Microreactortechnology: Real-Time Flow Measurements in Organic Synthesis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Apparatus
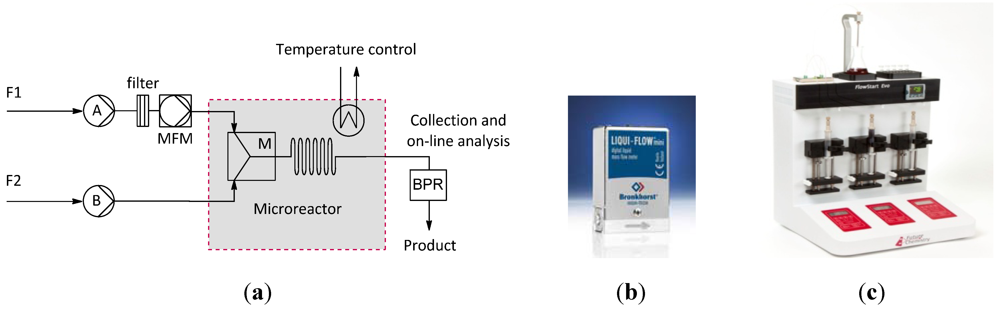
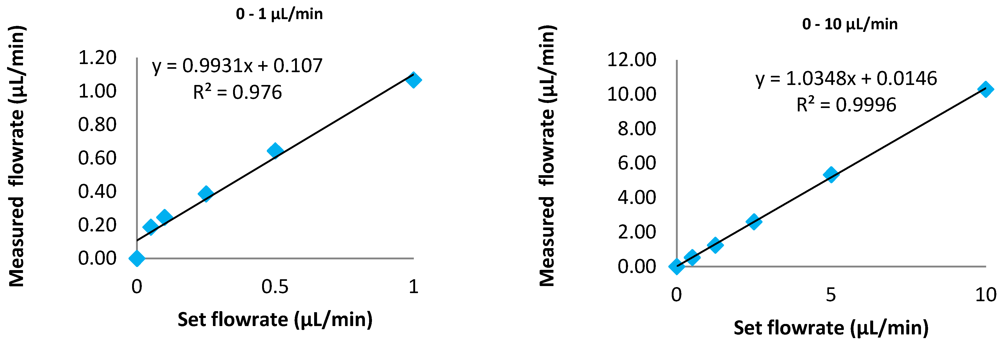
2.2. Fluidic Activation: Accuracy of Pumping Devices



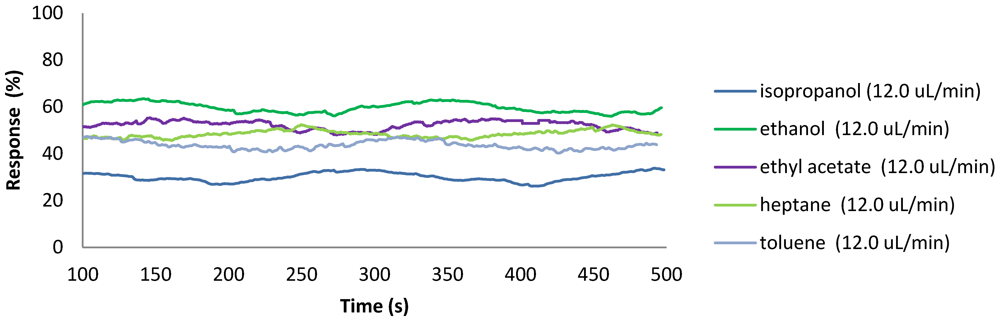
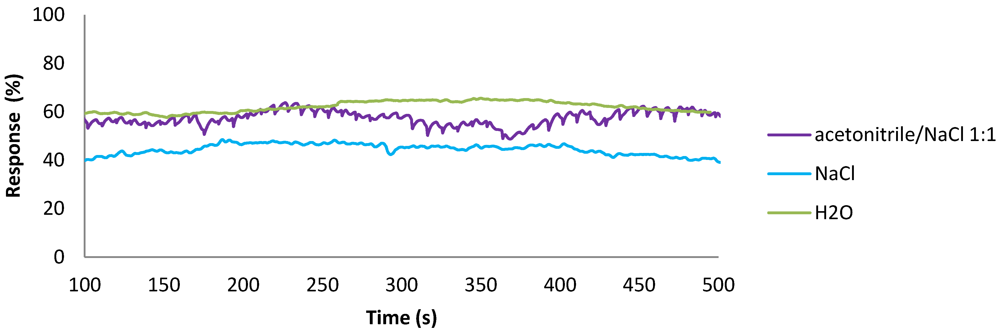
2.4. Application of Flow Meters in Chemical Processes
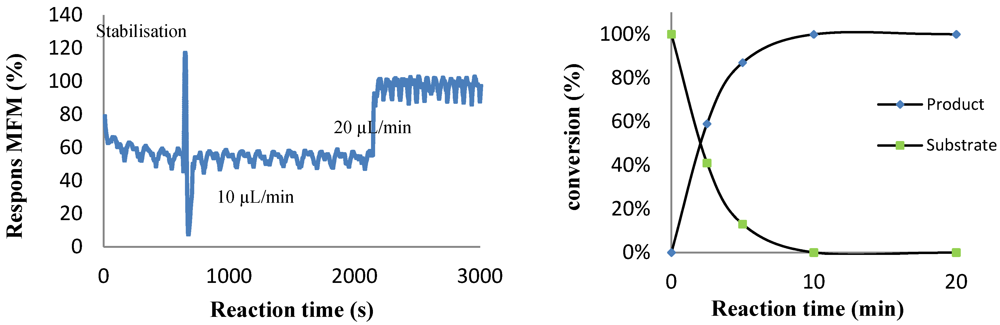
2.4.1. Photochemical Oxidation of Furanone
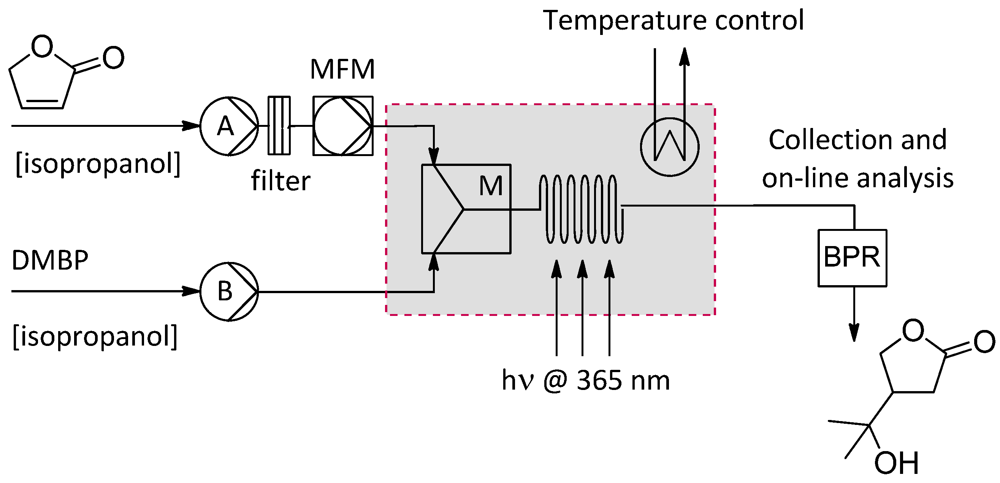
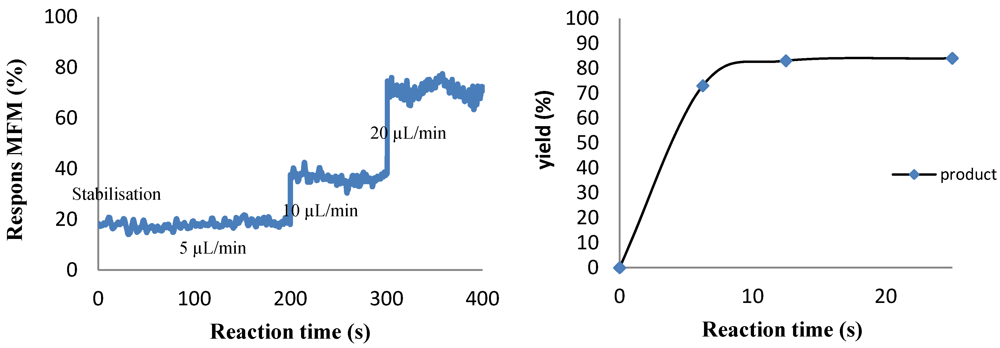
2.4.2. Grignard Reactions of Benzaldehyde
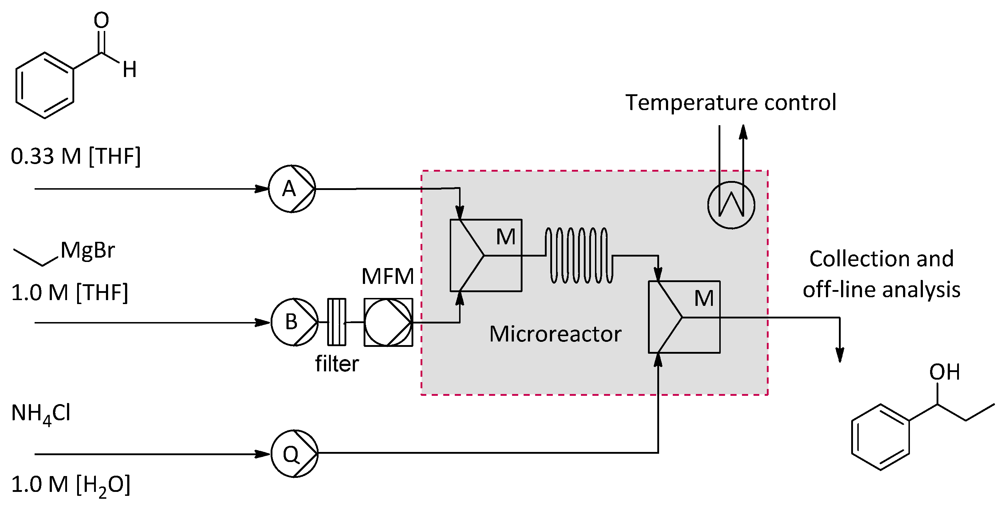
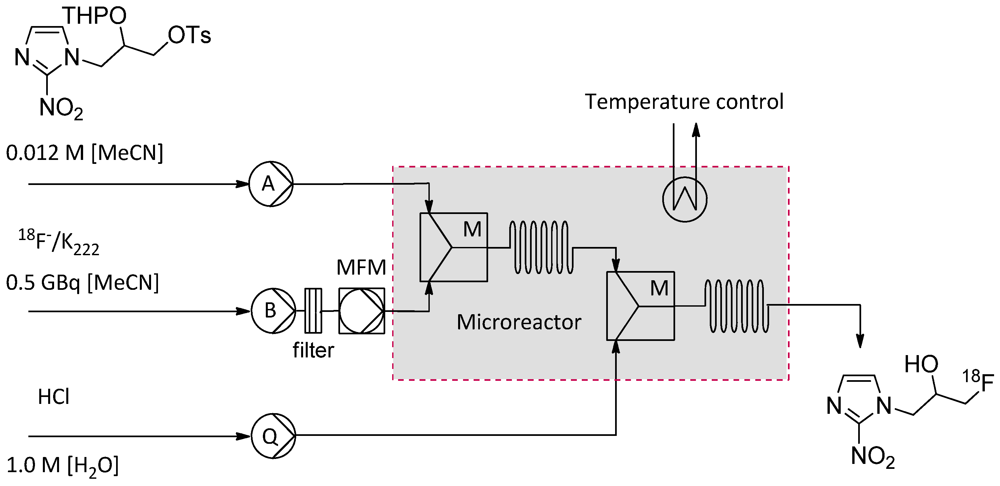
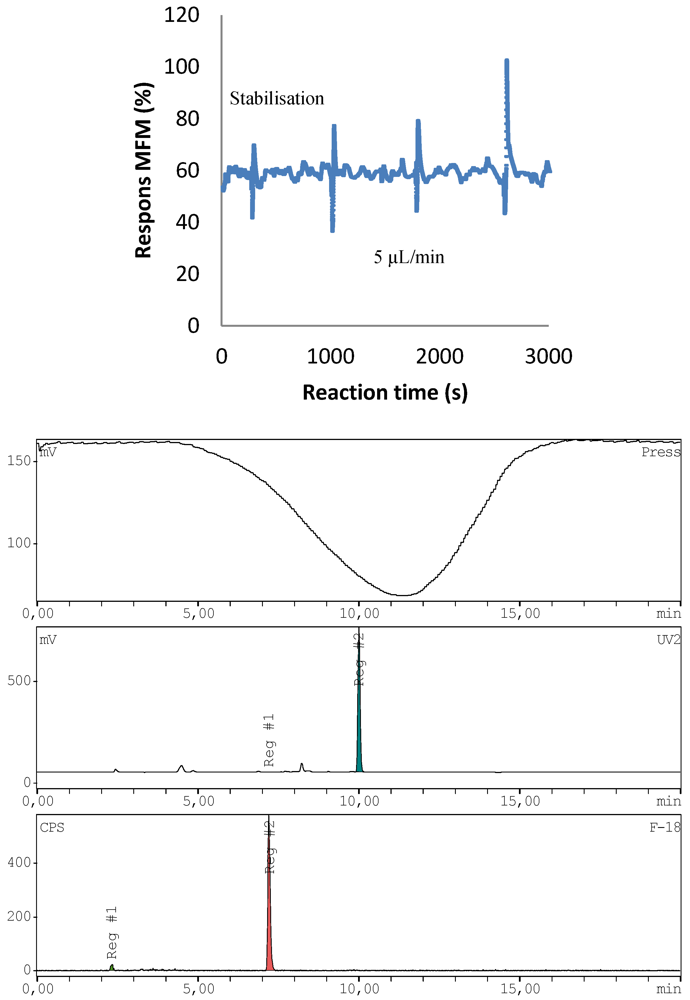
2.4.3. 18F-Radiolabeling
3. Conclusion
Acknowledgements
References
- Nobis, M.; Roberge, D.M. Mastering ozonolysis: Production from laboratory to ton scale in continuous flow. Chim. Oggi. 2011, 29, 56–58. [Google Scholar]
- Braune, S. From lab to production—An example from industry for the use of a microreactor in a multi-ton scale. In Proceeding ISPE Milan Congress on Science and Risk Assessment for Business Success, Milan, Italy, 22–25 March 2010.
- Braune, S.; Pochlauer, P.; Reintjens, R.; Steinhofer, S.; Winter, M.; Lobet, O.; Guidat, R.; Woehl, P.; Guermeur, C. Industry perspective—Selective nitration in a microreactor for pharmaceutical production under cGMP conditions. Chim. Oggi. 2009, 27, 26–29. [Google Scholar]
- van den Broek, S.A.M.W.; Becker, R.; Kőssl, F.; Mariëlle, M.E.; Delville, P.J.; Nieuwland, K.; Rutjes, F.P.J.T. Prilezhaev dihydroxyilation of olefins in a continuous flow process. ChemSusChem 2011, 4, 1–5. [Google Scholar] [CrossRef]
- Fox, M.; Esveld, E.; Luttge, R.; Boom, R. A new pulsed electric field microreactor: Comparison between the laboratory and microtechnology scale. Lab Chip 2005, 5, 943–948. [Google Scholar] [CrossRef]
- Matsushita1, Y.; Ichimura, T.; Ohba, N.; Kumada, S.; Sakeda, K.; Suzuki, T.; Tanibata, H.; Murata, T. Recent progress on photoreactions in microreactors. Pure Appl. Chem. 2007, 79, 1959–1968. [Google Scholar] [CrossRef]
- Jaehnisch, K.; Baerns, M.; Hessel, V.; Ehrfeld, W.; Haverkamp, V.; Loewe, H.; Wille, C.; Guber, A. Direct fluorination of toluene using elemental fluorine in gas/liquid microreactors. J. Fluor. Chem. 2000, 105, 117–128. [Google Scholar] [CrossRef]
- Okafor, O.C.; Tadepalli, S.; Tampy, G.; Lawal, A. Cycloaddition of isoamylene and methylstyrene in a microreactor using filtrol-24 catalyst: Microreactor performance study and comparison with semi-batch reactor performance. Int. J. Chem. React. Eng. 2010, 8, A71. [Google Scholar]
- Roberge, D.M.; Ducry, L.; Bieler, N.; Cretton, P.; Zimmermann, B. Microreactor technology: A revolution for the fine chemical and pharmaceutical industries? Chem. Eng. Technol. 2005, 28, 318–323. [Google Scholar] [CrossRef]
- Riego, J.M.; Sedin, Z.; Zaldivar, J.M.; Marziano, N.C.; Tortato, C. Sulfuric acid on silica-gel: An inexpensive catalyst for aromatic nitration. Tetrahedron Lett. 1996, 37, 513–517. [Google Scholar]
- Bollyn, M. Thermal hazards of the Vilsmeier-Haack reacdtion on N,N-dimethylaniline. Org. Process Res. Dev. 2005, 9, 982–996. [Google Scholar] [CrossRef]
- Nolan, P.F. Some lessons from thermal-runaway incidents. J. Haz. Mat. 1987, 14, 233–239. [Google Scholar] [CrossRef]
- FlowStart Evo/FutureChemistry. Available online: http://www.futurechemistry.com/flowstart-evo (accessed on 15 February 2012).
- MacInnes, J.M.; Vikhansky, A.; Allen, R.W.K. Numerical characterisation of folding flow microchannel mixers. Chem. Eng. Sci. 2007, 62, 2718–2727. [Google Scholar] [CrossRef]
- LIQUI-FLOW Mini Micro Fluidic Mass Flow Meters for Liquids—Bronkhorst®. Available online: http://www.bronkhorst.com/en/products/liquid_flow_meters___controllers/liqui-flow_mini/ (accessed on 15 February 2012).
- Shvydkiv, O.; Yavorskyy, A.; Nolan, K.; Youssef, A.; Riguet, E.; Hoffmann, N.; Oelgemöller, M. Photosensitized addition of isopropanol to furanones in a 365 nm UV-LED microchip. Photochem. Photobiol. Sci. 2010, 9, 1601–1603. [Google Scholar]
- Tilstam, U.; Weinmann, H. Activation of Mg metal for safe formation of Grignard reagents on plant scale. Org. Process Res. Dev. 2002, 6, 906–910. [Google Scholar] [CrossRef]
- Wang, C.W.; Chou, T.K.; Liu, R.S.; Wang, S.J.; Jiu, W.J.; Chen, C.H.; Wang, H.E. A robotic synthesis of [18F]fluoromisonidazole ([18F]FMISO). Appl. Radiat. Isot. 2007, 65, 862–686. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Van den Broek, S.A.M.W.; Becker, R.; Koch, K.; Nieuwland, P.J. Microreactortechnology: Real-Time Flow Measurements in Organic Synthesis. Micromachines 2012, 3, 244-254. https://doi.org/10.3390/mi3020244
Van den Broek SAMW, Becker R, Koch K, Nieuwland PJ. Microreactortechnology: Real-Time Flow Measurements in Organic Synthesis. Micromachines. 2012; 3(2):244-254. https://doi.org/10.3390/mi3020244
Chicago/Turabian StyleVan den Broek, Sebastiaan A. M. W., René Becker, Kaspar Koch, and Pieter J. Nieuwland. 2012. "Microreactortechnology: Real-Time Flow Measurements in Organic Synthesis" Micromachines 3, no. 2: 244-254. https://doi.org/10.3390/mi3020244
APA StyleVan den Broek, S. A. M. W., Becker, R., Koch, K., & Nieuwland, P. J. (2012). Microreactortechnology: Real-Time Flow Measurements in Organic Synthesis. Micromachines, 3(2), 244-254. https://doi.org/10.3390/mi3020244



