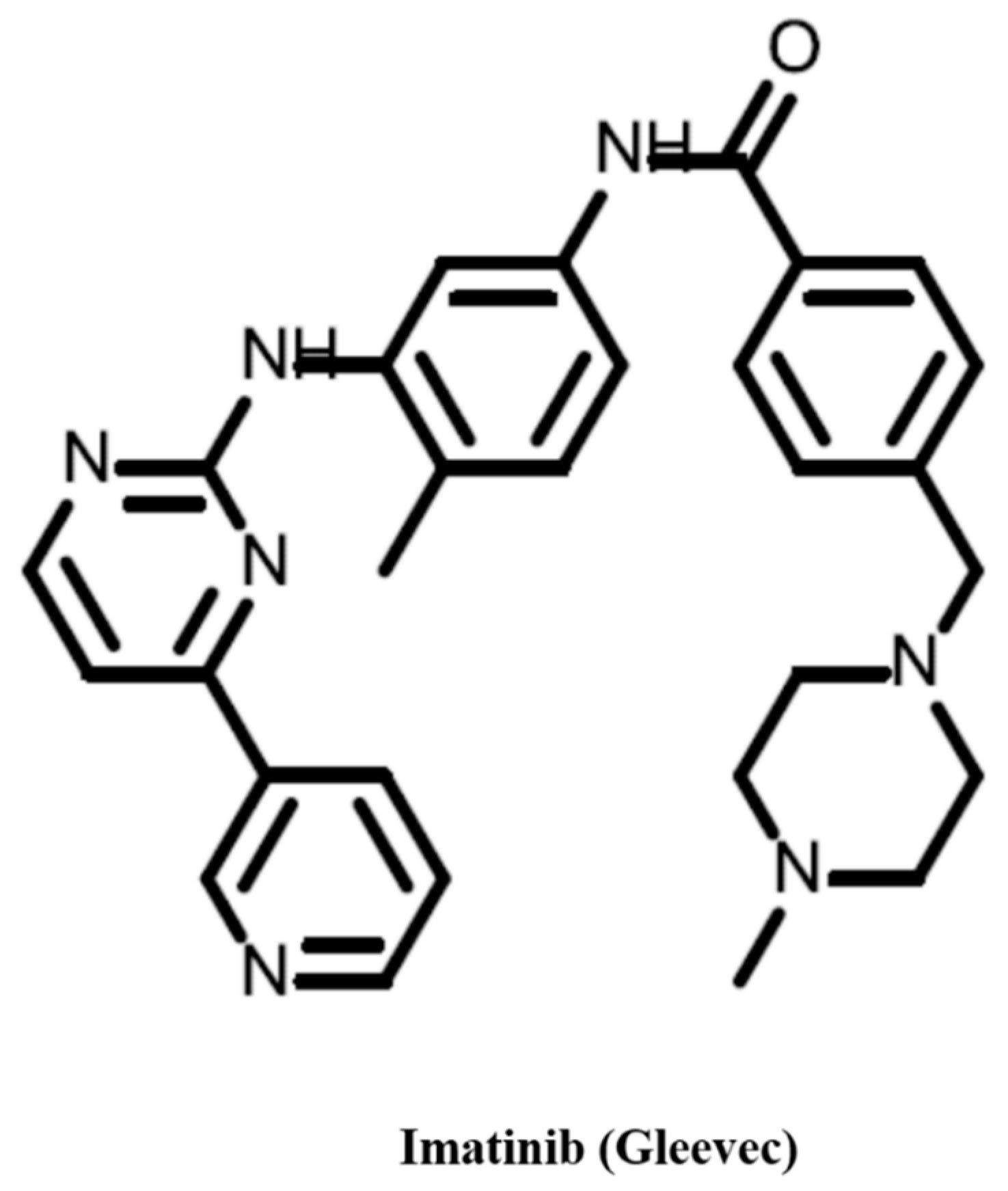
- Druker, B.J. Sti571 (gleevec) as a paradigm for cancer therapy. Trends Mol. Med. 2002, 8, S14–S18. [Google Scholar] [CrossRef]
- Gharwan, H.; Groninger, H. Kinase inhibitors and monoclonal antibodies in oncology: Clinical implications. Nat. Rev. Clin. Oncol. 2016, 13, 209–227. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Valius, M. The cdk inhibitors in cancer research and therapy. J. Cancer Res. Clin. Oncol. 2011, 137, 1409–1418. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Kalyan, K.; Sorokinas, A.; Stankunas, E.; Levy, J.; Meskinyte, I.; Stankevicius, V.; Kaupinis, A.; Valius, M. Roscovitine in cancer and other diseases. Ann. Transl. Med. 2015, 3, 135. [Google Scholar] [PubMed]
- Cicenas, J.; Kalyan, K.; Sorokinas, A.; Jatulyte, A.; Valiunas, D.; Kaupinis, A.; Valius, M. Highlights of the latest advances in research on cdk inhibitors. Cancers 2014, 6, 2224–2242. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J. The aurora kinase inhibitors in cancer research and therapy. J. Cancer Res. Clin. Oncol. 2016, 142, 1995–2012. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J.; Cicenas, E. Multi-kinase inhibitors, aurks and cancer. Med. Oncol. 2016, 33, 43. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Wang, X.; Proud, C.G. Mtor inhibitors in cancer therapy. F1000Res 2016. [Google Scholar] [CrossRef] [PubMed]
- Cicenas, J. Jnk inhibitors: Is there a future? MAP Kinases 2015, 4, 7. [Google Scholar] [CrossRef]
- Hudis, C.A. Trastuzumab--mechanism of action and use in clinical practice. N. Engl. J. Med. 2007, 357, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.B.; Lum, P.; Chen, A.; Arends, R.; Roskos, L.; Smith, B.; Perez Ruixo, J.J. Pharmacokinetic and pharmacodynamic perspectives on the clinical drug development of panitumumab. Clin. Pharmacokinet. 2010, 49, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Krupitskaya, Y.; Wakelee, H.A. Ramucirumab, a fully human mab to the transmembrane signaling tyrosine kinase vegfr-2 for the potential treatment of cancer. Curr. Opin. Investig. Drugs 2009, 10, 597–605. [Google Scholar] [PubMed]
- Vincenzi, B.; Badalamenti, G.; Napolitano, A.; Spalato Ceruso, M.; Pantano, F.; Grignani, G.; Russo, A.; Santini, D.; Aglietta, M.; Tonini, G. Olaratumab: Pdgfr-alpha inhibition as a novel tool in the treatment of advanced soft tissue sarcomas. Crit. Rev. Oncol. Hematol. 2017, 118, 1–6. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cicenas, J.; Zalyte, E.; Bairoch, A.; Gaudet, P. Kinases and Cancer. Cancers 2018, 10, 63. https://doi.org/10.3390/cancers10030063
Cicenas J, Zalyte E, Bairoch A, Gaudet P. Kinases and Cancer. Cancers. 2018; 10(3):63. https://doi.org/10.3390/cancers10030063
Chicago/Turabian StyleCicenas, Jonas, Egle Zalyte, Amos Bairoch, and Pascale Gaudet. 2018. "Kinases and Cancer" Cancers 10, no. 3: 63. https://doi.org/10.3390/cancers10030063
APA StyleCicenas, J., Zalyte, E., Bairoch, A., & Gaudet, P. (2018). Kinases and Cancer. Cancers, 10(3), 63. https://doi.org/10.3390/cancers10030063





