Fabrication of Stromal Cell-Derived Factor-1 Contained in Gelatin/Hyaluronate Copolymer Mixed with Hydroxyapatite for Use in Traumatic Bone Defects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Basic Materials
2.2. Synthesis of Hydroxyapatite
2.3. Synthesis of Gel/HA–HAP–SDF-1 Composite
2.4. Characterization of Material
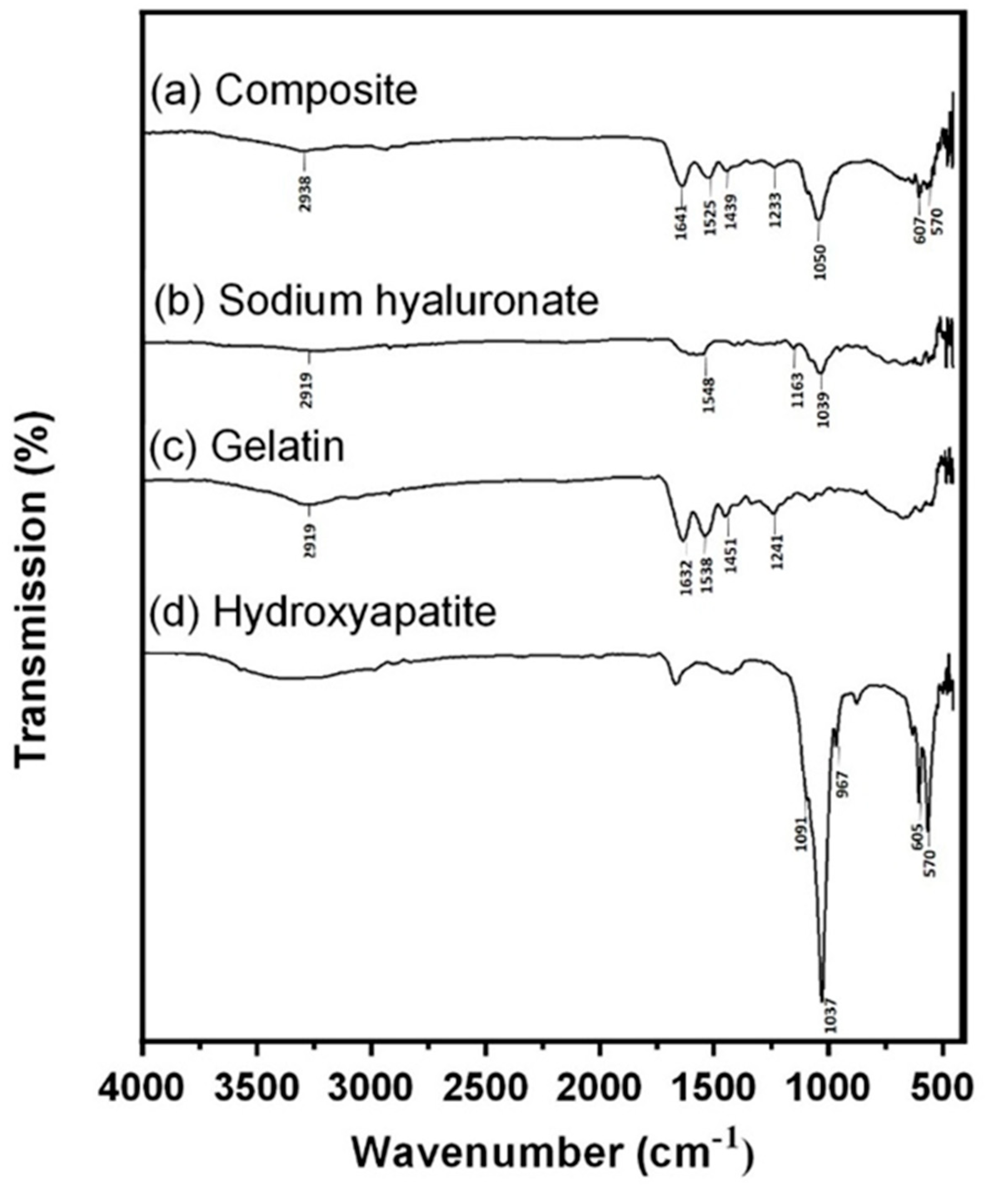
2.4.1. Fourier-Transform Infrared (FTIR) Spectroscopy Analysis
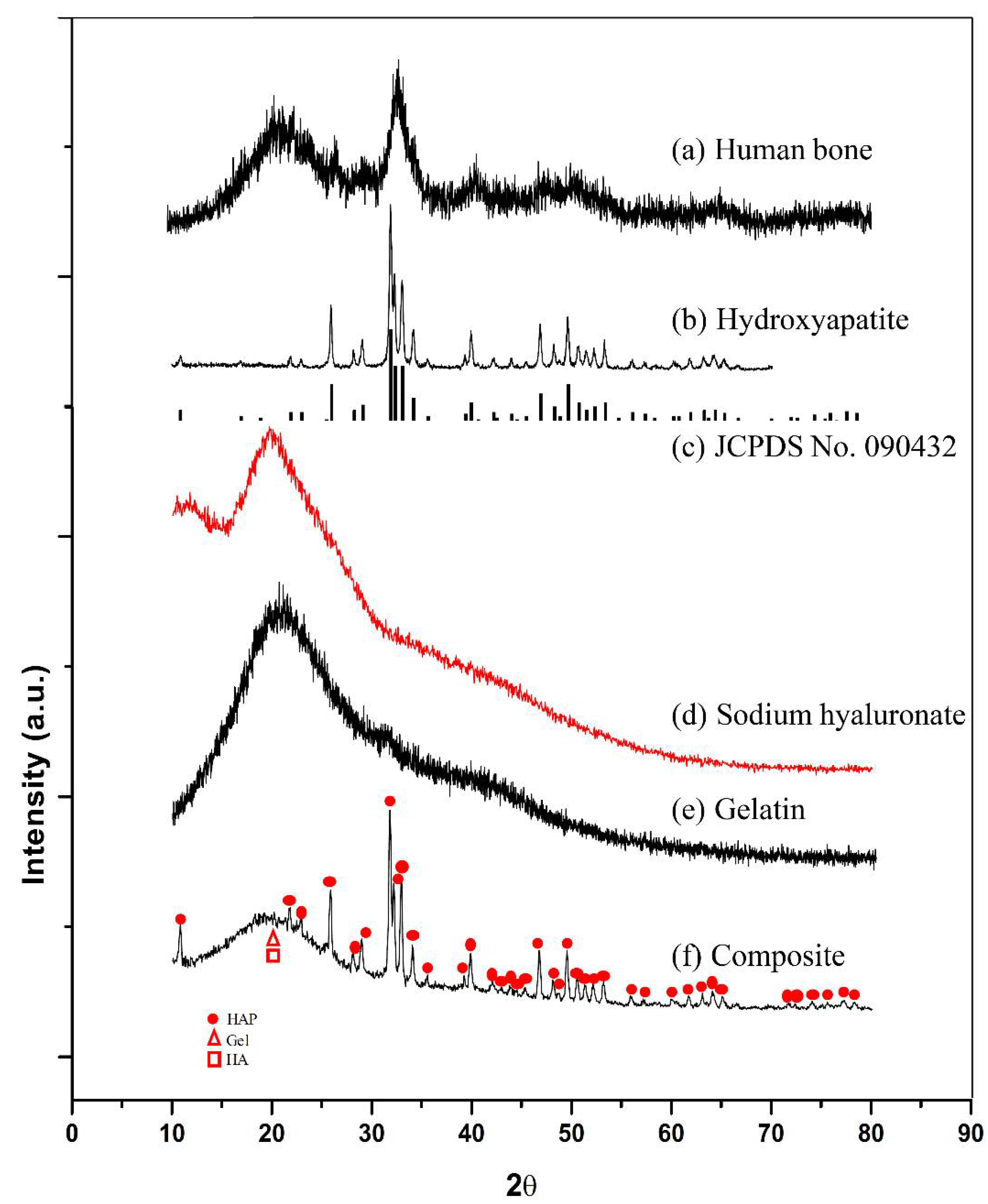
2.4.2. X-ray Diffraction (XRD) Analysis
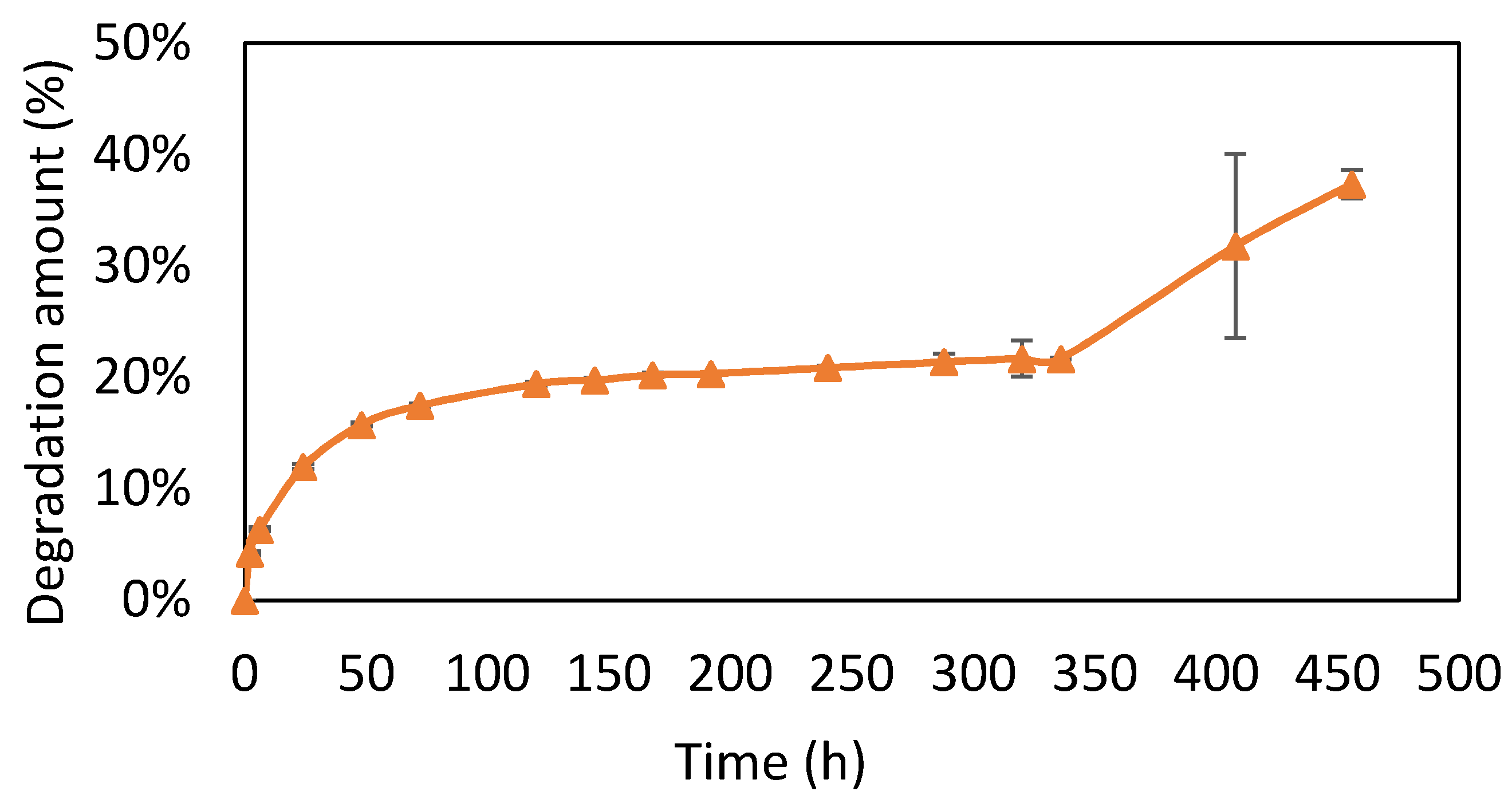
2.4.3. Degradation Profile
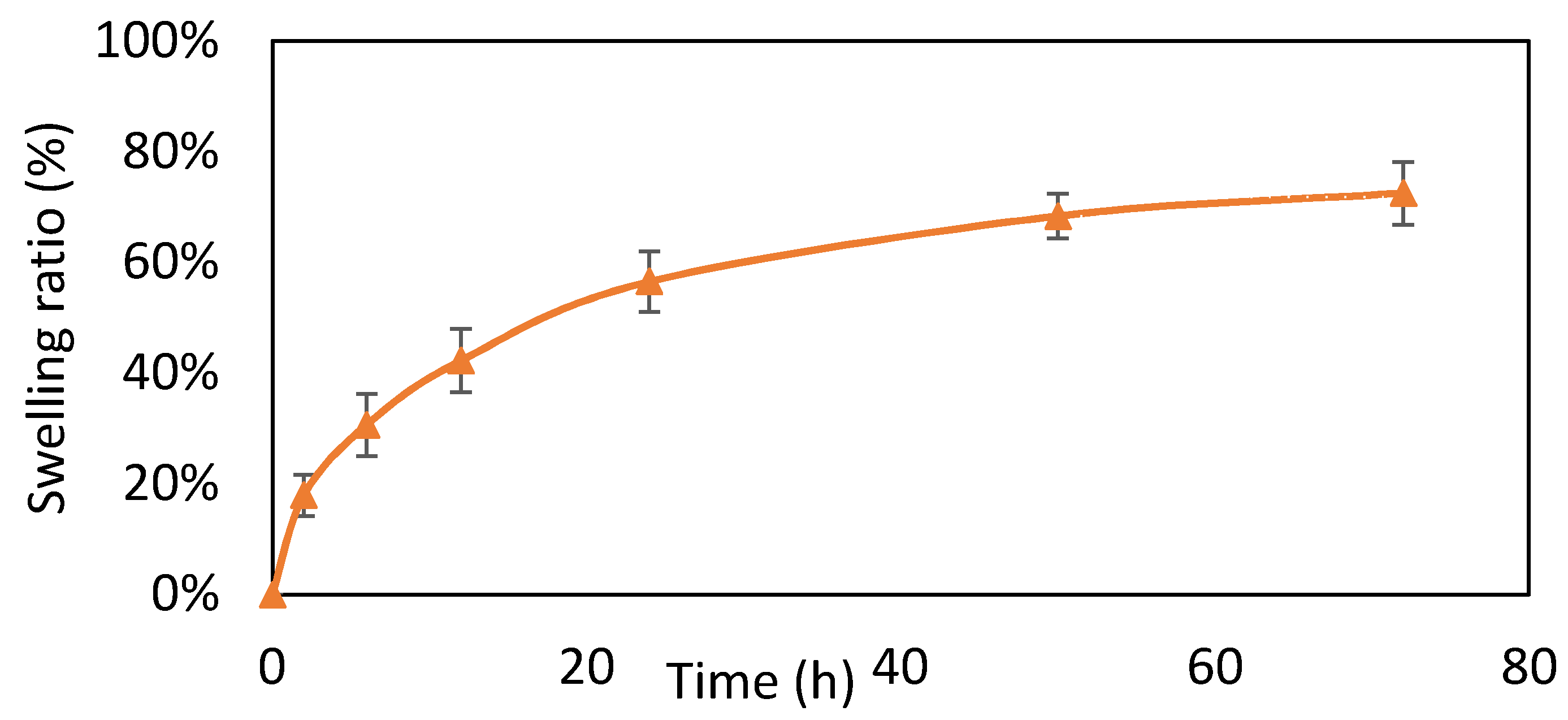
2.4.4. Swelling Ratio Test
2.4.5. Insulin Release Profile
2.5. In Vitro Studies
2.5.1. Culture of Cells
2.5.2. Viability of Cells
2.5.3. Mesenchymal Stem Cell (MSC) Recruitment Test
2.6. In Vivo Studies
2.6.1. Animal Model
2.6.2. Second Harmonic Generation (SHG) Imaging
2.6.3. Micro-CT Imaging
2.6.4. Histological Imaging
2.6.5. Blood Tests
2.7. Statistics
3. Results
3.1. FTIR Analysis
3.2. X-ray Diffraction Analysis
3.3. Degradation Test
3.4. Swelling Test
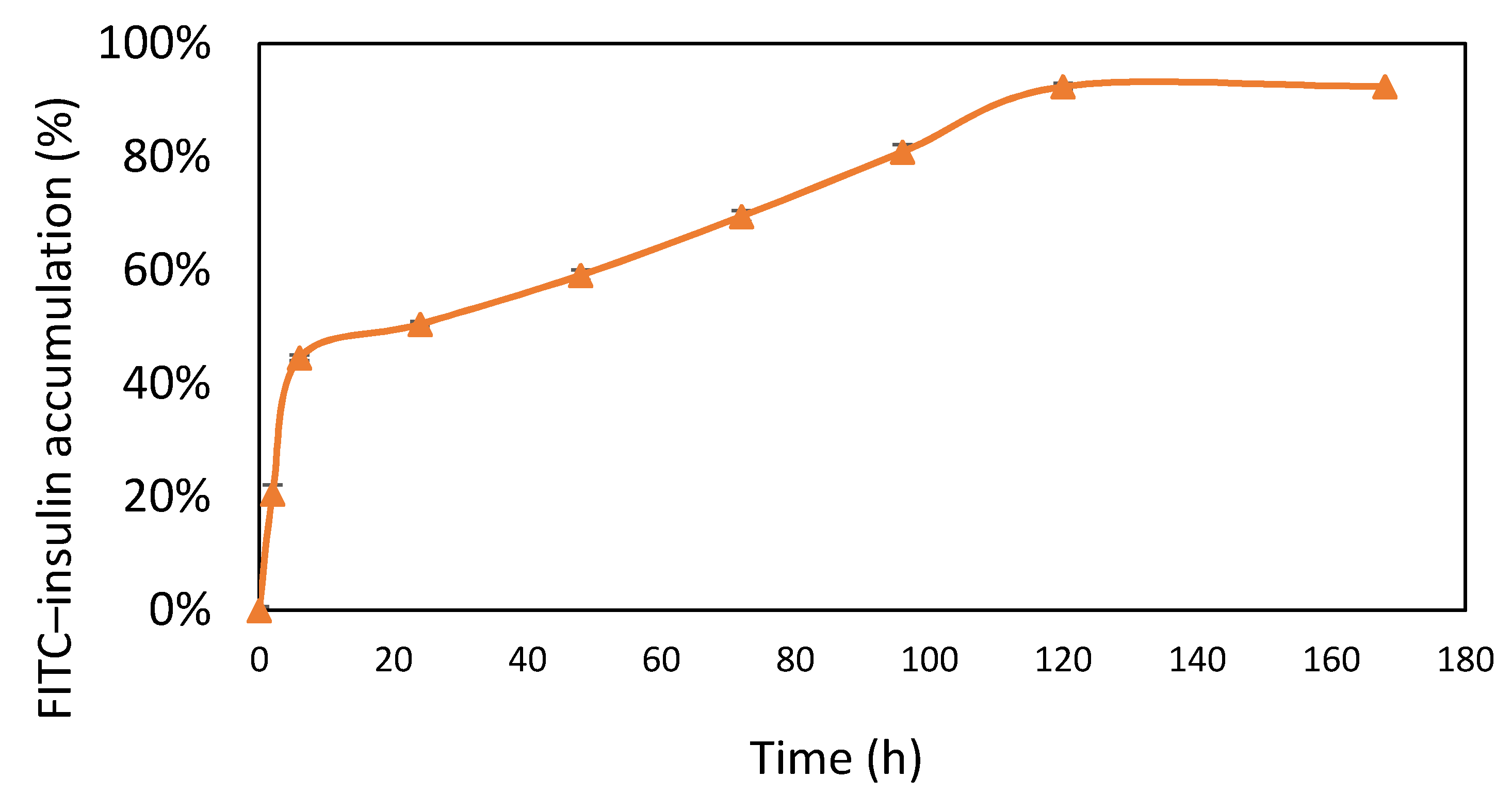
3.5. Insulin Release Profile
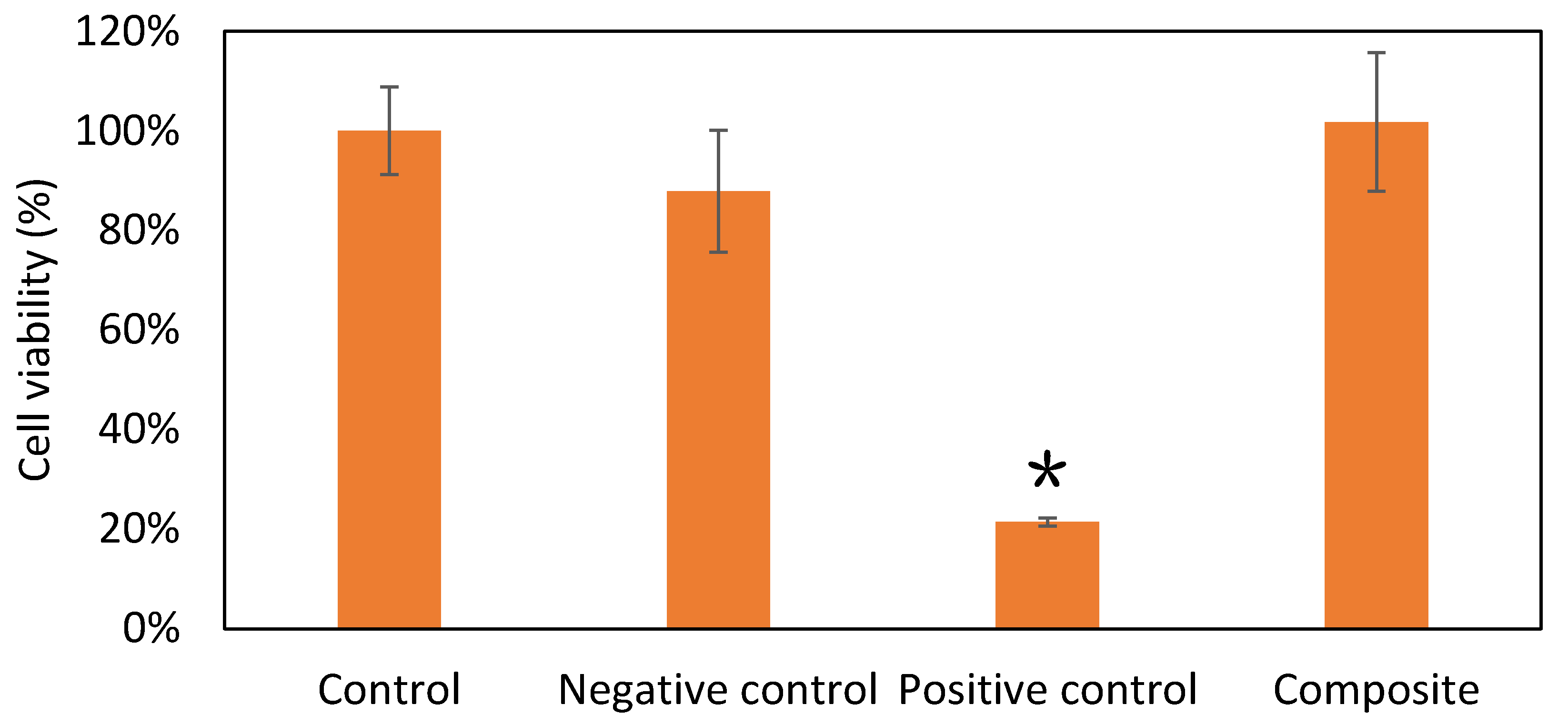
3.6. Viability of Cells
3.7. Mesenchymal Stem Cell Recruitment Test
3.8. Second Harmonic Generation Imaging
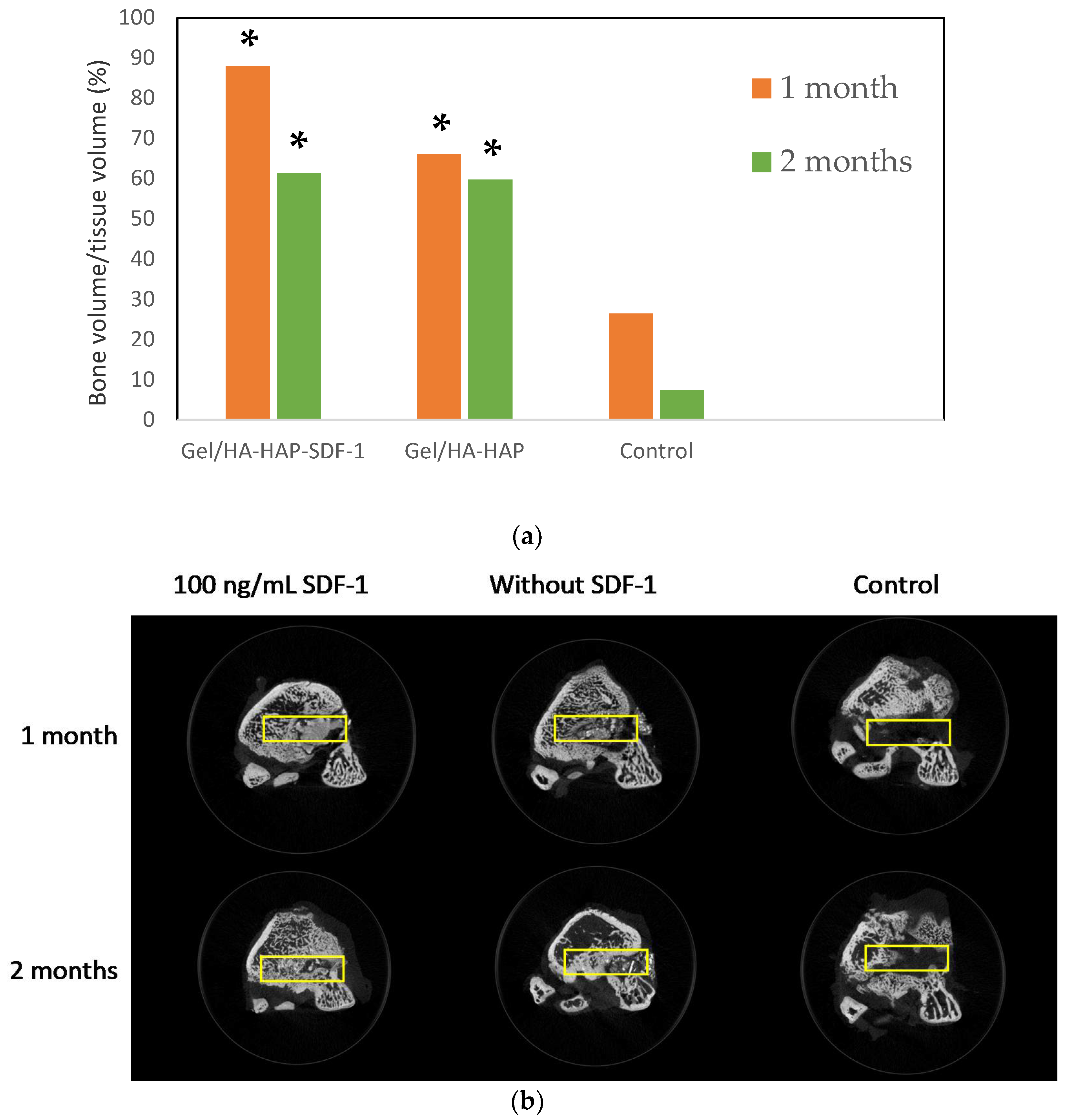
3.9. Micro-CT Imaging
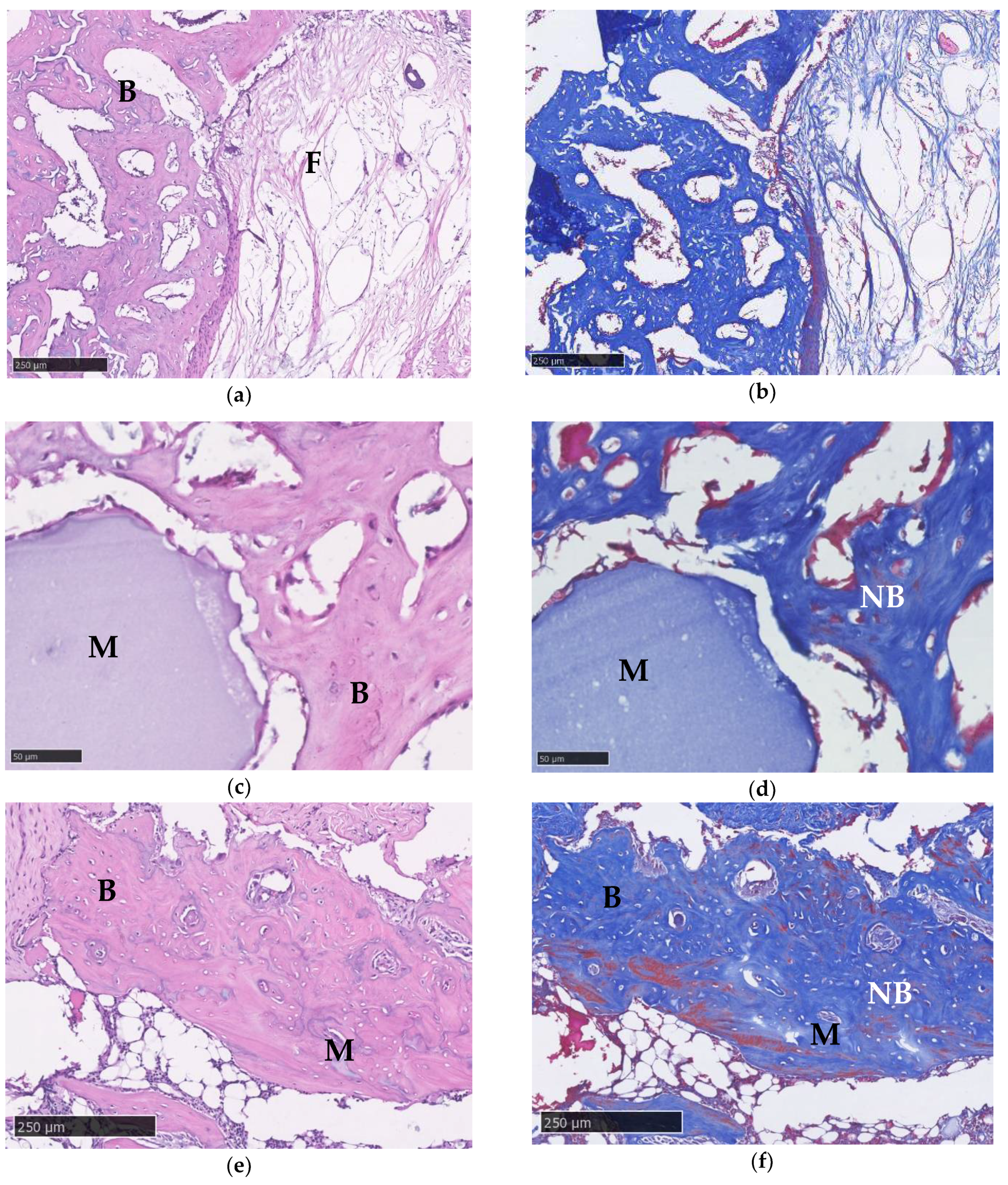
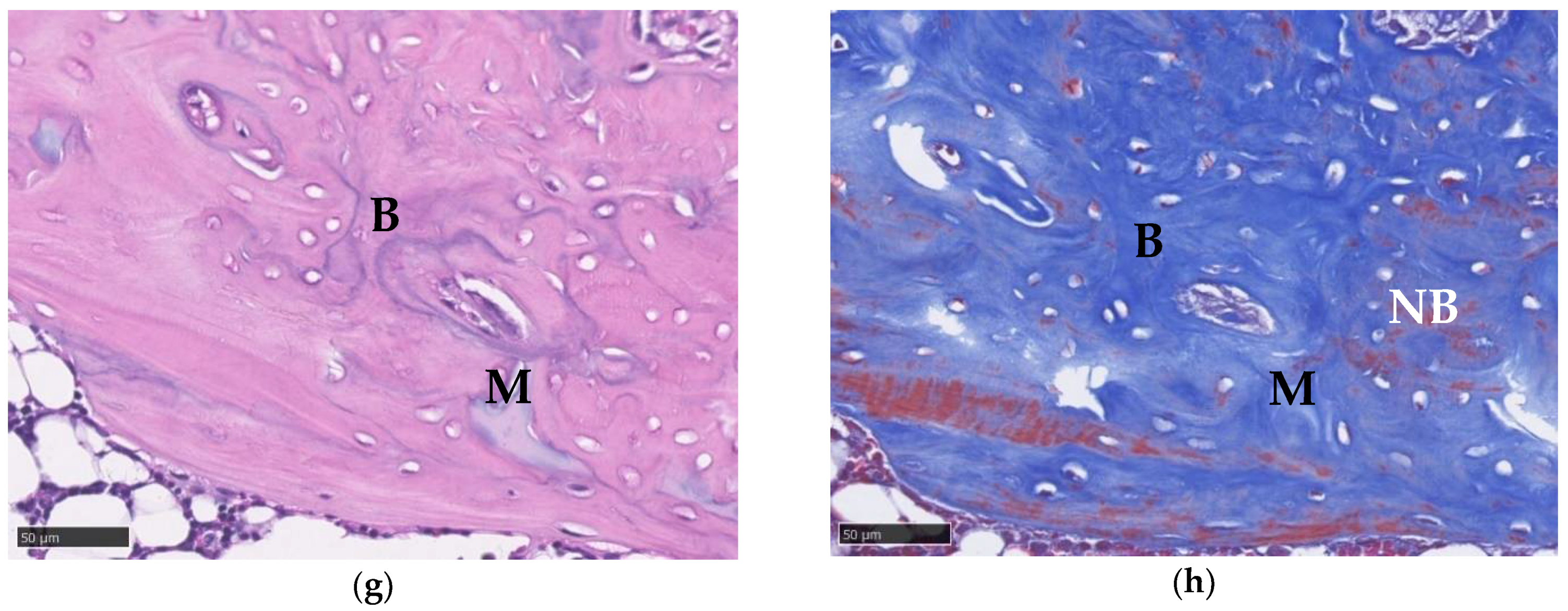
3.10. Histological Imaging
3.11. Blood Tests
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef]
- Langer, R.; Vacanti, J.P.; Vacanti, C.A.; Atala, A.; Freed, L.; Vunjak-Novakovic, G. Tissue Engineering: Biomedical Applications. Tissue Eng. 1995, 1, 151–161. [Google Scholar] [CrossRef]
- Einhorn, T.A. Enhancement of fracture-healing. J. Bone Jt. Surg. Am. 1995, 77, 940–956. [Google Scholar] [CrossRef] [PubMed]
- Sauer, H.D.; Schoettle, H. The stability of osteosyntheses bridging defects. Arch. Orthop. Trauma. Surg. 1979, 95, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Lobb, D.C.; DeGeorge, B.R.; Chhabra, A.B. Bone Graft Substitutes: Current Concepts and Future Expectations. J. Hand Surg. 2019, 44, 497–505.e2. [Google Scholar] [CrossRef]
- Mauffrey, C.; Barlow, B.T.; Smith, W. Management of segmental bone defects. J. Am. Acad. Orthop. Surg. 2015, 23, 143–153. [Google Scholar]
- Glenske, K.; Donkiewicz, P.; Köwitsch, A.; Milosevic-Oljaca, N.; Rider, P.; Rofall, S.; Franke, J.; Jung, O.; Smeets, R.; Schnettler, R.; et al. Applications of Metals for Bone Regeneration. Int. J. Mol. Sci. 2018, 19, 826. [Google Scholar] [CrossRef] [PubMed]
- Westhauser, F.; Essers, C.; Karadjian, M.; Reible, B.; Schmidmaier, G.; Hagmann, S.; Moghaddam, A. Supplementation with 45s5 bioactive glass reduces in vivo resorption of the beta-tricalcium-phosphate-based bone substitute material vitoss. Int. J. Mol. Sci. 2019, 20, 4253. [Google Scholar] [CrossRef] [PubMed]
- Kohn, J.; Welsh, W.J.; Knight, D. A new approach to the rationale discovery of polymeric biomaterials. Biomaterials 2007, 28, 4171–4177. [Google Scholar] [CrossRef]
- Laurencin, C.T.; Ambrosio, A.M.A.; Borden, M.D.; Cooper, J.A. Tissue Engineering: Orthopedic Applications. Annu. Rev. Biomed. Eng. 1999, 1, 19–46. [Google Scholar] [CrossRef]
- Zeng, J.-H.; Liu, S.-W.; Xiong, L.; Qiu, P.; Ding, L.-H.; Xiong, S.-L.; Li, J.-T.; Liao, X.-G.; Tang, Z.-M. Scaffolds for the repair of bone defects in clinical studies: A systematic review. J. Orthop. Surg. Res. 2018, 13, 1–14. [Google Scholar] [CrossRef]
- Kang, H.-W.; Tabata, Y.; Ikada, Y. Fabrication of porous gelatin scaffolds for tissue engineering. Biomaterials 1999, 20, 1339–1344. [Google Scholar] [CrossRef]
- Olsen, D.; Yang, C.L.; Bodo, M.; Chang, R.; Leigh, S.; Baez, J.; Carmichael, D.; Perala, M.; Hamalainen, E.R.; Jarvinen, M.; et al. Recombinant collagen and gelatin for drug delivery. Adv. Drug Deliver Rev. 2003, 55, 1547–1567. [Google Scholar] [CrossRef]
- Chen, M.-H.; Chen, P.-R.; Chen, M.-H.; Hsieh, S.-T.; Huang, J.-S.; Lin, F.-H. Gelatin-Tricalcium phosphate membrane modified with ngf and cultured schwann cells for peripheral nerve repair: A tissue engineering approach. Biomed. Eng. Appl. Basis Commun. 2006, 18, 47–54. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Kuo, S.-M.; Yao, C.-H. A review for gelatin used for artificial nerve and bone implants—10-year retrospection. Biomed. Eng. Appl. Basis Commun. 2009, 21, 233–238. [Google Scholar] [CrossRef]
- Luo, Y.; Li, Y.; Qin, X.; Wa, Q. 3D printing of concentrated alginate/gelatin scaffolds with homogeneous nano apatite coating for bone tissue engineering. Mater. Des. 2018, 146, 12–19. [Google Scholar] [CrossRef]
- Chen, W.Y.J.; Abatangelo, G. Functions of hyaluronan in wound repair. Wound Repair Regen. 1999, 7, 79–89. [Google Scholar] [CrossRef]
- Day, A.J.; de la Motte, C.A. Hyaluronan cross-linking: A protective mechanism in inflammation? Trends Immunol. 2005, 26, 637–643. [Google Scholar] [CrossRef] [PubMed]
- West, D.C.; Hampson, I.N.; Arnold, F.; Kumar, S. Angiogenesis induced by degradation products of hyaluronic acid. Science 1985, 228, 1324–1326. [Google Scholar] [CrossRef] [PubMed]
- De Boulle, K.; Glogau, R.; Kono, T.; Nathan, M.; Tezel, A.; Roca-Martinez, J.-X.; Paliwal, S.; Stroumpoulis, D. A Review of the Metabolism of 1,4-Butanediol Diglycidyl Ether-Crosslinked Hyaluronic Acid Dermal Fillers. Dermatol. Surg. 2013, 39, 1758–1766. [Google Scholar] [CrossRef]
- Holmes, R.; Mooney, V.; Bucholz, R.; Tencer, A. A coralline hydroxyapatite bone graft substitute. Preliminary report. Clin. Orthop. Relat. Res. 1984, 188, 252–262. [Google Scholar]
- Kumar, A.; Negi, Y.S.; Choudhary, V.; Bhardwaj, N.K. Microstructural and mechanical properties of porous biocomposite scaffolds based on polyvinyl alcohol, nano-hydroxyapatite and cellulose nanocrystals. Cellulose 2014, 21, 3409–3426. [Google Scholar] [CrossRef]
- Kumar, A.; Kargozar, S.; Baino, F.; Han, S.S. Additive manufacturing methods for producing hydroxyapatite and hydroxyapatite-based composite scaffolds: A review. Front. Mater. 2019, 6, 313. [Google Scholar] [CrossRef]
- Lin, W.; Xu, L.; Zwingenberger, S.; Gibon, E.; Goodman, S.B.; Li, G. Mesenchymal stem cells homing to improve bone healing. J. Orthop. Transl. 2017, 9, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Otsuru, S.; Tamai, K.; Yamazaki, T.; Yoshikawa, H.; Kaneda, Y. Circulating Bone Marrow-Derived Osteoblast Progenitor Cells Are Recruited to the Bone-Forming Site by the CXCR4/Stromal Cell-Derived Factor-1 Pathway. Stem Cells 2008, 26, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Segers, V.; Tokunou, T.; Higgins, L.J.; MacGillivray, C.; Gannon, J.; Lee, R.T. Local Delivery of Protease-Resistant Stromal Cell Derived Factor-1 for Stem Cell Recruitment After Myocardial Infarction. Circulation 2007, 116, 1683–1692. [Google Scholar] [CrossRef]
- Linh, N.T.B.; Min, Y.K.; Lee, B.-T. Fabrication and in vitro evaluations with osteoblast-like MG-63 cells of porous hyaluronic acid-gelatin blend scaffold for bone tissue engineering applications. J. Mater. Sci. 2013, 48, 4233–4242. [Google Scholar] [CrossRef]
- Akao, M.; Aoki, H.; Kato, K. Mechanical-properties of sintered hydroxyapatite for prosthetic applications. J. Mater. Sci. 1981, 16, 809–812. [Google Scholar] [CrossRef]
- Shyong, Y.-J.; Wang, M.-H.; Kuo, L.-W.; Su, C.-F.; Kuo, W.-T.; Chang, K.-C.; Lin, F.-H. Mesoporous hydroxyapatite as a carrier of olanzapine for long-acting antidepression treatment in rats with induced depression. J. Control. Release 2017, 255, 62–72. [Google Scholar] [CrossRef]
- Hung, C.J.; Yao, C.L.; Cheng, F.C.; Wu, M.L.; Wang, T.H.; Hwang, S.M. Establishment of immortalized mesenchymal stromal cells with red fluorescence protein expression for in vivo transplantation and tracing in the rat model with traumatic brain injury. Cytotherapy 2010, 12, 455–465. [Google Scholar] [CrossRef]
- Hollinger, J.O.; Kleinschmidt, J.C. The Critical Size Defect as an Experimental Model To Test Bone Repair Materials. J. Craniofacial Surg. 1990, 1, 60–68. [Google Scholar] [CrossRef]
- Kondo, N.; Ogose, A.; Tokunaga, K.; Ito, T.; Arai, K.; Kudo, N.; Inoue, H.; Irie, H.; Endo, N. Bone formation and resorption of highly purified beta-tricalcium phosphate in the rat femoral condyle. Biomaterials 2005, 26, 5600–5608. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, S.-K.; Li, L.; Qin, L.; Wang, X.-L.; Lai, Y.-X. Bone defect animal models for testing efficacy of bone substitute biomaterials. J. Orthop. Transl. 2015, 3, 95–104. [Google Scholar] [CrossRef]
- Schmitz, J.P.; Hollinger, J.O.; Schmitz, J.P.; Hollinger, J.O. The critical size defect as an experimental model for craniomandibulofacial nonunions. Clin. Orthop. Relat. Res. 1986, 205, 299–308. [Google Scholar] [CrossRef]
- Liu, M.; Lv, Y. Reconstructing Bone with Natural Bone Graft: A Review of In Vivo Studies in Bone Defect Animal Model. Nanomaterials 2018, 8, 999. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Mou, C.; Shi, Q.; Chen, B.; Hou, X.; Zhang, W.; Li, X.; Zhuang, Y.; Shi, J.; Chen, Y.; et al. Controlled release of collagen-binding SDF-1α from the collagen scaffold promoted tendon regeneration in a rat Achilles tendon defect model. Biomaterials 2018, 162, 22–33. [Google Scholar] [CrossRef]
- Sun, J.; Zhao, Y.; Li, Q.; Chen, B.; Hou, X.; Xiao, Z.; Dai, J. Controlled Release of Collagen-Binding SDF-1α Improves Cardiac Function after Myocardial Infarction by Recruiting Endogenous Stem Cells. Sci. Rep. 2016, 6, 26683. [Google Scholar] [CrossRef]
- Wang, W.; Li, W.; Ong, L.-L.; Furlani, D.; Kaminski, A.; Liebold, A.; Lützow, K.; Lendlein, A.; Wang, J.; Li, R.-K.; et al. Localized SDF-1alpha gene release mediated by collagen substrate induces CD117+ stem cells homing. J. Cell. Mol. Med. 2008, 14, 392–402. [Google Scholar] [CrossRef]
- Subramaniam, S.; Fang, Y.-H.; Sivasubramanian, S.; Lin, F.-H.; Lin, C.-P. Hydroxyapatite-calcium sulfate-hyaluronic acid composite encapsulated with collagenase as bone substitute for alveolar bone regeneration. Biomaterials 2016, 74, 99–108. [Google Scholar] [CrossRef]
- Faruq, O.; Kim, B.; Padalhin, A.; Lee, G.H.; Lee, B.-T. A hybrid composite system of biphasic calcium phosphate granules loaded with hyaluronic acid–gelatin hydrogel for bone regeneration. J. Biomater. Appl. 2017, 32, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yeung, K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact. Mater. 2017, 2, 224–247. [Google Scholar] [CrossRef]
- Marquez-Curtis, L.A.; Janowska-Wieczorek, A. Enhancing the migration ability of mesenchymal stromal cells by targeting the sdf-1/cxcr4 axis. Biomed Res. Int. 2013, 2013, 561098. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liu, W.; Du, M.; Yang, C.; Li, X.; Yang, P. The differential effect of basic fibroblast growth factor and stromal cellderived factor1 pretreatment on bone morrow mesenchymal stem cells osteogenic differentiation potency. Mol. Med. Rep. 2018, 17, 3715–3721. [Google Scholar] [PubMed]
- Zhang, H.; Li, X.; Li, J.; Zhong, L.; Chen, X.; Chen, S. SDF-1 mediates mesenchymal stem cell recruitment and migration via the SDF-1/CXCR4 axis in bone defect. J. Bone Miner. Metab. 2021, 39, 126–138. [Google Scholar] [CrossRef]
- Yang, F.; Xue, F.; Guan, J.; Zhang, Z.; Yin, J.; Kang, Q. Stromal-Cell-Derived Factor (SDF) 1-Alpha Overexpression Promotes Bone Regeneration by Osteogenesis and Angiogenesis in Osteonecrosis of the Femoral Head. Cell. Physiol. Biochem. 2018, 46, 2561–2575. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Zhang, Y.; Gu, Y.; Xu, Y.; Liu, Y.; Li, B.; Chen, L. Sequential and sustained release of SDF-1 and BMP-2 from silk fibroin-nanohydroxyapatite scaffold for the enhancement of bone regeneration. Biomaterials 2016, 106, 205–216. [Google Scholar] [CrossRef]
- Lauer, A.; Wolf, P.; Mehler, D.; Götz, H.; Rüzgar, M.; Baranowski, A.; Henrich, D.; Rommens, P.M.; Ritz, U. Biofabrication of SDF-1 Functionalized 3D-Printed Cell-Free Scaffolds for Bone Tissue Regeneration. Int. J. Mol. Sci. 2020, 21, 2175. [Google Scholar] [CrossRef] [PubMed]
- Sano, H.; Kikuta, J.; Furuya, M.; Kondo, N.; Endo, N.; Ishii, M. Intravital bone imaging by two-photon excitation microscopy to identify osteocytic osteolysis in vivo. Bone 2015, 74, 134–139. [Google Scholar] [CrossRef]
- Campagnola, P.J.; Loew, L. Second-harmonic imaging microscopy for visualizing biomolecular arrays in cells, tissues and organisms. Nat. Biotechnol. 2003, 21, 1356–1360. [Google Scholar] [CrossRef]
- Ishii, M.; Fujimori, S.; Kaneko, T.; Kikuta, J. Dynamic live imaging of bone: Opening a new era with ‘bone histodynametry’. J. Bone Miner Metab. 2013, 31, 507–511. [Google Scholar] [CrossRef]
- Genthial, R.; Beaurepaire, E.; Schanne-Klein, M.-C.; Peyrin, F.; Farlay, D.; Olivier, C.; Bala, Y.; Boivin, G.; Vial, J.-C.; Débarre, D.; et al. Label-free imaging of bone multiscale porosity and interfaces using third-harmonic generation microscopy. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pendleton, E.G.; Tehrani, K.F.; Barrow, R.P.; Mortensen, L.J. Second harmonic generation characterization of collagen in whole bone. Biomed. Opt. Express 2020, 11, 4379–4396. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-L.; Hsieh, C.-Y.; Yeh, C.-Y.; Lin, F.-H. The Development of Gelatin/Hyaluronate Copolymer Mixed with Calcium Sulfate, Hydroxyapatite, and Stromal-Cell-Derived Factor-1 for Bone Regeneration Enhancement. Polymers 2019, 11, 1454. [Google Scholar] [CrossRef] [PubMed]
- Jepegnanam, T.S.; von Schroeder, H.P. Rapid resorption of calcium sulfate and hardware failure following corrective radius osteotomy: 2 case reports. J. Hand Surg. Am. 2012, 37, 477–480. [Google Scholar] [CrossRef]
- Lee, G.H.; Khoury, J.G.; Bell, J.-E.; Buckwalter, J.A. Adverse Reactions to OsteoSet Bone Graft Substitute, the Incidence in a Consecutive Series. Iowa Orthop. J. 2002, 22, 35–38. [Google Scholar]
- Lapidot, T. Mechanism of Human Stem Cell Migration and Repopulation of NOD/SCID and B2mnull NOD/SCID Mice: The Role of sdf-1/cxcr4 Interactions. Ann. N. Y. Acad. Sci. 2006, 938, 83–95. [Google Scholar] [CrossRef] [PubMed]



| Component | Gel/HA–HAP–SDF-1 | Gel/HA–HAP | Control | Reference * |
|---|---|---|---|---|
| RBC (M/µL) | 8.28 (0.5) | 8.46 (0.32) | 8.09 (0.37) | 7.37–9.25 |
| HGB (g/dL) | 14.57 (0.35) | 14.3 (0.45) | 14.27 (0.45) | 14.4–17.6 |
| HCT (%) | 42.93 (1.8) | 43.33 (1.76) | 42.8 (0.62) | 36–46 |
| MCV (fL) | 51.87 (2) | 51.23 (0.38) | 53 (2.02) | 47–52 |
| MCH (pg) | 17.6 (0.75) | 16.9 (0.17) | 17.67 (0.25) | 17–21 |
| MCHC (g/dL) | 33.97 (0.61) | 33 (0.26) | 33.33 (0.81) | 35–43 |
| WBC (K/µL) | 12.38 (2.31) | 12.33 (1.57) | 12.2 (3.43) | 6.19–12.55 |
| NEUT (%) | 8.93 (7.24) | 13.53 (3.31) | 13.57 (2.03) | 1–29 |
| LYMPH (%) | 82.8 (7.23) | 78.4 (4.45) | 79.43 (3.11) | 70–99 |
| MONO (%) | 7.13 (0.85) | 5.8 (2.01) | 5.2 (1.5) | 0–6 |
| EO (%) | 1.07 (0.9) | 2.17 (1) | 1.67 (0.68) | 0–3 |
| BASO (%) | 0.07 (0.06) | 0.1 (0) | 0.13 (0.06) | 0–2 |
| ALKP (U/L) | 191.33 (10.02) | 162 (21.07) | 184.33 (88.95) | 39–216 |
| Ca (mg/dL) | 9.77 (0.45) | 9.77 (0.49) | 9.1 (0.3) | 8–15 |
| LDH (U/L) | 585.67 (100.95) | 659 (161.15) | 609 (193.9) | 300–700 |
| Component | Gel/HA–HAP–SDF-1 | Gel/HA–HAP | Control | Reference * |
|---|---|---|---|---|
| RBC (M/µL) | 8.33 (0.49) | 8.42 (0.23) | 8.27 (0.32) | 7.37–9.25 |
| HGB (g/dL) | 14.77 (0.65) | 14.5 (0.53) | 14.63 (0.67) | 14.4–17.6 |
| HCT (%) | 43.67 (2.5) | 43.4 (2.43) | 43.67 (1.57) | 36–46 |
| MCV (fL) | 52.43 (1.16) | 51.53 (1.53) | 52.8 (0.4) | 47–52 |
| MCH (pg) | 17.73 (0.35) | 17.23 (0.21) | 17.7 (0.44) | 17–21 |
| MCHC (g/dL) | 33.8 (0.46) | 33.43 (0.71) | 33.53 (1) | 35–43 |
| WBC (K/µL) | 12.53 (2.16) | 12.3 (1.69) | 11.97 (1.61) | 6.19–12.55 |
| NEUT (%) | 14.8 (6.15) | 14.3 (1.39) | 20.87 (11.82) | 1–29 |
| LYMPH (%) | 76.6 (4.97) | 79.4 (1.4) | 72.03 (12.42) | 70–99 |
| MONO (%) | 4.87 (0.45) | 4.03 (0.58) | 4.27 (0.15) | 0–6 |
| EO (%) | 3.6 (1.42) | 2.07 (0.72) | 2.63 (0.51) | 0–3 |
| BASO (%) | 0.13 (0.06) | 0.2 (0.1) | 0.2 (0.1) | 0–2 |
| ALKP (U/L) | 173.67 (24.54) | 193.33 (36.9) | 218.67 (45.32) | 39–216 |
| Ca (mg/dL) | 10.77 (0.42) | 10.43 (0.21) | 10.77 (0.31) | 8–15 |
| LDH (U/L) | 625.33 (276.3) | 414.67 (24.79) | 526 (121.9) | 300–700 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, Y.-L.; Hsieh, C.-Y.; Yeh, C.-Y.; Chang, C.-H.; Lin, F.-H. Fabrication of Stromal Cell-Derived Factor-1 Contained in Gelatin/Hyaluronate Copolymer Mixed with Hydroxyapatite for Use in Traumatic Bone Defects. Micromachines 2021, 12, 822. https://doi.org/10.3390/mi12070822
Chang Y-L, Hsieh C-Y, Yeh C-Y, Chang C-H, Lin F-H. Fabrication of Stromal Cell-Derived Factor-1 Contained in Gelatin/Hyaluronate Copolymer Mixed with Hydroxyapatite for Use in Traumatic Bone Defects. Micromachines. 2021; 12(7):822. https://doi.org/10.3390/mi12070822
Chicago/Turabian StyleChang, Yun-Liang, Chia-Ying Hsieh, Chao-Yuan Yeh, Chih-Hao Chang, and Feng-Huei Lin. 2021. "Fabrication of Stromal Cell-Derived Factor-1 Contained in Gelatin/Hyaluronate Copolymer Mixed with Hydroxyapatite for Use in Traumatic Bone Defects" Micromachines 12, no. 7: 822. https://doi.org/10.3390/mi12070822
APA StyleChang, Y.-L., Hsieh, C.-Y., Yeh, C.-Y., Chang, C.-H., & Lin, F.-H. (2021). Fabrication of Stromal Cell-Derived Factor-1 Contained in Gelatin/Hyaluronate Copolymer Mixed with Hydroxyapatite for Use in Traumatic Bone Defects. Micromachines, 12(7), 822. https://doi.org/10.3390/mi12070822







